Submitted:
27 June 2025
Posted:
30 June 2025
You are already at the latest version
Abstract
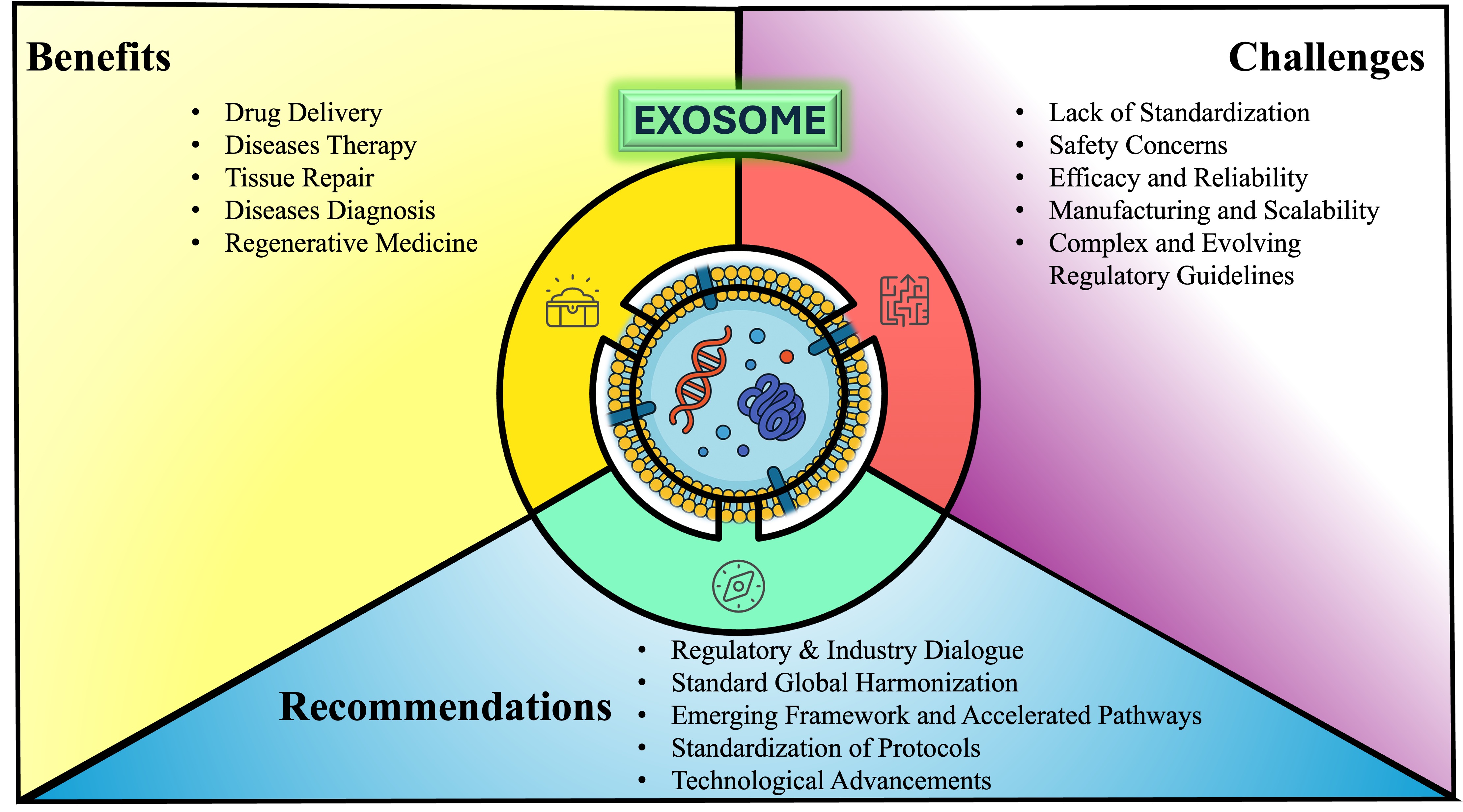
Keywords:
1. Introduction
2. Exosome-Based Therapeutics in Clinical Trials
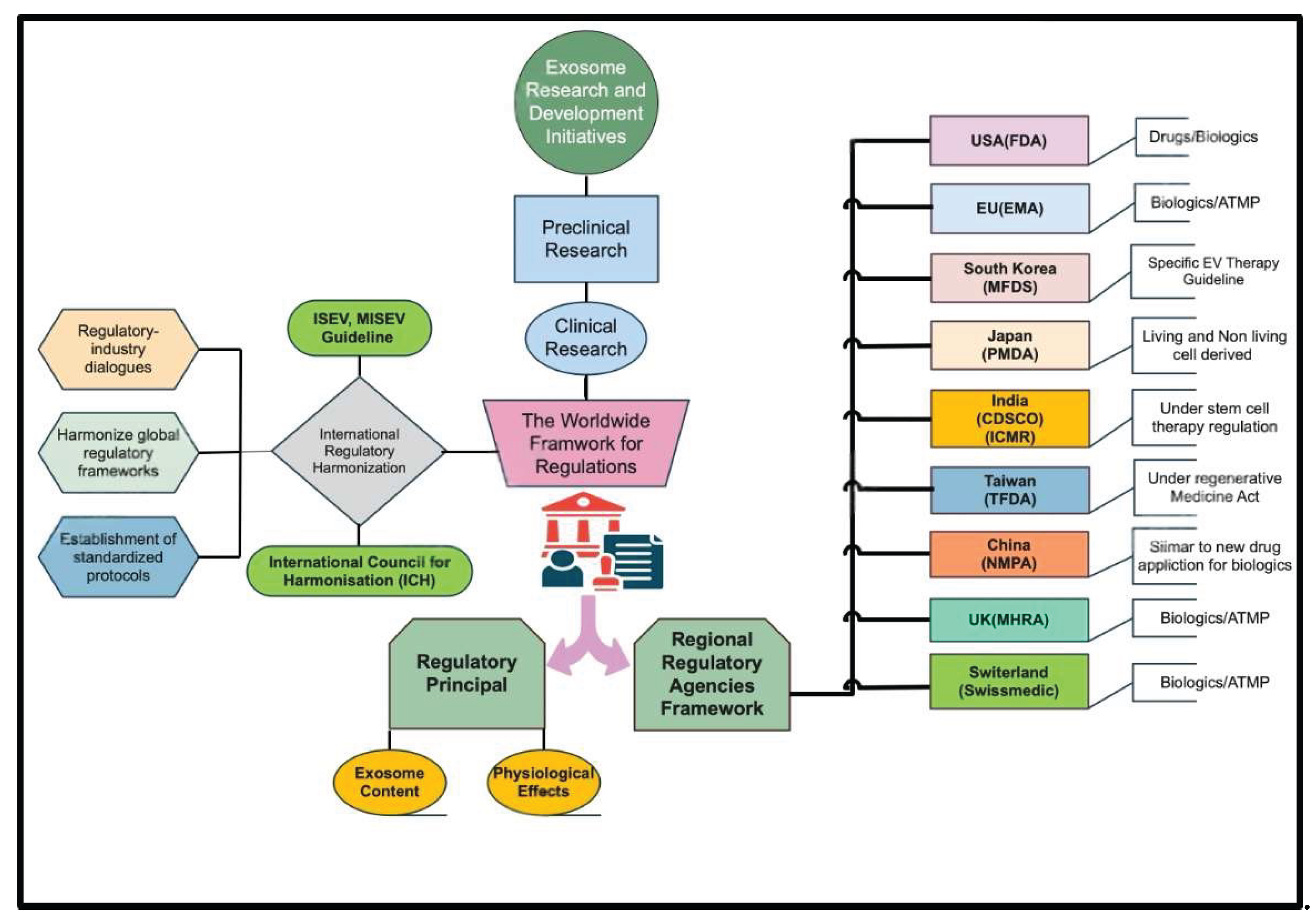
3. Global Regulatory Frameworks for Exosomes
| Region/Country | Regulatory Authority | Regulatory Status & Classification | Classification Focus |
|---|---|---|---|
| United States | U.S. Food and Drug Administration (FDA) |
Exosomes are regulated as biologics/drugs and subject to premarket review; no products approved to date | Content characterization; physiological function |
| European Union | European Medicines Agency (EMA) |
Exosomes fall under the Advanced Therapy Medicinal Products (ATMP) regulation; classification criteria remain unclear | Cargo composition; functional (RNA) content |
| Japan | Pharmaceuticals and Medical Devices Agency (PMDA) | Dedicated subcommittees evaluate safety and quality of extracellular vesicle therapies | Source of manufacture; living vs. nonliving |
| South Korea | Ministry of Food and Drug Safety (MFDS) | Published specific guidelines for extracellular vesicle–based therapies | Manufacturing source |
| Taiwan | Taiwan Food and Drug Administration (TFDA) | Regenerative Medicine Development Act encompasses exosomes; cosmetic use explicitly permitted | Manufacturing source; regenerative applications |
| India | Central Drugs Standard Control Organization (CDSCO) & ICMR | Stem-cell therapies regulated; no exosome-specific therapeutic guidelines established | Nascent and evolving |
| Australia | Therapeutic Goods Administration (TGA) | Stem-cell and tissue therapies regulated since 2019; no dedicated exosome guidelines | Nascent and evolving |
| China | National Medical Products Administration (NMPA) | Exosome products regulated under the same framework as biological new drug applications | Nascent and evolving |
| Switzerland | Swiss Agency for Therapeutic Products (Swissmedic) | Exosome-derived products classified as biological medicines; may be regulated as ATMPs when cells are extensively manipulated | Nascent and evolving |
