Submitted:
10 July 2025
Posted:
11 July 2025
You are already at the latest version
Abstract
Keywords:
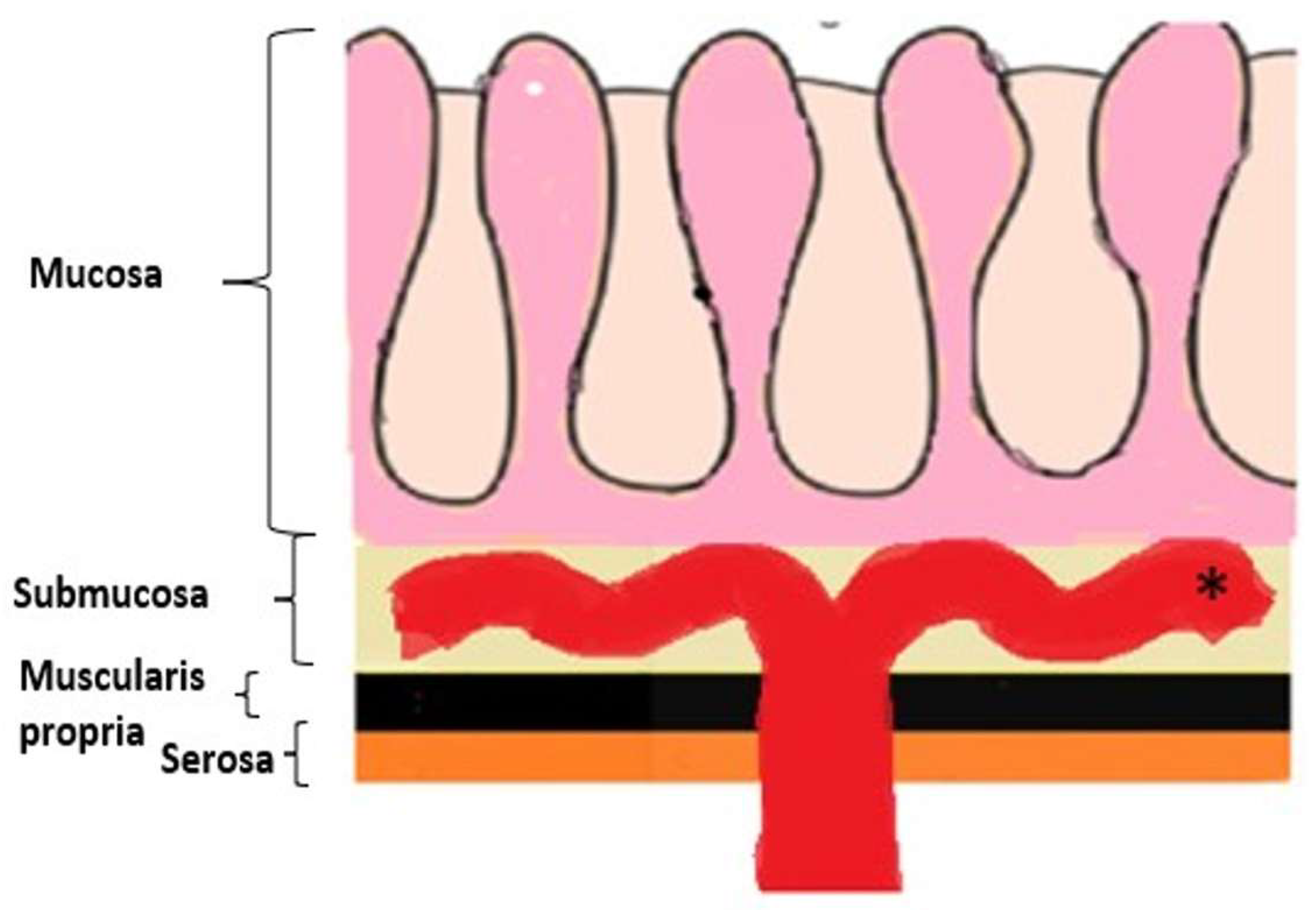
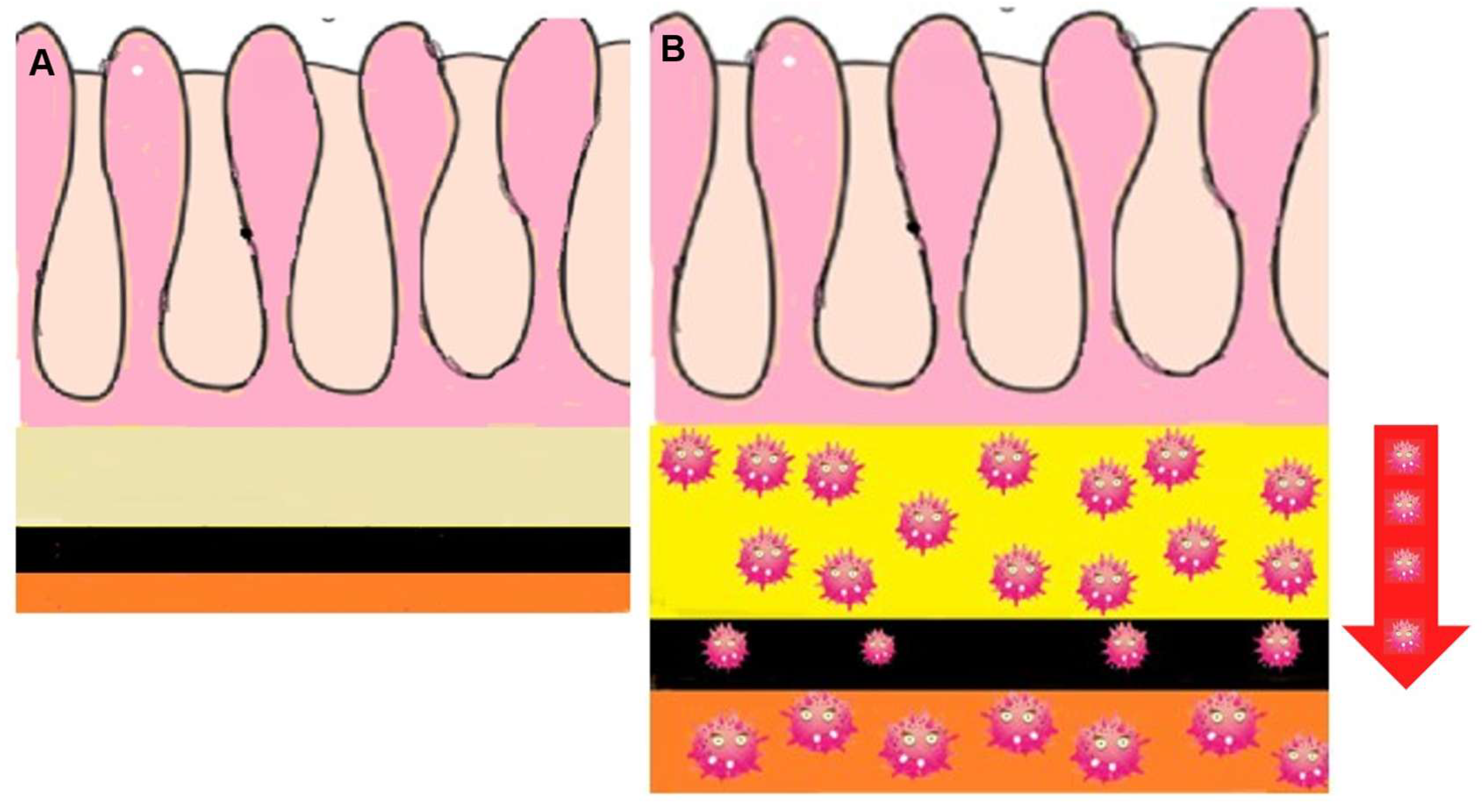
1. Background
2. Main text
