Submitted:
27 June 2025
Posted:
30 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
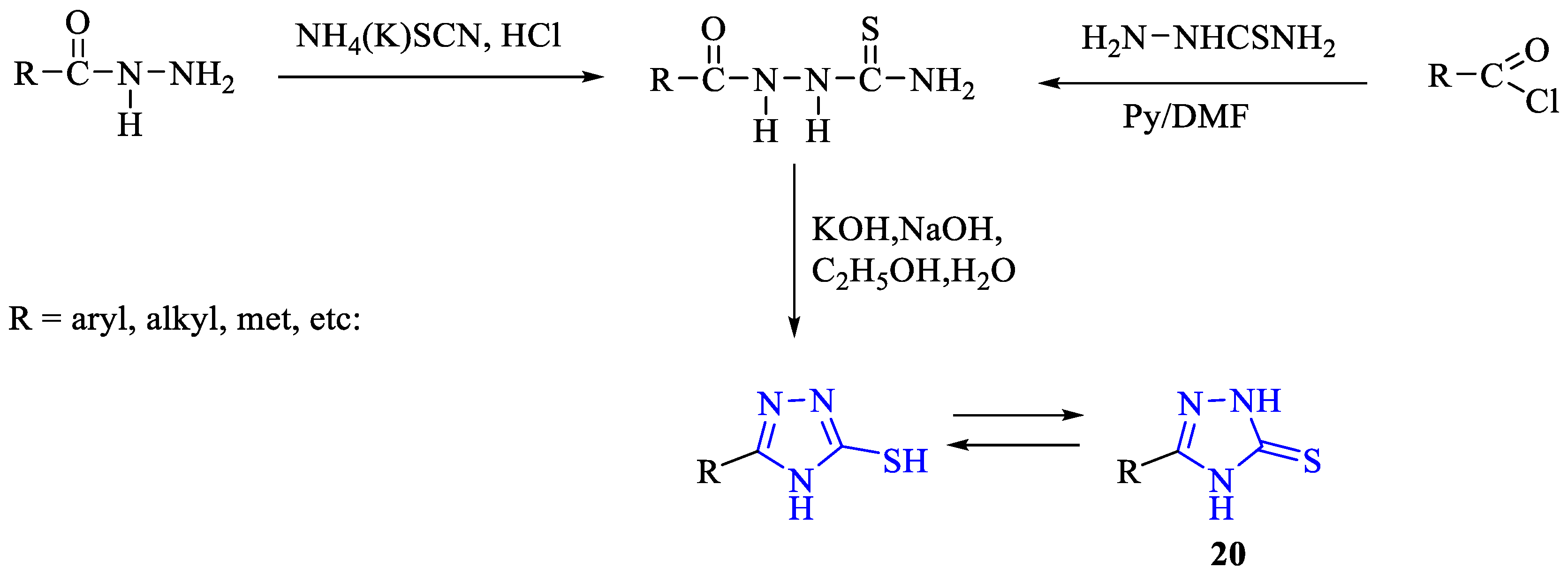
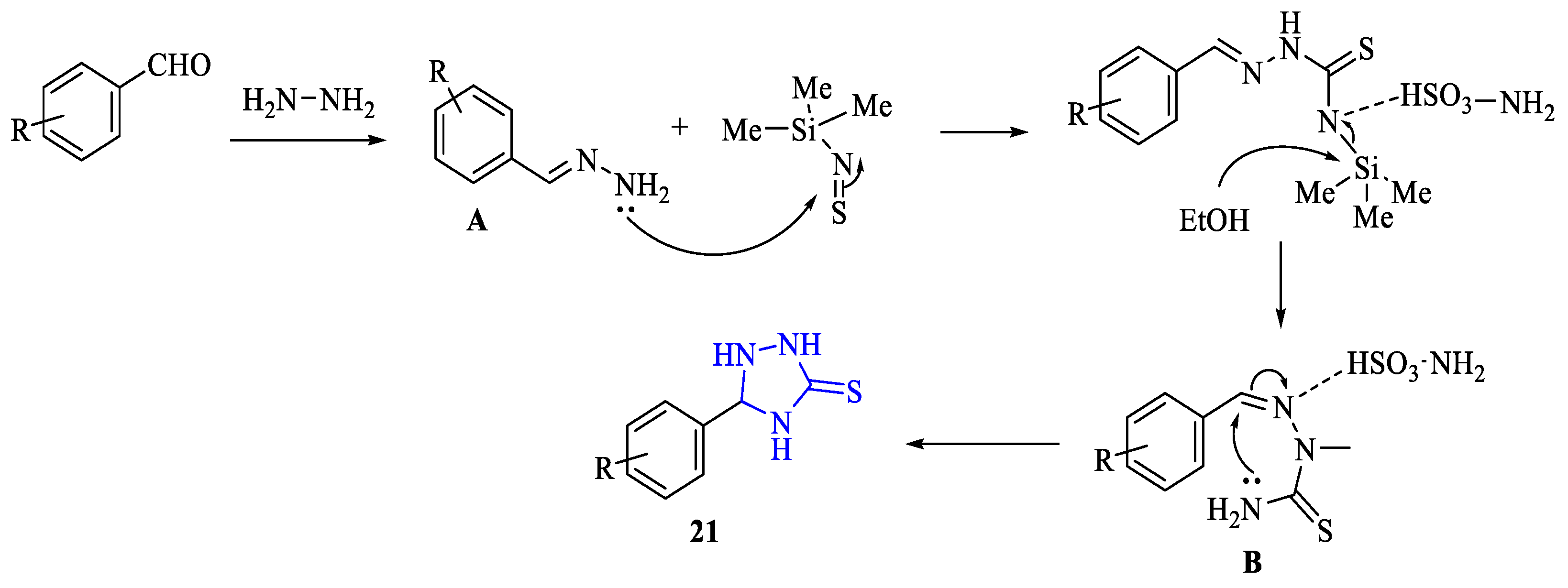
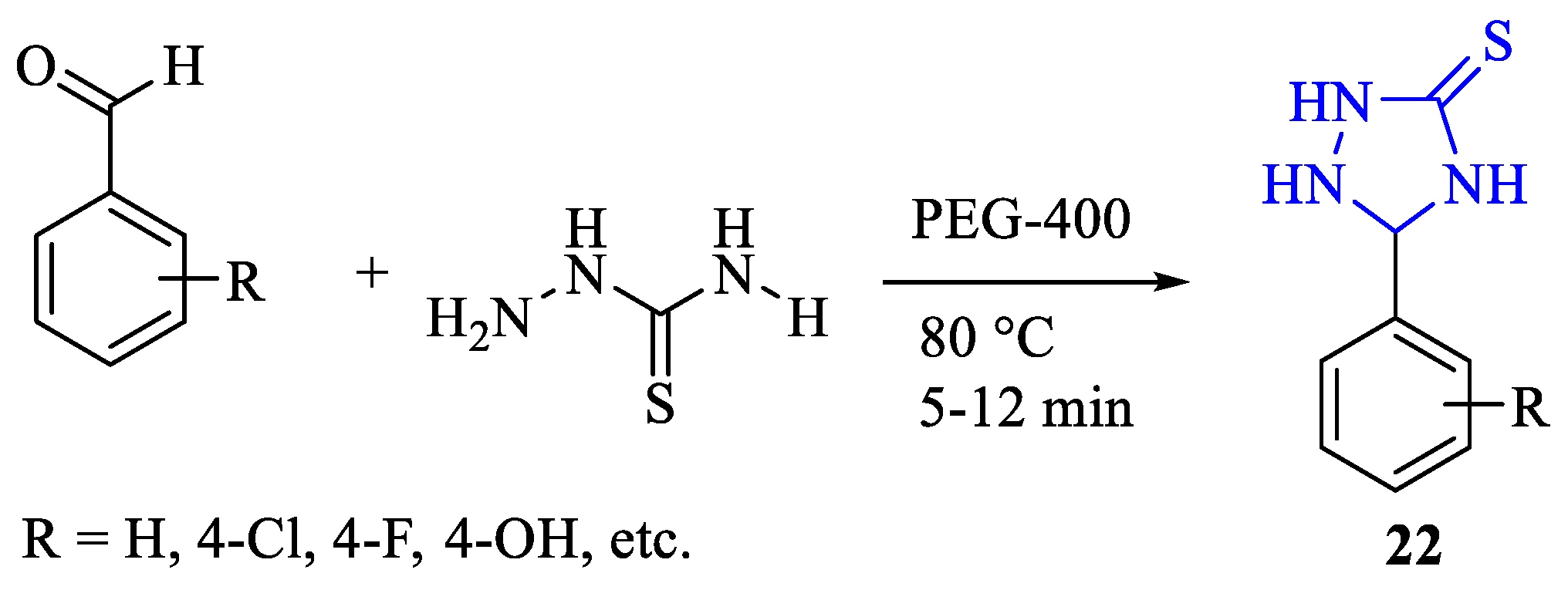
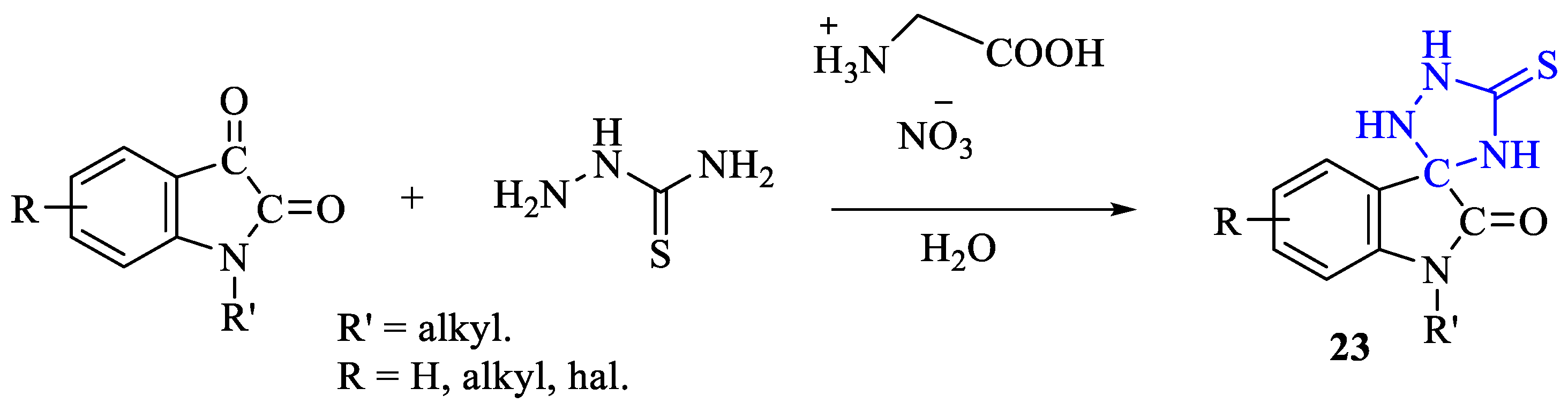
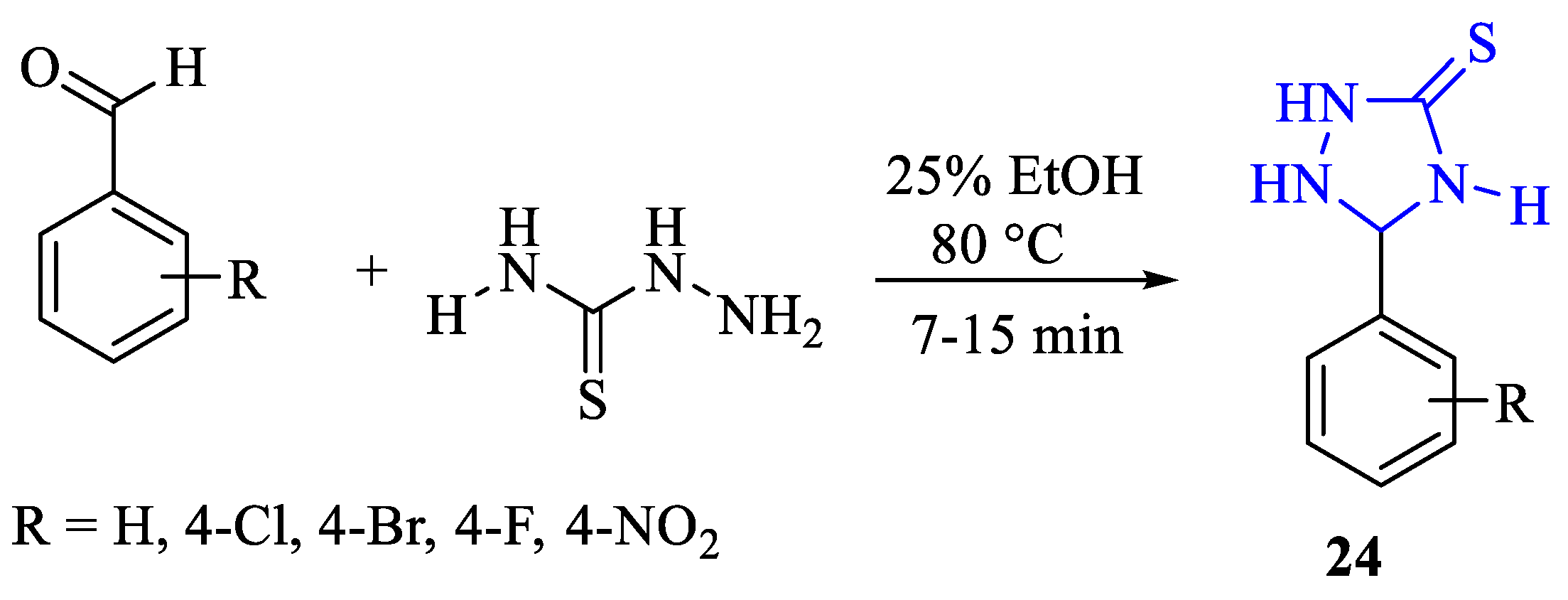
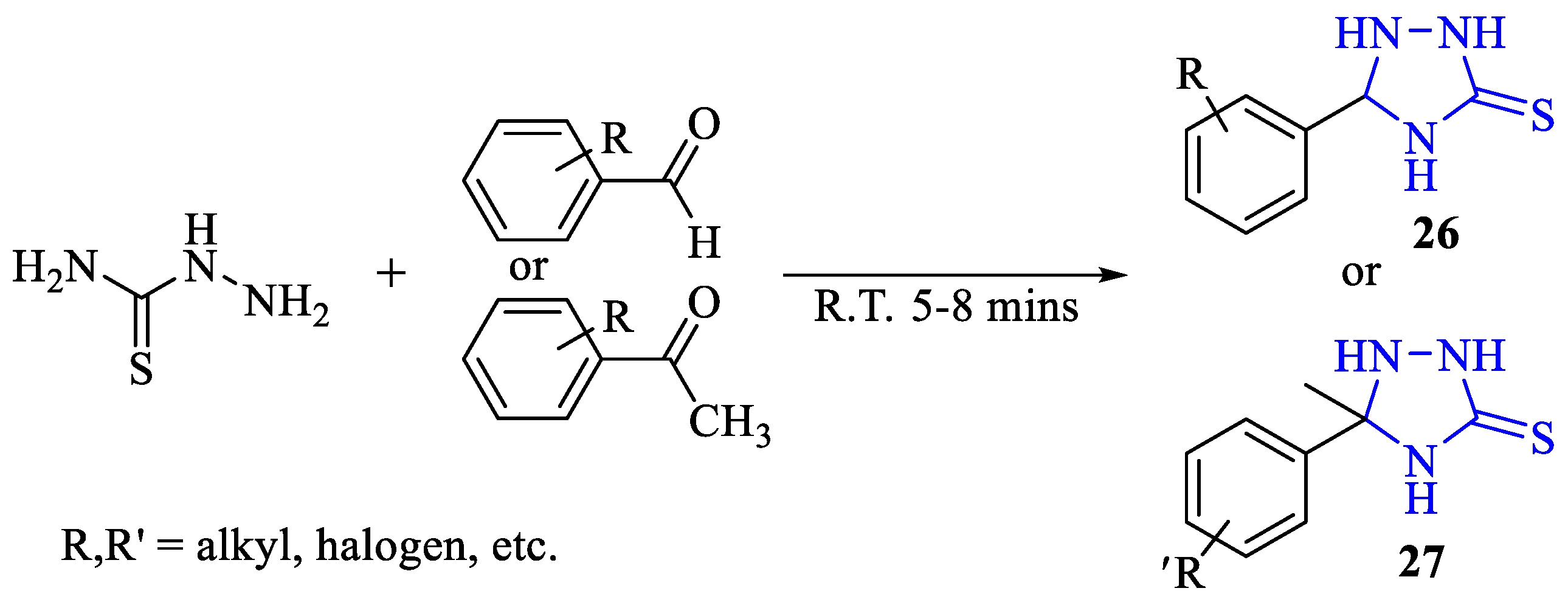
