Submitted:
12 July 2025
Posted:
14 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
| Species | AFDW (% DW) |
Protein (% AFDW) |
Lipid (% AFDW) |
Carbohydrate (% AFDW) |
| Benthosema suborbitale | 94.3 | 55.4 | 22.8 | 1.0 |
| Ceratoscopelus townsendi | 86.7 | 48.9 | 13.6 | 1.2 |
| Hygophum taaningi | 80.5 | 54.0 | 14.5 | 1.0 |
| Myctophum selenops | 83.9 | 55.4 | 13.3 | 1.3 |
| Notolychnus valdiviae | 83.5 | 58.4 | 11.7 | 0.8 |
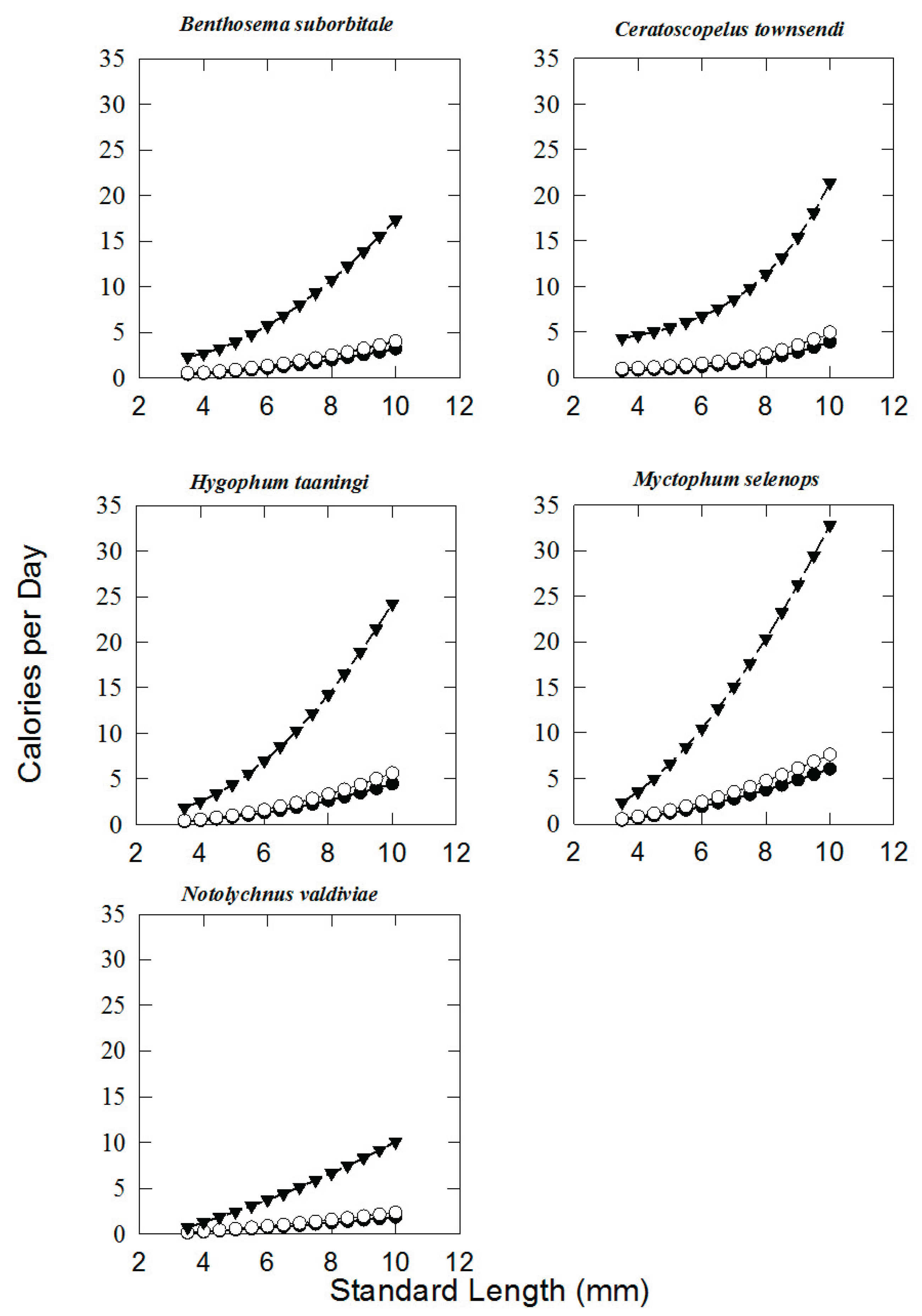
| SL (mm) |
W µgDW |
R µg O2 h –1 |
m x 10-6cal µg –1 h –1 |
Qm cal day-1 |
Qg cal day-1 |
Qm + Qg cal day-1 |
AvgQc cal day-1 |
MinQc cal day-1 |
MaxQc cal day-1 |
AvgPrey no. day-1 |
MinPrey no. day-1 |
MaxPrey no. day-1 |
| 3.5 | 80 | 0.70 | 40 | 0.13 | 0.27 | 0.39 | 0.54 | 0.42 | 2.30 | 7 | 5 | 29 |
| 4.0 | 130 | 1.05 | 37 | 0.19 | 0.27 | 0.45 | 0.62 | 0.49 | 2.67 | 8 | 6 | 34 |
| 4.5 | 210 | 1.56 | 35 | 0.28 | 0.27 | 0.55 | 0.75 | 0.60 | 3.22 | 9 | 8 | 41 |
| 5.0 | 320 | 2.23 | 32 | 0.40 | 0.27 | 0.67 | 0.91 | 0.73 | 3.92 | 12 | 9 | 50 |
| 5.5 | 460 | 3.02 | 30 | 0.55 | 0.27 | 0.81 | 1.11 | 0.88 | 4.77 | 9 | 7 | 60 |
| 6.0 | 630 | 3.93 | 29 | 0.71 | 0.27 | 0.97 | 1.34 | 1.06 | 5.73 | 10 | 8 | 45 |
| 6.5 | 830 | 4.95 | 28 | 0.89 | 0.27 | 1.16 | 1.59 | 1.26 | 6.82 | 12 | 10 | 53 |
| 7.0 | 1060 | 6.08 | 27 | 1.10 | 0.27 | 1.36 | 1.87 | 1.48 | 8.02 | 15 | 12 | 62 |
| 7.5 | 1320 | 7.31 | 26 | 1.32 | 0.27 | 1.58 | 2.17 | 1.72 | 9.32 | 17 | 13 | 72 |
| 8.0 | 1610 | 8.63 | 25 | 1.56 | 0.27 | 1.82 | 2.50 | 1.98 | 10.72 | 19 | 15 | 80 |
| 8.5 | 1930 | 10.04 | 24 | 1.81 | 0.27 | 2.08 | 2.85 | 2.26 | 12.23 | 21 | 17 | 91 |
| 9.0 | 2280 | 11.55 | 23 | 2.09 | 0.27 | 2.35 | 3.22 | 2.56 | 13.83 | 24 | 19 | 103 |
| 9.5 | 2660 | 13.14 | 23 | 2.37 | 0.27 | 2.64 | 3.61 | 2.87 | 15.52 | 27 | 21 | 115 |
| 10.0 | 3070 | 14.82 | 22 | 2.68 | 0.27 | 2.94 | 4.03 | 3.20 | 17.30 | 30 | 24 | 129 |
| SL (mm) |
W µgDW |
R µg O2 h –1 |
m x 10-6 cal µg –1 h –1 |
Qm cal day-1 |
Qg cal day-1 |
Qm + Qg cal day-1 |
AvgQc cal day-1 |
MinQc cal day-1 |
MaxQc cal day-1 |
AvgPrey no. day-1 |
MinPrey no. day-1 |
MaxPrey no. day-1 |
| 3.5 | 199 | 1.36 | 32 | 0.24 | 0.49 | 0.74 | 1.01 | 0.80 | 4.34 | 15 | 12 | 64 |
| 4.0 | 243 | 1.65 | 31 | 0.30 | 0.49 | 0.79 | 1.08 | 0.86 | 4.65 | 16 | 13 | 68 |
| 4.5 | 297 | 2.01 | 31 | 0.36 | 0.49 | 0.86 | 1.17 | 0.93 | 5.03 | 17 | 14 | 74 |
| 5.0 | 363 | 2.44 | 31 | 0.44 | 0.49 | 0.93 | 1.28 | 1.02 | 5.49 | 21 | 16 | 89 |
| 5.5 | 444 | 2.97 | 31 | 0.54 | 0.49 | 1.03 | 1.41 | 1.12 | 6.06 | 23 | 18 | 98 |
| 6.0 | 543 | 3.62 | 31 | 0.65 | 0.49 | 1.15 | 1.57 | 1.25 | 6.74 | 25 | 20 | 109 |
| 6.5 | 664 | 4.40 | 31 | 0.79 | 0.49 | 1.29 | 1.76 | 1.40 | 7.58 | 29 | 23 | 123 |
| 7.0 | 811 | 5.36 | 31 | 0.97 | 0.49 | 1.46 | 2.00 | 1.59 | 8.59 | 32 | 26 | 139 |
