Case Presentation
A 70-year-old Japanese male with a history of smoking and alcohol consumption underwent an annual health check-up, during which a fecal occult blood test returned positive for the first time. Subsequent investigations led to a diagnosis of transverse colon cancer with peritoneal metastases. Contrast-enhanced computed tomography (CECT) revealed an irregular mass in the transverse colon near the hepatic flexure, along with multiple peritoneal nodules primarily localized to the greater omentum [
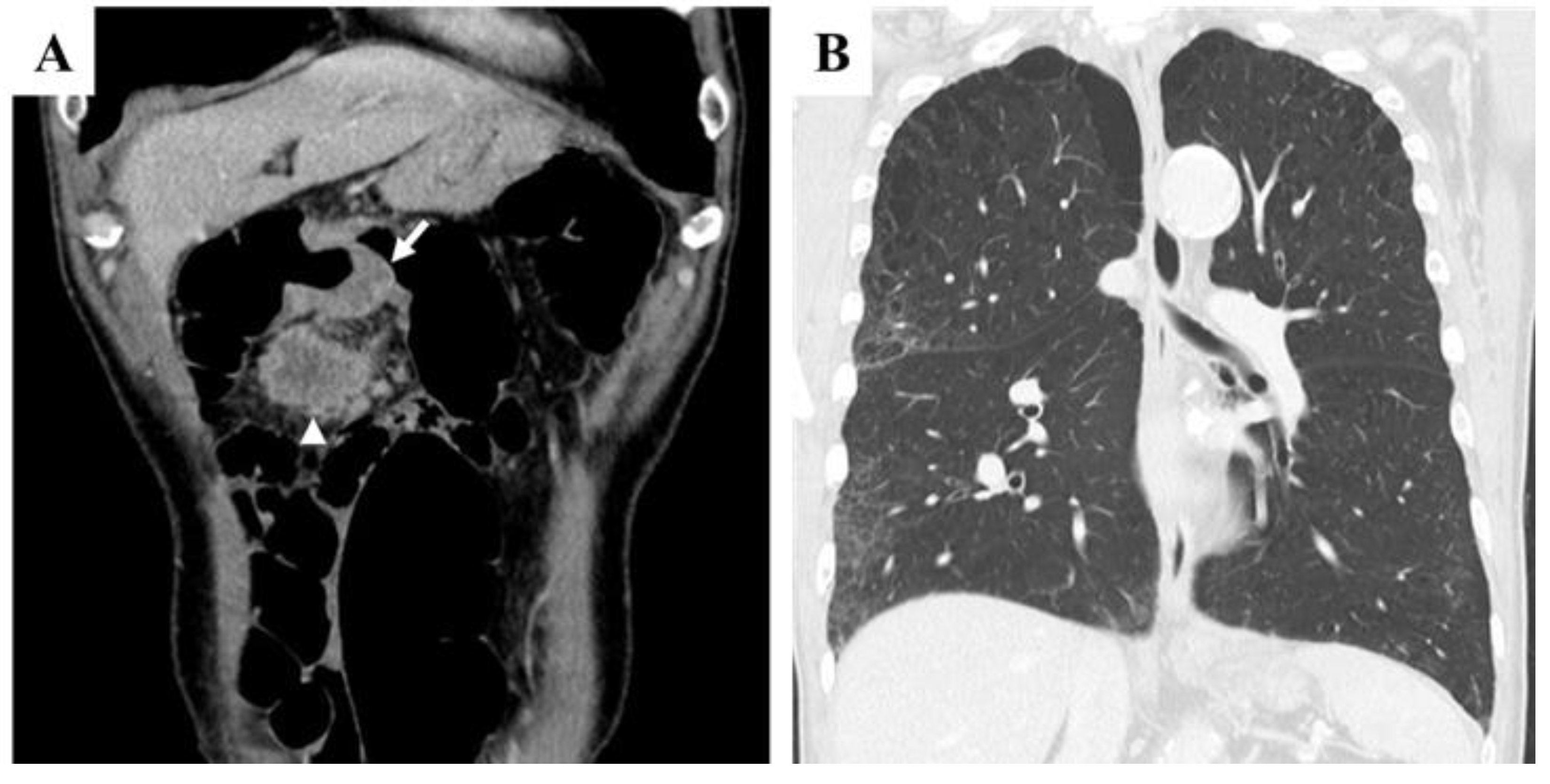
Figure 1A]. Chest CT revealed multiple blebs in both lung apices and a reticular pattern in the right lung [
Figure 1B]. Despite peritoneal dissemination, no hematogenous or lymphatic metastases were evident at this stage.