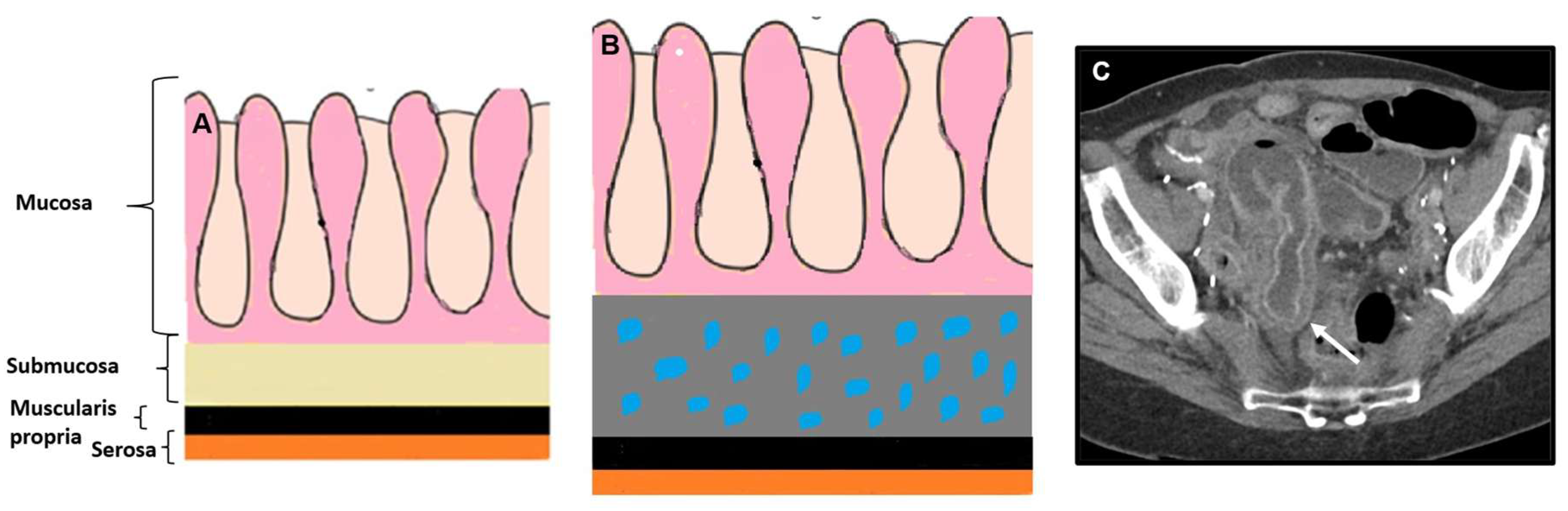
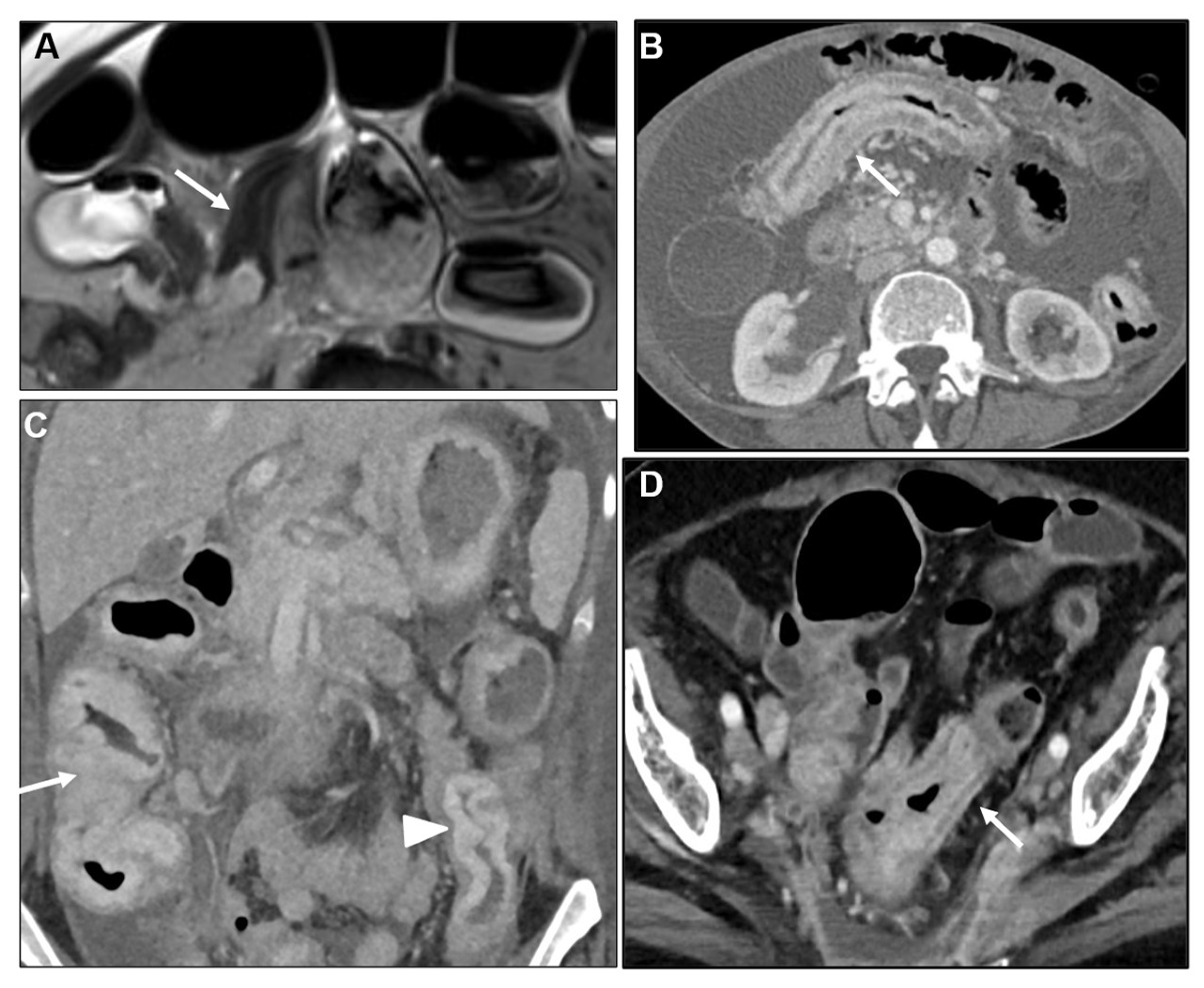
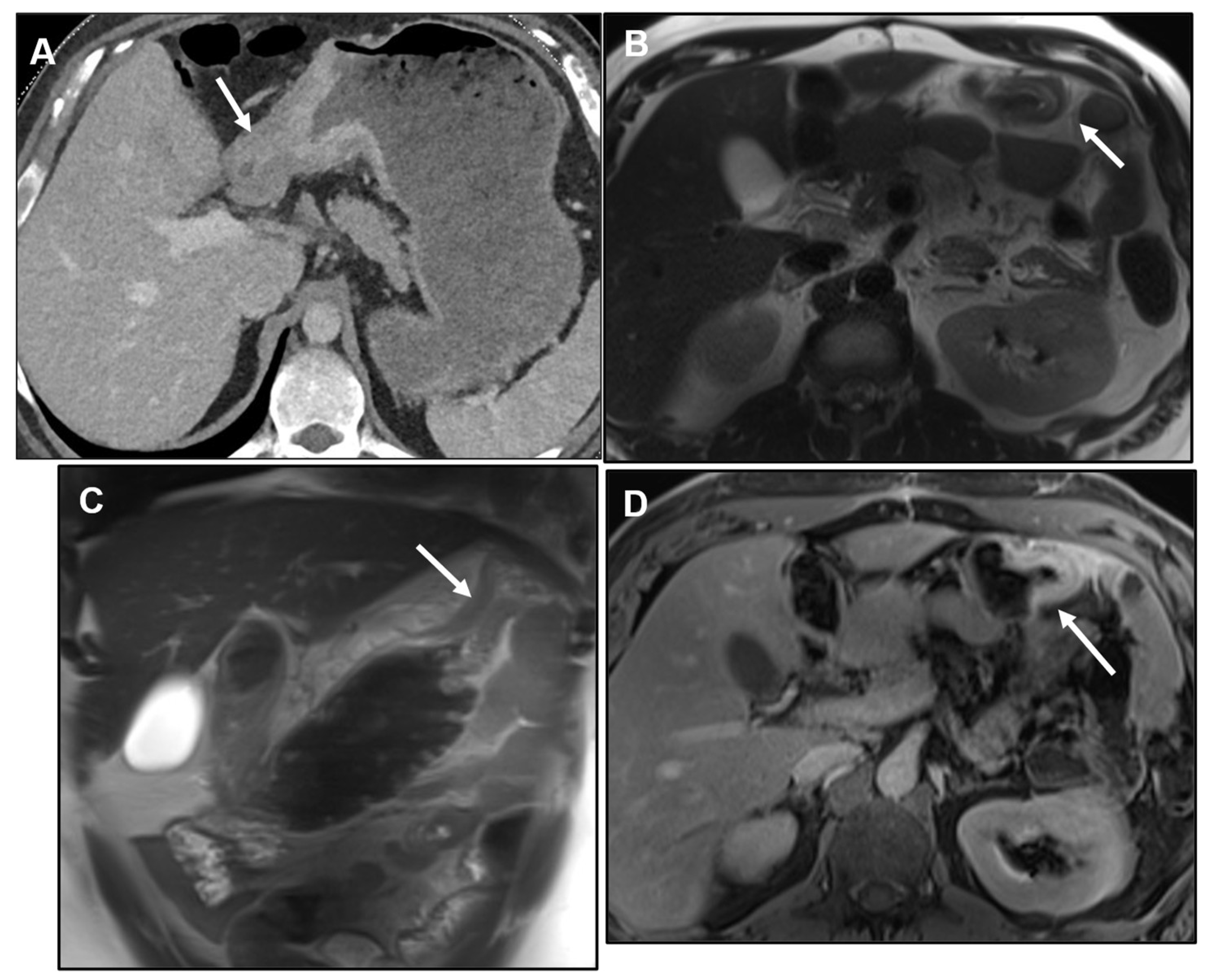
2.1. Primary gastric tumour with LP appearance
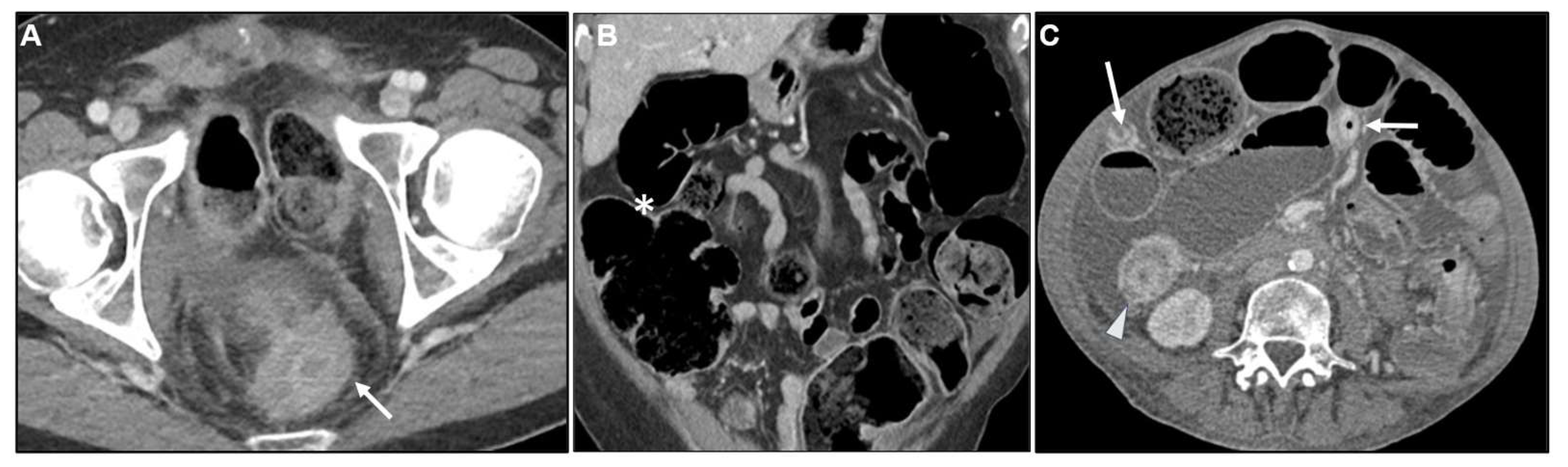
