Submitted:
25 July 2025
Posted:
28 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Overview of the Evolving Landscape of Nephron Replacement Strategies
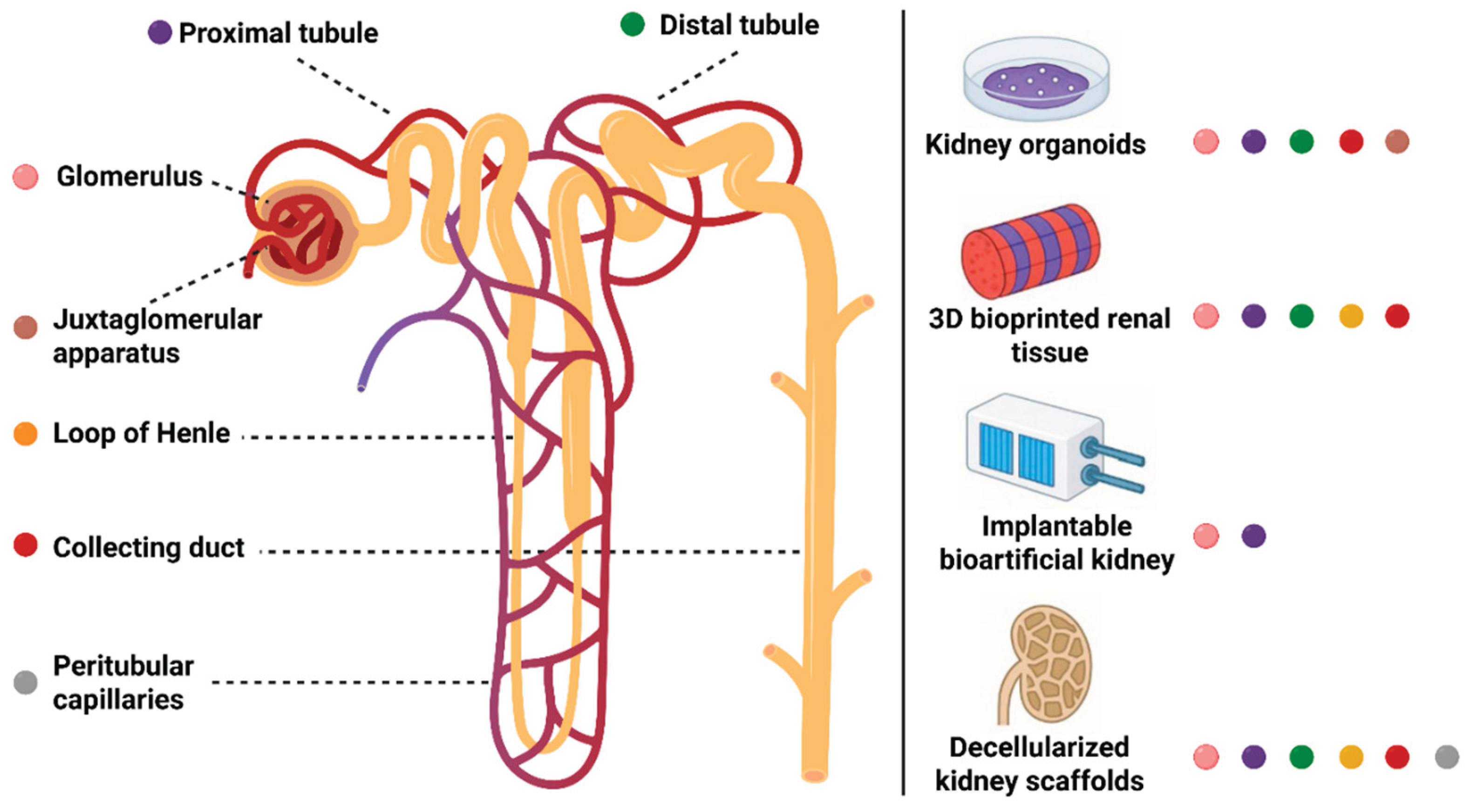
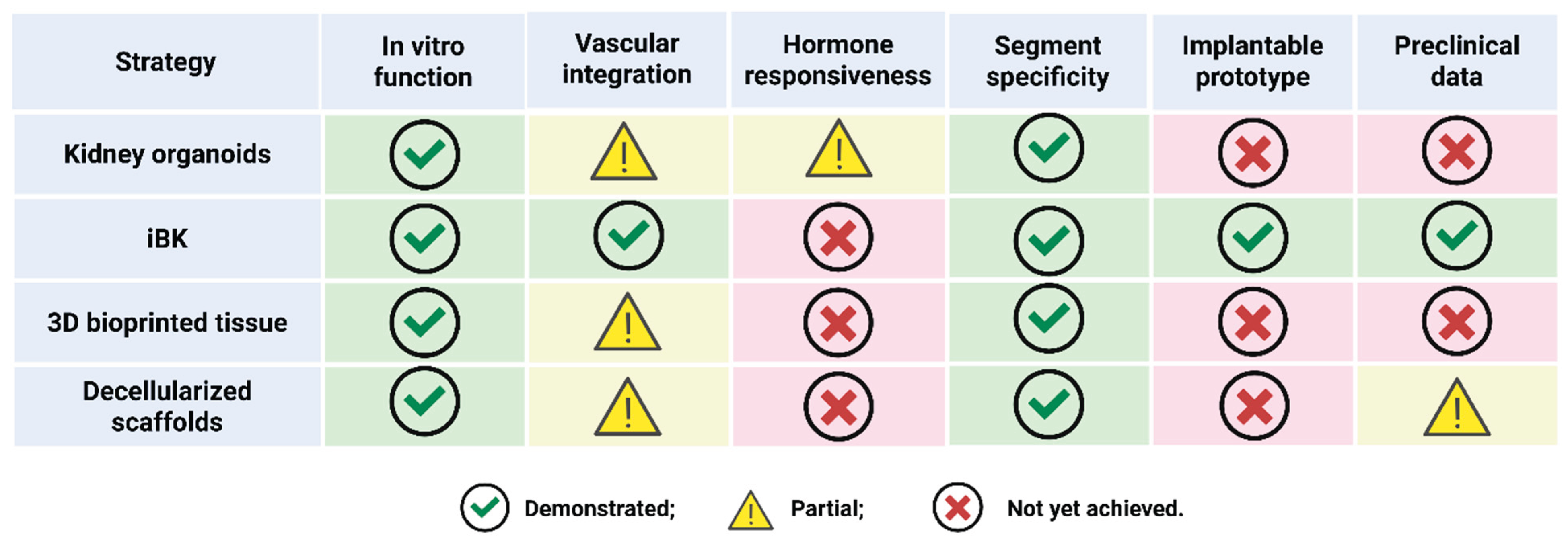
2.1. Kidney Organoids
- Absence of a vascular network, which restricts nutrient delivery and size,
- Lack of a urine drainage system, precluding physiological excretion,
2.2. Implantable Bioartificial Kidney (iBK) Devices
- A hemofilter, constructed from silicon nanopore membranes, emulates the glomerular filtration barrier by permitting selective ultrafiltration of plasma under native arterial pressure. These membranes are fabricated through microelectromechanical systems engineering, enabling high precision and resistance to fouling, without the need for anticoagulation,
- Sustaining epithelial cell viability and function under physiological shear stress,
- Preventing fibrosis and immune rejection in the absence of systemic immunosuppression,
- Ensuring long-term durability of membrane materials and preventing device fouling or occlusion,
2.3. 3D Bioprinted Renal Tissues
- Achieving hierarchical vascularization;
- Maintaining long-term cell viability under physiological pressure;
- Scaling constructs to clinically relevant sizes without compromising function;
2.4. Decellularized Kidney Scaffolds
- Efficient and selective recellularization of glomerular, tubular, and vascular compartments,
- Sourcing sufficient quantities of autologous or immunocompatible cells,
- Achieving functional integration and perfusion after implantation,
3. The Nephron as a Blueprint for Modular Kidney Replacement
3.1. Glomerulus (Filtration)
3.2. Proximal Tubule (Reabsorption and Secretion)
3.3. Loop of Henle (Countercurrent Concentration)
3.4. Distal Tubule (Electrolyte Regulation)
3.5. Collecting Duct (Water and Acid–Base Regulation)
3.6. Juxtaglomerular Apparatus (Endocrine and Autoregulatory Function)
3.7. Peritubular Capillaries (Microvascular Support)
4. From Technologies to Translation: Integration Challenges and Clinical Pathways
5. Conclusions and Ethical Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Li, M.-J.; Liu, H.-Y.; Zhang, Y.-Q.; Li, S.-R.; Zhang, J.-H.; Li, R. Global Burden of Chronic Kidney Disease and Its Attributable Risk Factors (1990-2021): An Analysis Based on the Global Burden of Disease Study. Front Endocrinol (Lausanne) 2025, 16, 1563246. [CrossRef]
- Francis, A.; Harhay, M.N.; Ong, A.C.M.; Tummalapalli, S.L.; Ortiz, A.; Fogo, A.B.; Fliser, D.; Roy-Chaudhury, P.; Fontana, M.; Nangaku, M.; et al. Chronic Kidney Disease and the Global Public Health Agenda: An International Consensus. Nature Reviews Nephrology 2024 20:7 2024, 20, 473–485. [CrossRef]
- Nkunu, V.; Tungsanga, S.; Diongole, H.M.; Sarki, A.; Arruebo, S.; Caskey, F.J.; Damster, S.; Donner, J.A.; Jha, V.; Levin, A.; et al. Landscape of Kidney Replacement Therapy Provision in Low- and Lower-Middle Income Countries: A Multinational Study from the ISN-GKHA. PLOS Global Public Health 2024, 4, e0003979. [CrossRef]