Figure 1.
Pretreatment findings from abdominal contrast-enhanced CT and chest CT. A: Coronal section view showing a tumor in the transverse colon near the hepatic flexure (arrow) and a peritoneal metastatic nodule located just caudal to the primary tumor (arrowhead). Several additional peritoneal metastatic nodules were observed along the greater omentum (not shown). B: Chest CT revealed multiple blebs in both lung apices, along with a reticular pattern in the right lung lobes.
Figure 1.
Pretreatment findings from abdominal contrast-enhanced CT and chest CT. A: Coronal section view showing a tumor in the transverse colon near the hepatic flexure (arrow) and a peritoneal metastatic nodule located just caudal to the primary tumor (arrowhead). Several additional peritoneal metastatic nodules were observed along the greater omentum (not shown). B: Chest CT revealed multiple blebs in both lung apices, along with a reticular pattern in the right lung lobes.
Initial laboratory findings revealed elevated tumor markers, including carcinoembryonic antigen (CEA) at 55.0 ng/ml and carbohydrate antigen 19-9 (CA19-9) at 2527.6 U/ml. Genetic analysis confirmed that the tumor was RAS/BRAF wild-type and microsatellite instability (MSI)-negative. The patient had a history of emphysema but did not require supplemental oxygen therapy.
The initial treatment regimen consisted of capecitabine, oxaliplatin, and cetuximab. However, after three cycles, imaging indicated disease progression, with an increase in peritoneal disseminated nodules. Chemotherapy was subsequently modified to capecitabine plus irinotecan and bevacizumab. Despite eight cycles, significant ascites accumulation and mesenteric shortening were observed [
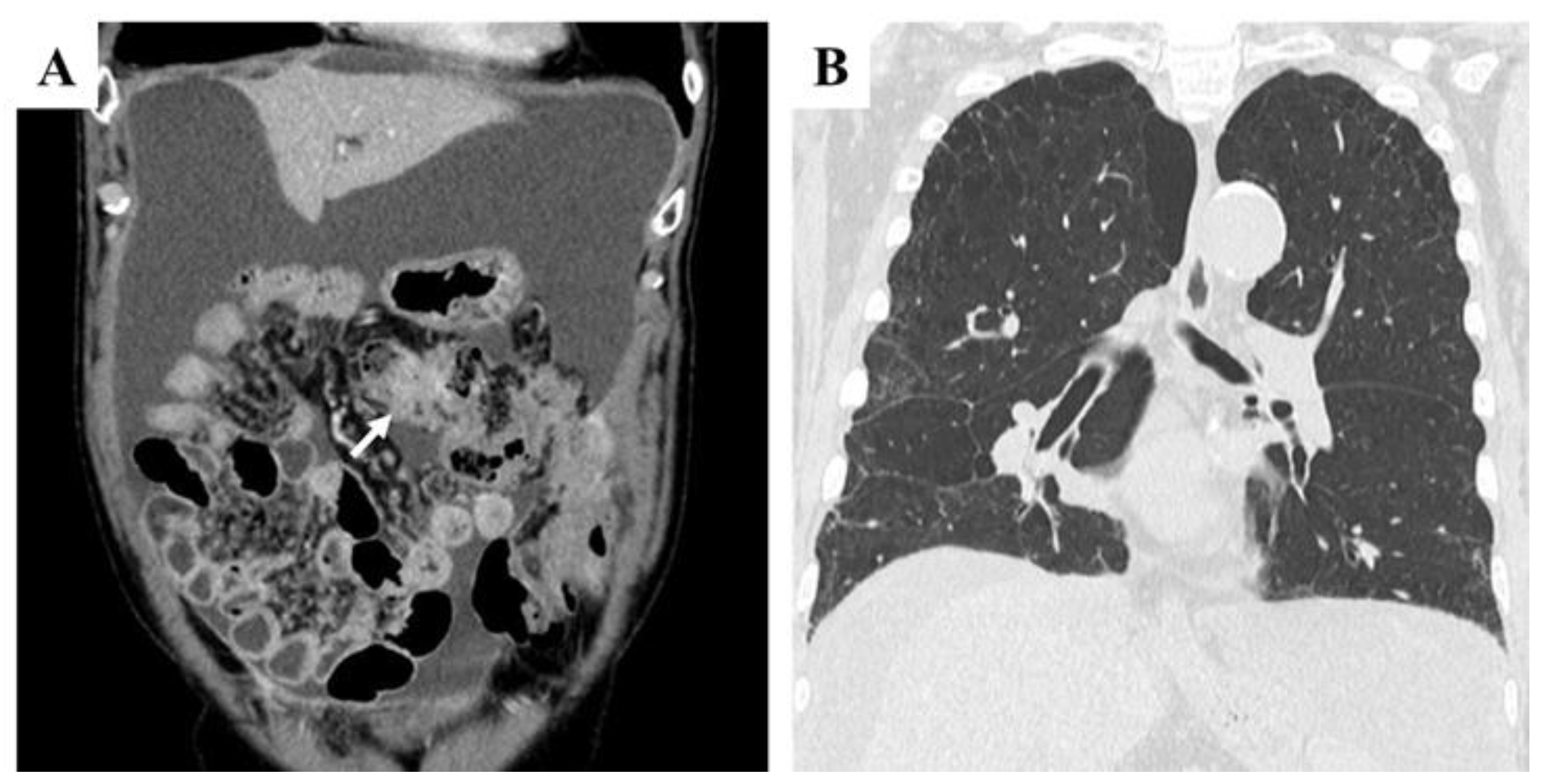
Figure 2A]. At this stage, the tumor remained an irregular mass without evidence of hematogenous metastases, although concerns about rapid disease progression persisted. Compared with pretreatment findings, chest CT revealed no significant worsening of reticular or interstitial shadows [
