Submitted:
21 July 2025
Posted:
24 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
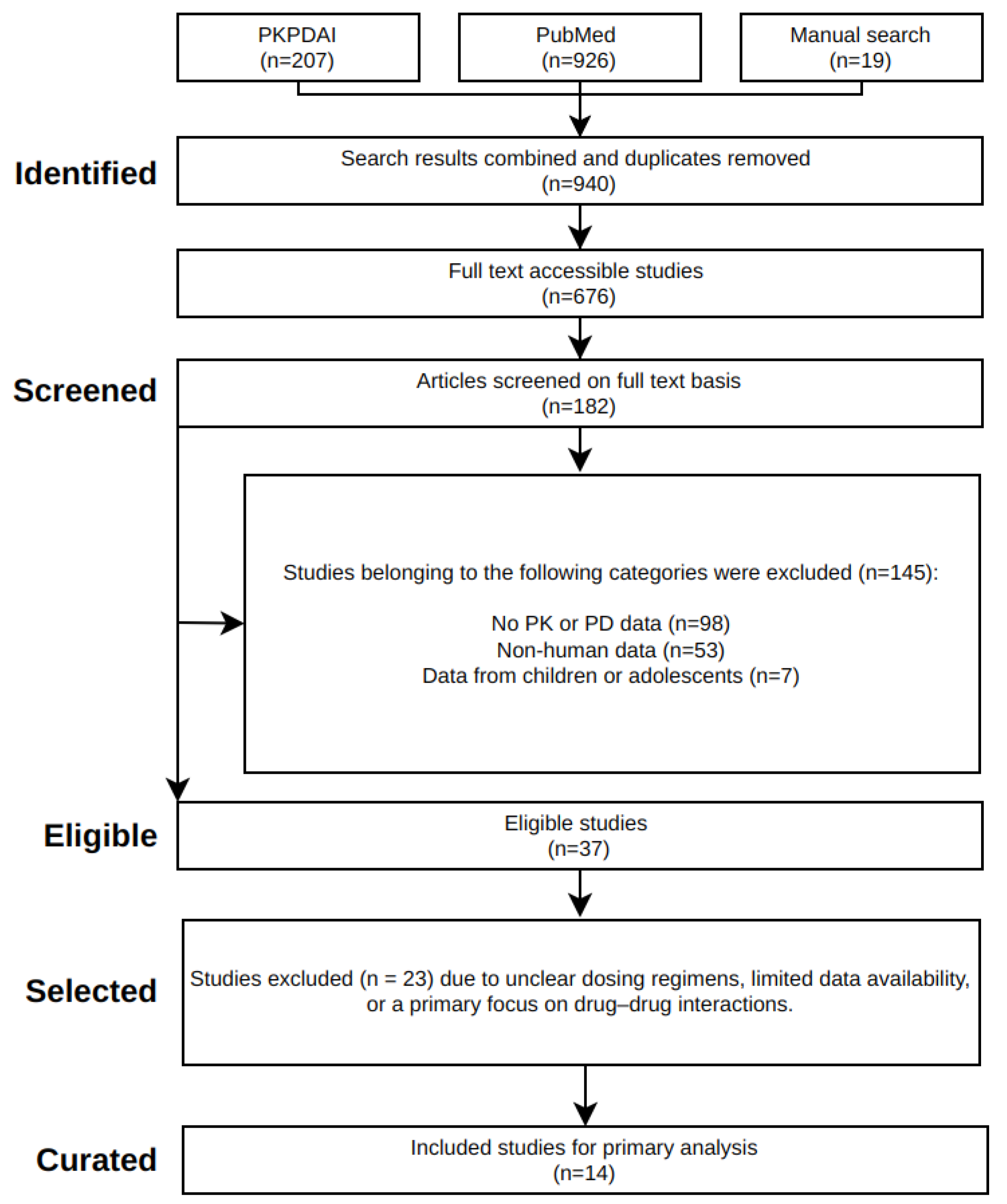
2.1. Systematic Literature Research
2.2. Data Curation
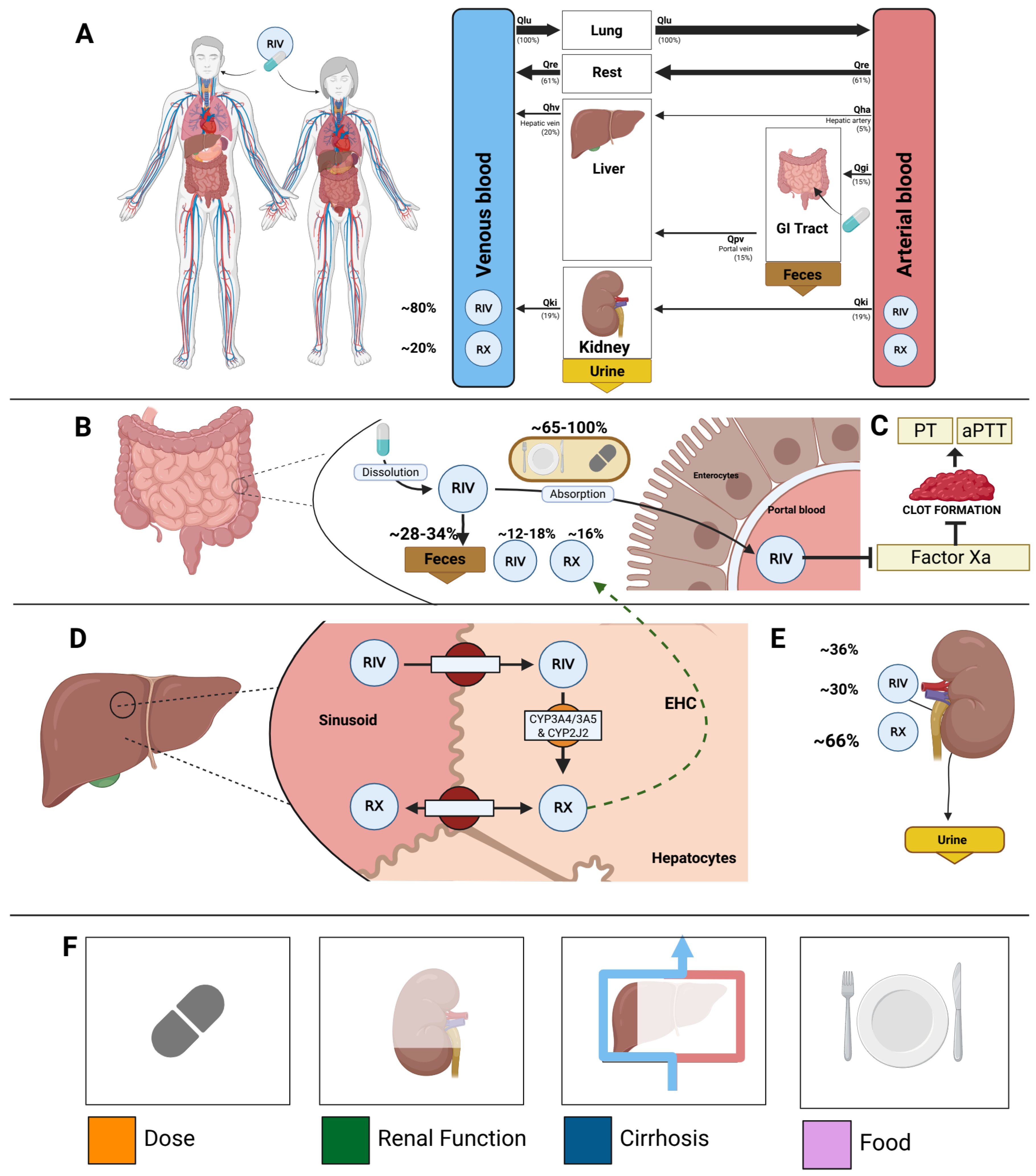
2.3. Model
2.4. Parameter Optimization
3. Results
3.1. Rivaroxaban Database
3.2. Computational Model
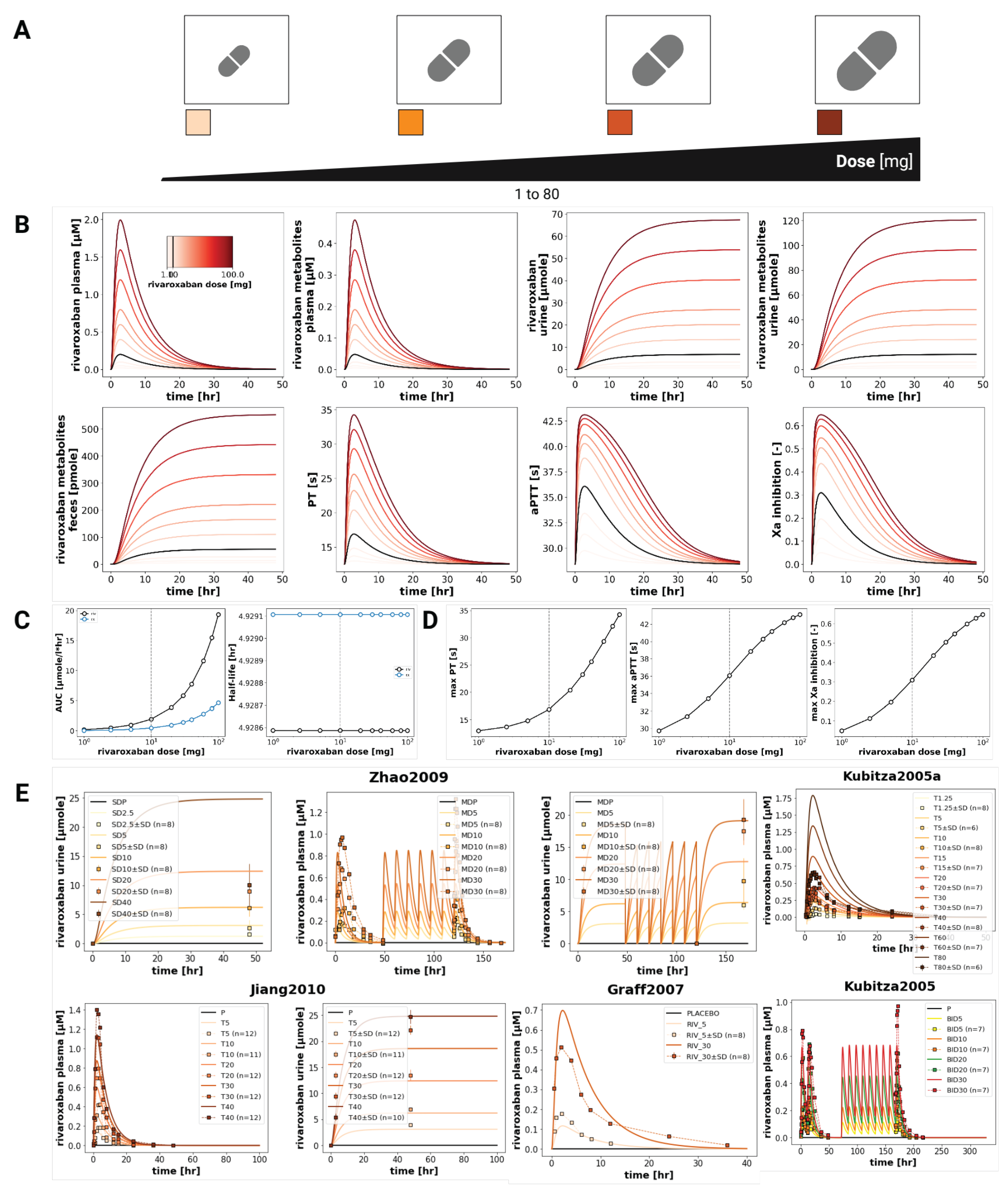
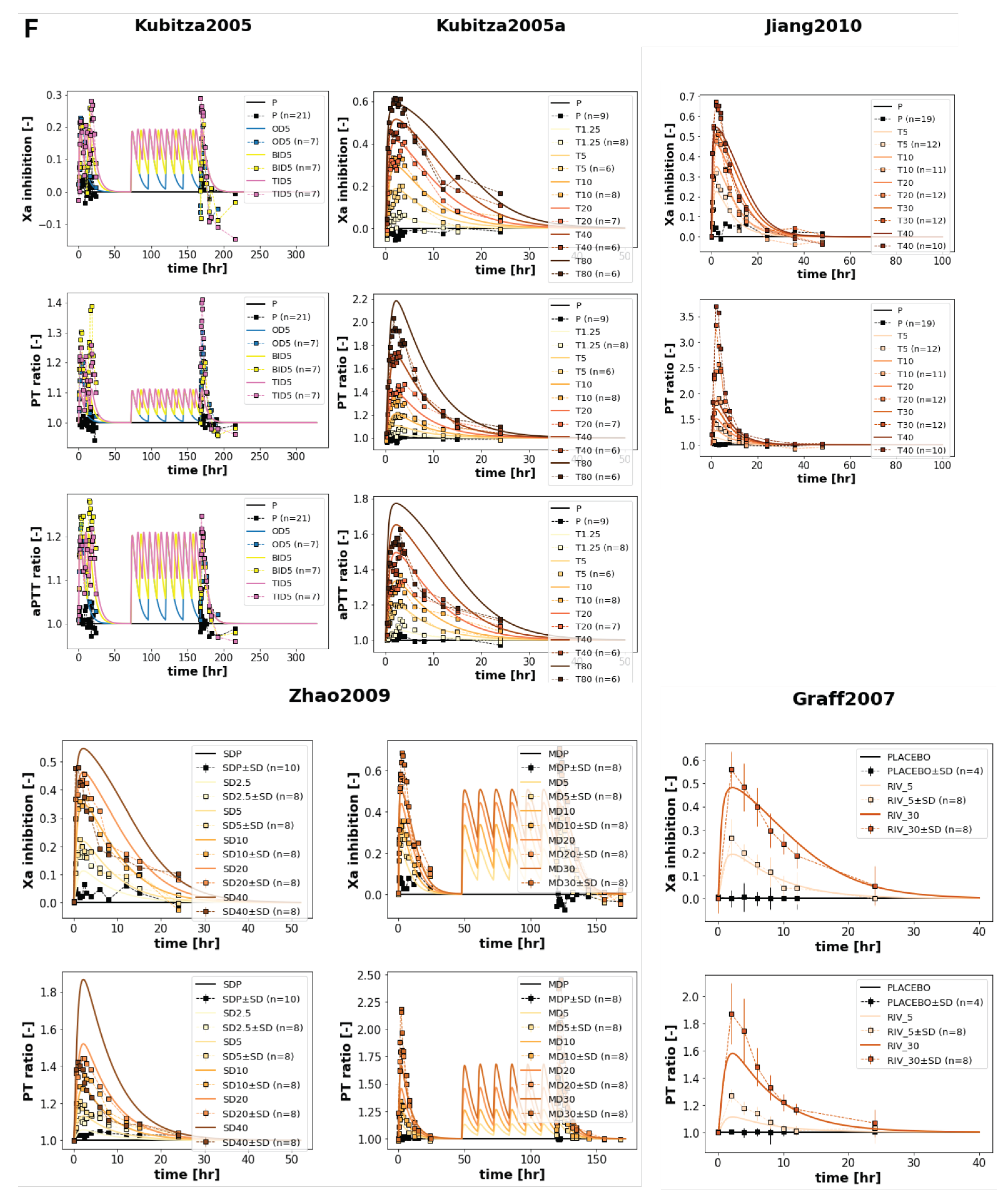
3.3. Dose Dependency
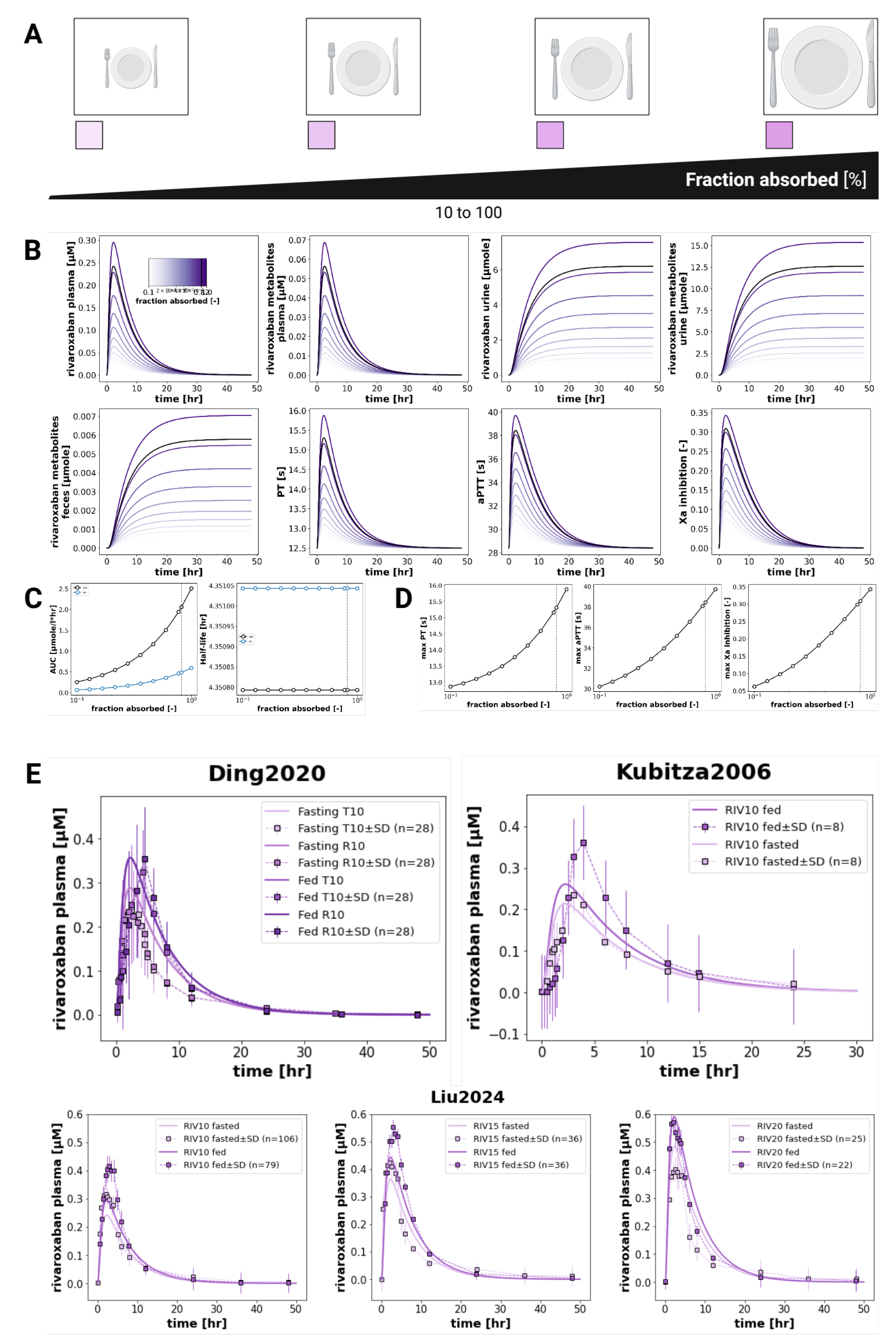
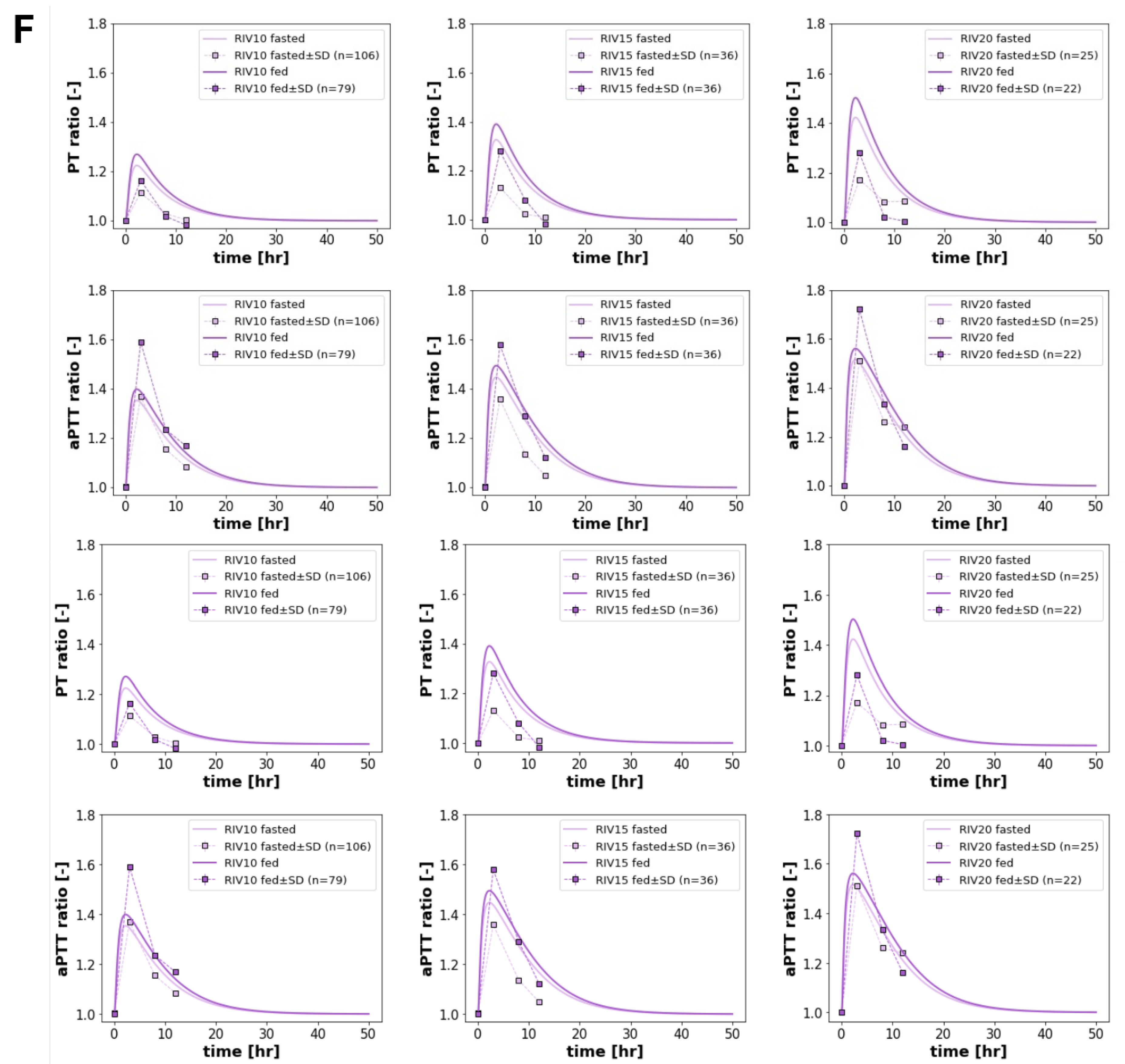
3.4. Food Effect
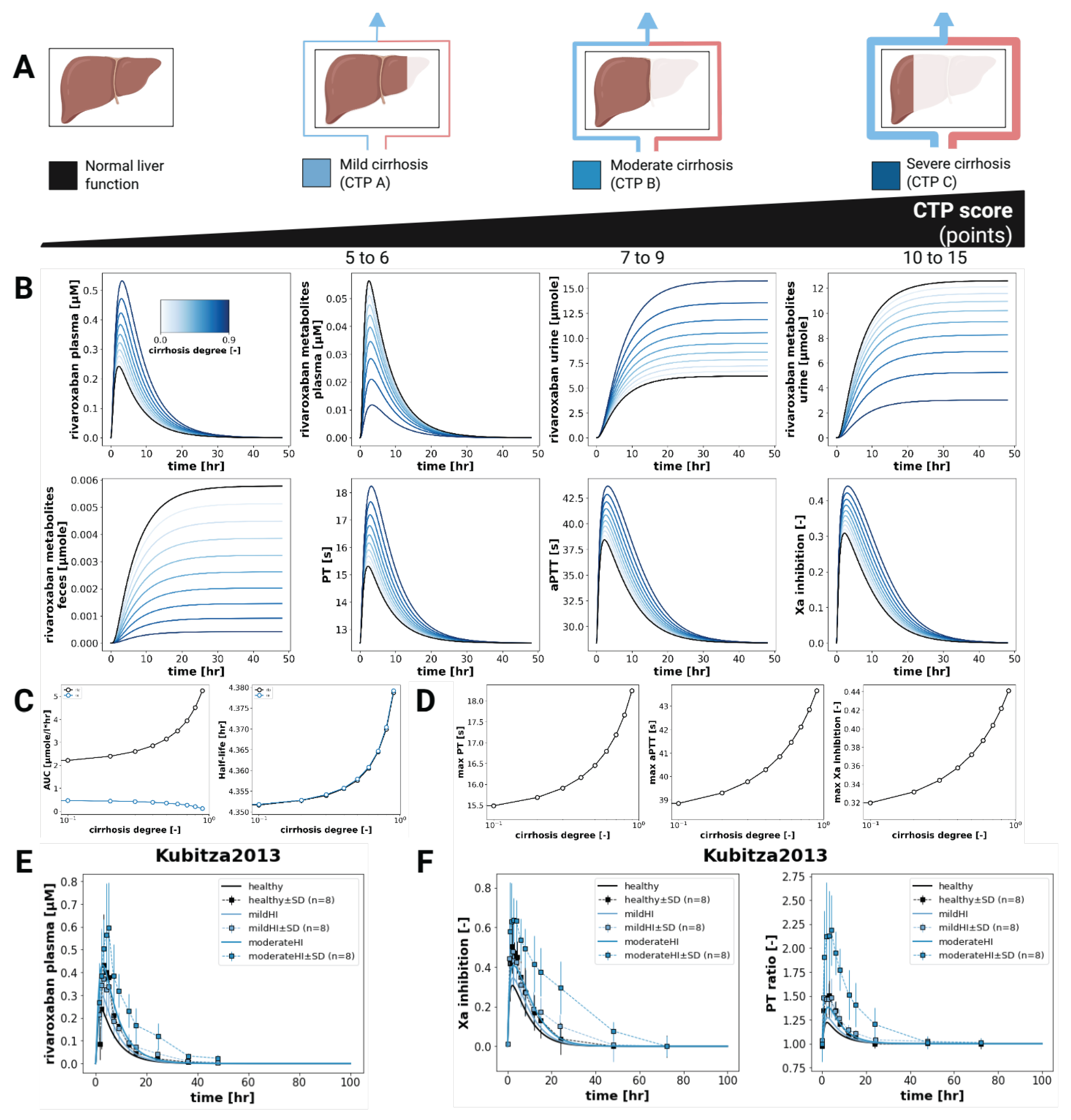
3.5. Hepatic Impairment
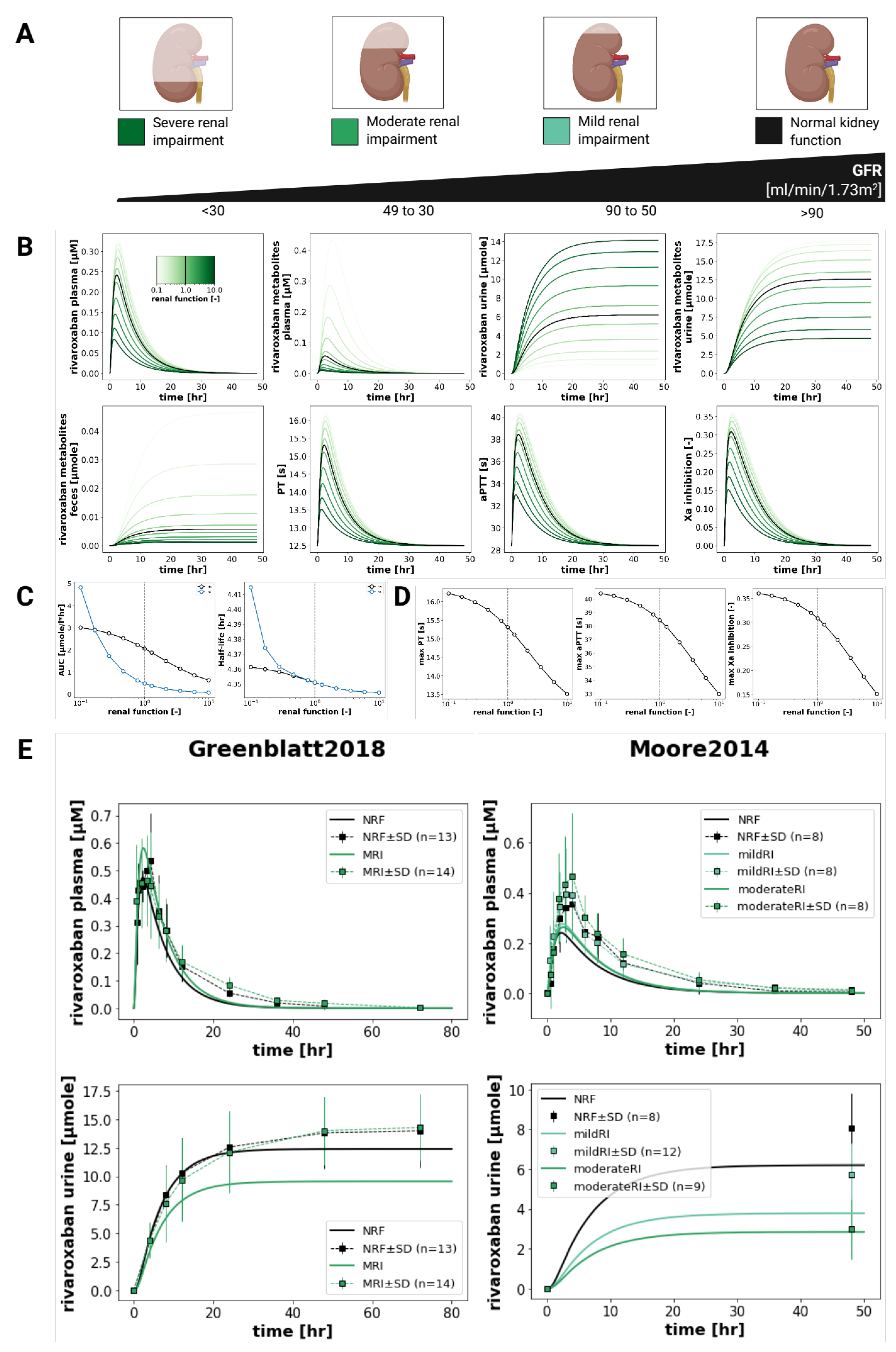
3.6. Renal Impairment
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Samama, M.M. The Mechanism of Action of Rivaroxaban – an Oral, Direct Factor Xa Inhibitor – Compared with Other Anticoagulants. Thrombosis Research 2011, 127, 497–504. [Google Scholar] [CrossRef]
