Submitted:
30 June 2025
Posted:
03 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
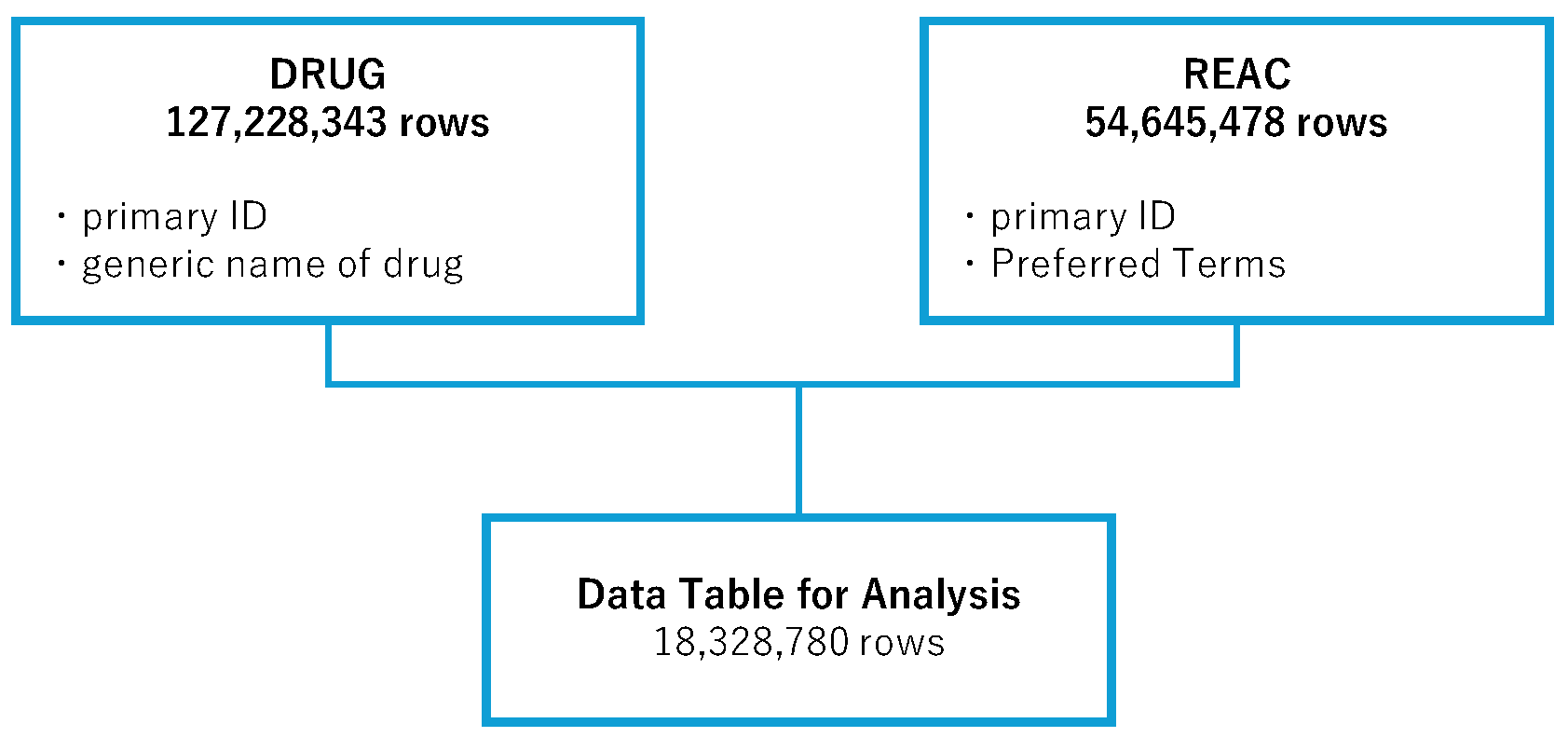
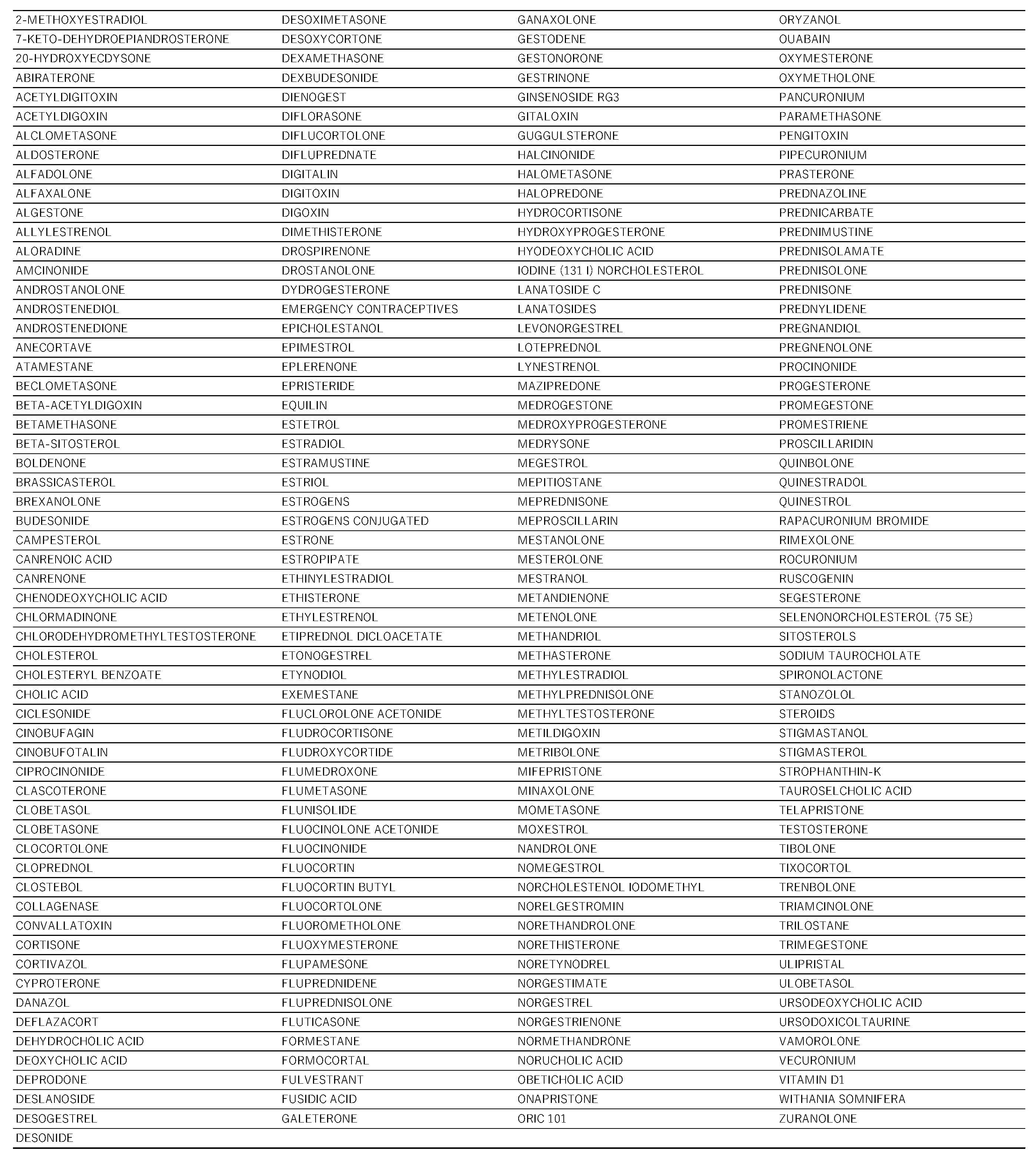
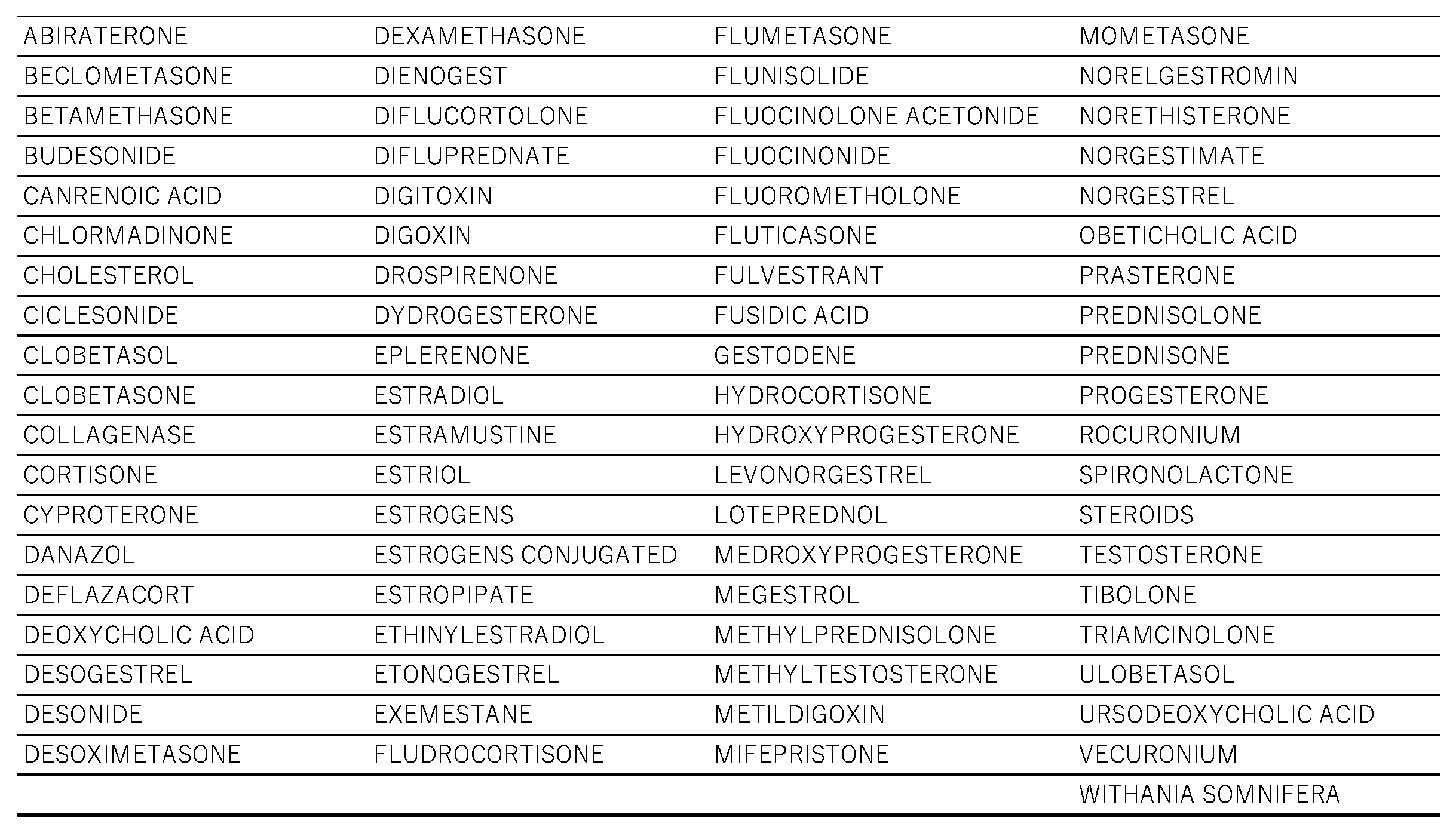
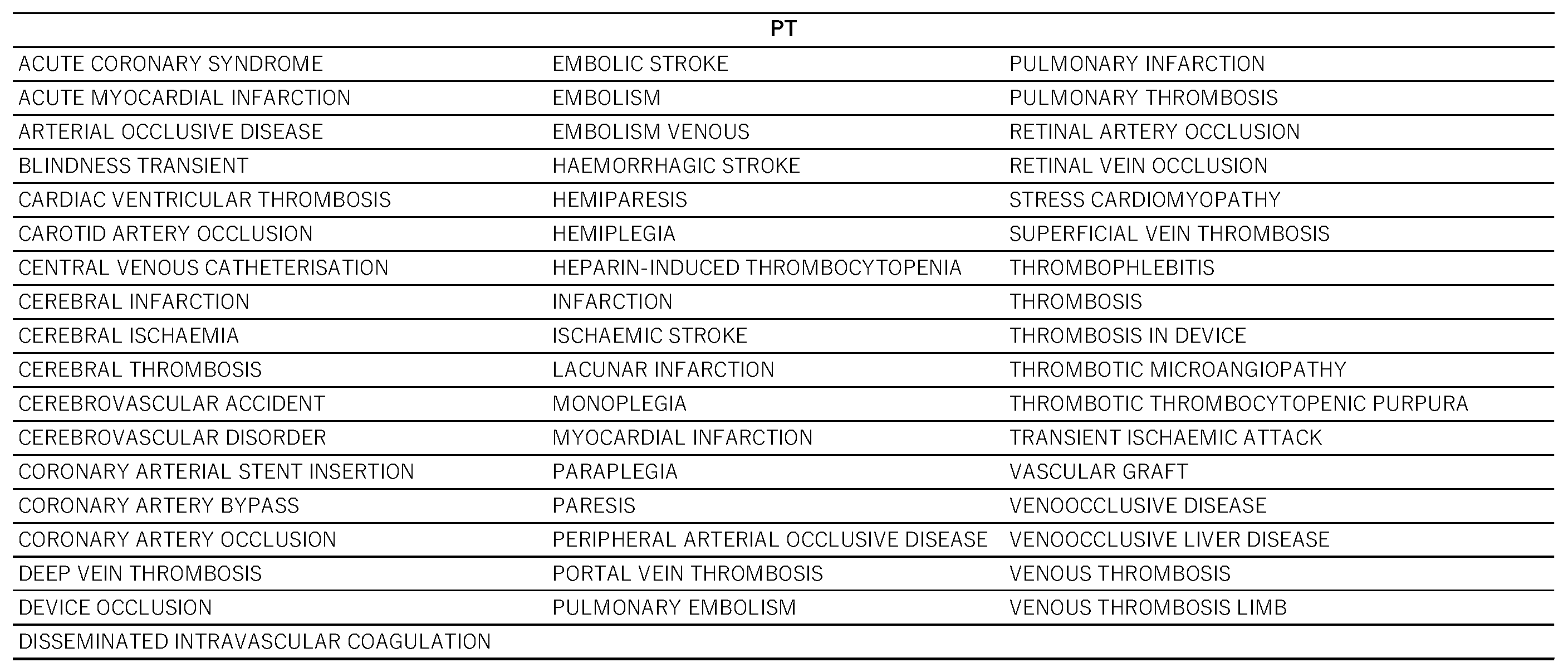
2.1. Creation of the Data Table
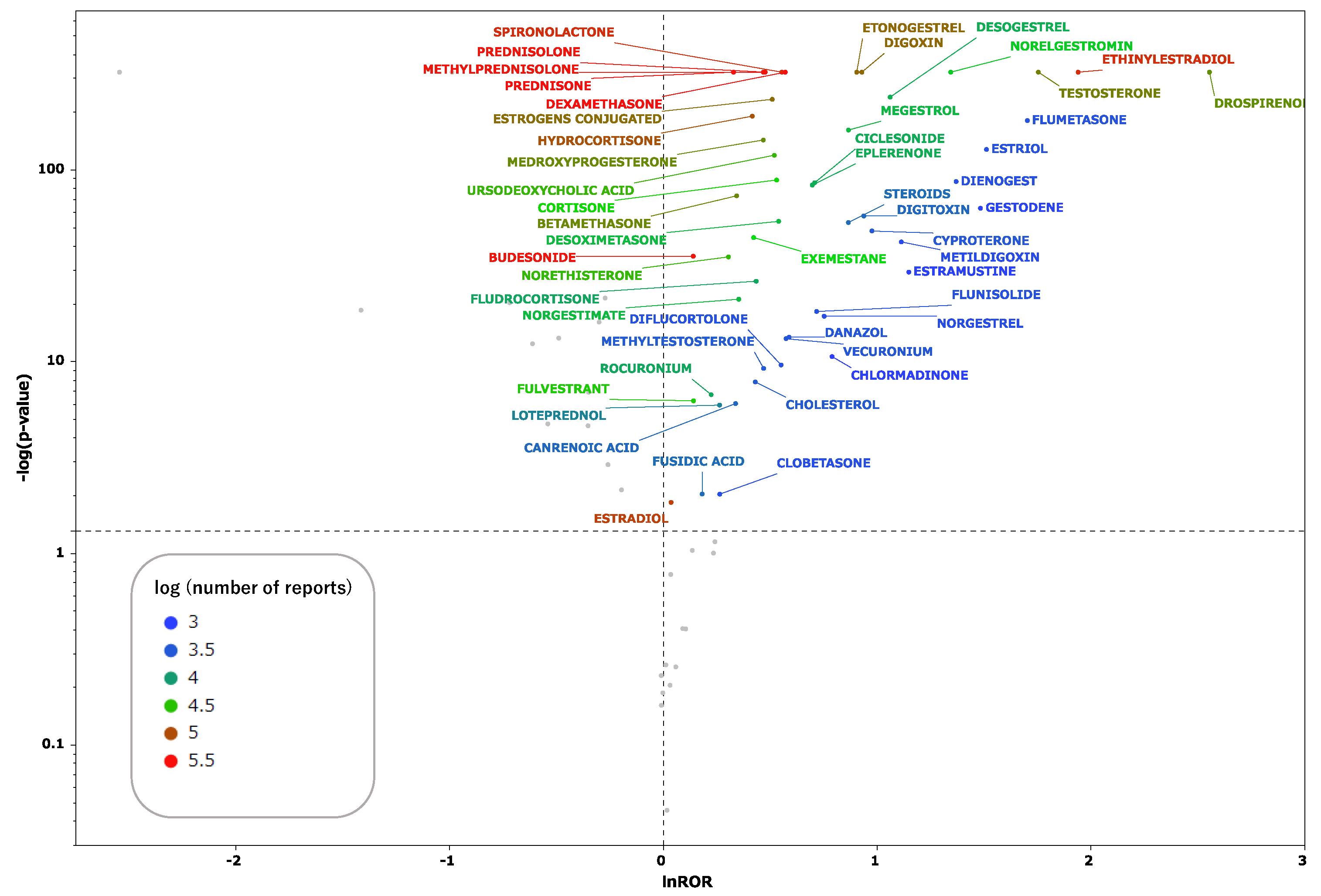
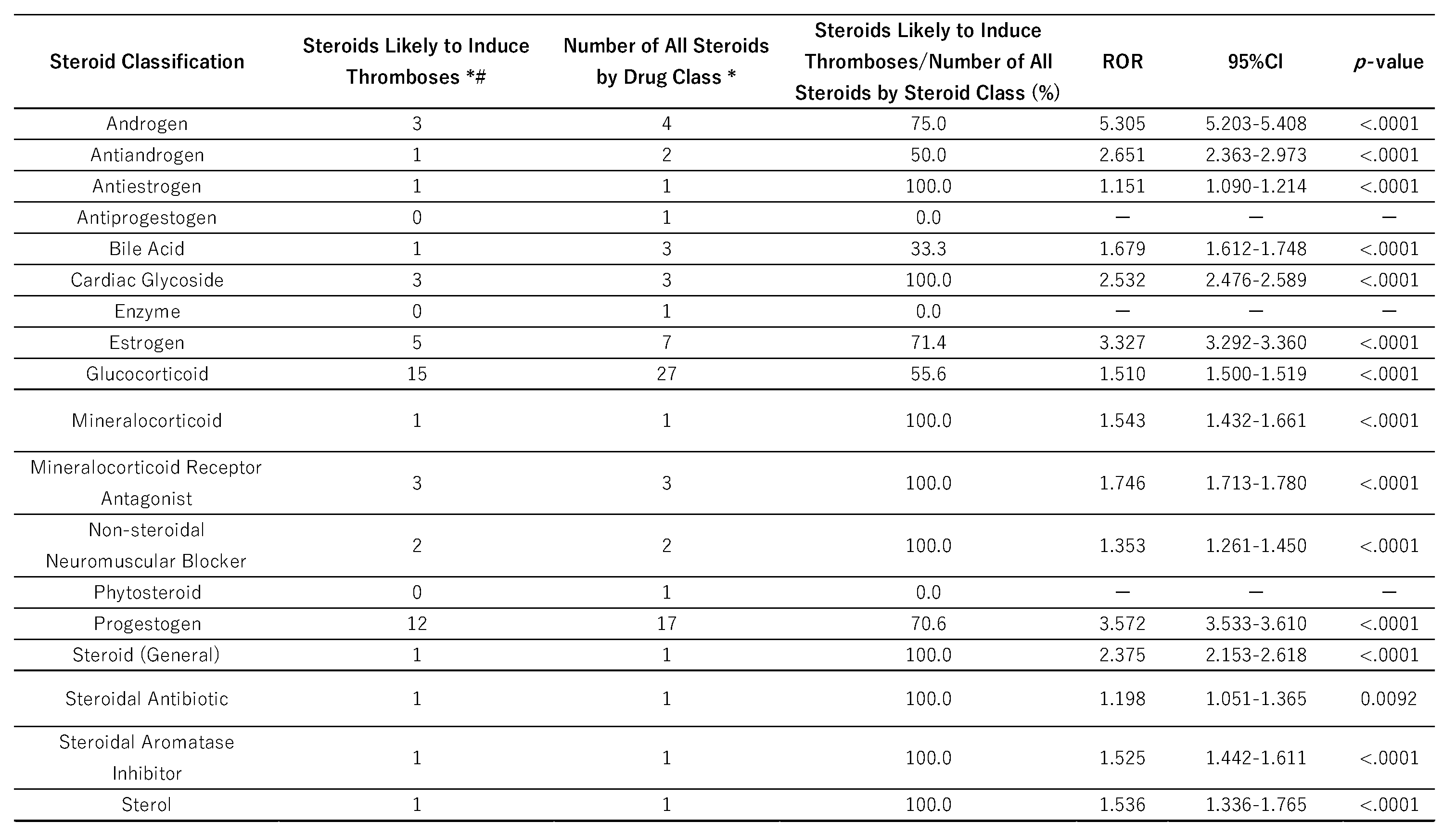
2.2. Steroid Drugs That Induce Thrombosis
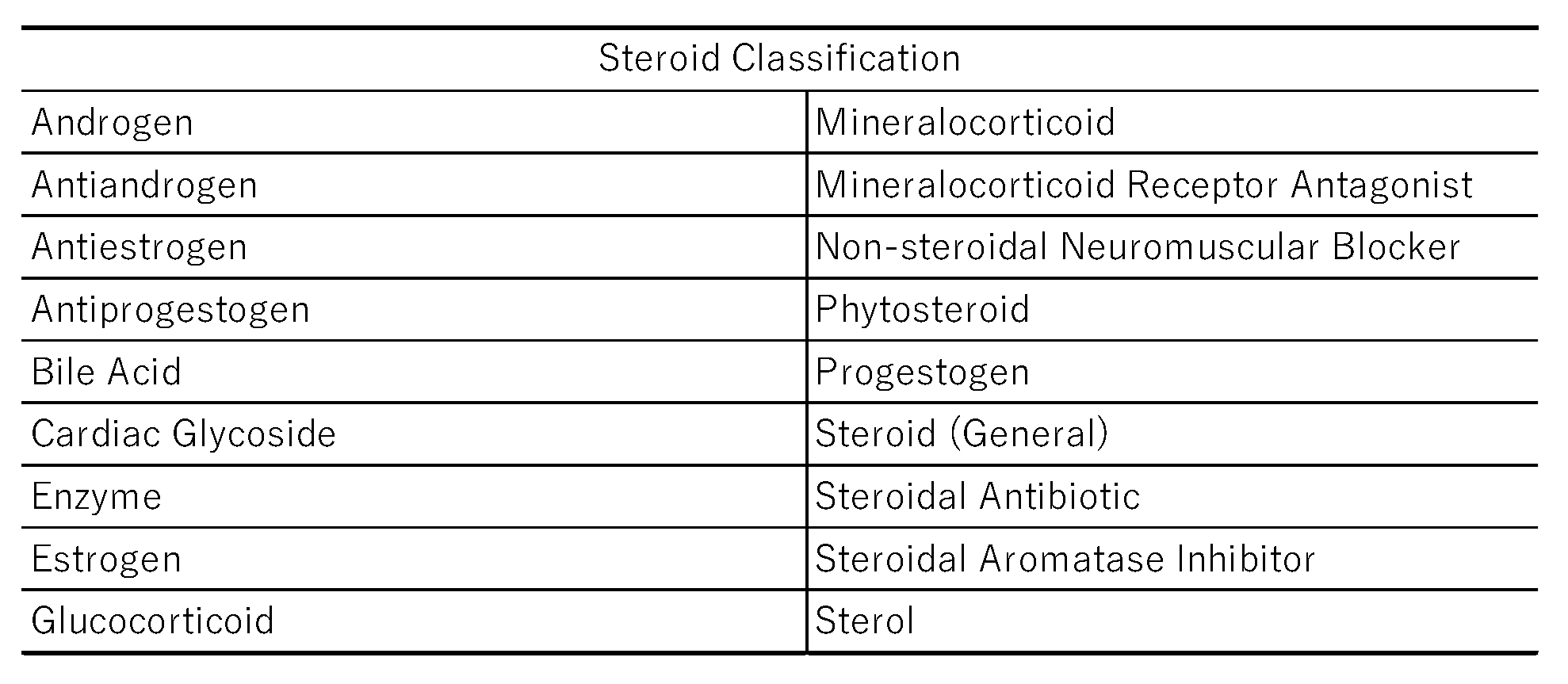
