Submitted:
07 July 2025
Posted:
08 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Patient Demographics Overall
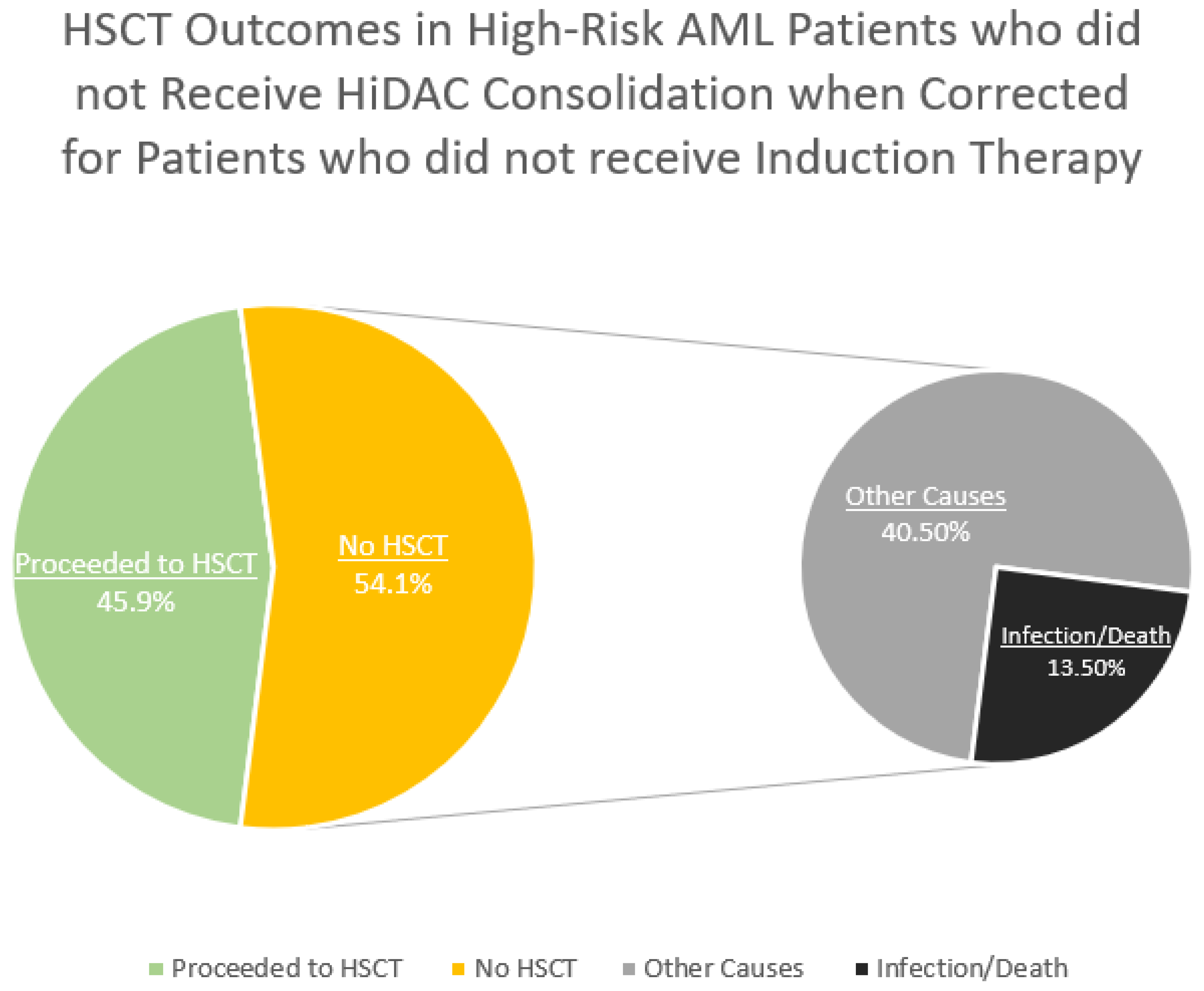
3.2. Characteristics of Patients Who Received Induction Therapy Versus Those Who Did Not
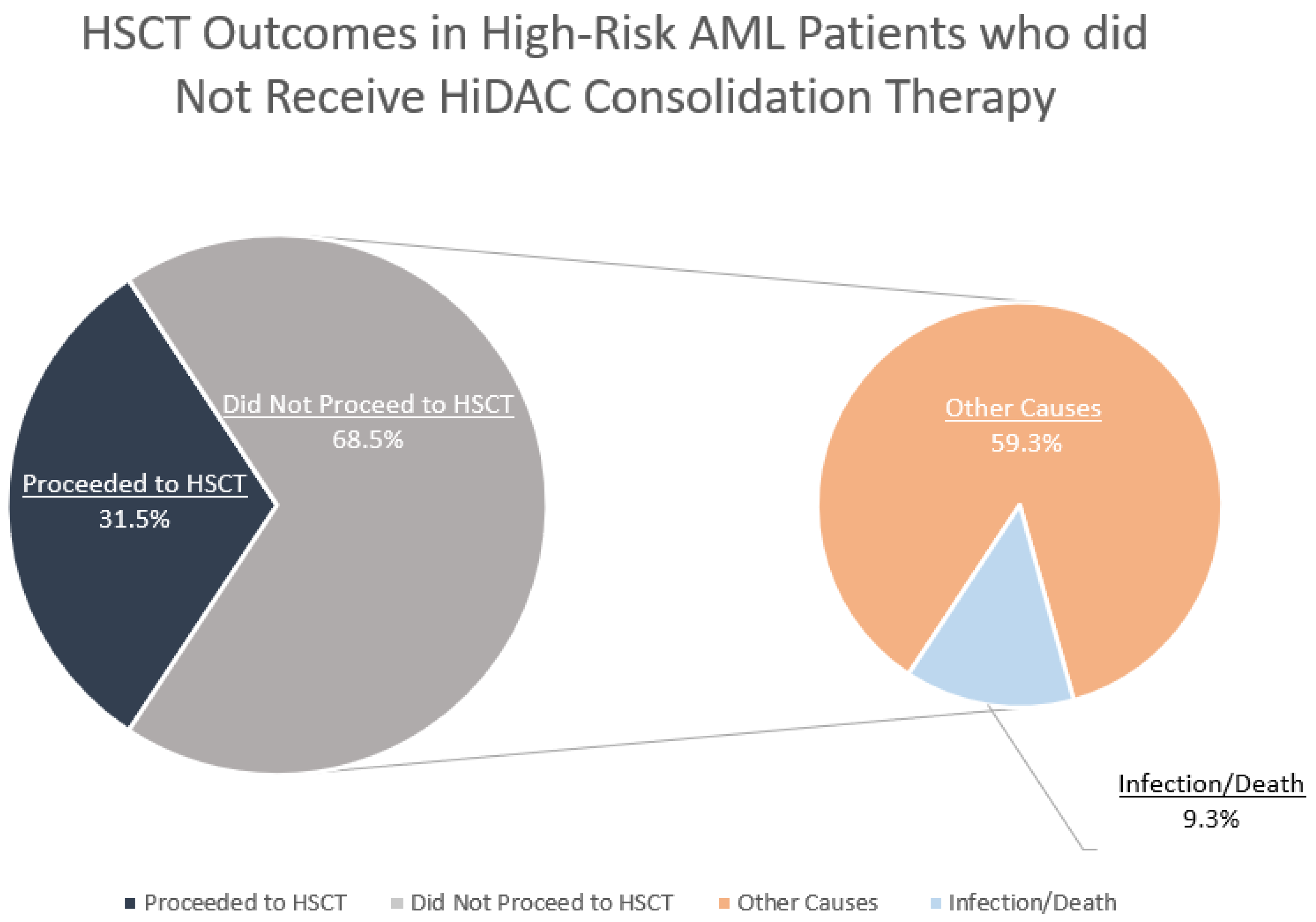
3.3. Characteristics of Patients Who Did Not Receive HiDAC Consolidation Therapy and Reasons for no HSCT
3.4. Comparing Patients Who Did Not Receive HiDAC Consolidation Therapy Based on Their HSCT Status
3.5. Characteristics of Patients Who Did Receive HiDAC Consolidation Therapy
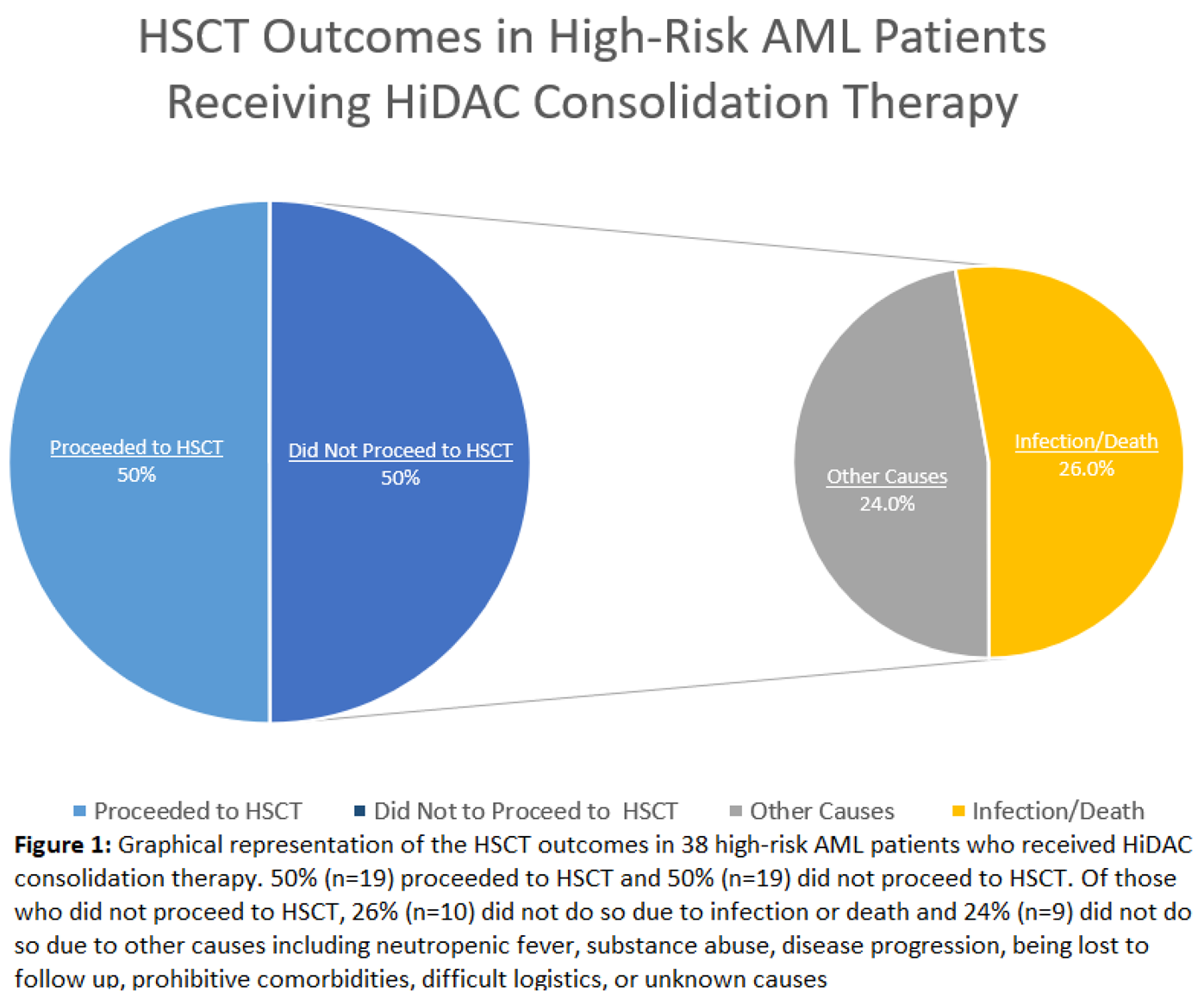
3.6. Characteristics of Patients Who Received HiDAC Consolidation and Reasons for Not Undergoing HSCT
3.7. Characteristics of Patients Who Received IDAC Consolidation Therapy
3.8. Comparing Time from Diagnosis to HSCT in Patients Who Did Versus Did Not Receive HiDAC Consolidation Therapy
3.2. Figures, Tables and Schemes
| Age (Range, Median, Mean,) | Race | Sex |
| Entire Cohort (n=92) | ||
| Range: 14-85 | White: 64 (69.6%) | Male: 43 (46.7%) |
| Median: 60 | Black: 25 (27.2%) | Female: 49 (53.3%) |
| Mean: 55.3 | Hispanic: 1 (1.1%) | |
| Other: 2 (2.1%) | ||
| Patients who received Induction: n=75 (81.5%) | ||
| Range: 14-77 | White: 52 (69.3%) | Male: 31 (41.3%) |
| Median: 55 | Black: 20 (26.7%) | Female: 44 (58.7%) |
| Mean: 50.8 | Hispanic: 1 (1.3%) | |
| Other: 2 (2.7%) | ||
| Patients who did not receive Induction: n=17 (18.5%) | ||
| Range: 64-85 | White: 12 (70.5%) | Male: 12 (70.5%) |
| Median: 77 | Black: 5 (29.5%) | Female: 5 (29.5%) |