Figure 2B].
Figure 2.
Abdominal contrast-enhanced CT (CECT) and chest CT findings after eight cycles of chemotherapy with capecitabine, irinotecan, and bevacizumab, following resistance to capecitabine, oxaliplatin, and cetuximab. A: Coronal section view showing a large volume of ascitic fluid and mesenteric shortening. The tumor in the transverse colon appeared as an irregular mass (arrow). B: Chest CT revealed no significant progression of reticular or interstitial shadows compared with pretreatment findings.
Figure 2.
Abdominal contrast-enhanced CT (CECT) and chest CT findings after eight cycles of chemotherapy with capecitabine, irinotecan, and bevacizumab, following resistance to capecitabine, oxaliplatin, and cetuximab. A: Coronal section view showing a large volume of ascitic fluid and mesenteric shortening. The tumor in the transverse colon appeared as an irregular mass (arrow). B: Chest CT revealed no significant progression of reticular or interstitial shadows compared with pretreatment findings.
A final attempt at chemotherapy with trifluridine/tipiracil (FTD/TPI) plus bevacizumab was initiated. Despite three additional cycles, the disease continued to progress. Given the need for alternative therapeutic strategies, a genetic panel analysis was performed, revealing an NTRK1-LMNA gene fusion. Considering the established efficacy of TRK inhibitors in NTRK fusion-positive malignancies, entrectinib treatment was promptly initiated.
Within six weeks of entrectinib treatment, the patient exhibited a remarkable response. Tumor marker levels decreased significantly, with CA19-9 levels decreasing from over 24,000 U/ml to 2,470.2 U/ml. Clinically, ascites accumulation stabilized, and three rounds of cell-free and concentration ascites reinfusion therapy (CART) were performed to manage symptoms. Throughout this period, the patient remained relatively stable and continued outpatient care.