2.3. Classification of Steroid Drugs and Their Relationships to Thrombogenesis
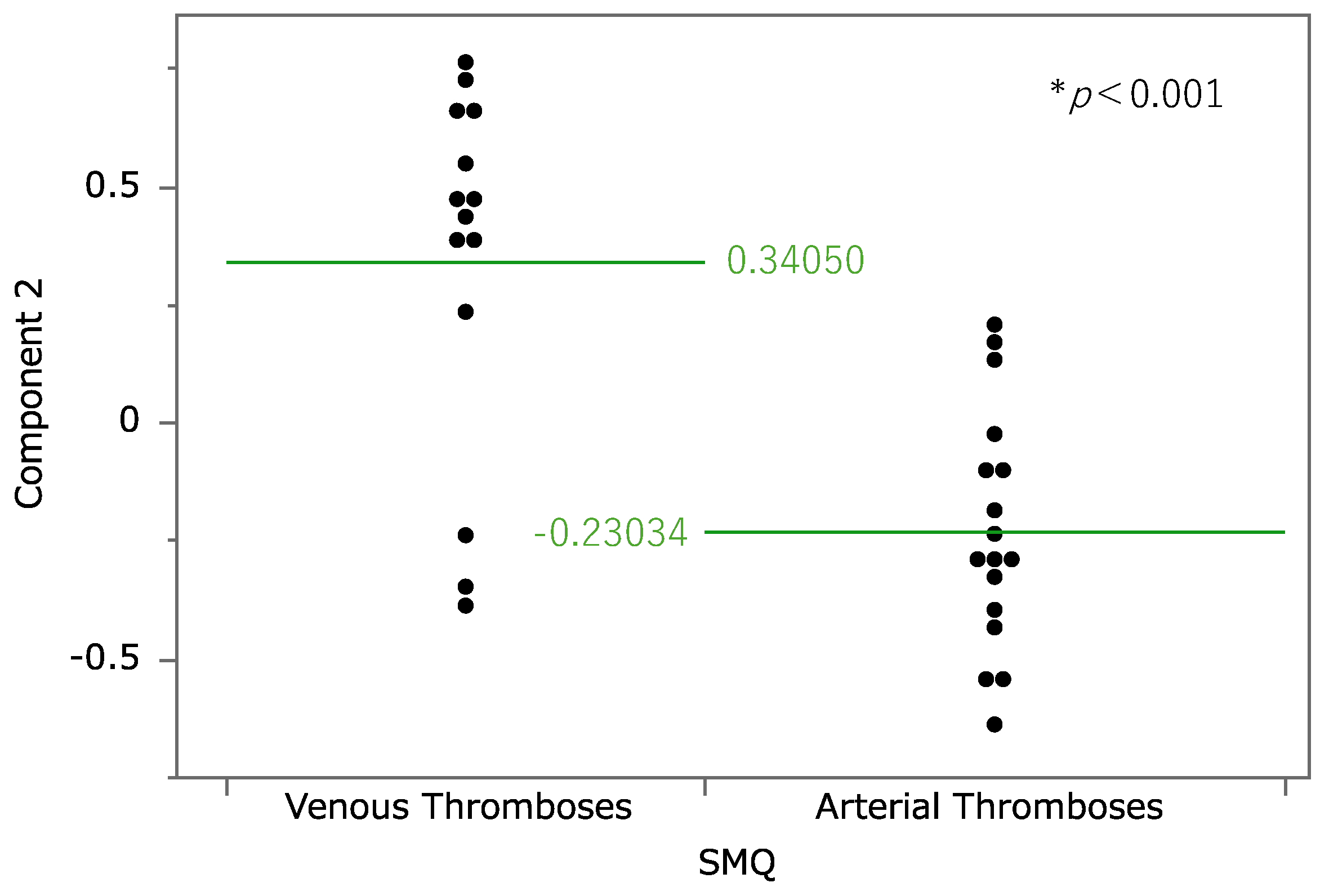
2.4. Principal Component Analysis
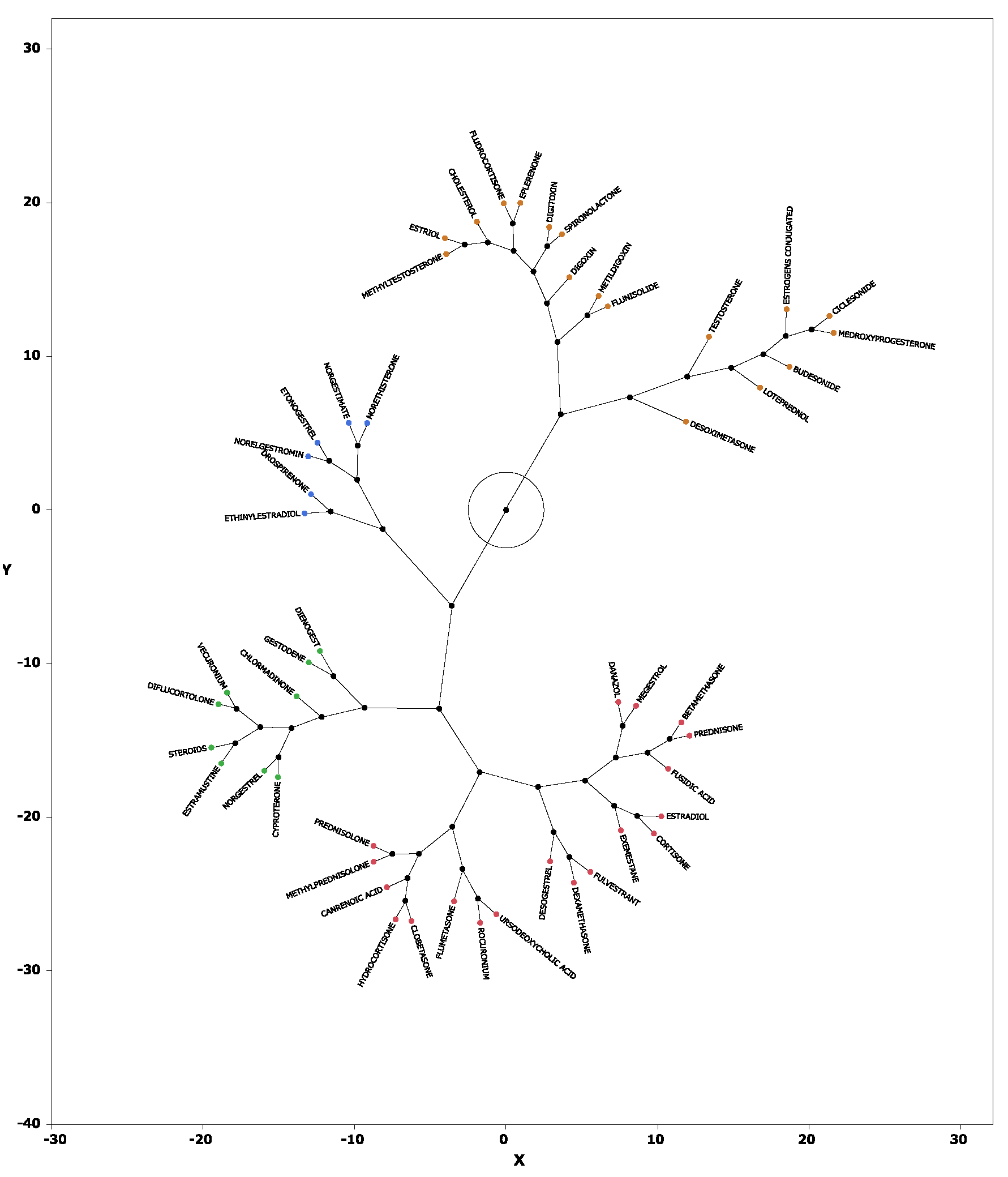
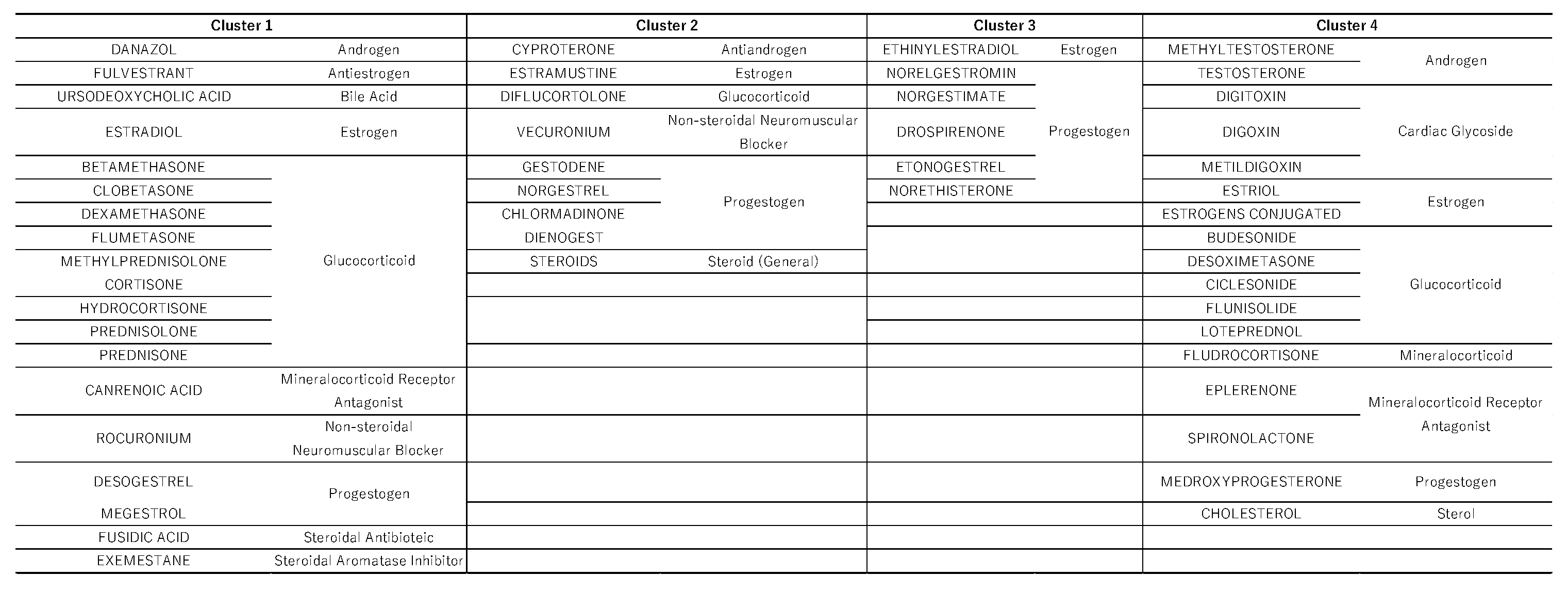
2.5. Hierarchical Cluster Analysis
3. Discussion
3.1. Classification of Steroid Drugs with High Thrombogenic Potential
3.2. Principal Component Analysis
3.3. Cluster Analysis
3.4. Study Limitations
4. Materials and Methods
4.1. FAERS Database