- Bello, A.K.; Okpechi, I.G.; Osman, M.A.; Cho, Y.; Htay, H.; Jha, V.; Wainstein, M.; Johnson, D.W. Epidemiology of Haemodialysis Outcomes. Nat Rev Nephrol 2022, 18, 378-395. [CrossRef]
- Lanini, I.; Samoni, S.; Husain-Syed, F.; Fabbri, S.; Canzani, F.; Messeri, A.; Mediati, R.D.; Ricci, Z.; Romagnoli, S.; Villa, G. Palliative Care for Patients with Kidney Disease. J Clin Med 2022, 11, 3923. [CrossRef]
- Kupiec-Weglinski, J.W. Grand Challenges in Organ Transplantation. Frontiers in Transplantation 2022, 1, 897679. [CrossRef]
- Kolesnyk, M.; Korol, L.; Shifris, I.; Stepanova, N.; Voroniak, O.; Shuba, I. Immunological determinants of long-term kidney graft survival as therapeutic targets. Ukr J Nephrol Dialys. 2025, 2, 98-111. [CrossRef]
- Salani, M.; Roy, S.; Fissell, W.H. Innovations in Wearable and Implantable Artificial Kidneys. Am J Kidney Dis. 2018, 72, 745–751. [CrossRef]
- Copur, S.; Tanriover, C.; Yavuz, F.; Soler, M.J.; Ortiz, A.; Covic, A.; Kanbay, M. Novel Strategies in Nephrology: What to Expect from the Future? Clin Kidney J 2023, 16, 230–244. [CrossRef]
- Bonventre, J. V.; Hurst, F. P.; West ,M.; Wu I.; Roy-Chaudhury, P.; Sheldon M. A Technology Roadmap for Innovative Approaches to Kidney Replacement Therapies: A Catalyst for Change. Clin J Am Soc Nephrol. 2019, 14, 1539-1547. [CrossRef]
- Guo, X.; Liu, B.; Zhang, Y.; Cheong, S.; Xu, T.; Lu, F.; He, Y. Decellularized Extracellular Matrix for Organoid and Engineered Organ Culture. J Tissue Eng 2024, 15, 20417314241300386. [CrossRef]
- Zheng, K.; Chai, M.; Luo, B.; Cheng, K.; Wang, Z.; Li, N.; Shi, X. Recent Progress of 3D Printed Vascularized Tissues and Organs. Smart Mater Med 2024, 5, 183–195. [CrossRef]
- Christou, C.D.; Vasileiadou, S.; Sotiroudis, G.; Tsoulfas, G. Three-Dimensional Printing and Bioprinting in Renal Transplantation and Regenerative Medicine: Current Perspectives. J Clin Med 2023, 12, 6520. [CrossRef]
- Chambers, B.E.; Weaver, N.E.; Wingert, R.A. The “3Ds” of Growing Kidney Organoids: Advances in Nephron Development, Disease Modeling, and Drug Screening. Cells 2023, 12, 549. [CrossRef]
- Tekguc, M.; Gaal, R.C.V.; Uzel, S.G.M.; Gupta, N.; Riella, L. V.; Lewis, J.A.; Morizane, R. Kidney Organoids: A Pioneering Model for Kidney Diseases. Transl Res 2022, 250, 1-17.. [CrossRef]
- Unbekandt, M.; Davies, J.A. Dissociation of Embryonic Kidneys Followed by Reaggregation Allows the Formation of Renal Tissues. Kidney Int 2010, 77, 407–416. [CrossRef]
- Xia, Y.; Nivet, E.; Sancho-Martinez, I.; Gallegos, T.; Suzuki, K.; Okamura, D.; Wu, M.Z.; Dubova, I.; Esteban, C.R.; Montserrat, N.; et al. Directed Differentiation of Human Pluripotent Cells to Ureteric Bud Kidney Progenitor-Like Cells. Nat Cell Biol 2013, 15, 1507–1515. [CrossRef]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Maier, B.; Baillie, G.J.; Ferguson, C.; Parton, R.G.; Wolvetang, E.J.; Roost, M.S.; De Sousa Lopes, S.M.C.; et al. Kidney Organoids from Human IPS Cells Contain Multiple Lineages and Model Human Nephrogenesis. Nature 2015, 526, 564–568. [CrossRef]
