Submitted:
21 July 2025
Posted:
22 July 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Discussion
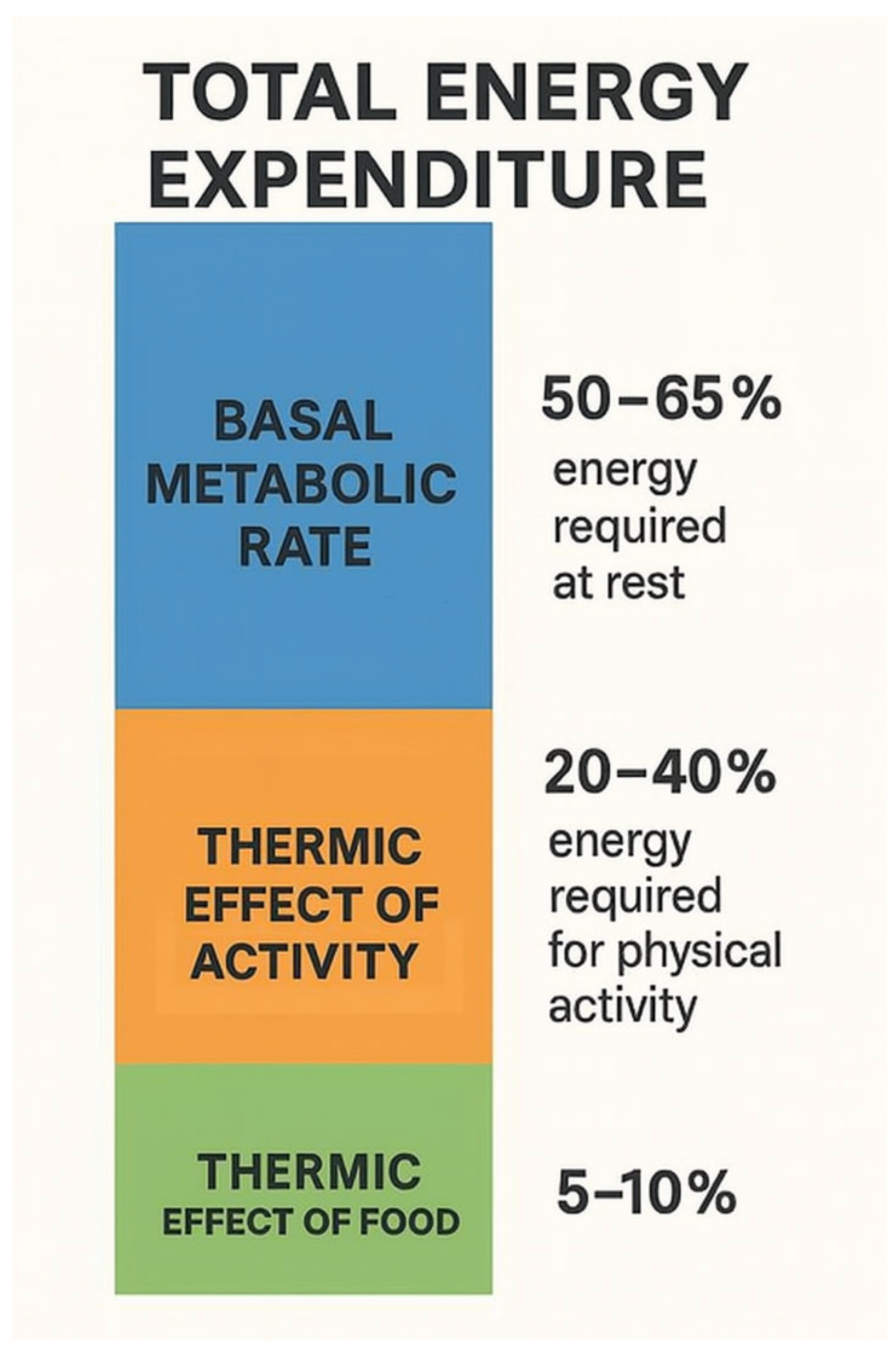
3.1. Mechanisms of Energy Expenditure
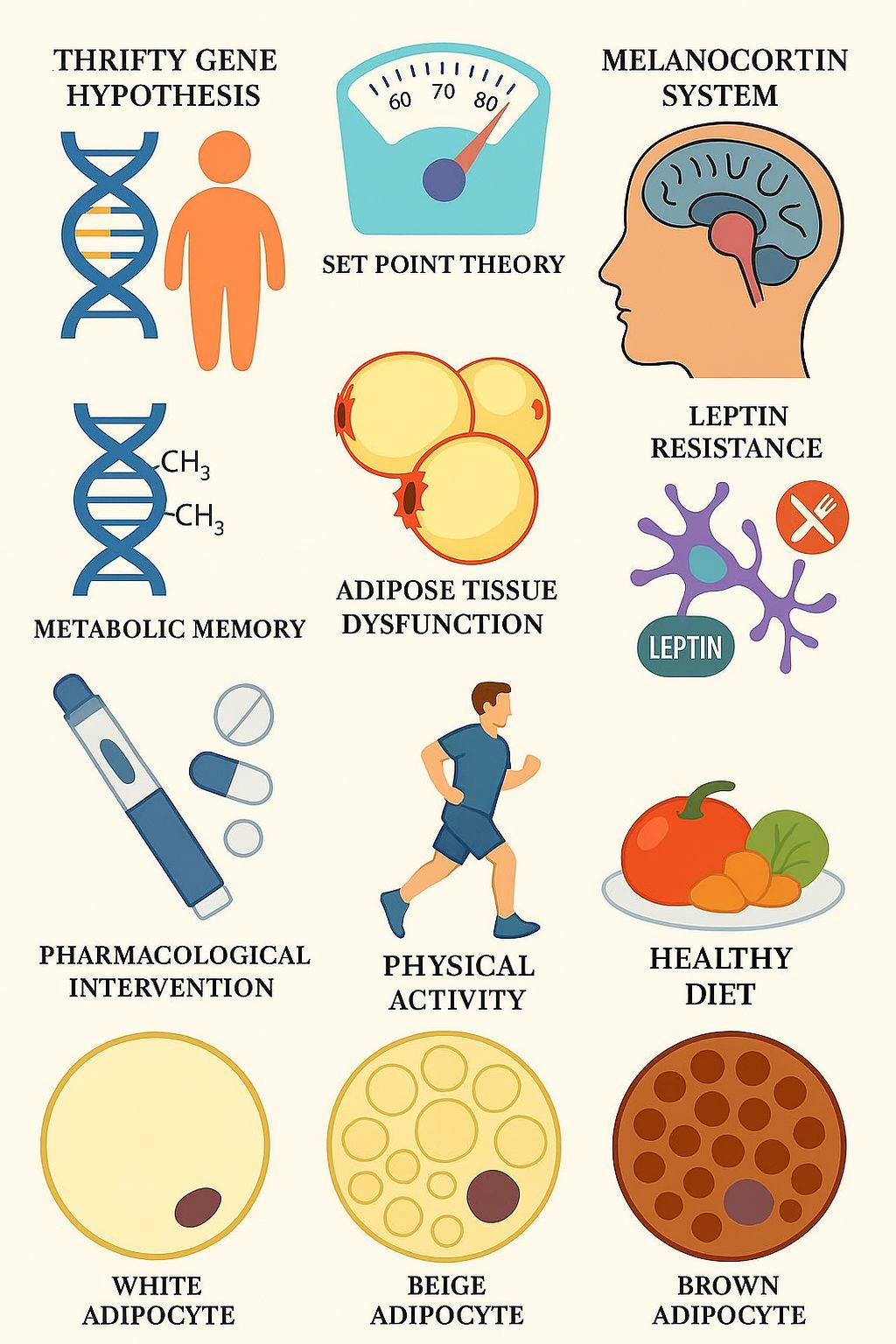
3.2. Thrifty Gene Hypothesis
3.3. Set Point Theory
3.4. Melanocortin System and Hypothalamic Regulation
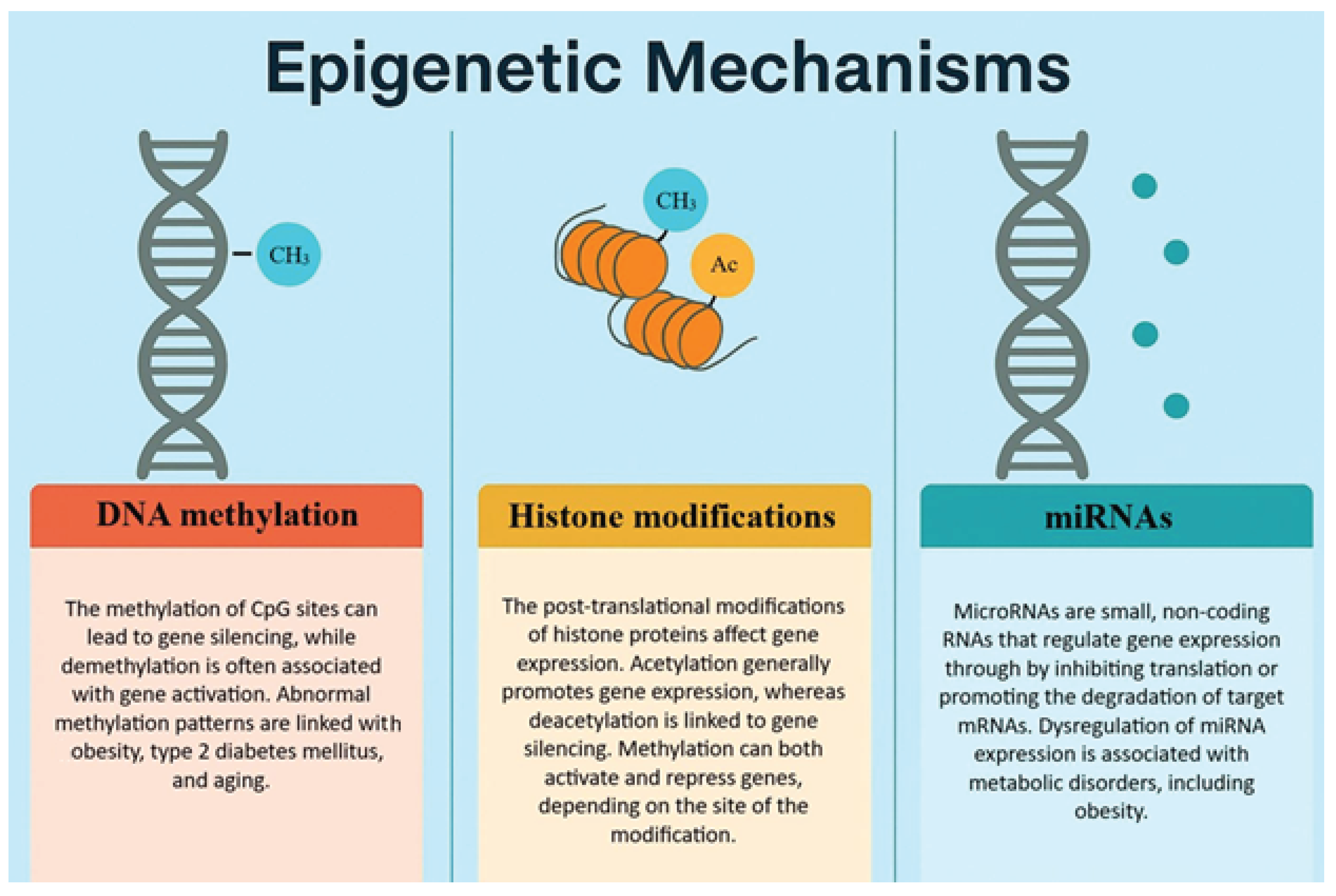
3.5. Metabolic Memory
3.6. Adipose Tissue Dysfunction
3.7. White, Brown, and Beige Adipose Tissue
3.8. Lifestyle Interventions
3.9. Pharmacological Interventions
| Drug Class | Example | Approved for Obesity? | Weight Loss Effect |
|---|---|---|---|
| Biguanides | Metformin | No | Subtle |
| β3-AR agonists | Mirabegron | No | Subtle |
| SGLT2 inhibitors | Canagliflozin | No | Moderate |
| Opioid antagonist/Dopamine reuptake inhibitor | Naltrexone/bupropion | Yes | Moderate |
| GLP-1RA | Liraglutide | Yes | Substantial |
| GLP-1/GIP dual agonists | Tirzepatide | Yes | Substantial |
| NMDA antagonist/GLP-1RA | Not available | Not available | Substantial |
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| NCDs | Noncommunicable Diseases |
| WHO | World Health Organization |
| CVDs | Cardiovascular Diseases |
| BMR | Basal Metabolic Rate |
| TEF | Thermic Effect of Food |
| TEA | Thermic Effect of Activity |
| FFM | Fat-free Mass |
| LBM | Lean Body Mass |
