1. Introduction
Chronic kidney disease (CKD) affects more than 850 million people globally, with the number of patients needing kidney replacement therapy (KRT) expected to reach 5.4 million by 2030 [
1,
2]. These burdens fall disproportionately on low- and middle-income countries, where access to dialysis and transplantation is often limited [
3]. Existing options, dialysis and kidney transplantation, are life-sustaining but not curative. Dialysis only partially replaces kidney function and is associated with high morbidity, poor quality of life, and reduced survival [
4,
5]. Kidney transplantation offers superior outcomes but is constrained by a severe shortage of donor organs, risk of graft failure, and the need for lifelong immunosuppression [
6,
7].
In response to these limitations, a new wave of technologies has emerged aimed at replicating key nephron-level functions using biological or bioengineered systems [
8,
9]. Rather than attempting to recreate an entire kidney, many of these approaches focus on modular restoration of specific physiological functions, leveraging advances in stem cell biology, biofabrication, and device engineering.
Several efforts have already laid the foundation for this conceptual shift. In 2018, the Kidney Health Initiative (KHI) released a landmark roadmap calling for transformative innovation in KRT [
10]. That same year, a Perspective by Salani et al., published in the American Journal of Kidney Diseases, highlighted advances in wearable and implantable artificial kidneys, efforts that reflect a parallel push to improve dialysis through engineering and miniaturization [
8]. More recently, Bonandrini et al. provided a comprehensive review of cell-based and bioartificial kidney technologies, marking a shift toward regenerative strategies aimed at restoring nephron-level function [
9].
Building on these efforts, this review introduces a function-based, modular framework for assessing emerging nephron replacement strategies. In contrast to earlier reviews that primarily examined individual technological platforms [
11,
12,
13], we adopt a physiological lens. We focus on four main approaches: kidney organoids, implantable bioartificial kidneys (iBK), three-dimensional (3D) bioprinted renal tissues, and decellularized scaffolds, and evaluate how effectively each replicates key nephron functions. We also consider the translational maturity of each approach and explore paths toward integrated, clinically viable therapies. Ethical and regulatory considerations are briefly addressed to support their responsible development and application.
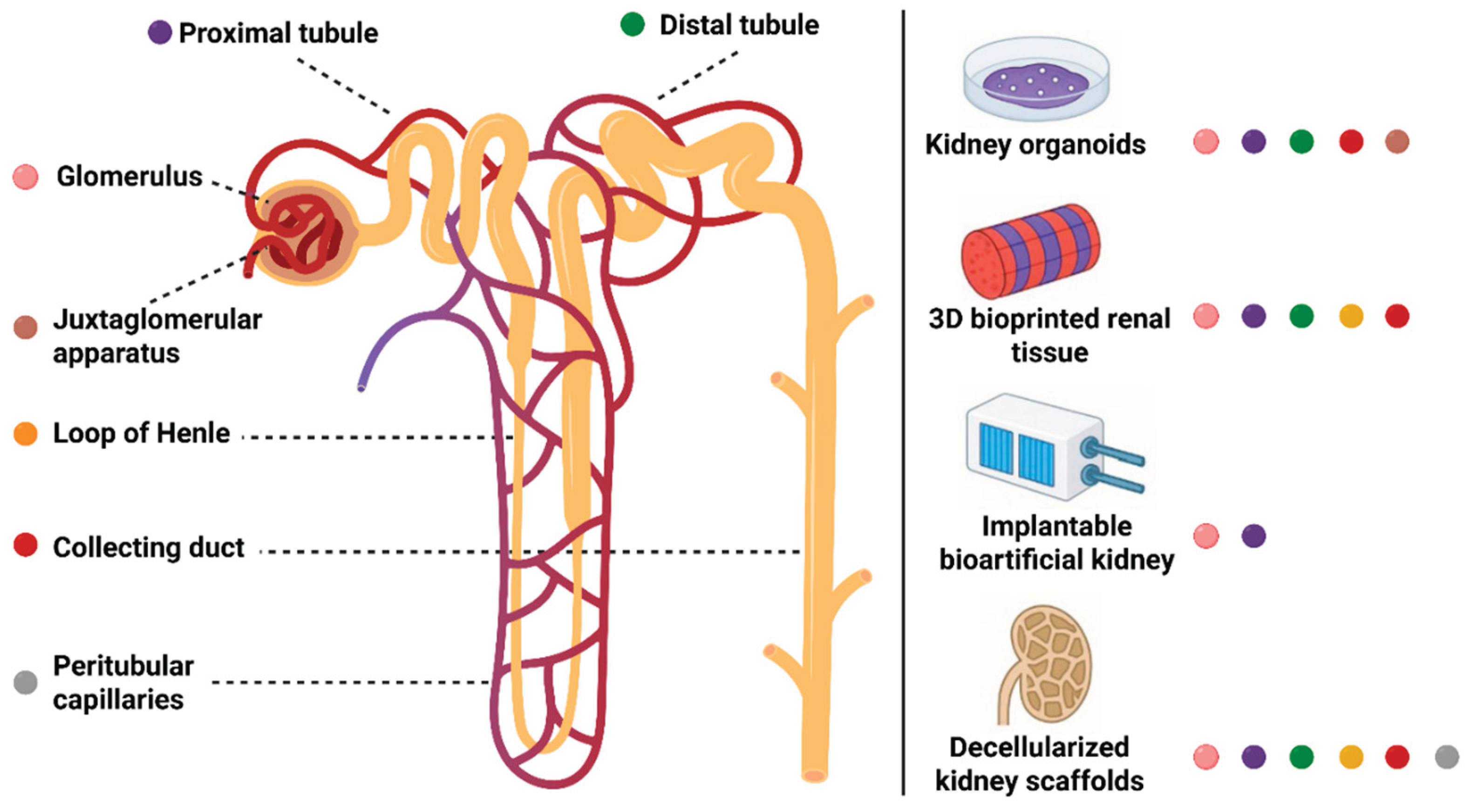
Although not yet formalized in clinical nomenclature, we use the term “nephron replacement strategies” to describe biologically or bioengineered interventions focused on restoring kidney function beyond the limits of conventional KRT. To orient the reader within this framework,
Figure 1 maps each nephron segment to corresponding bioengineering strategies, offering a visual guide to the review’s organization.
2. Overview of the Evolving Landscape of Nephron Replacement Strategies
Currently, the nephron replacement landscape includes four major platforms: kidney organoids, iBK devices, 3D-bioprinted renal tissues, and decellularized kidney scaffolds. Although these technologies differ in their mechanisms, maturity, and functional focus, they collectively represent the most promising avenues for next-generation, modular kidney support.
2.1. Kidney Organoids
Kidney organoids are 3D structures derived from pluripotent stem cells (PSCs) that mimic aspects of early nephron development and architecture [
14,
15]. Over the past decade, they have become central to regenerative nephrology, serving as platforms for disease modeling, drug testing, and potentially future transplantation [
14,
15].
Initial progress began in 2010 with self-organizing aggregates from dissociated mouse embryonic kidney cells [
16], though these lacked PSC origin and structural organization. A major advance came in 2013 with the generation of ureteric bud organoids from human PSCs, cocultured with mouse embryonic cells to form branching structures [
17]. In 2015, Takasato et al. published a reproducible protocol for differentiating human PSCs into kidney organoids containing organized nephrons, collecting ducts, interstitial cells, and vascular progenitors [
18]. This marked a turning point by showing that complex renal structures could be generated entirely from human stem cells without coculture, establishing organoids as a practical tool for kidney modeling and sparking widespread adoption across the field.
Modern kidney organoids typically contain glomerular-like podocytes, proximal and distal tubule segments, loop of Henle–like regions, and interstitial cell populations [
19,
20]. They are morphologically similar to first- and second-trimester fetal kidneys but remain developmentally immature and lack complete functional integration with vasculature and other organ systems [
18,
20]. This immaturity is a recognized limitation, as organoids do not fully recapitulate adult kidney structure or function and are not yet capable of supporting full physiological renal activity [
18,
19,
20]. Key limitations include:
Absence of a vascular network, which restricts nutrient delivery and size,
Lack of a urine drainage system, precluding physiological excretion,
Cellular heterogeneity and off-target populations, affecting reproducibility[
19,
21,
22].
