Submitted:
25 July 2025
Posted:
29 July 2025
You are already at the latest version
Abstract
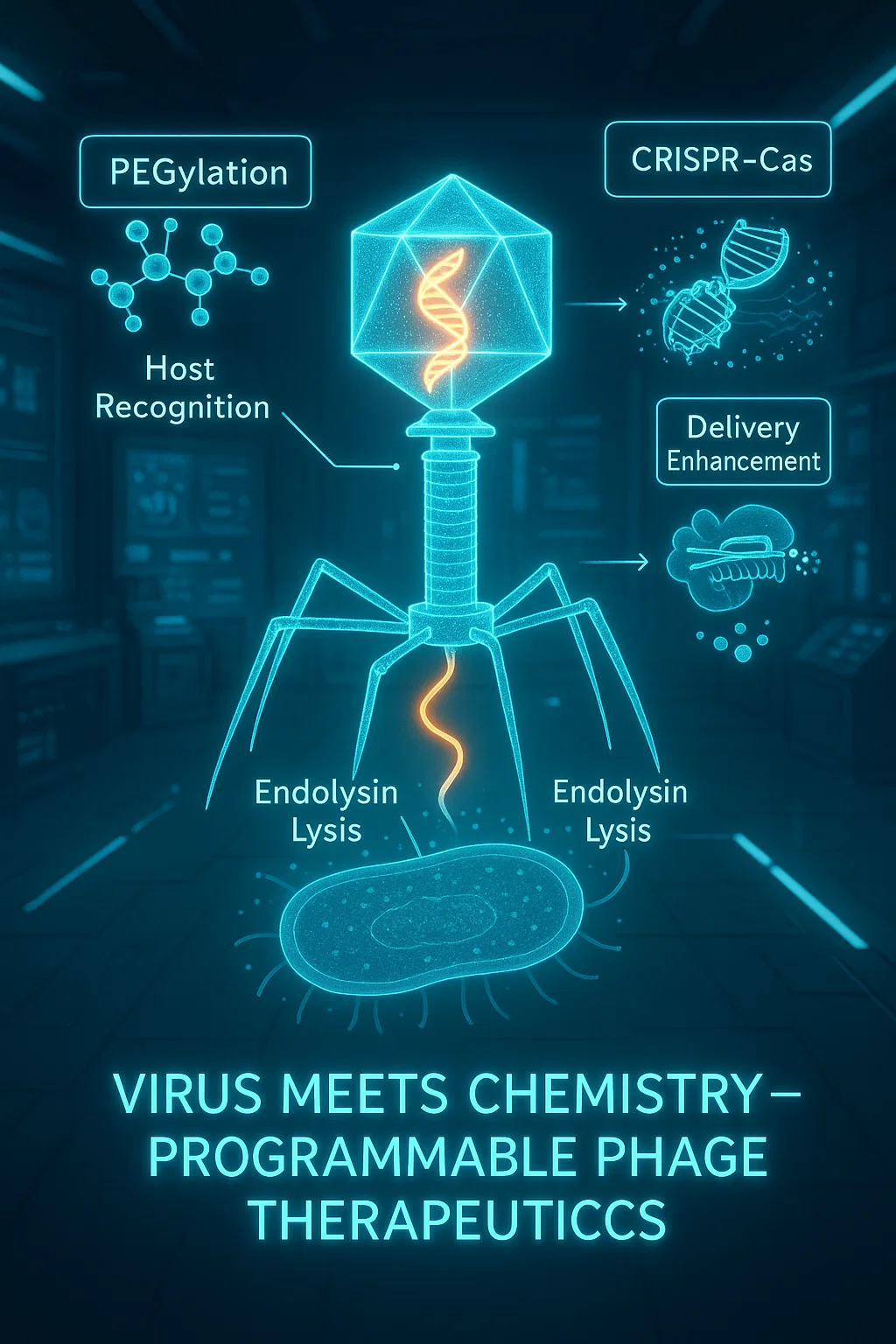
Keywords:
Introduction