| MAPK | Mitogen-Activated Protein Kinase |
| TGF-β | Transforming Growth Factor Beta |
| T2DM | Type 2 Diabetes Mellitus |
| POMC | Pro-opiomelanocortin |
| CART | Cocaine- and Amphetamine- Regulated Transcript |
| AgRP | Agouti-related Peptide |
| NPY | Neuropeptide Y |
| α-MSH | α-Melanocyte Stimulating Hormone |
| MC4R | Melanocortin-4 Receptor |
| DCCT | Diabetes Control and Complications Trial |
| CpG | Cytosine linked by a phosphate to guanine |
| CGIs | CpG Islands |
| DMRs | Differentially Methylated Regions |
| TFs | Transcription factors |
| PPARγ | Peroxisome Proliferator-Activated Receptor Gamma |
| miRNAs | microRNAs |
| WHR | Waist-to-Hip |
| WAT | White Adipose Tissue |
| BAT | Brown Adipose Tissue |
| BeAT | Beige Adipose Tissue |
| UCP1 | Uncoupling Protein 1 |
| ATP | Adenosine Triphosphate |
| CR | Calorie Restriction |
| β3-AR | β3-Adrenergic Receptor |
| GLP1-RA | Glucagon-like Peptide 1 Receptor Agonists |
| FDA | Food and Drug Administration |
| GIP | Glucose-Dependent Insulinotropic Polypeptide |
| NMDA | N-methyl-D-aspartate |
References
- World Obesity Atlas 2025. Available online: https://www.worldobesity.org/resources/resource-library/world-obesity-atlas-2025 (accessed on 09 July 2025).
- World Obesity: Prevalence of Obesity. Available online: https://www.worldobesity.org/about/about-obesity/prevalence-of-obesity (accessed on 09 July 2025).
