Submitted:
31 July 2025
Posted:
06 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
Study Design
IRB or Ethics Approval
Patient Population
Testing Procedure
Data Collection and Outcome Measures
Statistical Analysis and Model Development
3. Results
Patient Population and Data Overview
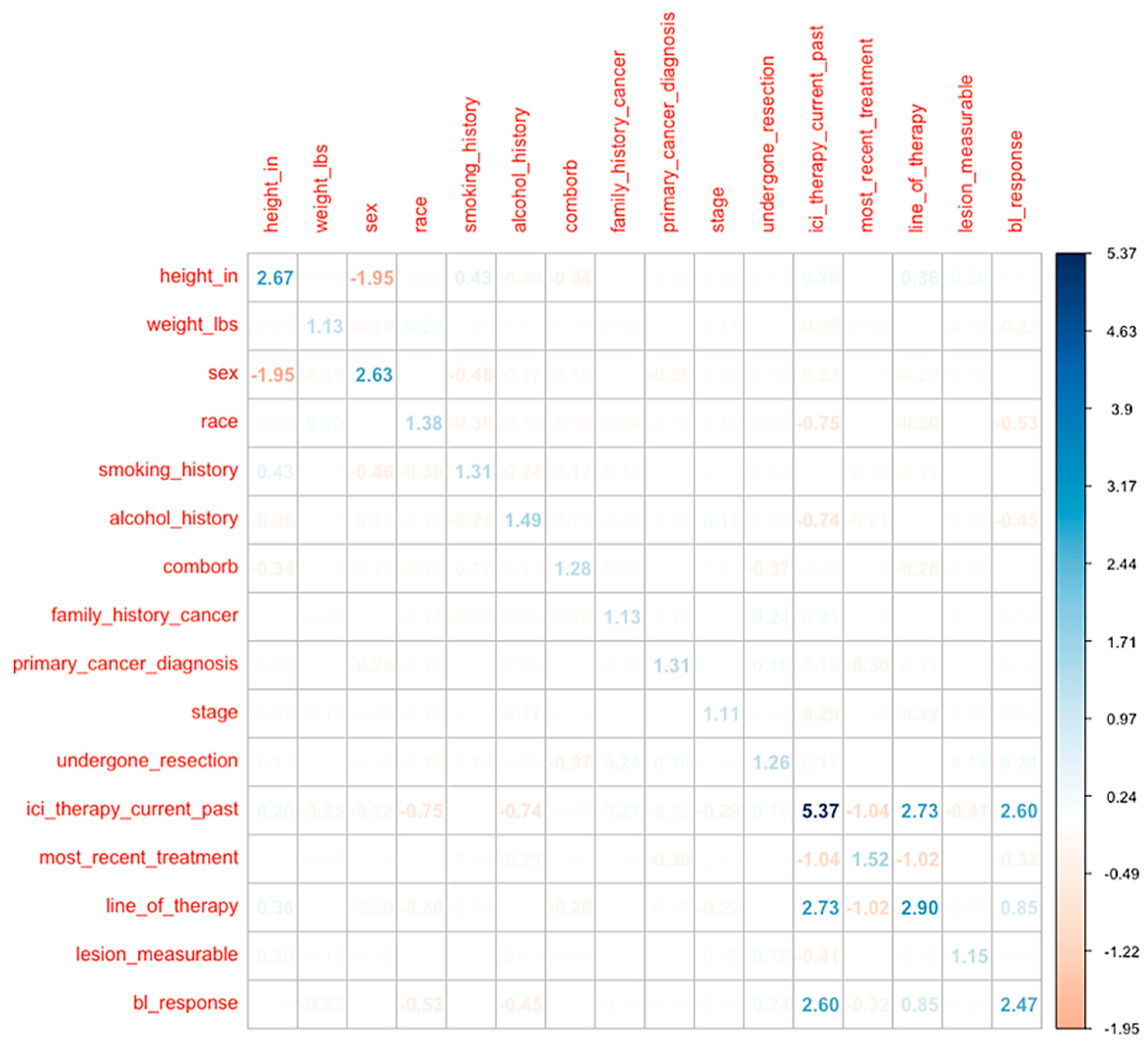
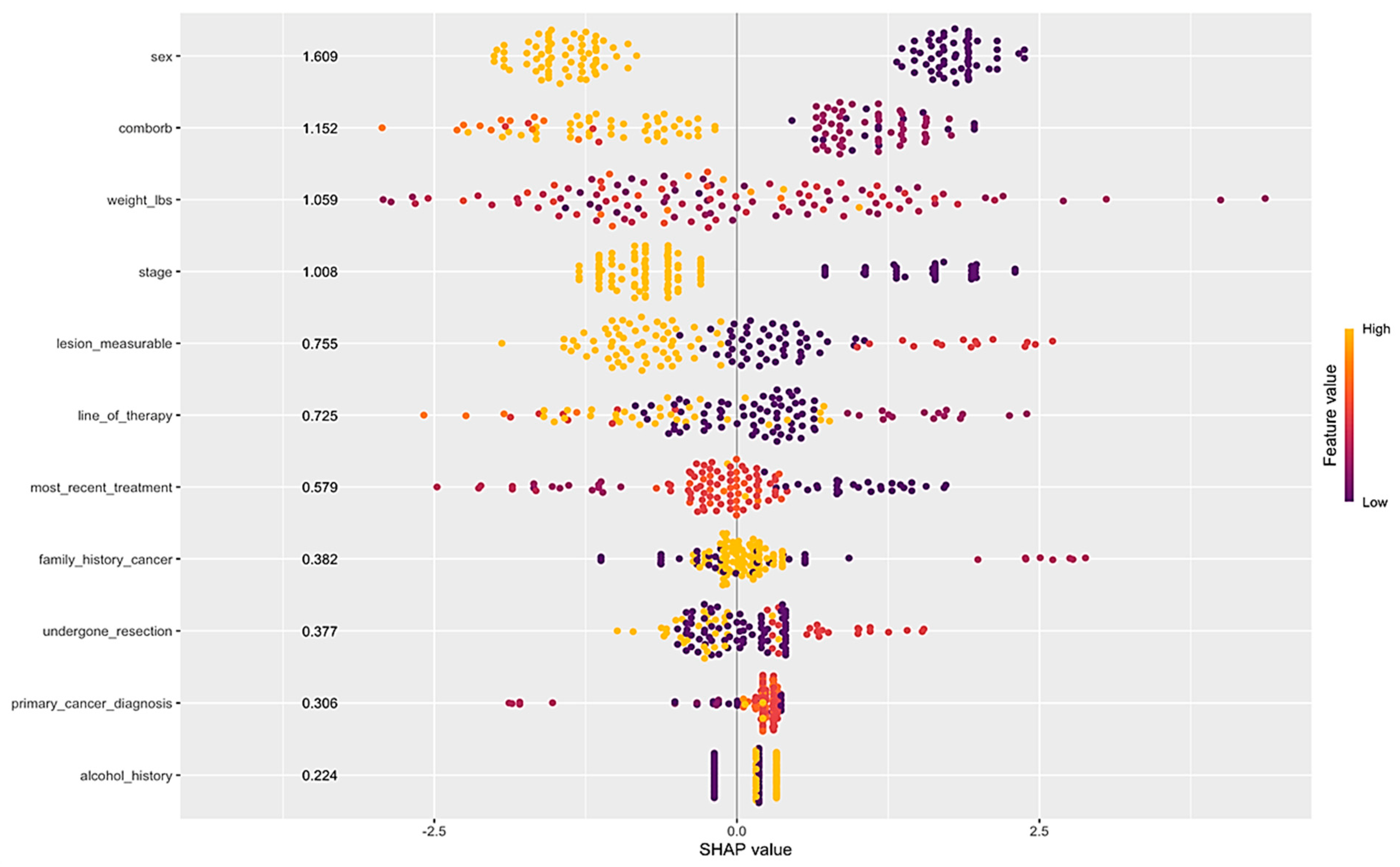
Clinical Predictors of CiRT Classification
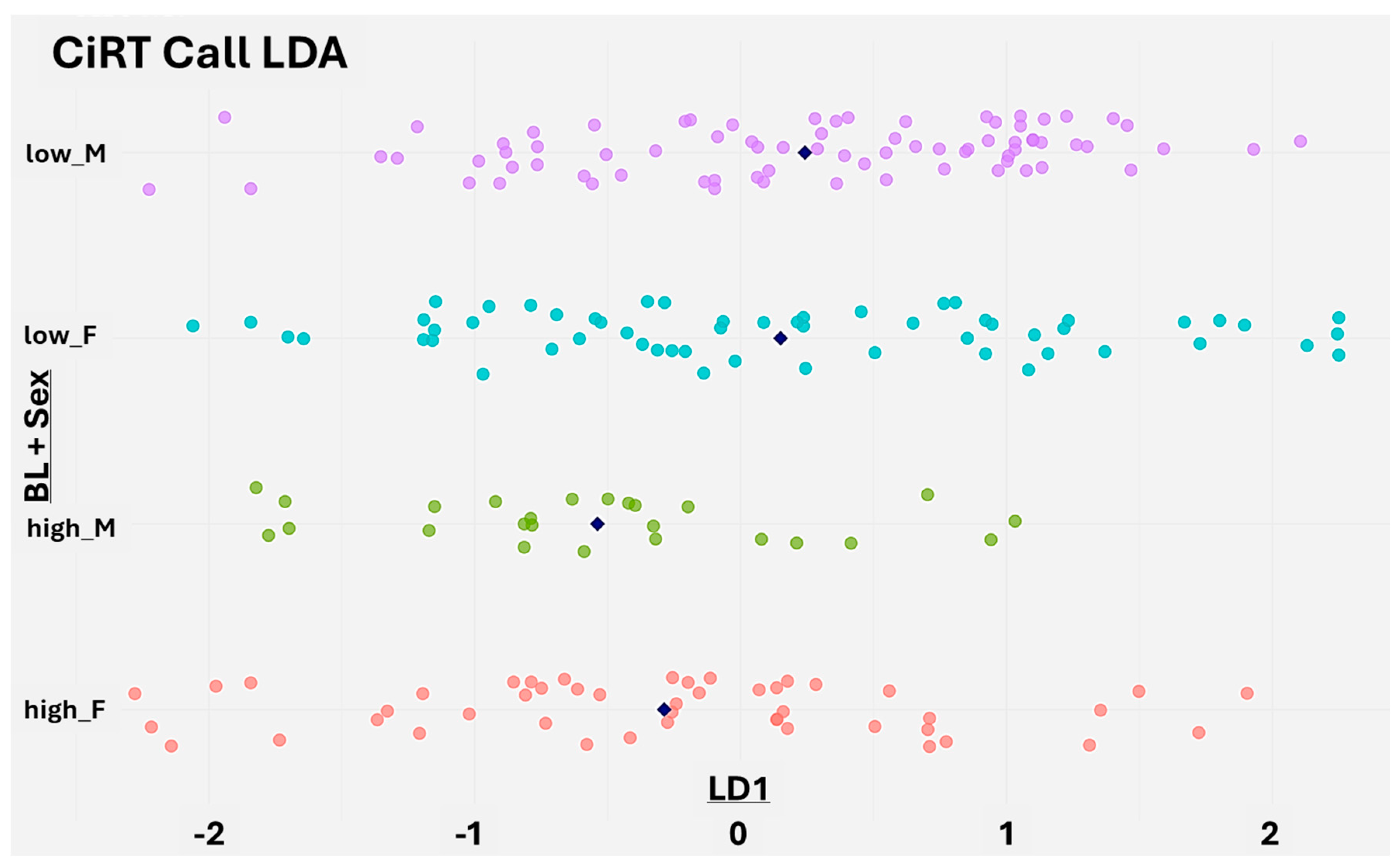
Linear Discriminant Analysis (LDA) of CiRT Classification