| 7.5 | 992 | 6.52 | 30 | 1.18 | 0.49 | 1.67 | 2.29 | 1.82 | 9.82 | 37 | 29 | 159 |
| 8.0 | 1213 | 7.93 | 30 | 1.43 | 0.49 | 1.93 | 2.64 | 2.09 | 11.32 | 42 | 33 | 178 |
| 8.5 | 1483 | 9.65 | 30 | 1.74 | 0.49 | 2.24 | 3.06 | 2.43 | 13.15 | 48 | 38 | 207 |
| 9.0 | 1813 | 11.74 | 30 | 2.12 | 0.49 | 2.61 | 3.58 | 2.84 | 15.38 | 56 | 45 | 242 |
| 9.5 | 2216 | 14.29 | 30 | 2.58 | 0.49 | 3.07 | 4.21 | 3.34 | 18.08 | 66 | 53 | 285 |
| 10.0 | 2709 | 17.39 | 30 | 3.14 | 0.49 | 3.63 | 4.98 | 3.95 | 21.37 | 78 | 62 | 336 |
| SL (mm) |
W µgDW |
R µg O2 h –1 |
m x 10-6 cal µg –1 h –1 |
Qm cal day-1 |
Qg cal day-1 |
Qm + Qg cal day-1 |
AvgQc cal day-1 |
MinQc cal day-1 |
MaxQc cal day-1 |
| 3.5 | 100 | 1.09 | 51 | 0.20 | 0.12 | 0.32 | 0.43 | 0.34 | 1.86 |
| 4.0 | 160 | 1.70 | 49 | 0.31 | 0.12 | 0.43 | 0.58 | 0.46 | 2.51 |
| 4.5 | 240 | 2.49 | 48 | 0.45 | 0.12 | 0.57 | 0.78 | 0.62 | 3.35 |
| 5.0 | 340 | 3.46 | 47 | 0.62 | 0.12 | 0.74 | 1.02 | 0.81 | 4.37 |
| 5.5 | 460 | 4.60 | 46 | 0.83 | 0.12 | 0.95 | 1.30 | 1.03 | 5.58 |
| 6.0 | 600 | 5.91 | 46 | 1.07 | 0.12 | 1.19 | 1.62 | 1.29 | 6.97 |
| 6.5 | 760 | 7.38 | 45 | 1.33 | 0.12 | 1.45 | 1.99 | 1.58 | 8.54 |
| 7.0 | 940 | 9.01 | 44 | 1.63 | 0.12 | 1.75 | 2.39 | 1.90 | 10.27 |
| 7.5 | 1140 | 10.81 | 44 | 1.95 | 0.12 | 2.07 | 2.84 | 2.25 | 12.18 |
| 8.0 | 1360 | 12.76 | 43 | 2.30 | 0.12 | 2.42 | 3.32 | 2.63 | 14.25 |
| 8.5 | 1600 | 14.87 | 43 | 2.69 | 0.12 | 2.80 | 3.84 | 3.05 | 16.50 |
| 9.0 | 1860 | 17.14 | 43 | 3.09 | 0.12 | 3.21 | 4.40 | 3.49 | 18.90 |
| 9.5 | 2140 | 19.55 | 42 | 3.53 | 0.12 | 3.65 | 5.00 | 3.97 | 21.47 |
| 10.0 | 2440 | 22.13 | 42 | 4.00 | 0.12 | 4.11 | 5.64 | 4.47 | 24.20 |
| SL (mm) |
W µgDW |
R µg O2 h –1 |
m x 10-6 cal µg –1 h –1 | Qm cal day-1 |
Qg cal day-1 |
Qm + Qg cal day-1 |
AvgQc cal day-1 |
MinQc cal day-1 |
MaxQc cal day-1 |
AvgPrey no. day-1 |
MinPrey no. day-1 |
MaxPrey no. day-1 |
| 3.5 | 153 | 1.20 | 36 | 0.27 | 0.14 | 0.40 | 0.55 | 0.44 | 2.36 | 5 | 4 | 22 |
| 4.0 | 300 | 2.11 | 33 | 0.47 | 0.14 | 0.60 | 0.83 | 0.66 | 3.55 | 8 | 6 | 33 |
| 4.5 | 493 | 3.20 | 30 | 0.71 | 0.14 | 0.85 | 1.16 | 0.92 | 4.97 | 11 | 8 | 46 |
| 5.0 | 730 | 4.45 | 28 | 0.99 | 0.14 | 1.12 | 1.54 | 1.22 | 6.61 | 9 | 7 | 40 |
| 5.5 | 1013 | 5.85 | 27 | 1.30 | 0.14 | 1.43 | 1.97 | 1.56 | 8.44 | 12 | 9 | 51 |
| 6.0 | 1340 | 7.40 | 26 | 1.64 | 0.14 | 1.78 | 2.44 | 1.93 | 10.47 | 15 | 12 | 63 |
| 6.5 | 1713 | 9.09 | 25 | 2.02 | 0.14 | 2.15 | 2.95 | 2.34 | 12.67 | 18 | 14 | 76 |
| 7.0 | 2130 | 10.91 | 24 | 2.42 | 0.14 | 2.56 | 3.51 | 2.78 | 15.06 | 21 | 17 | 91 |
| 7.5 | 2593 | 12.86 | 23 | 2.86 | 0.14 | 2.99 | 4.10 | 3.25 | 17.61 | 25 | 20 | 106 |
| 8.0 | 3100 | 14.94 | 22 | 3.32 | 0.14 | 3.46 | 4.73 | 3.76 | 20.33 | 28 | 22 | 120 |
| 8.5 | 3653 | 17.14 | 22 | 3.81 | 0.14 | 3.95 | 5.40 | 4.29 | 23.21 | 32 | 25 | 137 |