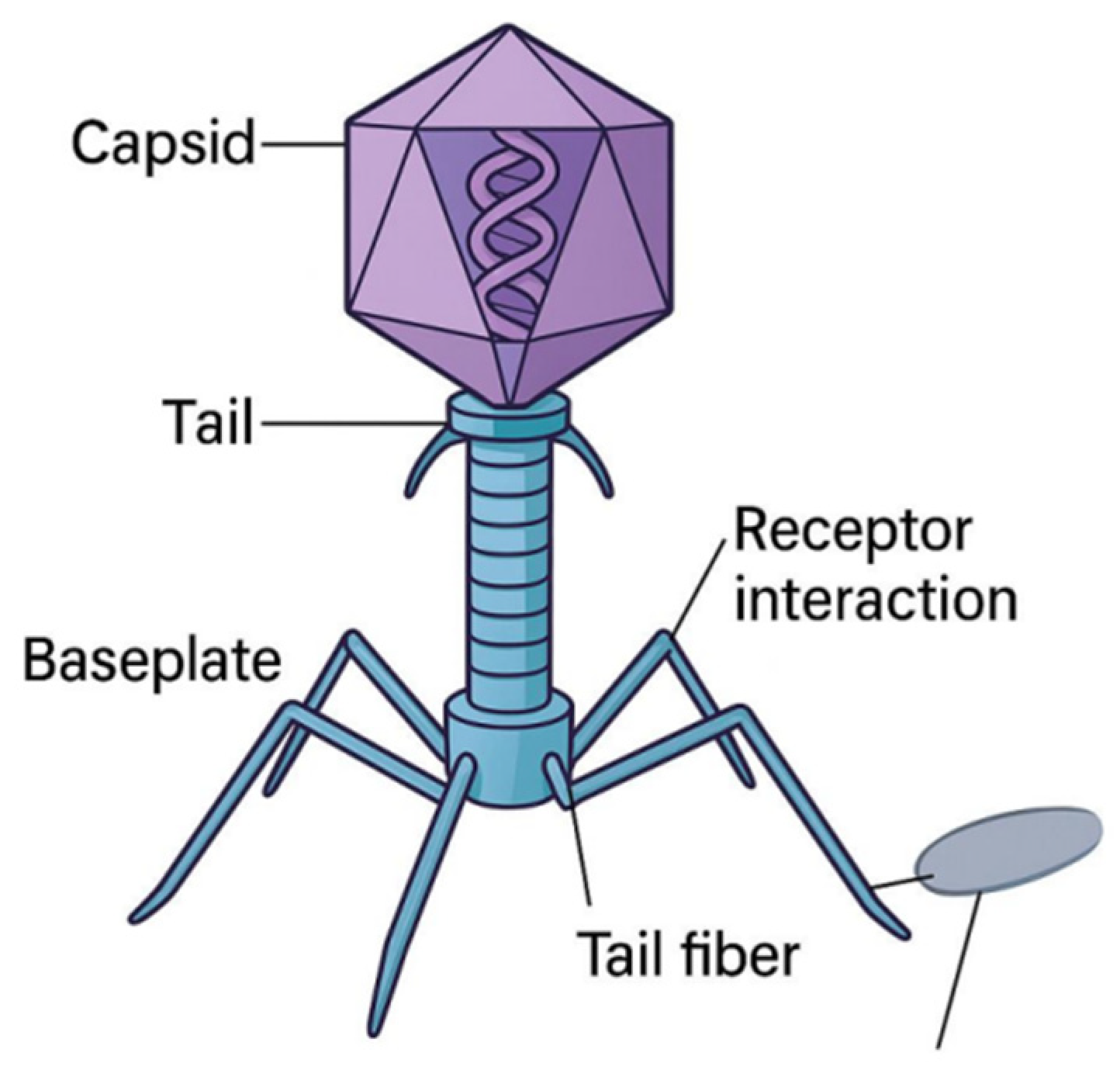
Molecular Architecture of Bacteriophages
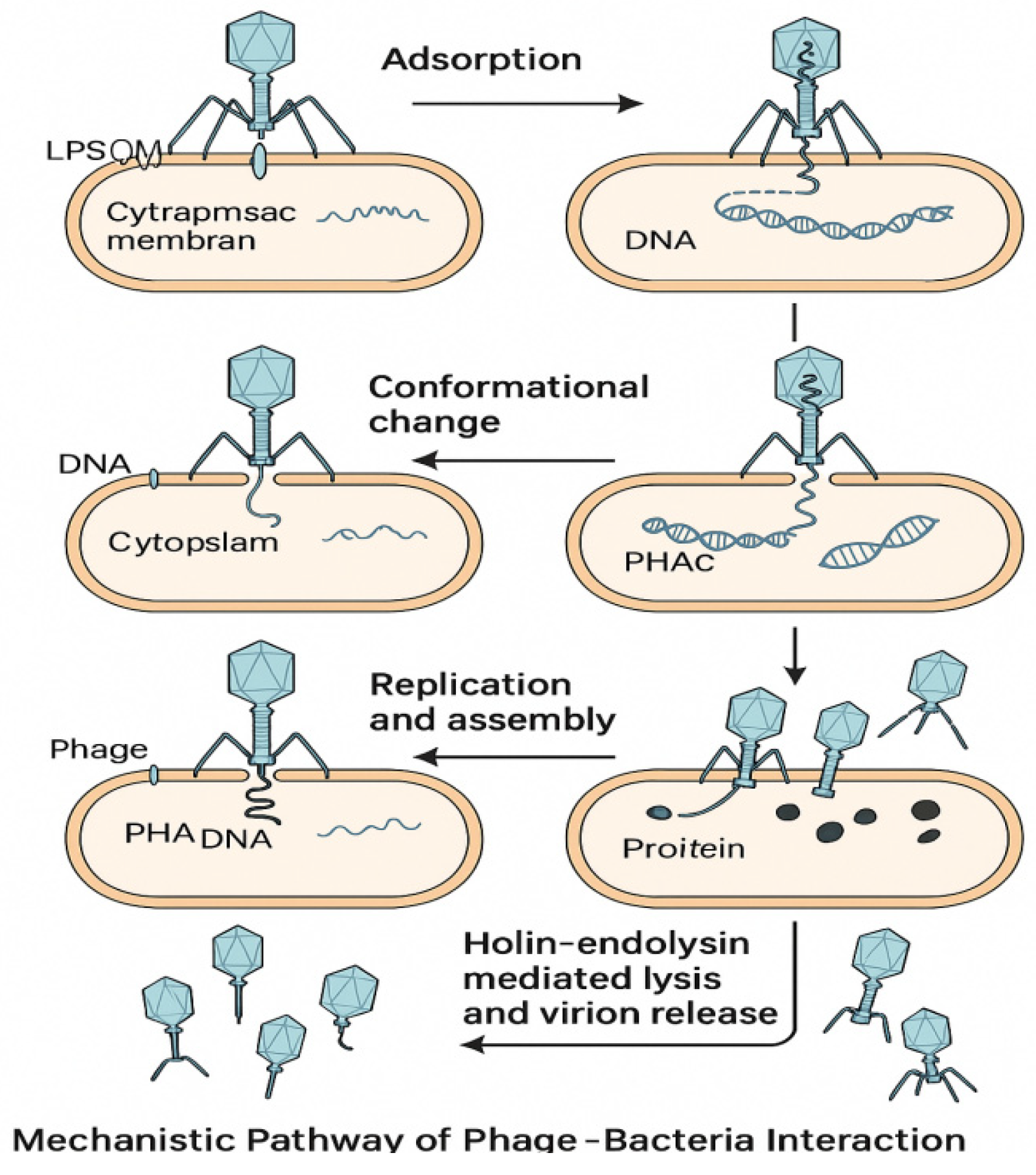
Mechanistic Chemistry of Phage–Bacteria Interaction