- Orfeo, T.; Butenas, S.; Brummel-Ziedins, K.E.; Gissel, M.; Mann, K.G. Anticoagulation by Factor Xa Inhibitors. Journal of thrombosis and haemostasis : JTH 2010, 8, 1745–1753. [Google Scholar] [CrossRef]
- Kvasnicka, T.; Malikova, I.; Zenahlikova, Z.; Kettnerova, K.; Brzezkova, R.; Zima, T.; Ulrych, J.; Briza, J.; Netuka, I.; Kvasnicka, J. Rivaroxaban - Metabolism, Pharmacologic Properties and Drug Interactions. Current Drug Metabolism 2017, 18, 636–642. [Google Scholar] [CrossRef]
- Tittl, L.; Marten, S.; Naue, C.; Beyer-Westendorf, J. 5-Year Outcomes from Rivaroxaban Therapy in Atrial Fibrillation: Results from the Dresden NOAC Registry. Thrombosis Research 2021, 202, 24–30. [Google Scholar] [CrossRef]
- Ding, S.; Wang, L.; Xie, L.; Zhou, S.; Chen, J.; Zhao, Y.; Deng, W.; Liu, Y.; Zhang, H.; Shao, F. Bioequivalence Study of 2 Formulations of Rivaroxaban, a Narrow-Therapeutic-Index Drug, in Healthy Chinese Subjects Under Fasting and Fed Conditions. Clinical pharmacology in drug development 2020, 9, 346–352. [Google Scholar] [CrossRef] [PubMed]