- World Health Organization: Diabetes. Available online: https://www.who.int/news-room/fact-sheets/detail/diabetes (accessed on 09 July 2025).
- Xie, F.; Xiong, F.; Yang, B.; Yan, Z.; Shen, Y.; Qin, H.; Chen, L.; Chen, T.; Chen, J.; Zhu, S.; et al. Global, regional, and national burden of mortality and DALYs attributable to high body mass index from 1990 to 2021 with projections to 2036. BMC Public Health 2025, 25. [CrossRef]
- Hall, K.D.; Guo, J. Obesity Energetics: Body Weight Regulation and the Effects of Diet Composition. Gastroenterology 2017, 152, 1718-1727.e1713. [CrossRef]
- van Baak, M.A.; Mariman, E.C.M. Mechanisms of weight regain after weight loss - the role of adipose tissue. Nat Rev Endocrinol 2019, 15, 274-287. [CrossRef]
- Di Francesco, A.; Deighan, A.G.; Litichevskiy, L.; Chen, Z.; Luciano, A.; Robinson, L.; Garland, G.; Donato, H.; Vincent, M.; Schott, W.; et al. Dietary restriction impacts health and lifespan of genetically diverse mice. Nature 2024, 634, 684-692. [CrossRef]
- Reddy, M.A.; Zhang, E.; Natarajan, R. Epigenetic mechanisms in diabetic complications and metabolic memory. Diabetologia 2015, 58, 443-455. [CrossRef]
- Medeiros Denis, M.; Wildman, R.E.C. Advanced Human Nutrition, 4 ed.; Jones & Bartlett Learning: Burlington, MA, USA, 2019.
