Submitted:
22 July 2025
Posted:
23 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Neural tissUE ENGINEERing and the iN VITRO GAP
1.2. Role of Composite Materials and AI
2. Bioinspiration in Neural Architecture
2.1. Hierarchical Brain Structures & Functional Cues
2.2. Biomimetic Principles for Regenerative Neuroscience
3. Advanced Composites & 4D Bioprinting for NeuroPods
3.1. Smart Nanocomposites & Hybrid Bioinks
3.2. Stimuli-Responsive & Shape-Memory Approaches
3.3. Fabrication Methodologies & Programming
4. AI-Driven Modeling and Digital Twins
4.1. Predictive Multiscale Modeling
4.2. Continuous Optimization and Learning
4.3. Digital Twins and Real-Time Adaptation
5. Integration into the Brain-on-Chip Platforms
5.1. Microfluidics & Organ-Level Synergy
5.2. Real-Time Sensing, Actuation & Drug Testing
6. Key Challenges & Future Directions
6.1. Scalability & Manufacturing Bottlenecks
6.2. Long-Term Stability & Translation
6.3. Visionary Concepts
7. Conclusions
8. Unresolved Questions
Author Information
Funding
Ethics approval and consent to participate
Competing interests
Availability of data and material
Consent for publication
Declaration Regarding the Use of AI-Assisted Readability Enhancement
References
- Fu, J. , & Tan, W. Stochastic reconstruction of multiphase composite microstructures using statistics-encoded neural network for poro/micromechanical modeling. Computer Methods in Applied Mechanics and Engineering 2025, 441, 117986. [Google Scholar] [CrossRef]
- Yan, Y. , Li, X., Gao, Y., Mathivanan, S., Kong, L., Tao, Y.,... & Zhang, S.C. 3D bioprinting of human neural tissues with functional connectivity. Cell Stem Cell 2024, 31, 260–274. [Google Scholar] [PubMed]
- Wu, X. , Shi, W., Liu, X., & Gu, Z. Recent advances in 3D-printing-based organ-on-a-chip. EngMedicine 2024, 1, 100003. [Google Scholar] [CrossRef]
- Kennedy, S.M. , M, S., A, V., & K, A. 4D bioprinting for personalized medicine, innovations in implant fabrication and regenerative therapies. Polymer-Plastics Technology and Materials 2025, 1–26. [Google Scholar] [CrossRef]
- Salmina, A.B. , Alexandrova, O.P., Averchuk, A.S., Korsakova, S.A., Saridis, M.R., Illarioshkin, S.N., & Yurchenko, S.O. Current progress and challenges in the development of brain tissue models: How to grow up the changeable brain in vitro? Journal of Tissue Engineering 2024, 15, 20417314241235527. [Google Scholar]
- Han, S. , Zhao, X., Cheng, L., & Fan, J. Recent progresses in neural tissue engineering using topographic scaffolds. American Journal of Stem Cells 2024, 13, 1. [Google Scholar] [PubMed]
- Arif, Z.U. , Khalid, M.Y., Ahmed, W., & Arshad, H. A review on four-dimensional (4D) bioprinting in pursuit of advanced tissue engineering applications. Bioprinting 2022, 27, e00203. [Google Scholar] [CrossRef]
- Papetti, A.V. , Jin, M., Ma, Z., Stillitano, A.C., & Jiang, P. (2025). Chimeric brain models: Unlocking insights into human neural development, aging, diseases, and cell therapies. Neuron.
- Oncu, E. , Ayanoglu, K.Y.U., & Ciftci, F. Comparative analysis of deep learning models for predicting biocompatibility in tissue scaffold images. Computers in Biology and Medicine 2025, 192, 110281. [Google Scholar] [CrossRef]
- Papamichail, L. , Koch, L.S., Veerman, D., Broersen, K., & van der Meer, A.D. Organoids-on-a-chip: microfluidic technology enables culture of organoids with enhanced tissue function and potential for disease modeling. Frontiers in bioengineering and biotechnology 2025, 13, 1515340. [Google Scholar] [CrossRef]
- Aili, Y. , Maimaitiming, N., Wang, Z., & Wang, Y. Brain organoids: A new tool for modeling of neurodevelopmental disorders. Journal of Cellular and Molecular Medicine 2024, 28, e18560. [Google Scholar] [CrossRef]
- Urrestizala-Arenaza, N. , Cerchio, S., Cavaliere, F., & Magliaro, C. Limitations of human brain organoids to study neurodegenerative diseases: a manual to survive. Frontiers in Cellular Neuroscience 2024, 18, 1419526. [Google Scholar] [CrossRef]
- Mu, Y. , Jin, Y., Ji, H., Luo, J., Li, G., Xu, M.,... & Du, J. Mechanical performance of interpenetrating phase composites with multisheet lattice structures. International Journal of Mechanical Sciences 2024, 276, 109369. [Google Scholar] [CrossRef]
- Parvin, N. , Joo, S.W., Jung, J.H., & Mandal, T.K. Multimodal AI in Biomedicine: Pioneering the Future of Biomaterials, Diagnostics, and Personalized Healthcare. Nanomaterials 2025, 15, 895. [Google Scholar] [CrossRef] [PubMed]
- Orr, A. , Kalantarnia, F., Nazir, S., Bolandi, B., Alderson, D., O’Grady, K.,... & Willerth, S.M. Recent advances in 3D bioprinted neural models: A systematic review on the applications to drug discovery. Advanced Drug Delivery Reviews 2025, 115524. [Google Scholar] [CrossRef]
- Lai, J. , Liu, Y., Lu, G., Yung, P., Wang, X., Tuan, R.S., & Li, Z.A. 4D bioprinting of programmed dynamic tissues. Bioactive Materials 2024, 37, 348–377. [Google Scholar] [CrossRef] [PubMed]
- Xie, C. , Chen, Y., Wang, L., Liao, K., Xue, B., Han, Y.,... & Jiang, Q. Recent research of peptide-based hydrogel in nervous regeneration. Bioactive Materials 2024, 40, 503–523. [Google Scholar] [CrossRef]
- Ferreira, M.J. , Colombani, S., Bernardin, A., Lacampagne, A., Pasquié, J.L., Costa, P.F.,... & Meli, A.C. Advancing organ-on-chip systems: the role of microfluidics in neuro-cardiac research. Current Research in Pharmacology and Drug Discovery 2025, 100227. [Google Scholar] [CrossRef]
- Moreau, S. , Flores-Berdines, R., Jalkh, T.E., Simon, A., Taret, G., Fomina, A.,... & Salmon, H. Reversible and reusable compartmentalized microfluidic chip for coculture of dorsal root ganglion neurons. bioRxiv.