2.1.1. Primary GI tumours with LP appearance: Cross-sectional imaging
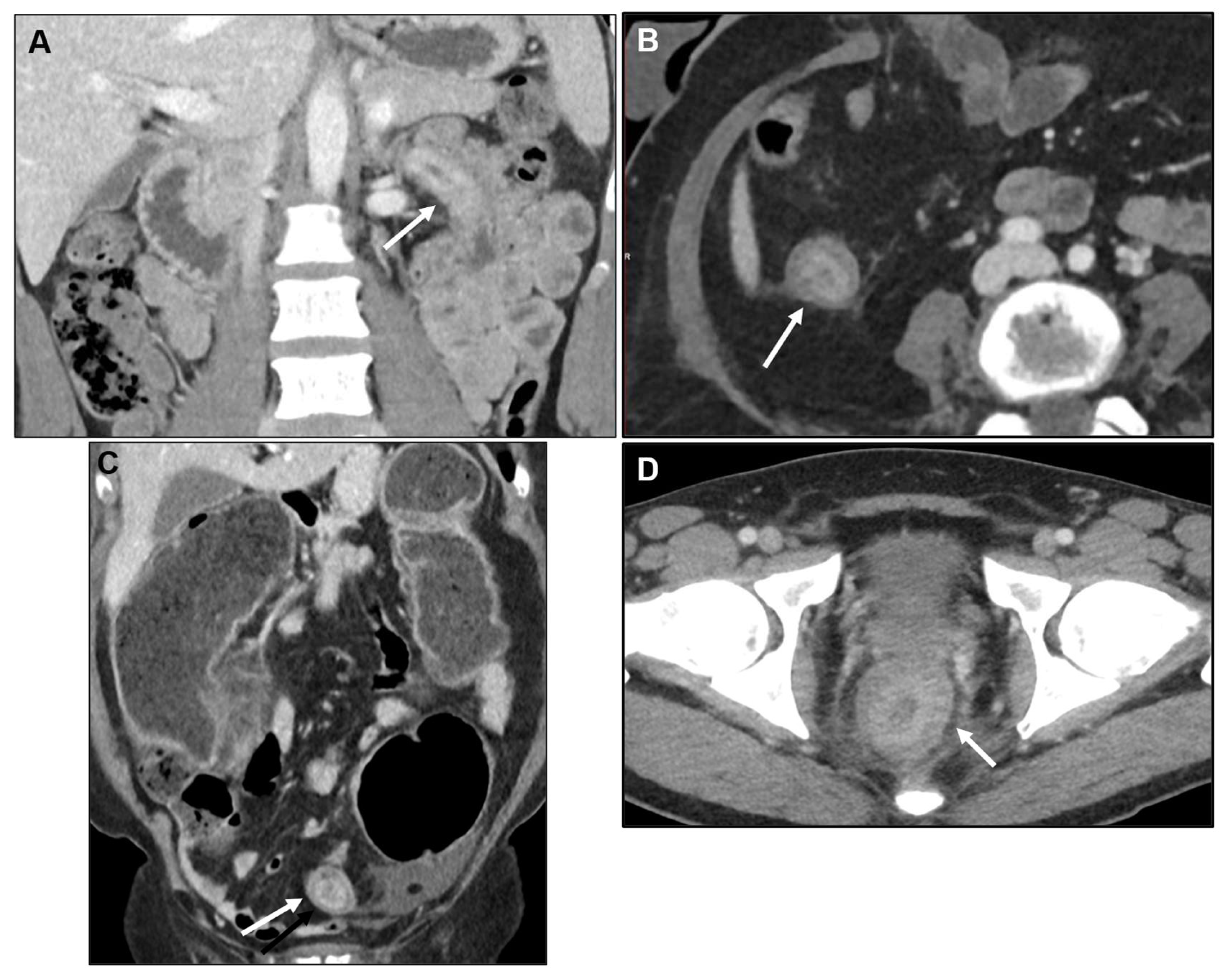
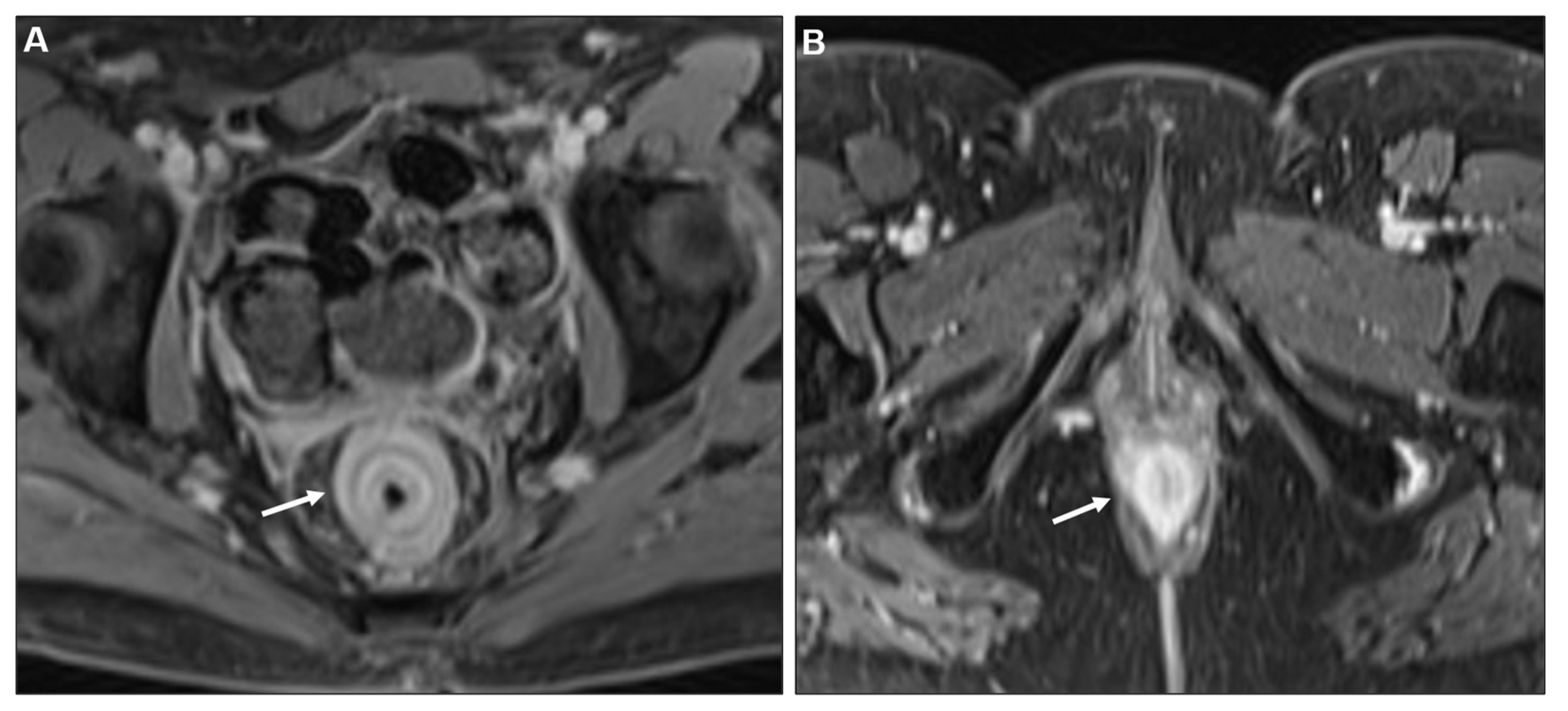
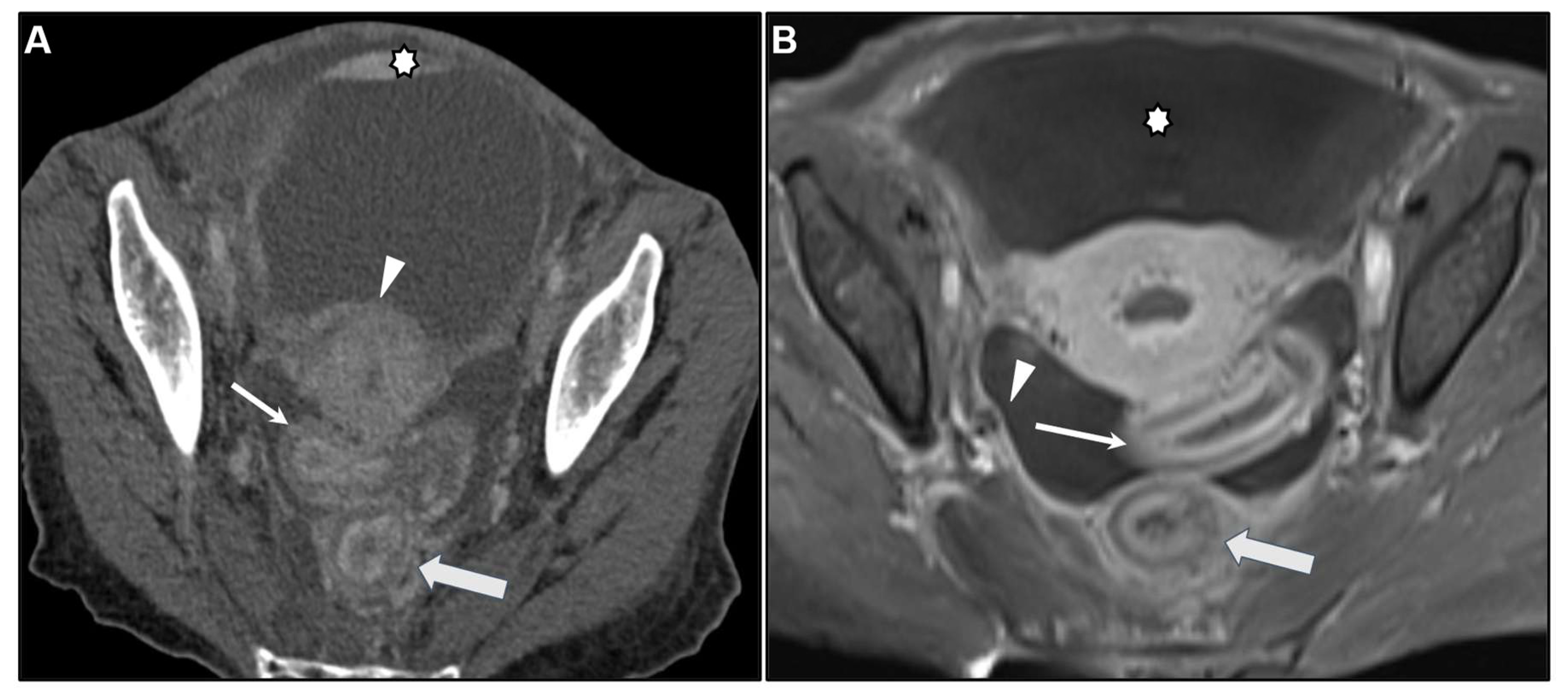