| 9.0 | 4250 | 19.47 | 21 | 4.33 | 0.14 | 4.46 | 6.11 | 4.85 | 26.24 | 36 | 29 | 155 |
| 9.5 | 4893 | 21.90 | 21 | 4.87 | 0.14 | 5.00 | 6.85 | 5.44 | 29.43 | 40 | 32 | 173 |
| 10.0 | 5580 | 24.46 | 20 | 5.44 | 0.14 | 5.57 | 7.63 | 6.05 | 32.77 | 45 | 36 | 193 |
| SL (mm) |
W µgDW |
R µg O2 h –1 |
m x 10-6cal µg –1 h –1 |
Qm cal day-1 |
Qg cal day-1 |
Qm + Qg cal day-1 |
AvgQc cal day-1 |
MinQc cal day-1 |
MaxQc cal day-1 |
AvgPrey no. day-1 |
MinPrey no. day-1 |
MaxPrey no. day-1 |
| 3.5 | 20 | 0.22 | 50 | 0.04 | 0.08 | 0.12 | 0.16 | 0.13 | 0.68 | 3 | 2 | 12 |
| 4.0 | 90 | 0.77 | 40 | 0.14 | 0.08 | 0.22 | 0.30 | 0.23 | 1.27 | 5 | 4 | 23 |
| 4.5 | 170 | 1.31 | 36 | 0.24 | 0.08 | 0.31 | 0.43 | 0.34 | 1.85 | 8 | 6 | 33 |
| 5.0 | 260 | 1.87 | 33 | 0.34 | 0.08 | 0.41 | 0.57 | 0.45 | 2.44 | 8 | 7 | 36 |
| 5.5 | 360 | 2.46 | 32 | 0.44 | 0.08 | 0.52 | 0.71 | 0.57 | 3.06 | 10 | 8 | 45 |
| 6.0 | 470 | 3.07 | 30 | 0.56 | 0.08 | 0.63 | 0.87 | 0.69 | 3.72 | 13 | 10 | 54 |
| 6.5 | 590 | 3.72 | 29 | 0.67 | 0.08 | 0.75 | 1.03 | 0.81 | 4.40 | 15 | 12 | 64 |
| 7.0 | 720 | 4.40 | 28 | 0.79 | 0.08 | 0.87 | 1.19 | 0.95 | 5.12 | 17 | 14 | 75 |
| 7.5 | 860 | 5.10 | 27 | 0.92 | 0.08 | 1.00 | 1.37 | 1.08 | 5.87 | 20 | 16 | 86 |
| 8.0 | 1010 | 5.84 | 27 | 1.05 | 0.08 | 1.13 | 1.55 | 1.23 | 6.65 | 20 | 16 | 86 |
| 8.5 | 1170 | 6.60 | 26 | 1.19 | 0.08 | 1.27 | 1.74 | 1.38 | 7.47 | 22 | 18 | 97 |
| 9.0 | 1340 | 7.40 | 26 | 1.34 | 0.08 | 1.41 | 1.94 | 1.54 | 8.31 | 25 | 20 | 108 |
| 9.5 | 1520 | 8.22 | 25 | 1.48 | 0.08 | 1.56 | 2.14 | 1.70 | 9.19 | 28 | 22 | 119 |
| 10.0 | 1710 | 9.08 | 25 | 1.64 | 0.08 | 1.72 | 2.35 | 1.86 | 10.09 | 30 | 24 | 131 |
4. Discussion
Acknowledgements
References
- Gjøsaeter, H.; Kawaguchi, K. A review of the world resources of mesopelagic fishes. FAO Fish Tech Pap 1980, 193, 1–151. [Google Scholar]
- Beamish, R.J.; McFarlane, G.A. The forgotten requirements for age validation in fisheries biology. Trans Am Fish Soc 1983, 112, 735–743. [Google Scholar] [CrossRef]
- Irigoien, X.; Klevjer, T.A.; Røstad, A.; Martinez, U.; Boyra, G.; Acuña, J.L.; Kaartvedt, S. Large mesopelagic fishes biomass and trophic efficiency in the open ocean. Nature communications 2014, 5, 3271. [Google Scholar] [CrossRef] [PubMed]
- Maynard, S.D.; Riggs, F.V.; Walters, J.F. Mesopelagic micronekton in the Hawaiian waters: Faunal composition, standing stock, and diel vertical migration. Fish Bull US 1975, 73, 726–736. [Google Scholar]
- Hulley, P.A. Results of the research cruises of FRV ‘Walther Herwig’ to South America. LVIII. Family Myctophidae (Osteichthyes, Myctophiformes). Archiv. Fisch.Wiss. 1981, 31, 1–300. [Google Scholar]
- Gartner, J.V. Jr.; Hopkins, T.L.; Baird, R.C.; Milliken, D.M. The lanternfishes [Pisces: Myctophidae] of the eastern Gulf of Mexico. Fish Bull US 1987, 85, 81–98. [Google Scholar]
