Submitted:
30 June 2025
Posted:
01 July 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Kaplan–Meier Survival Analysis
2.2. Cell lines and cell culture
2.3. Recombinant Antibody Production
2.4. Western Blot Analysis
2.5. Proliferation Assay In Vitro
2.6. Flow Cytometry
2.7. Determination of the Dissociation Constant (KD) via Flow Cytometry
2.8. ADCC
2.9. CDC
2.10. Antitumor Activity In Vivo
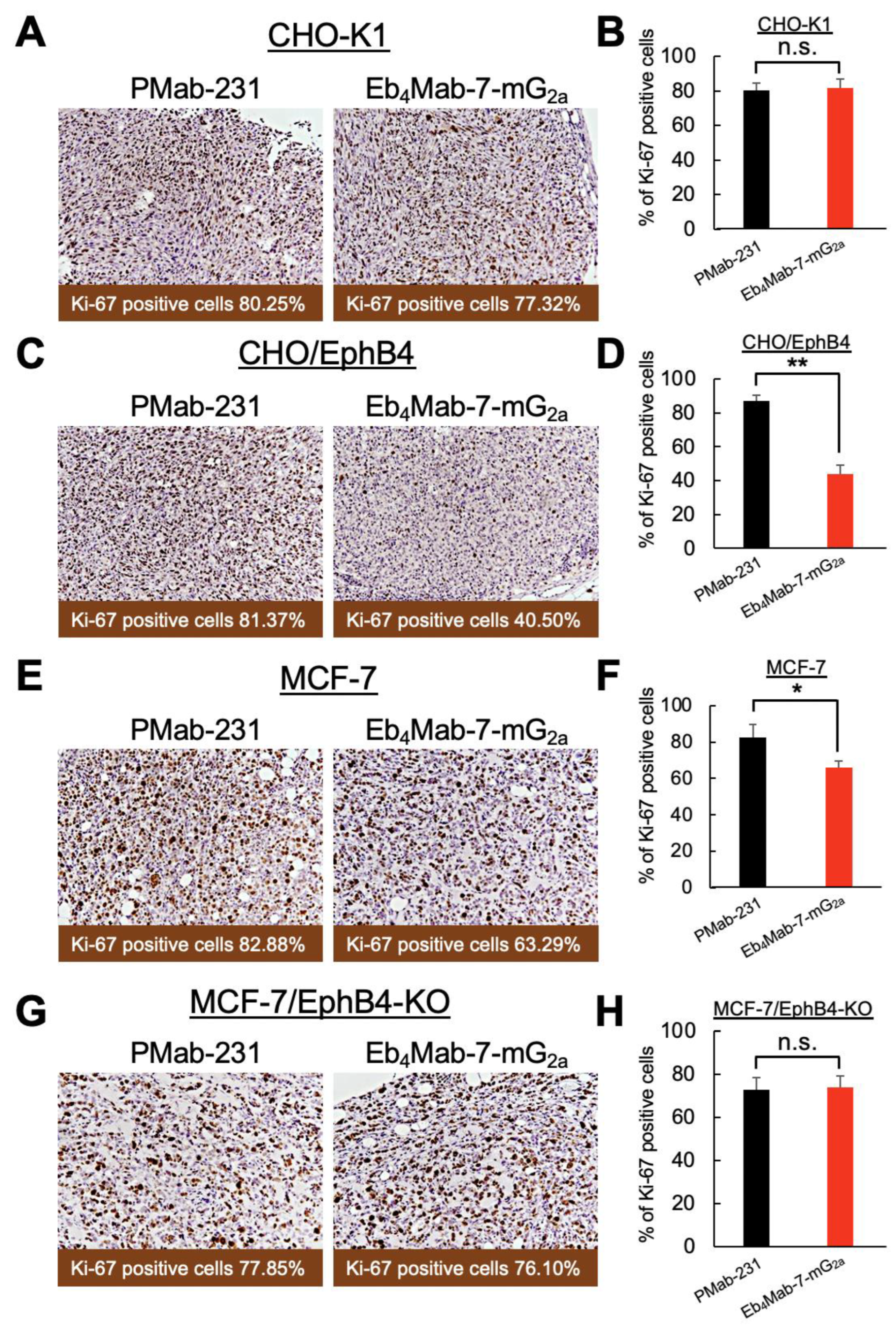
2.11. Immunohistochemical analysis
2.12. Statistical Analysis
3. Results
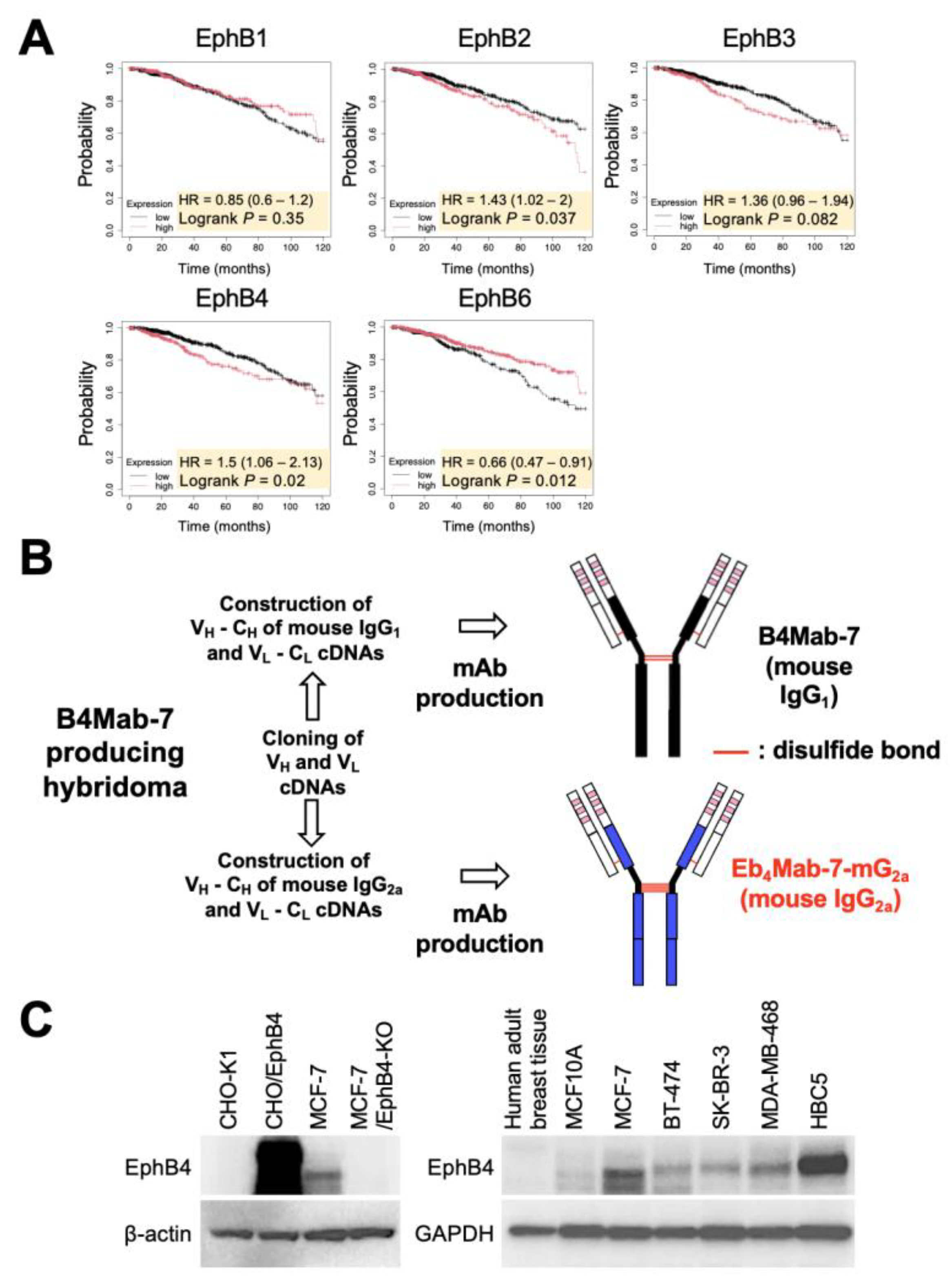
3.1. Development of a Class-Switched Anti-EphB4 Monoclonal Antibody and Analysis of EphB4 Expression Across Breast Cancer Subtypes