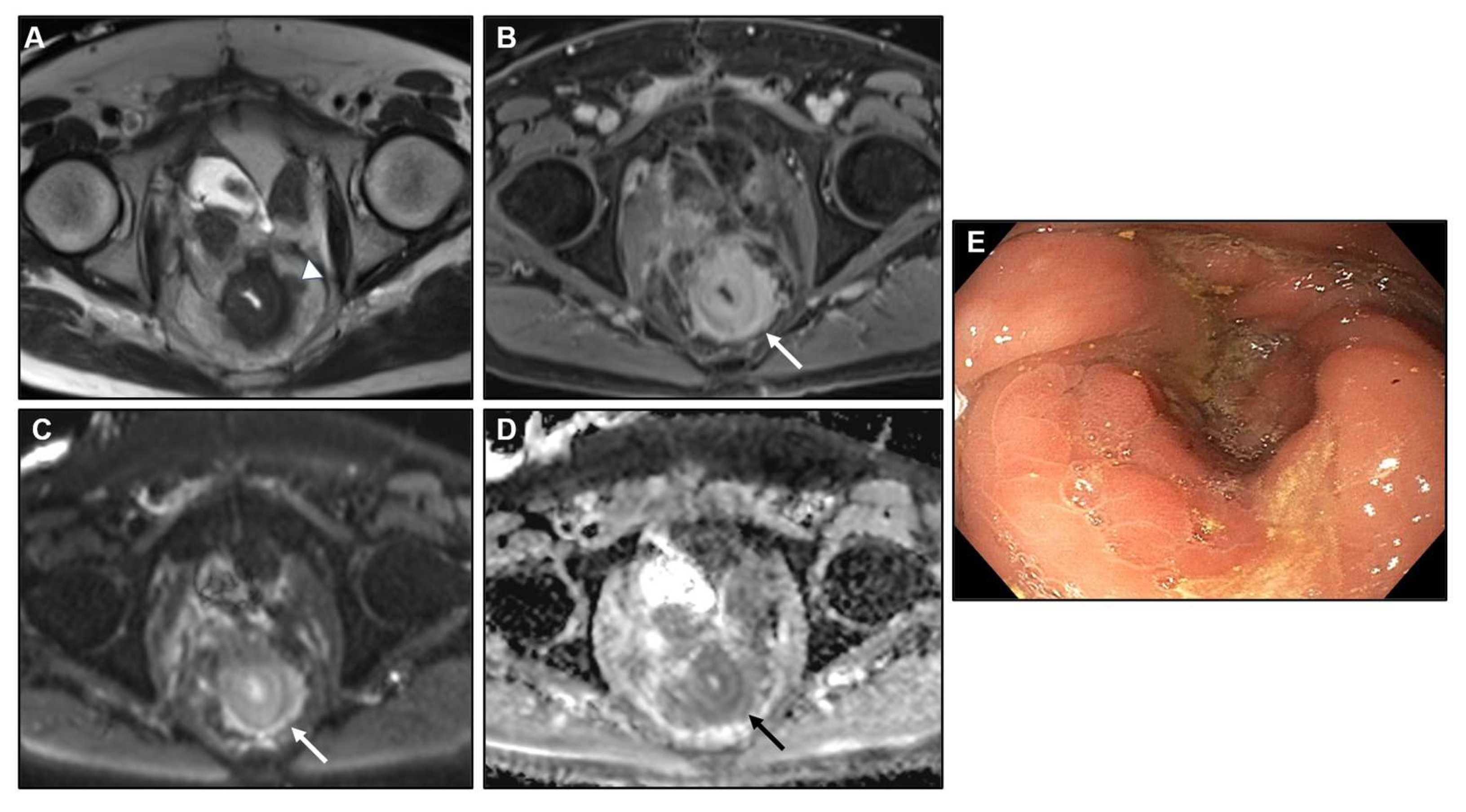
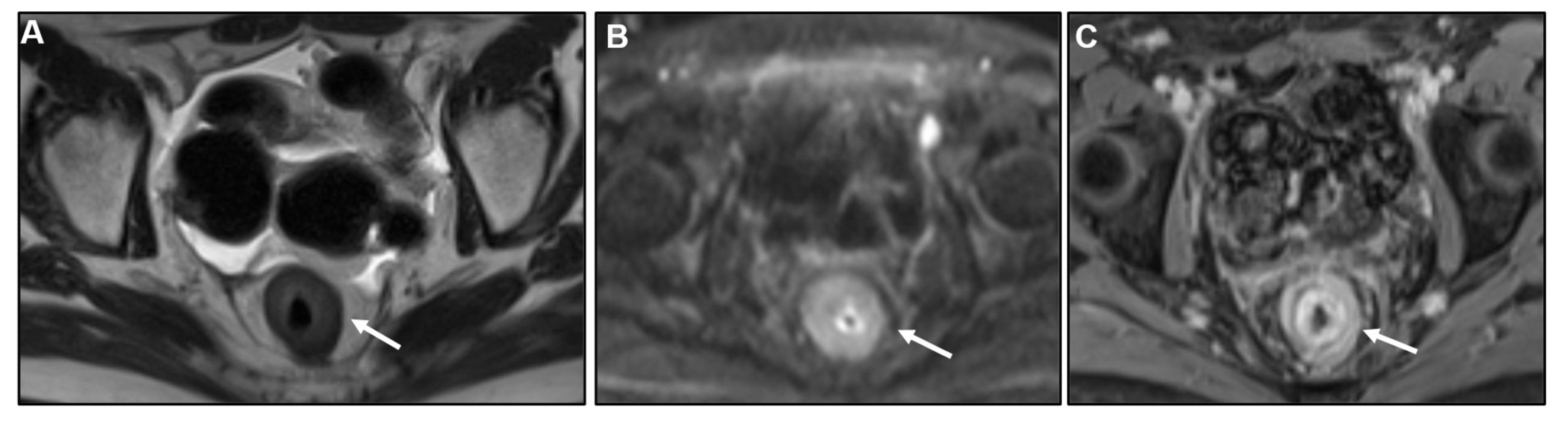
2.2. Metastases to the GI tract with LP appearance
2.2.1. Imaging features

2.2.2. Associated imaging features (Table 2)