| United Kingdom | Medicines and Healthcare Products Regulatory Agency (MHRA) | Exosome therapies classified as biological medicinal products; ATMP framework applies if derived from manipulated cells | Nascent and evolving |
3.1. United States Regulatory Framework
3.2. European Union Regulatory Framework
3.3. Japan Regulatory Framework
3.4. South Korea Regulatory Framework
3.5. Taiwan Regulatory Framework
3.6. Chinese Regulatory Framework
3.7. Indian Regulatory Framework
3.8. United Kingdom Regulatory Framework
3.9. Switzerland Regulatory Framework
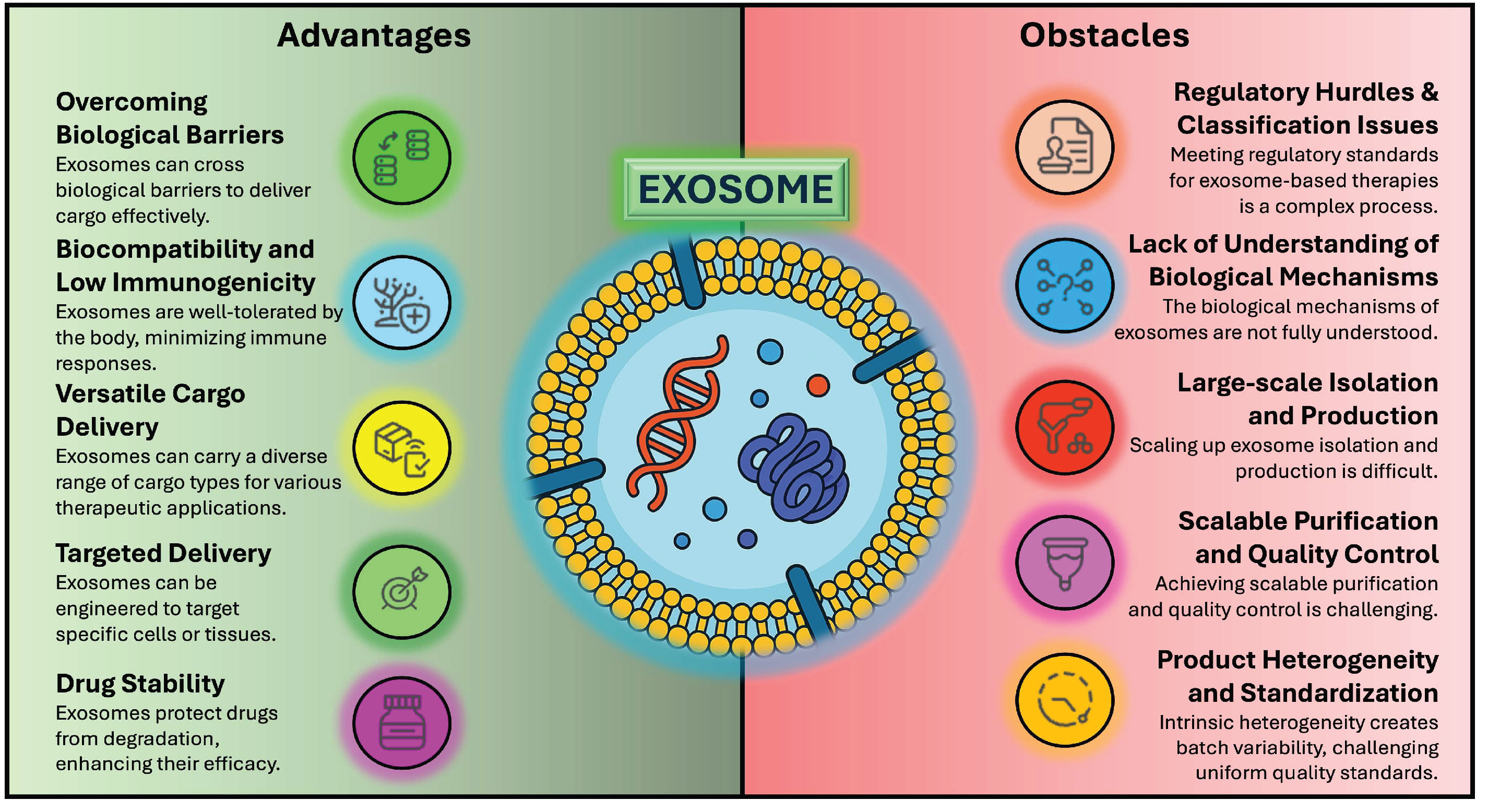
4. Regulatory Challenges in Clinical Trials and Exosome Research
5. Harmonization of the exosome regulatory framework
6. Future Directions, Opportunities, and Policy Implications
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATMP | Advanced Therapy Medicinal Products |
| CBER | Center for Biologics Evaluation and Research |
| CDER | Center for Drug Evaluation and Research |
| ISEV | International Society for Extracellular |
| FDA | Food and Drug Administration |
| GMP | Good Manufacturing Practices |
| MSC | Mesenchymal Stem Cell |
| PMD | Pharmaceuticals and Medical Devices |
| ASRM | Act on the Safety of Regenerative Medicine |
| EMA | European Medicines Agency |
| PMDA | Pharmaceuticals and Medical Devices Agency |
| MFDS | Ministry of Food and Drug Safety |
| TFDA | Taiwan Food and Drug Administration |
| CDSCO | Central Drugs Standard Control Organization |
| ICMR | Indian Council of Medical Research |
| TGA | Therapeutic Goods Administration |
| NMPA | National Medical Products Administration |
| MHRA | Medicines and Healthcare Products Regulatory Agency |
| PHS | Public Health Service |
| IND | Investigational New Drug |
| BLA | Biologics License Application |
| CMC | Chemical, Manufacturing, and Control |
| CAT | Committee for Advanced Therapies |
| RMA | Regenerative Medicine Act |
| GTP | Good Tissue Practice |
| GCP | Good Clinical Practice |
| MISEV | Minimal Information for Studies of Extracellular Vesicles |
| EV | Extracellular Vesicles |
| MOA | Mechanism of Action |