Recent advances partially address these issues. Microfluidic “organ-on-chip” platforms improve endothelial integration and podocyte maturation [
23,
24]. Genetic co-differentiation with endothelial progenitors enables kidney-specific vascularization and renin production [
25]. Co-culture and in vivo transplantation into immunodeficient mice further enhance integration with host vasculature and structural maturation [
26]. Lastly, refined protocols and ECM manipulation have improved tissue organization and nephron segmentation [
26,
27]. Collectively, these innovations are pushing organoids closer to physiological and translational relevance, though they remain preclinical tools.
2.2. Implantable Bioartificial Kidney (iBK) Devices
iBK devices represent a cutting-edge class of hybrid technologies designed to replicate essential nephron-level functions through a combination of engineered membranes and living renal epithelial cells [
28,
29]. Unlike conventional dialysis, which substitutes for excretory function, these systems aim to deliver continuous, physiologic support without external power sources, vascular access maintenance, or immunosuppression [
8,
28,
30]. Among the various nephron replacement strategies under development, the iBK platform is the most clinically advanced.
The concept of a bioartificial kidney originated in the late 1990s with extracorporeal systems like the Renal Assist Device (RAD), which coupled hollow-fiber hemofiltration with porcine renal tubular cells to augment dialysis [
28,
30]. Although these early devices showed metabolic and immunomodulatory benefits in acute kidney injury [
31,
32], they were designed for short-term use and lacked autonomous, long-term nephron-level function. As such, they are not classified within current nephron replacement strategies. Still, they laid crucial groundwork for next-generation implantable systems [
30].
The most prominent and mature example in this domain is the iBK developed by the Kidney Project, a collaborative initiative between researchers at the University of California, San Francisco, and Vanderbilt University. The device is composed of two key modules designed to function together in replicating core nephron tasks:
A hemofilter, constructed from silicon nanopore membranes, emulates the glomerular filtration barrier by permitting selective ultrafiltration of plasma under native arterial pressure. These membranes are fabricated through microelectromechanical systems engineering, enabling high precision and resistance to fouling, without the need for anticoagulation,
A bioreactor, seeded with human renal proximal tubule epithelial cells, performs essential solute reabsorption and metabolic functions. This compartment mimics the tubular segment of the nephron, processing the ultrafiltrate produced by the hemofilter [
8,
28,
29].
Together, these modules are implanted in the peritoneal cavity, connected to systemic circulation and draining into the bladder, eliminating the need for external dialysis infrastructure [
8,
30]. The system operates entirely passively, powered by the patient’s own hemodynamics, enabling a continuous and physiologic mode of therapy.
Significant experimental milestones have been achieved in recent years. In 2023, the iBK’s bioreactor module was successfully tested in a large-animal (porcine) model, demonstrating up to 7 days of stable performance without thrombosis, device occlusion, or mechanical failure [
33]. This preclinical trial validated the hemofilter’s biocompatibility, durability, and functional integration under physiologic conditions.
Despite its promise, the iBK faces several key engineering and biological challenges:
Sustaining epithelial cell viability and function under physiological shear stress,
Preventing fibrosis and immune rejection in the absence of systemic immunosuppression,
Ensuring long-term durability of membrane materials and preventing device fouling or occlusion,
Achieving tight coupling between filtration and reabsorption to fully mimic nephron-level homeostasis [
34,
35,
36].
Nevertheless, the iBK is widely recognized as the most mature nephron replacement strategy progressing toward clinical use, supported by KidneyX, NIH, and FDA engagement.
2.3. 3D Bioprinted Renal Tissues
3D-bioprinting quickly emerges as one of the most exciting and promising approaches in the effort to create functional kidney tissue [
37,
38]. Unlike organoid or scaffold-based methods, bioprinting enables bottom-up construction of tissue with high control over architecture and composition [
37,
39]. This process uses so-called “bioinks», carefully formulated blends of living cells and supportive hydrogels, which are deposited layer by layer to mimic the kidney’s complex architecture [
37]. In the context of kidney bioengineering, 3D bioprinting seeks to replicate essential nephron structures, including glomeruli, tubules, and surrounding interstitial components, in configurations that support physiological function and vascular integration [
38].
Initial proof-of-concept studies in the 2010s focused on tubular structures using inkjet or extrusion-based systems loaded with kidney-derived cells [
40,
41]. Though simple, they demonstrated the feasibility of printing nephron geometry with living cells.
By 2015, researchers created perfusable constructs containing proximal tubule cells capable of transporter activity and nephrotoxin response, marking a shift toward functional models [
40,
42]. Recent advances have focused on multi-compartmental bioprinting, combining epithelial and endothelial cells to model the filtration–reabsorption axis. Some constructs now incorporate glomerular-like elements, including podocyte–endothelial interfaces, though physiological filtration remains difficult to replicate [
43,
44,
45].
Innovations in multi-material printing, spatial patterning, and bioink customization have enabled the inclusion of stromal support cells (e.g., fibroblasts, mesangial cells) and ECM-derived signals that better simulate the renal microenvironment [
43,
46]. Despite these advances, several critical barriers remain:
Achieving hierarchical vascularization;
Maintaining long-term cell viability under physiological pressure;
Scaling constructs to clinically relevant sizes without compromising function;
Integrating bioprinted tissues with host vasculature and immune environments [
37,
45,
47].
Still, 3D bioprinting offers a modular, customizable platform. In the near term, bioprinted tissue may be used for ex vivo testing, hybrid constructs, or vascularized modules integrated with other systems [
39,
47,
48].
2.4. Decellularized Kidney Scaffolds
Decellularized kidney scaffolds are created by removing all cellular material from donor kidneys, using chemical, enzymatic, or physical methods, while preserving the organ’s 3D ECM structure and vascular architecture [
49,
50]. The ECM serves not only as a structural framework but also as a bioactive template that supports cell adhesion, proliferation, migration, and differentiation, owing to its retention of essential proteins, as well as embedded signaling molecules and growth factors [
49,
51].
The concept emerged in the late 2000s, following successful decellularization of rat and porcine kidneys, which retained their native macro- and microanatomy. These scaffolds were shown to support reseeding with renal epithelial and endothelial cells, enabling partial reconstruction of nephron compartments in vitro [
52,
53]. Recent advances in perfusion decellularization and tissue-specific recellularization have improved scaffold biocompatibility and functionality [
50,
51].
Modern methods better preserve ECM integrity and minimize immunogenic residue. Refined perfusion decellularization methods now better maintain ECM integrity while minimizing immunogenic residue [
54]. Recellularization efforts increasingly use organ-specific cell types such as podocytes, proximal tubule epithelial cells, and glomerular endothelial cells, often delivered through vascular perfusion routes to promote uniform distribution and functional zonation [
55]. In parallel, induced pluripotent stem cell (iPSC)-derived renal progenitors have shown promise in repopulating multiple nephron segments, while endothelial progenitors have been used to reconstruct functional vascular networks [
54,
56]. Perfusion bioreactors, which provide controlled flow, oxygenation, and mechanical cues, are now a critical component in fostering cellular engraftment, maturation, and phenotypic maintenance within the decellularized scaffolds [
54].
Despite promising progress, major challenges persist:
Efficient and selective recellularization of glomerular, tubular, and vascular compartments,
Sourcing sufficient quantities of autologous or immunocompatible cells,
Achieving functional integration and perfusion after implantation,
Controlling immune responses and fibrosis in vivo [
49,
50,
51].
Despite remaining hurdles, decellularized scaffolds are emerging as viable biological chassis for building patient-specific, transplantable kidneys [
51,
54,
55]. If current barriers can be overcome, this strategy may ultimately bridge the gap between regenerative and replacement paradigms in nephrology.
3. The Nephron as a Blueprint for Modular Kidney Replacement
The nephron comprises anatomically and functionally distinct subunits, including the glomerulus, proximal and distal tubules, loop of Henle, collecting duct, juxtaglomerular apparatus, and peritubular capillaries, that together mediate filtration, selective reabsorption, secretion, endocrine regulation, and urine excretion [
57,
58,
59]. Each segment is characterized by region-specific cell populations, membrane transporters, and hemodynamic interfaces.
The nephron’s complex architecture and functional specialization pose both challenges and opportunities for tissue engineering and regenerative nephrology [
60]. Framing nephron functions as discrete, addressable units helps guide the development of focused replacement strategies.
Table 1 provides an overview of this modular landscape, outlining physiological roles, cellular and molecular requirements, technological approaches, and the current status of each functional domain.