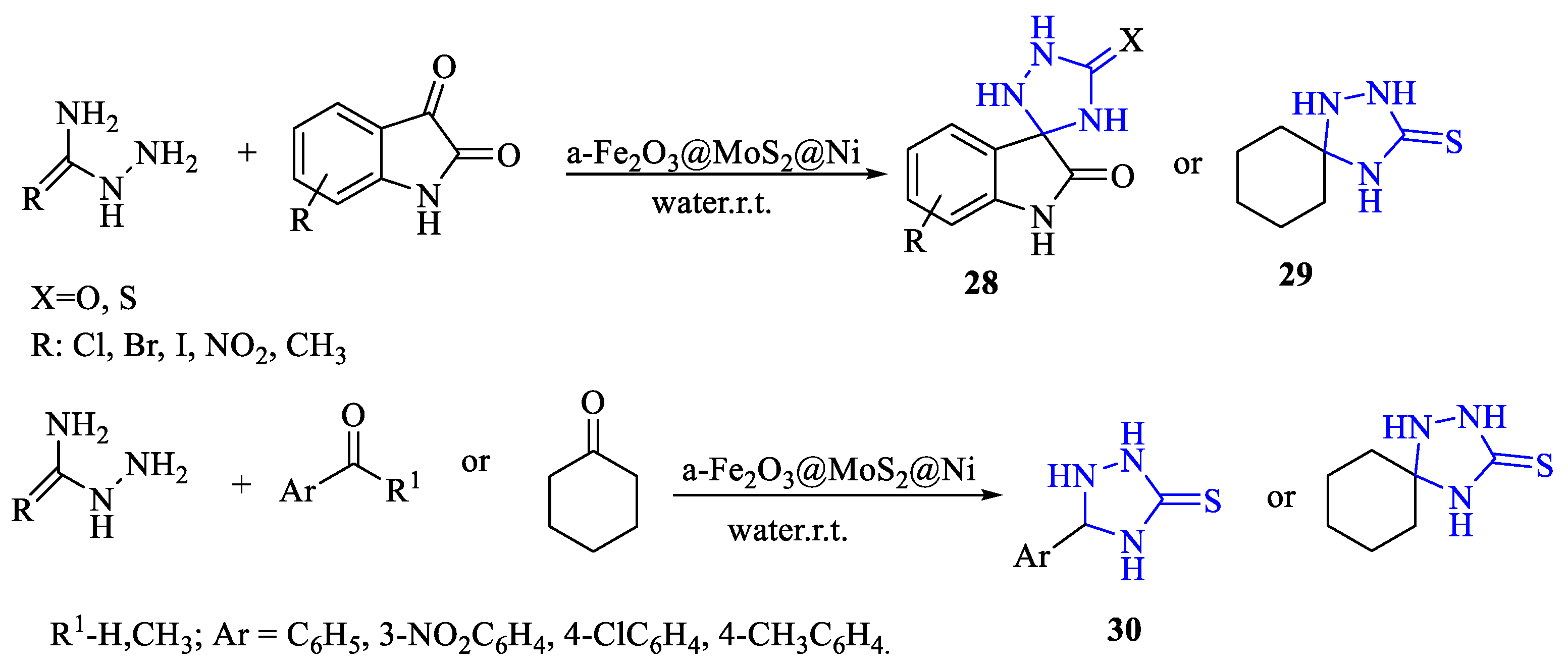
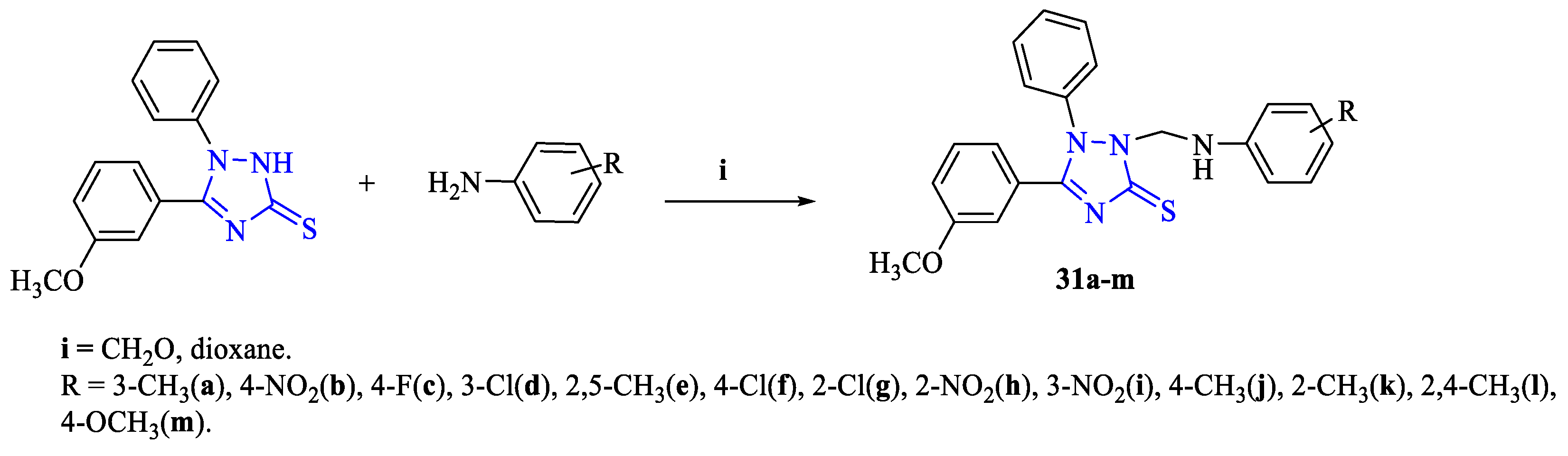
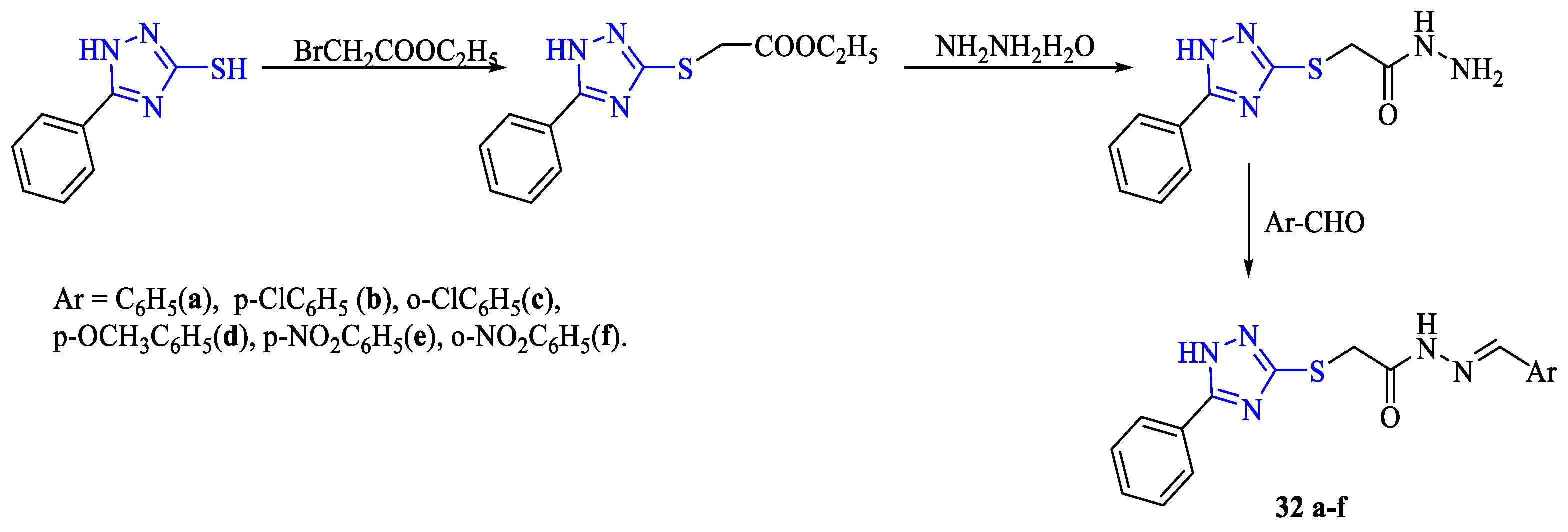
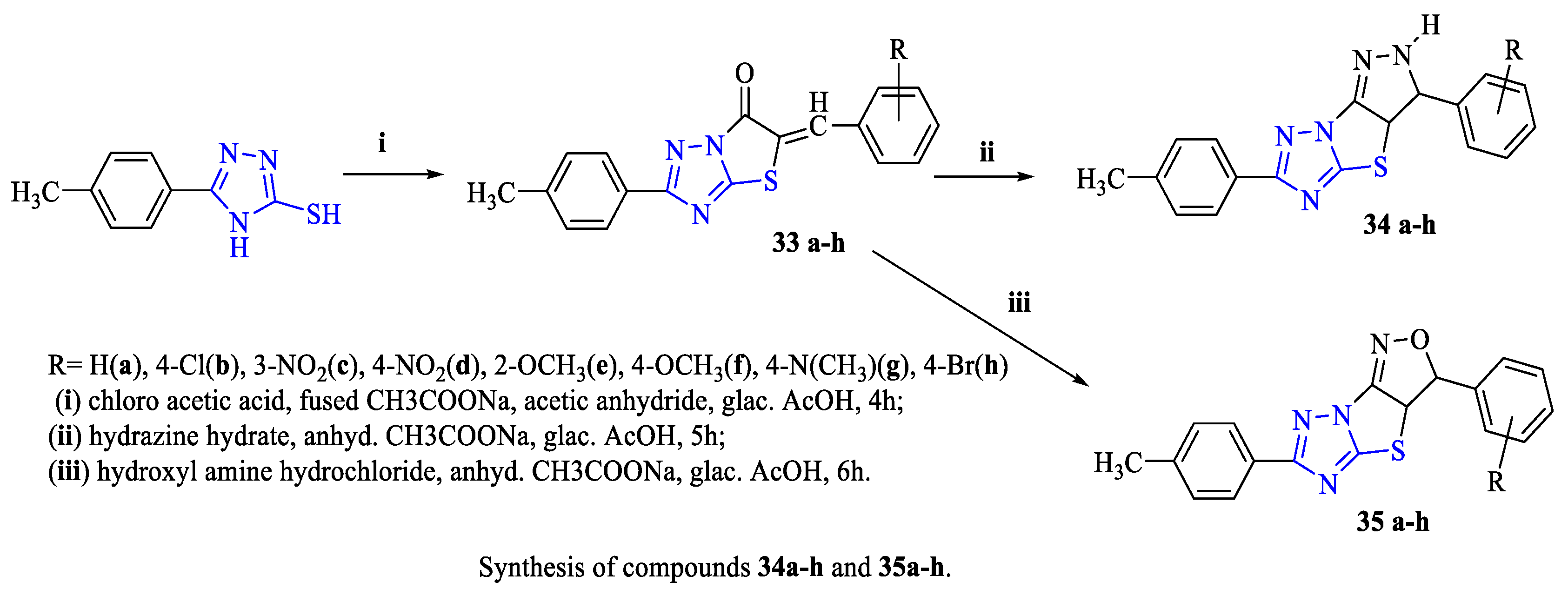
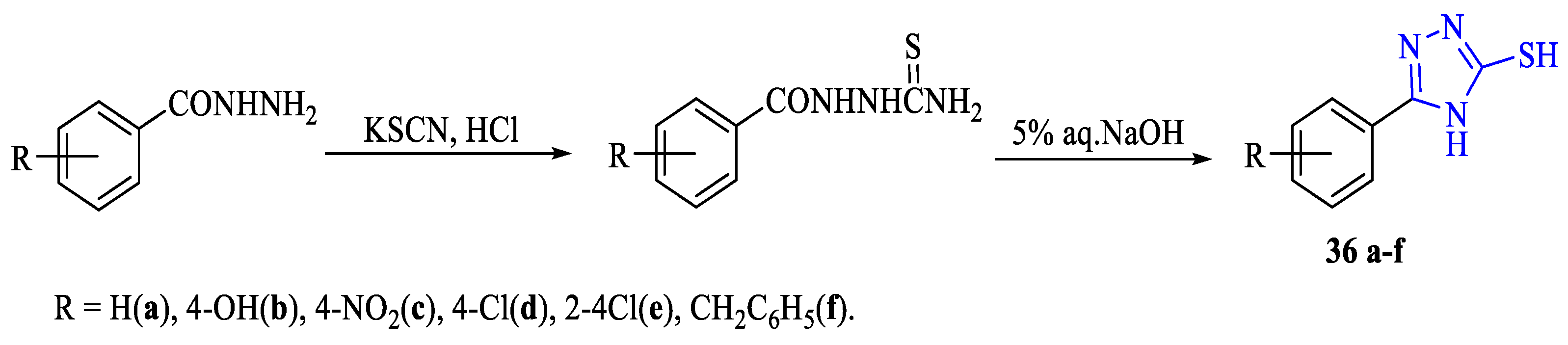
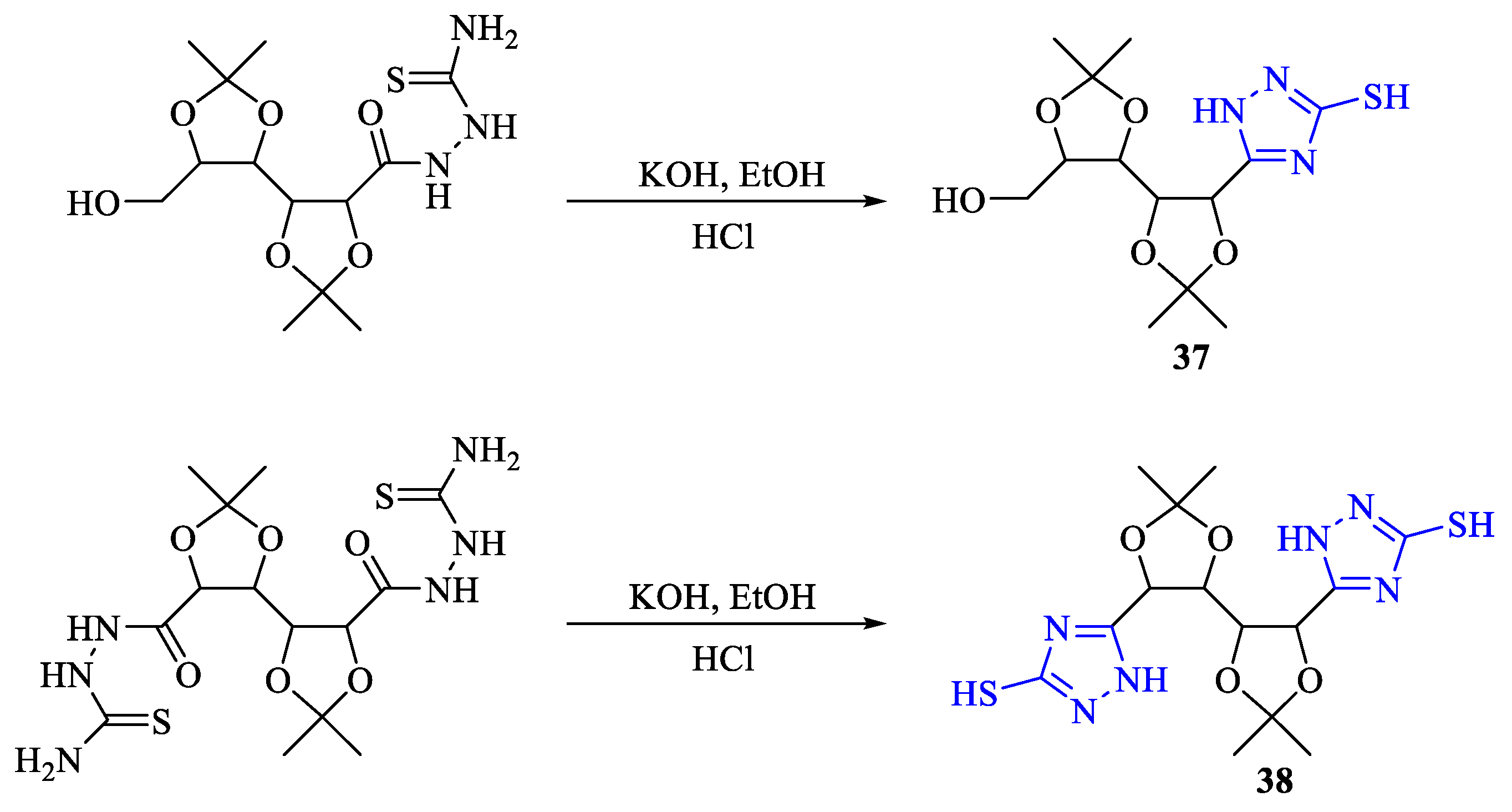
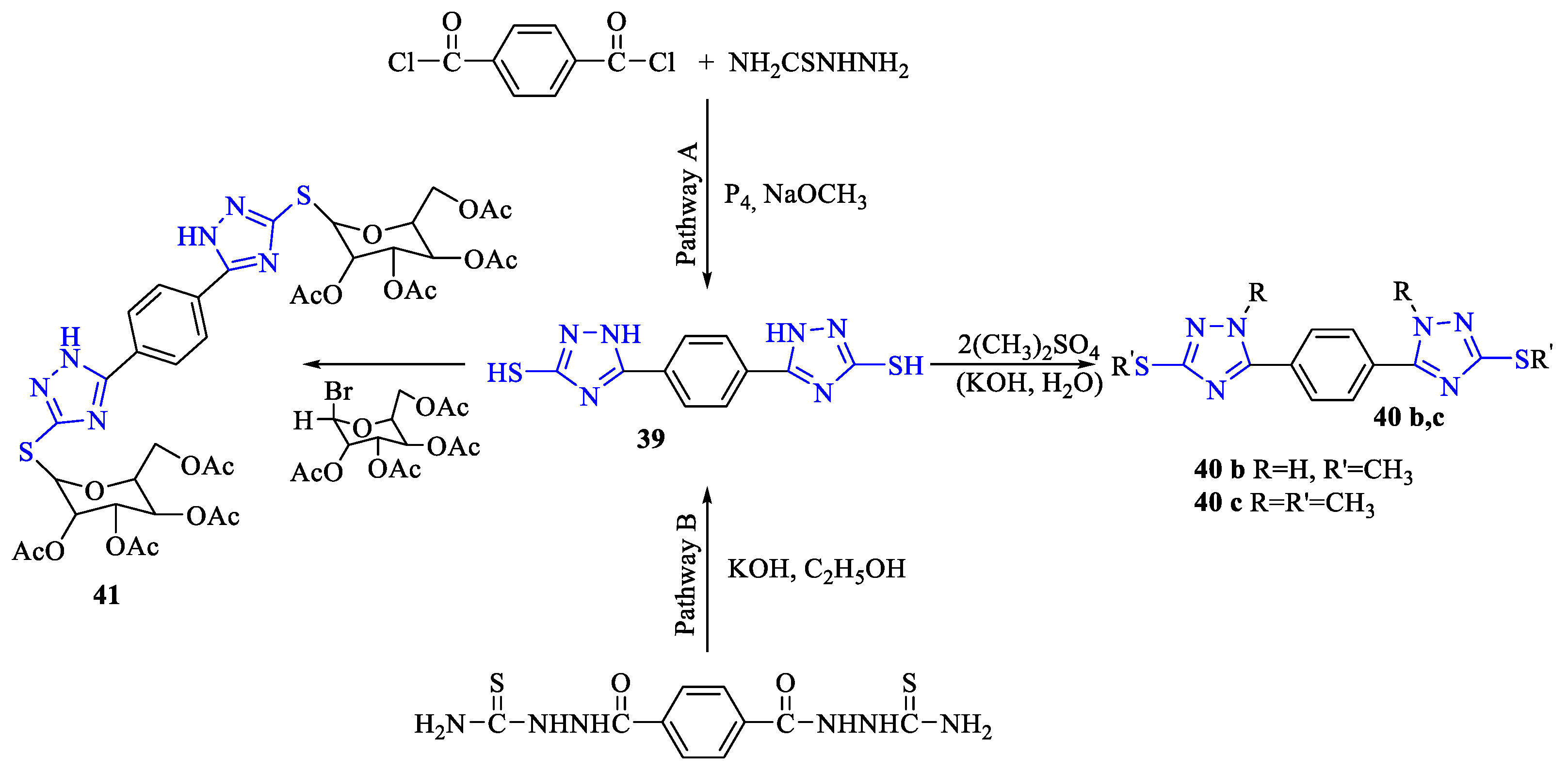
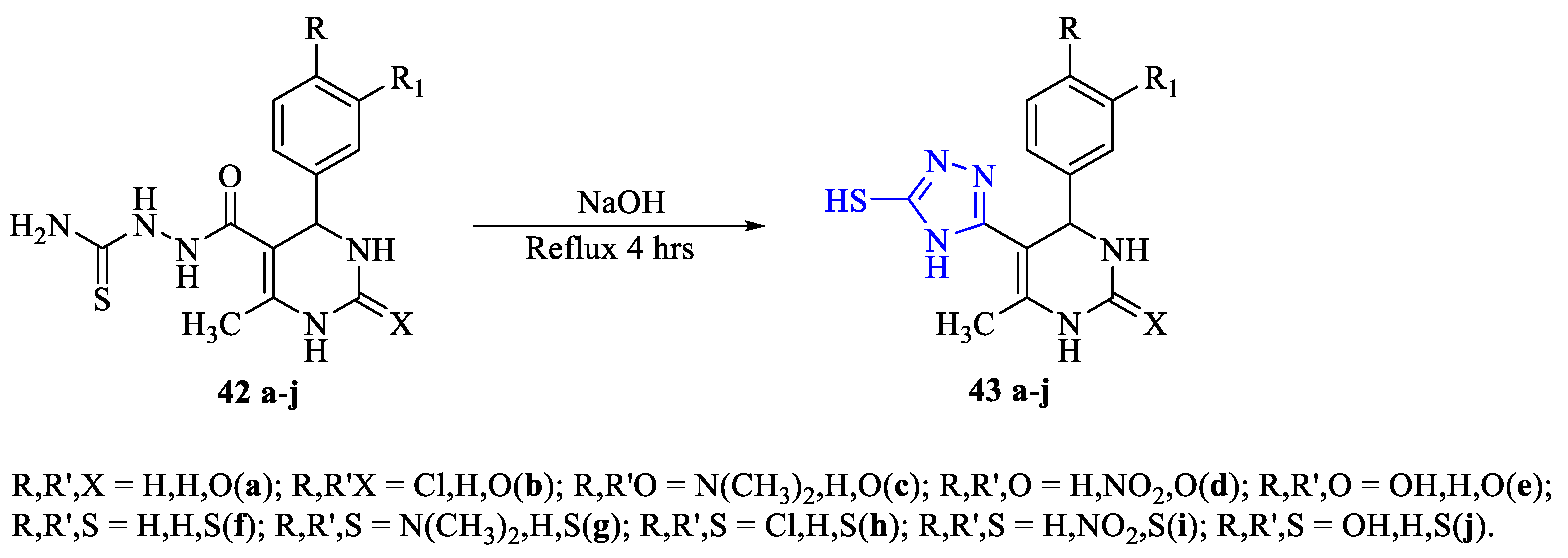