Two months after receiving entrectinib therapy, the patient developed sudden dyspnea, fever, and hypoxia, necessitating emergency hospitalization. Upon transport, his peripheral oxygen saturation was critically low at 50% on room air, improving only marginally to the low 80s with 15 liters of oxygen. Upon admission, his vital signs included a blood pressure of 102/56 mmHg, a pulse rate of 80 beats per minute, a respiratory rate of 20 breaths per minute, and a temperature of 38.5°C.
High-flow oxygen therapy was initiated; however, oxygen saturation remained precarious. The laboratory findings revealed worsening anemia, elevated liver enzymes, and significantly increased C-reactive protein (CRP) levels, suggesting a systemic inflammatory response. Chest CT imaging revealed marked progression of interstitial pneumonia in both lungs, which was most pronounced in the right lobe [
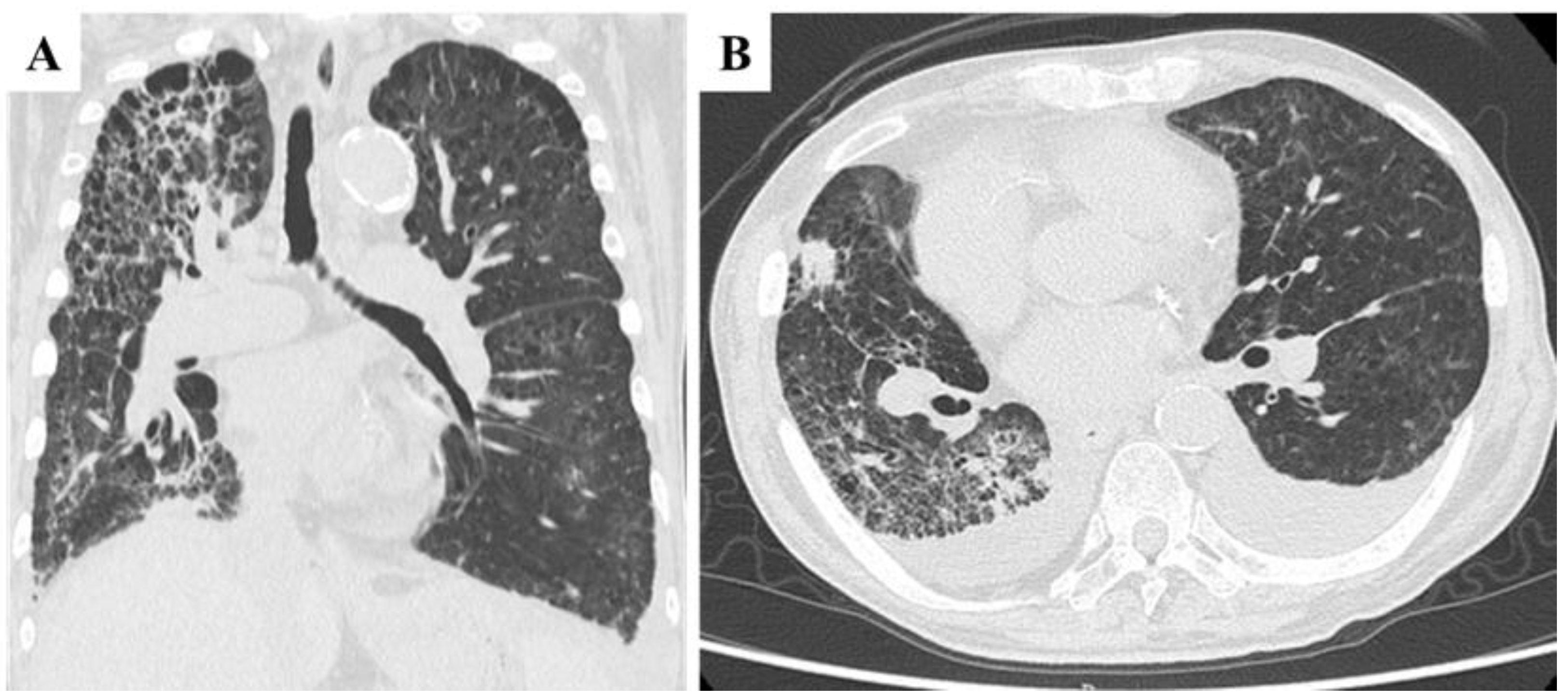
Figure 3A]. Newly developed ground-glass opacities appeared in previously normal lung regions amid areas of emphysema. Additional findings included multiple infiltrative shadows, mild right lung volume reduction, and bilateral pleural effusion [
Figure 3B]. These observations suggested worsening interstitial pneumonia with possible concomitant bacterial infection or drug-related lung toxicity. CT imaging of the chest and abdomen revealed no evidence of hematogenous metastasis, such as lung or liver involvement.