3.1. Glomerulus (Filtration)
The glomerulus serves as the nephron’s primary filtration barrier, forming an ultrafiltrate of plasma through a specialized tri-layered structure that excludes cells and high-molecular-weight proteins while allowing passage of water and small solutes [
57,
61]. This barrier consists of fenestrated glomerular endothelial cells, the glomerular basement membrane (GBM) composed of laminin, type IV collagen, and heparan sulfate proteoglycans, and podocyte foot processes bridged by slit diaphragm proteins such as nephrin and podocin. Filtration selectivity is governed by molecular size (~5–10 nm cutoff), electrostatic charge, and shape, while hydraulic permeability is modulated by capillary hydrostatic pressure, GBM thickness, and matrix compliance [
61].
Technologies attempting to reproduce glomerular filtration fall into two major categories: biological constructs and mechanical analogues. Among the latter, the iBK hemofilter remains the most clinically advanced. It uses precision silicon nanopore membranes to achieve selective ultrafiltration under arterial pressure, operating without external pumps [
8,
34,
35]. In vivo studies in large animals have shown short-term patency and protein retention, though challenges remain around long-term biocompatibility and fouling [
33].
On the biological side, organoid-derived glomeruli and bioprinted glomerular units express structural markers and partially replicate podocyte architecture, but lack continuous capillary loops and show immature filtration capacity [
19,
62]. Decellularized glomerular scaffolds preserve native ECM and spatial architecture, and support limited reseeding with podocytes and endothelial cells, though integration with upstream flow and physiologic pressure gradients remains lacking [
49,
50,
63]. Full recapitulation of selective, pressure-responsive filtration has yet to be achieved biologically.
3.2. Proximal Tubule (Reabsorption and Secretion)
The proximal tubule reabsorbs the majority of filtered water, electrolytes, glucose, and amino acids, and serves as the primary site for organic ion secretion [
64]. Its function depends on dense mitochondria, apical brush borders, and key transport proteins such as the sodium-glucose co-transporter 2 (SGLT2), sodium-hydrogen exchanger 3 (NHE3), and organic anion transporters 1 and 3 (OAT1/3), which together coordinate active reabsorption and secretion [
64,
65]. Proper function also requires strong apical-basal polarity and extensive vascular support.
Of all nephron segments, the proximal tubule has been modeled most successfully. Bioprinted proximal tubules and kidney-on-chip platforms reliably demonstrate transporter expression, polarized morphology, and functional reabsorption of glucose and albumin [
41,
42]. Under perfusion, epithelial monolayers form tight junctions and respond to fluid shear stress, simulating aspects of tubular flow [
41,
66]. Some platforms even allow drug screening and nephrotoxicity assays using human-derived cells [
41,
66,
67].
Organoid-derived proximal segments also exhibit brush border markers, SGLT2 expression, and uptake of low-molecular-weight proteins [
23,
68,
69]. However, they often lack proper tubule alignment, vascular proximity, and segmental specificity [
22,
68]. Recellularized proximal tubule scaffolds, using decellularized ECM, support improved polarity and survival, but still require external flow and do not yet show long-term viability [
50,
70]. Functional integration with downstream segments is an active area of development.
3.3. Loop of Henle (Countercurrent Concentration)
The loop of Henle plays a pivotal role in generating the corticomedullary osmotic gradient, which enables water reabsorption in the collecting duct and supports urine concentration [
58]. This is accomplished through a countercurrent multiplier mechanism, involving water efflux via aquaporin-1 (AQP1) in the thin descending limb, passive NaCl reabsorption in the thin ascending limb, and active Na⁺-K⁺-2Cl⁻ cotransport (NKCC2) in the thick ascending limb of the loop. The net result is an axial osmolarity gradient that increases toward the inner medulla, allowing the kidney to concentrate urine up to ~1200 mOsm/kg H₂O [
58]. Replicating this gradient spatially and functionally remains a major bioengineering challenge.
Reconstructing this gradient-generating architecture remains one of the most formidable challenges in nephron bioengineering. Technologies targeting the loop have focused on microfluidic “loop-on-chip” systems that simulate opposing tubular flow and allow precise control of solute gradients. These platforms support unidirectional Na⁺ transport, segment-specific differentiation, and fluid shear exposure, which influence transporter expression and epithelial polarization [
71,
72]. However, while NKCC2 and AQP1 expression have been observed in some organoid-derived constructs, these models lack true anatomical curvature, peritubular capillaries, and interstitial fibroblasts, all essential for the spatial organization of the osmotic gradient [
15,
37,
72]. None of the existing platforms successfully recreates the medullary interstitium, nor do they demonstrate dynamic osmotic concentration, water permeability gradients, or passive solute equilibration across countercurrent limbs [
62,
73]. Moreover, feedback regulation from the juxtaglomerular apparatus via the renin–angiotensin–aldosterone system (RAAS) or sodium sensing is absent, limiting physiological authenticity. Incorporation of gradient-generating scaffolds, ECM-based solute barriers, or co-culture with medullary interstitial cells, such as fibroblasts expressing urea transporters or prostaglandins, may help simulate the native environment and enable integration with downstream water-conserving modules [
72,
73]. Gradient-generating synthetic matrices or co-culture with medullary fibroblasts may be required to simulate the native interstitium.
3.4. Distal Tubule (Electrolyte Regulation)
The distal convoluted tubule plays a central role in fine-tuning sodium, potassium, calcium, and acid–base balance, acting as a key effector segment under hormonal regulation. It expresses the epithelial sodium channel (ENaC), the thiazide-sensitive sodium–chloride cotransporter (NCC), and TRPV5 calcium channels, and responds dynamically to aldosterone and parathyroid hormone to regulate systemic electrolyte and volume status [
59,
74]. The distal convoluted tubule also contributes to local feedback mechanisms, influencing JGA-mediated renin secretion via alterations in sodium load delivery.
In vitro systems, primarily organoid- and monolayer-based, have demonstrated segmental marker expression of ENaC, NCC, and TRPV5, with some models exhibiting partial transcriptional activation in response to hormone stimulation [
56,
71,
72]. However, these responses are often transient and lack sustained ion flux measurements, limiting functional validation. Engineered epithelial sheets offer more stable polarization and transporter localization but fail to mimic the hormone–receptor coupling dynamics and downstream signaling pathways seen in vivo [
77,
78].
Critically, existing models do not recapitulate the interplay between the distal tubule and JGA via macula densa–mediated feedback. The absence of tubular–vascular alignment, sodium-sensing signal transduction, and renin modulation severely limits physiological realism. Integration of the distal convoluted tubule with upstream (loop of Henle) and downstream (collecting duct) segments, along with vascularized macula densa-like interfaces, will be necessary to support functional nephron replacement and endocrine crosstalk. Moreover, mimicking flow-dependent ENaC activation and intracellular calcium dynamics remains an unmet technical goal [
59].
3.5. Collecting Duct (Water and Acid–Base Regulation)
The collecting duct is the nephron’s terminal regulatory segment for water reabsorption, sodium handling, and acid–base homeostasis. It comprises principal cells, which respond to antidiuretic hormone (ADH) by translocating AQP2 channels to the apical membrane, and intercalated cells, which mediate proton secretion (via H⁺-ATPase) and bicarbonate reabsorption to regulate systemic pH [
79]. Aldosterone also acts on this segment to promote sodium reabsorption via ENaC and potassium secretion through renal outer medullary potassium (ROMK) channels.
Organoid and chip-based systems have demonstrated inducible AQP2 expression in response to ADH analogs, with some showing transient aquaporin trafficking and cell-surface insertion [
77,
78]. ENaC and H⁺-ATPase expression has also been observed, suggesting partial replication of segmental identity. However, these models lack essential physiological drivers: the axial osmotic gradient generated by the loop of Henle and proper interstitial compartmentalization to permit passive water movement.
Furthermore, intercalated cells remain underdeveloped in most systems, with limited data on their acid-base transport kinetics or regulation via systemic pH shifts. The collecting duct’s responsiveness to aldosterone and ADH remains poorly sustained over time, and no current model has integrated real-time electrolyte sensing, vasopressin-cAMP signaling cascades, or acid-base buffering feedback loops [
62].