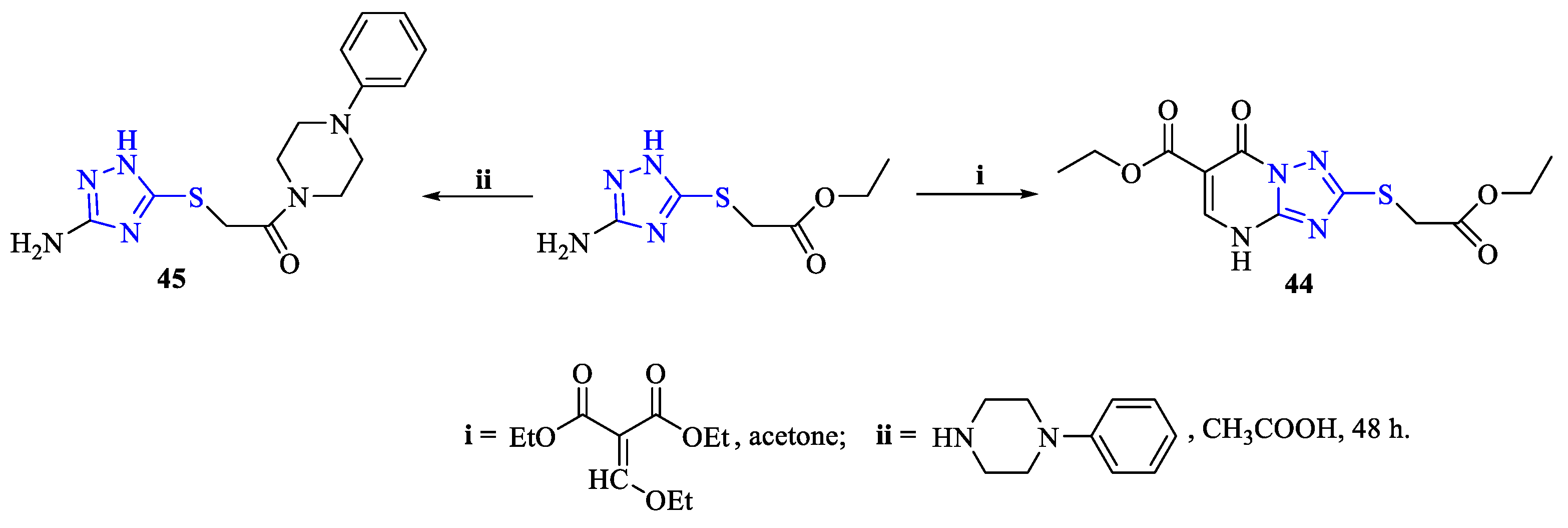
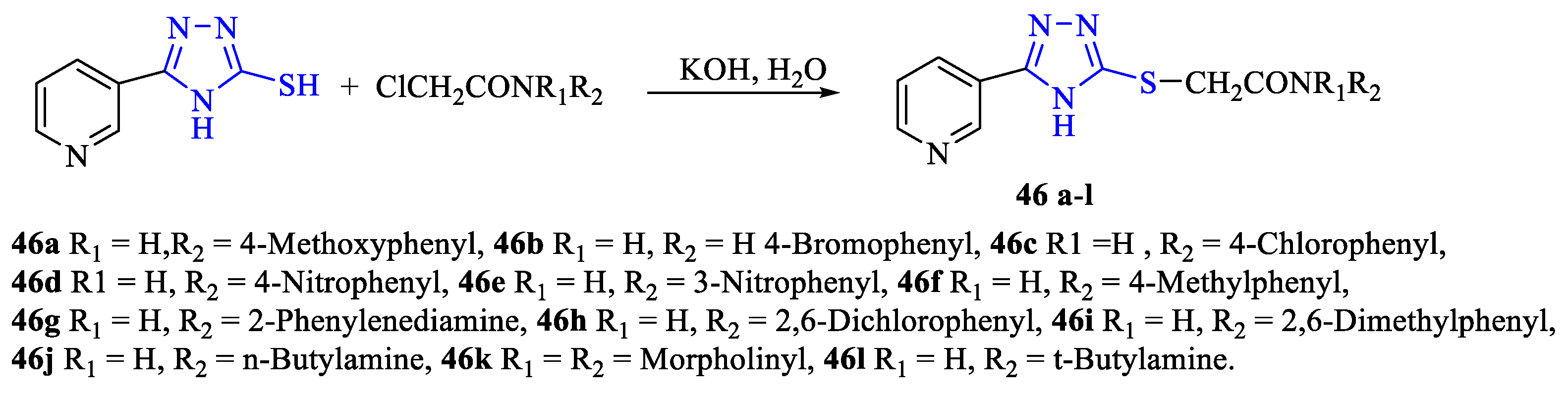
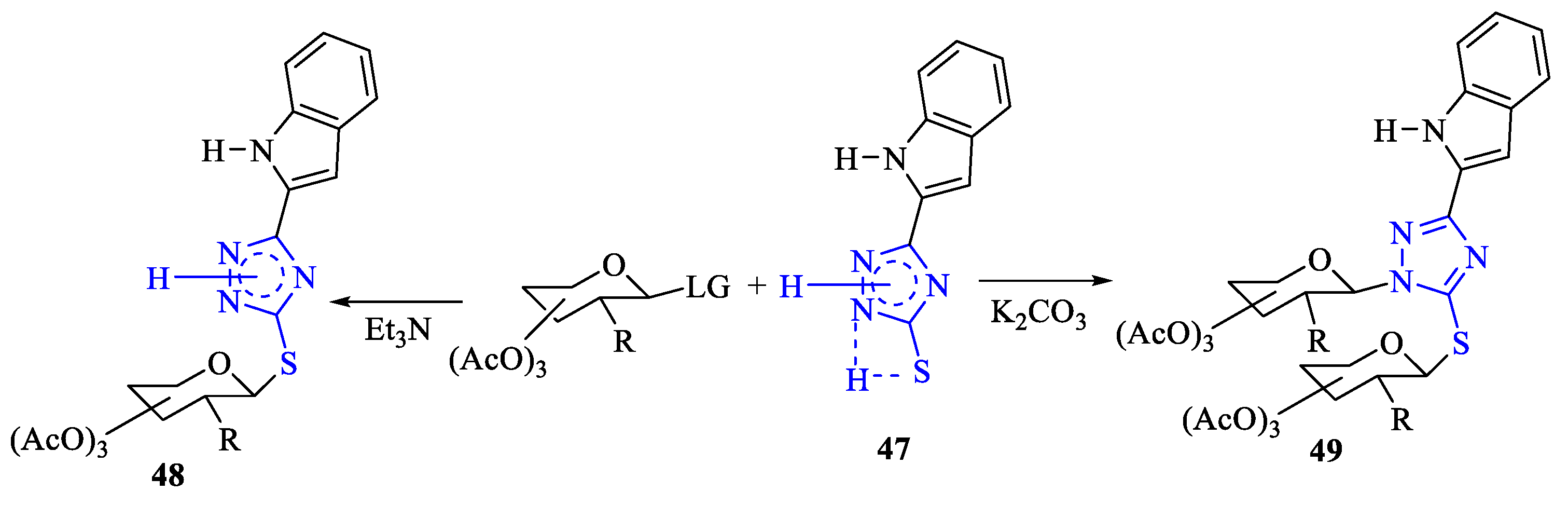
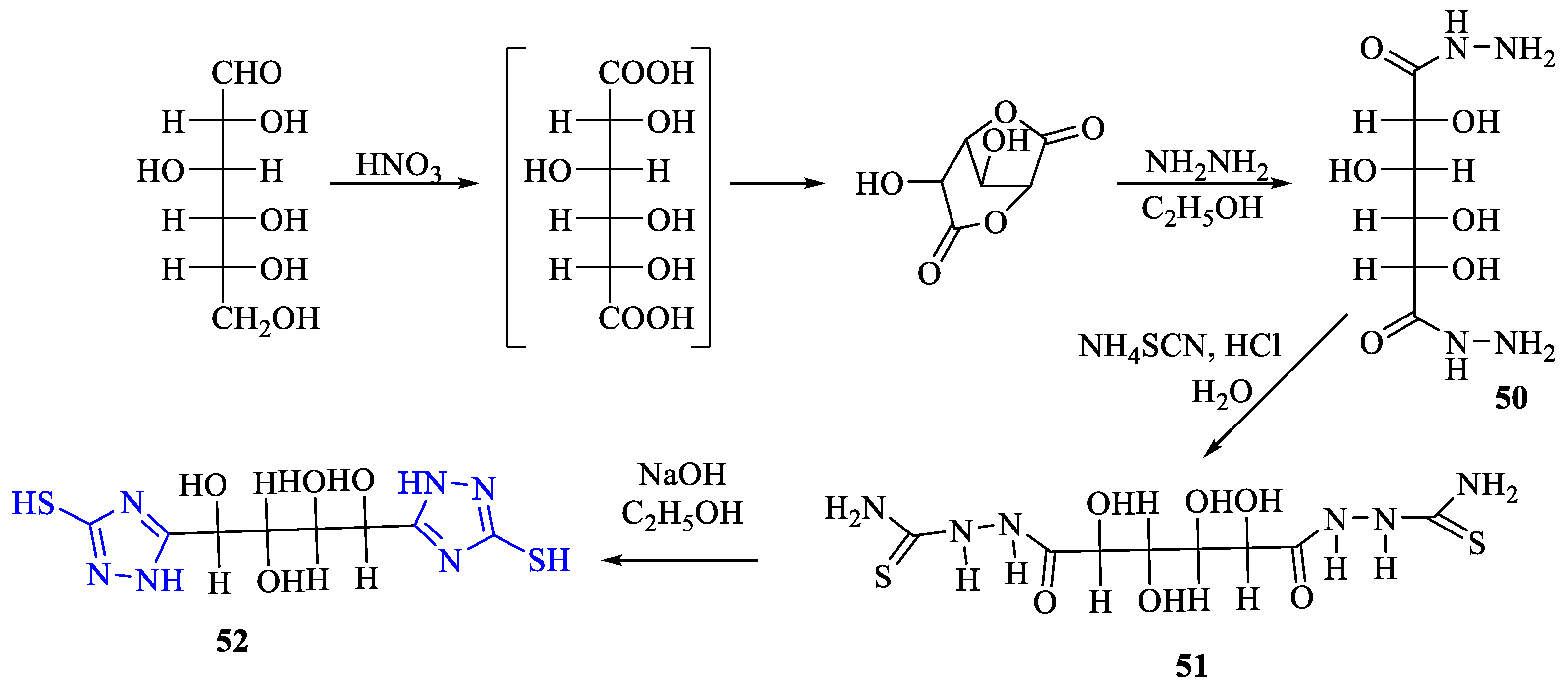
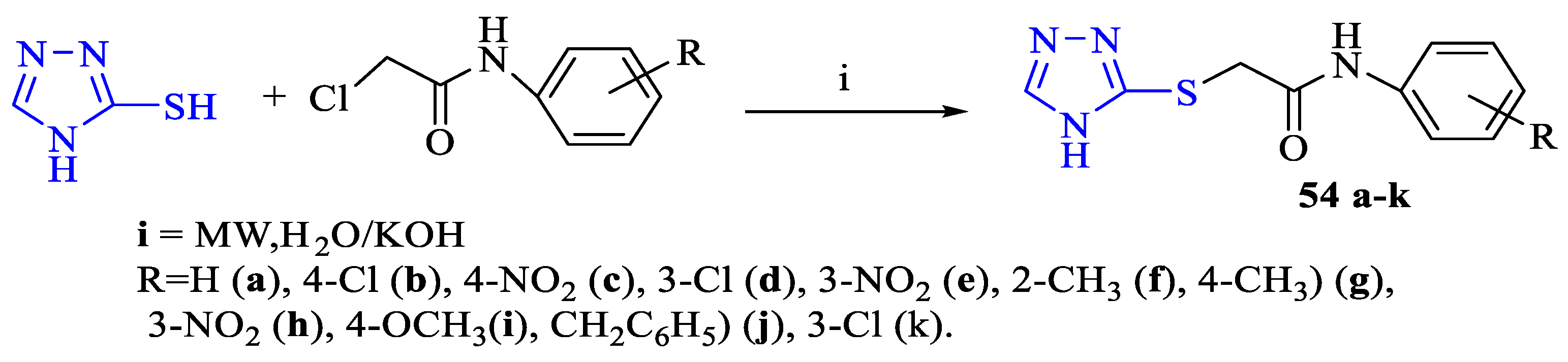
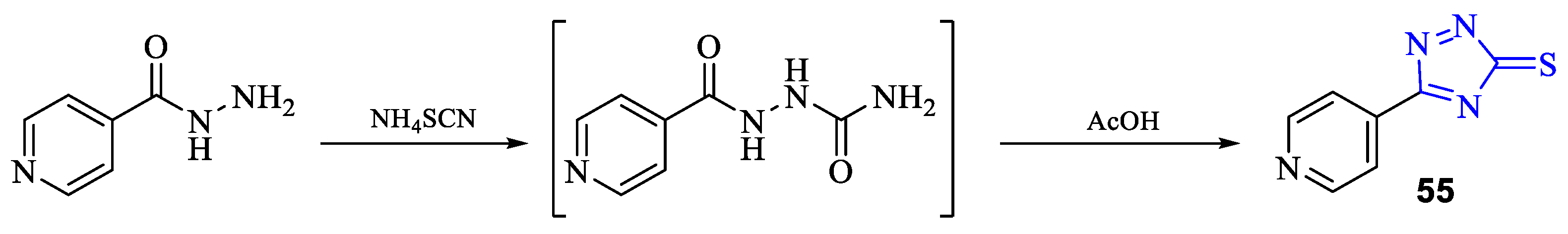
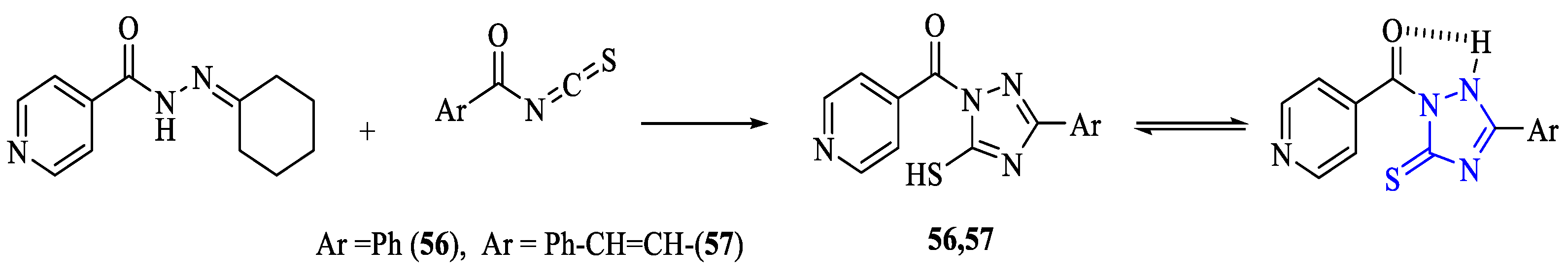
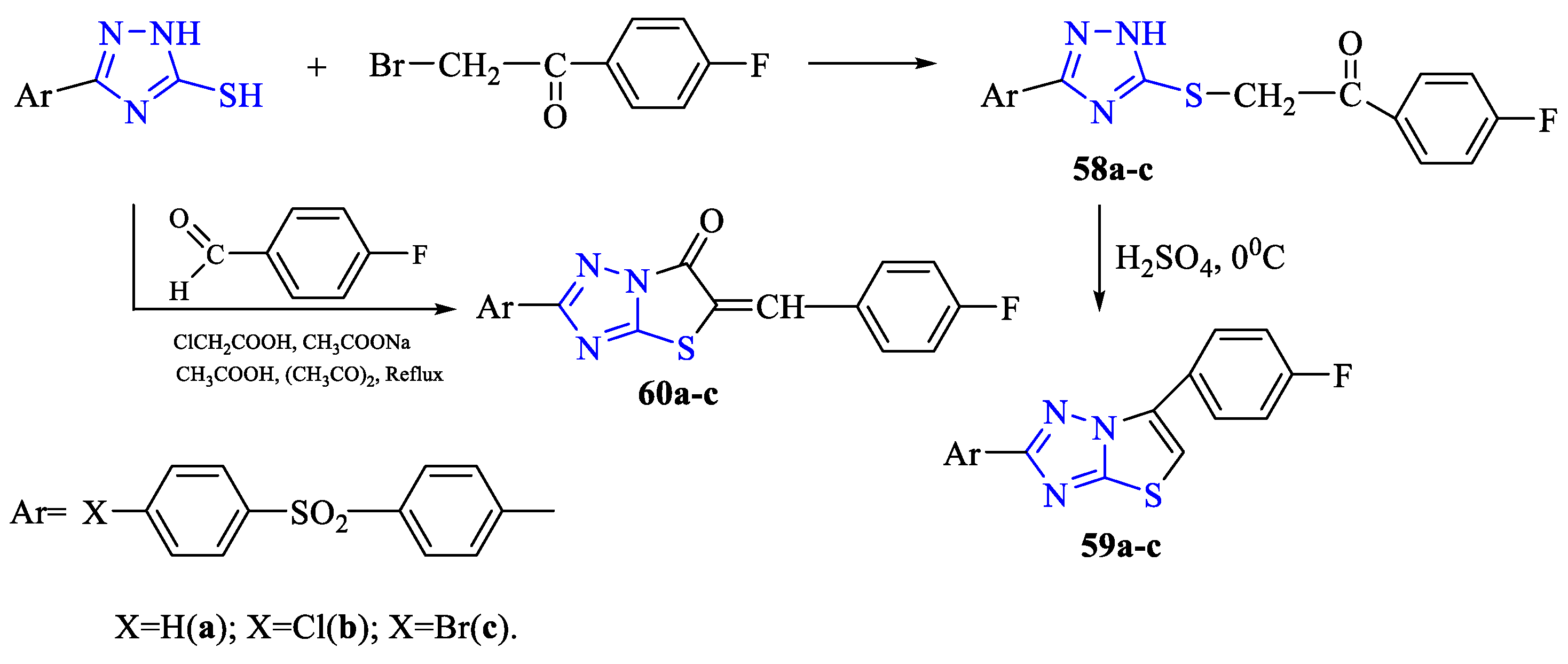
