Submitted:
12 July 2025
Posted:
14 July 2025
You are already at the latest version
Abstract
Keywords:
1. The Acquisition of Current Scientific Knowledge in Biomedicine
2. The Principles Behind It
2. Direct Experimental Method
3. Indirect Algorithmic Analysis
4. The Role of the Experimental Method in the Era of Big Data
5. Dubious Interpretations in Literature: Examples of Failures Because of Overreliance on Computational Predictions
6. Achieving Reliable Scientific Knowledge Through Balanced Approaches
7. Computer and Automatic Methods in Biology
8. The Contribution of AI
9. How to Mitigate Pollution of Scientific Knowledge and Enhance the Effectiveness of Computational Algorithms
- Replication of experiments: Repeating studies under varying conditions to confirm findings.
- Use of Adequate Controls: Ensuring experimental designs account for confounding factors.
- Transparency: Sharing raw data, protocols, and analysis methods openly for peer validation.
- Promotion of Data Sharing: Encouraging researchers to deposit datasets in accessible repositories facilitates cross-verification.
- Role of Algorithms in Interaction Networks: Computational algorithms are central to constructing and refining interactomes and comprehensive maps of molecular interactions. We can evaluate their performance based on several key criteria:
- Accuracy: Correctly predicting true biological interactions while minimizing false positives [73].
- Computational Speed: Rapid processing is vital given the vast size of biological datasets.
- Scalability: Algorithms should handle increasing data volumes without performance loss [74].
- Resource Efficiency: Optimizing memory and processing power to facilitate workable analysis [75].
- Robustness: Maintaining performance despite noisy, incomplete, or conflicting data [76].
10. Practical Approaches
- Dealing with Noisy Data: Biological datasets often contain experimental artifacts or context-dependent interactions; algorithms must distinguish genuine signals.
- Handling Incomplete Data: We do not know all interactions. Algorithms should predict missing links without overfitting known data.
- Resource Management: Developers must optimize advanced algorithms, such as graph-based machine learning models, for efficient operation on multi-terabyte datasets.
10. Advances and Future Directions
11. The Need for a Control Tool
11.1. Interactomics as a Tool for Controlling Metabolic Analyses
12. Recommendations and Practical Frameworks for Ensuring Balance
- Prioritize High-Confidence Predictions for Validation: Use computational scoring systems (like confidence scores in STRING) to select the most promising hypotheses for experimental validation. Focus on interactions or targets with high confidence, reducing costs associated with testing less likely candidates.
- Sequential Validation Strategy:
- Step 1: Use in silico methods to generate hypotheses [97].
- Step 2: Apply secondary computational filters (e.g., cross-validation across datasets, orthogonal methods) to refine predictions [98].
- Step 3: Experimentally validate only the top-tier predictions, such as through targeted assays or minimal confirmatory experiments.
- 3.
- Use Collaborations and Shared Resources: Partner with institutions or consortia that can offer access to specialized experimental platforms or datasets, which help to lessen individual resource burdens. Taking part in shared repositories or consortium initiatives can make validation more cost-effective.
- 4.
- Implement a Hypothesis-Driven Approach [99]: Restrict computational analysis to well-defined hypotheses rather than broad exploratory searches. This focused approach minimizes unnecessary experiments and maximizes resource efficiency.
- 5.
- Emphasize Open Science and Data Sharing: Share datasets and validation results transparently. This prevents redundant efforts, helps refine models collectively, and speeds up validation efforts [100].
- 6.
- Use Computational Validation as a Filter, Not an Ultimate Authority: Recognize computational predictions as hypotheses rather than conclusions. Establish a workflow incorporating initial predictions, systematic prioritization, and targeted experimental validation. This layered approach optimizes resource expenditure.
- 7.
- Incorporate training on data interpretation and biases. Researchers should receive training on data interpretation and biases. To avoid misinterpreting computational results, they must understand algorithms' limitations and potential biases and interpret findings critically.
13. Interactomics as a Criterion for Scientific Validation
- Identify the target protein (e.g., VCAM-1) and pathological/biological context (e.g., vascular inflammation). Determine if the proposed function is observed or merely hypothesized.