| ICH | International Conference on Harmonization |
References
- Kalluri, R.; LeBleu, V.S. The biology, function, and biomedical applications of exosomes. Science 2020, 367. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.A.; Chan, L.K.W.; Hung, L.C.; Lam, P.K.W.; Park, Y.; Yi, K.H. Clinical Applications of Exosomes: A Critical Review. International Journal of Molecular Sciences 2024, 25, 7794. [Google Scholar] [CrossRef] [PubMed]
- Cheng, C.H.; Hao, W.R.; Cheng, T.H. Stem cell exosomes: New hope for recovery from diabetic brain hemorrhage. World J Diabetes 2024, 15, 2264–2271. [Google Scholar] [CrossRef] [PubMed]
- Zhou, B.; Xu, K.; Zheng, X.; Chen, T.; Wang, J.; Song, Y.; Shao, Y.; Zheng, S. Application of Exosomes as Liquid Biopsy in Clinical Diagnosis. Signal Transduction and Targeted Therapy 2020, 5. [Google Scholar] [CrossRef]
- Rezaie, J.; Feghhi, M.; Etemadi, T. A Review on Exosomes Application in Clinical Trials: Perspective, Questions, and Challenges. Cell Communication and Signaling 2022, 20. [Google Scholar] [CrossRef]
- Chen, Y.F.; Luh, F.; Ho, Y.S.; Yen, Y. Exosomes: a review of biologic function, diagnostic and targeted therapy applications, and clinical trials. J Biomed Sci 2024, 31, 67. [Google Scholar] [CrossRef]
- Choudhery, M.S.; Arif, T.; Mahmood, R.; Harris, D.T. Stem Cell-Based Acellular Therapy: Insight into Biogenesis, Bioengineering and Therapeutic Applications of Exosomes. Biomolecules 2024, 14, 792. [Google Scholar] [CrossRef]
- Chu, C.-H.; Lee, R.P.; Wu, W.T.; Chen, I.-H.; Yeh, K.T.; Wang, C.C. Advancing Osteoarthritis Treatment: The Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes and Biomaterial Integration. Biomedicines 2024, 12, 2478. [Google Scholar] [CrossRef]
- Newton, W.C.; Kim, J.W.; Luo, J.Z.Q.; Luo, L. Stem cell-derived exosomes: a novel vector for tissue repair and diabetic therapy. J Mol Endocrinol 2017, 59, R155–R165. [Google Scholar] [CrossRef]
- Kar, R.; Dhar, R.; Mukherjee, S.; Nag, S.; Gorai, S.; Mukerjee, N.; Mukherjee, D.; Vatsa, R.; Jadhav, M.; Ghosh, A.; et al. Exosome-Based Smart Drug Delivery Tool for Cancer Theranostics. Acs Biomaterials Science & Engineering 2023, 9, 577–594. [Google Scholar] [CrossRef]