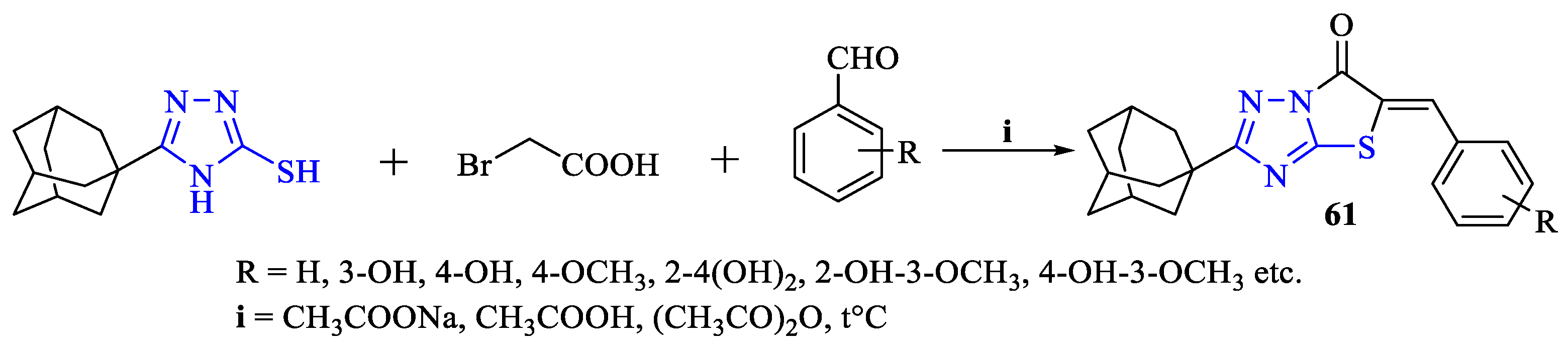
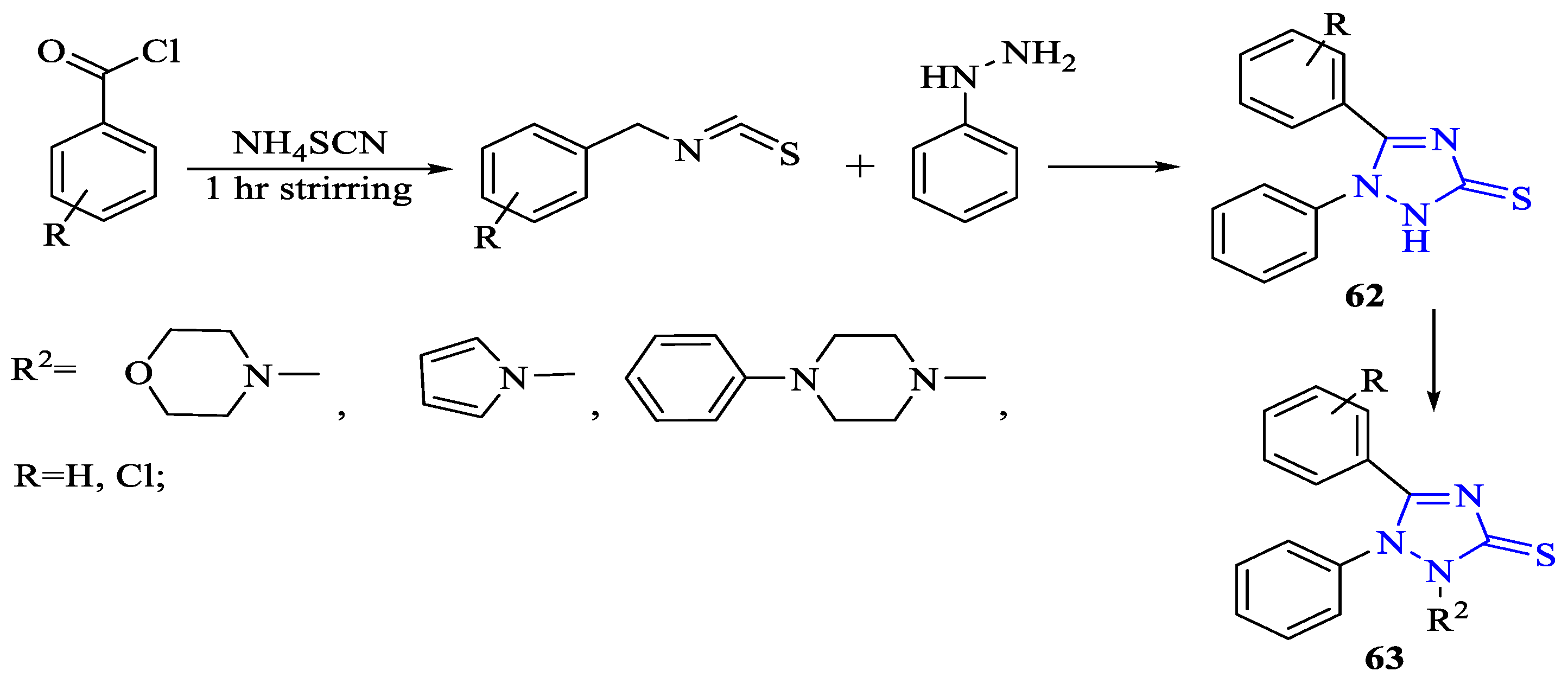
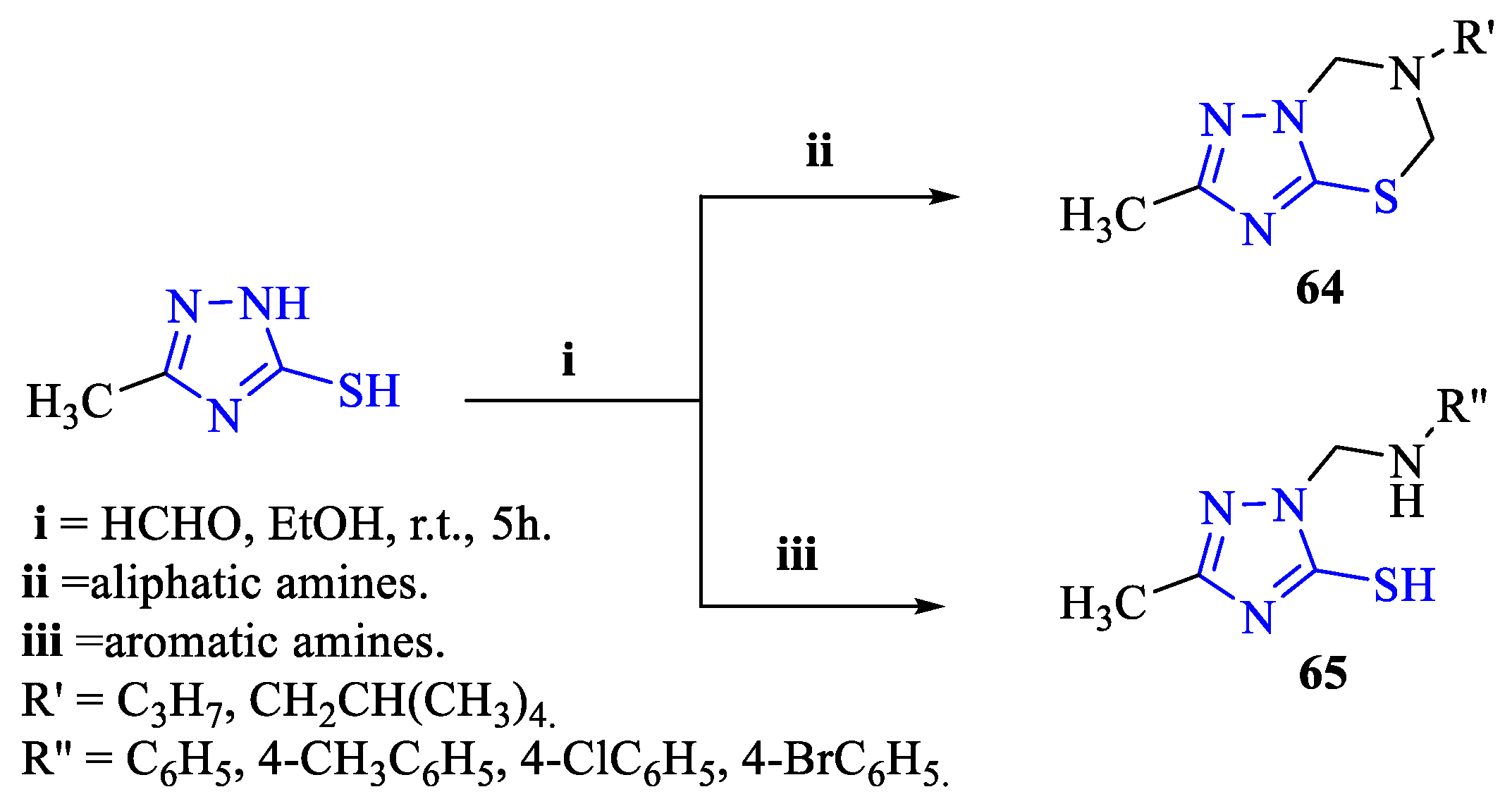
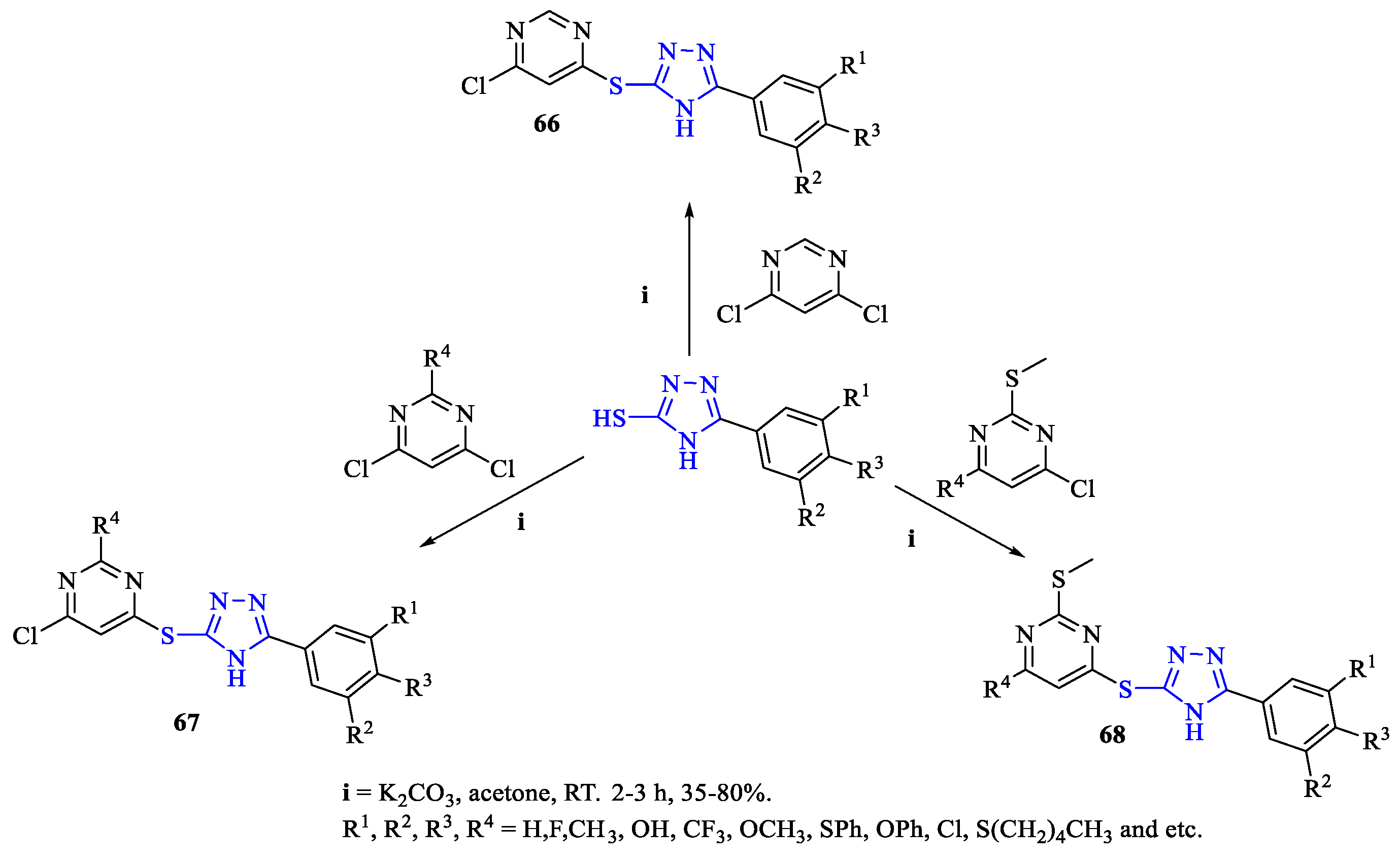
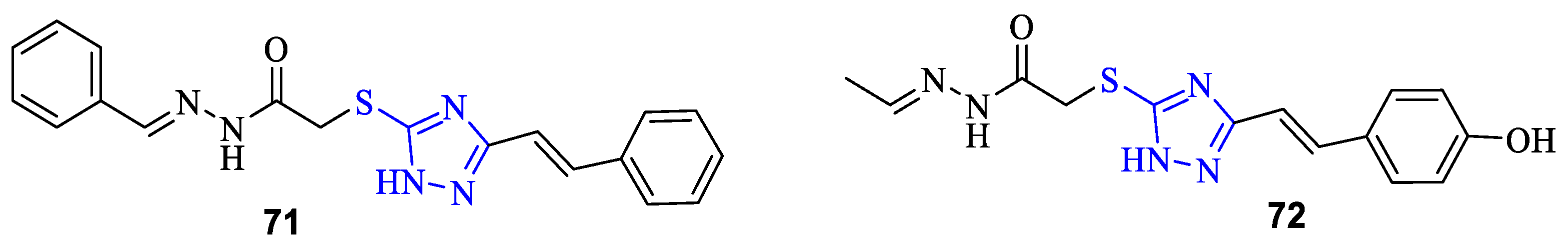
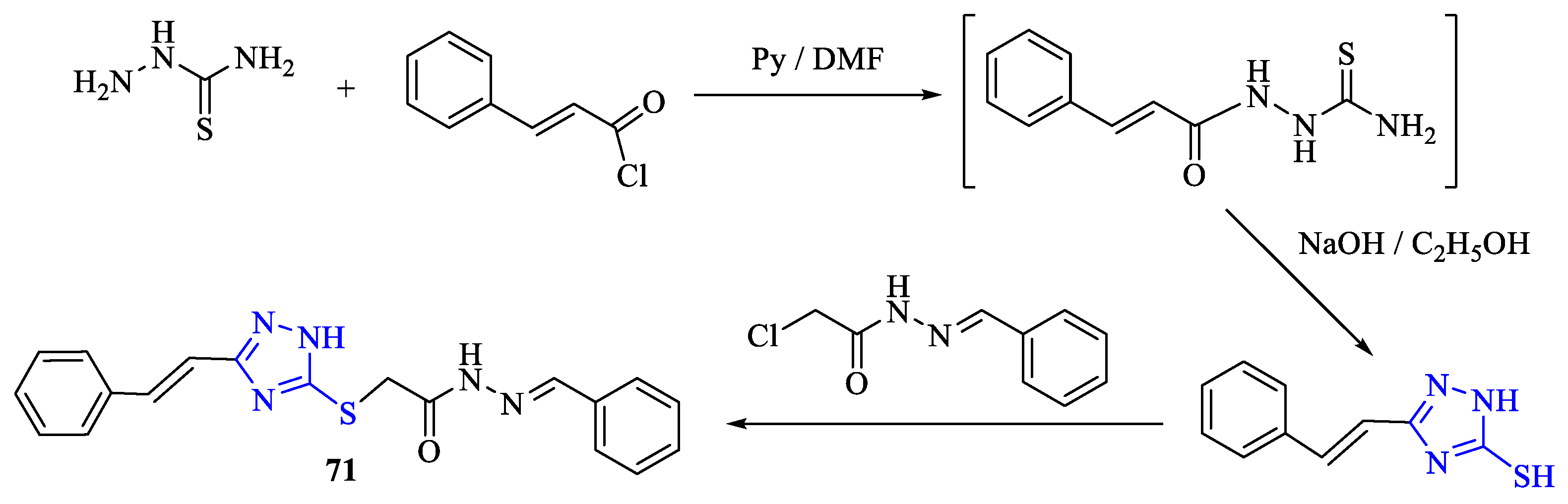
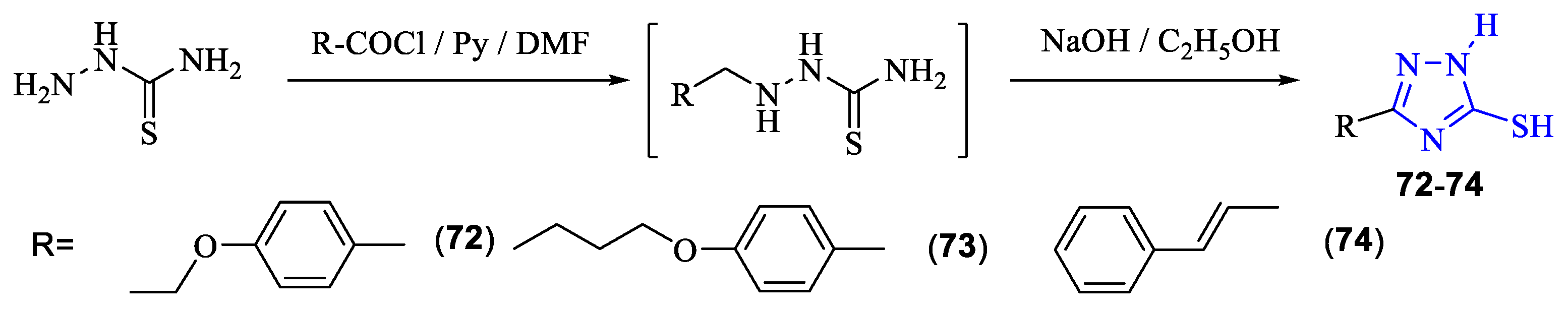
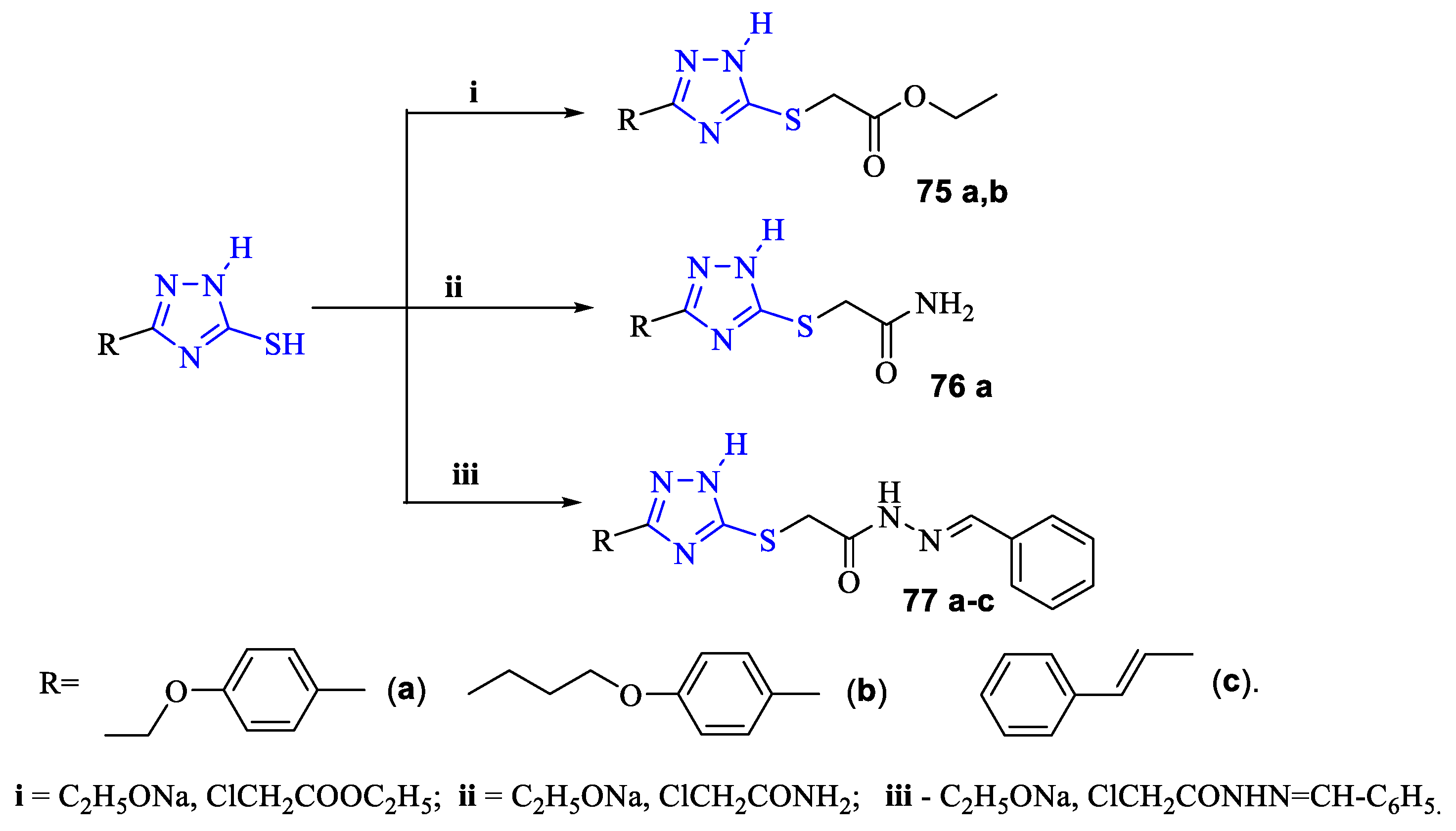