14. The Principle of Falsification
References
- Yang, X., Huang, K., Yang, D., Zhao, W., Zhou, X. "Biomedical big data technologies, applications, and challenges for precision medicine: a review." Global Challenges 8.1 (2024): 2300163. [CrossRef]
- Jafari, M., Guan, Y., Wedge, D.C., Ansari-Pour, N. "Re-evaluating experimental validation in the Big Data Era: a conceptual argument." Genome Biology 22 (2021): 1-6. [CrossRef]
- Iqbal, R., Doctor, F., More, B., Mahmud, S., & Yousuf, U. (2020). Big Data analytics and Computational Intelligence for Cyber–Physical Systems: Recent trends and state of the art applications. Future Generation Computer Systems, 105, 766-778. [CrossRef]
- Martin-Sanchez, F., Iakovidis, I., Nørager, S., Maojo, V., de Groen, P., Van der Lei, J., Jones, T., Abraham-Fuchs, K., Apweiler, R., Babic, A., et al. (2004). Synergy between medical informatics and bioinformatics: facilitating genomic medicine for future health care. Journal of biomedical informatics, 37(1), 30-42. [CrossRef]
- Preim, Bernhard, and Charl P. Botha. Visual computing for medicine: theory, algorithms, and applications. Second Edition (2014), Elsevier. ISBN: 978-0-12-415873-3.
- Altaf-Ul-Amin, M., Afendi, F. M., Kiboi, S. K., & Kanaya, S. (2014). Systems biology in the context of big data and networks. BioMed research international, 2014(1), 428570. [CrossRef]
- Kirschner, Marc W. "The meaning of systems biology." Cell 121.4 (2005): 503-504. [CrossRef]
- Kaiser, Marie I. Reductive explanation in the biological sciences. (2015) History, Philosophy and Theory of the Life Sciences (HPTL), Cham: Springer International Publishing. [CrossRef]
- Colonna, G. Overcoming Barriers in Cancer Biology Research: Current Limitations and Solutions. Cancers 2025, 17, 2102. [CrossRef]
- Van Segbroeck, S.; De Jong, S.; Nowé, A.; Santos, F.C.; Lenaerts, T. Learning to coordinate in complex networks. Adapt. Behav. (2010), 18, 416–427. [CrossRef]
- Ioannidis, John PA. "Limitations are not properly acknowledged in the scientific literature." Journal of clinical epidemiology 60.4 (2007): 324-329. [CrossRef]
- Strasser, B. J. (2012). Data-driven sciences: From wonder cabinets to electronic databases. Studies in History and Philosophy of Science Part C: Studies in History and Philosophy of Biological and Biomedical Sciences, 43(1), 85-87. [CrossRef]
- Windelband, Wilhelm. "History and natural science." Theory & Psychology 8.1 (1998): 5-22. [CrossRef]
- Stoelinga, M. T., Hobbs, P. V., Mass, C. F., Locatelli, J. D., Colle, B. A., Houze Jr, R. A., Rangno, A.L., Bond, N.A., Smull, B.F., Rasmussen, R.M., et al. (2003). Improvement of microphysical parameterization through observational verification experiment. Bulletin of the American Meteorological Society, 84(12), 1807-1826. [CrossRef]
- McGregor, S. L. (2017). Understanding and evaluating research: A critical guide. Sage Publications.
- Mullins, Simon, and Sean A. Spence. "Re-examining thought insertion: Semi-structured literature review and conceptual analysis." The British Journal of Psychiatry 182.4 (2003): 293-298. [CrossRef]
- Dahdul, Wasila M., et al. "Evolutionary characters, phenotypes and ontologies: curating data from the systematic biology literature." PLoS One 5.5 (2010): e10708. ttps://doi.org/10.1371/journal.pone.0010708.
- Szalay, Alexander, and Jim Gray. "Science in an exponential world." Nature 440.7083 (2006): 413-414. http://research.microsoft.com/towards2020science.