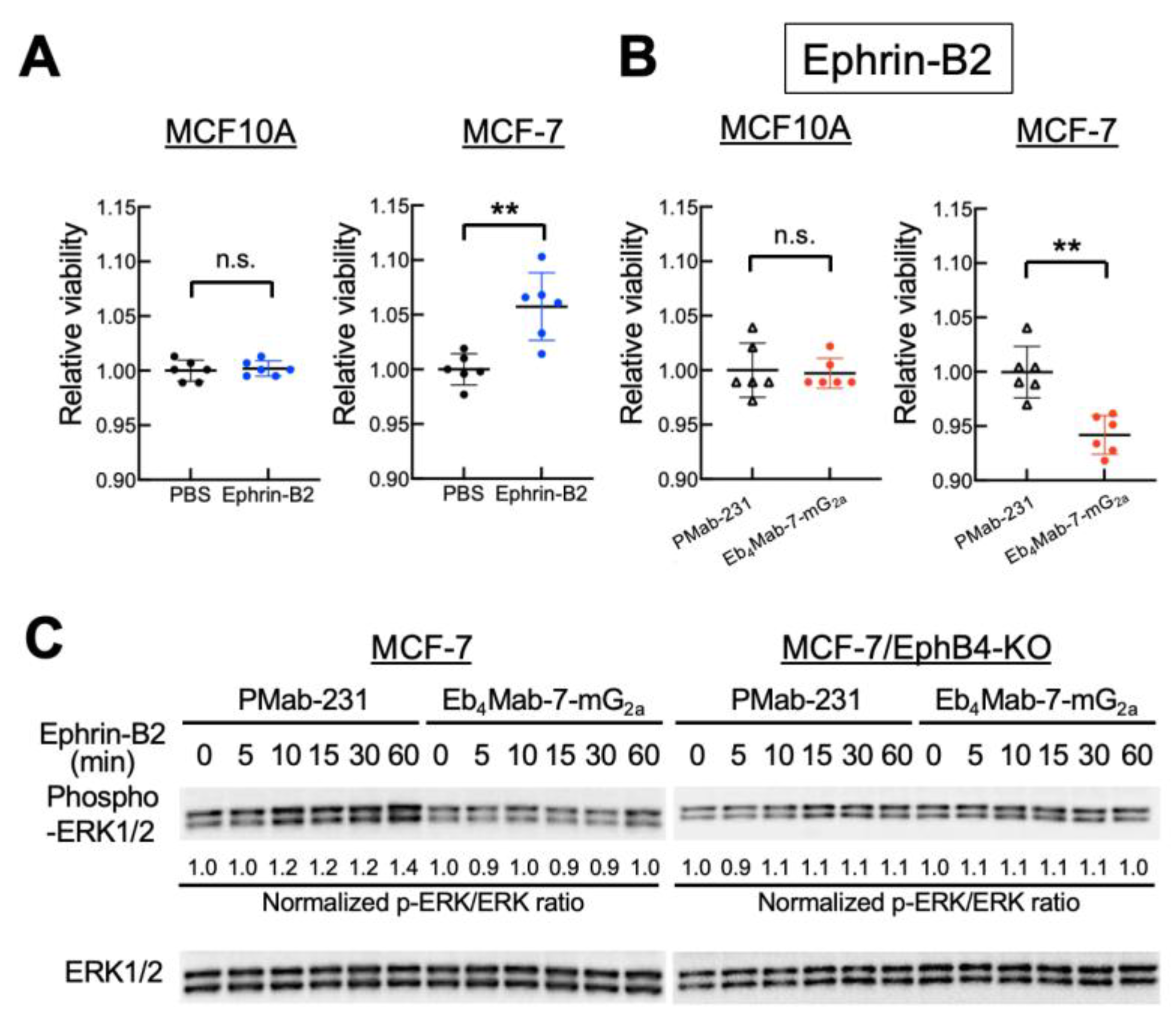
3.2. Effect of Eb4Mab-7-mG2a on Ephrin-B2-Induced Cell Proliferation and ERK Signaling
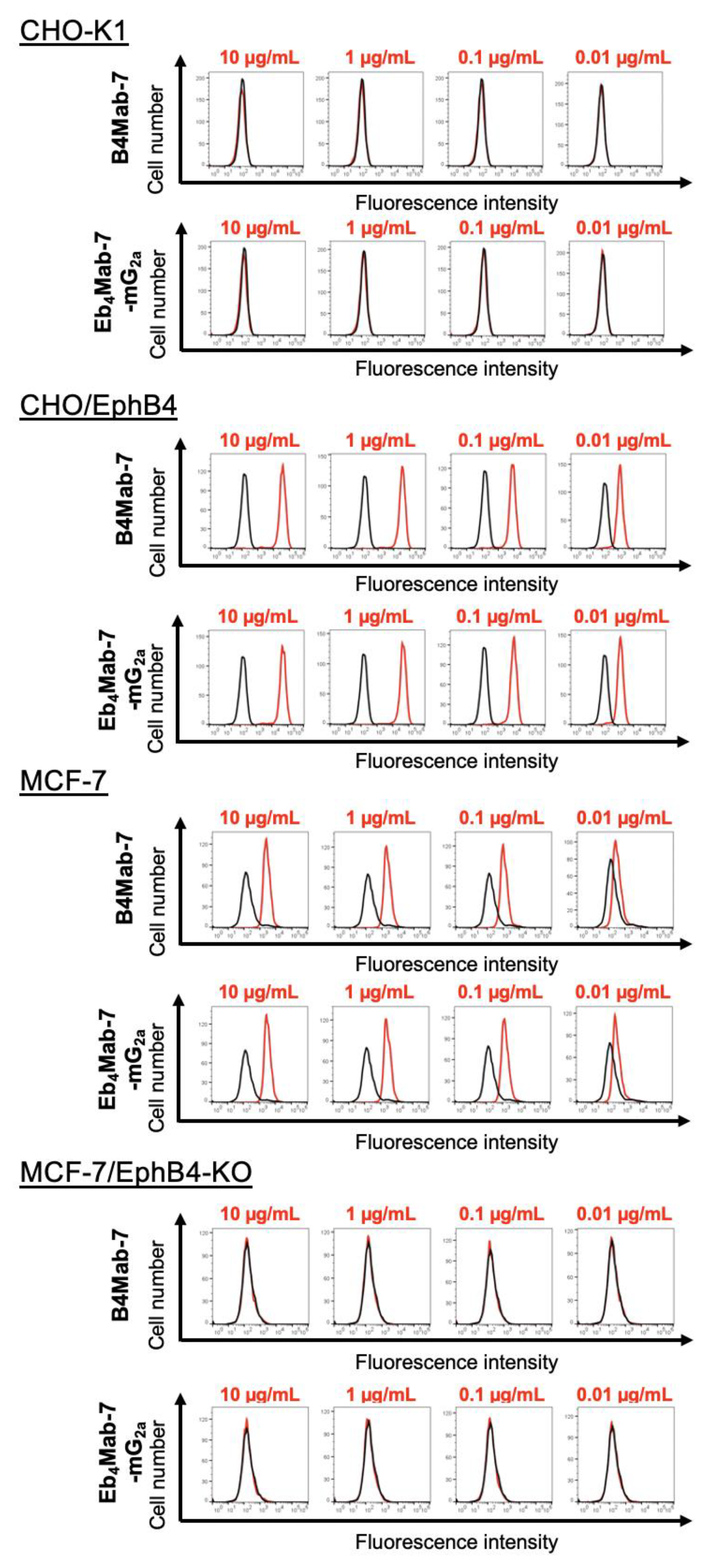
3.3. Binding Activity of Eb4Mab-7-mG2a and B4Mab-7 Against EphB4-Expressing and Control Cells
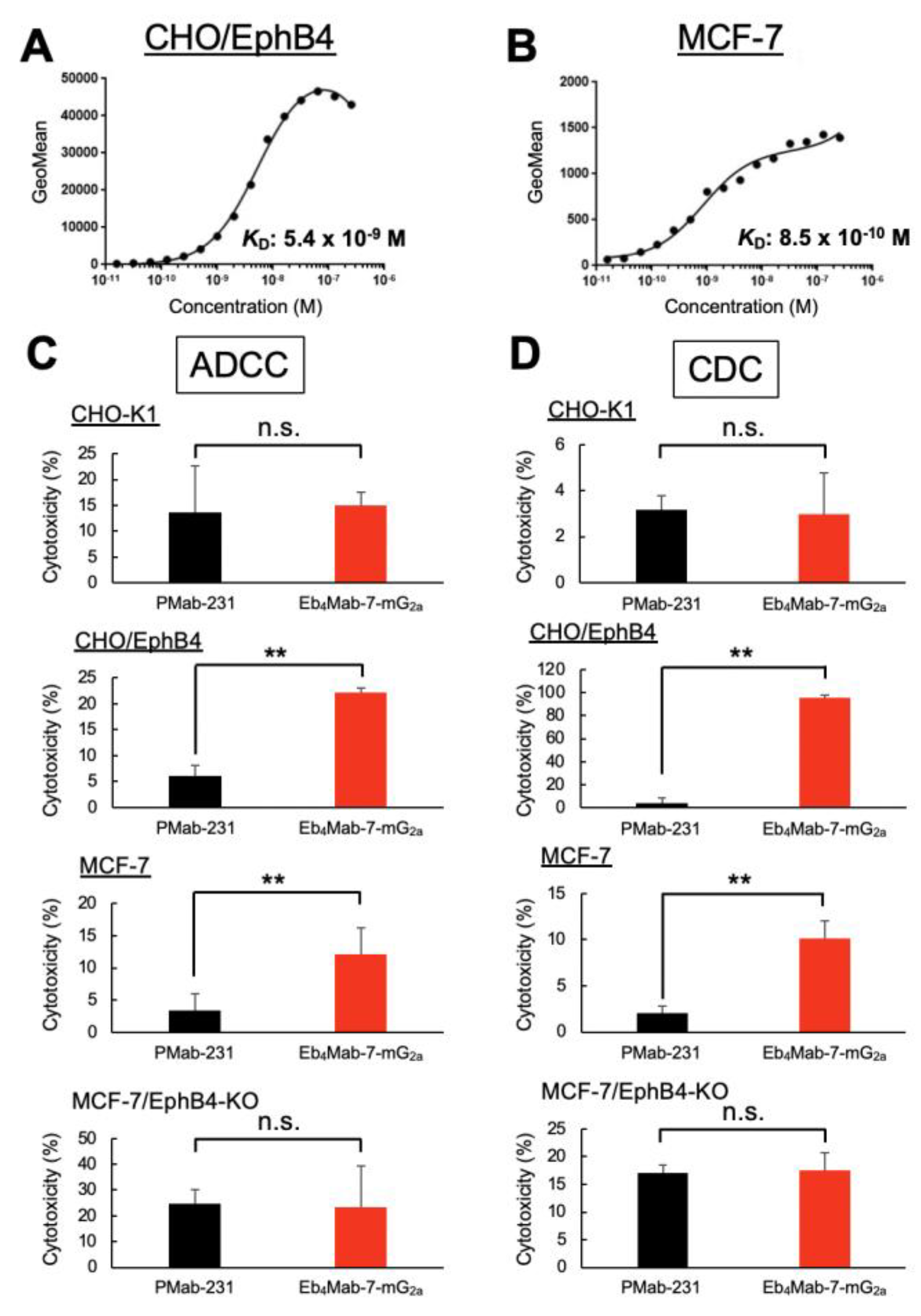
3.4. Binding Affinity of Eb4Mab-7-mG2a for EphB4-Expressing Cells
3.5. Evaluating the ADCC and CDC Activities of Eb4Mab-7-mG2a
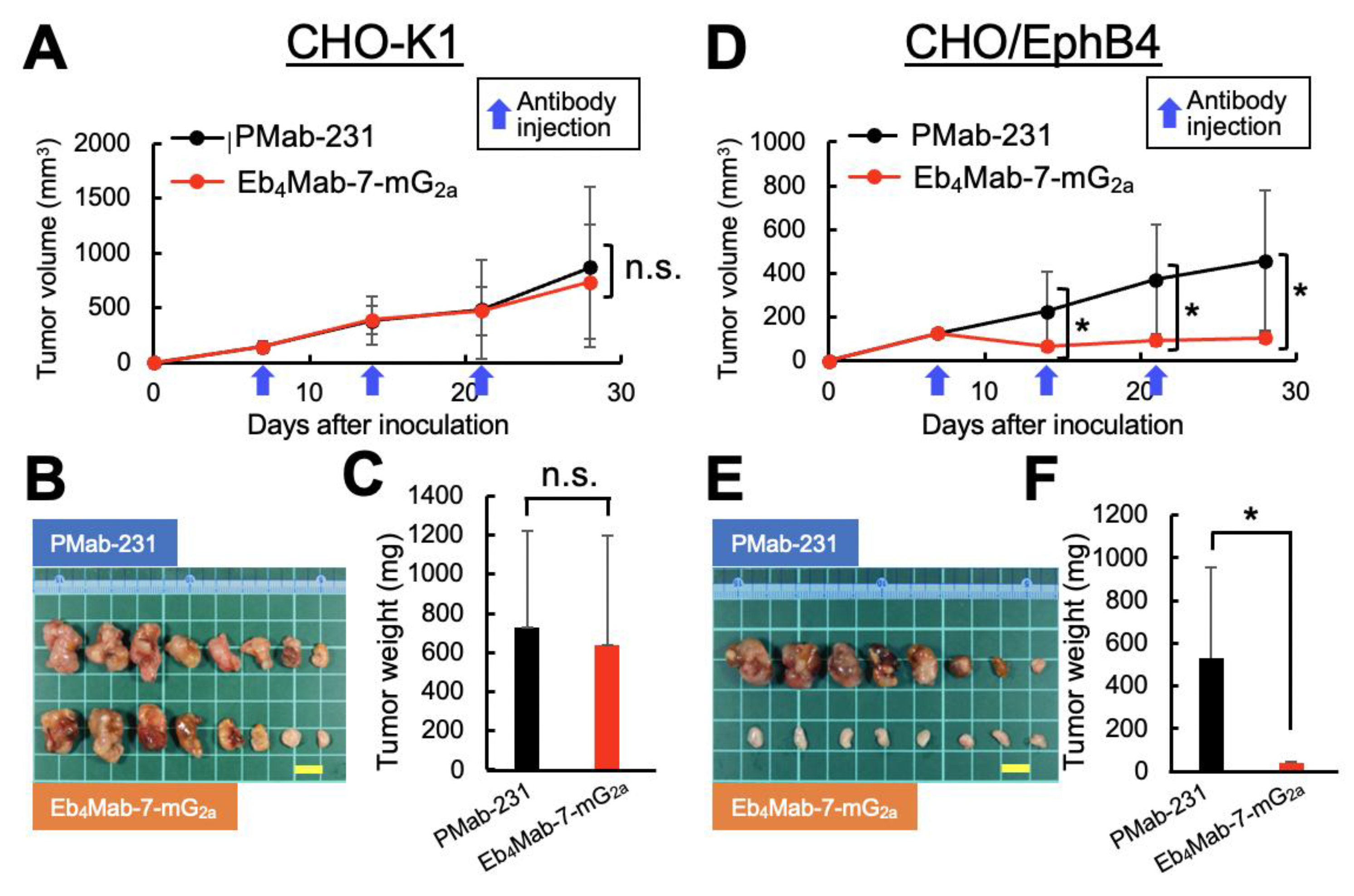
3.6. Antitumor Efficacy of Eb4Mab-7-mG2a in CHO-Derived Xenograft Models
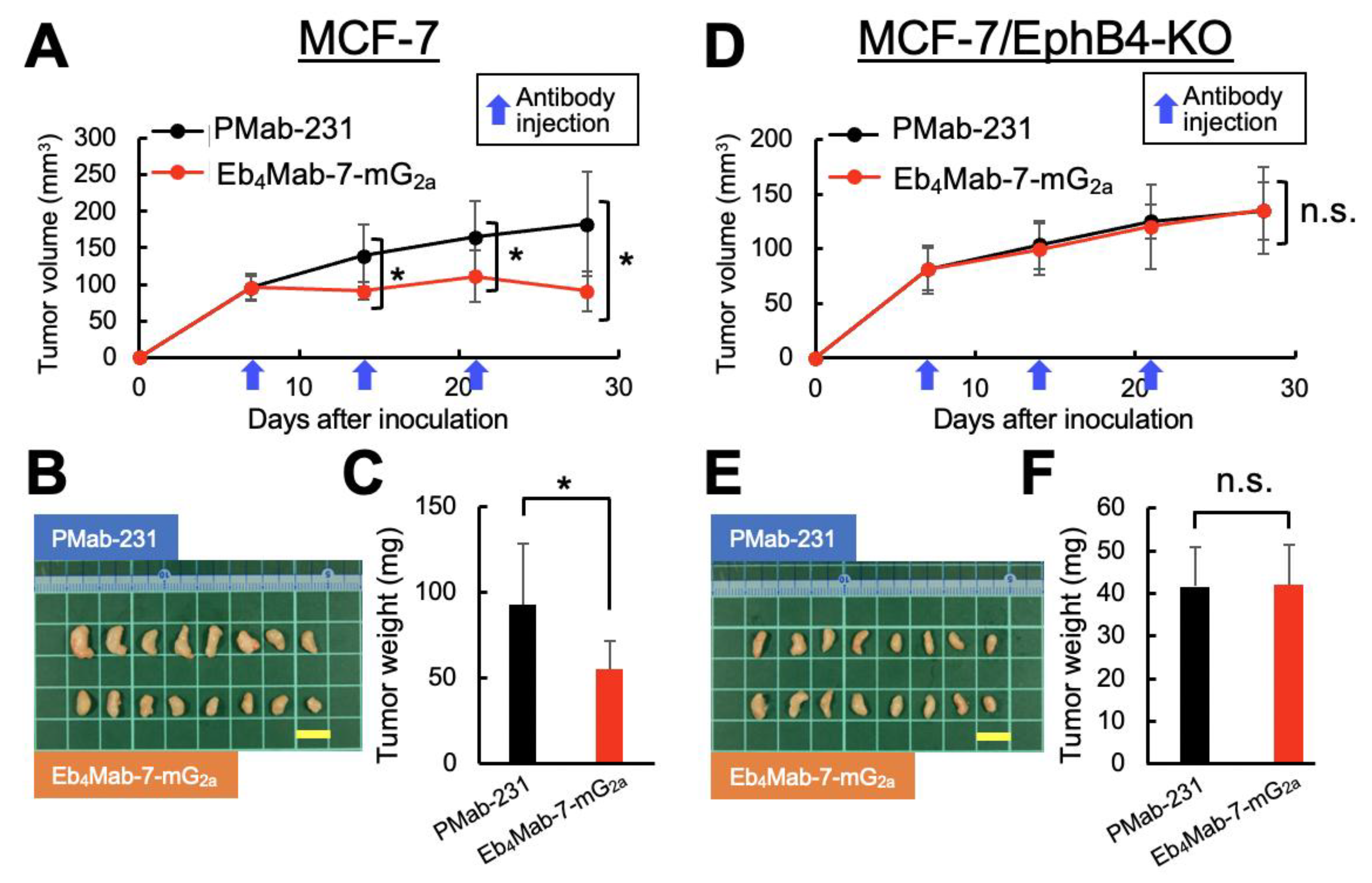
3.7. Antitumor Efficacy of Eb4Mab-7-mG2a in MCF-7-Derived Xenograft Models