- Kahveci, B. , Polatli, E., Evranos, A.E., Güner, H., Bastanlar, Y., Karakülah, G., & Güven, S. BrAIn: A comprehensive artificial intelligence-based morphology analysis system for brain organoids and neuroscience. bioRxiv 2025, 2025–02. [Google Scholar]
- Akcay, G. , & Luttge, R. Microenvironments matter: advances in brain-on-chip. Biosensors 2023, 13, 551. [Google Scholar] [CrossRef]
- da Silva, G.G. , Sacomani, D.P., de Carvalho, B.G., Porcionatto, M.A., Gobbi, A., Lima, R.S., & de la Torre, L.G. Microfluidic Systems to Mimic the Blood–Brain Barrier: from Market to Engineering Challenges and Perspectives. ACS Biomaterials Science & Engineering 2025. [Google Scholar]
- Pei, N. , Hao, Z., Wang, S., Pan, B., Fang, A., Kang, J.,... & Wang, L. 3D printing of layered gradient pore structure of brain-like tissue. International Journal of Bioprinting 2021, 7, 359. [Google Scholar] [CrossRef]
- Rybachuk, O. , Nesterenko, Y., & Zhovannyk, V. Modern advances in spinal cord regeneration: hydrogel combined with neural stem cells. Frontiers in Pharmacology 2024, 15, 1419797. [Google Scholar] [CrossRef] [PubMed]
- Parvin, N. , Joo, S.W., Jung, J.H., & Mandal, T.K. Multimodal AI in Biomedicine: Pioneering the Future of Biomaterials, Diagnostics, and Personalized Healthcare. Nanomaterials 2025, 15, 895. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y. , Li, X., Gao, Y., Mathivanan, S., Kong, L., Tao, Y.,... & Zhang, S.C. 3D bioprinting of human neural tissues with functional connectivity. Cell Stem Cell 2024, 31, 260–274. [Google Scholar] [CrossRef]
- Castiglione, H. , Vigneron, P.A., Baquerre, C., Yates, F., Rontard, J., & Honegger, T. Human brain organoids-on-chip: advances, challenges, and perspectives for preclinical applications. Pharmaceutics 2022, 14, 2301. [Google Scholar]
- Carvalho, V. , Rodrigues, R.O., Shin, S.R., Lima, R., & Teixeira, S.F. Advancing blood–brain barrier-on-a-chip models through numerical simulations. BioChip Journal 2024, 18, 546–565. [Google Scholar] [CrossRef]
- Amartumur, S. , Nguyen, H., Huynh, T., Kim, T.S., Woo, R.S., Oh, E.,... & Heo, C. Neuropathogenesis-on-chips for neurodegenerative diseases. Nature Communications 2024, 15, 2219. [Google Scholar] [CrossRef] [PubMed]
- Boufidis, D. , Garg, R., Angelopoulos, E., Cullen, D.K., & Vitale, F. Bioinspired electronics: Soft, biohybrid, and “living” neural interfaces. Nature Communications 2025, 16, 1861. [Google Scholar]
- De Vitis, E.; La Pesa, V.; Gervaso, F.; Romano, A.; Quattrini, A.; Gigli, G.; Moroni, L.; Polini, A. A microfabricated multi-compartment device for neuron and Schwann cell differentiation. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhou, X.; Fang, Y.; Xiong, Z.; Zhang, T. AI-driven 3D bioprinting for regenerative medicine: From bench to bedside. Bioact. Mater. 2025, 45, 201–230. [Google Scholar] [CrossRef]
- Lai, J.; Liu, Y.; Lu, G.; Yung, P.; Wang, X.; Tuan, R.S.; Li, Z.A. 4D bioprinting of programmed dynamic tissues. Bioact. Mater. 2024, 37, 348–377. [Google Scholar] [CrossRef]
- You, D.; Lin, L.; Dong, M.; Wu, Y.; Hu, Y.; Hu, X.; Shao, Y.; Xie, Y.; Xu, M.; Chen, G.; et al. Recent Advances in Shape Memory Polymers for Biomedical Applications: Bridging Macro- and Micro-Scale Effects. Smart Mater. Med. 2025. [Google Scholar] [CrossRef]
- Aizarna-Lopetegui, U.; Bittinger, S.C.; Álvarez, N.; Henriksen-Lacey, M.; de Aberasturi, D.J. Stimuli-responsive hybrid materials for 4D in vitro tissue models. Mater. Today Bio 2025, 33, 102035. [Google Scholar] [CrossRef] [PubMed]
- Krishna, D.V.; Sankar, M.R. Machine learning-assisted extrusion-based 3D bioprinting for tissue regeneration applications. Ann. 3D Print. Med. 2023, 12, 100132. [Google Scholar] [CrossRef]
- Lai, J. , Chen, X. , Lu, H.H., & Wang, M. 3D bioprinting of graphene oxide-incorporated hydrogels for neural tissue regeneration. 3D Printing and Additive Manufacturing 2024, 11, e2022–e2032. [Google Scholar] [PubMed]
- Ye, L.; Ji, H.; Liu, J.; Tu, C.; Kappl, M.; Koynov, K.; Vogt, J.; Butt, H. Carbon Nanotube–Hydrogel Composites Facilitate Neuronal Differentiation While Maintaining Homeostasis of Network Activity. Adv. Mater. 2021, 33, 2102981. [Google Scholar] [CrossRef]
- Shin, J.; Kang, R.; Hyun, K.; Li, Z.; Kumar, H.; Kim, K.; Park, S.S.; Kim, K. Machine Learning-Enhanced Optimization for High-Throughput Precision in Cellular Droplet Bioprinting. Adv. Sci. 2025, 12, e2412831. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wallmersperger, T.; Ehrenhofer, A. Prediction of hydrogel swelling states using machine learning methods. Eng. Rep. 2024, 6, e12893. [Google Scholar] [CrossRef]
- Esworthy, T.J.; Miao, S.; Lee, S.-J.; Zhou, X.; Cui, H.; Zuo, Y.Y.; Zhang, L.G. Advanced 4D-bioprinting technologies for brain tissue modeling and study. Int. J. Smart Nano Mater. 2019, 10, 177–204. [Google Scholar] [CrossRef]
- Freeman, S.; Calabro, S.; Williams, R.; Jin, S.; Ye, K. Bioink Formulation and Machine Learning-Empowered Bioprinting Optimization. Front. Bioeng. Biotechnol. 2022, 10, 913579. [Google Scholar] [CrossRef]
- Mofrad, Y.M.; Shamloo, A. The effect of conductive aligned fibers in an injectable hydrogel on nerve tissue regeneration. Int. J. Pharm. 2023, 645, 123419. [Google Scholar] [CrossRef]
- Yeh, Y.-C.; Chen, P.-Y.; Chen, K.-T.; Lee, I.-C. Innovative MXene/SilMA-Based Conductive Bioink for Three Dimensional Bioprinting of Neural Stem Cell Spheroids in Neural Tissue Engineering. ACS Appl. Mater. Interfaces 2025, 17, 10402–10416. [Google Scholar] [CrossRef] [PubMed]
- Jamshidi, M.B. , Hoang, D. T., Nguyen, D.N., Niyato, D., & Warkiani, M.E. Revolutionizing biological digital twins: Integrating internet of bionano things, convolutional neural networks, and federated learning. Computers in Biology and Medicine 2025, 189, 109970. [Google Scholar]
- Gangwal, A.; Lavecchia, A. Artificial intelligence in preclinical research: enhancing digital twins and organ-on-chip to reduce animal testing. Drug Discov. Today 2025, 30, 104360. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, N.; Narayanan, L.K.; Shirwaiker, R. A framework for digital twin integration in biofabrication and a scaffold 3D bioplotting case study. Manuf. Lett. 2024, 41, 1182–1191. [Google Scholar] [CrossRef]
- Wang, S.; Wang, X.; Holland, M.A. Multi-physics modeling and finite-element formulation of neuronal dendrite growth with electrical polarization. Brain Multiphysics 2023, 4, 100071. [Google Scholar] [CrossRef]
- Zheng, H. , Xie, W., Wang, K., & Li, Z. Opportunities of Hybrid Model-based Reinforcement Learning for Cell Therapy Manufacturing Process Control. arXiv 2022, arXiv:2201.03116. [Google Scholar]
- Xiong, H.; Chu, C.; Fan, L.; Song, M.; Zhang, J.; Ma, Y.; Zheng, R.; Zhang, J.; Yang, Z.; Jiang, T. The Digital Twin Brain: A Bridge between Biological and Artificial Intelligence. Intell. Comput. 2023, 2. [Google Scholar] [CrossRef]
- Zahedmanesh, H.; Lally, C. A multiscale mechanobiological modelling framework using agent-based models and finite element analysis: application to vascular tissue engineering. Biomech. Model. Mechanobiol. 2011, 11, 363–377. [Google Scholar] [CrossRef]
- Ramesh, S.; Deep, A.; Tamayol, A.; Kamaraj, A.; Mahajan, C.; Madihally, S. Advancing 3D bioprinting through machine learning and artificial intelligence. Bioprinting 2024, 38, e00331. [Google Scholar] [CrossRef]
- Treloar, N.J.; Fedorec, A.J.H.; Ingalls, B.; Barnes, C.P.; You, L. Deep reinforcement learning for the control of microbial co-cultures in bioreactors. PLOS Comput. Biol. 2020, 16, e1007783. [Google Scholar] [CrossRef]
- Xiong, H.; Chu, C.; Fan, L.; Song, M.; Zhang, J.; Ma, Y.; Zheng, R.; Zhang, J.; Yang, Z.; Jiang, T. The Digital Twin Brain: A Bridge between Biological and Artificial Intelligence. Intell. Comput. 2023, 2, 0055. [Google Scholar] [CrossRef]
- Fekonja, L.S.; Schenk, R.; Schröder, E.; Tomasello, R.; Tomšič, S.; Picht, T. The digital twin in neuroscience: from theory to tailored therapy. Front. Neurosci. 2024, 18, 1454856. [Google Scholar] [CrossRef]
- An, J.; Chua, C.K.; Mironov, V. Application of Machine Learning in 3D Bioprinting: Focus on Development of Big Data and Digital Twin. Int. J. Bioprinting 2021, 7, 342. [Google Scholar] [CrossRef]
- E Wang, H.; Triebkorn, P.; Breyton, M.; Dollomaja, B.; Lemarechal, J.-D.; Petkoski, S.; Sorrentino, P.; Depannemaecker, D.; Hashemi, M.; Jirsa, V.K. Virtual brain twins: from basic neuroscience to clinical use. Natl. Sci. Rev. 2024, 11, nwae079. [Google Scholar] [CrossRef]