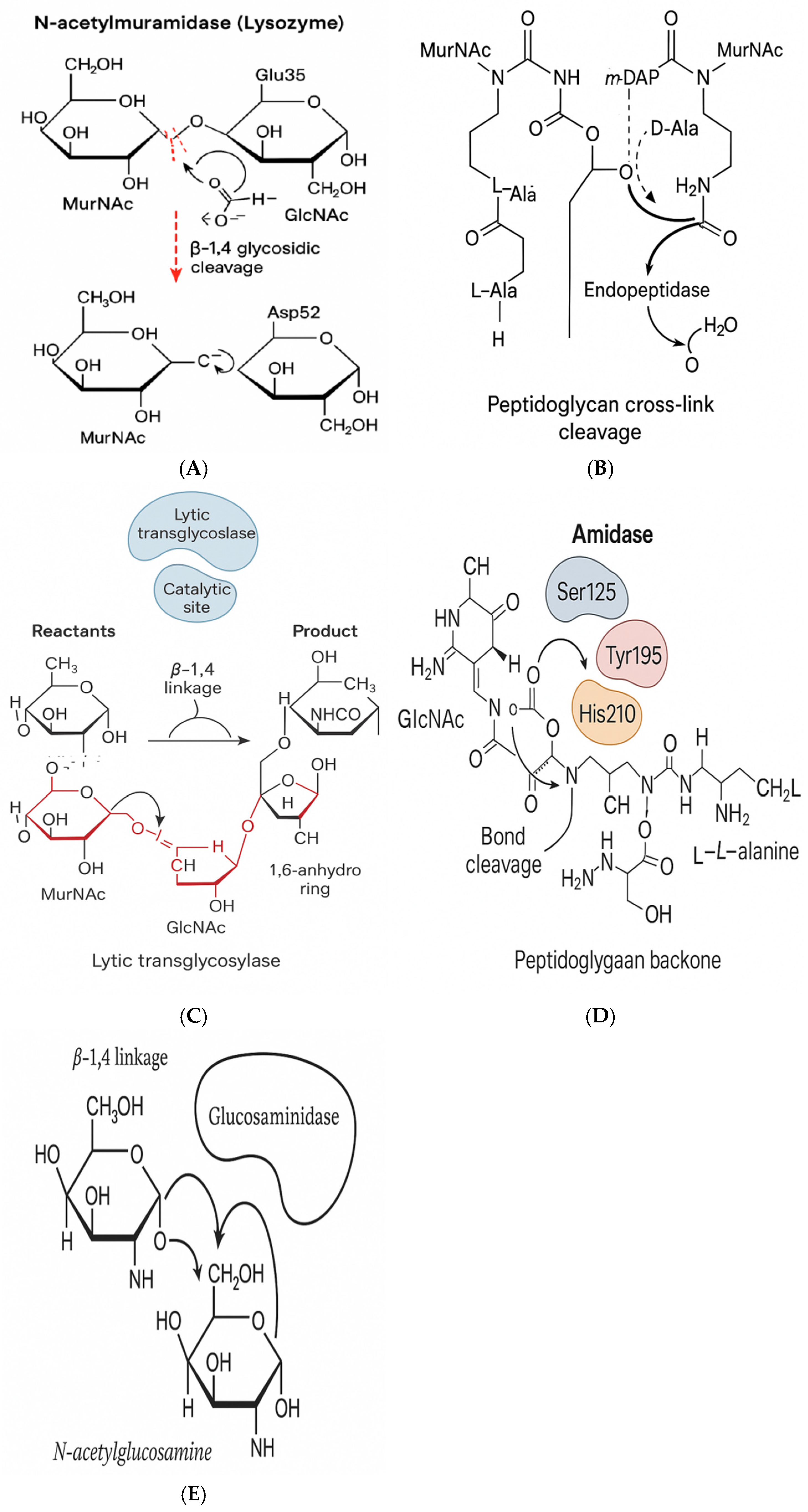
Enzymatic Lysis: Endolysins, Holins, and Catalytic Strategies
- Endolysins are peptidoglycan hydrolases that cleave bonds within the bacterial cell wall. They are categorized by their catalytic targets:
- (a)
- N-acetylmuramidases: cleave β-1,4 bonds between MurNAc and GlcNAc.