3.6. Juxtaglomerular Apparatus (Endocrine and Autoregulatory Function)
The juxtaglomerular apparatus (JGA) is a specialized multi-cellular structure that couples tubular sodium sensing, arteriolar hemodynamics, and systemic endocrine signaling to regulate glomerular filtration and maintain circulatory homeostasis [
80]. It comprises juxtaglomerular (granular) cells of the afferent arteriole, which synthesize and secrete renin; the macula densa of the distal tubule, which detects luminal NaCl concentrations; and extraglomerular mesangial cells that mediate paracrine communication. Through this architecture, the JGA orchestrates both tubuloglomerular feedback and activation of the RAAS in response to decreased renal perfusion, volume depletion, or sympathetic stimulation [
80,
81].
Functionally, the JGA serves as the kidney’s primary endocrine gateway. Upon renin release, a cascade ensues: angiotensinogen is converted to angiotensin I, then to angiotensin II, which promotes systemic vasoconstriction, sodium retention, aldosterone secretion, and restoration of blood pressure and volume [
80,
81]. The JGA thus provides the interface between local nephron sensing and systemic hemodynamic control, making its recreation essential for any nephron replacement system aiming to move beyond mechanical filtration.
Current in vitro models have made limited progress toward replicating this complexity. Kidney organoids have successfully generated renin-expressing cells from pluripotent stem cells, indicating partial endocrine differentiation [
82]. However, these cells lack functional coupling to flow or sodium-sensing signals, and their secretory dynamics remain poorly defined [
62]. Moreover, macula densa–like regions appear disorganized or absent, precluding accurate reconstruction of the sodium-feedback loop [
83,
84].
Efforts to mimic the functional topology of the JGA are underway in microfluidic and organ-on-chip systems, where pressure-sensitive flow interfaces and localized signaling gradients have been introduced. Yet these remain conceptual prototypes, and no model to date demonstrates regulated, feedback-responsive renin release, nor integration with downstream endocrine axes (e.g., aldosterone, ADH) [
25,
77].
3.7. Peritubular Capillaries (Microvascular Support)
Peritubular capillaries are essential for maintaining nephron function by supplying oxygen and nutrients, removing reabsorbed solutes, and supporting active transport processes across the tubular epithelium [
85]. Originating from the efferent arteriole, these capillaries form an extensive, low-pressure vascular network around the tubules, enabling close exchange between blood and filtrate. They also participate in inflammatory signaling, endothelial–epithelial cross-talk, and regulation of renal oxygen tension, features critical for both kidney homeostasis and injury response [
85,
86].
Replicating peritubular microvasculature in vitro remains a major challenge. Current approaches have focused on vascularized organoids, endothelialized microfluidic chips, and scaffold reseeding with endothelial cells [
20]. In co-culture models, endothelial networks have been shown to improve tubular cell polarization, tight junction formation, and mitochondrial density, particularly under perfused conditions. Vascular flow enhances epithelial survival and function by mimicking physiological shear stress and facilitating oxygen and nutrient delivery [
87,
88,
89].
Despite this progress, most systems lack hierarchical vascular organization, long-term perfusion stability, and integration with functional tubular modules. While transient networks can be induced by co-culturing with angiogenic factors, they are often unstable and prone to regression. Whole-organ scaffold reseeding offers potential for anatomically accurate vascular regeneration but remains limited by incomplete endothelial coverage and thrombosis risk upon implantation [
87,
88,
89].
4. From Technologies to Translation: Integration Challenges and Clinical Pathways
Although individual platforms contribute promising capabilities to nephron replacement, their clinical potential must be evaluated through shared translational benchmarks rather than platform-specific achievements.
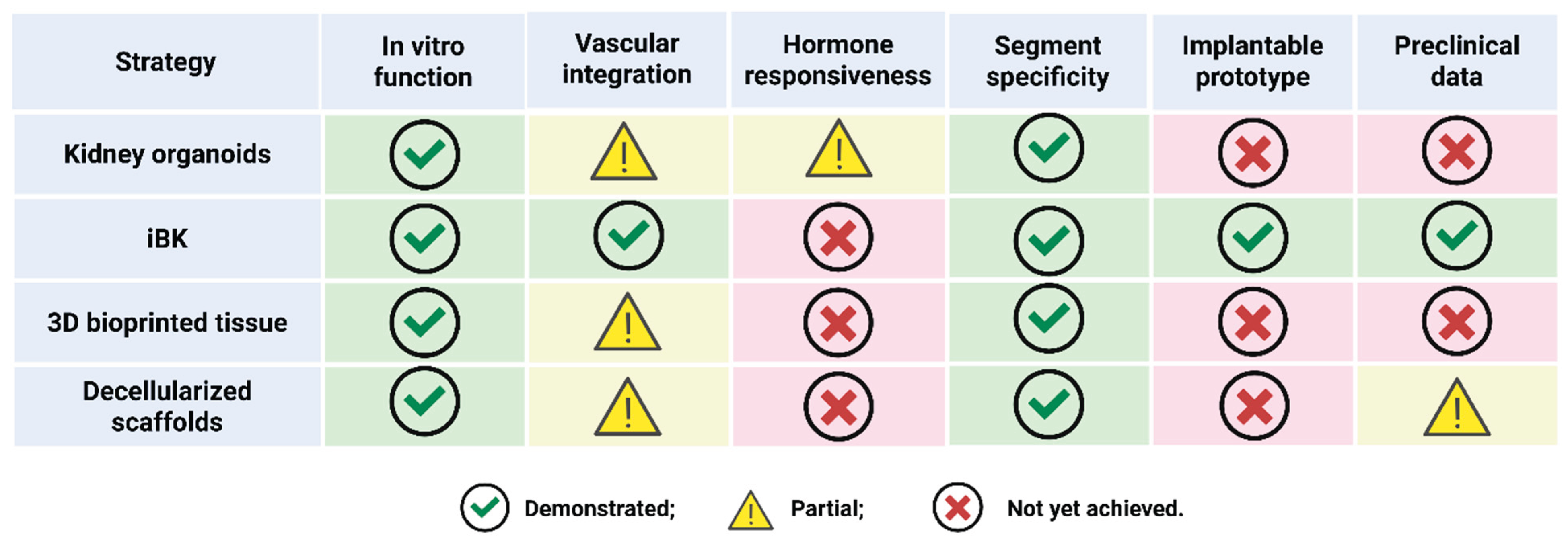
Figure 2 summarizes this comparative landscape, mapping four leading nephron replacement strategies against six critical domains: in vitro function, vascular integration, hormone responsiveness, segment specificity, implantability, and preclinical validation.
The matrix highlights both achievements and persistent gaps. While progress has been made in filtration and reabsorption, essential nephron functions such as endocrine signaling, acid-base regulation, and immune modulation remain poorly replicated or entirely absent [
36]. Key structures like the juxtaglomerular apparatus and peritubular capillaries are not yet recapitulated, and functional cross-talk between nephron segments is largely missing. These “orphan functions” must be addressed to achieve true physiological integration.
Three clear trends emerge. First, in vitro function and segment specificity have been successfully demonstrated across many platforms, particularly organoids, chip-based systems, and 3D-bioprinted constructs. These models capture transcriptional identity and localized function [
37]. However, they remain disconnected from the vascular, hormonal, and systemic integration needed for clinical translation.
Second, vascular integration and long-term perfusion remain major barriers. While endothelial co-culture and scaffold reseeding have produced partial capillary networks, no platform yet sustains stable, perfusable, hierarchical vasculature at a clinically relevant scale [
87,
89,
90].
Third, implantability and in vivo validation have progressed mainly within device-based approaches, particularly the iBK. These systems have demonstrated filtration and mechanical durability in large-animal models. However, they lack segmental complexity and endocrine responsiveness. In contrast, biologically advanced constructs, such as organoids and bioprinted tissues, offer richer cellular architecture but remain non-implantable due to limitations in scalability, perfusion, and immune shielding [
18,
41].
Addressing these limitations requires more than technological advancement; it also demands thoughtful integration into clinical contexts. Modular nephron replacement systems may first serve as adjuncts to dialysis or bridge therapies for patients awaiting transplantation [
30,
91]. They may also benefit populations with acute kidney injury or early-stage chronic disease, where even partial restoration of function can reduce dependence on conventional therapies [
8,
92]. These patient-centered applications should guide modular design, performance targets, and deployment models. In the long term, integrating complementary modules may offer the most viable path toward clinically functional systems. For example, iBK filtration units could be coupled with organoid-derived tubular modules to extend functionality, or vascularized scaffolds could serve as platforms for engineered glomerular and endocrine segments [
23,
37,
49].