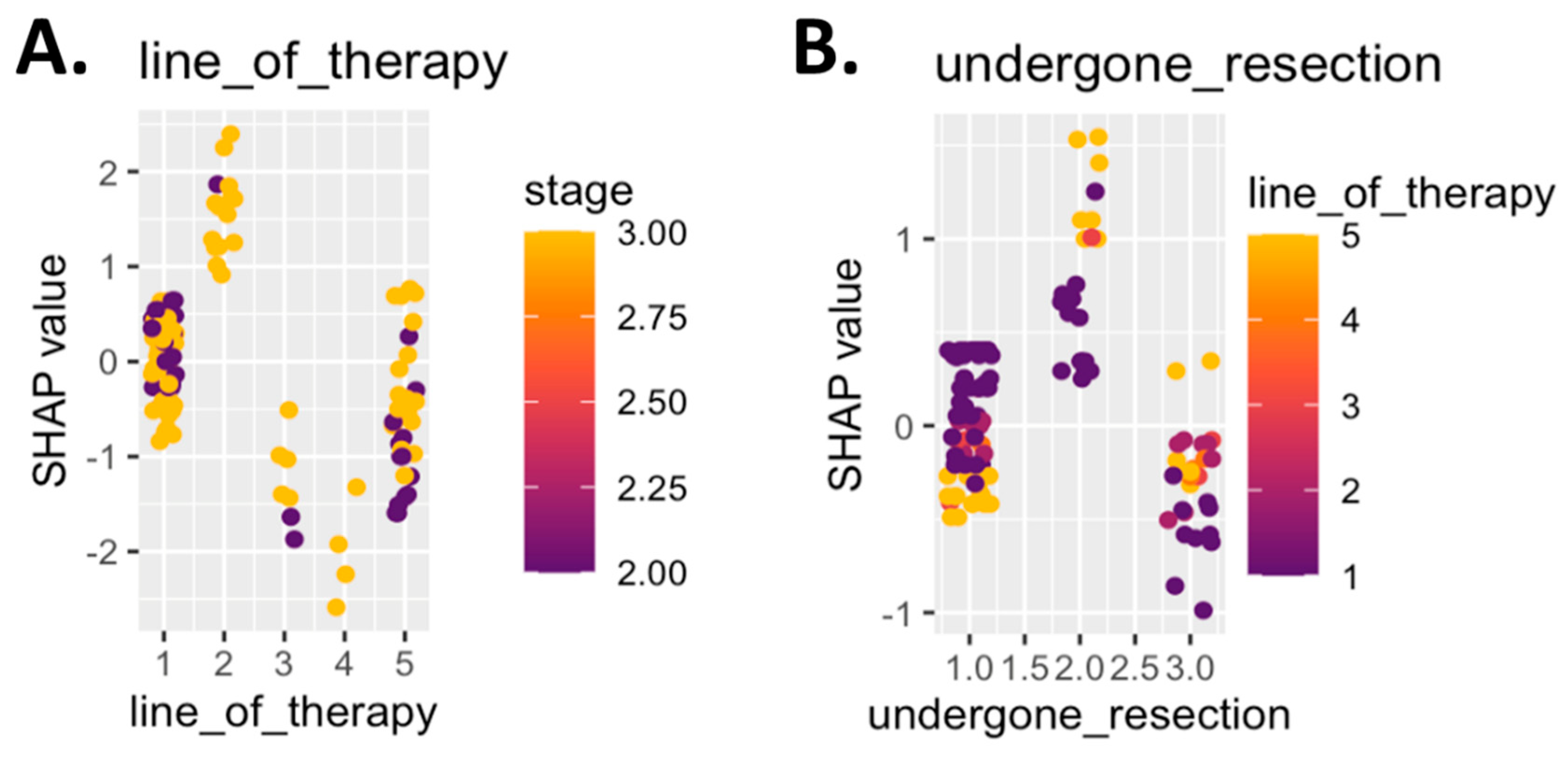
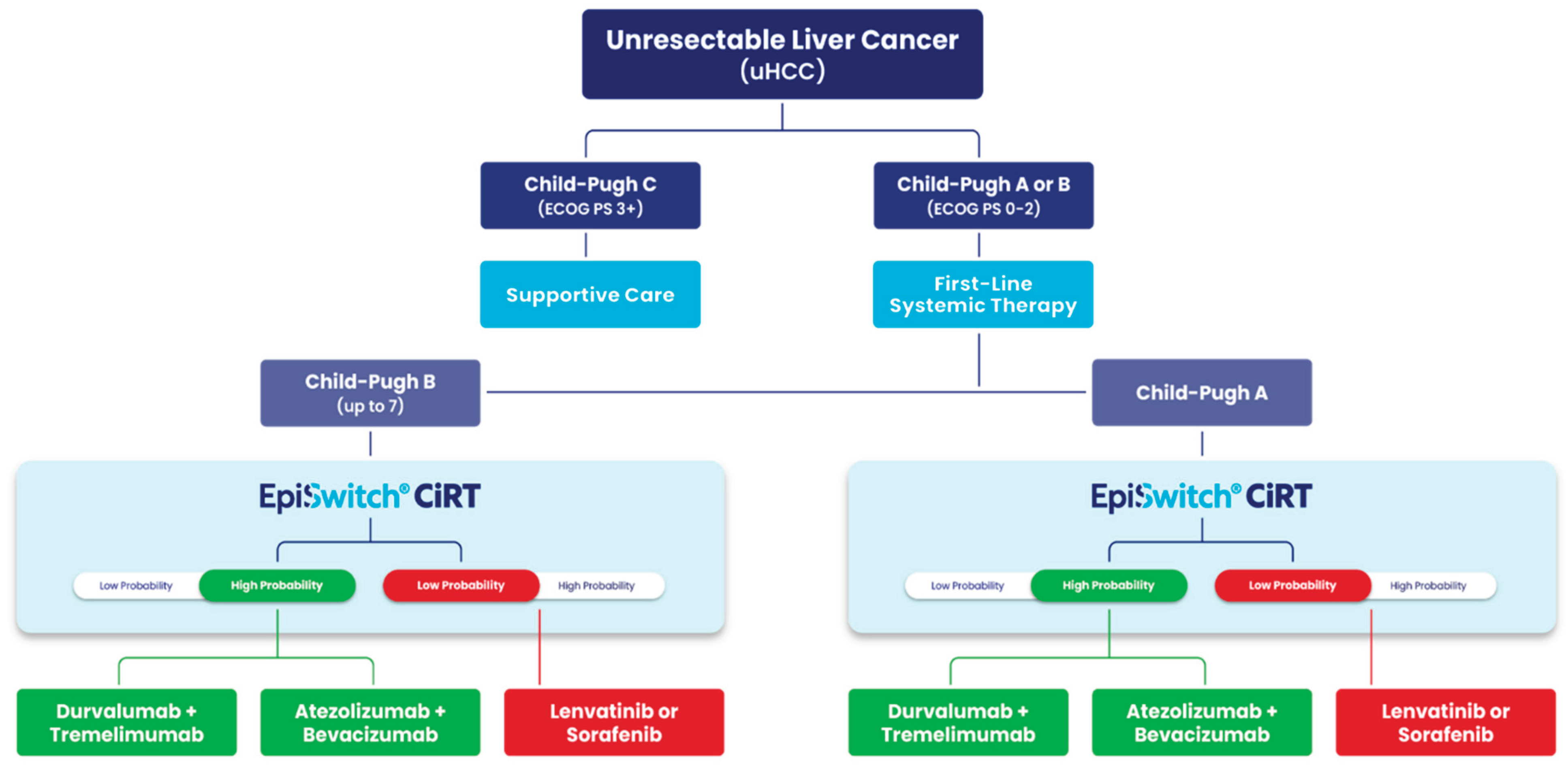
Impact of CiRT on Clinical Decision-Making
CiRT Result Distribution by Sex
Lesion Measurability and CiRT Status
Baseline Clinical Response and Lesion Measurability
CiRT Result and Baseline Response
Longitudinal and Monitoring Utility
Health Equity and Access Assessment
System-Level Utility and Cost Considerations
4. Discussion
Overview of Clinical Utility
Sex-Based Differences in CiRT Classification
Lesion Measurability and Immune Readiness