- Yong, T.; Zhang, X.; Bie, N.; Zhang, H.; Zhang, X.; Li, F.; Hakeem, A.; Hu, J.; Gan, L.; Santos, H.A.; et al. Tumor exosome-based nanoparticles are efficient drug carriers for chemotherapy. Nat Commun 2019, 10, 3838. [Google Scholar] [CrossRef] [PubMed]
- Sousa, P.; Lopes, B.; Sousa, A.C.; Moreira, A.; Coelho, A.; Alvites, R.; Alves, N.; Geuna, S.; Mauricio, A.C. Advancements and Insights in Exosome-Based Therapies for Wound Healing: A Comprehensive Systematic Review (2018-June 2023). Biomedicines 2023, 11, 2099. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.H.; Lee, R.P.; Wu, W.T.; Chen, I.H.; Yeh, K.T.; Wang, C.C. Advancing Osteoarthritis Treatment: The Therapeutic Potential of Mesenchymal Stem Cell-Derived Exosomes and Biomaterial Integration. Biomedicines 2024, 12, 2478. [Google Scholar] [CrossRef]
- Xu, Z.; Zeng, S.; Gong, Z.; Yan, Y. Exosome-based immunotherapy: a promising approach for cancer treatment. Mol Cancer 2020, 19, 160. [Google Scholar] [CrossRef]
- Fu, W.; Lei, C.; Liu, S.; Cui, Y.; Wang, C.; Qian, K.; Li, T.; Shen, Y.; Fan, X.; Lin, F.; et al. CAR exosomes derived from effector CAR-T cells have potent antitumour effects and low toxicity. Nat Commun 2019, 10, 4355. [Google Scholar] [CrossRef]
- Zhou, B.; Xu, K.; Zheng, X.; Chen, T.; Wang, J.; Song, Y.; Shao, Y.; Zheng, S. Application of exosomes as liquid biopsy in clinical diagnosis. Signal Transduct Target Ther 2020, 5, 144. [Google Scholar] [CrossRef]
- Wang, C.K.; Tsai, T.H.; Lee, C.H. Regulation of exosomes as biologic medicines: Regulatory challenges faced in exosome development and manufacturing processes. Clin Transl Sci 2024, 17, e13904. [Google Scholar] [CrossRef]
- Rezaie, J.; Feghhi, M.; Etemadi, T. A review on exosomes application in clinical trials: perspective, questions, and challenges. Cell Commun Signal 2022, 20, 145. [Google Scholar] [CrossRef]
- Mukherjee, R. The Silent Battle for Extracellular Vesicle Regulation: Why Extracellular Vesicle Therapies Remain in Legal Grey Zones Worldwide. 2025.