- Tabibzadeh, N.; Morizane, R. Advancements in Therapeutic Development: Kidney Organoids and Organs-on-a-Chip. Kidney Int 2024, 105, 702708. [CrossRef]
- Liu, Q.; Yue, L.; Deng, J.; Tan, Y.; Wu, C. Progress and Breakthroughs in Human Kidney Organoid Research. Biochem Biophys Rep 2024, 39, 101736. [CrossRef]
- Wilson, S.B.; Vanslambrouck, J.M.; Murphy, A.; Neavin, D.R.; Powell, J.E.; Howden, S.E.; Little, M.H. Classification of Indeterminate and Off-Target Cell Types within Human Kidney Organoid Differentiation. bioRxiv 2025, 2025.05.16.654519. [CrossRef]
- Goux Corredera, I.; Amato, G.; Moya-Rull, D.; Garreta, E.; Montserrat, N. Unlocking the Full Potential of Human Pluripotent Stem Cell–Derived Kidney Organoids through Bioengineering. Kidney Int 2025, 108, 38–47. [CrossRef]
- Papamichail, L.; Koch, L.S.; Veerman, D.; Broersen, K.; van der Meer, A.D. Organoids-on-a-Chip: Microfluidic Technology Enables Culture of Organoids with Enhanced Tissue Function and Potential for Disease Modeling. Front Bioeng Biotechnol 2025, 13, 1515340. [CrossRef]
- Menéndez, A.B.C.; Du, Z.; van den Bosch, T.P.P.; Othman, A.; Gaio, N.; Silvestri, C.; Quirós, W.; Lin, H.; Korevaar, S.; Merino, A.; et al. Creating a Kidney Organoid-Vasculature Interaction Model Using a Novel Organ-on-Chip System. Sci Rep 2022, 12, 20699. [CrossRef]
- Maggiore, J.C.; LeGraw, R.; Przepiorski, A.; Velazquez, J.; Chaney, C.; Vanichapol, T.; Streeter, E.; Almuallim, Z.; Oda, A.; Chiba, T.; et al. A Genetically Inducible Endothelial Niche Enables Vascularization of Human Kidney Organoids with Multilineage Maturation and Emergence of Renin Expressing Cells. Kidney Int 2024, 106, 1086–1100. [CrossRef]
- Raykhel, I.; Nishikawa, M.; Sakai, Y.; Vainio, S.J.; Skovorodkin, I. Vascularization of Kidney Organoids: Different Strategies and Perspectives. Frontiers in Urology 2024, 4, 1355042. [CrossRef]
- Wang, R.; Sui, Y.; Liu, Q.; Xiong, Y.; Li, S.; Guo, W.; Xu, Y.; Zhang, S. Recent Advances in Extracellular Matrix Manipulation for Kidney Organoid Research. Front Pharmacol 2024, 15, 1472361. [CrossRef]
- Corridon, P.R.; Ko, I.K.; Yoo, J.J.; Atala, A. Bioartificial Kidneys. Curr Stem Cell Rep 2017, 3, 68-76. [CrossRef]
- Nalesso, F.; Garzotto, F.; Cattarin, L.; Bettin, E.; Cacciapuoti, M.; Silvestre, C.; Stefanelli, L. F.; Furian, L.; Calò, L. A. The Future for End-Stage Kidney Disease Treatment: Implantable Bioartificial Kidney Challenge. Applied Sciences 2024; 14, 491. [CrossRef]
- van Gelder, M.K.; Mihaila, S.M.; Jansen, J.; Wester, M.; Verhaar, M.C.; Joles, J.A.; Stamatialis, D.; Masereeuw, R.; Gerritsen, K.G.F. From Portable Dialysis to a Bioengineered Kidney. Expert Rev Med Devices 2018, 15, 323–336. [CrossRef]
- Tumlin, J.; Wali, R.; Williams, W.; Murray, P.; Tolwani, A.J.; Vinnikova, A.K.; Szerlip, H.M.; Ye, J.; Paganini, E.P.; Dworkin, L.; et al. Efficacy and Safety of Renal Tubule Cell Therapy for Acute Renal Failure. J Am Soc Nephrol 2008, 19, 1034. [CrossRef]