Figure 3.
Chest CT findings at the time of transport due to acute respiratory failure. A and B: Marked deterioration of interstitial pneumonia was observed in both lobes, with the most pronounced changes in the right lobe. Ground-glass opacities appeared against a background of emphysematous lungs, accompanied by multiple infiltrative shadows, mild right lung volume reduction (A), and bilateral pleural effusion (B).
Figure 3.
Chest CT findings at the time of transport due to acute respiratory failure. A and B: Marked deterioration of interstitial pneumonia was observed in both lobes, with the most pronounced changes in the right lobe. Ground-glass opacities appeared against a background of emphysematous lungs, accompanied by multiple infiltrative shadows, mild right lung volume reduction (A), and bilateral pleural effusion (B).
By the third day of hospitalization, signs of right-sided heart failure, including jugular vein distention and peripheral edema, had become apparent. Echocardiography revealed a peak tricuspid regurgitation velocity (TRV) of 4.5 m/s, a tricuspid regurgitation pressure gradient (TRPG) of 82.8 mmHg, and an estimated systolic pulmonary artery pressure (SPAP) of 86 mm Hg, consistent with a diagnosis of severe pulmonary hypertension. Given the suspected presence of tumor emboli within pulmonary vessels, PTTM was considered a potential underlying cause.
Despite the discontinuation of entrectinib—due to the inability to rule out drug-induced lung injury—and the initiation of empirical antibiotic therapy with meropenem and methylprednisolone, the patient’s condition continued to deteriorate. The worsening of the pleural effusion necessitated bilateral thoracentesis for symptom relief. As pulmonary function decreases, entrectinib is reintroduced on the basis of the hypothesis that tumor embolization within pulmonary arteries drives disease progression, although clinically, there was no evidence of hematogenous metastasis to other organs.