- Salloum, S.A., Al-Emran, M., Monem, A.A., Shaalan, K. (2018). Using Text Mining Techniques for Extracting Information from Research Articles. In: Shaalan, K., Hassanien, A., Tolba, F. (eds) Intelligent Natural Language Processing: Trends and Applications. Studies in Computational Intelligence, vol 740. Springer, Cham. [CrossRef]
- Leonelli, Sabina, "Scientific Research and Big Data", The Stanford Encyclopedia of Philosophy (Summer 2020 Edition), Edward N. Zalta (ed.). https://plato.stanford.edu/archives/sum2020/entries/science-big-data/.
- Capurro, Rafael. "Past, present, and future of the concept of information." TripleC: communication, capitalism & critique. open access journal for a global sustainable information society 7.2 (2009): 125-141. [CrossRef]
- Mayernik, Matthew S. "Metadata." KO Knowledge Organization 47.8 (2021): 696-713. [CrossRef]
- Buneman, P., Cheney, J., Tan, W. C., & Vansummeren, S. (2008, June). Curated databases. In Proceedings of the twenty-seventh ACM SIGMOD-SIGACT-SIGART symposium on Principles of database systems (pp. 1-12). [CrossRef]
- Lemson, Joris, and Willem P. de Boode. "Importance of using the correct statistics." Archives of Disease in Childhood. Fetal and Neonatal Edition (2024). [CrossRef]
- Zhang, Peng, and Wanhua Su. "Statistical inference on recall, precision and average precision under random selection." 2012 9th international conference on fuzzy systems and knowledge discovery. IEEE, 2012. pp. 1348-1352. [CrossRef]
- Haase, Marco, Yvonne Seiler Zimmermann, and Heinz Zimmermann. "The impact of speculation on commodity futures markets–A review of the findings of 100 empirical studies." Journal of Commodity Markets 3.1 (2016): 1-15. [CrossRef]
- Zheng, H., Porebski, P. J., Grabowski, M., Cooper, D. R., & Minor, W. (2017). Databases, repositories, and other data resources in structural biology. Protein Crystallography: Methods and Protocols, 643-665. Methods in Molecular Biology ((MIMB, volume 1607)). [CrossRef]
- Storey, M. A. (2006). Theories, tools and research methods in program comprehension: past, present and future. Software Quality Journal, 14, 187-208. [CrossRef]
- Hepburn, Brian and Hanne Andersen, "Scientific Method", The Stanford Encyclopedia of Philosophy (Summer 2021 Edition), Edward N. Zalta (ed.). ISSN 1095-5054 https://plato.stanford.edu/archives/sum2021/entries/scientific-method/.
- Keskin, O., Gursoy, A., Ma, B., & Nussinov, R. (2008). Principles of protein− protein interactions: what are the preferred ways for proteins to interact? Chemical reviews, 108(4), 1225-1244. [CrossRef]
- Ramazi, S., & Zahiri, J. (2021). Post-translational modifications in proteins: resources, tools and prediction methods. Database, 2021, baab012. [CrossRef]
- Gordon, D. E., Hiatt, J., Bouhaddou, M., Rezelj, V. V., Ulferts, S., Braberg, H., Jureka, A., Obernier, K., Guo, J.Z., Batra, J., et al. (2020). Comparative host-coronavirus protein interaction networks reveal pan-viral disease mechanisms. Science, 370(6521), eabe9403. [CrossRef]
- Tolani, P., Gupta, S., Yadav, K., Aggarwal, S., & Yadav, A. K. (2021). Big data, integrative omics and network biology. Advances in protein chemistry and structural biology, 127, 127-160. [CrossRef]
- Lecca, P. (2021). Machine learning for causal inference in biological networks: perspectives of this challenge. Frontiers in Bioinformatics, 1, 746712. [CrossRef]
- Koyutürk, M. (2010). Algorithmic and analytical methods in network biology. Wiley Interdisciplinary Reviews: Systems Biology and Medicine, 2(3), 277-292. [CrossRef]
- Arab, Ali. "Knowledge discovery: from correlation to causation." (2024). (Thesis) Ph.D., Simon Fraser University, Computing Science Theses. Identifier etd23245.