- Malashin, I.; Masich, I.; Tynchenko, V.; Gantimurov, A.; Nelyub, V.; Borodulin, A.; Martysyuk, D.; Galinovsky, A. Machine Learning in 3D and 4D Printing of Polymer Composites: A Review. Polymers 2024, 16, 3125. [Google Scholar] [CrossRef] [PubMed]
- Akbarialiabad, H.; Seyyedi, M.S.; Paydar, S.; Habibzadeh, A.; Haghighi, A.; Kvedar, J.C. Bridging silicon and carbon worlds with digital twins and on-chip systems in drug discovery. npj Syst. Biol. Appl. 2024, 10, 150. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.; Pawar, A.; Liao, A.; Anitescu, C.; Webster-Wood, V.; Feinberg, A.W.; Rabczuk, T.; Zhang, Y.J. Modeling neuron growth using isogeometric collocation based phase field method. Sci. Rep. 2022, 12, 8120. [Google Scholar] [CrossRef] [PubMed]
- De Domenico, M.; Allegri, L.; Caldarelli, G.; D’aNdrea, V.; Di Camillo, B.; Rocha, L.M.; Rozum, J.; Sbarbati, R.; Zambelli, F. Challenges and opportunities for digital twins in precision medicine from a complex systems perspective. npj Digit. Med. 2025, 8, 37. [Google Scholar] [CrossRef]
- Park, S.-Y.; Park, C.-H.; Choi, D.-H.; Hong, J.K.; Lee, D.-Y. Bioprocess digital twins of mammalian cell culture for advanced biomanufacturing. Curr. Opin. Chem. Eng. 2021, 33, 100702. [Google Scholar] [CrossRef]
- Wu, Y.; Ding, X.; Wang, Y.; Ouyang, D. Harnessing the power of machine learning into tissue engineering: current progress and future prospects. Burn. Trauma 2024, 12, tkae053. [Google Scholar] [CrossRef]
- Spitz, S. , Schobesberger, S. , Brandauer, K., & Ertl, P. Sensor-integrated brain-on-a-chip platforms: improving the predictive validity in neurodegenerative research. Bioengineering & Translational Medicine 2024, 9, e10604. [Google Scholar]
- Zhang, H.; Huang, N.; Bian, S.; Sawan, M. Brain organoids-on-chip for neural diseases modeling: History, challenges and trends. J. Pharm. Anal. 2025, 101323. [Google Scholar] [CrossRef]
- Qiu, B. , Pompe, S., Xenaki, K.T., van Bergen en Henegouwen, P.M., Oliveira, S., Mastrobattista, E., & Caiazzo, M. Generation of a Three-dimensional Human Neurovascular Unit Model in a Microfluidic Chip. bioRxiv 2025, 2025-04. [Google Scholar]
- Berjaoui, C.; Kachouh, C.; Joumaa, S.; Ghayyad, M.H.; Bekele, B.A.; Ajirenike, R.; Al Maaz, Z.; Awde, S.; Wojtara, M.; Nazir, A.; et al. Neuroinflammation-on-a-chip for multiple sclerosis research: a narrative review. Ann. Med. Surg. 2024, 86, 4053–4059. [Google Scholar] [CrossRef]
- Rodrigues, R.O.; Shin, S.-R.; Bañobre-López, M. Brain-on-a-chip: an emerging platform for studying the nanotechnology-biology interface for neurodegenerative disorders. J. Nanobiotechnology 2024, 22, 573. [Google Scholar] [CrossRef] [PubMed]
- Sakai, K. , Teshima, T. F., Goto, T., Nakashima, H., & Yamaguchi, M. Self-Folding Graphene-Based Interface for Brain-Like Modular 3D Tissue. Advanced Functional Materials 2023, 33, 2301836. [Google Scholar]
- Wang, Z.; Ma, D.; Liu, J.; Xu, S.; Qiu, F.; Hu, L.; Liu, Y.; Ke, C.; Ruan, C. 4D printing polymeric biomaterials for adaptive tissue regeneration. Bioact. Mater. 2025, 48, 370–399. [Google Scholar] [CrossRef]
- Lai, J.; Liu, Y.; Lu, G.; Yung, P.; Wang, X.; Tuan, R.S.; Li, Z.A. 4D bioprinting of programmed dynamic tissues. Bioact. Mater. 2024, 37, 348–377. [Google Scholar] [CrossRef] [PubMed]
- Akbarialiabad, H.; Seyyedi, M.S.; Paydar, S.; Habibzadeh, A.; Haghighi, A.; Kvedar, J.C. Bridging silicon and carbon worlds with digital twins and on-chip systems in drug discovery. npj Syst. Biol. Appl. 2024, 10, 150. [Google Scholar] [CrossRef]
- A Shah, P.; Shrivastav, P.S.; Ghate, M.; Chavda, V. The fusion of microfluidics and artificial intelligence: a novel alliance for medical advancements. Bioanalysis 2024, 16, 927–930. [Google Scholar] [CrossRef]
- Xu, X.; Zuo, Y.; Chen, S.; Hatami, A.; Gu, H. Advancements in Brain Research: The In Vivo/In Vitro Electrochemical Detection of Neurochemicals. Biosensors 2024, 14, 125. [Google Scholar] [CrossRef]
- Yu, S.; Jiang, L.; Song, M.; Yang, T.; Yuan, M.; Chen, X.; Shu, H. Regulation of BBB function and pathological evolution of PD by microenvironment “spatiotemporal gradient”: unique advantages of microfluidic chips. Front. Aging Neurosci. 2025, 17, 1599509. [Google Scholar] [CrossRef]
- Ferreira, M.J.; Colombani, S.; Bernardin, A.; Lacampagne, A.; Pasquié, J.-L.; Costa, P.F.; Charlot, B.; Meli, A.C. Advancing organ-on-chip systems: the role of microfluidics in neuro-cardiac research. Curr. Res. Pharmacol. Drug Discov. 2025, 9, 100227. [Google Scholar] [CrossRef]
- Yan, Y.; Li, X.; Gao, Y.; Mathivanan, S.; Kong, L.; Tao, Y.; Dong, Y.; Li, X.; Bhattacharyya, A.; Zhao, X.; et al. 3D bioprinting of human neural tissues with functional connectivity. Cell Stem Cell 2024, 31, 260–274. [Google Scholar] [CrossRef]
- Kahveci, B. , Polatli, E. bioRxiv 2025, 2025-02. [Google Scholar]
- Dhar, S.; Ahmad, F.; Deshpande, A.; Rana, S.S.; A, T.A.; Priyadarsini, S. 3-Dimensional printing and bioprinting in neurological sciences: applications in surgery, imaging, tissue engineering, and pharmacology and therapeutics. J. Mater. Sci. Mater. Med. 2025, 36, 32. [Google Scholar] [CrossRef]
- Lai, J.; Liu, Y.; Lu, G.; Yung, P.; Wang, X.; Tuan, R.S.; Li, Z.A. 4D bioprinting of programmed dynamic tissues. Bioact. Mater. 2024, 37, 348–377. [Google Scholar] [CrossRef]
- Zhang, Z.; Zhou, X.; Fang, Y.; Xiong, Z.; Zhang, T. AI-driven 3D bioprinting for regenerative medicine: From bench to bedside. Bioact. Mater. 2025, 45, 201–230. [Google Scholar] [CrossRef]
- Li, K.; Huang, W.H.; Guo, H.T.; Liu, Y.Y.; Chen, S.; Liu, H.; Gu, Q. Advancements in robotic arm-based 3D bioprinting for biomedical applications. Life Med. 2023, 2, lnad046. [Google Scholar] [CrossRef] [PubMed]
- Ramesh, S.; Deep, A.; Tamayol, A.; Kamaraj, A.; Mahajan, C.; Madihally, S. Advancing 3D bioprinting through machine learning and artificial intelligence. Bioprinting 2024, 38, e00331. [Google Scholar] [CrossRef]
- Boufidis, D. , Garg, R. , Angelopoulos, E., Cullen, D.K., & Vitale, F. Bioinspired electronics: Soft, biohybrid, and “living” neural interfaces. Nature Communications 2025, 16, 1861. [Google Scholar]
- Ienca, M.; Valle, G.; Raspopovic, S. Clinical trials for implantable neural prostheses: understanding the ethical and technical requirements. Lancet Digit. Heal. 2025, 7, e216–e224. [Google Scholar] [CrossRef]
- Lindner, N.; Mejia-Wille, A.; Fritschen, A.; Blaeser, A. Development of a robotic-assisted handling and manipulation system for the high-scale bioproduction of 3D-bioprinted organ-on-a-chip devices. HardwareX 2024, 19, e00572. [Google Scholar] [CrossRef]
- Fritschen, A. , Lindner, N. , Scholpp, S., Richthof, P., Dietz, J., Linke, P.,... & Blaeser, A. High-Scale 3D-Bioprinting Platform for the Automated Production of Vascularized Organs-on-a-Chip. Advanced Healthcare Materials 2024, 13, 2304028. [Google Scholar]
- Kim, M.H.; Singh, Y.P.; Celik, N.; Yeo, M.; Rizk, E.; Hayes, D.J.; Ozbolat, I.T. High-throughput bioprinting of spheroids for scalable tissue fabrication. Nat. Commun. 2024, 15, 1–21. [Google Scholar] [CrossRef]
- Lv, S.; Xu, Z.; Mo, F.; Wang, Y.; Duan, Y.; Liu, Y.; Jing, L.; Shan, J.; Jia, Q.; Wang, M.; et al. Long-term stability strategies of deep brain flexible neural interface. npj Flex. Electron. 2025, 9, 1–17. [Google Scholar] [CrossRef]
- Pawlak, W.A.; Howard, N. Neuromorphic algorithms for brain implants: a review. Front. Neurosci. 2025, 19, 1570104. [Google Scholar] [CrossRef] [PubMed]
- Barjuei, E.S.; Shin, J.; Kim, K.; Lee, J. Precision improvement of robotic bioprinting via vision-based tool path compensation. Sci. Rep. 2024, 14, 17764. [Google Scholar] [CrossRef] [PubMed]
- Sirbu, R. , Morley, J., Schroder, T., Taddeo, M., Pothukuchi, R.P., Ugur, M.,... & Floridi, L. Regulating next-generation implantable brain-computer interfaces: Recommendations for ethical development and implementation. arXiv 2025, arXiv:2506.12540. [Google Scholar]
- Gao, H.; Shen, H.; Zhang, X.; Liu, Y.; Shang, Y.; Sun, S.; Guan, W.; Gu, X.; Yang, Y.; Li, G. Revolutionizing neural regeneration with smart responsive materials: Current insights and future prospects. Bioact. Mater. 2025, 52, 393–421. [Google Scholar] [CrossRef]
- Groff-Vindman, C.S.; Trump, B.D.; Cummings, C.L.; Smith, M.; Titus, A.J.; Oye, K.; Prado, V.; Turmus, E.; Linkov, I. The convergence of AI and synthetic biology: the looming deluge. npj Biomed. Innov. 2025, 2, 20. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration. (2024, –21). Executive summary for the Digital Health Advisory Committee meeting: Total product lifecycle considerations for generative AI-enabled devices [PDF]. U.S. Food and Drug Administration. https://www.fda. 20 November 1828.