- Beamish, R.; Leask, K.D.; Ivanov, O.A.; Balanov, A.A.; Orlov, A.M.; Sinclair, B. The ecology, distribution, and abundance of midwater fishes of the Subarctic Pacific gyres. Progress in Oceanography 1999, 43, 399–442. [Google Scholar] [CrossRef]
- Catul, V.; Gauns, M.; Karuppasamy, P.K. A review on mesopelagic fishes belonging to family Myctophidae. Reviews in Fish Biology and Fisheries 2011, 21, 339–54. [Google Scholar] [CrossRef]
- Eduardo, L.N.; Bertrand, A.; Mincarone, M.M.; Martins, J.R.; Fredou, T; Assuncao, R.V.; Lima, R.S.; Ménard, F.; Le Loc'h, F.; Lucena-Fredou, F. Distribution, vertical migration, and trophic ecology of lanternfishes (Myctophidae) in the Southwestern Tropical Atlantic. Progress in Oceanography 2021, 199, 102695. [Google Scholar] [CrossRef]
- Hopkins, T.L.; Baird, R.C. Aspects of the feeding ecology of oceanic midwater fishes. In Oceanic sound scattering prediction. Andersen, N.R., Zahuranec, B.J. eds.. Plenum Press. New York USA 1977, 325-360.
- Robinson, B.H.; Bailey, T.G. Sinking rates and dissolution of midwater fish fecal matter. Mar Biol 1981, 65, 135–142. [Google Scholar] [CrossRef]
- Hopkins, T.L.; Sutton, T.; Lancraft, T.M. The trophic structure and predation impact of a low latitude midwater fish assemblage. Prog Oceanogr 1996, 38, 205–239. [Google Scholar] [CrossRef]
- Gartner. J.V. Jr. Life histories of three species of lanternfishes [Pisces: Myctophidae] from the eastern Gulf of Mexico II. Age and growth patterns. Mar Biol 1991, 111, 21–27. [CrossRef]
- Conley, W.J.; Gartner, J.V. Jr. Growth among larvae of lanternfishes [Teleostei: Myctophidae] from the eastern Gulf of Mexico. Bull Mar Sci 2009, 84, 123–135. [Google Scholar]
- Nigmatullin, C.M.; Nesis, K.N.; Arkhipkin, A.I. A review of the biology of the jumbo squid Dosidicus gigas [Cephalopoda: Ommastrephidae]. Fisheries Research 2001, 54, 9–19. [Google Scholar] [CrossRef]
- Alverson, F.G. The food of yellowfin and skipjack tunas in the eastern tropical Pacific. Ocean Bull I-ATTC 1963, 7, 293–396. [Google Scholar]
- Scott, W.B.; Tibbo, S.N. Food and feeding habits of swordfish, Xiphias gladius, in the western North Atlantic. J Fish Res Bd Canada 1968, 25, 903–919. [Google Scholar] [CrossRef]
- Sutton, T.T.; Hopkins, T.L. Trophic ecology of the stomiid (Pisces: Stomiidae) fish assemblage of the eastern Gulf of Mexico: Strategies, selectivity and impact of a top mesopelagic predator group. Mar Biol 1996, 127, 179–192. [Google Scholar] [CrossRef]
- Sanchez-Velasco, L.; Contreras-Arredondo, I.; Esqueda-Escarcega, G. Diet composition of Euthynnus lineatus and Auxis sp. larvae [Pisces: Scombridae] in the Gulf of California. Bull Mar Sci 1999, 65, 687–698. [Google Scholar]
- Harrison, C.S.; Hida, T.; Seki, M.P. Hawaiian seabird feeding ecology. Wildlife Monographs 1983 1, 3–71.