2.3. Diagnostic challenges
3.1. Anamnesis
3.2. Clinical presentation
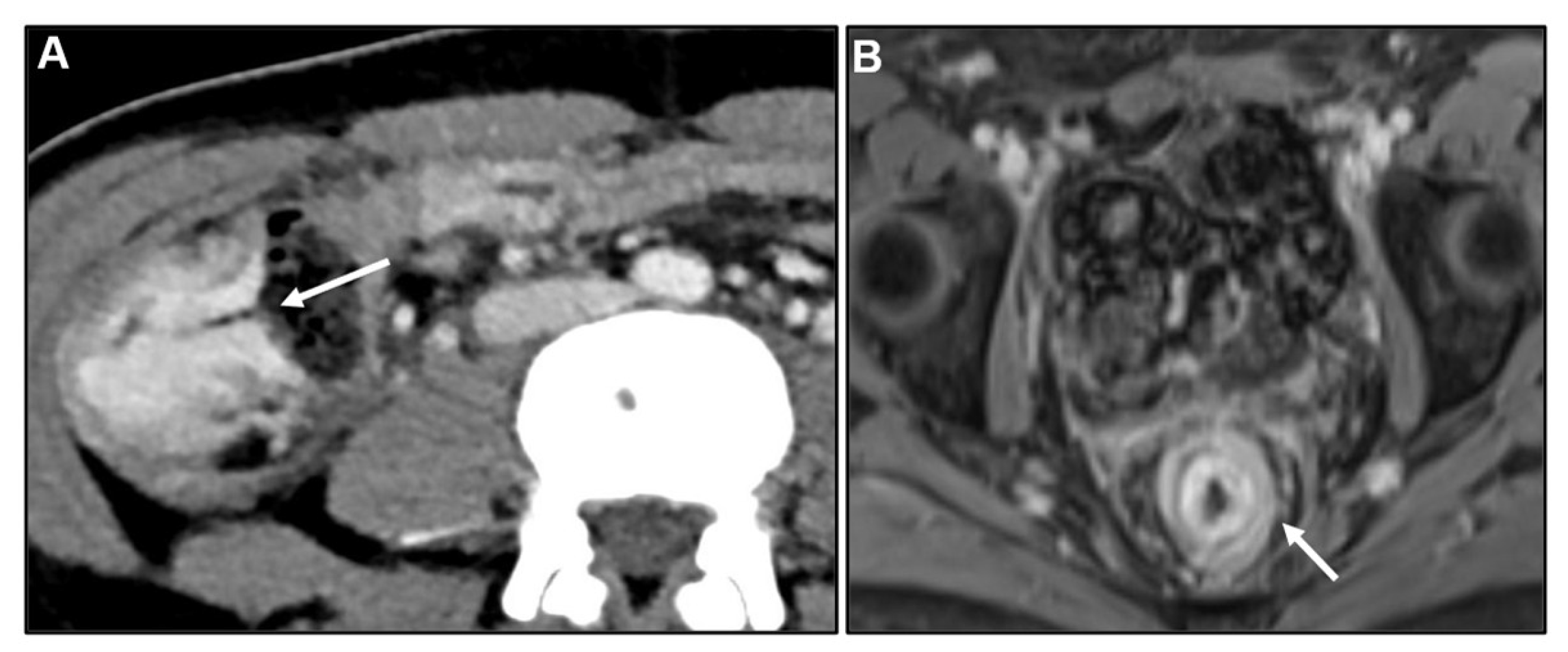
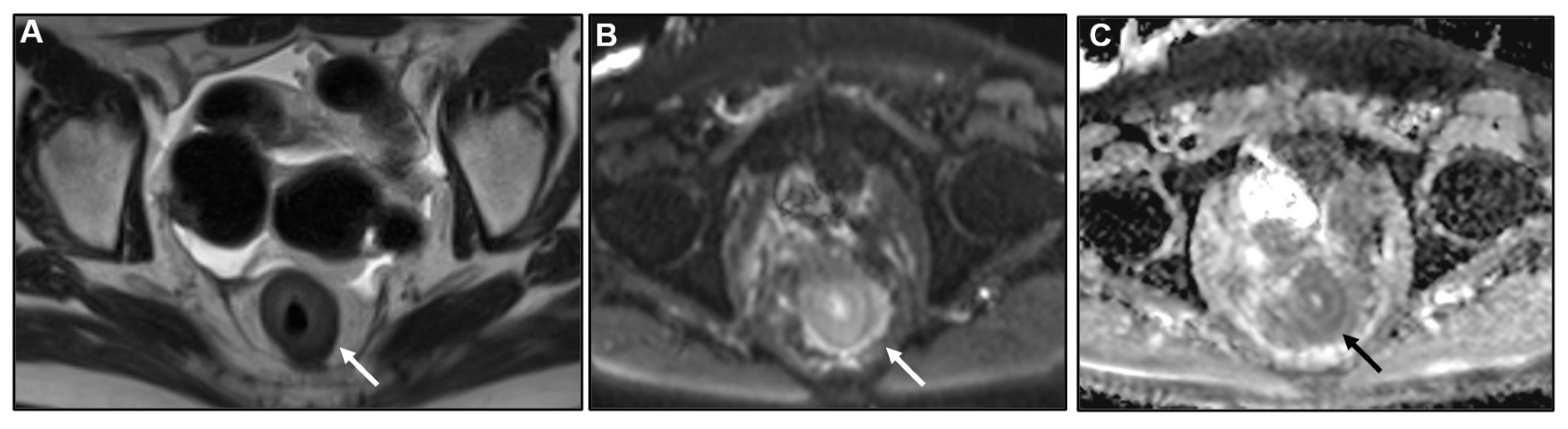
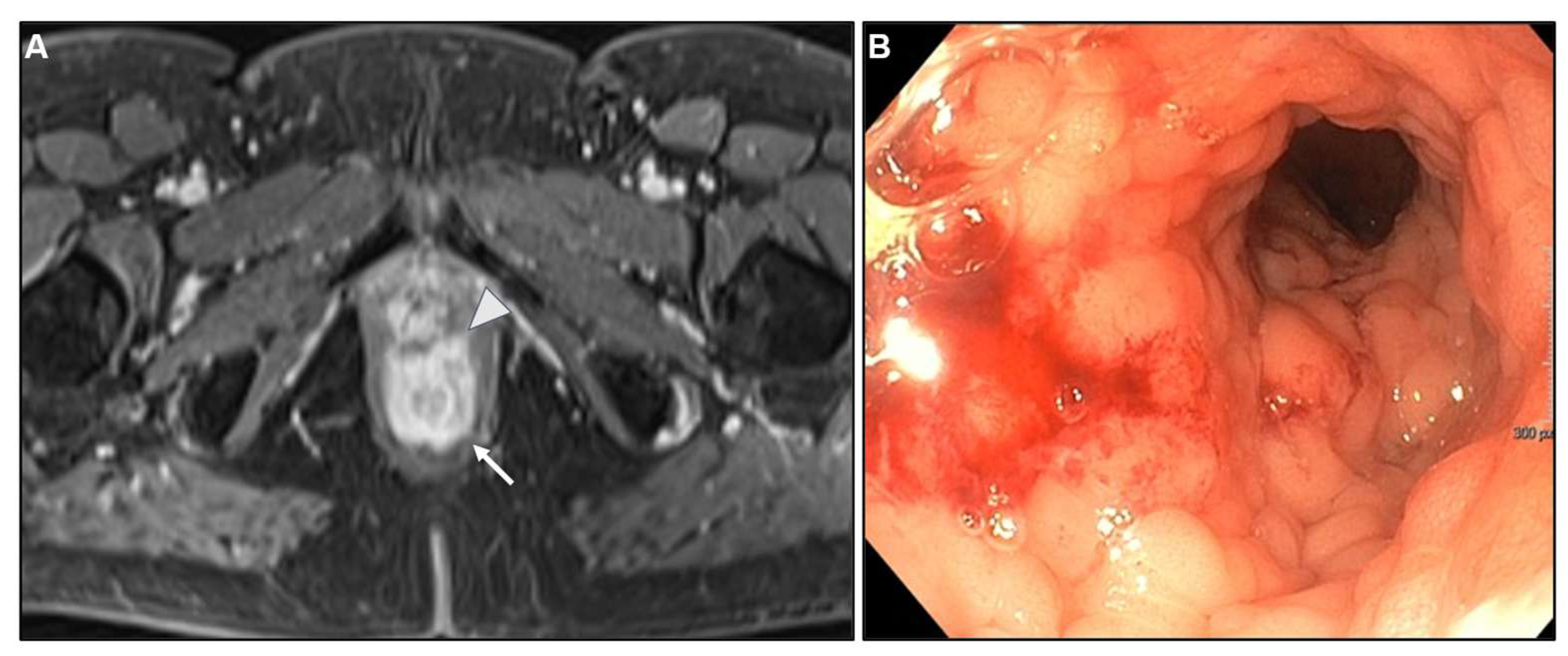
3.3. Imaging findings
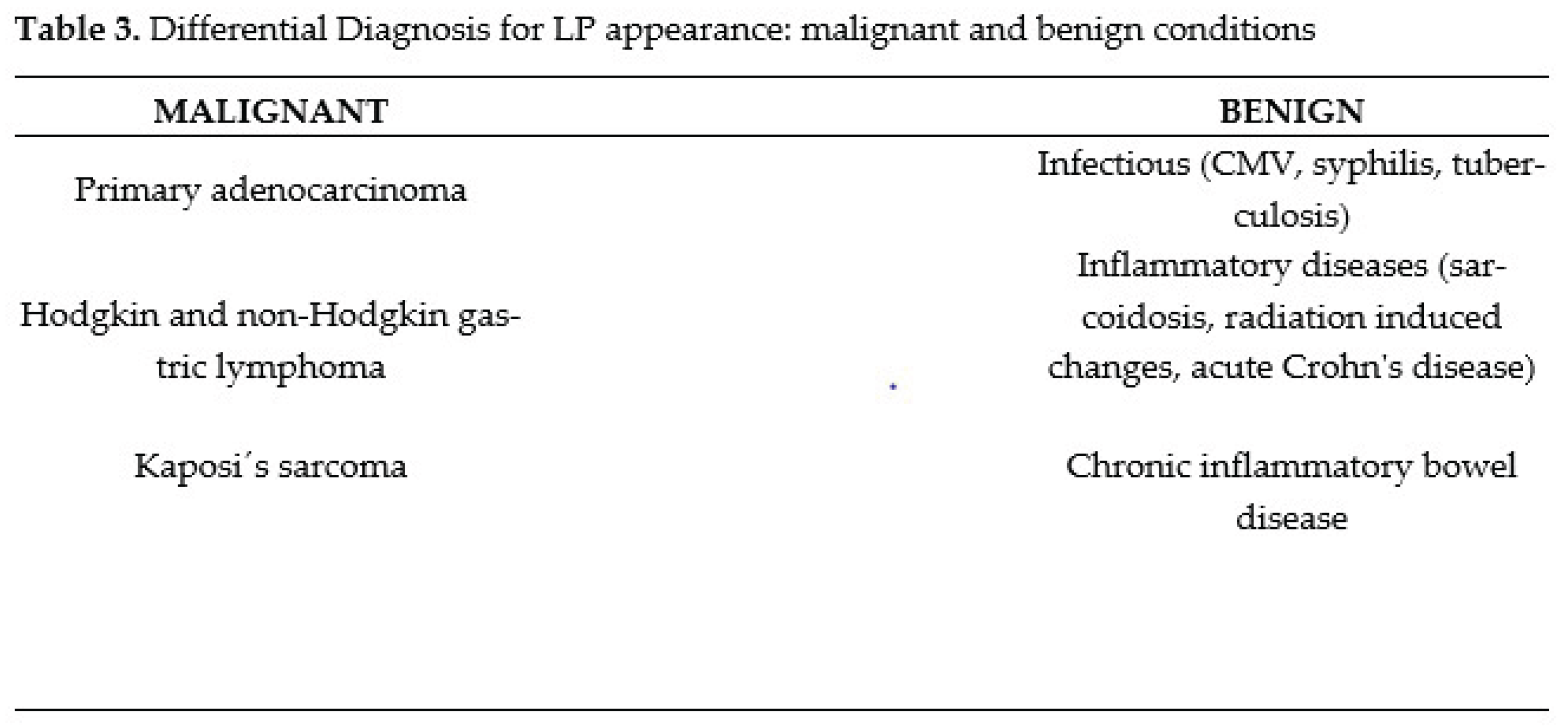
2.4. Differential diagnosis
2.4.1. Malignant causes.
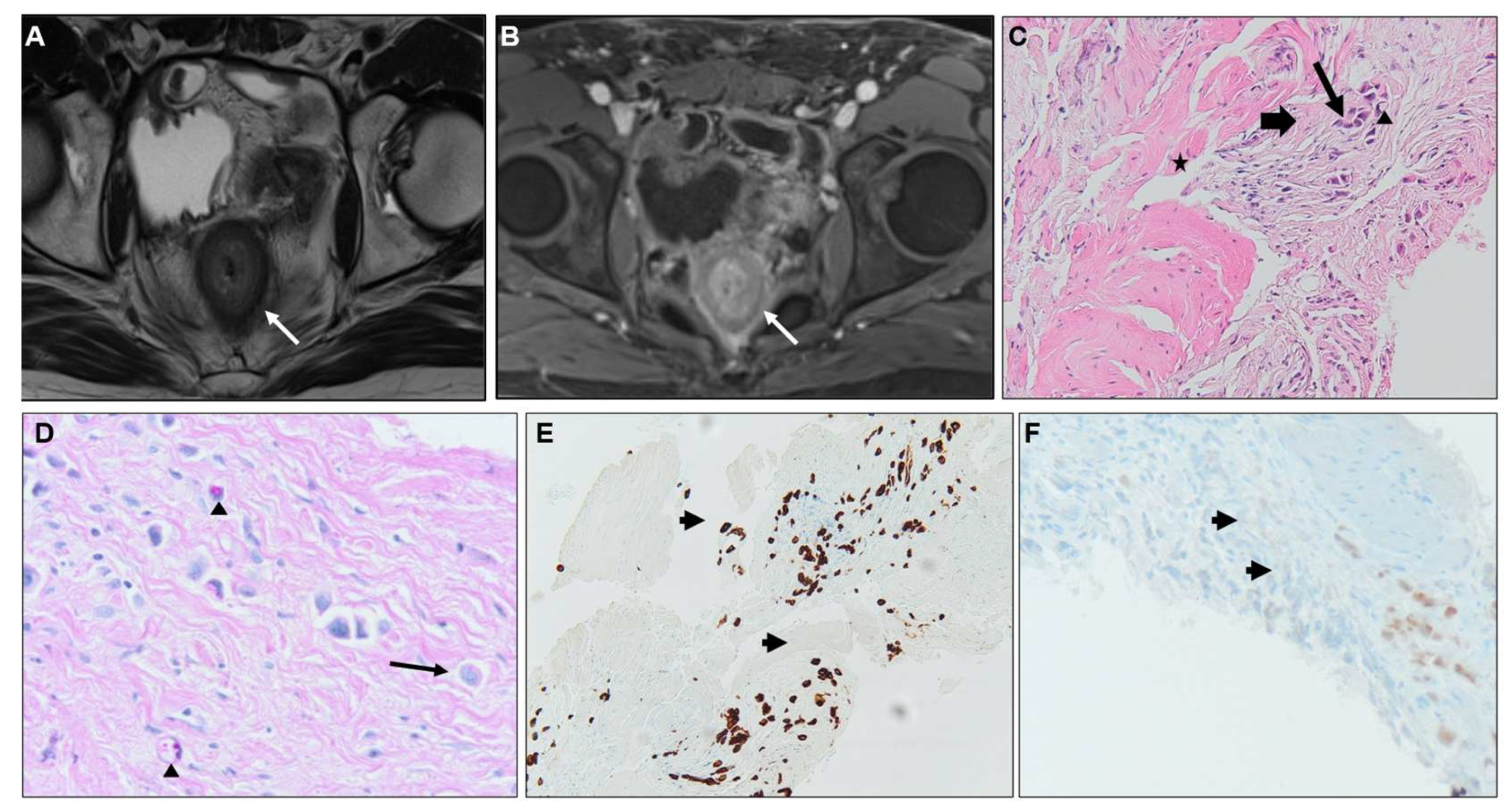
2.4.2. Benign causes.
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
| ADC | apparent diffusion coefficient |
| CE | contrast enhanced |
| CT | computer tomography |
| CK7 | cytokeratin 7 |
| DWI | diffusion weighted imaging |
| GI | gastrointestinal |
| GRE-T1WI | gradient recalled echo T1 weighted images |
| H&E | hematoxylin and eosin |
| IHC | immunohistochemistry |
| ILC | invasive lobular carcinoma |
| LP | linitis plastica |
| MP | muscularis propria |
| MPR | multiplanar reconstruction |
| MR | magnetic resonance |
| SB | small bowel |
| PC | peritoneal carcinomatosis |
| T2WI | T2 weighted imaging |
References
- Feczko, P.J.; Collins, D.D.; Mezwa, D.G. Metastatic disease involving the gastrointestinal tract. Radiol Clin North Am. 1993, 31, 1359–73. [Google Scholar] [CrossRef] [PubMed]
- Cormier, W.J.; Gaffey, T.A.; Welch, J.M.; Welch, J.S.; Edmonson, J.H. Linitis plastica caused by metastatic lobular carcinoma of the breast. Mayo Clin Proc. 1980, 55, 747–53. [Google Scholar] [PubMed]
- Mastoraki, A.; Papanikolaou, I.S.; Sakorafas, G.; Safioleas, M. Facing the challenge of managing linitis plastica-review of the literature. Hepatogastroenterology 2009, 56, 1773–8. [Google Scholar] [PubMed]
