Submitted:
03 August 2025
Posted:
04 August 2025
You are already at the latest version
Abstract
Keywords:
What Is Operation Stork Speed and What Are Its Goals?
Why Is an Update on Nutrient Requirements in Infant Formulas Needed?
Fats and Fatty Acids
Fatty Acids Regulations
Saturated and Monounsaturated Fatty Acids
Linoleic Acid and Alpha Linolenic Acid
DHA and ARA
Omega-6 for Body, Omega-3 for Brain
Carbohydrate Content of Infant Formula
Reassessment of the Recommended Protein Content in Infant Formula for Healthy Full-Term Infants
Protein Quantity
Amino Acid Profiles
| Dynamics of Amino Acid Profiles | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Source | His | Ile | Leu | Lys | SAA | AAA | Thr | Trp | Val | ||
| Total | Met | Cys | |||||||||
| (A) Bovine Casein | 26 | 90 | 51 | 75 | 28 | 25 | 3 | 100 | 41 | 13 | 61 |
| (B) Bovine Whey | 16 | 74 | 121 | 109 | 52 | 25 | 27 | 75 | 88 | 17 | 69 |
| (C) Soy Protein Isolate | 27 | 43 | 78 | 65 | 28 | 14 | 14 | 95 | 36 | 10 | 45 |
| (D) Human Milk | 23 | 51 | 94 | 63 | 35 | 14 | 21 | 87 | 43 | 18 | 50 |
| Ratio A:D | 1.13 | 1.76 | 0.54 | 1.19 | 0.80 | 1.79 | 0.14 | 1.15 | 0.95 | 0.72 | 1.22 |
| Ratio B:D | 0.70 | 1.45 | 1.29 | 1.73 | 1.49 | 1.79 | 1.29 | 0.86 | 2.05 | 0.94 | 1.38 |
| Ratio C:D | 1.17 | 0.84 | 0.83 | 1.03 | 0.80 | 1.00 | 0.67 | 1.09 | 0.84 | 0.56 | 0.90 |
Protein Quality
Micronutrient Considerations in Infant Formula Regulatory Review
Conclusions
Financial Support
Acknowledgments
Abbreviations
References
- US Dept of Health and Human Services. Operation Stork: Speeding up the process to bring infant formula to the U.S 2025. Available from: https://www.hhs.gov/about/news/operation-stork-speed.html Accessed July 28,2025.
- Kirchner L. We tested 41 Baby Formulas for Lead and Arsenic Consumer Reports. 2025.
- Video of FDA panel on infant formula, June 4, 2025 https://www.youtube.com/live/MmE6rlMJdwA). Accessed July 28, 2025.
- Porter D. Infant Formula: National Problems. In: Service CR, editor. Washington, DC: Library of Congress; 1984.
- Levy JS, Roy S, Hutcheson Jr RH, Gruskin AB, Hellerstein S, Linshaw M et al. Infant Metabolic Alkalosis and Soy-Based Formula -- United States. Morbidity and Mortality Weekly Report. 1979;28:358-9.
- Willoughby A, Moss HA, Hubbard VS, Bercu BB, Graubard BI, Vietze PM et al. Developmental outcome in children exposed to chloride-deficient formula. Pediatrics. 1987 Jun;79(6):851-7. [CrossRef]
- Malloy MH, Graubard B, Moss H, McCarthy M, Gwyn S, Vietze P et al. Hypochloremic metabolic alkalosis from ingestion of a chloride-deficient infant formula: outcome 9 and 10 years later. Pediatrics. 1991 Jun;87(6):811-22. [CrossRef]
- Kruse J, Foreman M, Denny S. US Seed Oil Ban Scenario. A scenario off the November 2024 baseline. World Agricultural Economic and Environmental Services, 2025.
- Keefe DM. GRN No. 553. Algal oil (40% docosahexaenoic acid) derived from Schizochytrium sp. In: CSFAN FDA_HHS, editor. Washington, DC2015.