- Springer, A.M.; Piatt, J.F.; Vliet, G.V. Sea birds as proxies of marine habitats and food webs in the western Aleutian Arc. Fisheries Oceanography 1996, 5, 45–55. [Google Scholar] [CrossRef]
- Olsson, O.; North, A.W. Diet of the King Penguin Aptenodytes patagonicus during three summers at South Georgia. Ibis 1997, 139, 504–512. [Google Scholar] [CrossRef]
- Dakwa, F. E.; Ryan, P. G.; Dyer, B. M.; Crawford, R. J. M.; Pistorius, P. A.; Makhado, A. B. [2021]. Long-term variation in the breeding diets of macaroni and eastern rockhopper penguins at Marion Island [1994–2018]. African Journal of Marine Science 2021, 43, 187–199. [Google Scholar] [CrossRef]
- Carey, P.W. Fish prey species of the New Zealand fur seal [Arctocephalus forsteri, Lesson]. New Zealand Journal of Ecology 1992, 41–46. [Google Scholar]
- Klages, N.T.W.; Bester, M.N. Fish prey of fur seals Arctocephalus spp. at subantarctic Marion Island. Mar Biol 1998, 131, 559–566. [Google Scholar] [CrossRef]
- Fitch, J.E.; Brownell Jr, R.L. Fish otoliths in cetacean stomachs and their importance in interpreting feeding habits. J Fish Res Bd Canada 1968, 25, 2561–2574. [Google Scholar] [CrossRef]
- Giménez, J.; Marçalo, A.; García-Polo, M.; García-Barón, I.; Castillo, J. J.; Fernández-Maldonado, C.; de Stephanis, R. Feeding ecology of Mediterranean common dolphins: The importance of mesopelagic fish in the diet of an endangered subpopulation. Marine Mammal Science, 2018, 34, 136–154. [Google Scholar] [CrossRef]
- Pereyra, W.T. , Pearcy, W.G.; Carvey Jr., F.E. Jr. Feeding on mesopelagic fauna, with consideration of the ecological implications. J Fish Res Bd Canada 1969, 26, 2211–2215. [Google Scholar] [CrossRef]
- Freer, J.J.; Tarling, G.A.; Collins, M.A.; Partridge, J.C.; Genner, M.J. 2019 Predicting future distributions of lanternfish, a significant ecological resource within the Southern Ocean. Diversity and Distributions 2019, 25, 1259–1272. [Google Scholar] [CrossRef]
- Shaviklo, A.R. A comprehensive review on animal feed, human food and industrial application of lanternfishes; from prototypes to products. Turkish Journal of Fisheries and Aquatic Sciences 2020, 20, 827–843. [Google Scholar] [CrossRef]
- Kourantidou, M.; Jin, D. Mesopelagic–epipelagic fish nexus in viability and feasibility of commercial-scale mesopelagic fisheries. Natural Resource Modeling 2022, 35, e12350. [Google Scholar] [CrossRef]
- Tierney, M.; Hindell, Mark A.; Goldsworthy, S. . Energy content of mesopelagic fish from Macquarie Island. Antarctic science 2002, 14, 225–230. [CrossRef]
- López-Pérez, C.; Olivar, M.P.; Tuset V.M.; Bernal A.; Hulley P.A. Energy density of mesopelagic fishes from the Atlantic Ocean. Journal of Fish Biology 2023, 102, 924–35. [CrossRef]
- Zhang B, Virtue P, Pethybridge H, Swadling KM, Nichols PD, Chang KJ. Nutritional composition and energetic values of mesopelagic fish from the Tasman Sea. Journal of Food Composition and Analysis. 2025 Jun 1;142:107509. 2025, 142, 107509.
- Stickney, D.G.; Torres, J.J. Proximate composition and energy content of mesopelagic fishes from the eastern Gulf of Mexico. Mar Biol 1989, 3, 13–24. [Google Scholar] [CrossRef]
- Childress, J.J.; Nygaard, M. H. ; The chemical composition of midwater fishes as a function of depth of occurrence off southern California. Deep Sea Research and Oceanographic Abstracts 1973, 20, 1093–1109. [Google Scholar] [CrossRef]
- Bailey, T.G.; Robison, B.H. Food availability as a selective factor on the chemical compositions of midwater fishes in the eastern North Pacific. Mar Biol 1986, 91, 131–141. [Google Scholar] [CrossRef]
- Hjort, J. Fluctuations in the great fisheries of northern Europe viewed in light of biological research. Rapp P-v Reun Cons Int Explor Mer 1914, 20, 1–228. [Google Scholar]
- Houde, E.D. Critical food concentrations for larvae of three species of subtropical marine fishes. Bull Mar Sci 1978, 28, 395–411. [Google Scholar]
- Hunter, J.R. Feeding ecology and predation of marine fish larvae. In: Lasker, R. [ed] Marine fish larvae. University of Washington Press. Seattle, USA. 1981, 1, 33–77. [Google Scholar]
- Sifa, L. and Matius J.A. 1987. The Critical Period of High Mortality of Larvae Fish: A Discussion Based on Current Research. Chinese Journal of Oceanology and Limnology 1987, 5, 80–96. [Google Scholar] [CrossRef]
- Letcher, B.H.; Rice, J.A.; Crowder, L.B.; Rose, K.A. Variability in survival of larval fish: Disentangling components with a generalized individual-based model. Can J Fish Aquat Sci 1996, 53, 787–801. [Google Scholar] [CrossRef]
- Claramunt, R.M.; Wahl, D.H. The effects of abiotic and biotic factors in determining larval fish growth rates: A comparison across species and reservoirs. Trans Amer Fish Soc 2000, 12, 835–851. [Google Scholar] [CrossRef]
- MacKenzie, B.R.; Kiørboe, T. Larval fish feeding and turbulence: A case for the downside. Limnol Oceanogr 2000, 45, 1–10. [Google Scholar] [CrossRef]
- McCasker, N.; Humphries, P.; Meredith, S.; Klomp, N. Contrasting Patterns of Larval Mortality in Two Sympatric Riverine Fish Species: A Test of the Critical Period Hypothesis. PLoS ONE 2014, 9, e109317. [Google Scholar] [CrossRef] [PubMed]
- Houde, E.D. Fish early life dynamics and recruitment variability. Amer Fish Soc Symp 1987, 2, 17–29. [Google Scholar]
- Luecke, C.; Rice, J.A.; Crowder, L.B.; Yeo, S.E.; Binkowski, F. P. Recruitment mechanisms of bloater in Lake Michigan: an analysis of the predatory gauntlet. Can J Fish Aquat Sci 1990, 47, 524–532. [Google Scholar] [CrossRef]
- Rice, J.A.; Miller, T.J.; Rose, K.A.; Crowder, L.B.; Marschall, E.A.; Trebitz, A.S.; DeAngelis, D.L. Growth rate variation and larval survival: Inferences from an individual-based size-dependent predation model. Can J Fish Aquat Sci 1993, 50, 133–142. [Google Scholar] [CrossRef]
- Jeffrey, J.; Taggart, C.T. Growth variation and water mass associations of larval silver hake (Merluccius bilinearis) on the Scotian Shelf. Can J Fish Aquat Sci 2000, 57, 1728–1738. [Google Scholar] [CrossRef]
- Lasker. R. Use of fish eggs and larvae in probing some major problems in fisheries and aquaculture. Amer Fish Soc Symp 1987, 2, 1–16.