Regulatory complexity also threatens to slow translation. Biological platforms, like organoids or decellularized constructs, face extensive review due to the use of living cells [
93]. Agencies such as the FDA require long-term data on safety, sourcing, and reproducibility, potentially delaying trials by 5–10 years [
94,
95]. In contrast, mechanical systems like iBKs, built on biocompatible materials, face faster review (typically 3–5 years), as seen in the Kidney Project. Hybrid systems will likely require new regulatory frameworks to evaluate interoperability, durability, and long-term integration. Programs like KidneyX and the FDA’s Breakthrough Device designation are pivotal, but standardized guidance for modular bio-devices remains lacking.
To support real-world modular assembly, practical design standards will be essential. Organoid segments might be coupled to iBK membranes using vascular connectors or shared flow systems. Scaffolds could serve as structured platforms for stacking segment-specific modules. Designing “plug-and-play” interfaces for vascular coupling, tubular continuity, and immune protection will be key to future integration.
In summary, no single platform replicates all core nephron functions or satisfies clinical readiness. A modular strategy that combines the mechanical strengths of engineered devices with the biological realism of cellular constructs offers the clearest path forward. By bridging device functionality with biological complexity, interoperable nephron modules may one day fulfill the clinical promise of systemic kidney function replacement [
90,
91].
5. Conclusions and Ethical Outlook
Nephron replacement strategies mark a paradigm shift in kidney care, offering modular, function-specific alternatives to dialysis and transplantation. Platforms such as kidney organoids, iBK, 3D-bioprinted tissues, and decellularized scaffolds have made substantial progress, especially in replicating filtration and reabsorption. Yet major gaps remain in replicating endocrine function, vascular integration, and immune regulation, processes critical to systemic homeostasis [
18,
40,
47].
Looking forward, the most promising path lies in combining platform strengths within modular architectures. Using the nephron’s architecture as a blueprint, modular assemblies can be designed to replicate specific functions and scaled according to patient need. This approach prioritizes flexibility, scalability, and personalization [
8,
35,
37].
However, achieving clinical viability will require more than technological refinement. Rigorous preclinical validation, functional interoperability, and ethically grounded deployment strategies must accompany innovation. Real-world impact will depend not only on what these systems can do, but also on how they are implemented, accessed, and trusted.
The ethical implications of these technologies are significant and must be addressed proactively [
96]. A central concern is the sourcing of iPSCs used in organoids and scaffolds. Although iPSCs from underrepresented populations may improve compatibility, they raise questions around informed consent, genetic privacy, and equitable participation in cell biobanks [
97,
98]. Transparent recruitment, robust safeguards, and community involvement will be essential to ensure fairness and trust.
Access to equity poses another major challenge. High development costs for bioengineered therapies, such as iBK’s silicon nanopore membranes or scaffold recellularization, could exacerbate inequities if pricing models mirror those of dialysis [
3,
99]. To avoid reinforcing disparities, open-access innovation, public–private partnerships, and tiered pricing strategies should be explored [
99].
Beyond logistics, nephron replacement introduces profound societal and moral questions. Who will benefit? How will costs be managed in the regions most burdened by CKD? How will consent and oversight be handled for autologous or genetically engineered cells? And as these systems become increasingly integrated into the human body, new boundaries will be tested, including those between therapy and enhancement, biology and bioengineering, treatment and commodification. To navigate this future responsibly, the development of a guiding ethical framework, centered on transparency, accessibility, proportionality, and therapeutic intent, will be essential.
In summary, nephron replacement is no longer a distant vision but an emerging frontier. As the field advances, ethical, regulatory, and social frameworks must evolve in parallel with science. Only by aligning innovation with values of equity and responsibility can nephron replacement strategies truly fulfill their transformative potential.
Author Contributions
Conceptualization, writing – original draft preparation, visualization, N.S.; Writing—review and editing, Ye.T.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Conflicts of Interest
The authors declare no conflicts of interest.
References
- Li, M.-J.; Liu, H.-Y.; Zhang, Y.-Q.; Li, S.-R.; Zhang, J.-H.; Li, R. Global Burden of Chronic Kidney Disease and Its Attributable Risk Factors (1990-2021): An Analysis Based on the Global Burden of Disease Study. Front Endocrinol (Lausanne) 2025, 16, 1563246. [CrossRef]
- Francis, A.; Harhay, M.N.; Ong, A.C.M.; Tummalapalli, S.L.; Ortiz, A.; Fogo, A.B.; Fliser, D.; Roy-Chaudhury, P.; Fontana, M.; Nangaku, M.; et al. Chronic Kidney Disease and the Global Public Health Agenda: An International Consensus. Nature Reviews Nephrology 2024 20:7 2024, 20, 473–485. [CrossRef]
- Nkunu, V.; Tungsanga, S.; Diongole, H.M.; Sarki, A.; Arruebo, S.; Caskey, F.J.; Damster, S.; Donner, J.A.; Jha, V.; Levin, A.; et al. Landscape of Kidney Replacement Therapy Provision in Low- and Lower-Middle Income Countries: A Multinational Study from the ISN-GKHA. PLOS Global Public Health 2024, 4, e0003979. [CrossRef]
- Bello, A.K.; Okpechi, I.G.; Osman, M.A.; Cho, Y.; Htay, H.; Jha, V.; Wainstein, M.; Johnson, D.W. Epidemiology of Haemodialysis Outcomes. Nat Rev Nephrol 2022, 18, 378-395. [CrossRef]
- Lanini, I.; Samoni, S.; Husain-Syed, F.; Fabbri, S.; Canzani, F.; Messeri, A.; Mediati, R.D.; Ricci, Z.; Romagnoli, S.; Villa, G. Palliative Care for Patients with Kidney Disease. J Clin Med 2022, 11, 3923. [CrossRef]
- Kupiec-Weglinski, J.W. Grand Challenges in Organ Transplantation. Frontiers in Transplantation 2022, 1, 897679. [CrossRef]
- Kolesnyk, M.; Korol, L.; Shifris, I.; Stepanova, N.; Voroniak, O.; Shuba, I. Immunological determinants of long-term kidney graft survival as therapeutic targets. Ukr J Nephrol Dialys. 2025, 2, 98-111. [CrossRef]