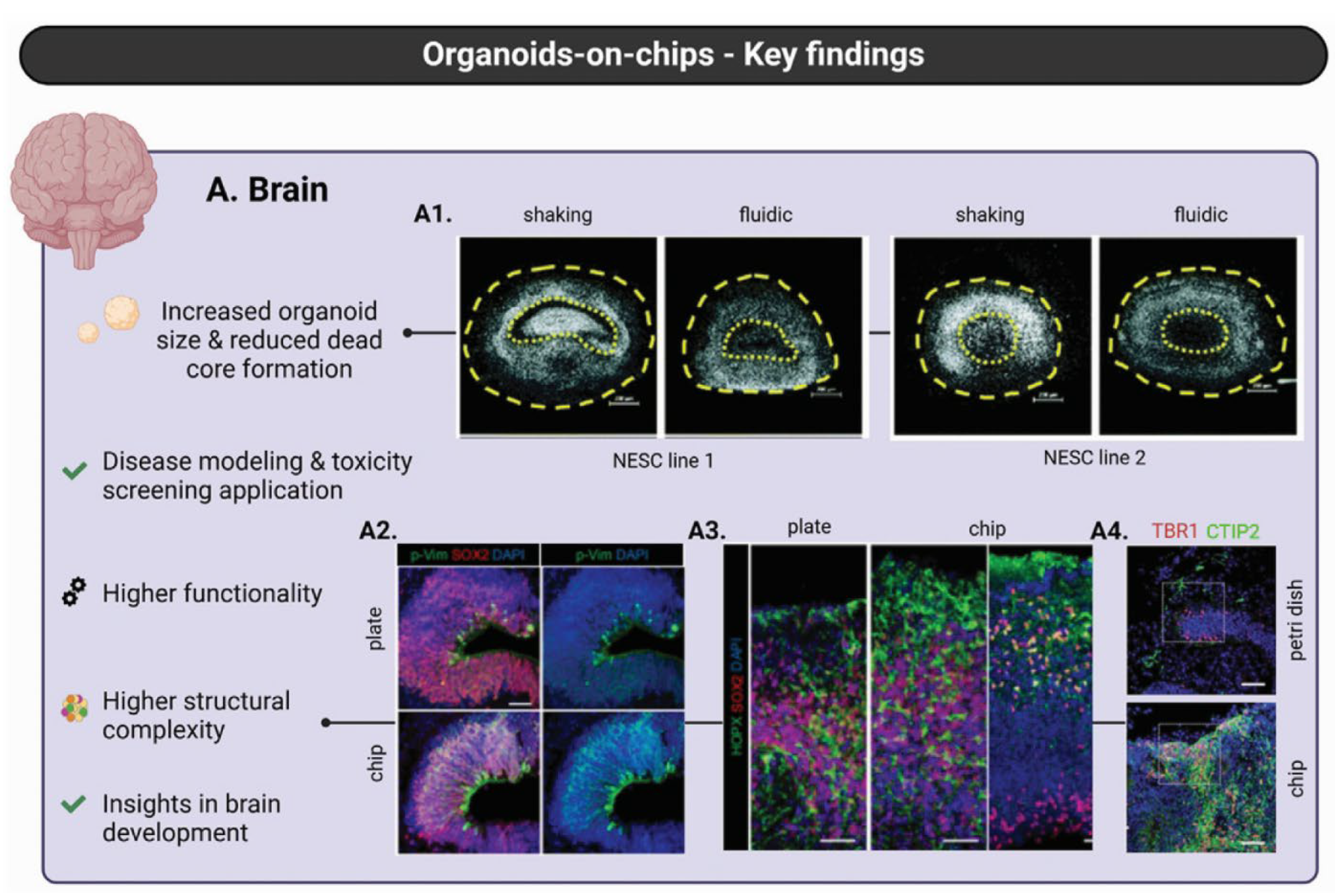
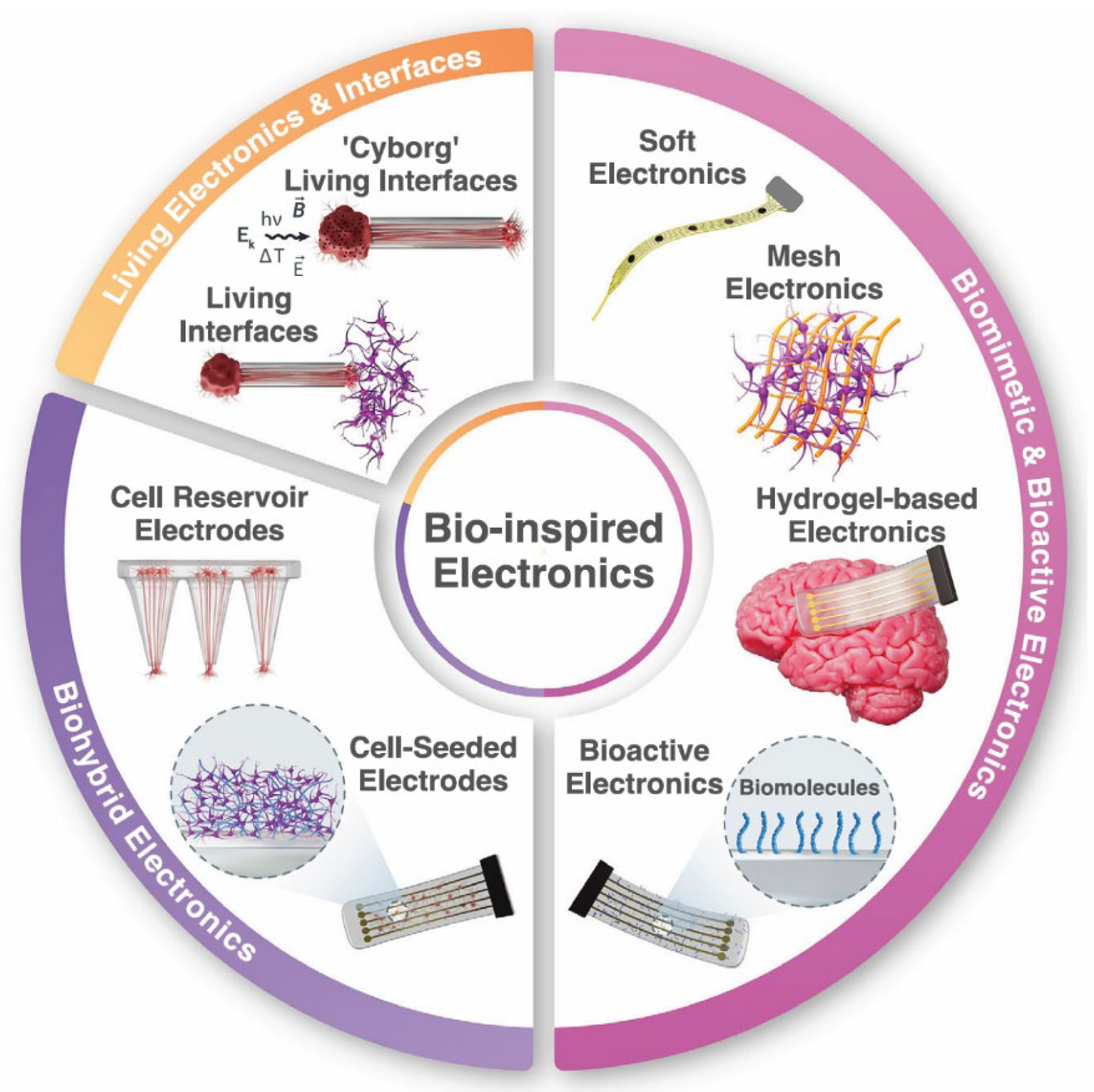
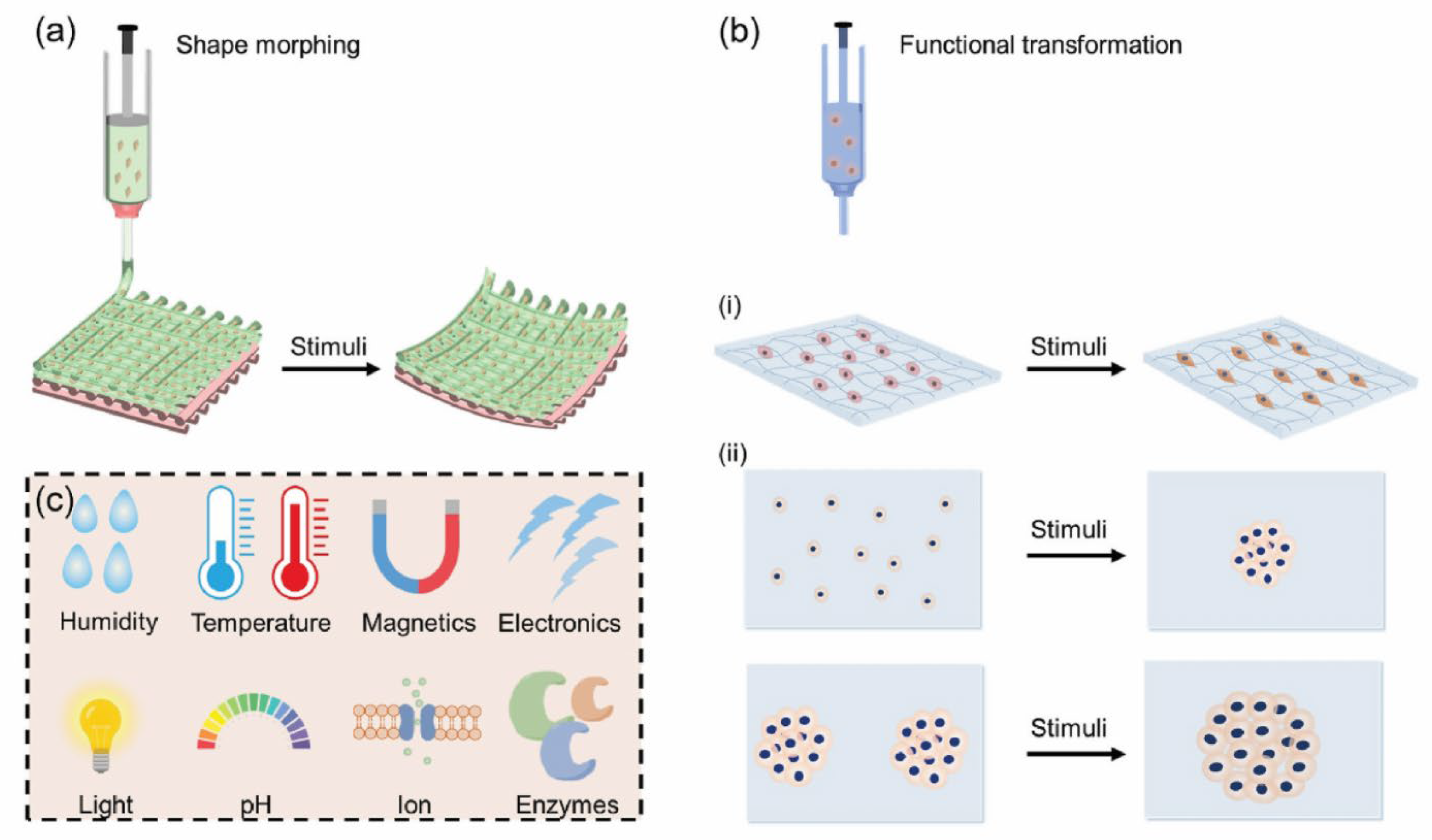
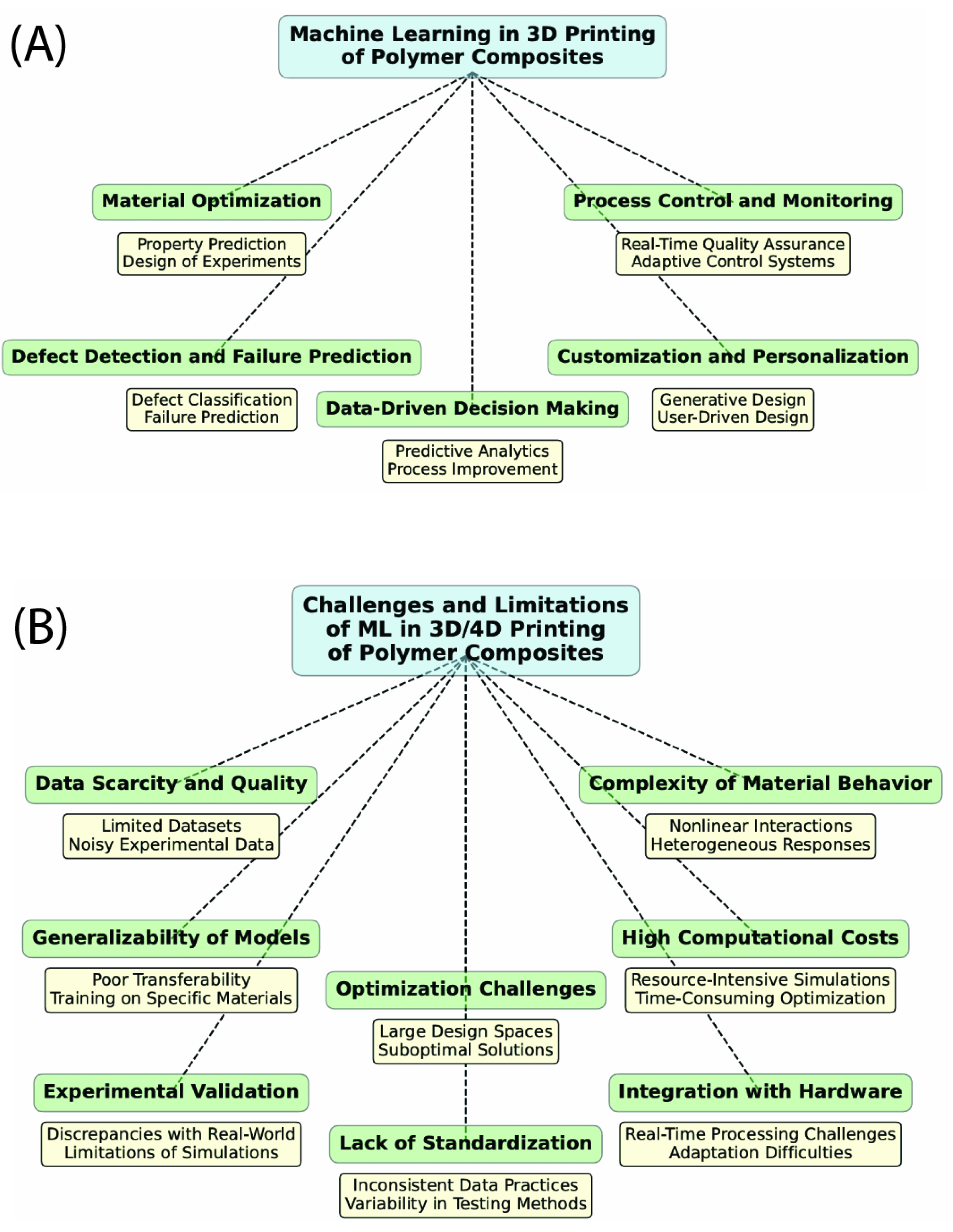
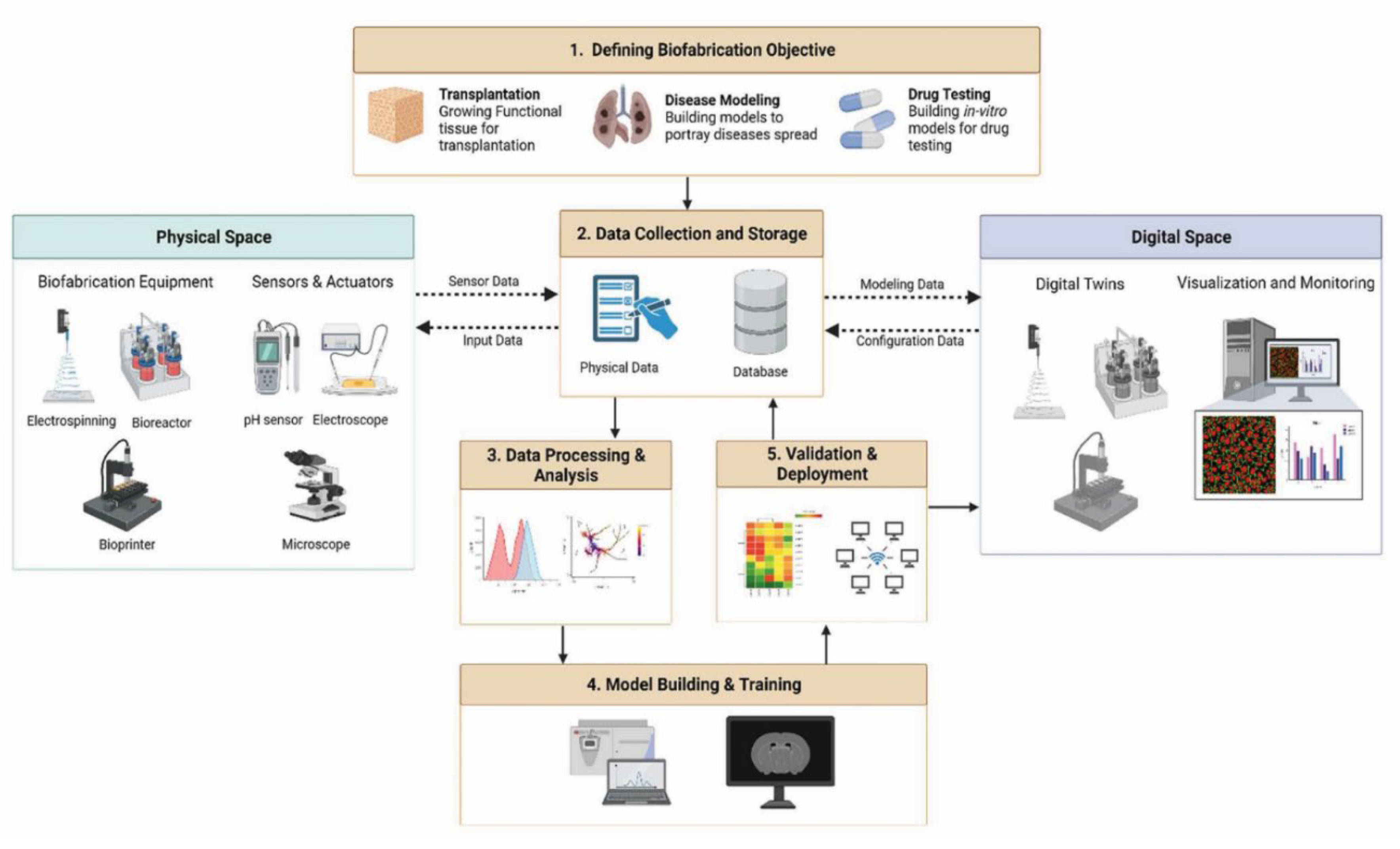
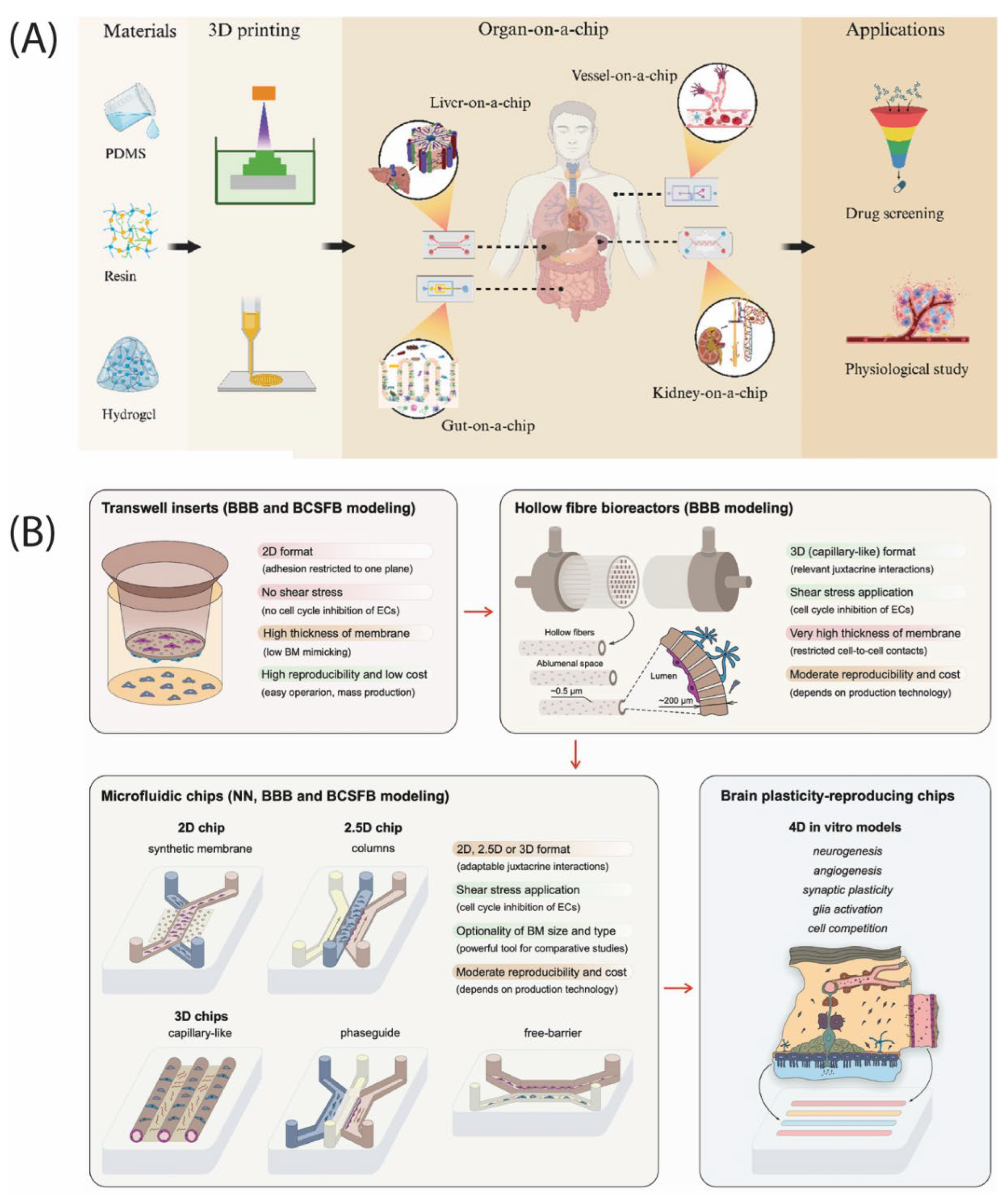
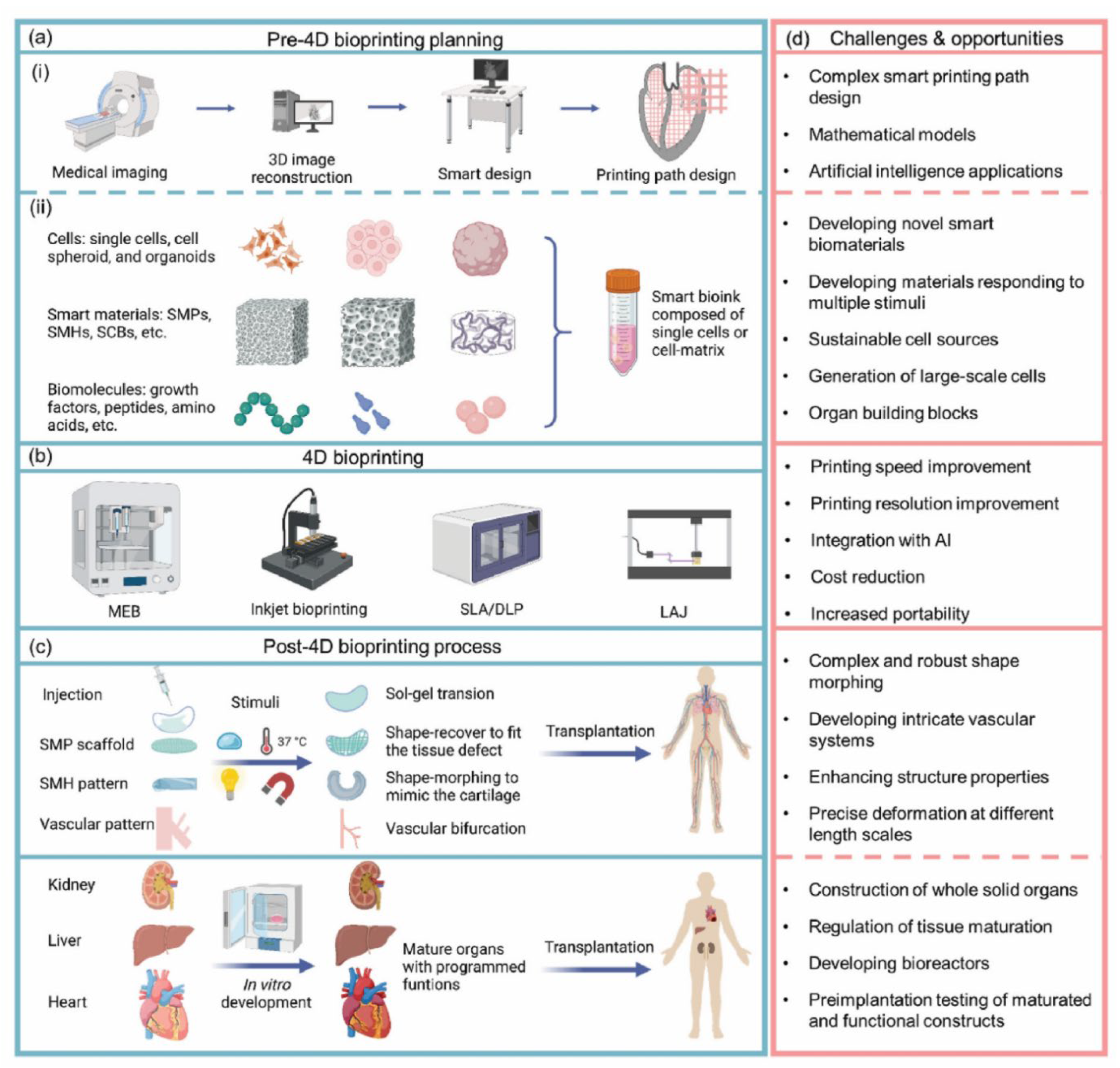
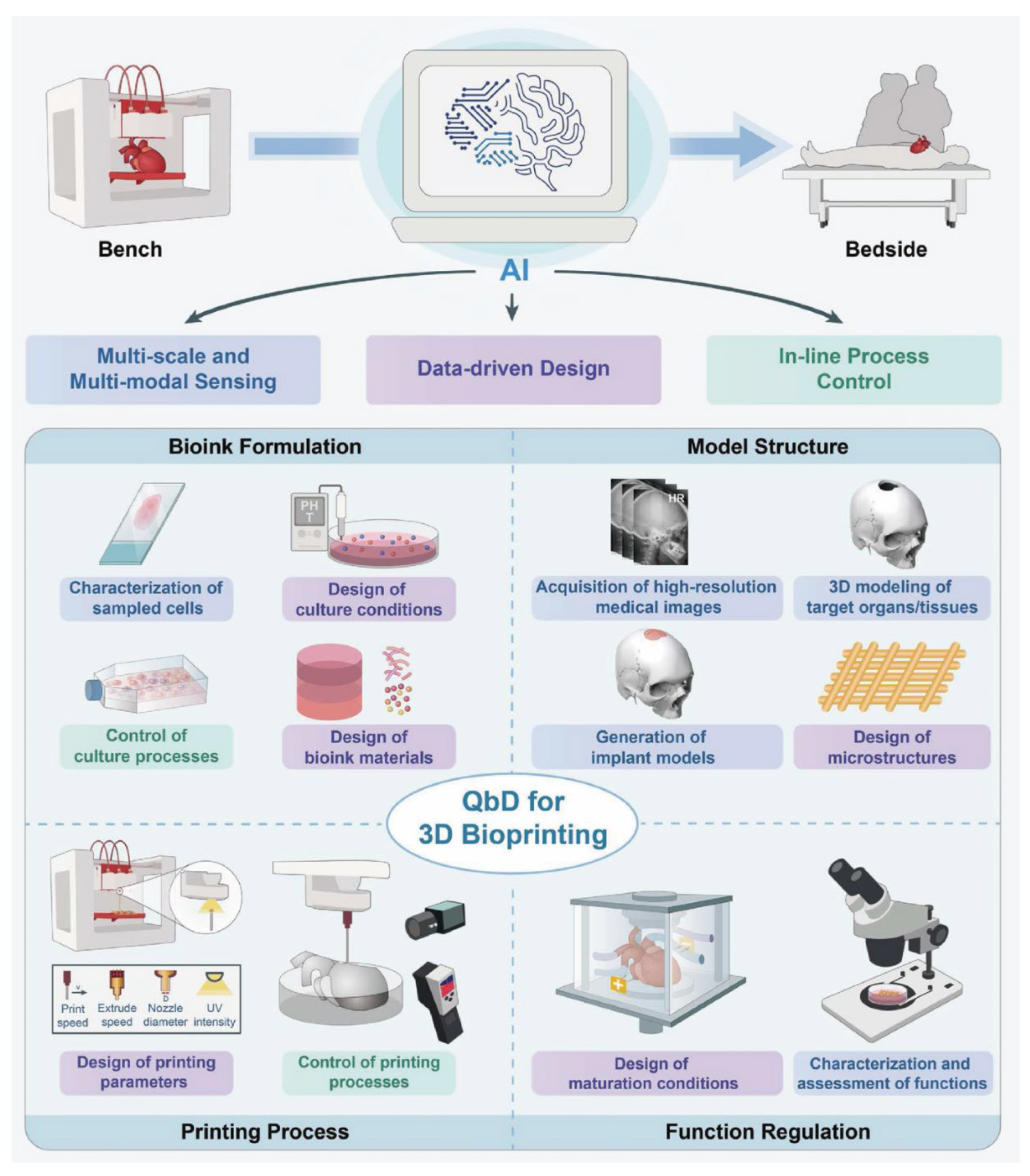
| Biological Cue/Engineering Target | Reported Experimental Range or Spec (as supported) | Source System Context | NeuroPod Design Translation (How to Use) | Ref. |
| Cortical layer analog thickness | Yan et al patterned adjacent printed strata ~50 µm thick and note that maintaining individual layers within ~100 to 200 µm supports diffusion and maturation in thick neural bioprints. | hiPSC-derived layered human neural constructs exhibiting cross-layer synaptic connectivity. | Stack multiple thin print passes (< ~200 µm each) to encode laminar identity; modulate stiffness or ligand density per layer while preserving nutrient/oxygen transport. | [26] |
| Minicolumn/banding pitch | Alternating ~50 µm cell-defined bands (neuronal vs support mixes) supported viability and cross-interface synaptic maturation; not a formal cortical minicolumn measurement but demonstrates functional benefit of fine-scale alternation. | Patterned human neural tissue bioprints. | Raster alternating neuron-enriched and glia-enriched filaments at fine pitch (≤ O(100 µm)) to create column-like microzones that promote interface signaling. | [26] |
| Gradient pore architecture | Layer-by-layer path spacing produced internal porosity gradients; 30-layer constructs; mean printed line width 0.48 ± 0.03 mm; ~94.5 percent post-print neural cell viability reported. (Pore diameters not directly quantified in micrometers.) | Layered gradient-pore hydrogel brain-like tissue print. | Use programmed nozzle path spacing and material blends to establish outward/inward pore gradients that manage diffusion and guide neurite trajectories; target high viability (~94 percent). | [23] |
| Brain-relevant modulus targeting | The mechanical mismatch between neural tissue (brain ~1 to 30 kPa) and rigid materials (for example Si ~180 GPa) drives inflammatory response, glial encapsulation, and signal degradation; soft tissue-like designs mitigate these effects. | Brain-on-chip microenvironment considerations; bio-inspired neural interface review. | Tune NeuroPod somatic regions into low-kPa modulus range; incorporate compliant gradients and supportive struts only where needed for handling to reduce chronic mechanical stress. | [21,30] |