- (b)
- Amidases: hydrolyze amide bonds between MurNAc and L-Ala.
- (c)
- Endopeptidases: target peptide bridges.
- (d)
- Transglycosylases: generate 1,6-anhydro rings [22].
- Enzybiotics: recombinant antimicrobials with high specificity.
- Fusion constructs: endolysins linked to targeting peptides.
- Synthetic holins: mimicked by amphipathic drug delivery peptides [25].
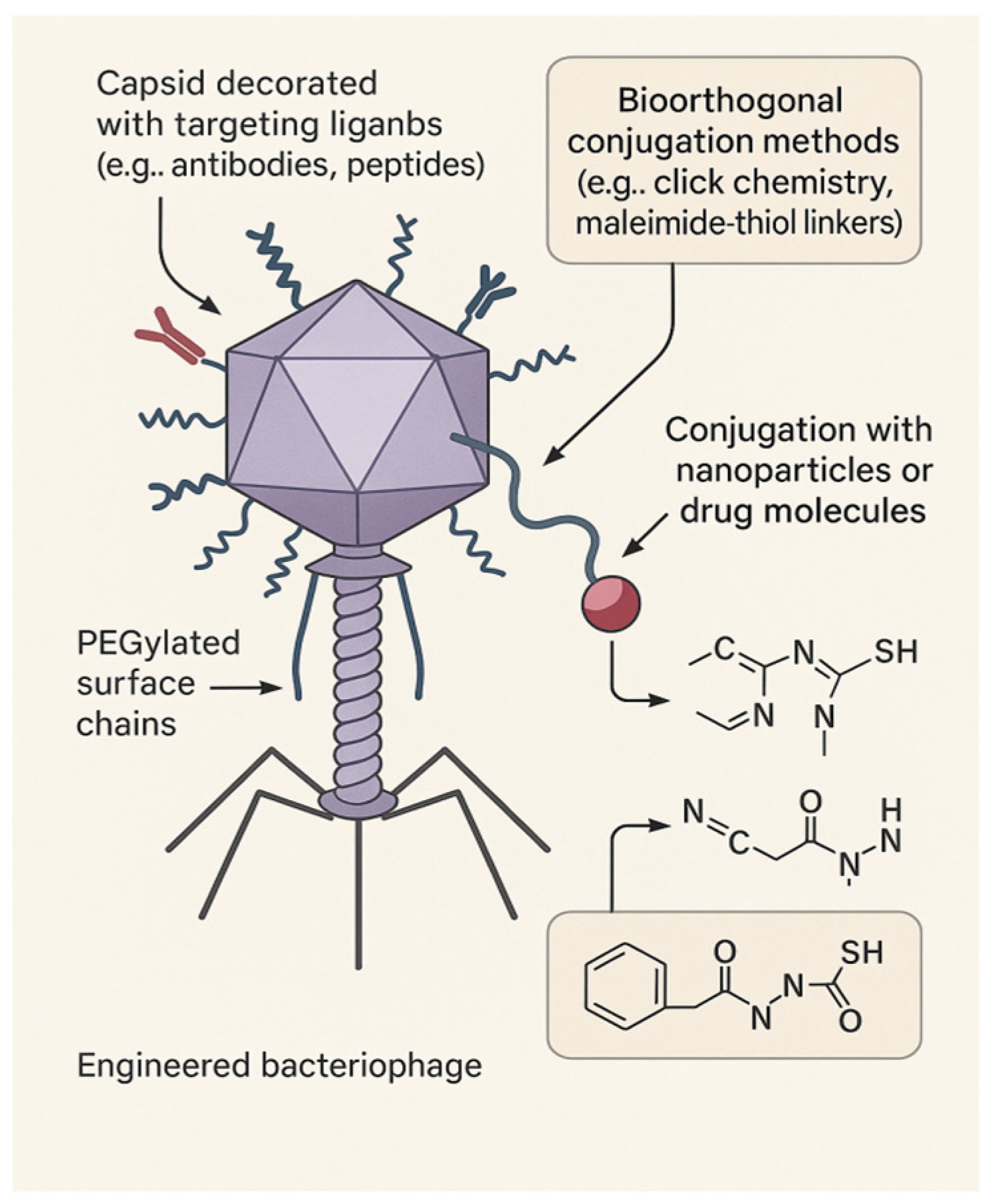
Capsid Engineering
PEGylation
Phage Display
Phage–Nanoparticle Hybrids
- ⮚
- Gold–phage: For photo-thermal ablation of biofilms.