- Salani, M.; Roy, S.; Fissell, W.H. Innovations in Wearable and Implantable Artificial Kidneys. Am J Kidney Dis. 2018, 72, 745–751. [CrossRef]
- Copur, S.; Tanriover, C.; Yavuz, F.; Soler, M.J.; Ortiz, A.; Covic, A.; Kanbay, M. Novel Strategies in Nephrology: What to Expect from the Future? Clin Kidney J 2023, 16, 230–244. [CrossRef]
- Bonventre, J. V.; Hurst, F. P.; West ,M.; Wu I.; Roy-Chaudhury, P.; Sheldon M. A Technology Roadmap for Innovative Approaches to Kidney Replacement Therapies: A Catalyst for Change. Clin J Am Soc Nephrol. 2019, 14, 1539-1547. [CrossRef]
- Guo, X.; Liu, B.; Zhang, Y.; Cheong, S.; Xu, T.; Lu, F.; He, Y. Decellularized Extracellular Matrix for Organoid and Engineered Organ Culture. J Tissue Eng 2024, 15, 20417314241300386. [CrossRef]
- Zheng, K.; Chai, M.; Luo, B.; Cheng, K.; Wang, Z.; Li, N.; Shi, X. Recent Progress of 3D Printed Vascularized Tissues and Organs. Smart Mater Med 2024, 5, 183–195. [CrossRef]
- Christou, C.D.; Vasileiadou, S.; Sotiroudis, G.; Tsoulfas, G. Three-Dimensional Printing and Bioprinting in Renal Transplantation and Regenerative Medicine: Current Perspectives. J Clin Med 2023, 12, 6520. [CrossRef]
- Chambers, B.E.; Weaver, N.E.; Wingert, R.A. The “3Ds” of Growing Kidney Organoids: Advances in Nephron Development, Disease Modeling, and Drug Screening. Cells 2023, 12, 549. [CrossRef]
- Tekguc, M.; Gaal, R.C.V.; Uzel, S.G.M.; Gupta, N.; Riella, L. V.; Lewis, J.A.; Morizane, R. Kidney Organoids: A Pioneering Model for Kidney Diseases. Transl Res 2022, 250, 1-17.. [CrossRef]
- Unbekandt, M.; Davies, J.A. Dissociation of Embryonic Kidneys Followed by Reaggregation Allows the Formation of Renal Tissues. Kidney Int 2010, 77, 407–416. [CrossRef]
- Xia, Y.; Nivet, E.; Sancho-Martinez, I.; Gallegos, T.; Suzuki, K.; Okamura, D.; Wu, M.Z.; Dubova, I.; Esteban, C.R.; Montserrat, N.; et al. Directed Differentiation of Human Pluripotent Cells to Ureteric Bud Kidney Progenitor-Like Cells. Nat Cell Biol 2013, 15, 1507–1515. [CrossRef]
- Takasato, M.; Er, P.X.; Chiu, H.S.; Maier, B.; Baillie, G.J.; Ferguson, C.; Parton, R.G.; Wolvetang, E.J.; Roost, M.S.; De Sousa Lopes, S.M.C.; et al. Kidney Organoids from Human IPS Cells Contain Multiple Lineages and Model Human Nephrogenesis. Nature 2015, 526, 564–568. [CrossRef]
- Tabibzadeh, N.; Morizane, R. Advancements in Therapeutic Development: Kidney Organoids and Organs-on-a-Chip. Kidney Int 2024, 105, 702708. [CrossRef]
- Liu, Q.; Yue, L.; Deng, J.; Tan, Y.; Wu, C. Progress and Breakthroughs in Human Kidney Organoid Research. Biochem Biophys Rep 2024, 39, 101736. [CrossRef]
- Wilson, S.B.; Vanslambrouck, J.M.; Murphy, A.; Neavin, D.R.; Powell, J.E.; Howden, S.E.; Little, M.H. Classification of Indeterminate and Off-Target Cell Types within Human Kidney Organoid Differentiation. bioRxiv 2025, 2025.05.16.654519. [CrossRef]
- Goux Corredera, I.; Amato, G.; Moya-Rull, D.; Garreta, E.; Montserrat, N. Unlocking the Full Potential of Human Pluripotent Stem Cell–Derived Kidney Organoids through Bioengineering. Kidney Int 2025, 108, 38–47. [CrossRef]
- Papamichail, L.; Koch, L.S.; Veerman, D.; Broersen, K.; van der Meer, A.D. Organoids-on-a-Chip: Microfluidic Technology Enables Culture of Organoids with Enhanced Tissue Function and Potential for Disease Modeling. Front Bioeng Biotechnol 2025, 13, 1515340. [CrossRef]
- Menéndez, A.B.C.; Du, Z.; van den Bosch, T.P.P.; Othman, A.; Gaio, N.; Silvestri, C.; Quirós, W.; Lin, H.; Korevaar, S.; Merino, A.; et al. Creating a Kidney Organoid-Vasculature Interaction Model Using a Novel Organ-on-Chip System. Sci Rep 2022, 12, 20699. [CrossRef]
- Maggiore, J.C.; LeGraw, R.; Przepiorski, A.; Velazquez, J.; Chaney, C.; Vanichapol, T.; Streeter, E.; Almuallim, Z.; Oda, A.; Chiba, T.; et al. A Genetically Inducible Endothelial Niche Enables Vascularization of Human Kidney Organoids with Multilineage Maturation and Emergence of Renin Expressing Cells. Kidney Int 2024, 106, 1086–1100. [CrossRef]
- Raykhel, I.; Nishikawa, M.; Sakai, Y.; Vainio, S.J.; Skovorodkin, I. Vascularization of Kidney Organoids: Different Strategies and Perspectives. Frontiers in Urology 2024, 4, 1355042. [CrossRef]
- Wang, R.; Sui, Y.; Liu, Q.; Xiong, Y.; Li, S.; Guo, W.; Xu, Y.; Zhang, S. Recent Advances in Extracellular Matrix Manipulation for Kidney Organoid Research. Front Pharmacol 2024, 15, 1472361. [CrossRef]
- Corridon, P.R.; Ko, I.K.; Yoo, J.J.; Atala, A. Bioartificial Kidneys. Curr Stem Cell Rep 2017, 3, 68-76. [CrossRef]
- Nalesso, F.; Garzotto, F.; Cattarin, L.; Bettin, E.; Cacciapuoti, M.; Silvestre, C.; Stefanelli, L. F.; Furian, L.; Calò, L. A. The Future for End-Stage Kidney Disease Treatment: Implantable Bioartificial Kidney Challenge. Applied Sciences 2024; 14, 491. [CrossRef]
- van Gelder, M.K.; Mihaila, S.M.; Jansen, J.; Wester, M.; Verhaar, M.C.; Joles, J.A.; Stamatialis, D.; Masereeuw, R.; Gerritsen, K.G.F. From Portable Dialysis to a Bioengineered Kidney. Expert Rev Med Devices 2018, 15, 323–336. [CrossRef]
- Tumlin, J.; Wali, R.; Williams, W.; Murray, P.; Tolwani, A.J.; Vinnikova, A.K.; Szerlip, H.M.; Ye, J.; Paganini, E.P.; Dworkin, L.; et al. Efficacy and Safety of Renal Tubule Cell Therapy for Acute Renal Failure. J Am Soc Nephrol 2008, 19, 1034. [CrossRef]
- Song, J.H.; Humes, H.D. The Bioartificial Kidney in the Treatment of Acute Kidney Injury. Curr Drug Targets 2009, 10, 1227. [CrossRef]
- Kim, E.J.; Chen, C.; Gologorsky, R.; Santandreu, A.; Torres, A.; Wright, N.; Goodin, M.S.; Moyer, J.; Chui, B.W.; Blaha, C.; et al. Feasibility of an Implantable Bioreactor for Renal Cell Therapy Using Silicon Nanopore Membranes. Nat Commun 2023, 14, 4890. [CrossRef]
- Groth, T.; Stegmayr, B. G.; Ash, S. R.; Kuchinka, J.; Wieringa, F. P.; Fissell, W. H.; Roy, S. Wearable and implantable artificial kidney devices for end-stage kidney disease treatment: Current status and review. Artif. Organs. 2023, 47, 649–666. [CrossRef]
- Karageorgos, F.F.; Neiros, S.; Karakasi, K.-E.; Vasileiadou, S.; Katsanos, G.; Antoniadis, N.; Tsoulfas, G. Artificial Kidney: Challenges and Opportunities. World J Transplant 2024, 14, 89025. [CrossRef]
- Jena, R.; Aggarwal, A.; Choudhary, G.R.; Bajpai, N.K.; Jena, R.; Aggarwal, A.; Choudhary, G.R.; Bajpai, N.K. Current Status and Future of Artificial Kidney in Humans. Indian J Nephrol 2022, 32, 531–538. [CrossRef]
- Rayat Pisheh, H.; Haghdel, M.; Jahangir, M.; Hoseinian, M.S.; Rostami Yasuj, S.; Sarhadi Roodbari, A. Effective and New Technologies in Kidney Tissue Engineering. Front Bioeng Biotechnol 2024, 12, 1476510. [CrossRef]