- Rosenthal, H.; Hempel, G. Experimental studies in feeding and food requirements of herring larvae [Clupea harengus L.] In: Steele JH [ed] Marine food chains. Univ of California Press, USA. 1970, 1, 344–364. [Google Scholar]
- Blaxter, J.H.S.; Staines, M.E. Food searching potential in marine fish larvae. In: Crisp DJ [ed] Fourth European marine biology symposium. University Press, Cambridge, UK. 1971, 1, 467–485. [Google Scholar]
- Hunter, J.R. Swimming and feeding behavior of larval anchovy Engraulis mordax. Fish Bull US 1972, 70, 821–838. [Google Scholar]
- May, R.C. Larval mortality in marine fishes and the critical period concept. In: Blaxter JHS [ed] The early life history of fish. Springer-Verlag, New York. 1974, 3-19.
- Hinrichsen, H.H.; Lehmann, A.; Möllmann, C.; Schmidt, J.O. Dependency of larval fish survival on retention/dispersion in food limited environments: the Baltic Sea as a case study. Fish Oceanogr 2003, 12, 425–433. [Google Scholar] [CrossRef]
- Govoni, J.J. , Terwilliger, M.; Markle, D.F. Age, growth, and long-distance dispersal in the western North Atlantic of vexillifer larvae of the pearlfish, Echiodon dawsoni, with notes on Carapus bermudensis [Ophidiiformes: Carapidae]. Ichthyol Res 2015, 62, 409–419. [Google Scholar] [CrossRef]
- Llopiz, J. K.; Hobday, A. J. A global comparative analysis of the feeding dynamics and environmental conditions of larval tunas, mackerels, and billfishes. Deep Sea Research Part II: Topical Studies in Oceanography 2015, 113, 113–124. [Google Scholar] [CrossRef]
- Quintanilla, J.M.; Borrego-Santos, R.; Malca, E.; Swalethorp, R.; Landry, M.R.; Gerard, T.; Laiz-Carrión, R. Maternal Effects and Trophodynamics Drive Interannual Larval Growth Variability of Atlantic Bluefin Tuna [Thunnus thynnus] from the Gulf of Mexico. Animals 2024, 14, 3–23. [Google Scholar] [CrossRef]
- Ferron, A.; Leggett, W.C. An appraisal of condition measures for marine fish larvae. Adv Mar Biol 1994, 30, 215–303. [Google Scholar]
- Ahlstrom, E.H. Kinds and abundance of fishes in the California Current region based on egg and larval surveys. Calif Mar Res Comm CalCOFI Rept 1965, 10, 31–52. [Google Scholar]
- Ahlstrom, E.H. Kinds and abundance of fish larvae in the eastern tropical Pacific on the second multivessel EASTROPAC survey, and observations on the annual cycle of larval abundance. Fish Bull US 1972, 70, 1153–1242. [Google Scholar]
- Loeb, V.J. Vertical distribution and development of larval fishes in the North Pacific central gyre during summer. Fish Bull US 1979, 77, 777–793. [Google Scholar]
- Loeb, V.J. Patterns of spatial and species abundance within the larval fish assemblage of the North Pacific central gyre during late summer. Mar Biol 1980, 60, 189–200. [Google Scholar] [CrossRef]
- Belyanina, T.N. Ichthyoplankton of the Gulf of Tonkin [composition, distribution and seasonal changes in populations]. J Ichthyo 1987, 7, 51–57. [Google Scholar]
- Perteseva-Ostroumova, T.A. New data on lanternfish larvae [Myctophidae, Pisces] with oval eyes from the Indian and Pacific Oceans. Tr Inst Okeanol SSSR 1974, 96, 77–142. [Google Scholar]
- Rodrıguez, J.M.; Barton, E.D.; Eve, L.; Hernández-León, S. Mesozooplankton and ichthyoplankton distribution around Gran Canaria, an oceanic island in the NE Atlantic. Deep Sea Research Part I: Oceanographic Research Papers 2001, 48, 2161–2183. [Google Scholar] [CrossRef]
- Gartner, J.V. Jr.; Conley, W.J.; Hopkins, T.L. Escapement by fishes from midwater trawls: A case study using lanternfishes [Pisces: Myctophidae]. Fish Bull US 1989, 87, 213–222. [Google Scholar]
- Sabatés, A.; Saiz, E. Intra-and interspecific variability in prey size and niche breadth of myctophiform fish larvae. Mar Ecol Prog Ser 2000, 201, 261–71. [Google Scholar] [CrossRef]
- Conley, W.J.; Hopkins, T.L. Feeding Ecology of Lanternfish [Pisces: Myctophidae] Larvae: Prey Preferences as a Reflection of Morphology. Bull Mar Sci 2004, 84, 123–135. [Google Scholar]
- Sassa, C.; Kawaguchi, K. 2005. Larval feeding habits of Diaphus theta, Protomyctophum thompsoni, and Tarletonbeania taylori (Pisces: Myctophidae) in the transition region of the western North Pacific. Mar Ecol Prog Ser 2005, 298, 261–276. [Google Scholar] [CrossRef]
- Contreras, T. , Olivar, M. P., Bernal, A., & Sabatés, A. Comparative feeding patterns of early stages of mesopelagic fishes with vertical habitat partitioning. Mar Biol, 2015, 162, 2265–2277. [Google Scholar]
- Methot, R.D. Jr. Spatial covariation of daily growth rates of larval northern anchovy, Engraulis mordax, and northern lampfish, Stenobranchius leucopsarus. Rapp P-v Reun Cons int Explor Mer 1981, 178, 424–431. [Google Scholar]
- Sassa, C.; Takahashi, M. Comparative larval growth and mortality of mesopelagic fishes and their predatory impact on zooplankton in the Kuroshio region. Deep Sea Research Part I: Oceanographic Research Papers 2018, 131, 121–32. [Google Scholar] [CrossRef]