- Denzer, C.M.; Young, J.C. The effect of resistance exercise on the thermic effect of food. Int J Sport Nutr Exerc Metab 2003, 13, 396-402. [CrossRef]
- Calcagno, M.; Kahleova, H.; Alwarith, J.; Burgess, N.N.; Flores, R.A.; Busta, M.L.; Barnard, N.D. The Thermic Effect of Food: A Review. J Am Coll Nutr 2019, 38, 547-551. [CrossRef]
- Colpani, V.; Oppermann, K.; Spritzer, P.M. Association between habitual physical activity and lower cardiovascular risk in premenopausal, perimenopausal, and postmenopausal women: a population-based study. Menopause 2013, 20, 525-531. [CrossRef]
- Wang, Z.; Ying, Z.; Bosy-Westphal, A.; Zhang, J.; Heller, M.; Later, W.; Heymsfield, S.B.; Müller, M.J. Evaluation of specific metabolic rates of major organs and tissues: comparison between men and women. Am J Hum Biol 2011, 23, 333-338. [CrossRef]
- Polotsky, H.N.; Polotsky, A.J. Metabolic implications of menopause. Semin Reprod Med 2010, 28, 426-434. [CrossRef]
- Vujović, N.; Piron, M.J.; Qian, J.; Chellappa, S.L.; Nedeltcheva, A.; Barr, D.; Heng, S.W.; Kerlin, K.; Srivastav, S.; Wang, W.; et al. Late isocaloric eating increases hunger, decreases energy expenditure, and modifies metabolic pathways in adults with overweight and obesity. Cell Metab 2022, 34, 1486-1498.e1487. [CrossRef]
- NEEL, J.V. Diabetes mellitus: a “thrifty” genotype rendered detrimental by “progress”? Am J Hum Genet 1962, 14, 353-362.
- Ayub, Q.; Moutsianas, L.; Chen, Y.; Panoutsopoulou, K.; Colonna, V.; Pagani, L.; Prokopenko, I.; Ritchie, G.R.; Tyler-Smith, C.; McCarthy, M.I.; et al. Revisiting the thrifty gene hypothesis via 65 loci associated with susceptibility to type 2 diabetes. Am J Hum Genet 2014, 94, 176-185. [CrossRef]
- Speakman, J.R. Thrifty genes for obesity, an attractive but flawed idea, and an alternative perspective: the ‘drifty gene’ hypothesis. Int J Obes (Lond) 2008, 32, 1611-1617. [CrossRef]
- Loos, R.J.F.; Yeo, G.S.H. The genetics of obesity: from discovery to biology. Nat Rev Genet 2022, 23, 120-133. [CrossRef]
- Wirtshafter, D.; Davis, J.D. Set points, settling points, and the control of body weight. Physiol Behav 1977, 19, 75-78. [CrossRef]
- KENNEDY, G.C. The role of depot fat in the hypothalamic control of food intake in the rat. Proc R Soc Lond B Biol Sci 1953, 140, 578-596. [CrossRef]
- Keesey, R.E.; Corbett, S.W. Metabolic defense of the body weight set-point. Res Publ Assoc Res Nerv Ment Dis 1984, 62, 87-96.
- Schwartz, M.W.; Woods, S.C.; Porte, D.; Seeley, R.J.; Baskin, D.G. Central nervous system control of food intake. Nature 2000, 404, 661-671. [CrossRef]
- Speakman, J.R.; Levitsky, D.A.; Allison, D.B.; Bray, M.S.; de Castro, J.M.; Clegg, D.J.; Clapham, J.C.; Dulloo, A.G.; Gruer, L.; Haw, S.; et al. Set points, settling points and some alternative models: theoretical options to understand how genes and environments combine to regulate body adiposity. Dis Model Mech 2011, 4, 733-745. [CrossRef]
- Huszar, D.; Lynch, C.A.; Fairchild-Huntress, V.; Dunmore, J.H.; Fang, Q.; Berkemeier, L.R.; Gu, W.; Kesterson, R.A.; Boston, B.A.; Cone, R.D.; et al. Targeted disruption of the melanocortin-4 receptor results in obesity in mice. Cell 1997, 88, 131-141. [CrossRef]
- Nathan, D.M.; Cleary, P.A.; Backlund, J.Y.; Genuth, S.M.; Lachin, J.M.; Orchard, T.J.; Raskin, P.; Zinman, B.; Group, D.C.a.C.T.E.o.D.I.a.C.D.E.S.R. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N Engl J Med 2005, 353, 2643-2653. [CrossRef]
- Samblas, M.; Milagro, F.I.; Martínez, A. DNA methylation markers in obesity, metabolic syndrome, and weight loss. Epigenetics 2019, 14, 421-444. [CrossRef]
- Ren, Y.; Huang, P.; Zhang, L.; Tang, Y.F.; Luo, S.L.; She, Z.; Peng, H.; Chen, Y.Q.; Luo, J.W.; Duan, W.X.; et al. Dual Regulation Mechanism of Obesity: DNA Methylation and Intestinal Flora. Biomedicines 2024, 12. [CrossRef]
- Wahl, S.; Drong, A.; Lehne, B.; Loh, M.; Scott, W.R.; Kunze, S.; Tsai, P.C.; Ried, J.S.; Zhang, W.; Yang, Y.; et al. Epigenome-wide association study of body mass index, and the adverse outcomes of adiposity. Nature 2017, 541, 81-86. [CrossRef]