- Research, C.f.D.E.a. International Regulatory Harmonization - FDA. Available online: https://www.fda.
- Chu, M.; Wang, H.; Bian, L.; Huang, J.; Wu, D.; Zhang, R.; Fei, F.; Chen, Y.; Xia, J. Nebulization Therapy with Umbilical Cord Mesenchymal Stem Cell-Derived Exosomes for COVID-19 Pneumonia. Stem Cell Rev Rep 2022, 18, 2152–2163. [Google Scholar] [CrossRef]
- Raghav, A.; Khan, Z.A.; Upadhayay, V.K.; Tripathi, P.; Gautam, K.A.; Mishra, B.K.; Ahmad, J.; Jeong, G.B. Mesenchymal Stem Cell-Derived Exosomes Exhibit Promising Potential for Treating SARS-CoV-2-Infected Patients. Cells 2021, 10, 587. [Google Scholar] [CrossRef]
- Zani-Ruttenstock, E.; Antounians, L.; Khalaj, K.; Figueira, R.L.; Zani, A. The Role of Exosomes in the Treatment, Prevention, Diagnosis, and Pathogenesis of COVID-19. Eur J Pediatr Surg 2021, 31, 326–334. [Google Scholar] [CrossRef] [PubMed]
- Chu, M.; Wang, H.; Bian, L.; Huang, J.; Wu, D.; Fei, F.; Zhang, R.; Chen, Y.; Xia, J. Nebulization Therapy for COVID-19 Pneumonia With Embryonic Mesenchymal Stem Cells-Derived Exosomes. 2020. [Google Scholar] [CrossRef]
- Park, H.S.; Cetin, E.; Siblini, H.; Seok, J.; Alkelani, H.; Alkhrait, S.; Liakath Ali, F.; Mousaei Ghasroldasht, M.; Beckman, A.; Al-Hendy, A. Therapeutic Potential of Mesenchymal Stem Cell-Derived Extracellular Vesicles to Treat PCOS. Int J Mol Sci 2023, 24, 11151. [Google Scholar] [CrossRef]
- Lee, B.C.; Kang, I.; Yu, K.R. Therapeutic Features and Updated Clinical Trials of Mesenchymal Stem Cell (MSC)-Derived Exosomes. J Clin Med 2021, 10, 711. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, Q.; Zhang, X.; Huang, H.; Tang, S.; Chai, Y.; Xu, Z.; Li, M.; Chen, X.; Liu, J.; et al. Recent advances in exosome-mediated nucleic acid delivery for cancer therapy. J Nanobiotechnology 2022, 20, 279. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Huang, W.; Li, M.; Zheng, A. Exosome-Based Carrier for RNA Delivery: Progress and Challenges. Pharmaceutics 2023, 15, 598. [Google Scholar] [CrossRef]
- Chew, F.Y.; Tsai, C.H.; Chang, K.H.; Chang, Y.K.; Chou, R.H.; Liu, Y.J. Exosomes as promising frontier approaches in future cancer therapy. World J Gastrointest Oncol 2025, 17, 100713. [Google Scholar] [CrossRef]
- Kar, R.; Dhar, R.; Mukherjee, S.; Nag, S.; Gorai, S.; Mukerjee, N.; Mukherjee, D.; Vatsa, R.; Chandrakanth Jadhav, M.; Ghosh, A.; et al. Exosome-Based Smart Drug Delivery Tool for Cancer Theranostics. ACS Biomater Sci Eng 2023, 9, 577–594. [Google Scholar] [CrossRef]
- Sonar, S. Clinical trial status of exosomes-based cancer theranostics. Clinical and Translational Discovery 2024, 4. [Google Scholar] [CrossRef]
- Li, Y.; Tang, Y.; Yang, G.Y. Therapeutic application of exosomes in ischaemic stroke. Stroke Vasc Neurol 2021, 6, 483–495. [Google Scholar] [CrossRef]
- Jiang, L.; Chen, W.; Ye, J.; Wang, Y. Potential Role of Exosomes in Ischemic Stroke Treatment. Biomolecules 2022, 12, 115. [Google Scholar] [CrossRef]
- Baghaei, K.; Tokhanbigli, S.; Asadzadeh, H.; Nmaki, S.; Reza Zali, M.; Hashemi, S.M. Exosomes as a novel cell-free therapeutic approach in gastrointestinal diseases. J Cell Physiol 2019, 234, 9910–9926. [Google Scholar] [CrossRef] [PubMed]
- Sharma, A.; Yadav, A.; Nandy, A.; Ghatak, S. Insight into the Functional Dynamics and Challenges of Exosomes in Pharmaceutical Innovation and Precision Medicine. Pharmaceutics 2024, 16. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.A.; Chan, L.K.W.; Hung, L.C.; Phoebe, L.K.W.; Park, Y.; Yi, K.H. Clinical Applications of Exosomes: A Critical Review. Int J Mol Sci 2024, 25, 7794. [Google Scholar] [CrossRef]
- Li, X.; Corbett, A.L.; Taatizadeh, E.; Tasnim, N.; Little, J.P.; Garnis, C.; Daugaard, M.; Guns, E.; Hoorfar, M.; Li, I.T.S. Challenges and opportunities in exosome research-Perspectives from biology, engineering, and cancer therapy. APL Bioeng 2019, 3, 011503. [Google Scholar] [CrossRef]
- Ren, K. Exosomes in perspective: a potential surrogate for stem cell therapy. Odontology 2019, 107, 271–284. [Google Scholar] [CrossRef]
- Ludwig, N.; Whiteside, T.L.; Reichert, T.E. Challenges in Exosome Isolation and Analysis in Health and Disease. Int J Mol Sci 2019, 20. [Google Scholar] [CrossRef]
- Martinez-Espinosa, I.; Serrato, J.A.; Ortiz-Quintero, B. The Role of Exosome-Derived microRNA on Lung Cancer Metastasis Progression. Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Shaun, S. Exosomes as therapeutics and drug delivery vehicles: global regulatory perspectives. 2020, 6, 1561 - 1569, publication_type = article.