Lack of Correlation with RECIST-Based Response
Role of Structured Analytics in Real-World Settings
Clinical Integration and Decision Impact
Validation in Hepatocellular Carcinoma and GI Tumors (Ouf, et al.)
Comparison with Standard Biomarkers
Evidence from Prospective Studies
Future Utility and System-Level Impact
Clinical Integration and Decision Impact
5. Conclusions
Author Contributions
Funding
Acknowledgements
Conflict of Interest and Disclaimer Statement
Abbreviations
References
- Yang, F., Wang, J. F., Wang, Y., Liu, B., & Molina, J. R. (2021). Comparative analysis of predictive biomarkers for PD-1/PD-L1 inhibitors in cancers: Developments and challenges. Cancers (Basel), 14(1), 109. [CrossRef]
- Shiravand Y, Khodadadi F, Kashani SMA, Hosseini-Fard SR, Hosseini S, Sadeghirad H, Ladwa R, O’Byrne K, Kulasinghe A. Immune Checkpoint Inhibitors in Cancer Therapy. Current Oncology. 2022; 29(5):3044-3060. [CrossRef]
- Postow MA, Sidlow R, Hellmann MD. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N Engl J Med. 2018 Jan 11;378(2):158-168. PMID: 29320654. [CrossRef]
- Zhao, B., Zhao, H., & Zhao, J. (2020). Efficacy of PD-1/PD-L1 blockade monotherapy in clinical trials. Therapeutic Advances in Medical Oncology, 12. [CrossRef]
- Licata L, Mariani M, Rossari F, et al. Tissue- and liquid biopsy-based biomarkers for immunotherapy in breast cancer. Breast. 2023;69:330-341. [CrossRef]
- Esfahani K, Roudaia L, Buhlaiga N, Del Rincon SV, Papneja N, Miller WH Jr. A review of cancer immunotherapy: from the past, to the present, to the future. Curr Oncol. 2020;27(Suppl 2):S87-S97. [CrossRef]
- Li JX, Huang JM, Jiang ZB, et al. Current Clinical Progress of PD-1/PD-L1 Immunotherapy and Potential Combination Treatment in Non-Small Cell Lung Cancer. Integr Cancer Ther. 2019;18:1534735419890020. [CrossRef]
- Migden MR, Rischin D, Schmults CD, et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N Engl J Med. 2018;379(4):341-351. [CrossRef]
- Mansh M. Ipilimumab and cancer immunotherapy: a new hope for advanced stage melanoma. Yale J Biol Med. 2011;84(4):381-389.