| Compartmental isolation gaps | Microchannels ~10 µm wide and ~2.5 µm high allowed axonal extension while restricting somata, enabling reversible fluidic isolation between DRG compartments. | Reversible, reusable compartmentalized DRG neuron coculture microfluidic chip. | Embed similar micro-slit guidance barriers between NeuroPod compartments to bias axonal exit without somatic leak; reversible designs allow post-culture access. | [19] |
| Neuron-Schwann separation and co-pattern | PDMS multi-compartment microdevice with interconnecting microchannels supports spatial segregation of neurons and Schwann cells and staged differentiation/myelination studies. | Microfabricated neuron/Schwann multi-compartment platform. | Incorporate peripheral glia corridors linked to central neuronal cores in NeuroPods to study myelination and trophic support across partitions. | [31] |
| Microenvironmental heterogeneity scales | Brain-on-chip studies show that microscale (tens to hundreds of micrometers) variations in channel geometry, local stiffness, ECM coatings, and soluble gradients strongly affect neuronal phenotype and barrier behavior; numeric ranges vary by device. | Brain-on-chip microenvironment review. | Deliberately pattern micro-niches (channels, wells, ECM islands) across ~10 to 500 µm scales inside NeuroPods to present localized shear, oxygen, and biochemical gradients. | [21] |
| Conformable soft interface footprint | Soft, flexible mesh and serpentine biohybrid electrodes improve mechanical matching (brain ~1 to 30 kPa) and reduce glial encapsulation compared with rigid probes; polymer and hydrogel strategies enhance conformal contact with 3D neural tissue. | Bio-inspired electronics review. | Integrate serpentine, ultra-soft conductive traces along NeuroPod layer planes for low-strain recording or stimulation; apply bioactive or biodegradable coatings to mitigate glial response. | [30] |
| Sensor/Data Stream | What It Measures | Inline vs Offline | How It Informs Control Loop | Example Algorithm/Use Case | Integration Considerations for NeuroPods | Ref |