- Pawanbir, S.; Laure, B.-D.; Satya, P.D. Exploratory assessment of the current EU regulatory framework for development of advanced therapies. Journal of Commercial Biotechnology 2010, 16, 331–336, publication_type = article. [Google Scholar]
- Daisuke, M.; Teruhide, Y.; Takami, I.; Masakazu, H.; Kazuhiro, T.; Daisaku, S. Regulatory Frameworks for Gene and Cell Therapies in Japan. 2015; Volume 871, pp. 147 - 162, publication_type = incollection.
- Lener, T.; Gimona, M.; Aigner, L.; Borger, V.; Buzas, E.; Camussi, G.; Chaput, N.; Chatterjee, D.; Court, F.A.; Del Portillo, H.A.; et al. Applying extracellular vesicles based therapeutics in clinical trials - an ISEV position paper. J Extracell Vesicles 2015, 4, 30087. [Google Scholar] [CrossRef]
- Batrakova, E.V.; Kim, M.S. Development and regulation of exosome-based therapy products. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2016, 8, 744–757. [Google Scholar] [CrossRef]
- Yoon, J.; Lee, S.; Kim, M.J.; Kim, J.H. Brief summary of the regulatory frameworks of regenerative medicine therapies. Front Pharmacol 2024, 15, 1486812. [Google Scholar] [CrossRef] [PubMed]
- Fujita, M.; Hatta, T.; Ikka, T.; Onishi, T. The urgent need for clear and concise regulations on exosome-based interventions. Stem Cell Reports 2024, 19, 1517–1519. [Google Scholar] [CrossRef] [PubMed]
- (FDA), U.F.a.D.A. Consumer Alert on Regenerative Medicine Products Including Stem Cells and Exosomes. 2024.
- Clinic. Exosomes & EU Regulations. 2024, 2025.
- Lysaght, T. Accelerating regenerative medicine: the Japanese experiment in ethics and regulation. Regen Med 2017, 12, 657–668. [Google Scholar] [CrossRef] [PubMed]
- Fujita, Y.; Kawamoto, A. Regenerative medicine legislation in Japan for fast provision of cell therapy products. Clin Pharmacol Ther 2016, 99, 26–29. [Google Scholar] [CrossRef]
- Tsuchiya, A.; Terai, S.; Horiguchi, I.; Homma, Y.; Saito, A.; Nakamura, N.; Sato, Y.; Ochiya, T.; Kino-Oka, M.; Working Group of Attitudes for, P.; et al. Basic points to consider regarding the preparation of extracellular vesicles and their clinical applications in Japan. Regen Ther 2022, 21, 19–24. [Google Scholar] [CrossRef]
- CiRA. The need for regulatory measures regarding exosome therapy. Available online: https://www.cira.kyoto-u.ac.jp/e/pressrelease/news/241025-110000.html (accessed on 05/22/2025).
- Verter, F. South Korea expands access to regenerative medicine for serious illnesses. Available online: https://parentsguidecordblood.org/en/news/south-korea-expands-access-regenerative-medicine-serious-illnesses. (accessed on 05/20/2025).
- patsnap, S.b. S&E Bio Gains Korea's First Approval for Exosome Therapy Trial. Available online: https://synapse.patsnap.com/article/se-bio-gains-koreas-first-approval-for-exosome-therapy-trial (accessed on 05/20/2025).
- (2025), T.-A.o. Association for the Advancement of Blood & Biotherapies - TFDA. Available online: https://www.aabb.org/regulatory-and-advocacy/regulatory-affairs/regulatory-for-cellular-therapies/international-competent-authorities/taiwan#:~:text=In%20Taiwan%2C%20the%20Food,these%20regulations.&text=primary%20pieces%20of%20legislation,these%20regulations.&text=tissues%20must%20comply%20with,these%20regulations.&text=cellular%20therapy%20product.%20The,these%20regulations. (accessed on 03/19/2025).
- Bridge, P. Taiwan’s Regulatory Framework for Regenerative Medicine. Available online: https://www.pacificbridgemedical.com/uncategorized/taiwan-regulatory-framework-for-regenerative-medicine/ (accessed on 05/18/2025).
- Chao, W.Y.; Chang, Y.T.; Tsai, Y.T.; Huang, M.C.; Lin, Y.C.; Wu, M.M.; Chi, J.F.; Lin, C.L.; Cheng, H.F.; Wu, S.M. Update on Regulation of Regenerative Medicine in Taiwan. Adv Exp Med Biol 2023, 1430, 211–219. [Google Scholar] [CrossRef]
- Chen, Y.C.; Cheng, H.F.; Yeh, M.K. Cell Therapy Regulation in Taiwan. Cell Transplant 2017, 26, 483–492. [Google Scholar] [CrossRef]
- healthcare, T. ExoOne has been granted approval by the Ministry of Health and Welfare to use human-derived exosomes as cosmetic ingredients, marking the first case in Taiwan. Available online: https://www.taiwan-healthcare.org/en/news-detail?id=0scizkifbr66p1ui (accessed on 03/18/2025).