- ⮚
- Magnetic–phage: For guided drug delivery.
- ⮚
- Drug–phage: Co-deliver antibiotics and matrix-degrading enzymes [29].
- ⮚
- Synthetic Phages
Therapeutic Applications and Challenges
Infectious Disease
Biofilm Disruption
Targeted Drug and Gene Delivery
- CRISPR–Cas antimicrobials,
- Antibiotics or peptides,
- Imaging or gene therapy payloads via ligand targeting [33].
Challenges
- Rapid clearance by immune cells
- Variable tissue distribution
- Lack of standardized regulatory pathways [34].
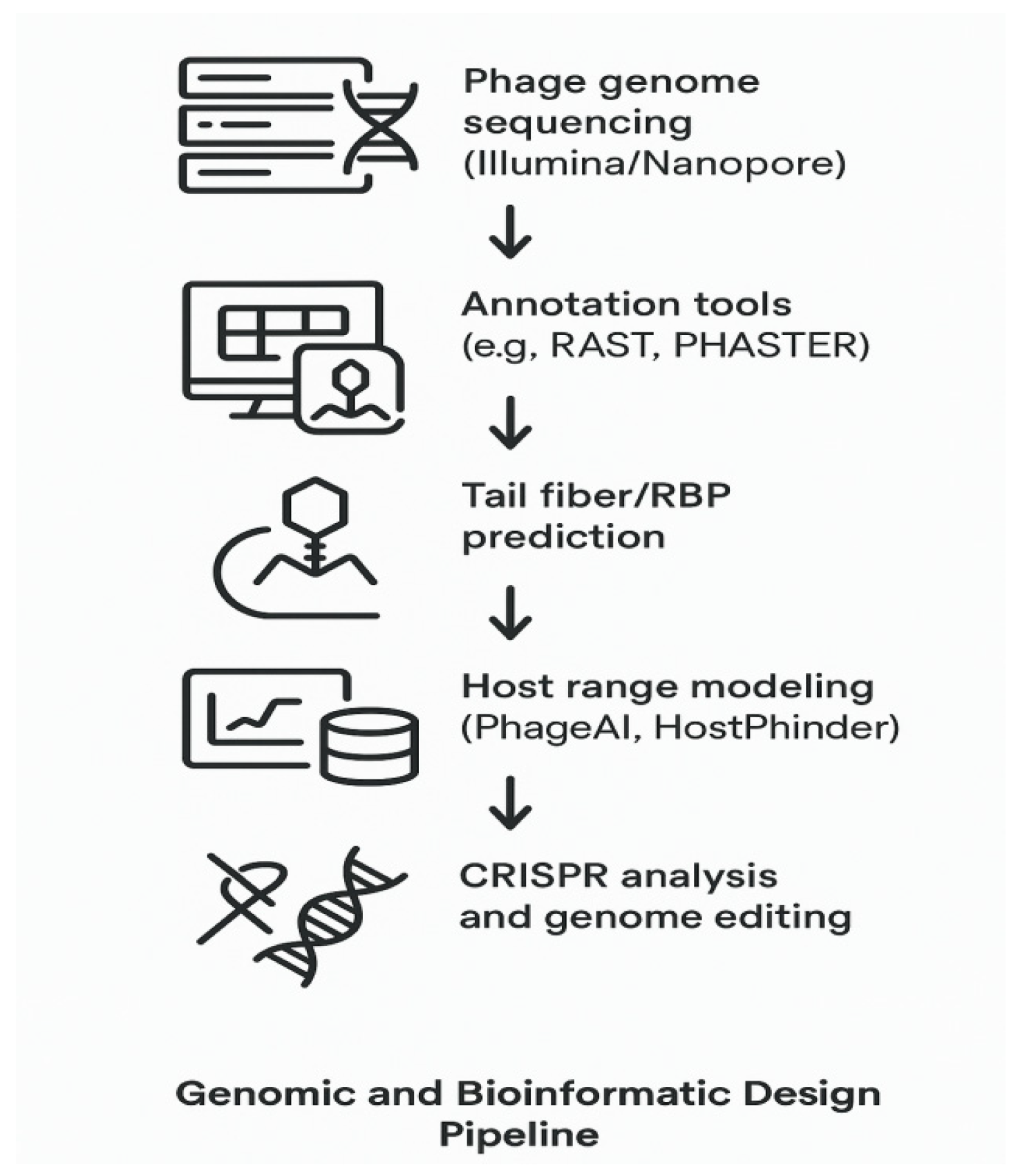
Genome Sequencing and Annotation
Comparative Genomics and Host Range Prediction
CRISPR–Phage Co-Evolution
Synthetic Genome Engineering
Toward Personalization
Conclusion and Outlook
- Prioritize collaboration between chemists, structural biologists, and microbiologists.
- Frame phage therapy as a modular chemical approach—not a fallback.
- Build curated libraries of phage structures and enzyme domains.
- Push for regulatory pathways tailored to customizable biologics.
- Integrate AI-driven selection and genomic matching into therapy design.
References
- Lee JH, Shin H, Ryu S. Bacteriophage-based antimicrobials and their potential use against multidrug-resistant bacteria. J Microbiol. 2022;60(2):91–98.
- Oduor JMO, Mabrouk MS, Elbadawy HM, et al. Bacteriophage therapy: a promising alternative in the age of antimicrobial resistance. Front Pharmacol. 2022;13:872816.