- Ratner, Bruce. Statistical and machine-learning data mining: Techniques for better predictive modeling and analysis of big data. Chapman and Hall/CRC, 2017. [CrossRef]
- Miteva, Yana V., Hanna G. Budayeva, and Ileana M. Cristea. "Proteomics-based methods for discovery, quantification, and validation of protein–protein interactions." Analytical chemistry 85.2 (2013): 749-768. [CrossRef]
- Cardelli, L. (2005). Abstract machines of systems biology. In Transactions on Computational Systems Biology III (pp. 145-168). Berlin, Heidelberg: Springer Berlin Heidelberg. [CrossRef]
- Sarker, I. H. (2021). Machine learning: Algorithms, real-world applications and research directions. SN computer science, 2(3), 160. [CrossRef]
- Agamah, F. E., Mazandu, G. K., Hassan, R., Bope, C. D., Thomford, N. E., Ghansah, A., & Chimusa, E. R. (2020). Computational/in silico methods in drug target and lead prediction. Briefings in bioinformatics, 21(5), 1663-1675. [CrossRef]
- Messina, F., Giombini, E., Agrati, C., Vairo, F., Ascoli Bartoli, T., Al Moghazi, S., Piacentini, M., Locatelli, F., Kobinger, G., Maeurer, M., et al. (2020). COVID-19: viral–host interactome analyzed by network based-approach model to study pathogenesis of SARS-CoV-2 infection. Journal of translational medicine, 18, 1-10. [CrossRef]
- Gaudelet, T. (2021). Integration of multi-scale protein interactions for biomedical data analysis (Doctoral dissertation), UCL (University College, London)). ttps://discovery.ucl.ac.uk/id/eprint/10125012.
- Röttjers, Lisa, and Karoline Faust. "From hairballs to hypotheses–biological insights from microbial networks." FEMS microbiology reviews 42.6 (2018): 761-780. [CrossRef]
- Ioannidis, John PA. "Biomarker failures." Clinical chemistry 59.1 (2013): 202-204. [CrossRef]
- Goossens, N., Nakagawa, S., Sun, X., & Hoshida, Y. (2015). Cancer biomarker discovery and validation. Translational cancer research, 4(3), 256. [CrossRef]
- Dimitrakopoulos, Christos M., and Niko Beerenwinkel. "Computational approaches for the identification of cancer genes and pathways." Wiley Interdisciplinary Reviews: Systems Biology and Medicine 9.1 (2017): e1364. [CrossRef]
- Rakedzon, S., Khoury, Y., Rozenberg, G., & Neuberger, A. (2020). Hydroxychloroquine and coronavirus disease 2019: A systematic review of a scientific failure. Rambam Maimonides medical journal, 11(3), e0025.. [CrossRef]
- O'malley, Maureen A., and Orkun S. Soyer. "The roles of integration in molecular systems biology." Studies in History and Philosophy of Science Part C: Studies in History and Philosophy of Biological and Biomedical Sciences 43.1 (2012): 58-68. [CrossRef]
- Luo, W., Phung, D., Tran, T., Gupta, S., Rana, S., Karmakar, C., Shilton, A., Yearwood, J., Dimitrova, N., Ho, T.B., et al. (2016). Guidelines for developing and reporting machine learning predictive models in biomedical research: a multidisciplinary view. Journal of medical Internet research, 18(12), e323. [CrossRef]
- Jaeger, J. (2017). The Importance of Being Dynamic: Systems Biology Beyond the Hairball. In: Green, S. (eds) Philosophy of Systems Biology. History, Philosophy and Theory of the Life Sciences, vol 20. Springer, Cham. [CrossRef]
- Brohee, Sylvain, and Jacques Van Helden. "Evaluation of clustering algorithms for protein-protein interaction networks." BMC bioinformatics 7 (2006): 1-19. [CrossRef]