- Bioscience, A. Navigating Cell & Gene Therapy Regulations in China – How does the dual-track system works? 2024.
- Ltd, A.I.P. Exosome research products market in China is rapidly expanding, driven by significant government funding, cutting-edge applications in diagnostics and drug delivery, and increasing collaborations among research institutions and biotech firms, positioning China as a leader in biomedical innovation. 2024.
- S, S.K.; Joga, R.; Srivastava, S.; Nagpal, K.; Dhamija, I.; Grover, P.; Kumar, S. Regulatory landscape and challenges in CAR-T cell therapy development in the US, EU, Japan, and India. Eur J Pharm Biopharm 2024, 201, 114361. [Google Scholar] [CrossRef]
- ClinRegs. Clinical Research Regulation For India - ClinRegs. 2025.
- AABB.org, I.-. https://www.aabb.org/regulatory-and-advocacy/regulatory-affairs/regulatory-for-cellular-therapies/international-competent-authorities/india#:~:text=All%20other%20forms%20of,ICMR%20registry.&text=marrow%20derived%20stem%20cells%2C,ICMR%20registry.&text=any%20embryonic%20stem%20cell,ICMR%20registry.&text=appropriately%20reviewed%20and%20monitored,ICMR%20registry. (accessed on 03/19/2025).
- Jain., V. Regulation of Biologics in India - Morulaa HealthTech. Available online: https://morulaa.com/regulation-of-biologics-in-india-2/#:~:text=Biologics%20are%20the%20medicinal%2F,in%20India.&text=spin%2Doff%20for%20human%20use.,in%20India.&text=tissues%20etc.%20Biotechnology%20is,in%20India.&text=Drugs%20Standard%20Control%20Organization,in%20India. (accessed on 05/19/2016).
- Thakur, A.; Rai, D. Global requirements for manufacturing and validation of clinical grade extracellular vesicles. J Liq Biopsy 2024, 6, 100278. [Google Scholar] [CrossRef]
- Agency, M.a.H.p.R. Advanced therapy medicinal products: regulation and licensing in UK. Available online: https://www.gov.uk/guidance/advanced-therapy-medicinal-products-regulation-and-licensing#:~:text=All%20ATMPs%20to%20be,the%20quality%2C&text=the%20market%20in%20the,the%20quality%2C&text=have%20a%20marketing%20authorisation,the%20quality%2C&text=%29.%20The%20MHRA%20is,the%20quality%2C (accessed on 05/21/2025).
- Asadpour, A.; Yahaya, B.H.; Bicknell, K.; Cottrell, G.S.; Widera, D. Uncovering the gray zone: mapping the global landscape of direct-to-consumer businesses offering interventions based on secretomes, extracellular vesicles, and exosomes. Stem Cell Res Ther 2023, 14, 111. [Google Scholar] [CrossRef]
- Basiadima, M. Can Exosomes Be Injected in the Face? - Premier Laser Clinic. 2024, 2025.
- Devlin, H. Beauty clinics in UK offering banned treatments derived from human cells. The Guardian 2025.
- Bukovac, P.K.; Hauser, M.; Lottaz, D.; Marti, A.; Schmitt, I.; Schochat, T. The Regulation of Cell Therapy and Gene Therapy Products in Switzerland. Adv Exp Med Biol 2023, 1430, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Swissmedic. Legal framework governing the use of tissues and cells of human origin. Available online: https://www.swissmedic.ch/swissmedic/en/home/humanarzneimittel/besondere-arzneimittelgruppen--ham-/innovation/publikationen/legal-basis-governing-the-use-of-tissues-and-cells-of-human-orig.html (accessed on 05/21/2025).