- Zhao, J.; Goldberg, J.; Vaccarino, V. Promoter methylation of serotonin transporter gene is associated with obesity measures: a monozygotic twin study. Int J Obes (Lond) 2013, 37, 140-145. [CrossRef]
- Abdul, Q.A.; Yu, B.P.; Chung, H.Y.; Jung, H.A.; Choi, J.S. Epigenetic modifications of gene expression by lifestyle and environment. Arch Pharm Res 2017, 40, 1219-1237. [CrossRef]
- Si, J.; Meir, A.Y.; Hong, X.; Wang, G.; Huang, W.; Pearson, C.; Adams, W.G.; Wang, X.; Liang, L. Maternal pre-pregnancy BMI, offspring epigenome-wide DNA methylation, and childhood obesity: findings from the Boston Birth Cohort. BMC Med 2023, 21, 317. [CrossRef]
- Sharp, G.C.; Lawlor, D.A.; Richmond, R.C.; Fraser, A.; Simpkin, A.; Suderman, M.; Shihab, H.A.; Lyttleton, O.; McArdle, W.; Ring, S.M.; et al. Maternal pre-pregnancy BMI and gestational weight gain, offspring DNA methylation and later offspring adiposity: findings from the Avon Longitudinal Study of Parents and Children. Int J Epidemiol 2015, 44, 1288-1304. [CrossRef]
- Jung, M.; Pfeifer, G.P. Aging and DNA methylation. BMC Biol 2015, 13, 7. [CrossRef]
- Klutstein, M.; Nejman, D.; Greenfield, R.; Cedar, H. DNA Methylation in Cancer and Aging. Cancer Res 2016, 76, 3446-3450. [CrossRef]
- Mikula, M.; Majewska, A.; Ledwon, J.K.; Dzwonek, A.; Ostrowski, J. Obesity increases histone H3 lysine 9 and 18 acetylation at Tnfa and Ccl2 genes in mouse liver. Int J Mol Med 2014, 34, 1647-1654. [CrossRef]
- Castillo, J.; López-Rodas, G.; Franco, L. Histone Post-Translational Modifications and Nucleosome Organisation in Transcriptional Regulation: Some Open Questions. Adv Exp Med Biol 2017, 966, 65-92. [CrossRef]
- Yang, H.; Yang, K.; Gu, H.; Sun, C. Dynamic post-translational modifications in obesity. J Cell Mol Med 2020, 24, 2384-2387. [CrossRef]
- Lorente-Cebrián, S.; González-Muniesa, P.; Milagro, F.I.; Martínez, J.A. MicroRNAs and other non-coding RNAs in adipose tissue and obesity: emerging roles as biomarkers and therapeutic targets. Clin Sci (Lond) 2019, 133, 23-40. [CrossRef]
- Esau, C.; Kang, X.; Peralta, E.; Hanson, E.; Marcusson, E.G.; Ravichandran, L.V.; Sun, Y.; Koo, S.; Perera, R.J.; Jain, R.; et al. MicroRNA-143 regulates adipocyte differentiation. J Biol Chem 2004, 279, 52361-52365. [CrossRef]
- Kim, S.Y.; Kim, A.Y.; Lee, H.W.; Son, Y.H.; Lee, G.Y.; Lee, J.W.; Lee, Y.S.; Kim, J.B. miR-27a is a negative regulator of adipocyte differentiation via suppressing PPARgamma expression. Biochem Biophys Res Commun 2010, 392, 323-328. [CrossRef]
- Takanabe, R.; Ono, K.; Abe, Y.; Takaya, T.; Horie, T.; Wada, H.; Kita, T.; Satoh, N.; Shimatsu, A.; Hasegawa, K. Up-regulated expression of microRNA-143 in association with obesity in adipose tissue of mice fed high-fat diet. Biochem Biophys Res Commun 2008, 376, 728-732. [CrossRef]
- Barrès, R.; Zierath, J.R. The role of diet and exercise in the transgenerational epigenetic landscape of T2DM. Nat Rev Endocrinol 2016, 12, 441-451. [CrossRef]
- Fuster, J.J.; Ouchi, N.; Gokce, N.; Walsh, K. Obesity-Induced Changes in Adipose Tissue Microenvironment and Their Impact on Cardiovascular Disease. Circ Res 2016, 118, 1786-1807. [CrossRef]
- Klein, S.; Allison, D.B.; Heymsfield, S.B.; Kelley, D.E.; Leibel, R.L.; Nonas, C.; Kahn, R.; Prevention, A.f.W.M.a.O.; NAASO; Society, O.; et al. Waist circumference and cardiometabolic risk: a consensus statement from shaping America’s health: Association for Weight Management and Obesity Prevention; NAASO, the Obesity Society; the American Society for Nutrition; and the American Diabetes Association. Diabetes Care 2007, 30, 1647-1652. [CrossRef]
- Cildir, G.; Akıncılar, S.C.; Tergaonkar, V. Chronic adipose tissue inflammation: all immune cells on the stage. Trends Mol Med 2013, 19, 487-500. [CrossRef]
- Goossens, G.H.; Bizzarri, A.; Venteclef, N.; Essers, Y.; Cleutjens, J.P.; Konings, E.; Jocken, J.W.; Cajlakovic, M.; Ribitsch, V.; Clément, K.; et al. Increased adipose tissue oxygen tension in obese compared with lean men is accompanied by insulin resistance, impaired adipose tissue capillarization, and inflammation. Circulation 2011, 124, 67-76. [CrossRef]