- Carlson SE, Schipper L, Brenna JT, Agostoni C, Calder PC, Forsyth S et al. Perspective: Moving Toward Desirable Linoleic Acid Content in Infant Formula. Adv Nutr. 2021 Dec 1;12(6):2085-98. [CrossRef]
- Einerhand AWC, Mi W, Haandrikman A, Sheng XY, Calder PC. The Impact of Linoleic Acid on Infant Health in the Absence or Presence of DHA in Infant Formulas. Nutrients. 2023 May 4;15(9). [CrossRef]
- Udell T, Gibson RA, Makrides M, Group PS. The effect of alpha-linolenic acid and linoleic acid on the growth and development of formula-fed infants: a systematic review and meta-analysis of randomized controlled trials. Lipids. 2005 Jan;40(1):1-11. [CrossRef]
- Brenna JT. Arachidonic acid needed in infant formula when docosahexaenoic acid is present. Nutr Rev. 2016 May;74(5):329-36. [CrossRef]
- Crawford MA, Wang Y, Forsyth S, Brenna JT. New European Food Safety Authority recommendation for infant formulae contradicts the physiology of human milk and infant development. Nutr Health. 2013 Apr;22(2):81-7. [CrossRef]
- Koletzko B, Bergmann K, Brenna JT, Calder PC, Campoy C, Clandinin MT et al. Should formula for infants provide arachidonic acid along with DHA? A position paper of the European Academy of Paediatrics and the Child Health Foundation. Am J Clin Nutr. 2020 Jan 1;111(1):10-6. [CrossRef]
- Hadley KB, Ryan AS, Forsyth S, Gautier S, Salem N, Jr. The Essentiality of Arachidonic Acid in Infant Development. Nutrients. 2016 Apr 12;8(4):216. [CrossRef]
- Tounian P, Bellaiche M, Legrand P. ARA or no ARA in infant formulae, that is the question. Arch Pediatr. 2021 Jan;28(1):69-74. [CrossRef]
- Cunnane SC, Francescutti V, Brenna JT. Docosahexaenoate requirement and infant development. Nutrition. 1999 Oct;15(10):801-2.
- Saldanha LG, Salem N, Jr., Brenna JT. Workshop on DHA as a required nutrient: overview. Prostaglandins, leukotrienes, and essential fatty acids. 2009 Aug-Sep;81(2-3):233-6. [CrossRef]
- Brenna JT, Lapillonne A. Background paper on fat and fatty acid requirements during pregnancy and lactation. Ann Nutr Metab. 2009;55(1-3):97-122. [CrossRef]
- Astrup AV, Bazinet RP, Brenna JT, Calder PC, Crawford MA, Dangour AD et al. Fats and fatty acids in human nutrition. Report of an expert consultation. Food and Agriculture Organization of the United Nations, 2010.
- Fan L, Wang X, Szeto IM, Liu B, Sinclair AJ, Li D. Dietary intake of different ratios of ARA/DHA in early stages and its impact on infant development. Food Funct. 2024 Apr 2;15(7):3259-73. [CrossRef]
- Tian A, Xu L, Szeto IM, Wang X, Li D. Effects of Different Proportions of DHA and ARA on Cognitive Development in Infants: A Meta-Analysis. Nutrients. 2025 Mar 20;17(6). [CrossRef]
- Ni X, Zhang Z, Deng Z, Li J. Optimizing ARA and DHA in infant formula: A systematic review of global trends, regional disparities, and considerations for precision nutrition. Food Res Int. 2024 Apr;182:114049. [CrossRef]
- Yuhas R, Pramuk K, Lien EL. Human milk fatty acid composition from nine countries varies most in DHA. Lipids. 2006 Sep;41(9):851-8. [CrossRef]
- Jensen RG. Handbook of Human Milk Composition. New York: Academic Press; 1995.
- Martin JC, Bougnoux P, Antoine JM, Lanson M, Couet C. Triacylglycerol structure of human colostrum and mature milk. Lipids. 1993 Jul;28(7):637-43. [CrossRef]
- Innis SM, Dyer R, Nelson CM. Evidence that palmitic acid is absorbed as sn-2 monoacylglycerol from human milk by breast-fed infants. Lipids. 1994 Aug;29(8):541-5.