- Fransen, M.F.J.; Addario, G.; Bouten, C.V.C.; Halary, F.; Moroni, L.; Mota, C. Bioprinting of Kidney in Vitro Models: Cells, Biomaterials, and Manufacturing Techniques. Essays Biochem 2021, 65, 587-602. [CrossRef]
- Mirshafiei, M.; Rashedi, H.; Yazdian, F.; Rahdar, A.; Baino, F. Advancements in Tissue and Organ 3D Bioprinting: Current Techniques, Applications, and Future Perspectives. Mater Des 2024, 240, 112853. [CrossRef]
- Jeong, H.J.; Nam, H.; Jang, J.; Lee, S.J. 3D Bioprinting Strategies for the Regeneration of Functional Tubular Tissues and Organs. Bioengineering 2020, 7, 32. [CrossRef]
- Homan, K.A.; Kolesky, D.B.; Skylar-Scott, M.A.; Herrmann, J.; Obuobi, H.; Moisan, A.; Lewis, J.A. Bioprinting of 3D Convoluted Renal Proximal Tubules on Perfusable Chips. Sci Rep 2016, 6, 34845. [CrossRef]
- Huang, W.; Chen, Y.Y.; He, F.F.; Zhang, C. Revolutionizing Nephrology Research: Expanding Horizons with Kidney-on-a-Chip and Beyond. Front Bioeng Biotechnol 2024, 12, 1373386. [CrossRef]
- Mou, X.; Shah, J.; Roye, Y.; Du, C.; Musah, S. An Ultrathin Membrane Mediates Tissue-Specific Morphogenesis and Barrier Function in a Human Kidney Chip. Sci Adv 2024, 10, eadn2689. [CrossRef]
- Singh, N.K.; Kim, J.Y.; Lee, J.Y.; Lee, H.; Gao, G.; Jang, J.; Kim, Y.K.; Cho, D.W. Coaxial Cell Printing of a Human Glomerular Model: An in Vitro Glomerular Filtration Barrier and Its Pathophysiology. Biofabrication 2023, 15, 10.1088/1758-5090/acad2c. [CrossRef]
- Carreno-Caleano, G.; Ali, M.; Yoo, J.J.; Lee, S.J.; Atala, A. 3D Bioprinted Renal Constructs Using Kidney-Specific ECM Bioink System on Kidney Regeneration. Adv Healthc Mater 2025, 2502576. [CrossRef]
- Yuan, Z.; Bai, X.; Li, S.; Fu, Y.; Wan, Z.; Guo, X.; Zhai, M.; Yi, J.; Liu, Y.; Zhou, Y.; et al. Multimaterial and Multidimensional Bioprinting in Regenerative Medicine: Advances, Limitations, and Future Directions. Adv Healthc Mater 2025, 2500475. [CrossRef]
- Agarwal, T.; Onesto, V.; Banerjee, D.; Guo, S.; Polini, A.; Vogt, C. D.; Viswanath, A.; Esworthy, T.; Cui, H.; O’Donnell, A.; Vajanthri, K. Y.; Moroni, L.; et al. 3D bioprinting in tissue engineering: current state-of-the-art and challenges towards system standardization and clinical translation. Biofabrication 2025, 10.1088/1758-5090/ade47a. Advance online publication. [CrossRef]
- 4Bonventre, J. V.; Hurst, F.P.; West, M.; Wu, I.; Roy-Chaudhury, P.; Sheldon, M. A Technology Roadmap for Innovative Approaches to Kidney Replacement Therapies a Catalyst for Change. Clin J Am Soc Nephrol. 2019, 14, 1539-1547. [CrossRef]
- de Haan, M.J.A.; Witjas, F.M.R.; Engelse, M.A.; Rabelink, T.J. Have We Hit a Wall with Whole Kidney Decellularization and Recellularization: A Review. Curr Opin Biomed Eng 2021, 20, 100335. [CrossRef]
- Destefani, A.C.; Sirtoli, G.M.; Nogueira, B.V. Advances in the Knowledge about Kidney Decellularization and Repopulation. Front Bioeng Biotechnol 2017, 5, 255314. [CrossRef]
- Khosropanah, M.H.; Torabinavid, P.; Azimzadeh, A.; Tanourlouee, S.B.; Kajbafzadeh, A.M. Efficient Decellularization of Human Fetal Kidneys through Optimized SDS Exposure. Sci Rep 2024, 14, 21545. [CrossRef]
- Sullivan, D.C.; Mirmalek-Sani, S.H.; Deegan, D.B.; Baptista, P.M.; Aboushwareb, T.; Atala, A.; Yoo, J.J. Decellularization Methods of Porcine Kidneys for Whole Organ Engineering Using a High-Throughput System. Biomaterials 2012, 33, 7756–7764. [CrossRef]
- Figliuzzi, M.; Bonandrini, B.; Remuzzi, A. Decellularized Kidney Matrix as Functional Material for Whole Organ Tissue Engineering. J Appl Biomater Funct Mater 2017, 15, e326–e333. [CrossRef]
- Torabinavid, P.; Khosropanah, M.H.; Azimzadeh, A.; Kajbafzadeh, A.M. Current Strategies on Kidney Regeneration Using Tissue Engineering Approaches: A Systematic Review. BMC Nephrol 2025, 26, 66. [CrossRef]
- Luque-Badillo, A.C.; Monjaras-Avila, C.U.; Adomat, H.; So, A.; Chavez-Muñoz, C. Evaluating Different Methods for Kidney Recellularization. Sci Rep 2024, 14, 23520. [CrossRef]
- Choi, M.; Al Fahad, M.A.; Shanto, P.C.; Park, S. su; Lee, B.T. Surface Modification of Decellularized Kidney Scaffold with Chemokine and AKI-CKD Cytokine Juice to Increase the Recellularization Efficiency of Bio-Engineered Kidney. Biomaterials 2025, 316, 123007. [CrossRef]
- Theodorou, C.; Leatherby, R.; Dhanda, R. Function of the Nephron and the Formation of Urine. Anaesthesia & Intensive Care Medicine 2021, 22, 434–438. [CrossRef]
- Koulouridis, E.; Koulouridis, I. The loop of Henle as the milestone of mammalian kindey concentrating ability: a historical review. Acta Med Hist Adriat. 2014, 12, 413-428.
- Olde Hanhof, C.J.A.; Yousef Yengej, F.A.; Rookmaaker, M.B.; Verhaar, M.C.; Van Der Wijst, J.; Hoenderop, J.G. Modeling Distal Convoluted Tubule (Patho)Physiology: An Overview of Past Developments and an Outlook Toward the Future. Tissue Eng Part C Methods 2021, 27, 200–212. [CrossRef]
- Lam, A.Q.; Bonventre, J. V. Regenerating the Nephron with Human Pluripotent Stem Cells. Curr Opin Organ Transplant 2015, 20, 187–192. [CrossRef]
- Haraldsson, B.; Nyström, J.; Deen, W.M. Properties of the Glomerular Barrier and Mechanisms of Proteinuria. Physiol Rev 2008, 88, 451–487. [CrossRef]
- van den Berg, C.W.; Dumas, S.J.; Little, M.H.; Rabelink, T.J. Challenges in Maturation and Integration of Kidney Organoids for Stem Cell–Based Renal Replacement Therapy. Kidney Int 2025, 107, 262–270. [CrossRef]
- Mazloomnejad, R.; Babajani, A.; Kasravi, M.; Ahmadi, A.; Shariatzadeh, S.; Bahrami, S.; Niknejad, H. Angiogenesis and Re-Endothelialization in Decellularized Scaffolds: Recent Advances and Current Challenges in Tissue Engineering. Front Bioeng Biotechnol 2023, 11, 1103727. [CrossRef]
- Wang, K.; Kestenbaum, B. Proximal Tubular Secretory Clearance: A Neglected Partner of Kidney Function. Clin J Am Soc Nephrol 2018, 13, 1291. [CrossRef]
- Wagner, C.A. Beyond SGLT2: Proximal Tubule Transporters as Potential Drug Targets for Chronic Kidney Disease. Nephrol Dial Transplant 2025, 40, i18–i28. [CrossRef]
- Ross, E.J.; Gordon, E.R.; Sothers, H.; Darji, R.; Baron, O.; Haithcock, D.; Prabhakarpandian, B.; Pant, K.; Myers, R.M.; Cooper, S.J.; et al. Three Dimensional Modeling of Biologically Relevant Fluid Shear Stress in Human Renal Tubule Cells Mimics in Vivo Transcriptional Profiles. Sci Rep 2021, 11, 14053. [CrossRef]
- Kim, H.; Lee, J.B.; Kim, K.; Sung, G.Y. Effect of Shear Stress on the Proximal Tubule-on-a-Chip for Multi-Organ Microphysiological System. Journal of Industrial and Engineering Chemistry 2022, 115, 279-286. [CrossRef]
- Karp, S.; Pollak, M.R.; Subramanian, B. Disease Modeling with Kidney Organoids. Micromachines 2022, 13, 1384. [CrossRef]