- Marcelin, G.; Gautier, E.L.; Clément, K. Adipose Tissue Fibrosis in Obesity: Etiology and Challenges. Annu Rev Physiol 2022, 84, 135-155. [CrossRef]
- Liu, X.; Zhao, L.; Chen, Y.; Gao, Y.; Tian, Q.; Son, J.S.; Chae, S.A.; de Avila, J.M.; Zhu, M.J.; Du, M. Obesity induces adipose fibrosis and collagen cross-linking through suppressing AMPK and enhancing lysyl oxidase expression. Biochim Biophys Acta Mol Basis Dis 2022, 1868, 166454. [CrossRef]
- Nakamura, K.; Fuster, J.J.; Walsh, K. Adipokines: a link between obesity and cardiovascular disease. J Cardiol 2014, 63, 250-259. [CrossRef]
- Suárez-Cuenca, J.A.; De La Peña-Sosa, G.; De La Vega-Moreno, K.; Banderas-Lares, D.Z.; Salamanca-García, M.; Martínez-Hernández, J.E.; Vera-Gómez, E.; Hernández-Patricio, A.; Zamora-Alemán, C.R.; Domínguez-Pérez, G.A.; et al. Enlarged adipocytes from subcutaneous vs. visceral adipose tissue differentially contribute to metabolic dysfunction and atherogenic risk of patients with obesity. Sci Rep 2021, 11, 1831. [CrossRef]
- Pan, W.W.; Myers, M.G. Leptin and the maintenance of elevated body weight. Nat Rev Neurosci 2018, 19, 95-105. [CrossRef]
- Sakers, A.; De Siqueira, M.K.; Seale, P.; Villanueva, C.J. Adipose-tissue plasticity in health and disease. Cell 2022, 185, 419-446. [CrossRef]
- Cedikova, M.; Kripnerová, M.; Dvorakova, J.; Pitule, P.; Grundmanova, M.; Babuska, V.; Mullerova, D.; Kuncova, J. Mitochondria in White, Brown, and Beige Adipocytes. Stem Cells Int 2016, 2016, 6067349. [CrossRef]
- Cai, J.; Wang, F.; Shao, M. The Emerging Importance of Mitochondria in White Adipocytes: Neither Last nor Least. Endocrinol Metab (Seoul) 2023, 38, 493-503. [CrossRef]
- McNeill, B.T.; Morton, N.M.; Stimson, R.H. Substrate Utilization by Brown Adipose Tissue: What’s Hot and What’s Not? Front Endocrinol (Lausanne) 2020, 11, 571659. [CrossRef]
- Labbé, S.M.; Caron, A.; Chechi, K.; Laplante, M.; Lecomte, R.; Richard, D. Metabolic activity of brown, “beige,” and white adipose tissues in response to chronic adrenergic stimulation in male mice. Am J Physiol Endocrinol Metab 2016, 311, E260-268. [CrossRef]
- Wu, J.; Boström, P.; Sparks, L.M.; Ye, L.; Choi, J.H.; Giang, A.H.; Khandekar, M.; Virtanen, K.A.; Nuutila, P.; Schaart, G.; et al. Beige adipocytes are a distinct type of thermogenic fat cell in mouse and human. Cell 2012, 150, 366-376. [CrossRef]
- Jung, S.M.; Sanchez-Gurmaches, J.; Guertin, D.A. Brown Adipose Tissue Development and Metabolism. Handb Exp Pharmacol 2019, 251, 3-36. [CrossRef]
- Loeliger, R.C.; Maushart, C.I.; Gashi, G.; Senn, J.R.; Felder, M.; Becker, A.S.; Müller, J.; Balaz, M.; Wolfrum, C.; Burger, I.A.; et al. Relation of diet-induced thermogenesis to brown adipose tissue activity in healthy men. Am J Physiol Endocrinol Metab 2021, 320, E93-E101. [CrossRef]
- Suchacki, K.J.; Stimson, R.H. Nutritional Regulation of Human Brown Adipose Tissue. Nutrients 2021, 13. [CrossRef]
- Schulz, T.J.; Huang, P.; Huang, T.L.; Xue, R.; McDougall, L.E.; Townsend, K.L.; Cypess, A.M.; Mishina, Y.; Gussoni, E.; Tseng, Y.H. Brown-fat paucity due to impaired BMP signalling induces compensatory browning of white fat. Nature 2013, 495, 379-383. [CrossRef]
- Rosen, E.D.; Spiegelman, B.M. What we talk about when we talk about fat. Cell 2014, 156, 20-44. [CrossRef]
- Bales, C.W.; Kraus, W.E. Caloric restriction: implications for human cardiometabolic health. J Cardiopulm Rehabil Prev 2013, 33, 201-208. [CrossRef]
- Lean, M.E.; Leslie, W.S.; Barnes, A.C.; Brosnahan, N.; Thom, G.; McCombie, L.; Peters, C.; Zhyzhneuskaya, S.; Al-Mrabeh, A.; Hollingsworth, K.G.; et al. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet 2018, 391, 541-551. [CrossRef]
- Fabbiano, S.; Suárez-Zamorano, N.; Rigo, D.; Veyrat-Durebex, C.; Dokic, S., Ana; Colin, J., Didier; Trajkovski, M. Caloric Restriction Leads to Browning of White Adipose Tissue through Type 2 Immune Signaling. Cell Metabolism 2016, 24, 434-446. [CrossRef]