| Mean: 75.4 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| No HiDAC Consolidation: n=54 (58.7%) | ||
| Range: 14-85 | White: 38 (70.3%) | Male: 29 (53.7%) |
| Median: 64.5 | Black: 14 (25.9%) | Female: 25 (46.3%) |
| Mean: 58.9 | Hispanic: 0 (0%) | |
| Other: 2 (3.8%) | ||
| No HiDAC Consolidation, with HSCT: n=17 (18.5%) | ||
| Range: 14-71 | White: 14 (82.4%) | Male: 7 (41.2%) |
| Median: 42 | Black: 3 (17.6%) | Female: 10 (58.8%) |
| Mean: 44.3 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| No HiDAC Consolidation or HSCT: n=37 (40.2%) | ||
| Range: 26-85 | White: 24 (64.9%) | Male: 22 (59.5%) |
| Median: 68 | Black: 11 (29.7%) | Female: 15 (40.5%) |
| Mean: 65.7 | Hispanic: 0 (0%) | |
| Other: 2 (5.4%) | ||
| HiDAC Consolidation: n=38 (41.3%) | ||
| Range: 19-70 | White: 26 (68.4%) | Male: 14 (36.8%) |
| Median: 53 | Black: 11 (28.9%) | Female: 24 (63.2%) |
| Mean: 50.1 | Hispanic: 1 (2.7%) | |
| Other: 0 (0%) | ||
| HiDAC with HSCT: n=19 (20.7%) | ||
| Range: 19-65 | White: 14 (73.7%) | Male: 6 (31.6%) |
| Median: 53 | Black: 5 (26.3%) | Female: 13 (68.4%) |
| Mean: 47.0 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| HiDAC without HSCT due to Infection or Death: n=10 (10.9%) | ||
| Range: 29-70 | White: 6 (60.0%) | Male: 2 (20.0%) |
| Median: 57 | Black: 3 (30.0%) | Female: 8 (80.0%) |
| Mean: 54.6 | Hispanic: 1 (10.0%) | |
| Other: 0 (0%) | ||
| HiDAC without HSCT due to Other Causes: n=9 (9.8%) | ||
| Range: 24-63 | White: 6 (66.7%) | Male: 6 (66.7%) |
| Median: 56 | Black: 3 (33.3%) | Female: 3 (33.3%) |
| Mean: 51.8 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| IDAC Total: n=5 (5.4%) | ||