- Yli-Jokipii KM, Schwab US, Tahvonen RL, Kurvinen JP, Mykkanen HM, Kallio HP. Chylomicron and VLDL TAG structures and postprandial lipid response induced by lard and modified lard. Lipids. 2003 Jul;38(7):693-703. [CrossRef]
- Parodi PW. Positional distribution of fatty acids in triglycerides from milk of several species of mammals. Lipids. 1982 Jun;17(6):437-42. [CrossRef]
- Brenna JT, Kothapalli KSD. New understandings of the pathway of long-chain polyunsaturated fatty acid biosynthesis. Curr Opin Clin Nutr Metab Care. 2022 Mar 1;25(2):60-6. [CrossRef]
- Holman RT, editor. A Long Scaly Tale - The Study of Essential Fatty Acid Deficiency at the University of Minnesota. Essential Fatty Acids and Eicosanoids Invited Papers from the Third International Congress; 1992; Adelaide, South Australia, Australia: American Oil Chemist Society.
- Hansen AE, Haggard ME, Boelsche AN, Adam DJ, Wiese HF. Essential fatty acids in infant nutrition. III. Clinical manifestations of linoleic acid deficiency. J Nutr. 1958 Dec 10;66(4):565-76.
- Hansen AE, Wiese HF, Boelsche AN, Haggard ME, Adam DJ, Davis H. Role of Linoleic Acid in Infant Nutrition: Clinical and Chemical Study of 428 Infants Fed on Milk Mixtures Varying in Kind and Amount of Fat. Pediatrics. 1963;31(1):171-92.
- Hansen HS. Essentiality of n-6 fatty acids. J Biol Chem. 2019 Apr 26;294(17):6692.
- Carlson SJ, O'Loughlin AA, Anez-Bustillos L, Baker MA, Andrews NA, Gunner G et al. A Diet With Docosahexaenoic and Arachidonic Acids as the Sole Source of Polyunsaturated Fatty Acids Is Sufficient to Support Visual, Cognitive, Motor, and Social Development in Mice. Front Neurosci. 2019;13:72. [CrossRef]
- Hansen HS, Jensen B, von Wettstein-Knowles P. Apparent in vivo retroconversion of dietary arachidonic to linoleic acid in essential fatty acid-deficient rats. Biochim Biophys Acta. 1986 Sep 12;878(2):284-7. [CrossRef]
- Clandinin MT, Chappell JE, Leong S, Heim T, Swyer PR, Chance GW. Extrauterine fatty acid accretion in infant brain: implications for fatty acid requirements. Early Hum Dev. 1980;4(2):131-8. [CrossRef]
- Carlson SE, Werkman SH, Peeples JM, Cooke RJ, Tolley EA. Arachidonic acid status correlates with first year growth in preterm infants. Proc Natl Acad Sci USA. 1993;90:1073-7. [CrossRef]
- Martinez M. Tissue levels of polyunsaturated fatty acids during early human development. J Pediatr. 1992 Apr;120(4 Pt 2):S129-38. [CrossRef]
- Carver JD, Benford VJ, Han B, Cantor AB. The relationship between age and the fatty acid composition of cerebral cortex and erythrocytes in human subjects. Brain Res Bull. 2001 Sep 15;56(2):79-85. [CrossRef]