Despite these efforts, the patient’s respiratory distress worsened, and he passed away seven weeks after hospitalization. An autopsy was performed with the family’s consent.
Autopsy Findings
Macroscopic Findings:
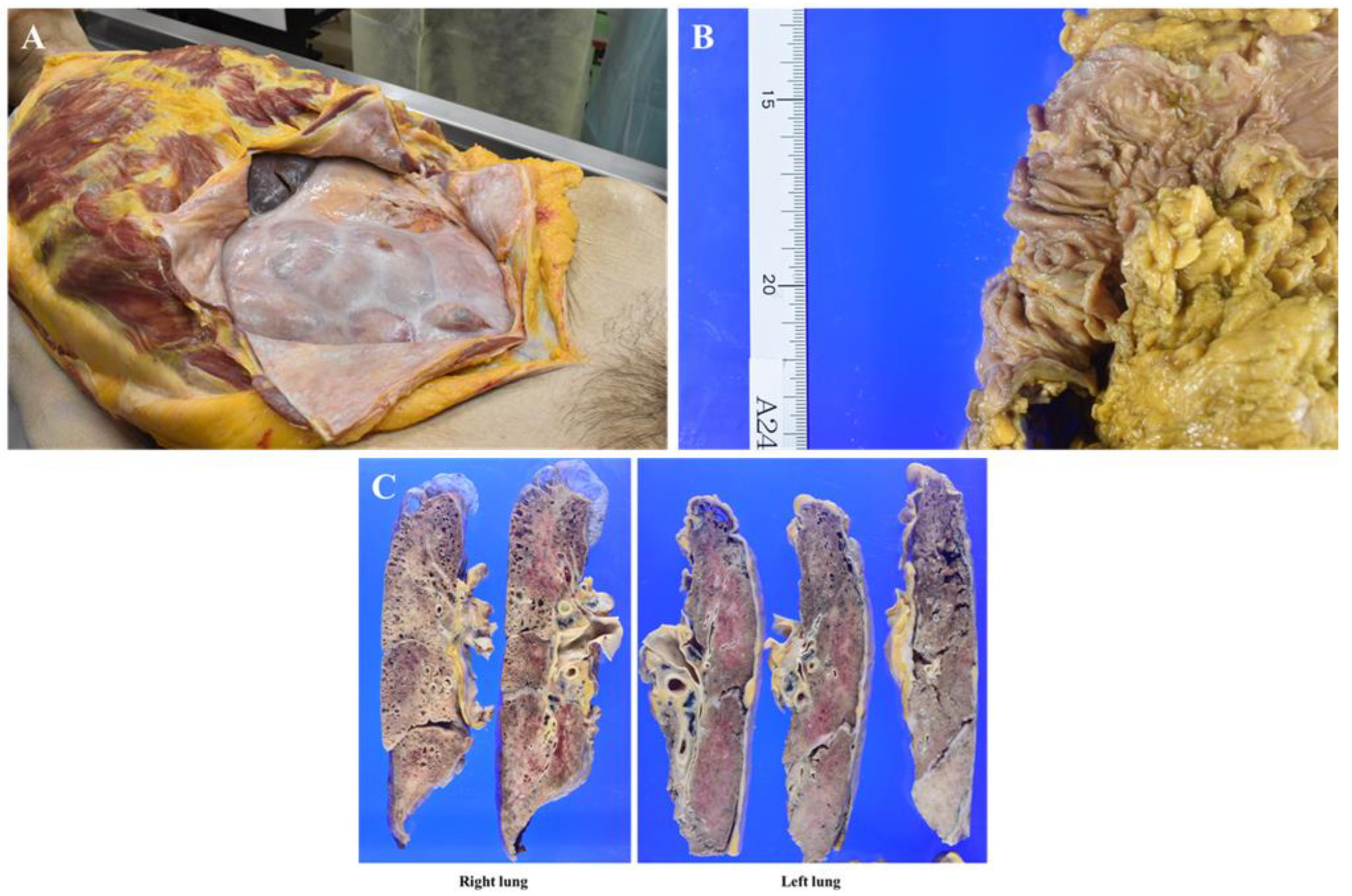
In the right thoracic cavity, minor adhesions were observed alongside 500 mL of clear yellowish pleural effusion. In contrast, the left thoracic cavity exhibited severe adhesions, making lung removal difficult, with almost no pleural effusion present. The abdominal cavity displayed extensive fibrous adhesions, with white fibrous tissue uniformly covering the serosal surfaces of the intestinal tract, leading to firm intersegmental adhesions (
Figure 4A). No discernible mass lesions were present on the serosal surfaces, and ascites was minimal. The primary transverse colon lesion had significantly regressed, leaving a 3 cm square mucosal area containing several polyps, the largest measuring up to 1 cm (
Figure 4B). No additional mass lesions, including disseminated foci, were detected in the abdominal cavity.
Prominent lymph node enlargement was noted in the abdominal cavity and along the periaorta. The lungs exhibited diffuse consolidation with areas of sclerosis, along with apical bullae and microcystic changes, which are likely associated with smoking (
Figure 4C). No metastatic mass lesions were found within the lung parenchyma; however, multiple hilar lymphadenopathies were observed. The heart, liver, and kidneys showed no significant abnormalities.
Figure 4.
Macroscopic autopsy findings. A: The intestinal tract is uniformly covered with white fibrous tissue, leading to extensive adhesion between structures. B: The primary lesion in the transverse colon has markedly decreased in size following effective treatment, appearing as clustered polyps within a 3 cm square area, the largest measuring up to 1 cm. C: Cross-sections of both lungs revealing diffuse consolidation, apical bullae, and multiple small cysts. The left lung was adherent to the parietal pleura, with enlargement of both hilar lymph nodes.