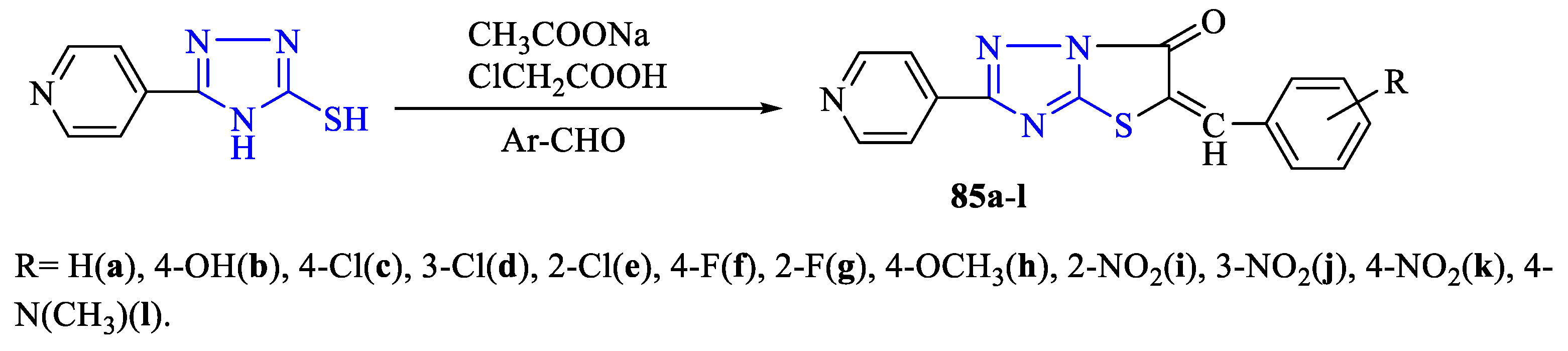
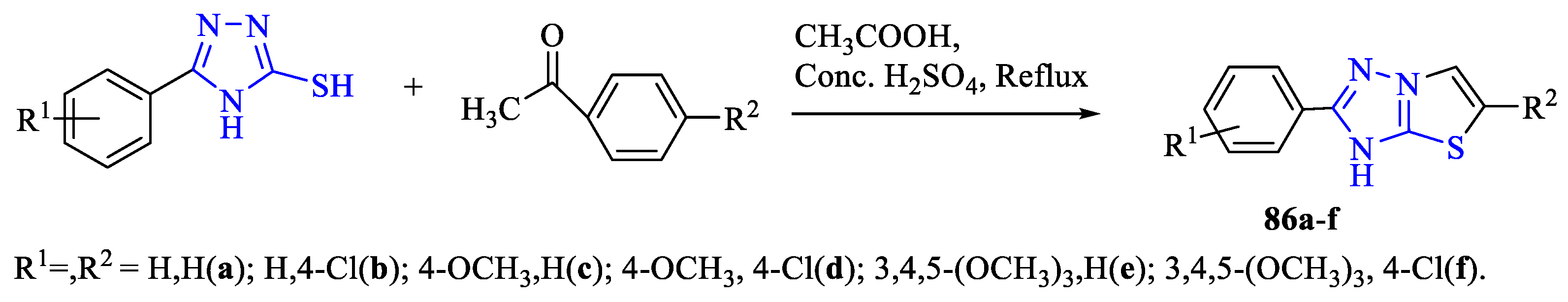
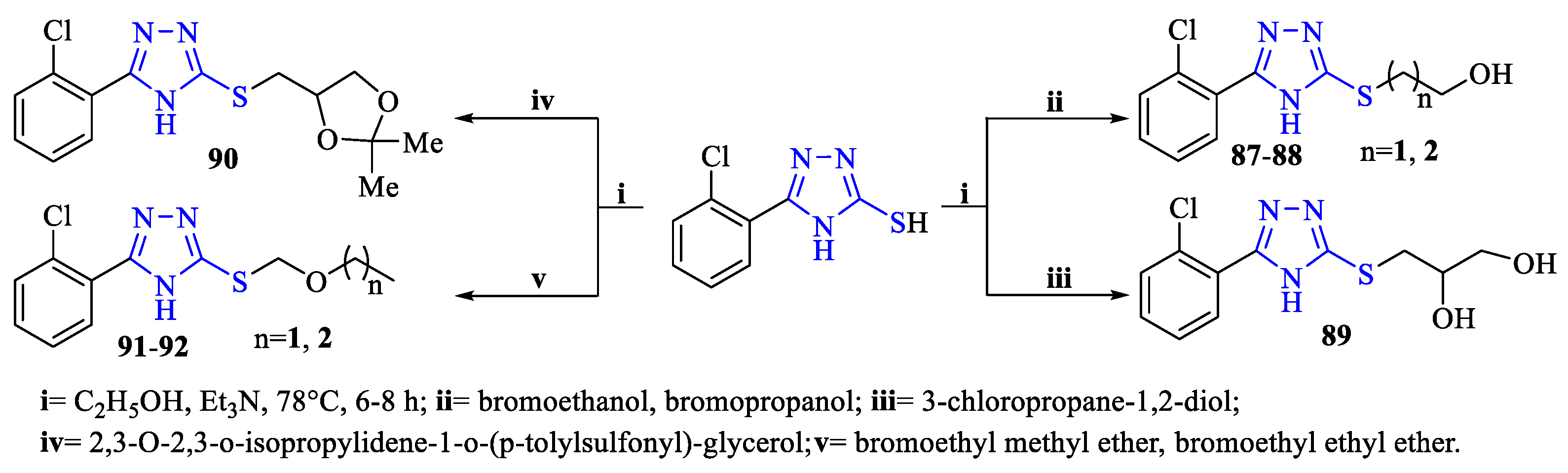
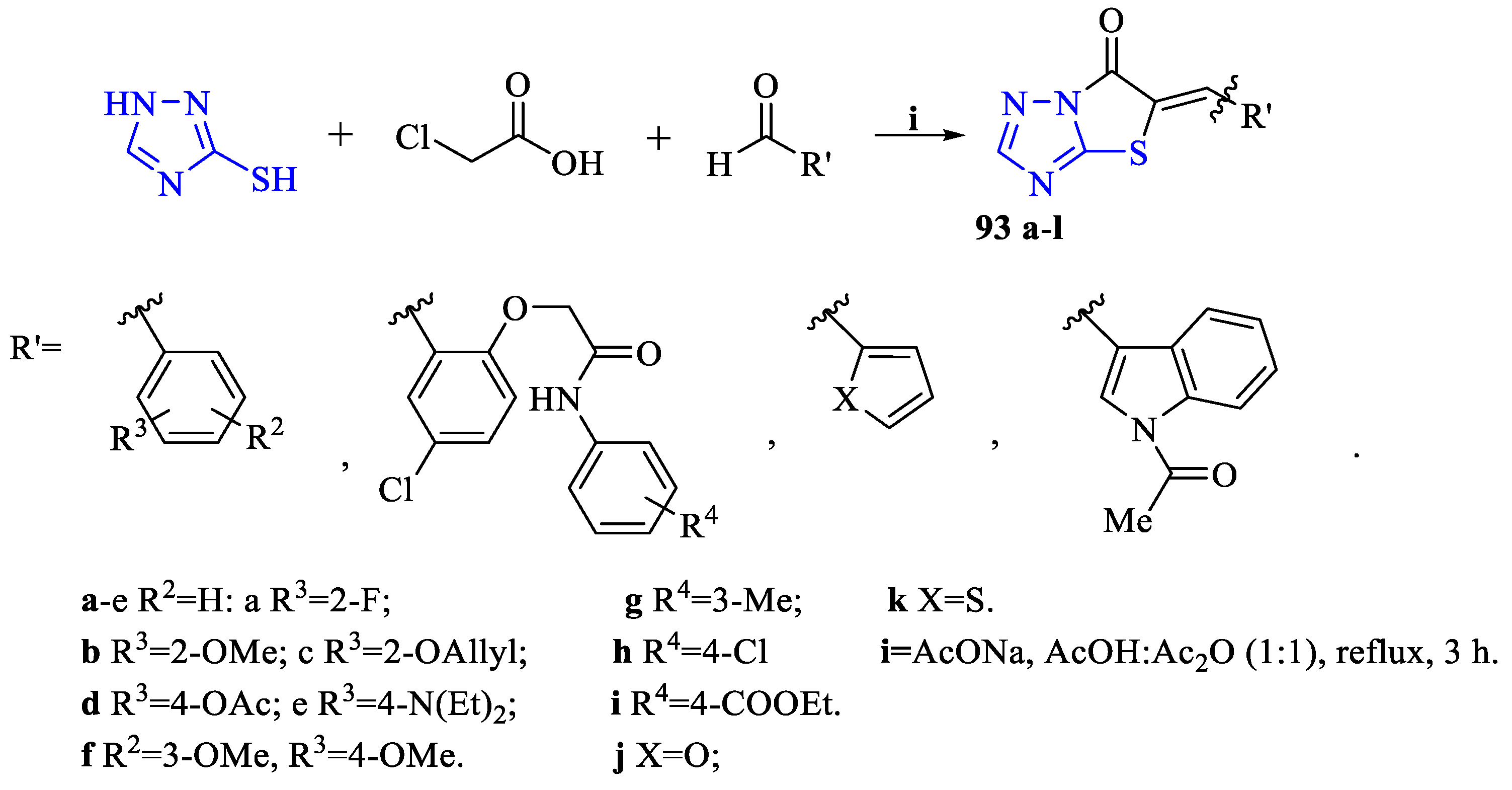
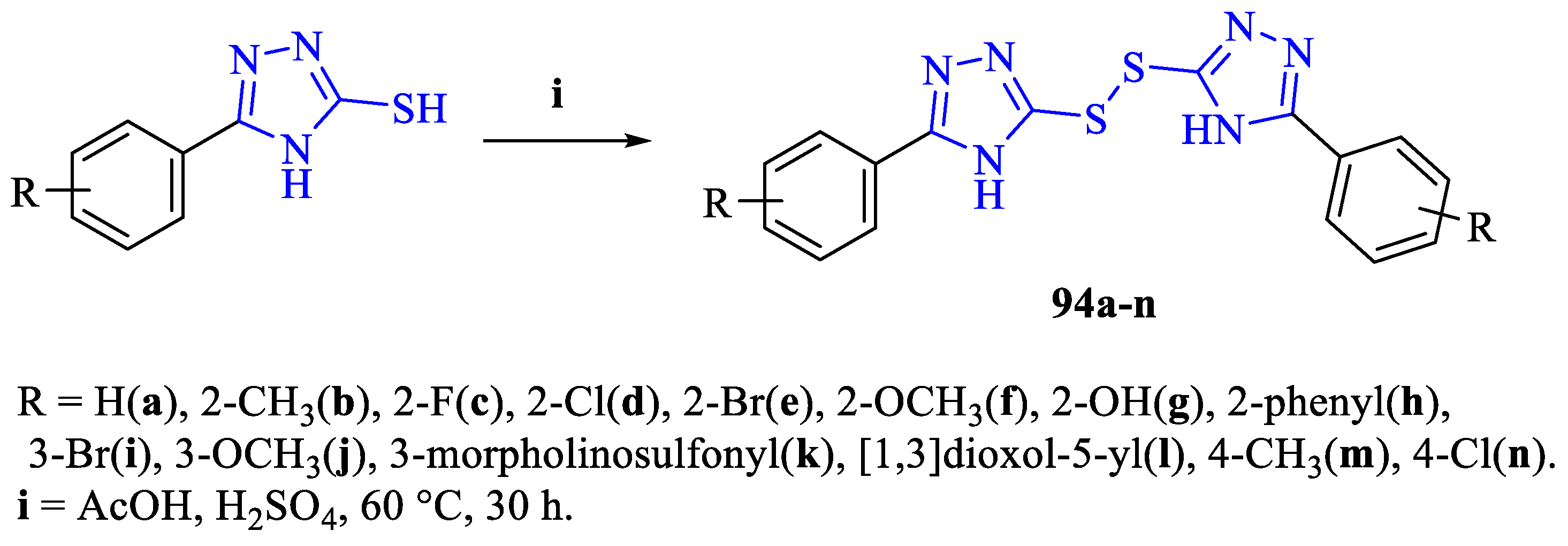
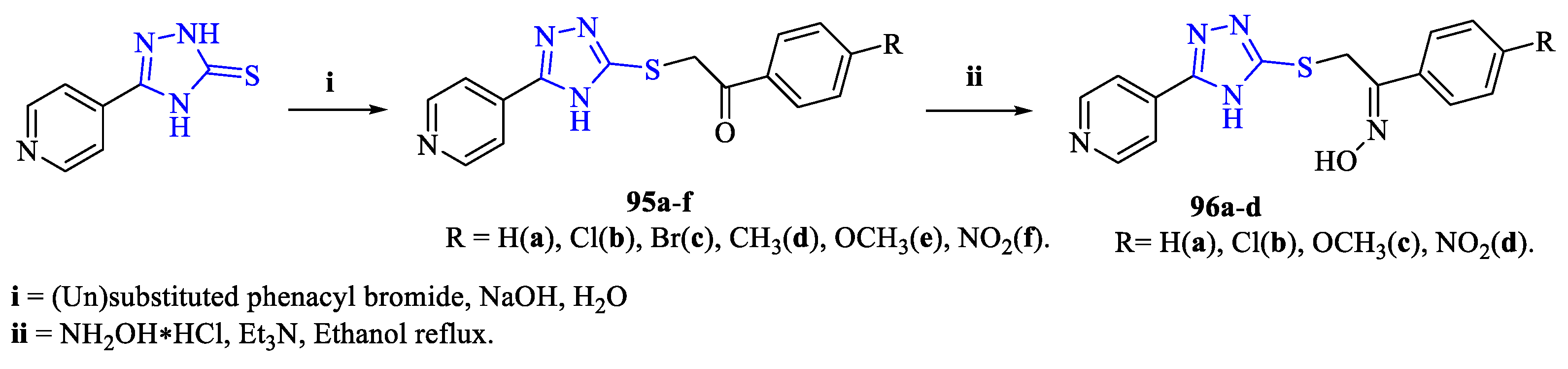
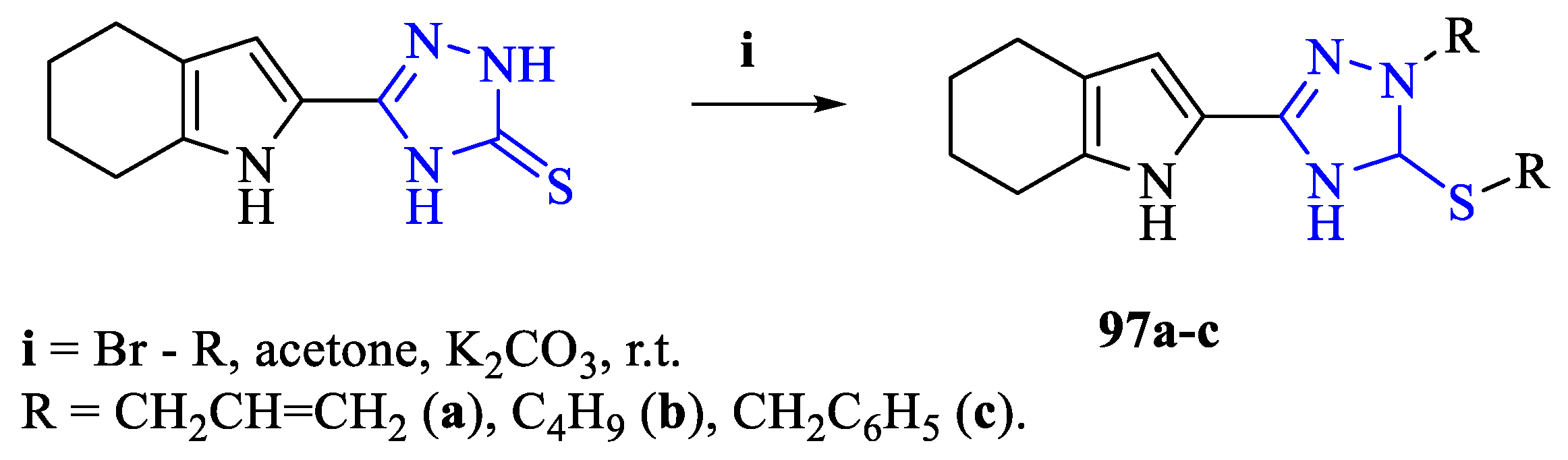
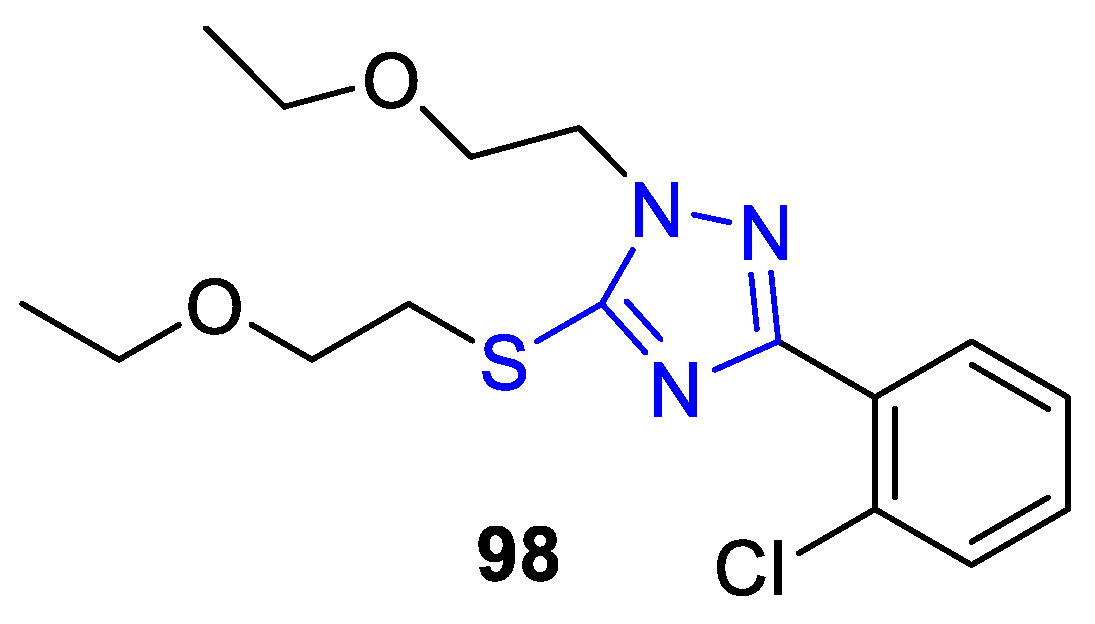