- Song, J.H.; Humes, H.D. The Bioartificial Kidney in the Treatment of Acute Kidney Injury. Curr Drug Targets 2009, 10, 1227. [CrossRef]
- Kim, E.J.; Chen, C.; Gologorsky, R.; Santandreu, A.; Torres, A.; Wright, N.; Goodin, M.S.; Moyer, J.; Chui, B.W.; Blaha, C.; et al. Feasibility of an Implantable Bioreactor for Renal Cell Therapy Using Silicon Nanopore Membranes. Nat Commun 2023, 14, 4890. [CrossRef]
- Groth, T.; Stegmayr, B. G.; Ash, S. R.; Kuchinka, J.; Wieringa, F. P.; Fissell, W. H.; Roy, S. Wearable and implantable artificial kidney devices for end-stage kidney disease treatment: Current status and review. Artif. Organs. 2023, 47, 649–666. [CrossRef]
- Karageorgos, F.F.; Neiros, S.; Karakasi, K.-E.; Vasileiadou, S.; Katsanos, G.; Antoniadis, N.; Tsoulfas, G. Artificial Kidney: Challenges and Opportunities. World J Transplant 2024, 14, 89025. [CrossRef]
- Jena, R.; Aggarwal, A.; Choudhary, G.R.; Bajpai, N.K.; Jena, R.; Aggarwal, A.; Choudhary, G.R.; Bajpai, N.K. Current Status and Future of Artificial Kidney in Humans. Indian J Nephrol 2022, 32, 531–538. [CrossRef]
- Rayat Pisheh, H.; Haghdel, M.; Jahangir, M.; Hoseinian, M.S.; Rostami Yasuj, S.; Sarhadi Roodbari, A. Effective and New Technologies in Kidney Tissue Engineering. Front Bioeng Biotechnol 2024, 12, 1476510. [CrossRef]
- Fransen, M.F.J.; Addario, G.; Bouten, C.V.C.; Halary, F.; Moroni, L.; Mota, C. Bioprinting of Kidney in Vitro Models: Cells, Biomaterials, and Manufacturing Techniques. Essays Biochem 2021, 65, 587-602. [CrossRef]
- Mirshafiei, M.; Rashedi, H.; Yazdian, F.; Rahdar, A.; Baino, F. Advancements in Tissue and Organ 3D Bioprinting: Current Techniques, Applications, and Future Perspectives. Mater Des 2024, 240, 112853. [CrossRef]
- Jeong, H.J.; Nam, H.; Jang, J.; Lee, S.J. 3D Bioprinting Strategies for the Regeneration of Functional Tubular Tissues and Organs. Bioengineering 2020, 7, 32. [CrossRef]
- Homan, K.A.; Kolesky, D.B.; Skylar-Scott, M.A.; Herrmann, J.; Obuobi, H.; Moisan, A.; Lewis, J.A. Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips. Sci Rep 2016, 6, 34845. [CrossRef]
- Huang, W.; Chen, Y.Y.; He, F.F.; Zhang, C. Revolutionizing Nephrology Research: Expanding Horizons with Kidney-on-a-Chip and Beyond. Front Bioeng Biotechnol 2024, 12, 1373386. [CrossRef]
- Mou, X.; Shah, J.; Roye, Y.; Du, C.; Musah, S. An Ultrathin Membrane Mediates Tissue-Specific Morphogenesis and Barrier Function in a Human Kidney Chip. Sci Adv 2024, 10, eadn2689. [CrossRef]
- Singh, N.K.; Kim, J.Y.; Lee, J.Y.; Lee, H.; Gao, G.; Jang, J.; Kim, Y.K.; Cho, D.W. Coaxial Cell Printing of a Human Glomerular Model: An in Vitro Glomerular Filtration Barrier and Its Pathophysiology. Biofabrication 2023, 15, 10.1088/1758-5090/acad2c. [CrossRef]
- Carreno-Caleano, G.; Ali, M.; Yoo, J.J.; Lee, S.J.; Atala, A. 3D Bioprinted Renal Constructs Using Kidney-Specific ECM Bioink System on Kidney Regeneration. Adv Healthc Mater 2025, 2502576. [CrossRef]