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2022. CA Cancer J Clin 2022, 72, 7–33. [Google Scholar] [CrossRef]
- Pasquale, E.B. Eph receptor signalling casts a wide net on cell behaviour. Nat Rev Mol Cell Biol 2005, 6, 462–475. [Google Scholar] [CrossRef]
- Pasquale, E.B. Eph-ephrin bidirectional signaling in physiology and disease. Cell 2008, 133, 38–52. [Google Scholar] [CrossRef] [PubMed]
- Pasquale, E.B. Eph receptors and ephrins in cancer: bidirectional signalling and beyond. Nat Rev Cancer 2010, 10, 165–180. [Google Scholar] [CrossRef]
- Noren, N.K.; Foos, G.; Hauser, C.A.; Pasquale, E.B. The EphB4 receptor suppresses breast cancer cell tumorigenicity through an Abl-Crk pathway. Nat Cell Biol 2006, 8, 815–825. [Google Scholar] [CrossRef]
- Barquilla, A.; Pasquale, E.B. Eph receptors and ephrins: therapeutic opportunities. Annu Rev Pharmacol Toxicol 2015, 55, 465–487. [Google Scholar] [CrossRef]
- Xiao, Z.; Carrasco, R.; Kinneer, K.; et al. EphB4 promotes or suppresses Ras/MEK/ERK pathway in a context-dependent manner: Implications for EphB4 as a cancer target. Cancer Biol Ther 2012, 13, 630–637. [Google Scholar] [CrossRef] [PubMed]
- Martiny-Baron, G.; Korff, T.; Schaffner, F.; et al. Inhibition of tumor growth and angiogenesis by soluble EphB4. Neoplasia 2004, 6, 248–257. [Google Scholar] [CrossRef]