- Kubitza, D.; Becka, M.; Zuehlsdorf, M.; Mueck, W. Effect of Food, an Antacid, and the H2 Antagonist Ranitidine on the Absorption of BAY 59-7939 (Rivaroxaban), an Oral, Direct Factor Xa Inhibitor, in Healthy Subjects. Journal of clinical pharmacology 2006, 46, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Xie, Q.; Zhao, X.; Tan, Y.; Wang, W.; Cao, Y.; Wei, X.; Mu, G.; Zhang, H.; Zhou, S.; et al. The Pharmacogenetic Variability Associated with the Pharmacokinetics and Pharmacodynamics of Rivaroxaban in Healthy Chinese Subjects: A National Multicenter Exploratory Study. Clinical therapeutics 2024, 46, 313–321. [Google Scholar] [CrossRef] [PubMed]
- Konicki, R.; Weiner, D.; Herbert Patterson, J.; Gonzalez, D.; Kashuba, A.; Cao, Y.C.; Gehi, A.K.; Watkins, P.; Powell, J.R. Rivaroxaban Precision Dosing Strategy for Real-World Atrial Fibrillation Patients. Clinical and translational science 2020, 13, 777–784. [Google Scholar] [CrossRef]
- Pereira Portela, C.; Gautier, L.A.; Zermatten, M.G.; Fraga, M.; Moradpour, D.; Bertaggia Calderara, D.; Aliotta, A.; Veuthey, L.; De Gottardi, A.; Stirnimann, G.; et al. Direct Oral Anticoagulants in Cirrhosis: Rationale and Current Evidence. JHEP Reports 2024, 6, 101116. [Google Scholar] [CrossRef]
- Suchman, A.L.; Griner, P.F. Diagnostic Uses of the Activated Partial Thromboplastin Time and Prothrombin Time. Annals of Internal Medicine 1986, 104, 810–816. [Google Scholar] [CrossRef]
- Kubitza, D.; Becka, M.; Voith, B.; Zuehlsdorf, M.; Wensing, G. Safety, Pharmacodynamics, and Pharmacokinetics of Single Doses of BAY 59-7939, an Oral, Direct Factor Xa Inhibitor. Clinical pharmacology and therapeutics 2005, 78, 412–421. [Google Scholar] [CrossRef]
- Jiang, J.; Hu, Y.; Zhang, J.; Yang, J.; Mueck, W.; Kubitza, D.; Bauer, R.J.; Meng, L.; Hu, P. Safety, Pharmacokinetics and Pharmacodynamics of Single Doses of Rivaroxaban - an Oral, Direct Factor Xa Inhibitor - in Elderly Chinese Subjects. Thrombosis and haemostasis 2010, 103, 234–241. [Google Scholar] [CrossRef]
- Zhao, X.; Sun, P.; Zhou, Y.; Liu, Y.; Zhang, H.; Mueck, W.; Kubitza, D.; Bauer, R.J.; Zhang, H.; Cui, Y. Safety, Pharmacokinetics and Pharmacodynamics of Single/Multiple Doses of the Oral, Direct Factor Xa Inhibitor Rivaroxaban in Healthy Chinese Subjects. British journal of clinical pharmacology 2009, 68, 77–88. [Google Scholar] [CrossRef]
- Graff, J.; von Hentig, N.; Misselwitz, F.; Kubitza, D.; Becka, M.; Breddin, H.K.; Harder, S. Effects of the Oral, Direct Factor Xa Inhibitor Rivaroxaban on Platelet-Induced Thrombin Generation and Prothrombinase Activity. Journal of clinical pharmacology 2007, 47, 1398–1407. [Google Scholar] [CrossRef]
- Mueck, W.; Stampfuss, J.; Kubitza, D.; Becka, M. Clinical Pharmacokinetic and Pharmacodynamic Profile of Rivaroxaban. Clinical Pharmacokinetics 2014, 53, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Kubitza, D.; Roth, A.; Becka, M.; Alatrach, A.; Halabi, A.; Hinrichsen, H.; Mueck, W. Effect of Hepatic Impairment on the Pharmacokinetics and Pharmacodynamics of a Single Dose of Rivaroxaban, an Oral, Direct Factor Xa Inhibitor. British journal of clinical pharmacology 2013, 76, 89–98. [Google Scholar] [CrossRef]
- Moore, K.T.; Vaidyanathan, S.; Natarajan, J.; Ariyawansa, J.; Haskell, L.; Turner, K.C. An Open-Label Study to Estimate the Effect of Steady-State Erythromycin on the Pharmacokinetics, Pharmacodynamics, and Safety of a Single Dose of Rivaroxaban in Subjects with Renal Impairment and Normal Renal Function. Journal of clinical pharmacology 2014, 54, 1407–1420. [Google Scholar] [CrossRef]
- Greenblatt, D.J.; Patel, M.; Harmatz, J.S.; Nicholson, W.T.; Rubino, C.M.; Chow, C.R. Impaired Rivaroxaban Clearance in Mild Renal Insufficiency With Verapamil Coadministration: Potential Implications for Bleeding Risk and Dose Selection. Journal of clinical pharmacology 2018, 58, 533–540. [Google Scholar] [CrossRef] [PubMed]
- Ashton, V.; Kerolus-Georgi, S.; Moore, K.T. The Pharmacology, Efficacy, and Safety of Rivaroxaban in Renally Impaired Patient Populations. Journal of clinical pharmacology 2021, 61, 1010–1026. [Google Scholar] [CrossRef] [PubMed]
- Mueck, W.; Kubitza, D.; Becka, M. Co-Administration of Rivaroxaban with Drugs That Share Its Elimination Pathways: Pharmacokinetic Effects in Healthy Subjects. British journal of clinical pharmacology 2013, 76, 455–466. [Google Scholar] [CrossRef]
- Grzegorzewski, J.; Brandhorst, J.; Green, K.; Eleftheriadou, D.; Duport, Y.; Barthorscht, F.; Köller, A.; Ke, D.Y.J.; De Angelis, S.; König, M. PK-DB: Pharmacokinetics Database for Individualized and Stratified Computational Modeling. Nucleic Acids Research 2021, 49, D1358–D1364. [Google Scholar] [CrossRef] [PubMed]
- Rohatgi, A. WebPlotDigitizer, 2024.