- Hoffman DR, Birch EE, Birch DG, Uauy RD. Effects of supplementation with omega 3 long-chain polyunsaturated fatty acids on retinal and cortical development in premature infants. Am J Clin Nutr. 1993 May;57(5 Suppl):807S-12S. [CrossRef]
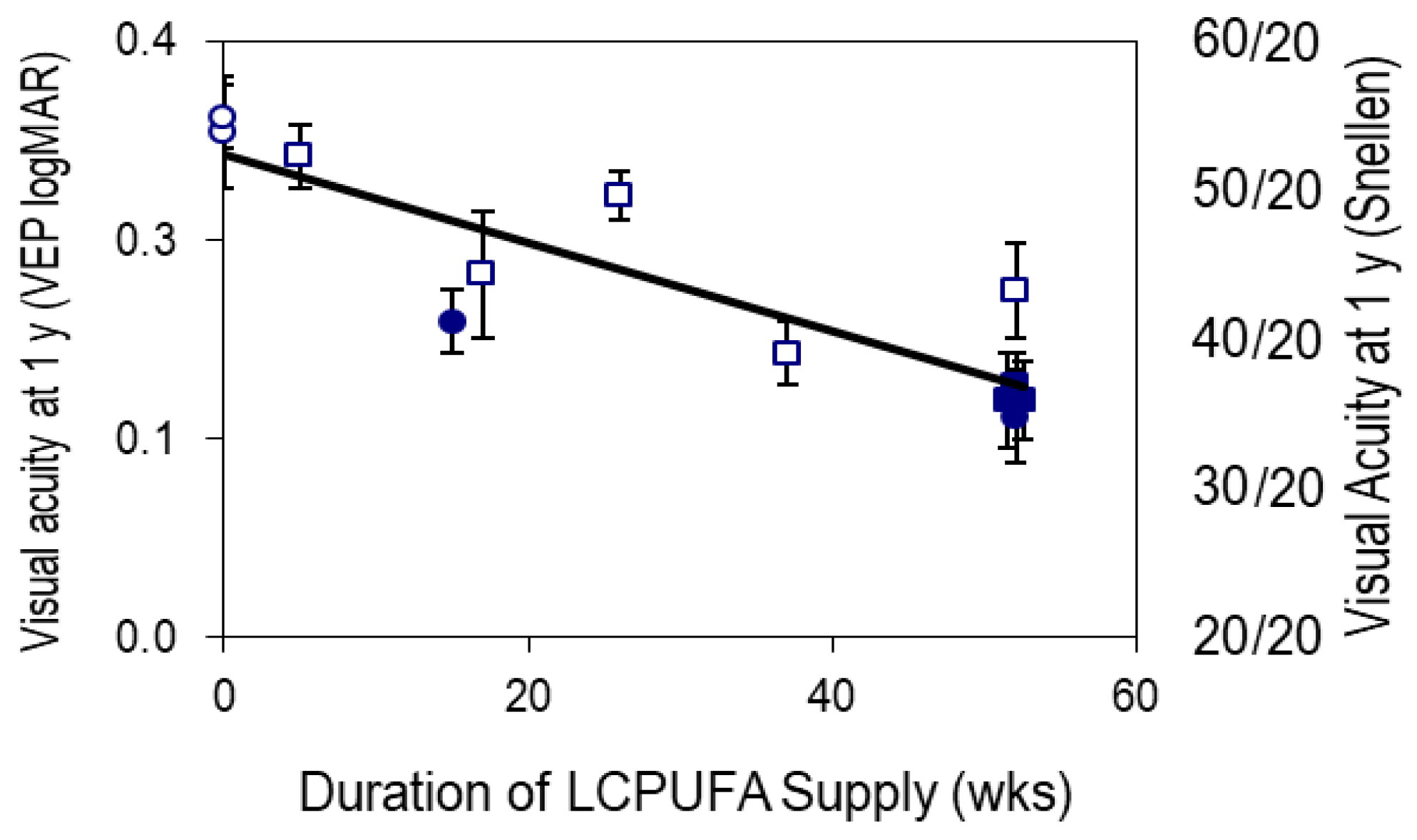
- Morale SE, Hoffman DR, Castaneda YS, Wheaton DH, Burns RA, Birch EE. Duration of long-chain polyunsaturated fatty acids availability in the diet and visual acuity. Early Hum Dev. 2005 Feb;81(2):197-203. [CrossRef]
- Brenna JT. Long-chain polyunsaturated fatty acids and the preterm infant: a case study in developmentally sensitive nutrient needs in the United States. Am J Clin Nutr. 2016 Feb;103(2):606S-15S. [CrossRef]
- Neuringer M, Connor WE, Van Petten C, Barstad L. Dietary omega-3 fatty acid deficiency and visual loss in infant rhesus monkeys. J Clin Invest. 1984;73(1):272-6. [CrossRef]
- Neuringer M, Connor WE, Lin DS, Barstad L, Luck S. Biochemical and functional effects of prenatal and postnatal omega 3 fatty acid deficiency on retina and brain in rhesus monkeys. Proc Natl Acad Sci U S A. 1986 Jun;83(11):4021-5. [CrossRef]
- Sarkadi-Nagy E, Huang MC, Diau GY, Kirwan R, Chueh Chao A, Tschanz C et al. Long chain polyunsaturate supplementation does not induce excess lipid peroxidation of piglet tissues. Eur J Nutr. 2003 Oct;42(5):293-6. [CrossRef]
- Strzalkowski AJ, Jarvinen KM, Schmidt B, Young BE. Protein and carbohydrate content of infant formula purchased in the United States. Clin Exp Allergy. 2022 Nov;52(11):1291-301. [CrossRef]
- Larson-Nath C, Bashir A, Herdes RE, Kiernan B, Lai J, Martin N et al. Term infant formula macronutrient composition: An update for clinicians. Journal of pediatric gastroenterology and nutrition. 2025 May;80(5):751-9. [CrossRef]
- DiMaggio DM, Abersone I, Porto AF. Infant consumption of 100% lactose-based and reduced lactose infant formula in the United States: Review of NHANES data from 1999 to 2020. Journal of pediatric gastroenterology and nutrition. 2024 Nov;79(5):1017-23. [CrossRef]
- Heyman MB, Committee on N. Lactose intolerance in infants, children, and adolescents. Pediatrics. 2006 Sep;118(3):1279-86.
- Abrams SA, Griffin IJ, Davila PM. Calcium and zinc absorption from lactose-containing and lactose-free infant formulas. Am J Clin Nutr. 2002 Aug;76(2):442-6. [CrossRef]
- Heubi J, Karasov R, Reisinger K, Blatter M, Rosenberg L, Vanderhoof J et al. Randomized multicenter trial documenting the efficacy and safety of a lactose-free and a lactose-containing formula for term infants. J Am Diet Assoc. 2000 Feb;100(2):212-7. [CrossRef]