- Queen, D.; Avram, M.R. Exosomes for Treating Hair Loss: A Review of Clinical Studies. Dermatol Surg 2025, 51, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Ahn, S.H.; Ryu, S.W.; Choi, H.; You, S.; Park, J.; Choi, C. Manufacturing Therapeutic Exosomes: from Bench to Industry. Mol Cells 2022, 45, 284–290. [Google Scholar] [CrossRef]
- Maitra, S.; Mukerjee, N.; Alharbi, H.M.; Ghosh, A.; Alexiou, A.; Thorat, N.D. Targeted therapies for HPV-associated cervical cancer: Harnessing the potential of exosome-based chipsets in combating leukemia and HPV-mediated cervical cancer. J Med Virol 2024, 96, e29596. [Google Scholar] [CrossRef]
- Willis, G.R.; Kourembanas, S.; Mitsialis, S.A. Toward Exosome-Based Therapeutics: Isolation, Heterogeneity, and Fit-for-Purpose Potency. Front Cardiovasc Med 2017, 4, 63. [Google Scholar] [CrossRef]
- Singh, K.; Nalabotala, R.; Koo, K.M.; Bose, S.; Nayak, R.; Shiddiky, M.J.A. Separation of distinct exosome subpopulations: isolation and characterization approaches and their associated challenges. Analyst 2021, 146, 3731–3749. [Google Scholar] [CrossRef]
- Wang, Z.; Wang, Q.; Qin, F.; Chen, J. Exosomes: a promising avenue for cancer diagnosis beyond treatment. Front Cell Dev Biol 2024, 12, 1344705. [Google Scholar] [CrossRef]
- Kibria, G.; Ramos, E.K.; Wan, Y.; Gius, D.R.; Liu, H. Exosomes as a Drug Delivery System in Cancer Therapy: Potential and Challenges. Mol Pharm 2018, 15, 3625–3633. [Google Scholar] [CrossRef]
- Zhang, P.; Zhou, X.; He, M.; Shang, Y.; Tetlow, A.L.; Godwin, A.K.; Zeng, Y. Ultrasensitive detection of circulating exosomes with a 3D-nanopatterned microfluidic chip. Nat Biomed Eng 2019, 3, 438–451. [Google Scholar] [CrossRef]
- Gurung, S.; Perocheau, D.; Touramanidou, L.; Baruteau, J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal 2021, 19, 47. [Google Scholar] [CrossRef] [PubMed]
- Milne, C.P.; Mittra, J.; Kojima, N.; Sugiyama, D.; Awatin, J.; Simmons, G. Prospects for Harmonizing Regulatory Science Programs in Europe, Japan, and the United States to Advance Regenerative Medicine. Ther Innov Regul Sci 2016, 50, 724–733. [Google Scholar] [CrossRef] [PubMed]
- Ndomondo-Sigonda, M.; Miot, J.; Naidoo, S.; Masota, N.; Ng'andu, B.; Ngum, N.; Kaale, E. Harmonization of Medical Products Regulation: A Key Factor for Improving Regulatory Capacity in the East African Community. 2020. [Google Scholar] [CrossRef]
- Lindstrom-Gommers, L.; Mullin, T. International Conference on Harmonization: Recent Reforms as a Driver of Global Regulatory Harmonization and Innovation in Medical Products. Clin Pharmacol Ther 2019, 105, 926–931. [Google Scholar] [CrossRef] [PubMed]
- Rosemann, A.; Chaisinthop, N. The pluralization of the international: Resistance and alter-standardization in regenerative stem cell medicine. Soc Stud Sci 2016, 46, 112–139. [Google Scholar] [CrossRef]
- Kleiderman, E.; Boily, A.; Hasilo, C.; Knoppers, B.M. Overcoming barriers to facilitate the regulation of multi-centre regenerative medicine clinical trials. Stem Cell Res Ther 2018, 9, 307. [Google Scholar] [CrossRef]
- Ghaffari, K.; Moradi-Hasanabad, A.; Sobhani-Nasab, A.; Javaheri, J.; Ghasemi, A. Application of cell-derived exosomes in the hematological malignancies therapy. Front Pharmacol 2023, 14, 1263834. [Google Scholar] [CrossRef]
- Li, J.; Wang, A.; Guo, H.; Zheng, W.; Chen, R.; Miao, C.; Zheng, D.; Peng, J.; Wang, J.; Chen, Z. Exosomes: innovative biomarkers leading the charge in non-invasive cancer diagnostics. Theranostics 2025, 15, 5277–5311. [Google Scholar] [CrossRef]
- Youssef, E.; Palmer, D.; Fletcher, B.; Vaughn, R. Exosomes in Precision Oncology and Beyond: From Bench to Bedside in Diagnostics and Therapeutics. Cancers (Basel) 2025, 17, 940. [Google Scholar] [CrossRef]
- Das, C.K.; Jena, B.C.; Banerjee, I.; Das, S.; Parekh, A.; Bhutia, S.K.; Mandal, M. Exosome as a Novel Shuttle for Delivery of Therapeutics across Biological Barriers. Mol Pharm 2019, 16, 24–40. [Google Scholar] [CrossRef]
- Sitbon, A.; Delmotte, P.R.; Pistorio, V.; Halter, S.; Gallet, J.; Gautheron, J.; Monsel, A. Mesenchymal stromal cell-derived extracellular vesicles therapy openings new translational challenges in immunomodulating acute liver inflammation. J Transl Med 2024, 22, 480. [Google Scholar] [CrossRef]
- Jaalouk, D.; Prasai, A.; Goldberg, D.J.; Yoo, J.Y. Regulatory Aspects of Regenerative Medicine in the United States and Abroad. Dermatological Reviews 2024, 5. [Google Scholar] [CrossRef]
- Jafari, N.; Zolfi Gol, A.; Shahabi Rabori, V.; Saberiyan, M. Exploring the role of exosomal and non-exosomal non-coding RNAs in Kawasaki disease: Implications for diagnosis and therapeutic strategies against coronary artery aneurysms. Biochem Biophys Rep 2025, 42, 101970. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tian, Z.; He, L.; Liu, C.; Wang, N.; Rong, L.; Liu, B. Exosomes derived from miR-26a-modified MSCs promote axonal regeneration via the PTEN/AKT/mTOR pathway following spinal cord injury. Stem Cell Res Ther 2021, 12, 224. [Google Scholar] [CrossRef] [PubMed]
- Vakil, D.; Doshi, R.; McKinnirey, F.; Sidhu, K. Stem Cell-Derived Exosomes as New Horizon for Cell-Free Therapeutic Development: Current Status and Prospects. 2023. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