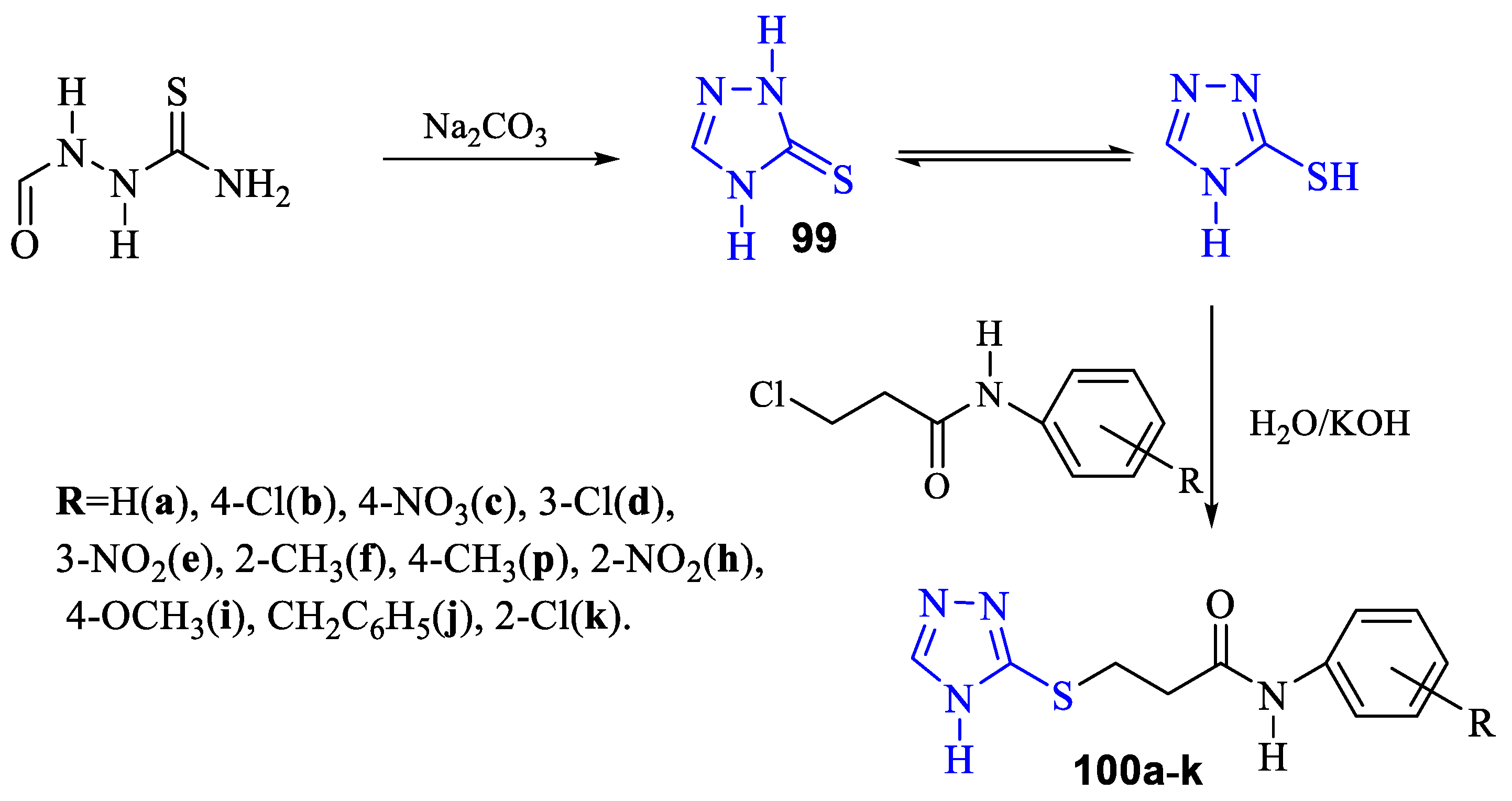
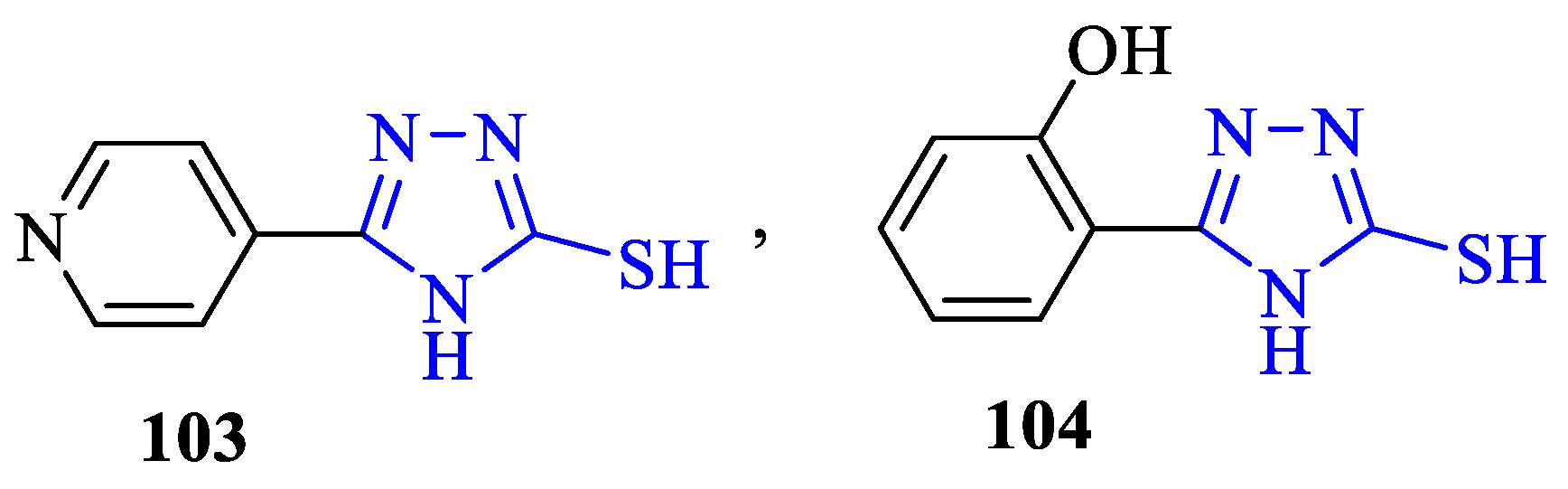
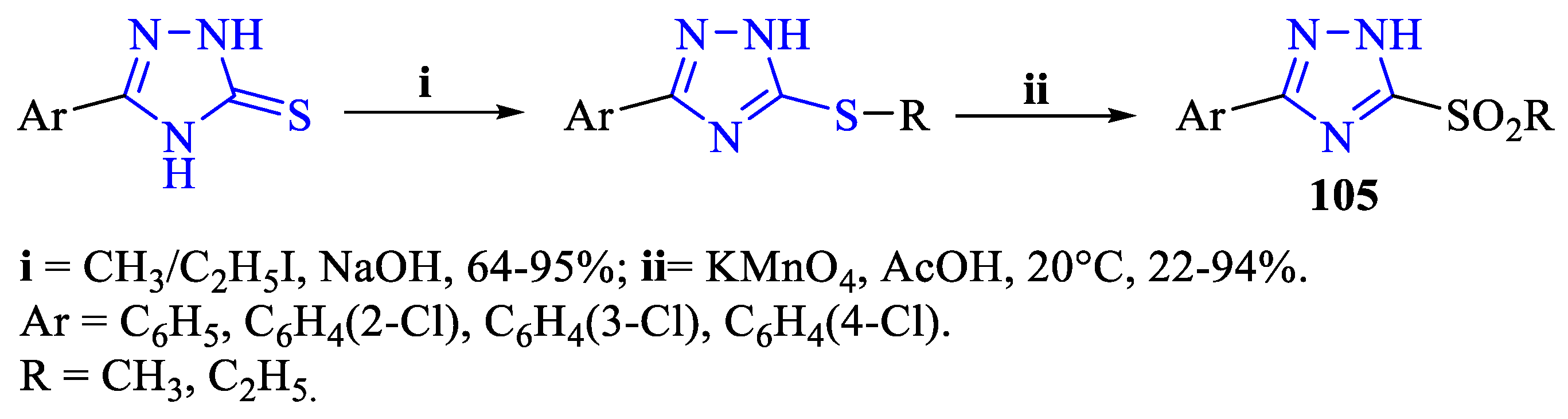
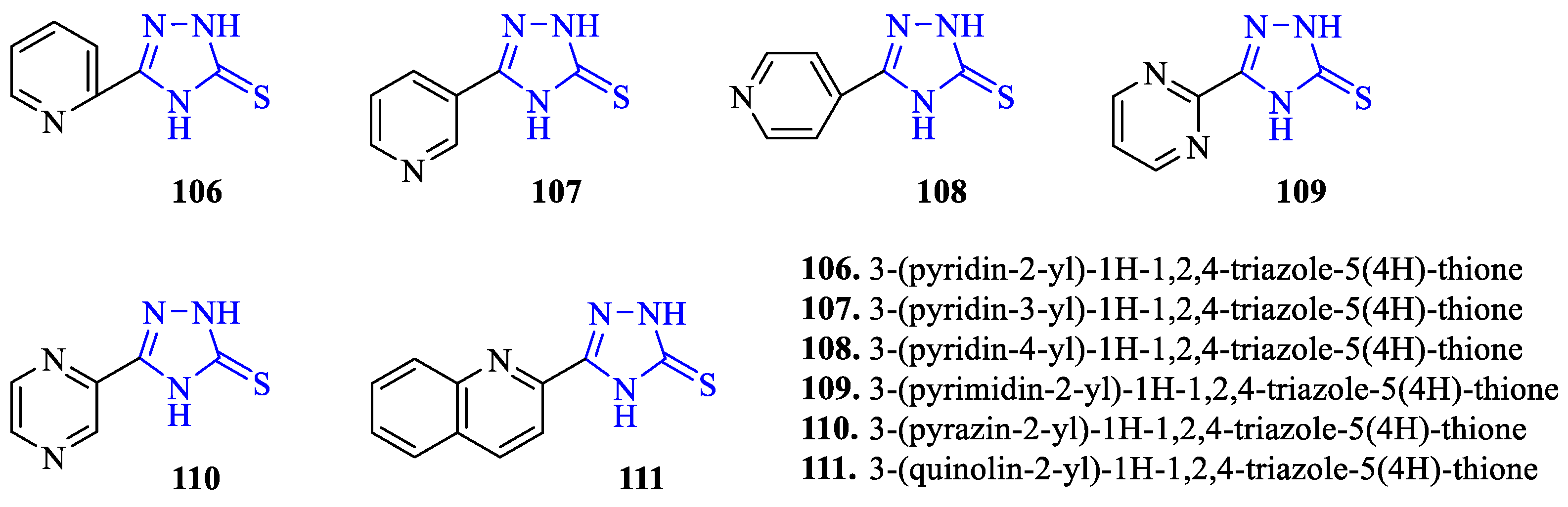
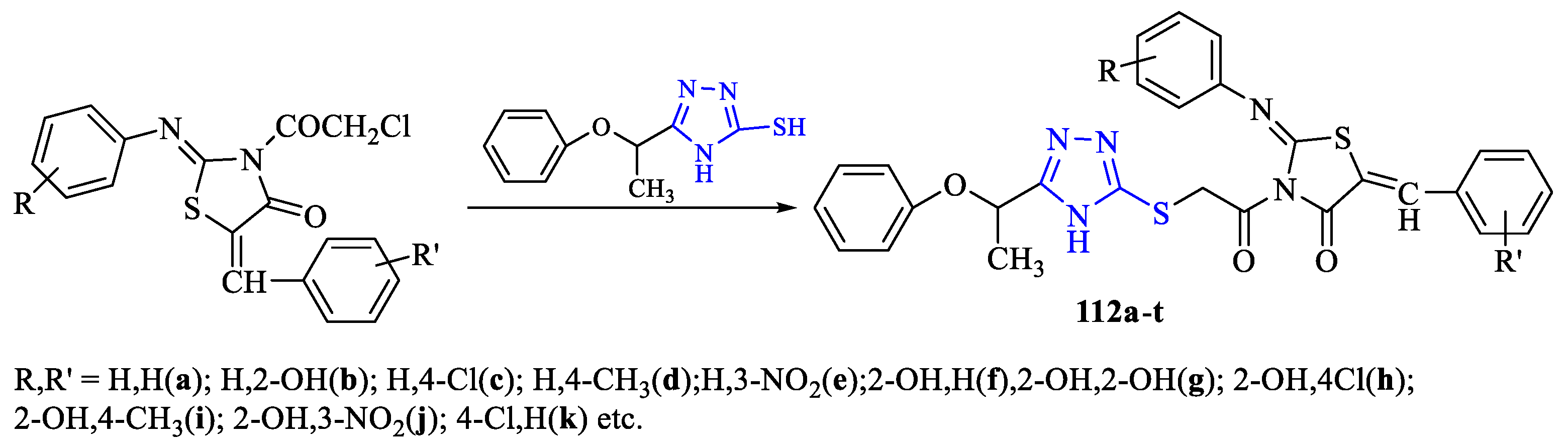
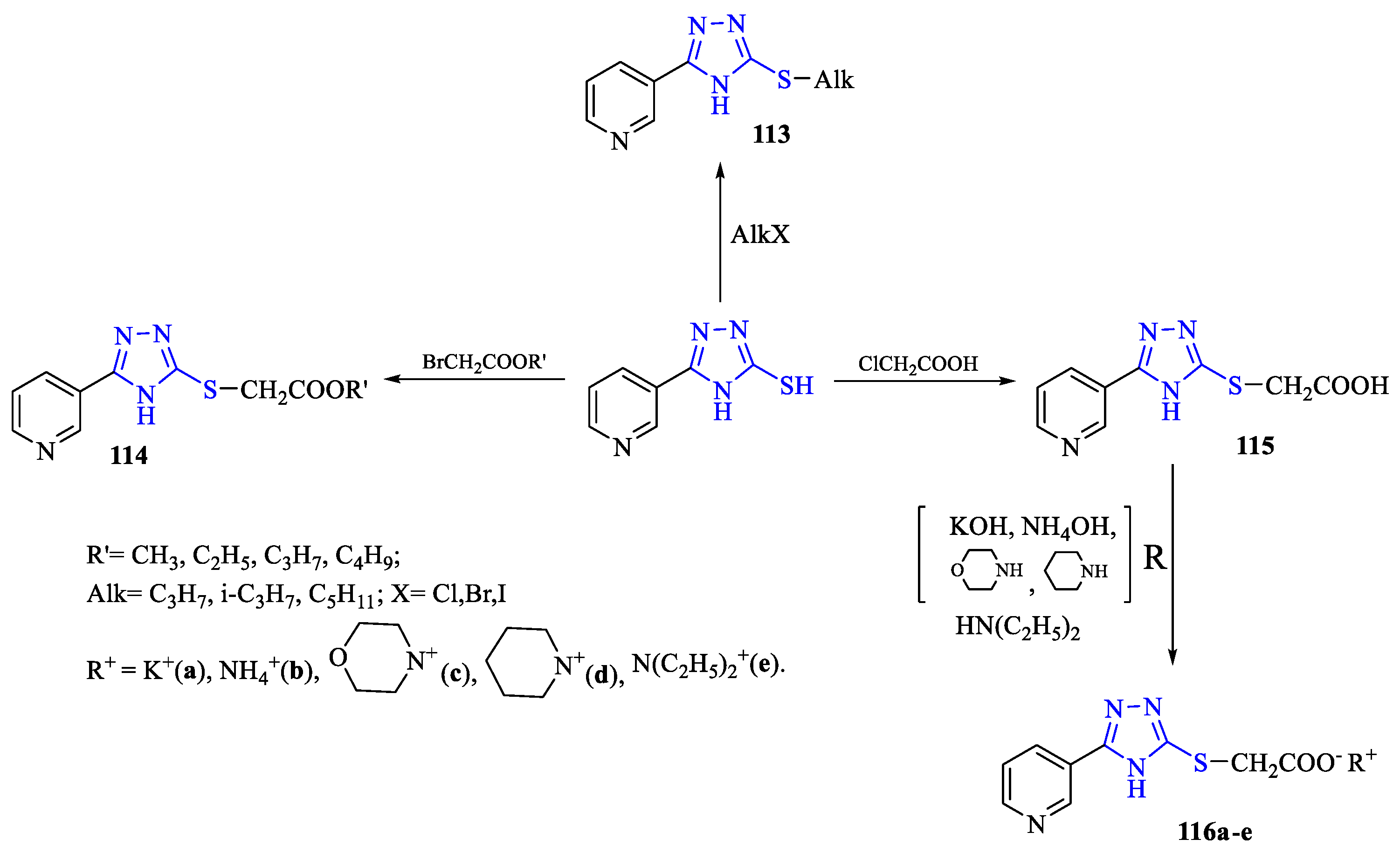
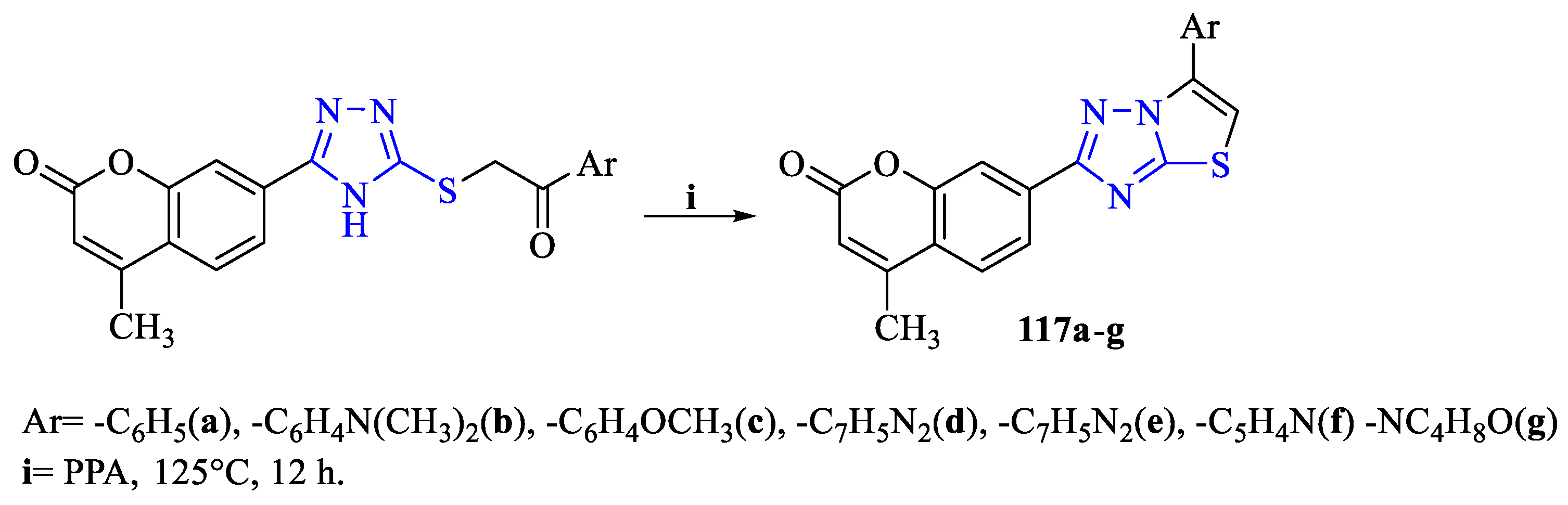
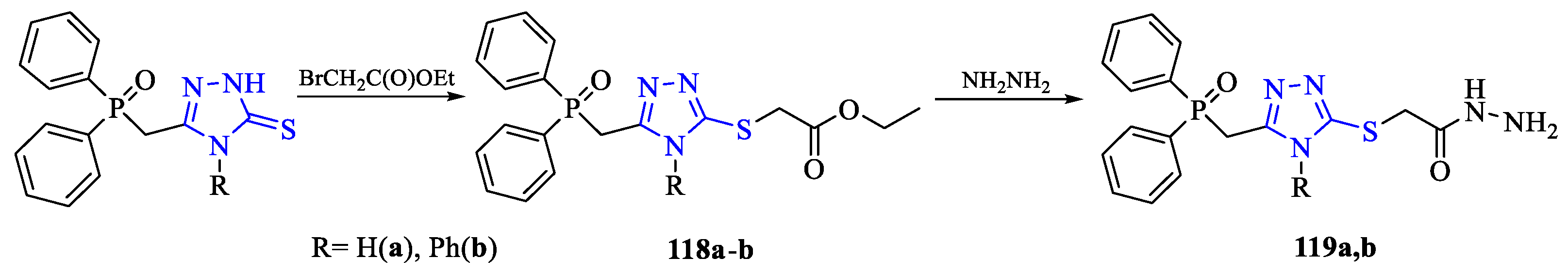