- Yuan, Z.; Bai, X.; Li, S.; Fu, Y.; Wan, Z.; Guo, X.; Zhai, M.; Yi, J.; Liu, Y.; Zhou, Y.; et al. Multimaterial and Multidimensional Bioprinting in Regenerative Medicine: Advances, Limitations, and Future Directions. Adv Healthc Mater 2025, 2500475. [CrossRef]
- Agarwal, T.; Onesto, V.; Banerjee, D.; Guo, S.; Polini, A.; Vogt, C. D.; Viswanath, A.; Esworthy, T.; Cui, H.; O’Donnell, A.; Vajanthri, K. Y.; Moroni, L.; et al. 3D bioprinting in tissue engineering: current state-of-the-art and challenges towards system standardization and clinical translation. Biofabrication 2025, 10.1088/1758-5090/ade47a. Advance online publication. [CrossRef]
- 4Bonventre, J. V.; Hurst, F.P.; West, M.; Wu, I.; Roy-Chaudhury, P.; Sheldon, M. A Technology Roadmap for Innovative Approaches to Kidney Replacement Therapies a Catalyst for Change. Clin J Am Soc Nephrol. 2019, 14, 1539-1547. [CrossRef]
- de Haan, M.J.A.; Witjas, F.M.R.; Engelse, M.A.; Rabelink, T.J. Have We Hit a Wall with Whole Kidney Decellularization and Recellularization: A Review. Curr Opin Biomed Eng 2021, 20, 100335. [CrossRef]
- Destefani, A.C.; Sirtoli, G.M.; Nogueira, B.V. Advances in the Knowledge about Kidney Decellularization and Repopulation. Front Bioeng Biotechnol 2017, 5, 255314. [CrossRef]
- Khosropanah, M.H.; Torabinavid, P.; Azimzadeh, A.; Tanourlouee, S.B.; Kajbafzadeh, A.M. Efficient Decellularization of Human Fetal Kidneys through Optimized SDS Exposure. Sci Rep 2024, 14, 21545. [CrossRef]
- Sullivan, D.C.; Mirmalek-Sani, S.H.; Deegan, D.B.; Baptista, P.M.; Aboushwareb, T.; Atala, A.; Yoo, J.J. Decellularization Methods of Porcine Kidneys for Whole Organ Engineering Using a High-Throughput System. Biomaterials 2012, 33, 7756–7764. [CrossRef]
- Figliuzzi, M.; Bonandrini, B.; Remuzzi, A. Decellularized Kidney Matrix as Functional Material for Whole Organ Tissue Engineering. J Appl Biomater Funct Mater 2017, 15, e326–e333. [CrossRef]
- Torabinavid, P.; Khosropanah, M.H.; Azimzadeh, A.; Kajbafzadeh, A.M. Current Strategies on Kidney Regeneration Using Tissue Engineering Approaches: A Systematic Review. BMC Nephrol 2025, 26, 66. [CrossRef]
- Luque-Badillo, A.C.; Monjaras-Avila, C.U.; Adomat, H.; So, A.; Chavez-Muñoz, C. Evaluating Different Methods for Kidney Recellularization. Sci Rep 2024, 14, 23520. [CrossRef]
- Choi, M.; Al Fahad, M.A.; Shanto, P.C.; Park, S. su; Lee, B.T. Surface Modification of Decellularized Kidney Scaffold with Chemokine and AKI-CKD Cytokine Juice to Increase the Recellularization Efficiency of Bio-Engineered Kidney. Biomaterials 2025, 316, 123007. [CrossRef]
- Theodorou, C.; Leatherby, R.; Dhanda, R. Function of the Nephron and the Formation of Urine. Anaesthesia & Intensive Care Medicine 2021, 22, 434–438. [CrossRef]
- Koulouridis, E.; Koulouridis, I. The loop of Henle as the milestone of mammalian kindey concentrating ability: a historical review. Acta Med Hist Adriat. 2014, 12, 413-428.