- Moya M, Lifschitz C, Ameen V, Euler AR. A metabolic balance study in term infants fed lactose-containing or lactose-free formula. Acta Paediatr. 1999 Nov;88(11):1211-5. [CrossRef]
- Hampson HE, Jones RB, Berger PK, Plows JF, Schmidt KA, Alderete TL et al. Adverse Effects of Infant Formula Made with Corn-Syrup Solids on the Development of Eating Behaviors in Hispanic Children. Nutrients. 2022 Mar 7;14(5). [CrossRef]
- Griebel-Thompson AK, Fazzino TL, Cramer E, Paluch RA, Morris KS, Kong KL. Early exposure to added sugars via infant formula may explain high intakes of added sugars during complementary feeding beyond maternal modeling. Front Nutr. 2023;10:1188852. [CrossRef]
- Anderson CE, Whaley SE, Goran MI. Lactose-reduced infant formula with corn syrup solids and obesity risk among participants in the Special Supplemental Nutrition Program for Women, Infants, and Children (WIC). Am J Clin Nutr. 2022 Oct 6;116(4):1002-9. [CrossRef]
- Jones RB, Berger PK, Plows JF, Alderete TL, Millstein J, Fogel J et al. Lactose-reduced infant formula with added corn syrup solids is associated with a distinct gut microbiota in Hispanic infants. Gut Microbes. 2020 Nov 9;12(1):1813534. [CrossRef]
- Bode L. Human milk oligosaccharides: every baby needs a sugar mama. Glycobiology. 2012 Sep;22(9):1147-62. [CrossRef]
- Dinleyici M, Barbieur J, Dinleyici EC, Vandenplas Y. Functional effects of human milk oligosaccharides (HMOs). Gut Microbes. 2023 Jan-Dec;15(1):2186115. [CrossRef]
- Berger PK, Bansal R, Sawardekar S, Yonemitsu C, Furst A, Hampson HE et al. Associations of Human Milk Oligosaccharides with Infant Brain Tissue Organization and Regional Blood Flow at 1 Month of Age. Nutrients. 2022 Sep 16;14(18). [CrossRef]
- Ottino-Gonzalez J, Adise S, Machle CJ, Mokhtari P, Holzhausen EA, Furst A et al. Consumption of different combinations of human milk oligosaccharides in the first 6 mo of infancy is positively associated with early cognition at 2 y of age in a longitudinal cohort of Latino children. Am J Clin Nutr. 2024 Sep;120(3):593-601. [CrossRef]
- Moughan PJ, Deglaire A, Yan Y, Wescombe P, Lim WXJ, Stroebinger N et al. Amino acid requirements of the infant: the amino acid composition of human breast milk. Front Nutr. 2024;11:1446565. [CrossRef]
- Duffy B, Gunn T, Collinge J, Pencharz P. The effect of varying protein quality and energy intake on the nitrogen metabolism of parenterally fed very low birthweight (less than 1600 g) infants. Pediatr Res. 1981 Jul;15(7):1040-4. [CrossRef]
- Grummer-Strawn LM, Reinold C, Krebs NF, Centers for Disease C, Prevention. Use of World Health Organization and CDC growth charts for children aged 0-59 months in the United States. MMWR Recomm Rep. 2010 Sep 10;59(RR-9):1-15.