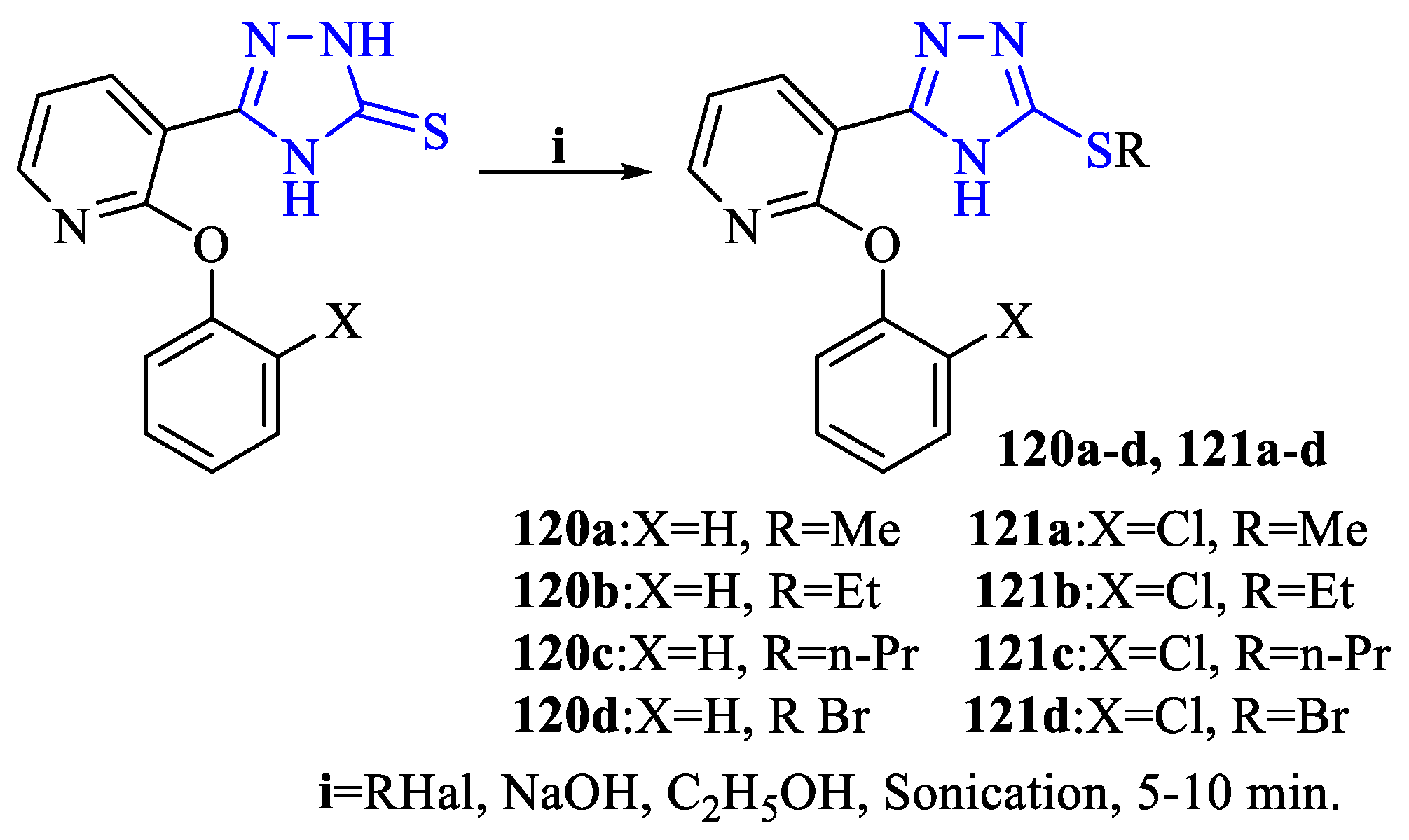
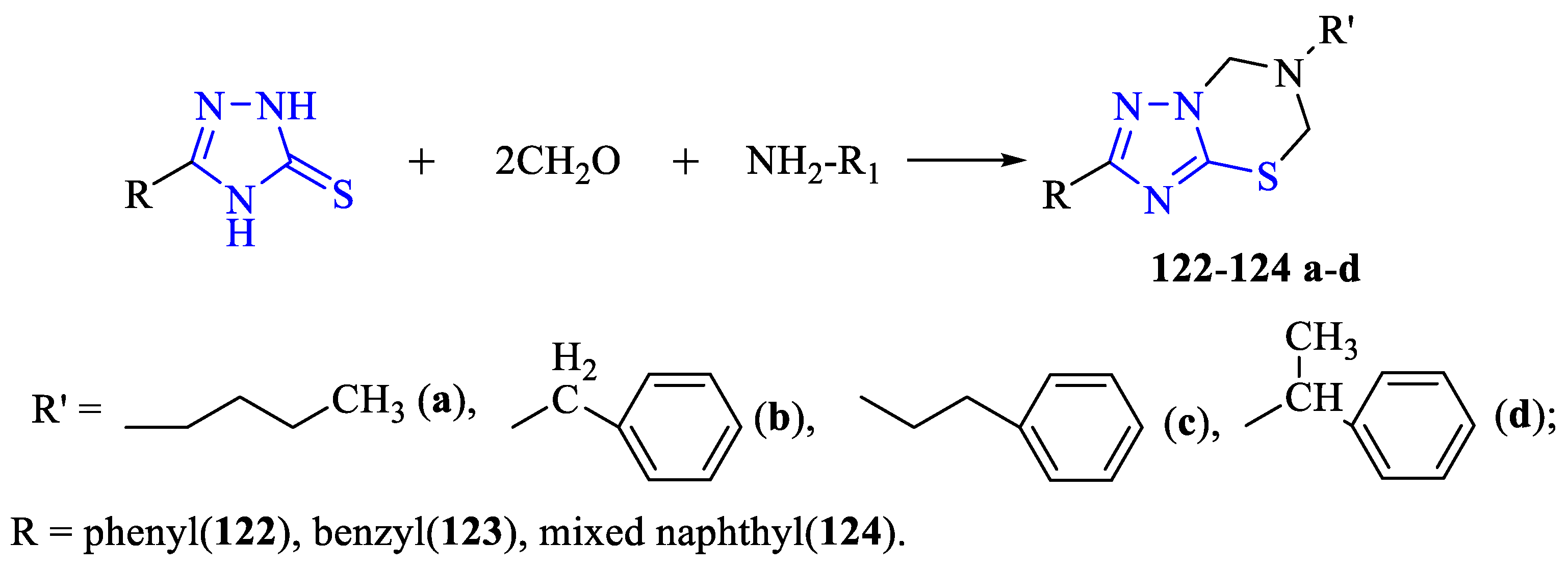
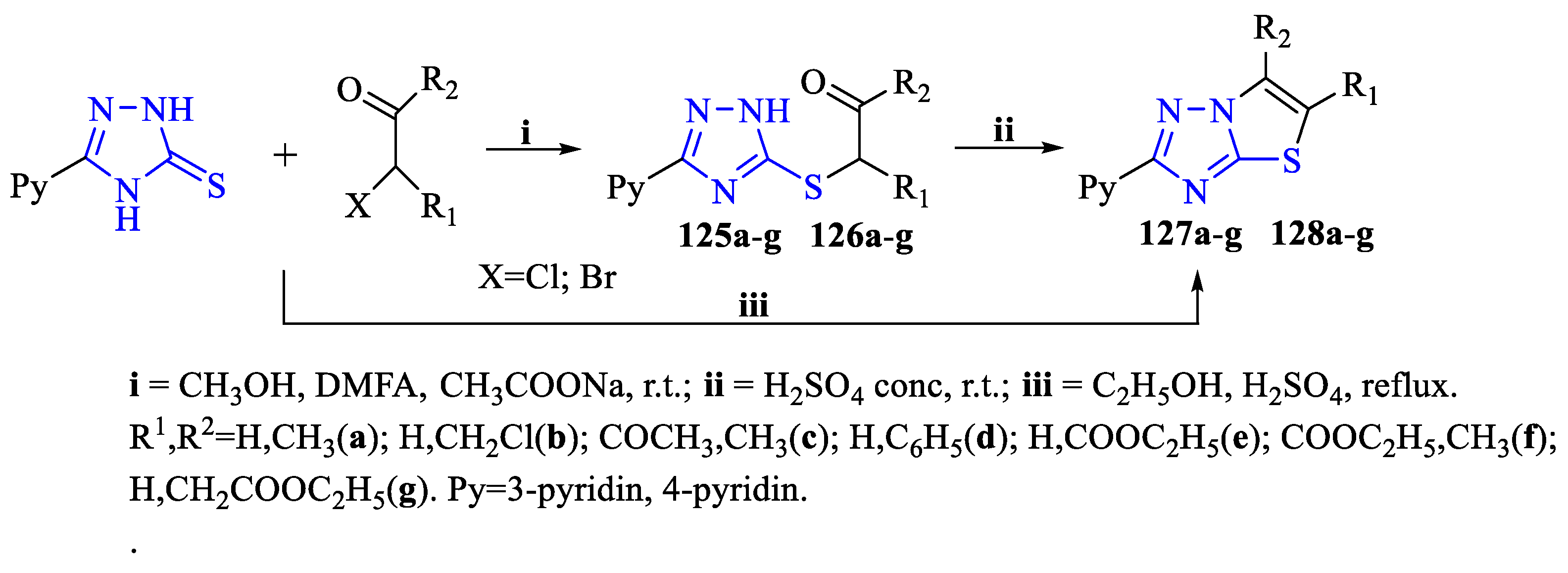
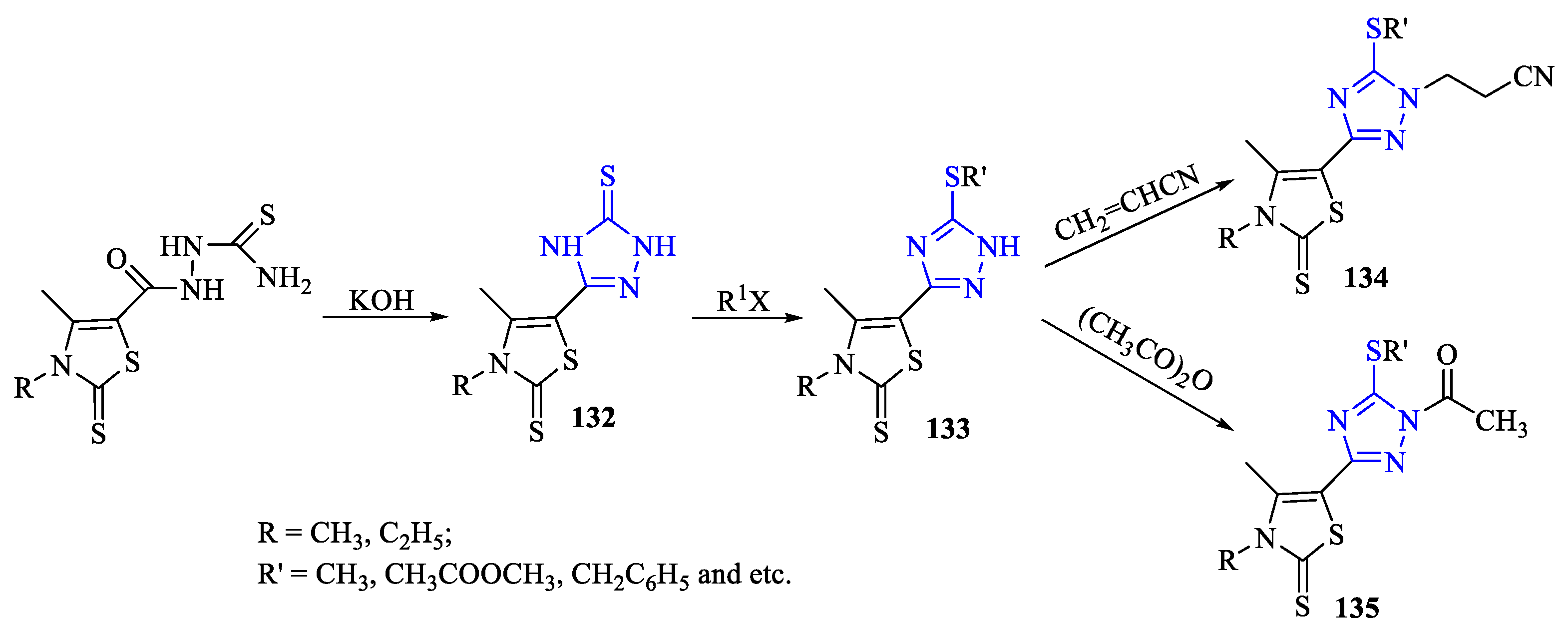
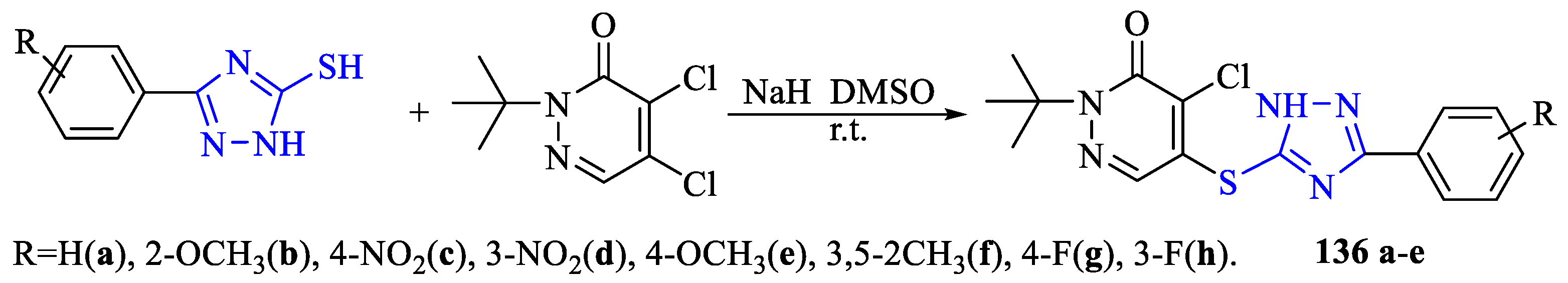
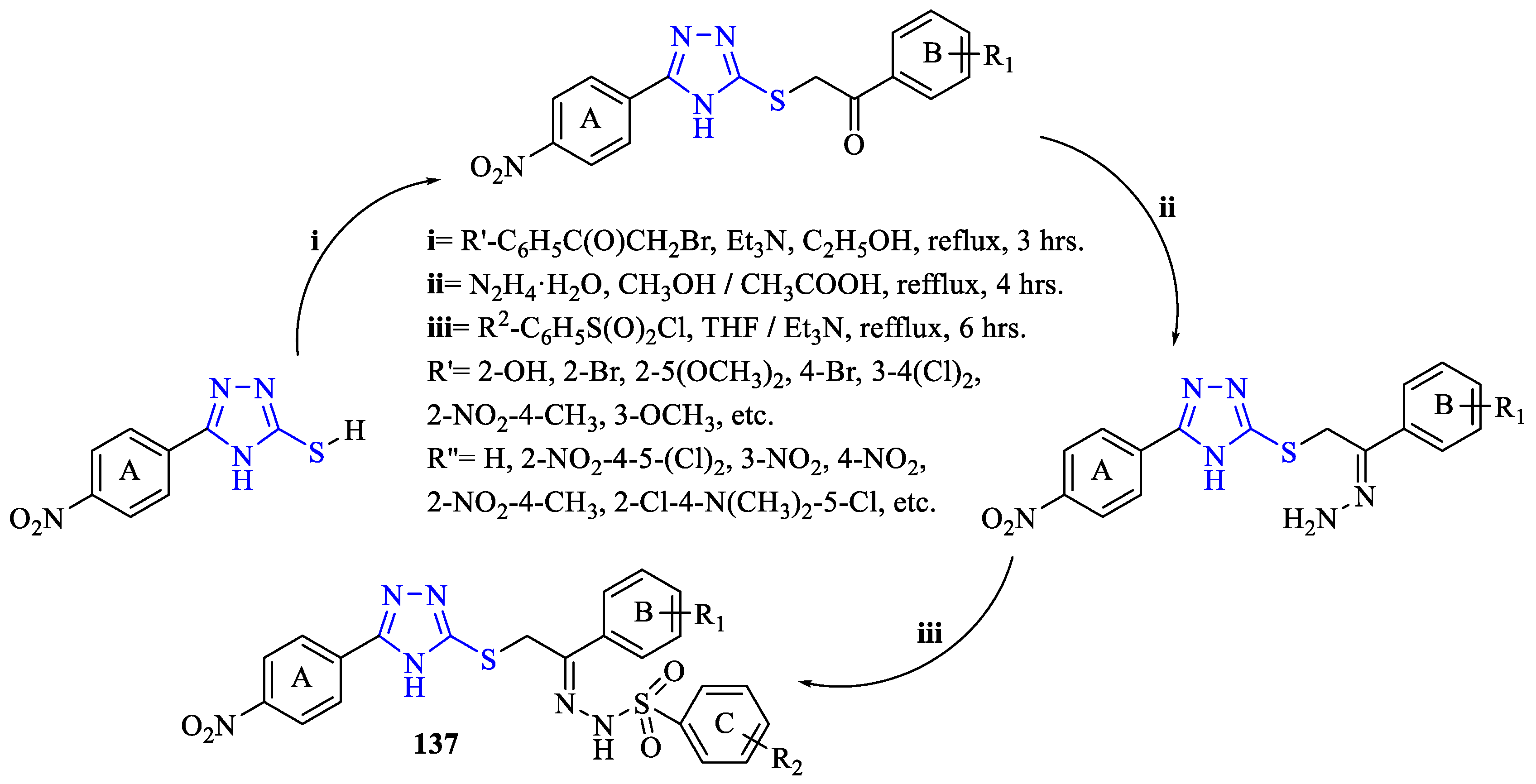
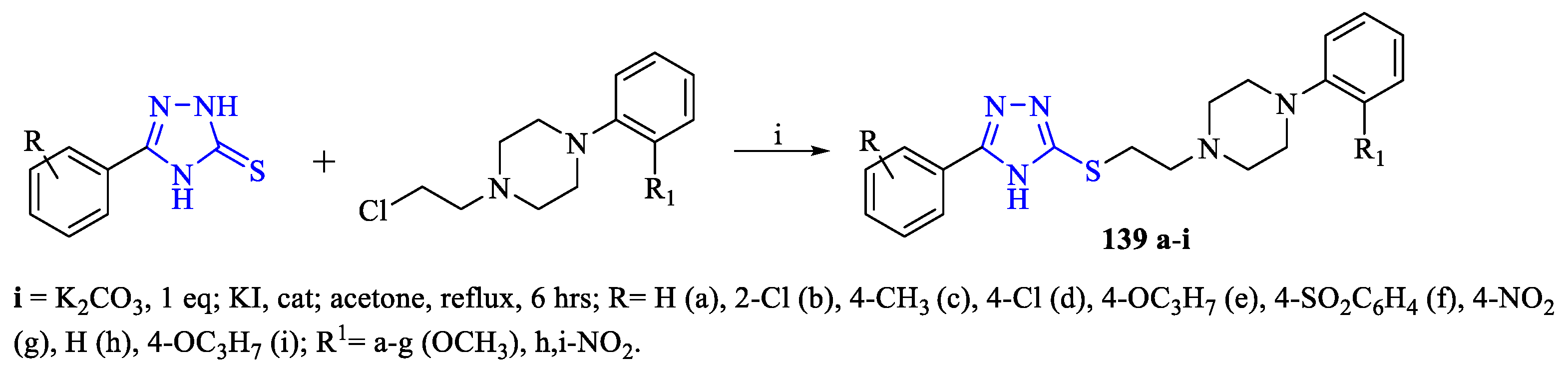
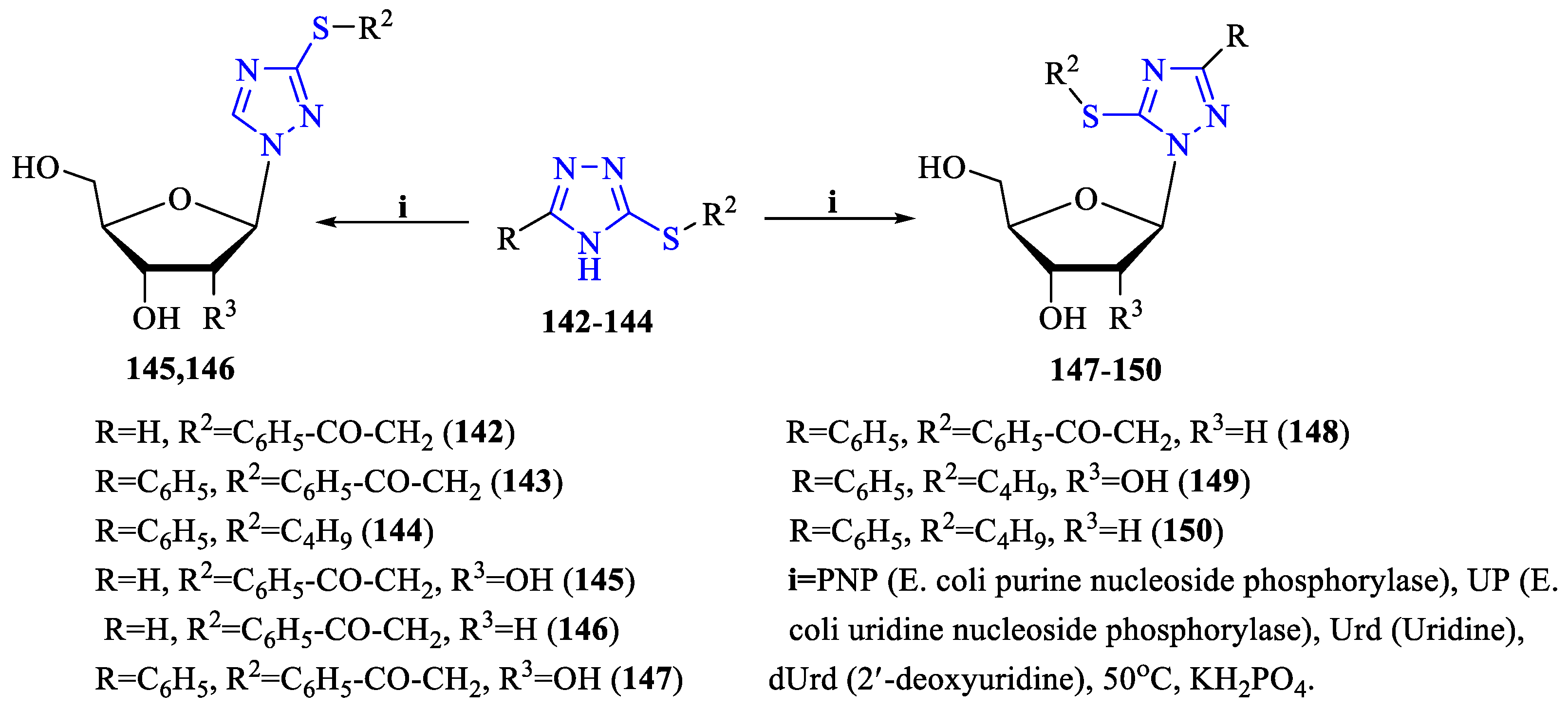