| Range: 27-69 | White: 5 | Male: 2 (40.0%) |
| Median: 62 | Black: 0 | Female: 3 (60.0%) |
| Mean: 50.2 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| IDAC with HSCT: n=3 (3.3%) | ||
| Range: 27-63 | White: 3 (100%) | Male: 1 (33.3%) |
| Median: 30 | Black: 0 (0%) | Female: 2 (66.7%) |
| Mean: 40 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| IDAC without HSCT: n=2 (2.2%) | ||
| Range: 62-69 | White: 2 (100%) | Male: 1 (50.0%) |
| Median: 65.5 | Black: 0 (0%) | Female: 1 (50.0%) |
| Mean: 65.5 | Hispanic: 0 (0%) | |
| Other: 0 (0%) | ||
| Table 1: Patient characteristics table including age, race, and sex of the High-Risk AML patients treated at the Georgia Cancer Center at Augusta University between November 2003 and December 2020. Patients were stratified based on whether they received induction therapy, HiDAC consolidation therapy, or HSCT. Patients who received HiDAC but did not undergo HSCT were further stratified based on the reasons why HSCT was not performed including infection/death and other causes. Characteristics of patients who received IDAC therapy are also listed. | ||
| Induction Therapy (n=75) | No Induction Therapy (n=17) | ||
| Age (Mean) | 50.8 | 75.4 | P<0.0000 |
| Race | |||
| White | 52 | 12 | P=0.5450 |
| Black | 20 | 5 | |
| Hispanic | 1 | 0 | |
| Other | 2 | 0 | |
| Sex | |||
| Male | 31 | 12 | P=0.0012 (Fischer Exact) |
| Female | 44 | 5 | |
| Table 2: Comparison of mean age, race demographics, and sex in those who received induction therapy versus those who did not. Hispanic and “Other” races were not included in the analysis due to low sample size. P-value comparing sex breakdown is a Fischer exact value. | |||