4.2. Selection of Target Drugs and Control of Adverse Events
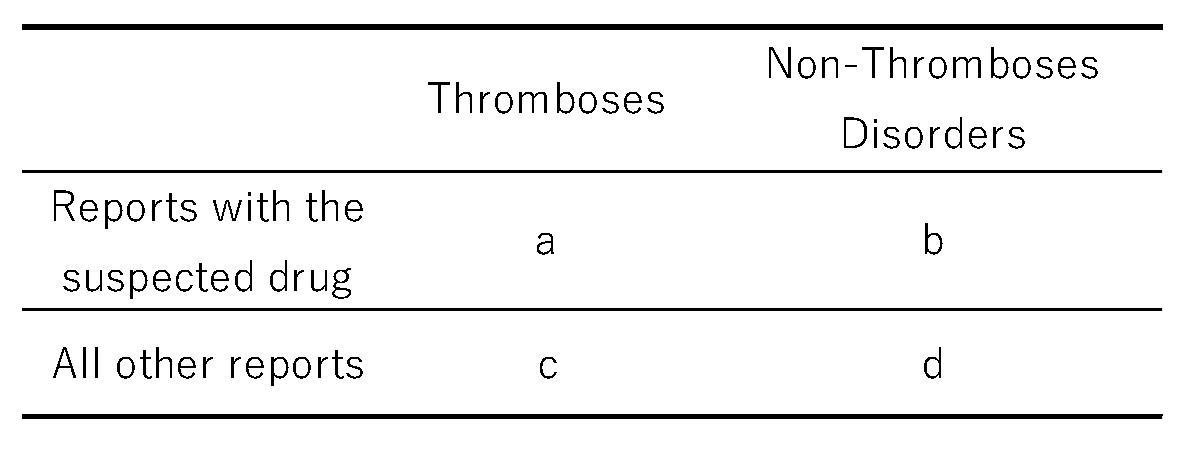
4.3. Calculation of RORs
4.5. Classification of Steroid Drugs with High Potential for Inducing Thrombosis
4.6. Principal Component Analysis
4.7. Hierarchical Cluster Analysis
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ryken, T.C.; McDermott, M.; Robinson, P.D.; Ammirati, M.; Andrews, D.W.; Asher, A.L.; Burri, S.H.; Cobbs, C.S.; Gaspar, L.E.; Kondziolka, D. The