- Li, S.R.; Gulieva, R.E.; Helms, L.; Cruz, N.M.; Vincent, T.; Fu, H.; Himmelfarb, J.; Freedman, B.S. Glucose Absorption Drives Cystogenesis in a Human Organoid-on-Chip Model of Polycystic Kidney Disease. Nat Commun 2022, 13, 7918. [CrossRef]
- Yu, Y.; Alkhawaji, A.; Ding, Y.; Mei, J. Decellularized Scaffolds in Regenerative Medicine. Oncotarget 2016, 7, 58671. [CrossRef]
- Marulli, M.; Edwards, A.; Milišić, V.; Vauchelet, N. On the Role of the Epithelium in a Model of Sodium Exchange in Renal Tubules. Math Biosci 2020, 321. [CrossRef]
- Pahuja, A.; Goux Corredera, I.; Moya-Rull, D.; Garreta, E.; Montserrat, N. Engineering Physiological Environments to Advance Kidney Organoid Models from Human Pluripotent Stem Cells. Curr Opin Cell Biol 2024, 86, 102306. [CrossRef]
- Liu, M.; Cardilla, A.; Ngeow, J.; Gong, X.; Xia, Y. Studying Kidney Diseases Using Organoid Models. Front Cell Dev Biol 2022, 10, 845401. [CrossRef]
- McCormick, J.A.; Ellison, D.H. Distal Convoluted Tubule. Compr Physiol 2015, 5, 2147-2163. [CrossRef]
- Dilmen, E.; Olde Hanhof, C.J.A.; Yousef Yengej, F.A.; Ammerlaan, C.M.E.; Rookmaaker, M.B.; Orhon, I.; Jansen, J.; Verhaar, M.C.; Hoenderop, J.G. A Semi-Permeable Insert Culture Model for the Distal Part of the Nephron with Human and Mouse Tubuloid Epithelial Cells. Exp Cell Res 2025, 444. [CrossRef]
- Yousef Yengej, F.A.; Pou Casellas, C.; Ammerlaan, C.M.E.; Olde Hanhof, C.J.A.; Dilmen, E.; Beumer, J.; Begthel, H.; Meeder, E.M.G.; Hoenderop, J.G.; Rookmaaker, M.B.; et al. Tubuloid Differentiation to Model the Human Distal Nephron and Collecting Duct in Health and Disease. Cell Rep 2024, 43, 113614. [CrossRef]
- Zeng, Z.; Huang, B.; Parvez, R.K.; Li, Y.; Chen, J.; Vonk, A.C.; Thornton, M.E.; Patel, T.; Rutledge, E.A.; Kim, A.D.; et al. Generation of Patterned Kidney Organoids That Recapitulate the Adult Kidney Collecting Duct System from Expandable Ureteric Bud Progenitors. Nat Commun 2021, 3641. [CrossRef]
- Howden, S.E.; Wilson, S.B.; Groenewegen, E.; Starks, L.; Forbes, T.A.; Tan, K.S.; Vanslambrouck, J.M.; Holloway, E.M.; Chen, Y.H.; Jain, S.; et al. Plasticity of Distal Nephron Epithelia from Human Kidney Organoids Enables the Induction of Ureteric Tip and Stalk. Cell Stem Cell 2021, 28, 671-684.e6. [CrossRef]
- Wang, W.; Hu, G.; Li, G.; Li, P.-L.; Li, N. Collecting Duct-Specific Deletion of Acid Ceramidase Promoted Sodium Retention and Salt-Sensitive Hypertension. Physiology 2025, 40, S1. [CrossRef]
- Higashihara, E.; Harada, T.; Fukuhara, H. Juxtaglomerular Apparatus-Mediated Homeostatic Mechanisms: Therapeutic Implication for Chronic Kidney Disease. Expert Opin Pharmacother 2024, 25, 819–832. 819-832. [CrossRef]
- Yamaguchi, H.; Gomez, R.A.; Sequeira-Lopez, M.L.S. Renin Cells, From Vascular Development to Blood Pressure Sensing. Hypertension 2023, 80, 1580-1589. [CrossRef]
- Shankar, A.S.; Du, Z.; Mora, H.T.; van den Bosch, T.P.P.; Korevaar, S.S.; Van den Berg-Garrelds, I.M.; Bindels, E.; Lopez-Iglesias, C.; Clahsen-van Groningen, M.C.; Gribnau, J.; et al. Human Kidney Organoids Produce Functional Renin. Kidney Int 2021, 99, 134–147. [CrossRef]
- Freedman, B.S. Physiology Assays in Human Kidney Organoids. Am J Physiol Renal Physiol 2022, 322, F625-F638. [CrossRef]
- Yanofsky, S.M.; Dugas, C.M.; Katsurada, A.; Liu, J.; Saifudeen, Z.; El-Dahr, S.S.; Satou, R. Angiotensin II Biphasically Regulates Cell Differentiation in Human IPSC-Derived Kidney Organoids. Am J Physiol Renal Physiol 2021, 321, F559-F571. [CrossRef]
- Kida, Y. Peritubular Capillary Rarefaction: An Underappreciated Regulator of CKD Progression Int J Mol Sci 2020, 21, 8255. [CrossRef]
- Choi, Y.J.; Chakraborty, S.; Nguyen, V.; Nguyen, C.; Kim, B.K.; Shim, S.I.; Suki, W.N.; Truong, L.D. Peritubular Capillary Loss Is Associated with Chronic Tubulointerstitial Injury in Human Kidney: Altered Expression of Vascular Endothelial Growth Factor. Hum Pathol 2000, 31, 1491–1497. [CrossRef]
- Meng, X.; Xing, Y.; Li, J.; Deng, C.; Li, Y.; Ren, X.; Zhang, D. Rebuilding the Vascular Network: In Vivo and in Vitro Approaches. Front Cell Dev Biol 2021, 9, 639299. [CrossRef]
- Shin, Y.J.; Safina, D.; Zheng, Y.; Levenberg, S. Microvascularization in 3D Human Engineered Tissue and Organoids. Annu Rev Biomed Eng 2025, 27, 473–498. [CrossRef]
- Liu, Q.; Ying, G.; Hu, C.; Du, L.; Zhang, H.; Wang, Z.; Yue, H.; Yetisen, A.K.; Wang, G.; Shen, Y.; et al. Engineering in Vitro Vascular Microsystems. Microsystems & Nanoengineering 2025 11:1 2025, 11, 100. [CrossRef]
- Lebedenko, C.G.; Banerjee, I.A. Enhancing Kidney Vasculature in Tissue Engineering—Current Trends and Approaches: A Review. Biomimetics 2021, 6, 40. [CrossRef]
- Wieringa, F.P.; Suran, S.; Søndergaard, H.; Ash, S.; Cummins, C.; Chaudhuri, A.R.; Irmak, T.; Gerritsen, K.; Vollenbroek, J. The Future of Technology-Based Kidney Replacement Therapies: An Update on Portable, Wearable, and Implantable Artificial Kidneys. Am J Kidney Dis 2025, 85, 787–796. [CrossRef]
- Araoka, T.; Toyohara, K.; Ryosaka, M.; Inui, C.; Matsuura, M.; Ma, C.; Watahiki, J.; Li, Z.; Iwasaki, M.; Watanabe, A.; et al. Human IPSC–Derived Nephron Progenitor Cells Treat Acute Kidney Injury and Chronic Kidney Disease in Mouse Models. Sci Transl Med 2025, 17, eadt5553. [CrossRef]
- Tsokas, K.; McFarland, R.; Burke, C.; Lynch, J.L.; Bollenbach, T.; Callaway II, D.A.; Siegel, J. Reducing Risks and Delays in the Translation of Cell and Gene Therapy Innovations into Regulated Products. NAM Perspectives 2019, 2019, 10.31478/201909d. [CrossRef]
- Lowdell, M.W. Considerations for Manufacturing of Cell and Gene Medicines for Clinical Development. Cytotherapy 2025, 27, 874–883. [CrossRef]
- Iglesias-Lopez, C.; Agustí, A.; Vallano, A.; Obach, M. Current Landscape of Clinical Development and Approval of Advanced Therapies. Mol Ther Methods Clin Dev 2021, 23, 606. [CrossRef]
- Sekar, M.P.; Budharaju, H.; Zennifer, A.; Sethuraman, S.; Vermeulen, N.; Sundaramurthi, D.; Kalaskar, D.M. Current Standards and Ethical Landscape of Engineered Tissues—3D Bioprinting Perspective. J Tissue Eng 2021, 12, 20417314211027677. [CrossRef]
- Zheng, Y.L. Some Ethical Concerns About Human Induced Pluripotent Stem Cells. Sci Eng Ethics 2016, 22, 1277–1284. [CrossRef]
- Arifa, N. Induced Pluripotent Stem Cells (IPSCs): The Ethical Alternative? Stem Cell Res Reg Med 2024, 7, 251-252.
- Cintron Pregosin, N.; Bronstein, R.; Mallipattu, S.K. Recent Advances in Kidney Bioengineering. Front Pediatr 2021, 9, 743301. [CrossRef]
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).