- König, M. Sbmlutils: Python Utilities for SBML. Zenodo, 2024. [CrossRef]
- König, M. Sbmlsim: SBML Simulation Made Easy. [object Object], 2021. [CrossRef]
- Welsh, C.; Xu, J.; Smith, L.; König, M.; Choi, K.; Sauro, H.M. libRoadRunner 2.0: A High Performance SBML Simulation and Analysis Library. Bioinformatics 2023, 39, btac770. [Google Scholar] [CrossRef]
- Somogyi, E.T.; Bouteiller, J.M.; Glazier, J.A.; König, M.; Medley, J.K.; Swat, M.H.; Sauro, H.M. libRoadRunner: A High Performance SBML Simulation and Analysis Library. Bioinformatics 2015, 31, 3315–3321. [Google Scholar] [CrossRef]
- König, M.; Dräger, A.; Holzhütter, H.G. CySBML: A Cytoscape Plugin for SBML. Bioinformatics 2012, 28, 2402–2403. [Google Scholar] [CrossRef]
- Köller, A.; Grzegorzewski, J.; König, M. Physiologically Based Modeling of the Effect of Physiological and Anthropometric Variability on Indocyanine Green Based Liver Function Tests. Frontiers in Physiology 2021, 12, 757293. [Google Scholar] [CrossRef]
- Stevens, P.E.; Ahmed, S.B.; Carrero, J.J.; Foster, B.; Francis, A.; Hall, R.K.; Herrington, W.G.; Hill, G.; Inker, L.A.; Kazancıoğlu, R.; et al. KDIGO 2024 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease. Kidney International 2024, 105, S117–S314. [Google Scholar] [CrossRef]
- Alalawneh, M.; Awaisu, A.; Abdallah, I.; Elewa, H.; Danjuma, M.; Matar, K.M.; ElKashlan, A.M.; Elshayep, Y.; Ibrahim, F.; Rachid, O. Pharmacokinetics of Single-Dose Rivaroxaban under Fed State in Obese vs. Non-Obese Subjects: An Open-Label Controlled Clinical Trial (RIVOBESE-PK). Clinical and translational science 2024, 17, e13853. [Google Scholar] [CrossRef]
- Genis-Najera, L.; Sañudo-Maury, M.E.; Moquete, T. A Single-blind, Randomized, Single-dose, Two-sequence, Two-period, Crossover Study to Assess the Bioequivalence between Two Oral Tablet Formulations of Rivaroxaban 20 Mg in Healthy Mexican Volunteers. Clinical pharmacology in drug development 2022, 11, 826–831. [Google Scholar] [CrossRef] [PubMed]
- Kubitza, D.; Becka, M.; Wensing, G.; Voith, B.; Zuehlsdorf, M. Safety, Pharmacodynamics, and Pharmacokinetics of BAY 59-7939–an Oral, Direct Factor Xa Inhibitor–after Multiple Dosing in Healthy Male Subjects. European Journal of Clinical Pharmacology 2005, 61, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Weinz, C.; Schwarz, T.; Kubitza, D.; Mueck, W.; Lang, D. Metabolism and Excretion of Rivaroxaban, an Oral, Direct Factor Xa Inhibitor, in Rats, Dogs, and Humans. Drug metabolism and disposition: the biological fate of chemicals 2009, 37, 1056–1064. [Google Scholar] [CrossRef]
- Kubitza, D.; Becka, M.; Roth, A.; Mueck, W. Dose-Escalation Study of the Pharmacokinetics and Pharmacodynamics of Rivaroxaban in Healthy Elderly Subjects. Current medical research and opinion 2008, 24, 2757–2765. [Google Scholar] [CrossRef] [PubMed]
- Speed, V.; Green, B.; Roberts, L.N.; Woolcombe, S.; Bartoli-Abdou, J.; Barsam, S.; Byrne, R.; Gee, E.; Czuprynska, J.; Brown, A.; et al. Fixed Dose Rivaroxaban Can Be Used in Extremes of Bodyweight: A Population Pharmacokinetic Analysis. Journal of thrombosis and haemostasis : JTH 2020, 18, 2296–2307. [Google Scholar] [CrossRef] [PubMed]
- Remko, M. Molecular Structure, Lipophilicity, Solubility, Absorption, and Polar Surface Area of Novel Anticoagulant Agents. Journal of Molecular Structure: THEOCHEM 2009, 916, 76–85. [Google Scholar] [CrossRef]
- Casabianca, E.; König, M. Physiologically Based Pharmacokinetic/Pharmacodynamic (PBPK/PD) Model of Rivaroxaban. Zenodo, 2025. [CrossRef]
| Study | PK-DB | Dosing | Dose [mg] | H | RI | HI | FA | FE | RP | RU | RF | Xa | PT | aP | S |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alalawneh2024 | PKDB00983 | single | 20 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [30] | |||||
| Ding2020 | PKDB00972 | single | 10 | ✓ | ✓ | ✓ | ✓ | [5] | |||||||
| GenisNajera2022 | PKDB00973 | single | 20 | ✓ | ✓ | ✓ | [31] | ||||||||
| Graff2007 | PKDB00984 | single | 5, 30 | ✓ | ✓ | ✓ | ✓ | ✓ | [14] | ||||||
| Greenblatt2018 | PKDB00985 | single | 20 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [18] | ||||
| Jiang2010 | PKDB00986 | single | 5, 10, 20, 30, 40 | ✓ | ✓ | ✓ | ✓ | ✓ | [12] | ||||||
| Kubitza2005 | PKDB00987 | multi | 5, 10, 20, 30 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [32] | |||||
| Kubitza2005a | PKDB00988 | single, multi | 1.25, 5, 10, 20, 40, 80 | ✓ | ✓ | ✓ | ✓ | ✓ | [11] | ||||||
| Kubitza2006 | PKDB00989 | single, multi | 5, 20 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [6] | |||||
| Kubitza2013 | PKDB00990 | single | 10 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [16] | |||||
| Liu2024 | PKDB00991 | single | 10, 15, 20 | ✓ | ✓ | ✓ | ✓ | ✓ | [7] | ||||||
| Moore2014 | PKDB00992 | single | 10 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [17] | |||||
| Weinz2009 | PKDB00993 | single | 10 | ✓ | ✓ | ✓ | ✓ | ✓ | [33] | ||||||
| Zhao2009 | PKDB00994 | single, multi | 2.5, 5, 10, 20, 40 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | [13] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