|---|---|---|---|---|---|---|
| In-line machine vision (camera) | Filament width, droplet volume, layer defects | Inline | Adjust pressure, speed, pause or redo layer when deviation detected | Image-based ML models trained on high-throughput droplet images; vision-based defect detection workflows | Stable illumination and calibrated optics needed for accurate image features | [39,42] |
| Rheology probes or at-line rheometry | Real-time (or batch) viscosity, yield stress, gel point drift | Inline (emerging) or batch | Tune extrusion pressure, temperature, and crosslink timing within defined design space | Data-driven printability mapping and QbD optimization using rheological inputs | Maintain sterility; limit shear exposure that can harm encapsulated neural cells | [32,42] |
| Impedance/EIS microelectrodes | Bulk and interfacial conductivity; charge transfer resistance indicating conductive network quality | Inline (embedded) or post-cure | Optimize conductive filler dispersion; assess evolution of the conductive network during and after crosslinking | Electrochemical impedance spectroscopy used to compare MXene-modified SilMA formulations | Embed removable or sacrificial microelectrodes in printbed or channels | [44] |
| Temperature probes/IR micro-imagers | Gelation front and thermal history in thermo-responsive gels | Inline | Control heating or cooling ramps to synchronize deposition with sol-gel transition and programmed 4D change | Thermally triggered injectable chitosan/β-GP systems; temperature-responsive 4D constructs reviewed | Integrate micro-thermocouples or catheter sensors for in situ neural delivery | [33,43] |
| UV/light intensity dosimetry | Spatial photocure dose mapping | Inline | Ensure uniform crosslink; compensate for light attenuation by nanofillers | Photocuring impacted by MXene opacity in SilMA and by GO light shielding in GG composites | Adjust exposure dwell; protect encapsulated cells from over-dose | [37,44] |
| Post-print viability and morphology imaging | Live/Dead, proliferation, morphology (neurite outgrowth where measured) | Offline (rapid) | Update ML models (active learning) to refine future print parameters | High-throughput printed droplet viability datasets; AI-driven QbD quality feedback; NSC spheroid viability/morphology analytics in MXene/SilMA prints | Standardize imaging pipelines and feature extraction across labs | [32,39,44] |