role of steroids in the management of brain metastases: a systematic review and evidence-based clinical practice guideline. J. Neurooncol. 2009, 96, 103–114.
- Möhlmann, J.E.; Ezzafzafi, S.; Lindemans, C.A.; Jansen, M.H.A.; Nierkens, S.; Huitema, A.D.R.; van Luin, M. Pharmacokinetics and pharmacodynamics of systemic corticosteroids in autoimmune and inflammatory diseases: a review of current evidence. Clin. Pharmacokinet. 2024, 63, 1251–1270.
- Shagufta, I.; Panda, G. Quest for steroidomimetics: amino acids derived steroidal and nonsteroidal architectures. Eur. J. Med. Chem. 2017, 133, 139–151.
- Ronchetti, S.; Ayroldi, E.; Gentili, M.; Migilorati, G.; Riccardi, C. A glance at the use of glucocorticoids in rare inflammatory and autoimmune diseases: still an indispensable pharmacological tool? Front. Immunol. 2021, 11, 613435.
- Sugiura, K.; Kobayashi, T.; Ojima, S. The actual status of thromboembolism caused by the use of female hormonal agents in Japan. July 14, 2017. Available online: https://www.jstage.jst.go.jp/article/shinzo/48/7/48_826/_article/-char/ja/.
- Zhang, G.-Q.; Chen, J.-L.; Luo, Y.; Mathur, M.B.; Anagnostis, P.; Nurmatov, U.; Talibov, M.; Zhang, J.; Hawrylowicz, C.M.; Lumsden, M.A.; Critchley, H.; Sheikh, A.; Lundbäck, B.; Lässer, C.; Kankaanranta, H.; Lee, S.H.; Nwaru, B.I. Menopausal hormone therapy and women’s health: an umbrella review. PLoS Med. 2021, 18, e1003731.
- Pan, M.; Pan, X.; Zhou, J.; Wang, J.; Qi, Q.; Wang, L. Update on hormone therapy for the management of postmenopausal women. Biosci. Trends 2022, 16, 46–57.
- Nelson, H.D. Postmenopausal osteoporosis and estrogen. 2003. Available online: https://pubmed.ncbi.nlm.nih.gov/12952379/.
- Khosla, S.; Monroe, D.G. Regulation of bone metabolism by sex steroids. Cold Spring Harb. Perspect. Med. 2018, 8, a031211.