- Arnesen EK, Thorisdottir B, Lamberg-Allardt C, Barebring L, Nwaru B, Dierkes J et al. Protein intake in children and growth and risk of overweight or obesity: A systematic review and meta-analysis. Food Nutr Res. 2022;66. [CrossRef]
- Stokes A, Campbell KJ, Yu HJ, Szymlek-Gay EA, Abbott G, He QQ et al. Protein Intake from Birth to 2 Years and Obesity Outcomes in Later Childhood and Adolescence: A Systematic Review of Prospective Cohort Studies. Adv Nutr. 2021 Oct 1;12(5):1863-76. [CrossRef]
- Abrams SA, Hawthorne KM, Pammi M. A systematic review of controlled trials of lower-protein or energy-containing infant formulas for use by healthy full-term infants. Adv Nutr. 2015 Mar;6(2):178-88. [CrossRef]
- Koletzko B, von Kries R, Closa R, Escribano J, Scaglioni S, Giovannini M et al. Can infant feeding choices modulate later obesity risk? Am J Clin Nutr. 2009 May;89(5):1502S-8S.
- Koletzko B, Baker S, Cleghorn G, Neto UF, Gopalan S, Hernell O et al. Global standard for the composition of infant formula: recommendations of an ESPGHAN coordinated international expert group. Journal of pediatric gastroenterology and nutrition. 2005 Nov;41(5):584-99. [CrossRef]
- Kuczmarski RJ, Ogden CL, Grummer-Strawn LM, Flegal KM, Guo SS, Wei R et al. CDC growth charts: United States. Adv Data. 2000 Jun 8(314):1-27.
- Appleton J, Russell CG, Laws R, Fowler C, Campbell K, Denney-Wilson E. Infant formula feeding practices associated with rapid weight gain: A systematic review. Matern Child Nutr. 2018 Jul;14(3):e12602. [CrossRef]
- Koletzko B, von Kries R, Closa R, Escribano J, Scaglioni S, Giovannini M et al. Lower protein in infant formula is associated with lower weight up to age 2 y: a randomized clinical trial. Am J Clin Nutr. 2009 Jun;89(6):1836-45. [CrossRef]
- Codex Alimentarius. Standard for Infant Formula and Formulas for Special Medical Purposes Intended for Infants. Codex Stan, 2007.
- Health Canada. Proposed compositional requirements for infant foods and foods currently regulated as foods for special dietary use. 2025.