- Nanamiya, R.; Suzuki, H.; Kaneko, M.K.; Kato, Y. Development of an Anti-EphB4 Monoclonal Antibody for Multiple Applications Against Breast Cancers. Monoclon Antib Immunodiagn Immunother 2023, 42, 166–177. [Google Scholar] [CrossRef]
- Manso, T.; Kushwaha, A.; Abdollahi, N.; et al. Mechanisms of action of monoclonal antibodies in oncology integrated in IMGT/mAb-DB. Front Immunol 2023, 14, 1129323. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Nava, C.; Ortuno-Pineda, C.; Illades-Aguiar, B.; et al. Mechanisms of Action and Limitations of Monoclonal Antibodies and Single Chain Fragment Variable (scFv) in the Treatment of Cancer. Biomedicines 2023, 11. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Ohishi, T.; Tanaka, T.; Kaneko, M.K.; Kato, Y. A Cancer-Specific Monoclonal Antibody against Podocalyxin Exerted Antitumor Activities in Pancreatic Cancer Xenografts. Int J Mol Sci 2023, 25. [Google Scholar] [CrossRef]
- Ohishi, T.; Masuda, T.; Abe, H.; et al. Monotherapy with a novel intervenolin derivative, AS-1934, is an effective treatment for Helicobacter pylori infection. Helicobacter 2018, 23, e12470. [Google Scholar] [CrossRef] [PubMed]
- Kaneko, M.K.; Suzuki, H.; Ohishi, T.; et al. A Cancer-Specific Monoclonal Antibody against HER2 Exerts Antitumor Activities in Human Breast Cancer Xenograft Models. Int J Mol Sci 2024, 25. [Google Scholar] [CrossRef]