| No HiDAC, No HSCT (n=37) | No HiDAC, with HSCT (n=17) | ||||
| Age (Mean) | 65.7 | 44.3 | P=0.0002 | ||
| Race | |||||
| White | 24 | 14 | P=0.4082 | ||
| Black | 11 | 3 | |||
| Hispanic | 0 | 0 | |||
| Other | 2 | 0 | |||
| Sex | |||||
| Male | 22 | 7 | P=0.1997 | ||
| Female | 15 | 10 | |||
| Table 3: Comparison of mean age, race demographics, and sex in those who did not receive HiDAC therapy or HSCT versus those who did not receive HiDAC but did get HSCT. Hispanic and “Other” races were not included in the analysis due to low sample size. | |||||
| HiDAC (n=38) | No HiDAC (n=54) | ||
| Age (Median) | 53 | 64.5 | P=0.0603 |
| Race | |||
| White | 26 | 38 | P=0.4054 |
| Black | 11 | 14 | |
| Hispanic | 1 | 0 | |
| Other | 0 | 2 | |
| Sex | |||
| Male | 14 | 29 | P=0.1414 |
| Female | 24 | 25 | |
| Table 4: Comparison of median age, race demographics, and sex in those who received HiDAC therapy versus those who did not. Hispanic and “Other” races were not included in the analysis due to low sample size. | |||
| HiDAC | No HiDAC | ||
| Time in Days | *P=0.00416 | ||
| Range | 97-1339 | 46-511 | P=0.4020 |
| Median | 362.5 | 127 | |
| Mean | 442.3 | 165.7 | |
| Table 5: Comparison of time from diagnosis to HSCT in days in patients treated with HiDAC consolidation therapy versus those who were not. *When utilizing ANOVA analysis, P value is 0.00416, which appears statistically significant. However, Barlett’s Test for Inequality of Population Variances yields a P value of 0.00002. Any P value less than 0.05 suggests that the variances are not homogenous between these two groups and that ANOVA is not appropriate. Mann-Whitney/Wilcoxon Two-Sample Test (Kruskai-Wallis test for two groups) has a non-significant P-value of 0.4020. | |||