- Olde Hanhof, C.J.A.; Yousef Yengej, F.A.; Rookmaaker, M.B.; Verhaar, M.C.; Van Der Wijst, J.; Hoenderop, J.G. Modeling Distal Convoluted Tubule (Patho)Physiology: An Overview of Past Developments and an Outlook Toward the Future. Tissue Eng Part C Methods 2021, 27, 200–212. [CrossRef]
- Lam, A.Q.; Bonventre, J. V. Regenerating the Nephron with Human Pluripotent Stem Cells. Curr Opin Organ Transplant 2015, 20, 187–192. [CrossRef]
- Haraldsson, B.; Nyström, J.; Deen, W.M. Properties of the Glomerular Barrier and Mechanisms of Proteinuria. Physiol Rev 2008, 88, 451–487. [CrossRef]
- van den Berg, C.W.; Dumas, S.J.; Little, M.H.; Rabelink, T.J. Challenges in Maturation and Integration of Kidney Organoids for Stem Cell–Based Renal Replacement Therapy. Kidney Int 2025, 107, 262–270. [CrossRef]
- Mazloomnejad, R.; Babajani, A.; Kasravi, M.; Ahmadi, A.; Shariatzadeh, S.; Bahrami, S.; Niknejad, H. Angiogenesis and Re-Endothelialization in Decellularized Scaffolds: Recent Advances and Current Challenges in Tissue Engineering. Front Bioeng Biotechnol 2023, 11, 1103727. [CrossRef]
- Wang, K.; Kestenbaum, B. Proximal Tubular Secretory Clearance: A Neglected Partner of Kidney Function. Clin J Am Soc Nephrol 2018, 13, 1291. [CrossRef]
- Wagner, C.A. Beyond SGLT2: Proximal Tubule Transporters as Potential Drug Targets for Chronic Kidney Disease. Nephrol Dial Transplant 2025, 40, i18–i28. [CrossRef]
- Ross, E.J.; Gordon, E.R.; Sothers, H.; Darji, R.; Baron, O.; Haithcock, D.; Prabhakarpandian, B.; Pant, K.; Myers, R.M.; Cooper, S.J.; et al. Three Dimensional Modeling of Biologically Relevant Fluid Shear Stress in Human Renal Tubule Cells Mimics in Vivo Transcriptional Profiles. Sci Rep 2021, 11, 14053. [CrossRef]
- Kim, H.; Lee, J.B.; Kim, K.; Sung, G.Y. Effect of Shear Stress on the Proximal Tubule-on-a-Chip for Multi-Organ Microphysiological System. Journal of Industrial and Engineering Chemistry 2022, 115, 279-286. [CrossRef]
- Karp, S.; Pollak, M.R.; Subramanian, B. Disease Modeling with Kidney Organoids. Micromachines 2022, 13, 1384. [CrossRef]
- Li, S.R.; Gulieva, R.E.; Helms, L.; Cruz, N.M.; Vincent, T.; Fu, H.; Himmelfarb, J.; Freedman, B.S. Glucose Absorption Drives Cystogenesis in a Human Organoid-on-Chip Model of Polycystic Kidney Disease. Nat Commun 2022, 13, 7918. [CrossRef]
- Yu, Y.; Alkhawaji, A.; Ding, Y.; Mei, J. Decellularized Scaffolds in Regenerative Medicine. Oncotarget 2016, 7, 58671. [CrossRef]
- Marulli, M.; Edwards, A.; Milišić, V.; Vauchelet, N. On the Role of the Epithelium in a Model of Sodium Exchange in Renal Tubules. Math Biosci 2020, 321. [CrossRef]
- Pahuja, A.; Goux Corredera, I.; Moya-Rull, D.; Garreta, E.; Montserrat, N. Engineering Physiological Environments to Advance Kidney Organoid Models from Human Pluripotent Stem Cells. Curr Opin Cell Biol 2024, 86, 102306. [CrossRef]
- Liu, M.; Cardilla, A.; Ngeow, J.; Gong, X.; Xia, Y. Studying Kidney Diseases Using Organoid Models. Front Cell Dev Biol 2022, 10, 845401. [CrossRef]
- McCormick, J.A.; Ellison, D.H. Distal Convoluted Tubule. Compr Physiol 2015, 5, 2147-2163. [CrossRef]
- Dilmen, E.; Olde Hanhof, C.J.A.; Yousef Yengej, F.A.; Ammerlaan, C.M.E.; Rookmaaker, M.B.; Orhon, I.; Jansen, J.; Verhaar, M.C.; Hoenderop, J.G. A Semi-Permeable Insert Culture Model for the Distal Part of the Nephron with Human and Mouse Tubuloid Epithelial Cells. Exp Cell Res 2025, 444. [CrossRef]
- Yousef Yengej, F.A.; Pou Casellas, C.; Ammerlaan, C.M.E.; Olde Hanhof, C.J.A.; Dilmen, E.; Beumer, J.; Begthel, H.; Meeder, E.M.G.; Hoenderop, J.G.; Rookmaaker, M.B.; et al. Tubuloid Differentiation to Model the Human Distal Nephron and Collecting Duct in Health and Disease. Cell Rep 2024, 43, 113614. [CrossRef]
- Zeng, Z.; Huang, B.; Parvez, R.K.; Li, Y.; Chen, J.; Vonk, A.C.; Thornton, M.E.; Patel, T.; Rutledge, E.A.; Kim, A.D.; et al. Generation of Patterned Kidney Organoids That Recapitulate the Adult Kidney Collecting Duct System from Expandable Ureteric Bud Progenitors. Nat Commun 2021, 3641. [CrossRef]
- Howden, S.E.; Wilson, S.B.; Groenewegen, E.; Starks, L.; Forbes, T.A.; Tan, K.S.; Vanslambrouck, J.M.; Holloway, E.M.; Chen, Y.H.; Jain, S.; et al. Plasticity of Distal Nephron Epithelia from Human Kidney Organoids Enables the Induction of Ureteric Tip and Stalk. Cell Stem Cell 2021, 28, 671-684.e6. [CrossRef]