| Swelling/dimensional tracking (vision or gravimetric) | Hydration-driven volume change and actuation magnitude | Inline or periodic | Compare observed swelling to ANN predictions; recalibrate triggers for 4D actuation | ML prediction of hydrogel swelling states from synthesis/process metadata | Control temperature and medium; use fiducial markers for optical tracking | [40] |
| Mechanical indentation/nano-DMA | Layer modulus vs neural tissue target; time-dependent softening | Offline calibration | Update print parameters and crosslink schedules to hit patient-specific mechanical windows | Mechanical CQAs in AI-QbD workflows; rheology-printability linkages; brain tissue modeling requires soft compliance | Use low-force probes; recalibrate frequently for ultra-soft constructs | [32,41,42] |
| DT Lifecycle Stage | Core Tasks | Data Ingest | Computational Stack | Outputs to Physical System | Update Frequency | Ref. |
|---|---|---|---|---|---|---|
| 1. Design intent capture | Specify cell types, bioink composites, target microarchitecture, stimulation plan | CAD, print recipes, material libraries | Parametric DB + ontology mapping | Requirements spec; preprint simulation scenarios | One-time then on design change | [47,52,56] |
| 2. Process DT linkage | Map printer logs and in situ sensors to as-built geometry and material state; reconcile deviations | G-code, temperature, crosslink dose, in-process imaging, PAT feeds | Data pipelines + process/quality models | Corrected geometry mesh; process quality dashboard | Each print run | [47,52,62] |
| 3. Initialization of mechanistic core | Generate FEM/poroelastic mesh, ABM/phase-field populations, initial morphogen fields | As-built mesh + assay data | Multiphysics solvers; ABM/phase-field engines | Baseline digital replica | Once per device then refined | [48,51,60] |
| 4. Multiscale data assimilation | Register live sensor feeds (strain, flow, electrophysiology, biochem) and on-chip readouts | Streaming sensor bus; chip analytics | Data fusion; state estimators; ML surrogates; federated update | Updated model states; parameter posteriors | Streaming to hourly | [45,59,62] |