- Paz-Ares L, Spira A, Raben D, et al. Outcomes with durvalumab by tumour PD-L1 expression in unresectable, stage III non-small-cell lung cancer in the PACIFIC trial. Ann Oncol. 2020;31(6):798-806. [CrossRef]
- Rolfo C, Caglevic C, Santarpia M, et al. Immunotherapy in NSCLC: A Promising and Revolutionary Weapon. Adv Exp Med Biol. 2017;995:97-125. [CrossRef]
- Gemelli M, Cortinovis D, Carola G, et al. Efficacy of immune checkpoint inhibitors (ICIs) in PD-L1 negative Non-Small Cell Lung Cancer (NSCLC) - A meta-analysis based on reconstructed individual participant data. Lung Cancer. 2025;205:108621. [CrossRef]
- Inomata M, Takata N, Mizushima I, et al. Immune Checkpoint Inhibitor for Non-small Cell Lung Cancer With Negative or Low Tumor PD-L1 Expression. Cancer Diagn Progn. 2021;1(3):173-177. Published 2021 Jul 3. [CrossRef]
- McGranahan N, Swanton C. Clonal Heterogeneity and Tumor Evolution: Past, Present, and the Future. Cell. 2017;168(4):613-628. [CrossRef]
- Scobie MR, Zhou KI, Ahmed S, Kelley MJ. Utility of Tumor Mutational Burden as a Biomarker for Response to Immune Checkpoint Inhibition in the VA Population. JCO Precis Oncol. 2023;7:e2300176. [CrossRef]
- Zhao P, Li L, Jiang X, Li Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J Hematol Oncol. 2019;12(1):54. Published 2019 May 31. [CrossRef]
- Hussaini S, Chehade R, Boldt RG, et al. Association between immune-related side effects and efficacy and benefit of immune checkpoint inhibitors - A systematic review and meta-analysis. Cancer Treat Rev. 2021;92:102134. [CrossRef]
- Champiat S, Dercle L, Ammari S, et al. Hyperprogressive Disease Is a New Pattern of Progression in Cancer Patients Treated by Anti-PD-1/PD-L1. Clin Cancer Res. 2017;23(8):1920-1928. [CrossRef]
- Kato S, Goodman A, Walavalkar V, Barkauskas DA, Sharabi A, Kurzrock R. Hyperprogressors after Immunotherapy: Analysis of Genomic Alterations Associated with Accelerated Growth Rate. Clin Cancer Res. 2017;23(15):4242-4250. [CrossRef]
- Saâda-Bouzid E, Defaucheux C, Karabajakian A, et al. Hyperprogression during anti-PD-1/PD-L1 therapy in patients with recurrent and/or metastatic head and neck squamous cell carcinoma. Ann Oncol. 2017;28(7):1605-1611. [CrossRef]
- Ferrara R, Mezquita L, Texier M, et al. Hyperprogressive Disease in Patients With Advanced Non-Small Cell Lung Cancer Treated With PD-1/PD-L1 Inhibitors or With Single-Agent Chemotherapy. JAMA Oncol. 2018;4(11):1543-1552. [CrossRef]
- Matos I, Martin-Liberal J, García-Ruiz A, et al. Capturing Hyperprogressive Disease with Immune-Checkpoint Inhibitors Using RECIST 1.1 Criteria. Clin Cancer Res. 2020;26(8):1846-1855. [CrossRef]
- Kim CG, Kim C, Yoon SE, et al. Hyperprogressive disease during PD-1 blockade in patients with advanced hepatocellular carcinoma. J Hepatol. 2021;74(2):350-359. [CrossRef]
- Hunter E, Salter M, Powell R, et al. Development and Validation of Blood-Based Predictive Biomarkers for Response to PD-1/PD-L1 Checkpoint Inhibitors: Evidence of a Universal Systemic Core of 3D Immunogenetic Profiling across Multiple Oncological Indications. Cancers (Basel). 2023;15(10):2696. Published 2023 May 10. [CrossRef]