- Wang, W.; Hu, G.; Li, G.; Li, P.-L.; Li, N. Collecting Duct-Specific Deletion of Acid Ceramidase Promoted Sodium Retention and Salt-Sensitive Hypertension. Physiology 2025, 40, S1. [CrossRef]
- Higashihara, E.; Harada, T.; Fukuhara, H. Juxtaglomerular Apparatus-Mediated Homeostatic Mechanisms: Therapeutic Implication for Chronic Kidney Disease. Expert Opin Pharmacother 2024, 25, 819–832. 819-832. [CrossRef]
- Yamaguchi, H.; Gomez, R.A.; Sequeira-Lopez, M.L.S. Renin Cells, From Vascular Development to Blood Pressure Sensing. Hypertension 2023, 80, 1580-1589. [CrossRef]
- Shankar, A.S.; Du, Z.; Mora, H.T.; van den Bosch, T.P.P.; Korevaar, S.S.; Van den Berg-Garrelds, I.M.; Bindels, E.; Lopez-Iglesias, C.; Clahsen-van Groningen, M.C.; Gribnau, J.; et al. Human Kidney Organoids Produce Functional Renin. Kidney Int 2021, 99, 134–147. [CrossRef]
- Freedman, B.S. Physiology Assays in Human Kidney Organoids. Am J Physiol Renal Physiol 2022, 322, F625-F638. [CrossRef]
- Yanofsky, S.M.; Dugas, C.M.; Katsurada, A.; Liu, J.; Saifudeen, Z.; El-Dahr, S.S.; Satou, R. Angiotensin II Biphasically Regulates Cell Differentiation in Human IPSC-Derived Kidney Organoids. Am J Physiol Renal Physiol 2021, 321, F559-F571. [CrossRef]
- Kida, Y. Peritubular Capillary Rarefaction: An Underappreciated Regulator of CKD Progression Int J Mol Sci 2020, 21, 8255. [CrossRef]
- Choi, Y.J.; Chakraborty, S.; Nguyen, V.; Nguyen, C.; Kim, B.K.; Shim, S.I.; Suki, W.N.; Truong, L.D. Peritubular Capillary Loss Is Associated with Chronic Tubulointerstitial Injury in Human Kidney: Altered Expression of Vascular Endothelial Growth Factor. Hum Pathol 2000, 31, 1491–1497. [CrossRef]
- Meng, X.; Xing, Y.; Li, J.; Deng, C.; Li, Y.; Ren, X.; Zhang, D. Rebuilding the Vascular Network: In Vivo and in Vitro Approaches. Front Cell Dev Biol 2021, 9, 639299. [CrossRef]
- Shin, Y.J.; Safina, D.; Zheng, Y.; Levenberg, S. Microvascularization in 3D Human Engineered Tissue and Organoids. Annu Rev Biomed Eng 2025, 27, 473–498. [CrossRef]
- Liu, Q.; Ying, G.; Hu, C.; Du, L.; Zhang, H.; Wang, Z.; Yue, H.; Yetisen, A.K.; Wang, G.; Shen, Y.; et al. Engineering in Vitro Vascular Microsystems. Microsystems & Nanoengineering 2025 11:1 2025, 11, 100. [CrossRef]
- Lebedenko, C.G.; Banerjee, I.A. Enhancing Kidney Vasculature in Tissue Engineering—Current Trends and Approaches: A Review. Biomimetics 2021, 6, 40. [CrossRef]
- Wieringa, F.P.; Suran, S.; Søndergaard, H.; Ash, S.; Cummins, C.; Chaudhuri, A.R.; Irmak, T.; Gerritsen, K.; Vollenbroek, J. The Future of Technology-Based Kidney Replacement Therapies: An Update on Portable, Wearable, and Implantable Artificial Kidneys. Am J Kidney Dis 2025, 85, 787–796. [CrossRef]
- Araoka, T.; Toyohara, K.; Ryosaka, M.; Inui, C.; Matsuura, M.; Ma, C.; Watahiki, J.; Li, Z.; Iwasaki, M.; Watanabe, A.; et al. Human IPSC–Derived Nephron Progenitor Cells Treat Acute Kidney Injury and Chronic Kidney Disease in Mouse Models. Sci Transl Med 2025, 17, eadt5553. [CrossRef]
- Tsokas, K.; McFarland, R.; Burke, C.; Lynch, J.L.; Bollenbach, T.; Callaway II, D.A.; Siegel, J. Reducing Risks and Delays in the Translation of Cell and Gene Therapy Innovations into Regulated Products. NAM Perspectives 2019, 2019, 10.31478/201909d. [CrossRef]
- Lowdell, M.W. Considerations for Manufacturing of Cell and Gene Medicines for Clinical Development. Cytotherapy 2025, 27, 874–883. [CrossRef]
- Iglesias-Lopez, C.; Agustí, A.; Vallano, A.; Obach, M. Current Landscape of Clinical Development and Approval of Advanced Therapies. Mol Ther Methods Clin Dev 2021, 23, 606. [CrossRef]
- Sekar, M.P.; Budharaju, H.; Zennifer, A.; Sethuraman, S.; Vermeulen, N.; Sundaramurthi, D.; Kalaskar, D.M. Current Standards and Ethical Landscape of Engineered Tissues—3D Bioprinting Perspective. J Tissue Eng 2021, 12, 20417314211027677. [CrossRef]
- Zheng, Y.L. Some Ethical Concerns About Human Induced Pluripotent Stem Cells. Sci Eng Ethics 2016, 22, 1277–1284. [CrossRef]
- Arifa, N. Induced Pluripotent Stem Cells (IPSCs): The Ethical Alternative? Stem Cell Res Reg Med 2024, 7, 251-252.