- Agnes, A.; Estrella, J.S.; Badgwell, B. The significance of a nineteenth century definition in the era of genomics: linitis plastica . World J Surg Oncol. 2017, 5, 15(1)-123. [CrossRef] [PubMed]
- Lyle, H.H. VIII.Linitis Plastica (Cirrhosis of Stomach): With a Report of a Case Cured by Gastro-Jejunostomy. Ann Surg. 1911, 54, 625–68. [Google Scholar] [CrossRef] [PubMed]
- Stout, A.P. Tumors of the stomach, Armed Forces Institute of Pathology, Washington DC, USA, 1953; pp. 1- 134.
- Consul, N.; DiSantis, D.J.; Dyer, R.B. The “leather bottle” stomach. Abdom Radiol (NY) 2018, 43, 2210–1. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, N.; Ulahannan, M.J.; Mandile, M.A.; Cayten, C.G.; Pitchumoni, C.S. Increased risk of colorectal cancer following breast cancer. Ann Surg 1986, 203, 307–10. [Google Scholar] [CrossRef] [PubMed]
- Christodoulidis, G.; Koumarelas, K.E.; Kouliou, M.N.; Samara, M.; Thodou, E.; Zacharoulis, D. The Genomic Signatures of Linitis Plastica Signal the Entrance into a New Era: Novel Approaches for Diagnosis and Treatment. Int J Mol Sci [Internet] 2023. [CrossRef]
- Balthazar, E.J.; Rosenberg, H.D.; Davidian, M.M. Primary and metastatic scirrrhous carcinoma of the rectum. AJR Am J Roentgenol 1979, 132, 711–5. [Google Scholar] [CrossRef] [PubMed]
- Mariette, C.; Carneiro, F.; Grabsch, H.I.; van der Post, R.S.; Allum, W.; de Manzoni, G.; et al. Consensus on the pathological definition and classification of poorly cohesive gastric carcinoma. Gastric Cancer 2019, 22, 1–9. [Google Scholar] [CrossRef] [PubMed]
- El-Nakeep, S.; Kasi, A. Linitis Plastica. StatPearls Publishing, Treasure Island, Florida, USA, 2023.