- Kawada, M.; Inoue, H.; Kajikawa, M.; et al. A novel monoclonal antibody targeting coxsackie virus and adenovirus receptor inhibits tumor growth in vivo. Sci Rep 2017, 7, 40400. [Google Scholar] [CrossRef]
- Jackovich, A.; Gitlitz, B.J.; Tiu-Lim, J.W.W.; et al. Improved efficacy of pembrolizumab combined with soluble EphB4-albumin in HPV-negative EphrinB2 positive head neck squamous cell carcinoma. Oncotarget 2024, 15, 444–458. [Google Scholar] [CrossRef]
- Sadeghi, S.; Quinn, D.; Dorff, T.; et al. EphrinB2 Inhibition and Pembrolizumab in Metastatic Urothelial Carcinoma. J Clin Oncol 2023, 41, 640–650. [Google Scholar] [CrossRef]
- VanderWeele, D.J.; Kocherginsky, M.; Munir, S.; et al. A Phase II Study of sEphB4-HSA in Metastatic Castration-Resistant Prostate Cancer. Clin Genitourin Cancer 2022, 20, 575–580. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K. A cancer-specific monoclonal antibody recognizes the aberrantly glycosylated podoplanin. Sci Rep 2014, 4, 5924. [Google Scholar] [CrossRef]
- Tanaka, T.; Suzuki, H.; Ohishi, T.; et al. Antitumor Activities by a Humanized Cancer-Specific Anti-Podoplanin Monoclonal Antibody humPMab-117 Against Human Tumors. Cancer Sci 2025. [Google Scholar] [CrossRef] [PubMed]
- Hou, A.J.; Chen, L.C.; Chen, Y.Y. Navigating CAR-T cells through the solid-tumour microenvironment. Nat Rev Drug Discov 2021, 20, 531–550. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