- Sornette, D., Davis, A. B., Ide, K., Vixie, K. R., Pisarenko, V., & Kamm, J. R. (2007). Algorithm for model validation: Theory and applications. Proceedings of the National Academy of Sciences, 104(16), 6562-6567. [CrossRef]
- Dougherty, Edward R., and Ulisses Braga-Neto. "Epistemology of computational biology: mathematical models and experimental prediction as the basis of their validity." Journal of Biological Systems 14.01 (2006): 65-90. [CrossRef]
- Ahady Dolatsara, H., Chen, Y. J., Leonard, R. D., Megahed, F. M., & Jones-Farmer, L. A. (2023). Explaining predictive model performance: An experimental study of data preparation and model choice. Big Data, 11(3), 199-214. [CrossRef]
- Kairys, V., Baranauskiene, L., Kazlauskiene, M., Matulis, D., & Kazlauskas, E. (2019). Binding affinity in drug design: experimental and computational techniques. Expert opinion on drug discovery, 14(8), 755-768. [CrossRef]
- Kopac T. Leveraging Artificial Intelligence and Machine Learning for Characterizing Protein Corona, Nanobiological Interactions, and Advancing Drug Discovery. Bioengineering (Basel). 2025 Mar 18;12(3):312. [CrossRef] [PubMed] [PubMed Central]
- Refaeilzadeh, P., Tang, L., & Liu, H. (2009). Cross-validation. In Encyclopedia of database systems (pp. 532-538). Springer, Boston, MA. [CrossRef]
- Adadi, A. (2021). A survey on data-efficient algorithms in big data era. Journal of Big Data, 8(1), 24. [CrossRef]
- Mahmud, M., Kaiser, M. S., McGinnity, T. M., & Hussain, A. (2021). Deep learning in mining biological data. Cognitive computation, 13(1), 1-33. [CrossRef]
- Altman, R. B., Bergman, C. M., Blake, J., Blaschke, C., Cohen, A., Gannon, F., ... & Valencia, A. (2008). Text mining for biology-the way forward: opinions from leading scientists. Genome biology, 9, 1-15. [CrossRef]
- Agarwal, S., Godbole, S., Punjani, D., & Roy, S. (2007, October). How much noise is too much: A study in automatic text classification. In Seventh IEEE International Conference on Data Mining (ICDM 2007) (pp. 3-12). IEEE. [CrossRef]
- Kar, A. K., & Dwivedi, Y. K. (2020). Theory building with big data-driven research–Moving away from the "What" towards the "Why". International Journal of Information Management, 54, 102205. [CrossRef]
- Johanson, A., & Hasselbring, W. (2018). Software engineering for computational science: Past, present, future. Computing in Science & Engineering, 20(2), 90-109. [CrossRef]
- Tracy, Sarah J. Qualitative research methods: Collecting evidence, crafting analysis, communicating impact. Third Edition. John Wiley & Sons, 2024. ePUB ISBN: 9781119988670.
- Aldoseri, A., Al-Khalifa, K. N., & Hamouda, A. M. (2023). Re-thinking data strategy and integration for artificial intelligence: concepts, opportunities, and challenges. Applied Sciences, 13(12), 7082. [CrossRef]
- Chelly Dagdia, Z., Avdeyev, P. & Bayzid, M.S. Biological computation and computational biology: survey, challenges, and discussion. Artif Intell Rev 54, 4169–4235 (2021). [CrossRef]
- Kaur, D., Uslu, S., Rittichier, K. J., & Durresi, A. (2022). Trustworthy artificial intelligence: a review. ACM computing surveys (CSUR), 55(2), 1-38. [CrossRef]
- Nickles, Thomas J. "Knowledge Pollution." NSF Award 95.9530174 (1996): 30174. https://www.nsf.gov/awardsearch/showAward?AWD_ID=9530174.