- Hursel, R.; Viechtbauer, W.; Dulloo, A.G.; Tremblay, A.; Tappy, L.; Rumpler, W.; Westerterp-Plantenga, M.S. The effects of catechin rich teas and caffeine on energy expenditure and fat oxidation: a meta-analysis. Obes Rev 2011, 12, e573-581. [CrossRef]
- Yoneshiro, T.; Matsushita, M.; Hibi, M.; Tone, H.; Takeshita, M.; Yasunaga, K.; Katsuragi, Y.; Kameya, T.; Sugie, H.; Saito, M. Tea catechin and caffeine activate brown adipose tissue and increase cold-induced thermogenic capacity in humans. Am J Clin Nutr 2017, 105, 873-881. [CrossRef]
- Gali Ramamoorthy, T.; Allen, T.J.; Davies, A.; Harno, E.; Sefton, C.; Murgatroyd, C.; White, A. Maternal overnutrition programs epigenetic changes in the regulatory regions of hypothalamic Pomc in the offspring of rats. Int J Obes (Lond) 2018, 42, 1431-1444. [CrossRef]
- Mahmoud, A.M.; Ali, M.M. Methyl Donor Micronutrients that Modify DNA Methylation and Cancer Outcome. Nutrients 2019, 11. [CrossRef]
- Anderson, O.S.; Sant, K.E.; Dolinoy, D.C. Nutrition and epigenetics: an interplay of dietary methyl donors, one-carbon metabolism and DNA methylation. J Nutr Biochem 2012, 23, 853-859. [CrossRef]
- Dolinoy, D.C. The agouti mouse model: an epigenetic biosensor for nutritional and environmental alterations on the fetal epigenome. Nutr Rev 2008, 66 Suppl 1, S7-11. [CrossRef]
- Wheatley, K.E.; Nogueira, L.M.; Perkins, S.N.; Hursting, S.D. Differential effects of calorie restriction and exercise on the adipose transcriptome in diet-induced obese mice. J Obes 2011, 2011, 265417. [CrossRef]
- Hinte, L.C.; Castellano-Castillo, D.; Ghosh, A.; Melrose, K.; Gasser, E.; Noé, F.; Massier, L.; Dong, H.; Sun, W.; Hoffmann, A.; et al. Adipose tissue retains an epigenetic memory of obesity after weight loss. Nature 2024, 636, 457-465. [CrossRef]
- Lu, S.C.; Huang, Z.Z.; Yang, H.; Mato, J.M.; Avila, M.A.; Tsukamoto, H. Changes in methionine adenosyltransferase and S-adenosylmethionine homeostasis in alcoholic rat liver. Am J Physiol Gastrointest Liver Physiol 2000, 279, G178-185. [CrossRef]
- Piyathilake, C.J.; Macaluso, M.; Hine, R.J.; Richards, E.W.; Krumdieck, C.L. Local and systemic effects of cigarette smoking on folate and vitamin B-12. Am J Clin Nutr 1994, 60, 559-566. [CrossRef]
- Thyfault, J.P.; Bergouignan, A. Exercise and metabolic health: beyond skeletal muscle. Diabetologia 2020, 63, 1464-1474. [CrossRef]
- García-Hermoso, A.; Ceballos-Ceballos, R.J.; Poblete-Aro, C.E.; Hackney, A.C.; Mota, J.; Ramírez-Vélez, R. Exercise, adipokines and pediatric obesity: a meta-analysis of randomized controlled trials. Int J Obes (Lond) 2017, 41, 475-482. [CrossRef]
- Stanford, K.I.; Goodyear, L.J. Exercise regulation of adipose tissue. Adipocyte 2016, 5, 153-162. [CrossRef]
- Rönn, T.; Volkov, P.; Davegårdh, C.; Dayeh, T.; Hall, E.; Olsson, A.H.; Nilsson, E.; Tornberg, A.; Dekker Nitert, M.; Eriksson, K.F.; et al. A six months exercise intervention influences the genome-wide DNA methylation pattern in human adipose tissue. PLoS Genet 2013, 9, e1003572. [CrossRef]
- Denham, J.; Marques, F.Z.; O’Brien, B.J.; Charchar, F.J. Exercise: putting action into our epigenome. Sports Med 2014, 44, 189-209. [CrossRef]
- Goel, S.; Singh, R.; Singh, V.; Singh, H.; Kumari, P.; Chopra, H.; Sharma, R.; Nepovimova, E.; Valis, M.; Kuca, K.; et al. Metformin: Activation of 5′ AMP-activated protein kinase and its emerging potential beyond anti-hyperglycemic action. Front Genet 2022, 13, 1022739. [CrossRef]
- Szekeres, Z.; Toth, K.; Szabados, E. The Effects of SGLT2 Inhibitors on Lipid Metabolism. Metabolites 2021, 11. [CrossRef]
- Hanssen, M.J.; Hoeks, J.; Brans, B.; van der Lans, A.A.; Schaart, G.; van den Driessche, J.J.; Jörgensen, J.A.; Boekschoten, M.V.; Hesselink, M.K.; Havekes, B.; et al. Short-term cold acclimation improves insulin sensitivity in patients with type 2 diabetes mellitus. Nat Med 2015, 21, 863-865. [CrossRef]
- Schena, G.; Caplan, M.J. Everything You Always Wanted to Know about β. Cells 2019, 8. [CrossRef]