Figure 4.
Macroscopic autopsy findings. A: The intestinal tract is uniformly covered with white fibrous tissue, leading to extensive adhesion between structures. B: The primary lesion in the transverse colon has markedly decreased in size following effective treatment, appearing as clustered polyps within a 3 cm square area, the largest measuring up to 1 cm. C: Cross-sections of both lungs revealing diffuse consolidation, apical bullae, and multiple small cysts. The left lung was adherent to the parietal pleura, with enlargement of both hilar lymph nodes.
Microscopic Findings:
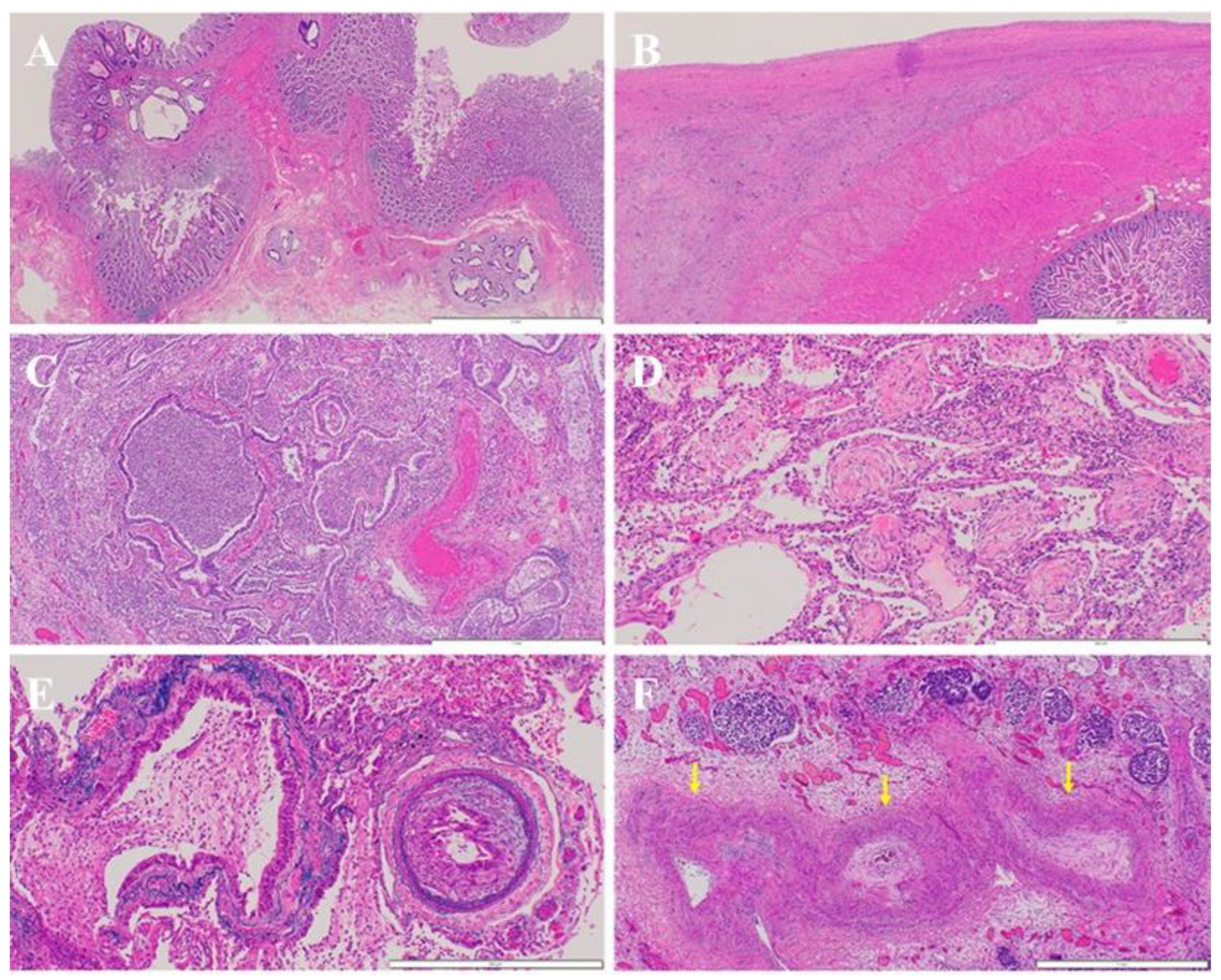
At the primary transverse colon site, chemotherapy has demonstrated significant efficacy, with well- to moderately differentiated tubular adenocarcinomas present in several polypoid areas within the mucosal layer. Scattered microscopic remnants were identified in the submucosal layer, although no evidence of lymphatic or venous invasion was observed (
Figure 5A). Adenocarcinoma with extensive fibrosis was diffusely spread across the intestinal surface, leading to adherence and direct invasion of adjacent organs, including the stomach, liver, bladder, and diaphragm (
Figure 5B). Additionally, widespread dissemination of adenocarcinoma was noted in the left thoracic cavity. Tumor emboli within lymphatic vessels have been identified across multiple organs, including the stomach, bladder, and pleura. With respect to lymph node metastasis, multiple metastatic lesions were present in the abdominal cavity, pulmonary hilum, and para-aortic region.
In the lungs, diffuse bronchopneumonia and organizing pneumonia were observed (
Figure 5C, D), likely resulting from aspiration pneumonia, although drug-related effects contributing to organizing pneumonia could not be entirely excluded. Histopathological findings consistent with PTTM were identified in both lungs, characterized by tumor emboli, fibrous intimal thickening, and thrombus formation within numerous small pulmonary arteries (
Figure 5E, F). Additionally, a small number of embolized lymphatic vessels were observed. No hematogenous metastatic lesions were found in the lung tissue, highlighting the distinct vascular involvement characteristic of PTTM.
Figure 5.