- Elouataoui, W. (2024). AI-Driven frameworks for enhancing data quality in big data ecosystems: Error_detection, correction, and metadata integration. arXiv preprint arXiv:2405.03870. [CrossRef]
- Samariya, D., & Thakkar, A. (2023). A comprehensive survey of anomaly detection algorithms. Annals of Data Science, 10(3), 829-850. [CrossRef]
- Paramesha, M., Rane, N. L., & Rane, J. (2024). Big data analytics, artificial intelligence, machine learning, internet of things, and blockchain for enhanced business intelligence. Partners Universal Multidisciplinary Research Journal, 1(2), 110-133. [CrossRef]
- Forstmeier, W., Wagenmakers, E. J., & Parker, T. H. (2017). Detecting and avoiding likely false-positive findings–a practical guide. Biological Reviews, 92(4), 1941-1968. [CrossRef]
- E. M. Hassib, A. I. El-Desouky, E. -S. M. El-Kenawy and S. M. El-Ghamrawy, "An Imbalanced Big Data Mining Framework for Improving Optimization Algorithms Performance," in IEEE Access, vol. 7, pp. 170774-170795, 2019. [CrossRef]
- Chedid, W., Yu, C., & Lee, B. (2005). Power analysis and optimization techniques for energy efficient computer systems. Advances in Computers, 63, 129-164. [CrossRef]
- Hamidzadeh, J., & Moradi, M. (2020). Enhancing data analysis: uncertainty-resistance method for handling incomplete data. Applied Intelligence, 50(1), 74-86. [CrossRef]
- Jiao, Y., & Du, P. (2016). Performance measures in evaluating machine learning based bioinformatics predictors for classifications. Quantitative Biology, 4, 320-330. [CrossRef]
- Lei, Jing. "Cross-validation with confidence." Journal of the American Statistical Association 115.532 (2020): 1978-1997. [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Nastou, K.C.; Lyon, D.; Kirsch, R.; Pyysalo, S.; Doncheva, N.T.; Legeay, M.; Fang, T.; Bork, P.; et al. The STRING database in 2021: Customizable protein–protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 2020, 49, D605–D612, Erratum in: Nucleic Acids Res. 2021, 49, 10800.. [CrossRef].
- Szklarczyk, D.; Kirsch, R.; Koutrouli, M.; Nastou, K.; Mehryary, F.; Hachilif, R.; Gable, A.L.; Fang, T.; Doncheva, N.T.; Pyysalo, S.; et al. The STRING database in 2023: Protein–protein association networks and functional enrichment analyses for any sequenced genome of interest. Nucleic Acids Res. 2022, 51, D638–D646. [CrossRef] [PubMed].
- Lin, J. S., & Lai, E. M. (2017). Protein–protein interactions: co-immunoprecipitation. Bacterial protein secretion systems: Methods and protocols, 211-219. [CrossRef]
- Galas, D. J., Nykter, M., Carter, G. W., Price, N. D., & Shmulevich, I. (2010). Biological information as set-based complexity. IEEE Transactions on Information Theory, 56(2), 667-677. [CrossRef]
- Kholodenko, B., Yaffe, M. B., & Kolch, W. (2012). Computational approaches for analyzing information flow in biological networks. Science signaling, 5(220), re1-re1. [CrossRef]
- Li, Y., Wu, F. X., & Ngom, A. (2018). A review on machine learning principles for multi-view biological data integration. Briefings in bioinformatics, 19(2), 325-340. [CrossRef]
- Sokouti, B., & Amjad, E. (2025). Validation strategies in systems biology research. In Systems Biology and In-Depth Applications for Unlocking Diseases (pp. 183-190). Academic Press. [CrossRef]
- Boué, S., Byrne, M., Hayes, A. W., Hoeng, J., & Peitsch, M. C. (2018). Embracing transparency through data sharing. International Journal of Toxicology, 37(6), 466-471. [CrossRef]
- Roberts, M. K., Fisher, D. M., Parker, L. E., Darnell, D., Sugarman, J., Carrithers, J., Weinfurt, K., Jurkovich, G., Zatzick, D. (2020). Ethical and regulatory concerns in pragmatic clinical trial monitoring and oversight. Ethics & Human Research, 42(5), 29-37. [CrossRef]
- Cannataro, Mario, Pietro H. Guzzi, and Pierangelo Veltri. "Protein-to-protein interactions: Technologies, databases, and algorithms." ACM Computing Surveys (CSUR) 43.1 (2010): 1-36. [CrossRef]
- Leung, M. K., Delong, A., Alipanahi, B., & Frey, B. J. (2015). Machine learning in genomic medicine: a review of computational problems and data sets. Proceedings of the IEEE, 104(1), 176-197. [CrossRef]
- Lualdi, Marta, and Mauro Fasano. "Statistical analysis of proteomics data: a review on feature selection." Journal of proteomics 198 (2019): 18-26. [CrossRef]
- Ferrell, M., Wang, Z., Anderson, J.T. et al. A terminal metabolite of niacin promotes vascular inflammation and contributes to cardiovascular disease risk. Nat Med 30, 424–434 (2024). [CrossRef]
- Karunakaran, Kalyani Bindu. Interactome-based framework to translate disease genetic data into biological and clinical insights. Diss. University of Reading, UK, 2024. School of Chemistry, Food & Pharmacy. [CrossRef]
- Singh, Vikram, and Vikram Singh. "Inferring interaction networks from transcriptomic data: methods and applications." Transcriptome Data Analysis. New York, NY: Springer US, 2024. 11-37. [CrossRef]
- MacKenzie, Donald. Mechanizing proof: computing, risk, and trust. MIT Press, London, 2004. ISBN 0-262-13393-8.