- Alshaker, H.; Mills, R.; Hunter, E.; Salter, M.; Ramadass, A.; Skinner, B.M.; Westra, W.; Green, J.; Akoulitchev, A.; Winkler, M.; et al. Chromatin conformation changes in peripheral blood can detect prostate cancer and stratify disease risk groups. J. Transl. Med. 2021, 19, 46. [CrossRef]
- Xu Y, He Z, Du J, et al. Epigenetic modulations of immune cells: from normal development to tumor progression. Int J Biol Sci. 2023;19(16):5120-5144. Published 2023 Oct 2. [CrossRef]
- Klein SL, Flanagan KL. Sex differences in immune responses. Nat Rev Immunol. 2016;16(10):626-638. [CrossRef]
- Gonzalez H, Hagerling C, Werb Z. Roles of the immune system in cancer: from tumor initiation to metastatic progression. Genes Dev. 2018;32(19-20):1267-1284. [CrossRef]
- Powles T, Sridhar SS, Bellmunt J, Sternberg CN, Grivas P, Hunter E, Salter M, Powell R, Dring A, Green J, et al. Blood-Epigenetic Biomarker Associations with Tumor Immunophenotype in Patients with Urothelial Carcinoma from JAVELIN Bladder 100. Cancers. 2025; 17(14):2332. [CrossRef]
- Mahmoud Ouf et al. Evaluation of EpiSwitch in predicting immunotherapy response in hepatocellular carcinoma and gastrointestinal tumors. JCO 43, 623-623(2025). [CrossRef]
- Hegde PS, Chen DS. Top 10 Challenges in Cancer Immunotherapy. Immunity. 2020;52(1):17-35. [CrossRef]
- The 2018 Nobel Prize is a reminder of the outrageous cost of curing cancer | Vox (accessed on July 29th, 2025).
- We Can’t Afford the Drugs That Could Cure Cancer - WSJ (accessed on July 29th, 2025).
- Mellor J, Hunter E, Akoulitchev A. Paradigm Lost. Cancers. 2025; 17(13):2187. [CrossRef]
- Hunter E, Alshaker H, Weston C, Issa M, Bautista S, Gebregzabhar A, Virdi A, Dring A, Powell R, Green J, Lal R, Velchuru V, Aryal K, Bin Abu Hassan MR, Meng GT, Patel JS, Mohamed Gani SP, Lim CR, Guiel T, Akoulitchev A, Pchejetski D. A New Blood-Based Epigenetic Diagnostic Biomarker Test (EpiSwitch®® NST) with High Sensitivity and Positive Predictive Value for Colorectal Cancer and Precancerous Polyps. Cancers (Basel). 2025 Feb 4;17(3):521. PMID: 39941889; PMCID: PMC11816175. [CrossRef]
- Ko LC, Gravina N, Berghausen J, Abdo J. Rising Trends in Prostate Cancer Among Asian Men: Global Concerns and Diagnostic Solutions. Cancers (Basel). 2025;17(6):1013. Published 2025 Mar 17. [CrossRef]
- Berghausen J, Abdo J, Mathis R, Hunter E, Akoulitchev A, Pohlman GD. EpiSwitch PSE Blood Test Reduces Unnecessary Prostate Biopsies: A Real-World Clinical Utility Study. Cancers. 2025; 17(13):2193. [CrossRef]
| Inclusion Criteria |
|
| Exclusion criteria |
|
| Outcome | Utility Domain | Measured As |
| Change in treatment decision | Decision support | Physician-reported pre/post CiRT result |
| Concordance of LPRR to no response | Clinical validity & utility | Radiographic response (RECIST v1.1) |
| Treatment avoidance | Cost & toxicity mitigation | % of LPRR patients not treated with ICI |
| Early discontinuation of ICI | Toxicity mitigation | % of LPRR patients de-escalated from ICI |
| Time to ICI start | Workflow efficiency | Days from diagnosis to treatment start |
| SDoH stratification | Equity and access | CiRT status vs. race/ethnicity/SES |
| Resistance detection | Longitudinal monitoring | Proportion shifting from HPRR to LPRR |
| HEOR value | Economic utility | Modeled cost savings from CiRT guidance |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