- Loh, R.K.C.; Formosa, M.F.; La Gerche, A.; Reutens, A.T.; Kingwell, B.A.; Carey, A.L. Acute metabolic and cardiovascular effects of mirabegron in healthy individuals. Diabetes Obes Metab 2019, 21, 276-284. [CrossRef]
- Szekeres, Z.; Nagy, A.; Jahner, K.; Szabados, E. Impact of Selected Glucagon-like Peptide-1 Receptor Agonists on Serum Lipids, Adipose Tissue, and Muscle Metabolism-A Narrative Review. Int J Mol Sci 2024, 25. [CrossRef]
- Zhu, E.; Yang, Y.; Zhang, J.; Li, Y.; Li, C.; Chen, L.; Sun, B. Liraglutide suppresses obesity and induces brown fat-like phenotype via Soluble Guanylyl Cyclase mediated pathway in vivo and in vitro. Oncotarget 2016, 7, 81077-81089. [CrossRef]
- Zhao, L.; Li, W.; Zhang, P.; Wang, D.; Yang, L.; Yuan, G. Liraglutide induced browning of visceral white adipose through regulation of miRNAs in high-fat-diet-induced obese mice. Endocrine 2024, 85, 222-232. [CrossRef]
- Beiroa, D.; Imbernon, M.; Gallego, R.; Senra, A.; Herranz, D.; Villarroya, F.; Serrano, M.; Fernø, J.; Salvador, J.; Escalada, J.; et al. GLP-1 agonism stimulates brown adipose tissue thermogenesis and browning through hypothalamic AMPK. Diabetes 2014, 63, 3346-3358. [CrossRef]
- Nreu, B.; Dicembrini, I.; Tinti, F.; Mannucci, E.; Monami, M. Pancreatitis and pancreatic cancer in patients with type 2 diabetes treated with glucagon-like peptide-1 receptor agonists: an updated meta-analysis of randomized controlled trials. Minerva Endocrinol (Torino) 2023, 48, 206-213. [CrossRef]
- Guo, H.; Guo, Q.; Li, Z.; Wang, Z. Association between different GLP-1 receptor agonists and acute pancreatitis: case series and real-world pharmacovigilance analysis. Front Pharmacol 2024, 15, 1461398. [CrossRef]
- Silverii, G.A.; Monami, M.; Gallo, M.; Ragni, A.; Prattichizzo, F.; Renzelli, V.; Ceriello, A.; Mannucci, E. Glucagon-like peptide-1 receptor agonists and risk of thyroid cancer: A systematic review and meta-analysis of randomized controlled trials. Diabetes Obes Metab 2024, 26, 891-900. [CrossRef]
- Baxter, S.M.; Lund, L.C.; Andersen, J.H.; Brix, T.H.; Hegedüs, L.; Hsieh, M.H.; Su, C.T.; Cheng, M.C.; Chang, Z.C.; Lai, E.C.; et al. Glucagon-Like Peptide 1 Receptor Agonists and Risk of Thyroid Cancer: An International Multisite Cohort Study. Thyroid 2025, 35, 69-78. [CrossRef]
- Wilding, J.P.H.; Batterham, R.L.; Calanna, S.; Davies, M.; Van Gaal, L.F.; Lingvay, I.; McGowan, B.M.; Rosenstock, J.; Tran, M.T.D.; Wadden, T.A.; et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med 2021, 384, 989-1002. [CrossRef]
- le Roux, C.W.; Fils-Aimé, N.; Camacho, F.; Gould, E.; Barakat, M. The relationship between early weight loss and weight loss maintenance with naltrexone-bupropion therapy. EClinicalMedicine 2022, 49, 101436. [CrossRef]
- Jastreboff, A.M.; Aronne, L.J.; Ahmad, N.N.; Wharton, S.; Connery, L.; Alves, B.; Kiyosue, A.; Zhang, S.; Liu, B.; Bunck, M.C.; et al. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med 2022, 387, 205-216. [CrossRef]
- Frías, J.P.; Davies, M.J.; Rosenstock, J.; Pérez Manghi, F.C.; Fernández Landó, L.; Bergman, B.K.; Liu, B.; Cui, X.; Brown, K.; Investigators, S.-. Tirzepatide versus Semaglutide Once Weekly in Patients with Type 2 Diabetes. N Engl J Med 2021, 385, 503-515. [CrossRef]
- Popik, P.; Kos, T.; Zhang, Y.; Bisaga, A. Memantine reduces consumption of highly palatable food in a rat model of binge eating. Amino Acids 2011, 40, 477-485. [CrossRef]
- Foltin, R.W.; Danysz, W.; Bisaga, A. A novel procedure for assessing the effects of drugs on satiation in baboons: effects of memantine and dexfenfluramine. Psychopharmacology (Berl) 2008, 199, 583-592. [CrossRef]
- Brennan, B.P.; Roberts, J.L.; Fogarty, K.V.; Reynolds, K.A.; Jonas, J.M.; Hudson, J.I. Memantine in the treatment of binge eating disorder: an open-label, prospective trial. Int J Eat Disord 2008, 41, 520-526. [CrossRef]
- Petersen, J.; Ludwig, M.Q.; Juozaityte, V.; Ranea-Robles, P.; Svendsen, C.; Hwang, E.; Kristensen, A.W.; Fadahunsi, N.; Lund, J.; Breum, A.W.; et al. GLP-1-directed NMDA receptor antagonism for obesity treatment. Nature 2024, 629, 1133-1141. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