- Mering, C. V., Huynen, M., Jaeggi, D., Schmidt, S., Bork, P., & Snel, B. (2003). STRING: a database of predicted functional associations between proteins. Nucleic acids research, 31(1), 258-261. [CrossRef]
- Colonna, G. Understanding the SARS-CoV-2–Human Liver Interactome Using a Comprehensive Analysis of the Individual Virus–Host Interactions. Livers 2024, 4, 209–239. [CrossRef]
- Klinke, David J. "In silico model-based inference: A contemporary approach for hypothesis testing in network biology." Biotechnology progress 30.6 (2014): 1247-1261. [CrossRef]
- Roberts, D. R., Bahn, V., Ciuti, S., Boyce, M. S., Elith, J., Guillera-Arroita, G., Hauenstein, S., Lahoz-Monfort, J.J., Schroeder, B., Thuiller, W., et al. (2017). Cross-validation strategies for data with temporal, spatial, hierarchical, or phylogenetic structure. Ecography, 40(8), 913-929. [CrossRef]
- Kalinichenko, L. A., Kovalev, D. Y. E., Kovaleva, D. A., & Malkov, O. Y. E. (2015). Methods and tools for hypothesis-driven research support: A survey. Informatics and its Applications, 9(1), 28-54. [CrossRef]
- Abele-Brehm, A. E., Gollwitzer, M., Steinberg, U., & Schönbrodt, F. D. (2019). Attitudes toward open science and public data sharing. Social Psychology. Vol 50, No4, Res. Report. [CrossRef]
- Campaner, Raffaella. "Complexity and Integration." Explaining Disease: Philosophical Reflections on Medical Research and Clinical Practice. European Studies in Philosophy of Science. Cham: Springer International Publishing, 2022. 65-88. [CrossRef]
- Sharma, A., Colonna, G. System-Wide Pollution of Biomedical Data: Consequence of the Search for Hub Genes of Hepatocellular Carcinoma Without Spatiotemporal Consideration. Mol Diagn Ther 25, 9–27 (2021). [CrossRef]
- Karl Popper, Conjectures and Refutations, London: Routledge and Keagan Paul, 1963, pp. 33-39; from Theodore Schick, ed., Readings in the Philosophy of Science, Mountain View, CA: Mayfield Publishing Company, 2000, pp. 9-13. Excerpt from Royal Institute of Technology (KTH), Sweden. https://www.kth.se/social/files/57da705ef276540c0f789308/KPopper_Falsification.pdf.