- Piessen, G; Messager, M; Leteurtre, E; Jean-Pierre, T; Mariette, C. Signet ring cell histology is an independent predictor of poor prognosis in gastric adenocarcinoma regardless of tumoral clinical presentation. Ann Surg. 2009, 250, 878–87. [Google Scholar]
- WHO Classification of Tumours of the Digestive System. 2010. International Agency for Research on Cancer (IARC). WHO Classification of Tumours of the Digestive System. 4th ed. Lyon: IARC; 2010.
- Landry, R.M. Linitis Plastica: A Clinical and Pathological Study of the Various Diseases which May Produce the Leather Bottle Stomach 1950, 140 p.
- Howard, C.P. Linitis plastica: a study of ten cases. QJM 1933, 2, 59–78. [Google Scholar]
- Fornasarig, M.; Capuano, A.; Maiero, S.; Pivetta, E.; Canzonieri, V.; Belluco, C.; et al. pCLE detects mucosal neoplastic vascular pattern in gastric linitis plastica. Clin Exp Med. 2023, 23, 547–51. [Google Scholar] [CrossRef] [PubMed]
- Muraoka, S.; Tsuchida, K.; Iwasaki, M.; Izawa, N.; Jinnai, H.; Komatsubara, T.; et al. A case report of gastric linitis plastica diagnosed by endoscopic ultrasound-guided fine needle aspiration. Medicine 2017, 96, e8937. [Google Scholar] [CrossRef] [PubMed]
- Meyers, M.A.; Oliphant, M.; Teixidor, H.; Weiser, P. Metastatic carcinoma simulating inflammatory colitis. Am J Roentgenol Radium Ther Nucl Med 1975, 123, 74–83. [Google Scholar] [CrossRef] [PubMed]
- Gollub, M.J.; Schwartz, M.B.; Shia, J. Scirrhous metastases to the gastrointestinal tract at CT: the malignant target sign. AJR Am J Roentgenol 2009, 192, 936–40. [Google Scholar] [CrossRef] [PubMed]
- Balthazar, E.J. CT of the gastrointestinal tract: principles and interpretation. AJR Am J Roentgenol 1991, 156, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Balthazar, E.J.; Siegel, S.E.; Megibow, A.J.; Scholes, J.; Gordon, R. CT in patients with scirrhous carcinoma of the GI tract: imaging findings and value for tumor detection and staging. AJR Am J Roentgenol 1995, 165, 839–45. [Google Scholar] [CrossRef] [PubMed]
- Laufman, H.; Saphir, O. Primary linitis plastica type of carcinoma of the colon. AMA Arch Surg 1951, 62, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Ha, H.K.; Cho, K.S.; Yu, E.; Kim, J.C.; Yoo, C.S.; et al. CT features of primary colorectal signet-ring cell carcinoma. J Comput Assist Tomogr 2001, 25, 225–30. [Google Scholar] [CrossRef] [PubMed]
- Rao, T.R.; Hambrick, E.; Abcarian, H.; Salgia, K.; Recant, W.M. Colorectal linitis plastica. Dis Colon Rectum 1982, 25, 239–44. [Google Scholar] [CrossRef] [PubMed]
- Brosnan, C.; Hannan, E.; Duggan, W.; Mullen, D.; Buckley, M.; Stafford, A.T. Primary colorectal linitis plastica presenting as rapid acute deterioration: a diagnostic dilemma. Ann R Coll Surg Engl 2020, 102, e187–9. [Google Scholar] [CrossRef] [PubMed]
- Shirouzu, K.; Isomoto, H.; Morodomi, T.; Ogata, Y.; Akagi, Y.; Kakegawa, T. Primary linitis plastica carcinoma of the colon and rectum. Cancer 1994, 74, 1863–8. [Google Scholar] [CrossRef] [PubMed]
- Yen, H.H.; Chen, Y.Y.; Soon, M.S. Primary linitis plastica of the jejunum. Gastrointest Endosc 2006, 63, 503–4. [Google Scholar] [CrossRef] [PubMed]
- Raskin, M.M.; Viamonte, M.; Viamonte, M.Jr. Primary linitis plastica carcinoma of the colon. Radiology 1974, 11, 17–22. [Google Scholar] [CrossRef] [PubMed]
- Papp, J.P.Jr.; Levine, E.J.; Thomas, F.B. Primary linitis plastica carcinoma of the colon and rectum. Am J Gastroenterol 1995, 90, 141–5. [Google Scholar] [PubMed]
- Jang, H.J.; Lim, H.K.; Kim, H.S.; Cho, E.Y.; Lee, S.J.; Kim, K.A.; et al. Intestinal metastases from gastric adenocarcinoma: helical CT findings. J Comput Assist Tomogr 2001, 25, 61–7. [Google Scholar] [CrossRef] [PubMed]
- Fisher, E.R.; Brown, C.H. Linitis plastica carcinoma of the stomach with extensive metastases simulating a colonic lesion. Gastroenterology 1952, 20, 503–8. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Usui, Y.; Matsukawa, M.; Yamada, S.; Negoro, T.; Kan, T.; et al. An autopsy case of gastric metastasis simulating linitis plastica carcinoma from primary linitis plastica carcinoma of the rectu]. Gan No Rinsho 1988, 34, 1996–2001. [Google Scholar] [PubMed]
- Eljabu, W.; Finch, G.; Nottingham, J.; Vaingankar, N. Metastatic deposits of breast lobular carcinoma to small bowel and rectum. Int J Breast Cancer 2011, 2011, 413949. [Google Scholar] [CrossRef] [PubMed]
- Dixon, J.M.; Anderson, T.J.; Page, D.L.; Lee, D.; Duffy, S.W. Infiltrating lobular carcinoma of the breast. Histopathology 1982, 6, 149–61. [Google Scholar] [CrossRef] [PubMed]
- Lamovec, J.; Bracko, M. Metastatic pattern of infiltrating lobular carcinoma of the breast: an autopsy study. J Surg Oncol 1991, 48, 28–33. [Google Scholar] [CrossRef] [PubMed]
- Winston, C.B.; Hadar, O.; Teitcher, J.B.; Caravelli, J.F.; Sklarin, N.T.; Panicek, D.M.; et al. Metastatic lobular carcinoma of the breast: patterns of spread in the chest, abdomen, and pelvis on CT. AJR Am J Roentgenol 2000, 175, 795–800. [Google Scholar] [CrossRef] [PubMed]
- El-Hage, A.; Ruel, C.; Afif, W.; Wissanji, H.; Hogue, J.C.; Desbiens, C.; et al. Metastatic pattern of invasive lobular carcinoma of the breast-Emphasis on gastric metastases. J Surg Oncol 2017, 115, 359. [Google Scholar] [CrossRef] [PubMed]
- McCart Reed, A.E.; Kutasovic, J.R.; Lakhani, S.R.; Simpson, PT. Invasive lobular carcinoma of the breast: morphology, biomarkers and ’omics. Breast Cancer Res 2015, 30, 17:12. [Google Scholar]
- McLemore, E.C.; Pockaj, B.A.; Reynolds, C.; Gray, R.J.; Hernandez, J.L.; Grant, C.S.; et al. Breast cancer: presentation and intervention in women with gastrointestinal metastasis and carcinomatosis. Ann Surg Oncol 2005, 12, 886–94. [Google Scholar] [CrossRef] [PubMed]
- Ambroggi, M.; Stroppa, E.M.; Mordenti, P.; Biasini, C.; Zangrandi, A.; Michieletti, E.; et al. Metastatic breast cancer to the gastrointestinal tract: report of five cases and review of the literature. Int J Breast Cancer, 2012. [Google Scholar]
- Janjic, O.; Labgaa, I.; Hübner, M.; Demartines, N.; Joliat, G.R. Metastasis to the rectum: A systematic review of the literature. Eur J Surg Oncol 2022, 48, 822–33. [Google Scholar] [CrossRef] [PubMed]
- Lior, T.; Chin Ng, S.; Ng, M.K. A rare case of linitis plastica of the colon from ovarian carcinoma. J Surg Case Rep 2019, 2019(3), rjz089. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.K.; Jee, K.R.; Yu, E.; Yu, C.S.; Rha, S.E.; Lee, I.J.; et al. CT features of metastatic linitis plastica to the rectum in patients with peritoneal carcinomatosis. AJR Am J Roentgenol 2000, 174, 463–6. [Google Scholar] [CrossRef] [PubMed]
- Fernet, P.; Azar, H.A.; Stout, A.P. Intramural (tubal) spread of linitis plastica along the alimentary tract. Gastroenterology 1965, 48, 419–24. [Google Scholar] [CrossRef] [PubMed]
- Burgain, C.; Germain, A.; Bastien, C.; Orry, X.; Choné, L.; Claudon, M.; et al. Computed tomography features of gastrointestinal linitis plastica: spectrum of findings in early and delayed phase imaging. Abdom Radiol 2016, 41, 1370–7. [Google Scholar] [CrossRef] [PubMed]
- Lau, L.C.; Wee, B.; Wang, S.; Thian, Y.L. Metastatic breast cancer to the rectum: A case report with emphasis on MRI features. Medicine 2017;96,e6739.