- Gartner. J.V. Jr. Life histories of three species of lanternfishes [Pisces: Myctophidae] from the eastern Gulf of Mexico I. Morphological and microstructural analysis of sagittal otoliths. Mar Biol 1991, 111, 11–20. [CrossRef]
- Gartner, J.V. Jr. Patterns of reproduction in the dominant lanternfish species [Pisces: Myctophidae] of the eastern Gulf of Mexico, with a review of reproduction among tropical-subtropical Myctophidae. Bull Mar Sci 1993, 52, 721–750. [Google Scholar]
- Hopkins, T.L. Gartner, J.V. Jr. Resource-partitioning and predation impact of a low-latitude myctophid community. Mar Biol 1992, 11, 185–197. [Google Scholar] [CrossRef]
- Hopkins, T.L.; Sutton, T.; Lancraft, T.M. The trophic structure and predation impact of a low latitude midwater fish assemblage. Prog Oceanogr 1996, 38, 205–239. [Google Scholar] [CrossRef]
- Sutton, T.T.; Hopkins, T.L. Species composition, abundance, and vertical distribution of the stomiid [Pisces: Stomiiformes] fish assemblage of the Gulf of Mexico. Bull Mar Sci 1996, 59, 530–542. [Google Scholar]
- Hopkins, T.L. The vertical distribution of zooplankton in the eastern Gulf of Mexico. Deep-Sea Res 1982, 29, 1069–1083. [Google Scholar] [CrossRef]
- Hopkins, T.L.; Lancraft, T.M. The composition and standing stock of mesopelagic micronekton at 27oN, 86oW in the eastern Gulf of Mexico. Contr Mar Sci 1984, 27, 143–158. [Google Scholar]
- Hopkins, T.L.; Flock, M.E.; Gartner, J.V. Jr.; Torres, J.J. Structure and trophic ecology of a low latitude midwater decapod and mysid assemblage. Mar Ecol Prog Ser 1994, 109, 143–156. [Google Scholar] [CrossRef]
- Hopkins, T.L.; Baird, R.C.; Milliken, D.M. A messenger-operated closing trawl. Limnol Oceanogr 1973, 18, 488–490. [Google Scholar] [CrossRef]
- Baird, R.C.; Hopkins, T.L. Trophodynamics of the fish Valenciennelus tripunctulatus. III. Energetics, resources and feeding strategy. Mar Ecol Prog Ser 1981, 5, 21–28. [Google Scholar] [CrossRef]
- Burke, B. J.; Rice, J. A. A linked foraging and bioenergetics model for southern flounder. Trans Amer Fish Soc 2002, 131, 120–131. [Google Scholar] [CrossRef]
- Laurence, G.C. Growth and survival of Haddock (Melanogrammus aeglefinus) larvae in relation to planktonic prey concentration. J. Fish. Res. Bd. Canada 1974, 31, 1415–1419. [Google Scholar] [CrossRef]
- Laurence, G.C. A bioenergetic model for the analysis of feeding and survival potential of winter flounder, Pseudopleuronectes americanus, larvae during the period from hatching to metamorphosis. Fish. Bull 1977, 75, 529–546. [Google Scholar]
- Eldridge, M.B.; Whipple, J.A.; Bowers, M.J. Bioenergetics and growth of striped bass, Morone saxatilis, embryos and larvae. Fish Bull US 1982, 80, 461–474. [Google Scholar]
- Brightman, R.V. Energetics and RNA/DNA ratio of red drum larvae Sciaenops ocellatus. PhD Dissertation, Univ of South Florida, Tampa, Florida. 1993, 1-178.
- Brightman, R.V.; Torres, J.J.; Donnelly, J.D.; Clarke, M.E. Energetics of larval red drum Sciaenops ocellatus. Part II, Growth and biochemical indicators. Fish Bull US 1997, 95, 431–444. [Google Scholar]
- Karjalainen, J.; Miserque, D.; Huuskonen, H. The estimates of food consumption in larval and juvenile fish: experimental evaluation of bioenergetic models. J Fish Biol 1997, 51, 39–51. [Google Scholar] [CrossRef]
- Morales, M.M.; Fiechter, J.; Field, J.C.; Kashef, N.S.; Hazen, E.L.; Carr, M.H. , Development and application of a bioenergetics growth model for multiple early life stages of an ecologically important marine fish. Ecological Modelling 2024, 488, 110575. [Google Scholar] [CrossRef]
- Houde, E.D.; Schekter, R.C. Oxygen uptake and comparative energetics among eggs and larvae of three subtropical marine fishes. Mar Biol 1983, 72, 283–293. [Google Scholar] [CrossRef]
- Donnelly, J.; Torres, J.J.; Hopkins, T.L.; Lancraft, T.M. Proximate composition of Antarctic mesopelagic fishes. Mar Biol 1990, 106, 13–23. [Google Scholar] [CrossRef]
- Winberg, G.G. The relation of the metabolic rate of fish to temperature. Role of metabolism and food requirements of fishes. 1956, 194, 21–38. [Google Scholar]
- Warren, C.E.; Davis, G.E. Laboratory studies on the feeding, bioenergetics and growth of fish. In: Gerking SD [ed.] The biological basis of fresh-water fish production. Wiley & Sons, New York, USA. 1967, 175-214. [Google Scholar]
- Deslauriers, D.; Chipps, S.R.; Breck, J.E.; Rice, J.A.; Madenjian, C.P. Fish Bioenergetics 4.0: An R-Based Modeling Application. Fisheries 2017, 42, 586–96. [Google Scholar] [CrossRef]
- Lowrey, O.H.; Roseborough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J Biol Chem 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Bligh, E.G.; Dyer, W.F. A rapid method of total lipid extraction and purification. Can J Biochem Physiol 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.A.; Smith, F. Colorimetric method for determination of sugars and related substances. Analyt Chem 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Brett, J.R.; Groves, T.D.D. Physiological Energetics. In: Hoar WS Randall DJ [eds.] Fish Physiology. Academic Press New York, NY, USA 1979, VIII, 279-351.