- Popper, Karl. "Science: Conjectures and refutations." Arguing about Science. Routledge, 2012. 15-43. Taylor & Francis Group. [CrossRef]
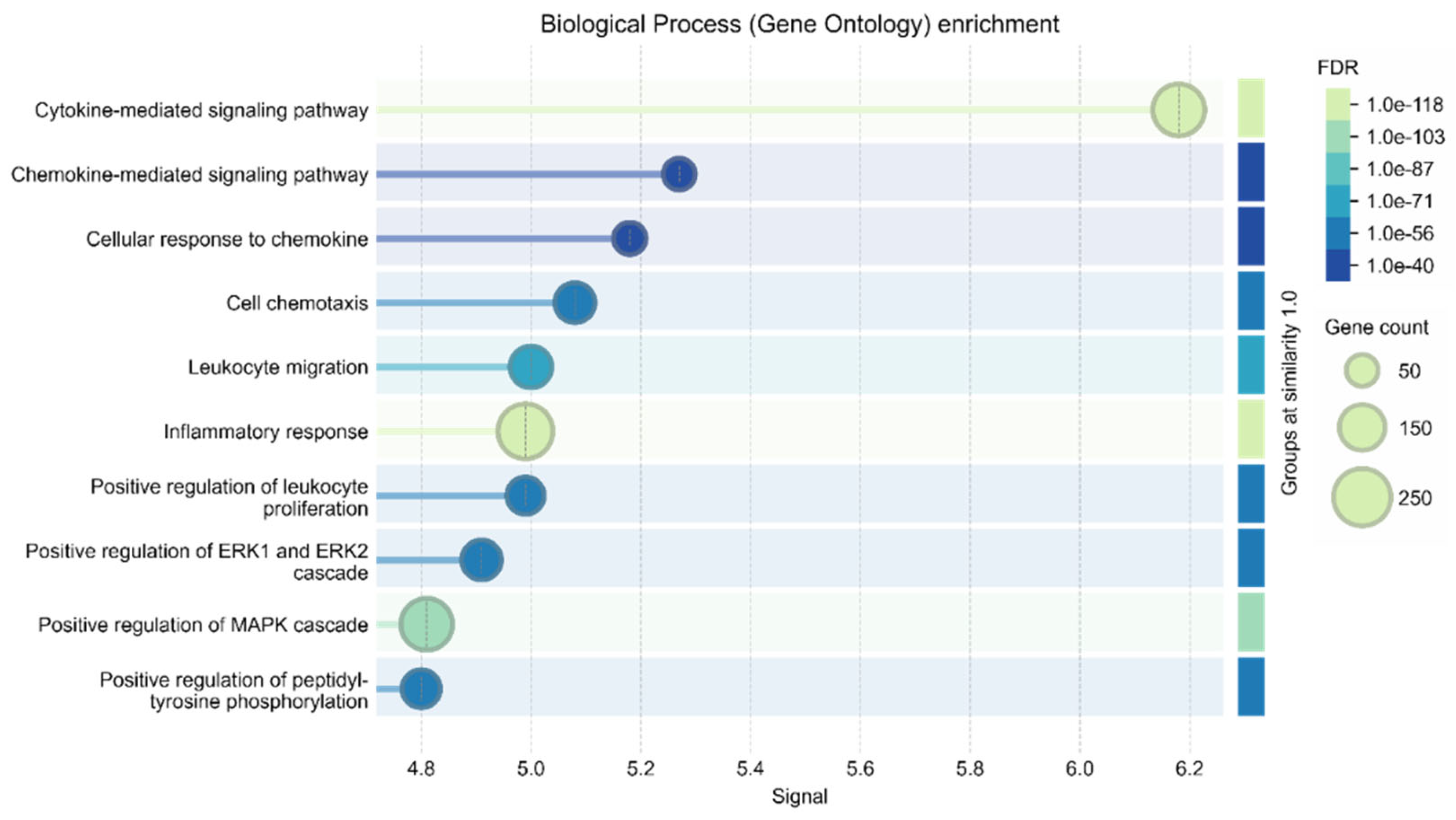

| Confidence score | Experimental validation | % |
| 0.900 | 0 | 0 |
| 0.800 | 0 | 0 |
| 0.700 | 0 | 0 |
| 0.600 | 0 | 0 |
| 0.500 | 0 | 0 |
| 0.400 | 2 | 0.42 |
| 0.300 | 7 | 1.48 |
| 0.200 | 1 | 0.02 |
| 0.100 | 81 | 17.19 |
| 0<x<0.1 | 108 | 22.92 |
| 0 | 272 | 57.74 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