- Lu TK, Chan BK, Sahay G. Advances in the engineering of phages and their applications in medicine. Trends Biotechnol. 2023;41(1):24–38.
- Li X, Li X, Bai X, et al. Tail fiber engineering of phages to alter host specificity. Biotechnol Adv. 2023;62:108061.
- Witte AK, Fieseler L. Lysis mechanisms of bacteriophages. Viruses. 2023;15(3):726.
- Alsaadi EA, Alsaedi MH, Almalki MA, et al. Structural insights into endolysin–peptidoglycan interactions. Int J Mol Sci. 2022;23(9):5106.
- Nam KH, Kim MS, Lee K, et al. PEGylation improves pharmacokinetics and therapeutic efficacy of phage-based therapies. Pharmaceutics. 2022;14(5):980.
- Akello J, Udaondo Z, Ross A, et al. Genomic insights into phage–host specificity. Microorganisms. 2022;10(4):679.
- Abdelkader K, Knecht LE, Bayer A, et al. Machine learning for personalized bacteriophage therapy. Nat Biotechnol. 2023;41(4):464–475.
- Shen Y, Wang D, Zhao Y, et al. Modular architecture and structural diversity of tailed bacteriophages. Microbiol Spectr. 2022;10(6):e01633–22.
- Persson T, Leung CY, O’Connell K, et al. Supramolecular principles in phage capsid assembly. ACS Nano. 2023;17(1):131–142.
- Batra H, Konieczny A. Internal pressure in viral capsids: lessons from phage DNA packaging. Viruses. 2022;14(9):1994.
- Vos M, Louwen R, Kropinski AM, et al. Phage tail contraction and genome injection mechanisms. Trends Microbiol. 2023;31(5):389–398.