| Reasons for Not Receiving HSCT after HiDAC | Number of Patients |
| Infections | 7 |
| Death prior to transplant | 3 |
| Prohibitive Comorbidities | 3 |
| Unknown | 2 |
| Neutropenic Fever without identifiable source | 2 |
| Drug Abuse | 1 |
| Logistics | 1 |
| Total | 19 |
| Types of Infections | Number of Patients |
| Bacteremia | |
| Klebsiella | 2 |
| Vancomycin Resistant E. Faecalis | 2 |
| E. Coli | 1 |
| Proteus | 1 |
| Group B Strep | 1 |
| Coagulase Negative Strep | 1 |
| Pneumonia | |
| Klebsiella | 1 |
| Urinary Tract Infections | |
| Extended Spectrum Beta-Lactamase | 1 |
| Proteus | 1 |
| Klebsiella | 1 |
| Fungal Sinusitis | 1 |
| Clostridium Difficile Colitis | 3 |
| Outcome of Patients Receiving IDAC Consolidation Therapy | Number of Patients |
| Able to Receive HSCT | 3 |
| Unable to Receive HSCT | 2 |
| Reasons | Number of Patients |
| Prohibitive Comorbidities/ECOG Performance Status | 17 |
| Failed Induction/Disease Progression | 12 |
| Infection/Death | 5 |
| Patient Declined | 2 |
| Lost to Follow-Up | 1 |
| Total | 37 |
| Types of Infection | Number of Patients |
| Septic Shock | |
| VRE Enterococcus Faecium | 1 |
| Unspecified | 2 |
| Pneumonia | |
| Burkholdeia | 1 |
| Unspecified | 1 |
4. Discussion
Why we use HiDAC
HiDAC and the timing of HSCT Evaluation
HiDAC Versus IDAC: Risk Versus Benefit
HiDAC Versus HSCT Alone
Is HiDAC Consolidation preventing some high-risk AML patients from receiving HSCT?
Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Institutional Board Review Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.J.; Le Beau, M.M.; Bloomfield, C.D.; Cazzola, M.; Vardiman, J.W. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [CrossRef] [PubMed]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef] [PubMed]
- Shepshelovich, D.; Edel, Y.; Goldvaser, H.; Dujovny, T.; Wolach, O.; Raanani, P. Pharmacodynamics of cytarabine induced leucopenia: a retrospective cohort study. Br. J. Clin. Pharmacol. 2015, 79, 685–691. [Google Scholar] [CrossRef] [PubMed]
- Chanswangphuwana, C.; Polprasert, C.; Owattanapanich, W.; Kungwankiattichai, S.; Rattarittamrong, E.; Rattanathammethee, T.; Limvorapitak, W.; Saengboon, S.; Niparuck, P.; Puavilai, T.; et al. Comparison of Three Doses of Cytarabine Consolidation for Intermediate- and Adverse-risk Acute Myeloid Leukemia: Real World Evidence From Thai Acute Myeloid Leukemia Registry. Clin. Lymphoma Myeloma Leuk. 2022, 22, e915–e921. [Google Scholar] [CrossRef] [PubMed]
- Ravikumar, D.; Saju, H.; Choudary, A.; Bhattacharjee, A.; Dubashi, B.; Ganesan, P.; Kayal, S. Outcomes of HIDAC 18 g Versus IDAC 9 g in Consolidation Therapy of Acute Myeloid Leukemia: A Retrospective Study. Indian J. Hematol. Blood Transfus. 2021, 38, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Tangchitpianvit, K.; Rattarittamrong, E.; Chai-Adisaksopha, C.; Piriyakhuntorn, P.; Rattanathammethee, T.; Hantrakool, S.; Tantiworawit, A.; Norasetthada, L. Efficacy and safety of consolidation therapy with intermediate and high dose cytarabine in acute myeloid leukemia patients. Hematology 2021, 26, 355–364. [Google Scholar] [CrossRef] [PubMed]
- Magina, K.N.; Pregartner, G.; Zebisch, A.; Wölfler, A.; Neumeister, P.; Greinix, H.T.; Berghold, A.; Sill, H. Cytarabine dose in the consolidation treatment of AML: a systematic review and meta-analysis. Blood 2017, 130, 946–948. [Google Scholar] [CrossRef] [PubMed]
- Hanoun, M.; Ruhnke, L.; Kramer, M.; Hanoun, C.; Schäfer-Eckart, K.; Steffen, B.; Sauer, T.; Krause, S.W.; Schliemann, C.; Mikesch, J.-H.; et al. Intensified cytarabine dose during consolidation in adult AML patients under 65 years is not associated with survival benefit: real-world data from the German SAL-AML registry. J. Cancer Res. Clin. Oncol. 2022, 149, 4611–4621. [Google Scholar] [CrossRef] [PubMed]
- Mayer, R.J. et al. (1997). Intensified Post-Remission Chemotherapy for Adults with Acute Myeloid Leukemia: An Update of CALGB 8525. In: Büchner, T., Schellong, G., Ritter, J., Creutzig, U., Hiddemann, W., Wörmann, B. (eds) Acute Leukemias VI. Haematology and Blood Transfusion / Hämatologie und Bluttransfusion, vol 38. Springer, Berlin, Heidelberg. [CrossRef]
- Wu, D.; Duan, C.; Chen, L.; Chen, S. Efficacy and safety of different doses of cytarabine in consolidation therapy for adult acute myeloid leukemia patients: a network meta-analysis. Sci. Rep. 2017, 7, 9509–9509. [Google Scholar] [CrossRef] [PubMed]
- Stasi, R.; Venditti, A.; Del Poeta, G.; Aronica, G.; Abruzzese, E.; Pisani, F.; Cecconi, M.; Masi, M.; Amadori, S. High-dose chemotherapy in adult acute myeloid leukemia: Rationale and results. Leuk. Res. 1996, 20, 535–549. [Google Scholar] [CrossRef] [PubMed]
- Zittoun, R.A.; Mandelli, F.; Willemze, R.; de Witte, T.; Labar, B.; Resegotti, L.; Leoni, F.; Damasio, E.; Visani, G.; Papa, G.; et al. Autologous or Allogeneic Bone Marrow Transplantation Compared with Intensive Chemotherapy in Acute Myelogenous Leukemia. New Engl. J. Med. 1995, 332, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Cornelissen, J.J.; Blaise, D. Hematopoietic stem cell transplantation for patients with AML in first complete remission. Blood 2016, 127, 62–70. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Gao, Q.; Du, J.; Hu, J.; Liu, X.; Zhang, F. Effects of post-remission chemotherapy before allo-HSCT for acute myeloid leukemia during first complete remission: a meta-analysis. Ann. Hematol. 2018, 97, 1519–1526. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