| 5. Predictive scenario testing | In silico perturbations: dosing, mechanical loading, field stimulation, network modulation | Current DT state | Ensemble simulations/surrogate sweeps | Ranked intervention strategies | On demand | [50,55,57] |
| 6. Control policy deployment | Translate chosen intervention to actuation commands; AI-assisted control (MPC/RL/federated update) | DT recommended setpoints | MPC/RL policy engines; supervisory controllers | Updated experiment or therapy protocol | Closed loops (min-hr) | [49,53,62] |
| V&V Level | Question Addressed | Evidence Types | Quantitative Metrics | Acceptance Criteria Example* | Regulatory/Translational Context | Ref. |
| Code Verification | Are numerical solvers implemented correctly | Benchmark problems; mesh refinement; cross-solver comparison | Residual norms; convergence rate; energy balance | <5 percent change with mesh refinement* | Good modeling practice baseline | [48,51] |
| Parameter Verification | Are material and biological parameters correctly estimated | Calibration sets; imaging inverse fits; process logs | RMSE vs imaging; posterior variance | Confidence intervals within biologically plausible range* | Preclinical DT qualification | [47,48,52] |
| Biological Validation (in vitro) | Does DT reproduce measured on-chip phenotypes | Side-by-side culture vs simulation | Morphology overlap; growth rate error; electrophysiology match | >80 percent match in growth trends over defined window* | Organ-on-chip acceptance; animal replacement | [46,59,63] |
| Scenario Prediction | Can DT rank interventions correctly | Prospective DOE with blinded predictions | Rank correlation; AUC for success classification | Correct top 3 interventions in 5 trial set* | Decision support gating to advanced studies | [46,52,63] |
| System Integration Validation | Multiscale coherence (micro to brain network) | Cross-scale datasets; imaging + NeuroPod activity | Functional connectivity error; phase coherence | Within tolerance bands derived from virtual brain benchmarks* | Translational neuroscience bridging | [50,55,57] |
| Real-Time Clinical Performance | Does adaptive DT improve outcome vs standard | Controlled study with adaptive stimulation/therapy | Clinical endpoint delta; safety events | Non-inferiority or superiority margin met* | Path to regulatory clearance | [55,57] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