- Ogilvie LA, Jones BV. Host recognition strategies in phage infection. Curr Opin Virol. 2022;54:101237.
- Ceyssens PJ, Lavigne R. Phage structural protein scaffolding and self-assembly. Front Microbiol. 2022;13:865229.
- Li F, Zhou W, Liang Y, et al. Phage–bacteria interface: binding, injection, and beyond. Microbiol Res. 2022;261:127042.
- Niu Y, Xu M, Liu Y, et al. Molecular modeling of phage tail fiber interactions. Comput Struct Biotechnol J. 2023;21:2223–2233.
- Murthy D, Podgorski J, Grayson P, et al. Mechanics of phage DNA injection under high pressure. Biophys J. 2022;121(4):785–793.
- Zhang Y, Wang Y, Zhu Z, et al. Catalytic profiling of phage endolysins. Biochem Biophys Res Commun. 2022;603:104–110.
- Doss J, Culbertson K, Hahn D, Camacho J. Chemical insights into timed lysis in phage therapy. Antibiotics. 2022;11(7):920.
- Gu J, Xu W, Lei L, et al. Advances in engineering lytic enzymes. Viruses. 2023;15(2):456.
- Lopes A, Oliveira H, Melo LDR, et al. Domain-specific activity of bacteriophage-derived endolysins. Microorganisms. 2022;10(3):521.
- Turner D, Topka G, Haggard-Ljungquist E. Structure–function of lytic phage enzymes. Front Microbiol. 2022;13:852323.
- Yin Y, Liu X, Zhao J, et al. Holins and spanins in Gram-negative lysis. Viruses. 2023;15(4):766.
- Ghosh D, Bhattacharya D. Synthetic holins and peptide analogues. ACS Synth Biol. 2023;12(2):317–325.
- Chen Y, Zhang C, Ma Y, et al. Chemical modification of phage capsids for precision delivery. ACS Chem Biol. 2023;18(3):524–534.
- Arndt D, Suh JW, Ryu J. PEGylation enhances phage longevity in systemic circulation. Nanomedicine. 2022;40:102504.
- Derda R, Tang SKY, Whitesides GM. Phage display for bacterial targeting. Bioconjug Chem. 2023;34(1):78–86.
- Ramesh N, Haldar S, Pillai S, et al. Phage–nanoparticle hybrids for antimicrobial applications. J Nanobiotechnology. 2022;20(1):248.
- Fernández-Ruiz I, Brown R, Marwick C, et al. Synthetic phages for targeted bacterial killing. Nat Chem Biol. 2024;20(2):142–152.
- Law N, Logan C, Yung G, et al. Clinical evidence for personalized phage therapy. Clin Infect Dis. 2023;76(8):1367–1375.
- Baker JL, Ma L, Kim M, et al. Anti-biofilm phage hydrogels and delivery systems. ACS Appl Mater Interfaces. 2023;15(7):9333–9345.
- Kim S, Jung J, Kim M, et al. Targeted phage delivery of CRISPR and antibiotics. Mol Ther. 2022;30(9):3119–3132.
- Tan D, Zhang Y, Ruan L, et al. Regulatory outlook on phage therapeutics. Trends Biotechnol. 2023;41(6):707–719.
- Jahn M, Bunk B, Schildkraut J, et al. Genomic annotation and biosafety of therapeutic phages. Microbiol Spectr. 2022;10(4):e00284-22.
- Rhoads DD, Wolcott RD, Kuskowski MA, et al. Host range prediction using AI-assisted phage tools. NPJ Biofilms Microbiomes. 2023;9(1):30.
- Díaz-Muñoz SL, Koskella B. Phage–CRISPR coevolution in the wild. Curr Opin Microbiol. 2022;66:25–31.
- Pinto R, Silva YJ, Costa L, et al. Genome editing and lysogeny control in synthetic phages. Viruses. 2022;14(11):2395.
- Kutter E, Debarbieux L. Toward precision phage therapy in the clinic. Microbiol Spectr. 2023;11(1):e03538-22.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