Microscopic findings at autopsy. A: At the primary site in the transverse colon, chemotherapy was highly effective, with well- to moderately differentiated adenocarcinomas observed in several polypoid regions within the mucosal layer. Microscopically, scattered remnants were detected in the submucosal layer, but no evidence of lymphatic or venous invasion was found (white bar at bottom right: 2 mm). B: Adenocarcinoma with severe fibrosis diffusely infiltrated the intestinal tract, leading to adhesion to its surface (white bar at bottom right: 2 mm). C and D: In the lungs, diffuse bronchopneumonia (C) and organizing pneumonia (D) were observed. E and F: Findings consistent with PTTM were identified. A small pulmonary artery was embolized by adenocarcinoma (E; Victoria blue and hematoxylin‒eosin staining), whereas another pulmonary artery exhibited a narrowed lumen due to intimal thickening (F; arrows) (E: white bar at bottom right: 500 µm; F: white bar at bottom right: 1 mm).
Figure 5.
Microscopic findings at autopsy. A: At the primary site in the transverse colon, chemotherapy was highly effective, with well- to moderately differentiated adenocarcinomas observed in several polypoid regions within the mucosal layer. Microscopically, scattered remnants were detected in the submucosal layer, but no evidence of lymphatic or venous invasion was found (white bar at bottom right: 2 mm). B: Adenocarcinoma with severe fibrosis diffusely infiltrated the intestinal tract, leading to adhesion to its surface (white bar at bottom right: 2 mm). C and D: In the lungs, diffuse bronchopneumonia (C) and organizing pneumonia (D) were observed. E and F: Findings consistent with PTTM were identified. A small pulmonary artery was embolized by adenocarcinoma (E; Victoria blue and hematoxylin‒eosin staining), whereas another pulmonary artery exhibited a narrowed lumen due to intimal thickening (F; arrows) (E: white bar at bottom right: 500 µm; F: white bar at bottom right: 1 mm).
The tumor mutational burden (TMB) was 1.26 mut/Mb, indicating a relatively low level of genetic alterations.
A definitive postmortem diagnosis of PTTM was established, suggesting that the condition led to severe PH and right heart failure, progressing rapidly over a short period.