- Ware, D.M. Bioenergetics of pelagic fish: Theoretical change in swimming speed and motion with body size. J Fish Res Bd Canada 1975, 35, 33–41. [Google Scholar] [CrossRef]
- Govoni, J.J.; Peters, D.S.; Merriner, J.V. Carbon assimilation during larval development of the marine teleost Leiostomus xanthurus Lacepede. J Exp Mar Biol Ecol 1982, 64, 287–299. [Google Scholar] [CrossRef]
- Govoni, J.J.; Boehlert, G.M.; Watanabe, Y. The physiology of digestion in fish larvae. Environ Biol Fishes 16, 59–77. [CrossRef]
- Hansson, S.; Rudstam, L.G.; Kitchell, J.F.; Peppard, P.E.; Hildén, M.; Johnson, B.L. Predation rates by North Sea cod [Gadus morhua]–predictions from models on gastric evacuation and bioenergetics. ICES Journal of Marine Science 1996, 53, 107–114. [Google Scholar] [CrossRef]
- Beers, J.R.; Reid, F.M.H.; Stewart, G.L. Microplankton of the North Pacific Central Gyre. Population structure and abundance, June 1973. Int Revue Gesamten Hydrobiol 1975, 60, 607–638. [Google Scholar]
- Cetta, C.M.; Capuzzo, J.M. Physiological and biochemical aspects of embryonic and larval development of the winter flounder Pseudopleuronectes americanus. Mar Biol 1982, 71, 327–337. [Google Scholar] [CrossRef]
- Torres, J.J.; Brightman, R.V.; Donnelly, J.; Harvey, J. Energetics of larval red drum Sciaenops ocellatus. Part I: Oxygen consumption, specific dynamic action, and nitrogen excretion. Fish Bull US 1996, 95, 431–444. [Google Scholar]
- Houde, E.D.; Schekter, R.C. Simulated food patches and survival of larval bay anchovy, Anchoa mitchilli, and sea bream, Archosargus rhomboidalis. Fish Bull US 1978, 76, 483–486. [Google Scholar]
- Theilacker, G.H. Feeding ecology and growth energetics of larval northern anchovy, Engraulis mordax. Fish Bull US 1987, 85, 213–228. [Google Scholar]
- Infante, J. Z.; Cahu, C. L. Ontogeny of the gastrointestinal tract of marine fish larvae. Comparative Biochemistry and Physiology. Part C: Toxicology & Pharmacology 2001, 130, 477–487. [Google Scholar]
- Cassie, R.M. Micro-distribution of plankton. NZJ Sci 1959, 2, 398–409. [Google Scholar]
- Owen, R.W. Microscale plankton patchiness in the larval anchovy environment. Rapp P-v Reun Cons int Explor Mer 1981, 178, 363–368. [Google Scholar]
- Owen, R.W. Microscale and finescale variations of small plankton in coastal and pelagic environments. J Mar Res 1989, 47, 197–240. [Google Scholar] [CrossRef]
- Conley, W.J.; Turner, J.T. The Zooplankton and Phytoplankton of the Westport River Estuary. Hydrobiologia 1991, 210, 225–232. [Google Scholar] [CrossRef]
- Arthur, D.K. Food and feeding of larvae of three fishes occurring in the California Current, Sardinops sagax, Engraulis mordax, and Trachurus symmetricus. Fish Bull US 1976, 74, 517–530. [Google Scholar]
- Govoni, J.J.; Hoss, D.E.; Chester, A.J. Comparative feeding of three species of larval fishes in the northern Gulf of Mexico: Brevoortia patronus, Leiostomus xanthurus, and Micropogonias undulatus. Mar Ecol Prog Ser 1983, 13, 189–199. [Google Scholar] [CrossRef]
- Govoni, J.J.; Ortner, P.B.; Al-Yamani, F.; Hill, L.C. Selective feeding of spot, Leiostomus xanthurus, and Atlantic croaker, Micropogonias undulatus, larvae in the northern Gulf of Mexico. Mar Ecol Prog Ser 1986, 28, 175–183. [Google Scholar] [CrossRef]
- Stoecker, D.K.; Govoni, J.J. Food selection by young larval gulf menhaden [Brevoortia patronus]. Mar Biol 1984, 80, 299–306. [Google Scholar] [CrossRef]
- Brothers, E.B.; Williams, D.M.; Sale, P.F. Length of larval life in twelve families of fishes at "One Tree Lagoon", Great Barrier Reef, Australia. Mar Biol 1983, 76, 319–324. [Google Scholar] [CrossRef]
- Cowan, J.H. Jr. Age and growth of Atlantic croaker, Micropogonias undulatus, larvae collected in the coastal waters of the northern Gulf of Mexico as determined by increments in saccular otoliths. Bull Mar Sci 1988, 42, 349–357. [Google Scholar]
- Peebles, E.B.; Tolley, S.G. Distribution, growth and mortality of larval spotted seatrout, Cynoscion nebulosus: a comparison between two adjacent estuarine areas of southwest Florida. Bull Mar Sci 1988, 42, 397–410. [Google Scholar]
- Searcy, S.P.; Sponaugle, S. Variable larval growth in a coral reef fish. Mar Ecol Prog Ser 2000, 206, 213–226. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