- Liotto N. Protein content of infant formula for the healthy full-term infant. Am J Clin Nutr. 2020 May 1;111(5):946-7. [CrossRef]
- Kouwenhoven SMP, Antl N, Finken MJJ, Twisk JWR, van der Beek EM, Abrahamse-Berkeveld M et al. A modified low-protein infant formula supports adequate growth in healthy, term infants: a randomized, double-blind, equivalence trial. Am J Clin Nutr. 2020 May 1;111(5):962-74. [CrossRef]
- Liotto N, Orsi A, Menis C, Piemontese P, Morlacchi L, Condello CC et al. Clinical evaluation of two different protein content formulas fed to full-term healthy infants: a randomized controlled trial. BMC Pediatr. 2018 Feb 13;18(1):59. [CrossRef]
- Gai N, Uniacke-Lowe T, O'Regan J, Faulkner H, Kelly AL. Effect of Protein Genotypes on Physicochemical Properties and Protein Functionality of Bovine Milk: A Review. Foods. 2021 Oct 11;10(10). [CrossRef]
- Meng F, Uniacke-Lowe T, Ryan AC, Kelly AL. The composition and physico-chemical properties of human milk: A review. Trends Food Sci Technol. 2021;112:608-21. [CrossRef]
- Baker RD, Merritt RJ. Welcome to a New Infant Formula. Journal of pediatric gastroenterology and nutrition. 2022 Oct 1;75(4):389-90. [CrossRef]
- Tinghall Nilsson U, Hernell O, Lonnerdal B, Hartvigsen ML, Jacobsen LN, Kvistgaard AS et al. Low-Protein Formulas with Alpha-Lactalbumin-Enriched or Glycomacropeptide-Reduced Whey: Effects on Growth, Nutrient Intake and Protein Metabolism during Early Infancy: A Randomized, Double-Blinded Controlled Trial. Nutrients. 2023 Feb 17;15(4). [CrossRef]
- Federal_Register. Alternate Foods for Meals: Enriched Macaroni Products with Fortified Protein; National School Lunch Program. Federal Register 59(194) Docket No 1994 October 7. 1004; 94-24902.
- Wallingford JC. Perspective: Assuring the Quality of Protein in Infant Formula. Adv Nutr. 2023 Jul;14(4):585-91. [CrossRef]
- Federal_Register. Protein Efficiency Ratio Rat Bioassay Studies to Demonstrate that a New Infant Formula Supports the Quality Factor of Sufficient Biological Quality of Protein. Draft Guidance for Industry. Federal Register Docket No 2023 February 10, 2023;2023-02836. 88 FR 8868:8868-72.
- AOAC. AOAC Official Method 982.30. Protein Efficiency Ratio: Calculation Method. In: Latimer J, G.W., editor. Official Methods of Analysis of AOAC INTERNATIONAL. 22 ed. New York: AOAC Publications; 2023.
- Boye J, Wijesinha-Bettoni R, Burlingame B. Protein quality evaluation twenty years after the introduction of the protein digestibility corrected amino acid score method. Br J Nutr. 2012 Aug;108 Suppl 2:S183-211. [CrossRef]
- Moughan PJ, Lim WXJ. Digestible indispensable amino acid score (DIAAS): 10 years on. Front Nutr. 2024;11:1389719. [CrossRef]
- Craddock JC, Genoni A, Strutt EF, Goldman DM. Limitations with the Digestible Indispensable Amino Acid Score (DIAAS) with Special Attention to Plant-Based Diets: a Review. Curr Nutr Rep. 2021 Mar;10(1):93-8. [CrossRef]
- USFDA. FDA Roadmap to Reducing Animal Testing in Preclinical Safety Studies, . [July 13, 2025 ]; Available from: https://www.fda.gov/media/187092. Accessed Aug 1, 2025.
- EFSA Panel on Nutrition NF, Food A, Turck D, Bohn T, Castenmiller J, de Henauw S et al. Scientific opinion on the tolerable upper intake level for iron. EFSA J. 2024 Jun;22(6):e8819.
- S. Gahagan, E. Delker, E. Blanco, R. Burrows, B. Lozoff, Randomized controlled trial of Iron-Fortified versus Low-Iron Infant Formula: developmental Outcomes at 16 years, J Pediatr 212 (2019) 124–130, e1. [CrossRef]
- M. Domellöf, C. Braegger, C. Campoy, V. Colomb, T. Decsi, M. Fewtrell, et al., Iron requirements of infants and toddlers. J Pediatr Gastroenterol Nutr. 2014. 58 (1):119–129. doi: 10.5167/uzh-106040. [CrossRef]
- Codex Alimentarius https://www.fao.org/fao-who-codexalimentarius/en/ accessed August 1, 2025.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