- Timmermans, S.; Souffriau, J.; Libert, C. A general introduction to glucocorticoid biology. Front. Immunol. 2019, 10, 1545.
- Bond, P.; Smit, D.L.; Ronde, W.D. Anabolic–androgenic steroids: How do they work and what are the risks?. Front. Endocrinol. 2022, 13, 1059473.
- Wenbo, Z.; Yan, Z. The uses of anabolic androgenic steroids among athletes; its positive and negative aspects-a literature review. J. Multidiscip. Healthc. 2023, 16, 4293–4305.
- Waljee, A.K.; Rogers, M.A.M.; Lin, P.; Singal, A.G.; Stein, J.S.; Marks, R.M.; Ayanian, J.Z.; Nallamothu, B.K. Short term use of oral corticosteroids and related harms among adults in the United States: population based cohort study. BMJ. 2017, 357.
- Ministry of Health, Labour and Welfare of Japan. Manual for serious adverse drug reactions by disease: thrombosis (thromboembolism, embolism, infarction). 2007.
- Ayyoub, S.; Orriols, R.; Oliver, E.; Ceide, O.T. Thrombosis models: An overview of common in vivo and in vitro models of thrombosis. Int. J. Mol. Sci. 2023, 24, 2569.
- Delluc, A.; Lacut, K.; Rodger, M.A. Arterial and venous thrombosis: what’s the link? A narrative review. Thromb. Res. 2020, 191, 97–102.
- Donadini, M.P.; Calcaterra, F.; Romualdi, E.; Ciceri, R.; Cancellara, A.; Lodigiani, C.; Bacci, M.; Bella, S.D.; Ageno, W.; Mavilio, D. The Link Between Venous and Arterial Thrombosis: Is There a Role for Endothelial Dysfunction?. Cells. 2025, 14, 144.
- Macleod, C.; Hadoke, P.W.F.; Nixon, M. Glucocorticoids: fuelling the fire of atherosclerosis or therapeutic extinguishers?. Int. J. Mol. Sci. 2021, 22, 7622.
- FDA. FDA Adverse event reporting system (FAERS). Available: https://www.fda.gov/drugs/fdas-adverse-event-reporting-system-faers/fda-adverse-event-reporting-system-faers-public-dashboard.
- Glueck, C.J.; Goldenberg, N.; Wang, P. Testosterone therapy, thrombophilia, venous thromboembolism, and thrombotic events. J. Clin. Med. 2018, 8, 11.
- [van Hylckama Vlieg, A.; Helmerhorst, F.M.; Vandenbroucke, J.P.; Doggen, C.J.M.; Rosendaal, F.R. The venous thrombotic risk of oral contraceptives, effects of oestrogen dose and progestogen type: results of the MEGA case-control study. BMJ 2009, 339.
- Jolliffe, I.T.; Cadima, J. Principal component analysis: a review and recent developments. Philos. Trans. Roy. Soc. A 2016, 374, 20150202.
- Chao, T.F.; Liu, C.J.; Chen, S.J.; Wang, K.L.; Lin, Y.J.; Chang, S.L.; Lo, L.W.; Tuan, T.C.; Chen, T.J.; Chiang, C.E.; Chen, S.A. Does digoxin increase the risk of ischemic stroke and mortality in atrial fibrillation? A nationwide population-based cohort study. Can. J. Cardiol. 2014, 30, 1190–1195.
- Everitt, B.S.; Landau, S.; Leese, M.; Stahl, D. Cluster Analysis, 5th ed.; Wiley-Blackwell; 2011.
- Pariente, A.; Avillach, P.; Salvo, F.; Thiessard, F.; Salamé, G.M.; Reglat, A.F.; Haramburu, F.; Bégaud, B.; Moore, N. Effect of competition bias in safety signal generation: analysis of a research database of spontaneous reports in France. Drug Saf. 2012, 35, 855–864.
- PubChemPy. Available: https://github.com/mcs07/pubchempy.
- Molecular Operating Environment. Available: https://www.chemcomp.com/en/Products.htm.
- Introductory guide for standardised MeDRA queries (SMQs) version 27.1. 9 2024. Available: https://www.jmo.pmrj.jp/download/2361.
- Introductory guide MeDRA version 27.1. 9 2024. Available: https://www.jmo.pmrj.jp/download/2360.
- Watanabe, Y.; Matsushita, Y.; Watanabe, A.; Maeda, T.; Nukui, H.; Ogawa, Y.; Sawa, A.; Maeda, H. Early detection of important safety information—recent methods for signal detection. Jpn. J. Biomet. 2004, 25, 37–60.
- Greenland, S.; Schwartzbaum, J.A.; Finkle, W.D. Problems due to small samples and sparse data in conditional logistic regression analysis. Am. J. Epidemiol. 2000, 151, 531–539.
- Kawabe, A.; Uesawa, Y. Analysis of corticosteroid-induced glaucoma using the Japanese adverse drug event reporting database. Pharmaceuticals. 2023, 16, 948.
- Okunaka, M.; Kano, D.; Matsui, R.; Kawasaki, T.; Uesawa, Y. Comprehensive analysis of chemotherapeutic agents that induce infectious neutropenia. Pharmaceuticals. 2021, 14, 681.
- Kan, Y.; Nagai, J.; Uesawa, Y. Evaluation of antibiotic-induced taste and smell disorders using the FDA adverse event reporting system database. Sci. Rep. 2021, 11, 9625.
- Hamazaki, R.; Uesawa, Y. Characterization of Antineoplastic Agents Inducing Taste and Smell Disorders Using the FAERS Database. Pharmaceuticals. 2024, 17, 1116.
- Kagaya, M.; Uesawa, Y. Nuclear Receptors and Stress Response Pathways Associated with the Development of Oral Mucositis Induced by Antineoplastic Agents. Pharmaceuticals. 2024, 17, 1086.
- Everitt, B.S. Unresolved problems in cluster analysis. Biometrics. 1979, 35, 169–181.


 |
 |
 |
 |
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