- Rudralingam, V.; Dobson, M.J.; Pitt, M.; Stewart, D.J.; Hearn, A.; Susnerwala, S. MR imaging of linitis plastica of the rectum. AJR Am J Roentgenol 2003, 181, 428–30. [Google Scholar] [CrossRef] [PubMed]
- Mazza, S.; Laurenza, C.; Elvo, B.; Tanzi, G.; Ungari, M.; Soro, S.; et al. Rectal linitis plastica as the first presentation of metastatic lobular breast cancer: an endoscopic ultrasound diagnosis. Clin J Gastroenterol 2022, 15, 1072–7. [Google Scholar] [CrossRef] [PubMed]
- Mommersteeg, M.C.; Kies, D.A.; van der Laan, J.; Wonders, J. Linitis plastica of the rectum secondary to prostate carcinoma. BMJ Case Rep 2022, 15. [Google Scholar] [CrossRef] [PubMed]
- Dresen, R.C.; Beets, G.L.; Rutten, H.J.T.; Engelen, S.M.E.; Lahaye, M.J.; Vliegen, R.F.A.; et al. Locally advanced rectal cancer: MR imaging for restaging after neoadjuvant radiation therapy with concomitant chemotherapy. Part I. Are we able to predict tumor confined to the rectal wall? Radiology 2009, 252, 71–80. [Google Scholar] [CrossRef] [PubMed]
- Franceschini, G.; Manno, A.; Mulè, A.; Verbo, A.; Rizzo, G.; Sermoneta, D.; et al. Gastro-intestinal symptoms as clinical manifestation of peritoneal and retroperitoneal spread of an invasive lobular breast cancer: report of a case and review of the literature. BMC Cancer 2006, 19, 193. [Google Scholar] [CrossRef] [PubMed]
- Ruymbeke, H.; Harlet, L.; Stragier, B.; Steenkiste, E.; Ryckx, M.; Marolleau, F. Anorectal metastasis from breast carcinoma: a case report and review of the literature. BMC Res Notes 2018, 11, 268. [Google Scholar] [CrossRef] [PubMed]
- Taal, B.G.; den Hartog Jager, F.C.; Steinmetz, R.; Peterse, H. The spectrum of gastrointestinal metastases of breast carcinoma: II. The colon and rectum. Gastrointest Endosc 1992, 38, 136–41. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Zhang, J. Gastric cancer with large bowel obstruction as the first presentation: A case report. Oncol Lett 2013, 6, 1377–9. [Google Scholar] [CrossRef] [PubMed]
- Tariq, T.; Turk, A.; Reaume, M.; Muddasani, A.; Parmar, M. Blocked by a Ring: A Case of Gastric Linitis Plastica Presenting as Large Bowel Obstruction Secondary to Rectal Stenosis. ACG Case Rep J 2019, 6:e00007.2. 57.
- Benfiguig, A.; Anciaux, M.L.; Eugène, C.I.; Benkémoun, G.; Etienne, J.C. Gastric metastasis of breast cancer occurring after a cancer-free interval of 30 years. Ann Gastroenterol Hepatol 1992, 28, 175–7. [Google Scholar]
- Fizazi, K.; Greco, F.A.; Pavlidis, N.; Daugaard, G. ; Oien, K; ; Pentheroudakis, G. et al. Cancers of unknown primary site: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2015 26 Suppl 5,v133–8. [Google Scholar]
- Fayemi, A.O.; Ali, M.; Braun, E.V. Metastatic carcinoma simulating linitis plastica of the colon. A case report. Am J Gastroenterol 1979, 71, 311–4. [Google Scholar] [PubMed]
- Meyers, M.A. Intraperitoneal spread of malignancies and its effect on the bowel. Clin Radiol 1981, 32, 129–46. [Google Scholar] [CrossRef] [PubMed]
- Haberstich, R.; Tuech, J.J.; Wilt, M.; Rodier, J.F. Anal localization as first manifestation of metastatic ductal breast carcinoma. Tech Coloproctol 2005, 9, 237–8. [Google Scholar] [CrossRef] [PubMed]
- Harris, M.; Howell, A.; Chrissohou, M.; Swindell, R.I.; Hudson, M.; Sellwood, R.A. A comparison of the metastatic pattern of infiltrating lobular carcinoma and infiltrating duct carcinoma of the breast. Br J Cancer 1984, 5, :23–30. [Google Scholar] [CrossRef] [PubMed]
- Ferri, L.E.; Onerheim, R.; Emond, C. Linitis plastica as the first indication of metastatic lobular carcinoma of the breast: case report and literature review. Can J Surg 1999, 42, 466–9. [Google Scholar] [PubMed]
- Hsieh, P.S; Yeh, C.Y.; Chen, J.R. Changchien, CR. Ileocecal breast carcinoma metastasis. Int J Colorectal Dis 2004, 19, 607–8. [Google Scholar] [CrossRef] [PubMed]
- Tot, T. The role of cytokeratins 20 and 7 and estrogen receptor analysis in separation of metastatic lobular carcinoma of the breast and metastatic signet ring cell carcinoma of the gastrointestinal tract. APMIS 2000, 108, 467–72. [Google Scholar] [CrossRef] [PubMed]
- Levine, M.S.; Pantongrag-Brown, L.; Aguilera, N.S.; Buck, J.L.; Buetow, P.C. Non-Hodgkin lymphoma of the stomach: a cause of linitis plastica. Radiology 1996, 201, 375–8. [Google Scholar] [CrossRef] [PubMed]
- Ozyilkan, O.; Ozyilkan, E. ; Gastric Hodgkin’s disease presenting with radiological appearance of linitis plastica. Am J Gastroenterol 1999, 94, 3661–2. [Google Scholar] [CrossRef] [PubMed]
- Lo Re, G.; Federica, V.; Midiri, F.; Picone, D.; La Tona, G.; Galia, M.; et al. Radiological Features of Gastrointestinal Lymphoma. Gastroenterol Res Pract 2016, 2016, 2498143. [Google Scholar] [CrossRef] [PubMed]
- Cervia, J.S. Gastric Kaposi’s sarcoma without skin lesions presenting as linitis plastica. Gastrointest Endosc 1992, 38, 96. [Google Scholar] [CrossRef] [PubMed]
- Hadjiyane, C.; Lee, Y.H.; Stein, L.; Jayagopal, S.; Shih, H.; Pellecchia, C. Kaposi’s sarcoma presenting as linitis plastica. Am J Gastroenterol 1991, 86, 1823–5. [Google Scholar] [PubMed]
- Murray, J.G.; Evans, S.J.; Jeffrey, P.B.; Halvorsen, R.A.Jr. Cytomegalovirus colitis in AIDS: CT features. AJR Am J Roentgenol 1995, 165, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Abdu, R.A.; Carter, K.; Pomidor, W.J. Gastric syphilis mimicking linitis plastica. Arch Surg 1993, 128, 103–4. [Google Scholar] [CrossRef] [PubMed]
- Rubesin, S.E.; Levine, M.S.; Laufer, I. Double-contrast upper gastrointestinal radiography: a pattern approach for diseases of the stomach. Radiology 2008, 246, 33–48. [Google Scholar] [CrossRef] [PubMed]
- Calafat, P.; de Diller, A.B.; Sanchez, C. Breast carcinoma metastasis in ileum-colon and gallbladder simulating inflammatory diseases. Rev Fac Cien Med Univ Nac Cordoba 1999, 56, 123–7. [Google Scholar] [CrossRef] [PubMed]
- Macari, M.; Balthazar, E.J. CT of bowel wall thickening: significance and pitfalls of interpretation. AJR Am J Roentgenol 2001, 176, 1105–16. [Google Scholar] [CrossRef] [PubMed]
- Macari, M.; Megibow, A.J.; Balthazar, E.J. A pattern approach to the abnormal small bowel: observations at MDCT and CT enterography. AJR Am J Roentgeno 2007, 188, 1344–55. [Google Scholar] [CrossRef] [PubMed]
- DiPiro, P.J.; Tirumani, S.H.; Cruz, G.P.; Ramaiya, N.H.; Lester, S.C.; Shinagare, A.B. Lobular breast cancer: patterns of intraabdominal metastatic spread on imaging and prognostic significance. Abdom Radiol 2019, 44, 362–9. [Google Scholar] [CrossRef] [PubMed]
- Hristova, L.; Soyer, P.; Hoeffel, C.; Marteau, P.; Oussalah, A.; Lavergne-Slove, A.; et al. Colorectal cancer in inflammatory bowel diseases: CT features with pathological correlation. Abdom Imaging 2013, 38, 421–35. [Google Scholar] [CrossRef] [PubMed]
- Dória, M.T.; Maesaka, J.Y.; Filho, S.N.; Silveira, T.P.; Boufelli, G.; Siqueira, S.A.C.; et al. Gastric metastasis as the first manifestation of an invasive lobular carcinoma of the breast. Autops Case Rep 2015, 5, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Manohar, P.M.; Davidson, N.E. Updates in endocrine therapy for metastatic breast cancer. Cancer Biol Med 2021, 19, 202–12. [Google Scholar] [CrossRef] [PubMed]
- Ethier, SP. Using functional genomics and artificial intelligence to reverse engineer human cancer cells; Cambridge Scholars Publishing: Newcastle upon Tyne, UK; 2023; p. 161. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