- Cintron Pregosin, N.; Bronstein, R.; Mallipattu, S.K. Recent Advances in Kidney Bioengineering. Front Pediatr 2021, 9, 743301. [CrossRef]
| Nephron function | Physiological role | Cellular/molecular requirements | Replacement technologies | Current status |
|---|---|---|---|---|
| Glomerulus (filtration) | Initiates urine formation via selective, high-pressure filtration of blood; retains proteins and cells while allowing passage of water and small solutes | Podocytes, glomerular endothelial cells, specialized GBM (collagen IV, laminin), slit diaphragm proteins (nephrin, podocin) | Decellularized glomerular scaffolds; bioprinted glomerular units; PSC-derived organoid glomeruli | Proof-of-concept filtration achieved in vitro and in small animal models; physiological selectivity and sustained filtration rates remain suboptimal. Integration with vascular networks is a key challenge. |
| Proximal tubule (reabsorption/secretion) | Reabsorbs ~65% of filtered water, glucose, amino acids, bicarbonate, and ions; secretes organic solutes and drugs | Proximal tubular epithelial cells with brush border, SGLT2, NHE3, rich mitochondrial content, tight junctions | 3D bioprinted tubules; organoid-derived proximal segments; recellularized scaffolds; microfluidic kidney-on-chip platforms | Functional reabsorption and secretion demonstrated in vitro; models support drug screening and nephrotoxicity studies. Long-term maturation, polarity, and integration with downstream segments are active areas of research. |
| Loop of Henle (countercurrent concentration) | Establishes medullary osmotic gradient via countercurrent multiplication, enabling urine concentration | Thin and thick limb epithelial cells, aquaporins (AQP1, AQP2), Na-K-2Cl cotransporter (NKCC2), medullary interstitium | Microengineered loop modules; segment-specific differentiation in organoids | Early-stage prototypes: partial recapitulation of countercurrent function. Full osmotic gradient generation and integration with adjacent segments remain to be achieved. |
| Distal tubule (electrolyte fine-tuning) | Regulates sodium, potassium, calcium, and acid-base balance under hormonal control (aldosterone, PTH) | Distal tubular epithelial cells, ENaC, NCC, calcium channels, hormone receptors | Segment-specific cell sheets; responsive bioartificial modules; engineered distal tubule constructs | Segment identity and hormonal responsiveness were demonstrated in vitro. Integration with upstream and downstream modules and dynamic regulation are under development. |
| Collecting duct (water reabsorption/excretion) | Final site for water reabsorption (ADH-regulated), acid-base homeostasis, and urine excretion | Principal and intercalated cells, aquaporins (AQP2), ADH and aldosterone receptors, tight junctions | Engineered collecting duct arrays; organoid-derived collecting duct segments; responsive bioartificial modules | Functional water reabsorption and hormone response are shown in vitro. Full integration with nephron modules and urine drainage systems remains a challenge. |
| Juxtaglomerular apparatus (endocrine/autoregulation) | Senses tubular flow and sodium; regulates renin secretion and blood pressure (RAAS system); autoregulates GFR | Juxtaglomerular cells (renin), macula densa, afferent arteriole, paracrine signaling molecules | Organoid-based models; microfluidic feedback systems | Experimental models recapitulate some aspects of renin secretion and feedback. Full endocrine and autoregulatory function has not yet been achieved. |
| Peritubular capillaries (microvascular support) | Supplies oxygen/nutrients, removes reabsorbed solutes, supports tubule metabolism and function | Endothelial cells, pericytes, angiogenic factors, basement membrane | Vascularized scaffolds; endothelialized microfluidic chips; co-culture systems | Microvascular networks established in vitro; perfusion and stability over time are improving. Full integration with nephron modules and host vasculature remains a barrier. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