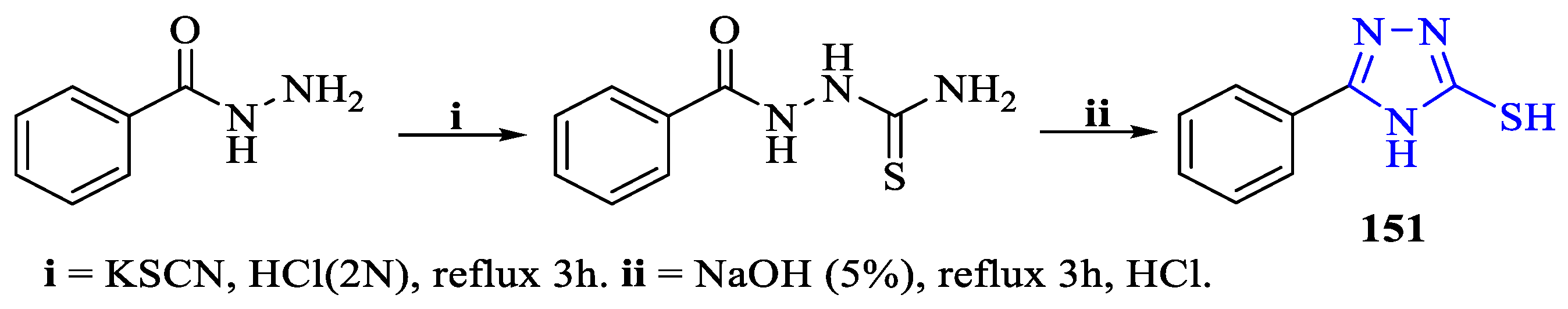
2. Synthesis of 5-substituted-2,4-dihydro-1,2,4-triazole-3-thiones
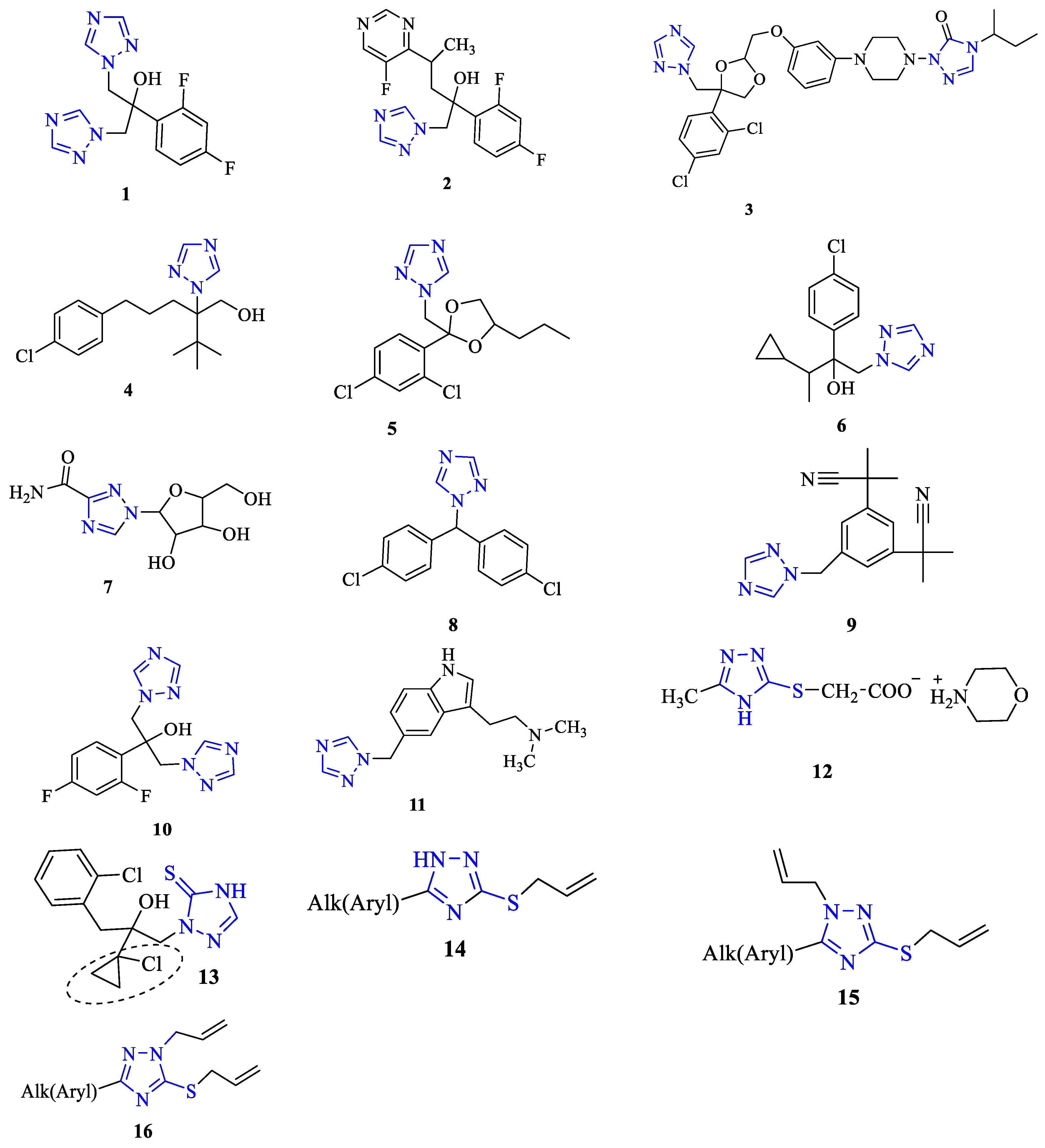
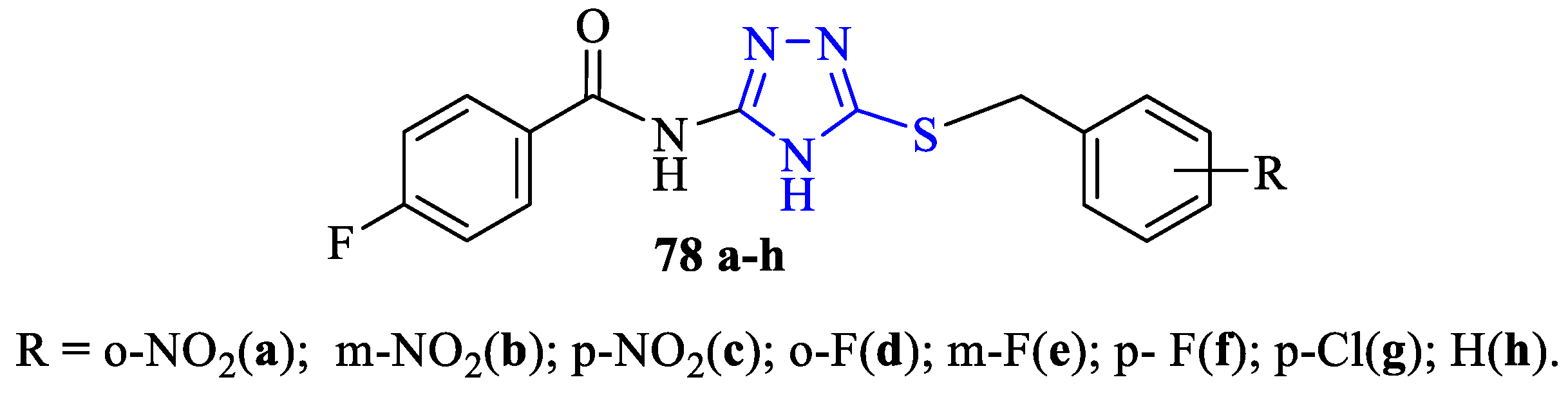
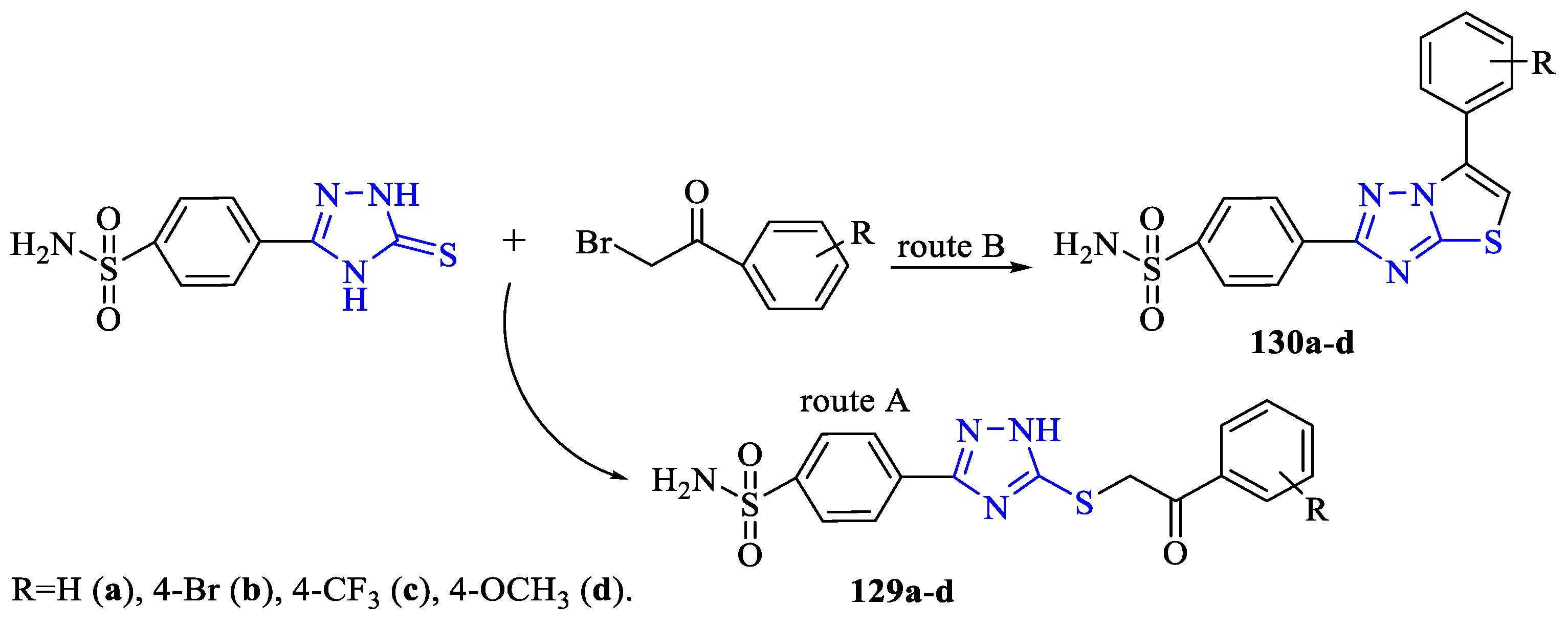
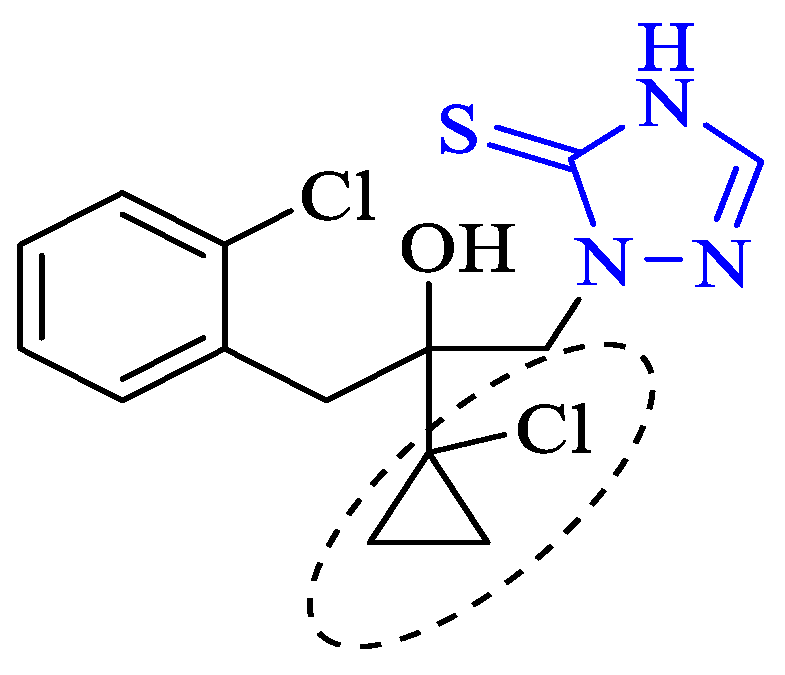
3. Antibacterial and Antifungal Activity
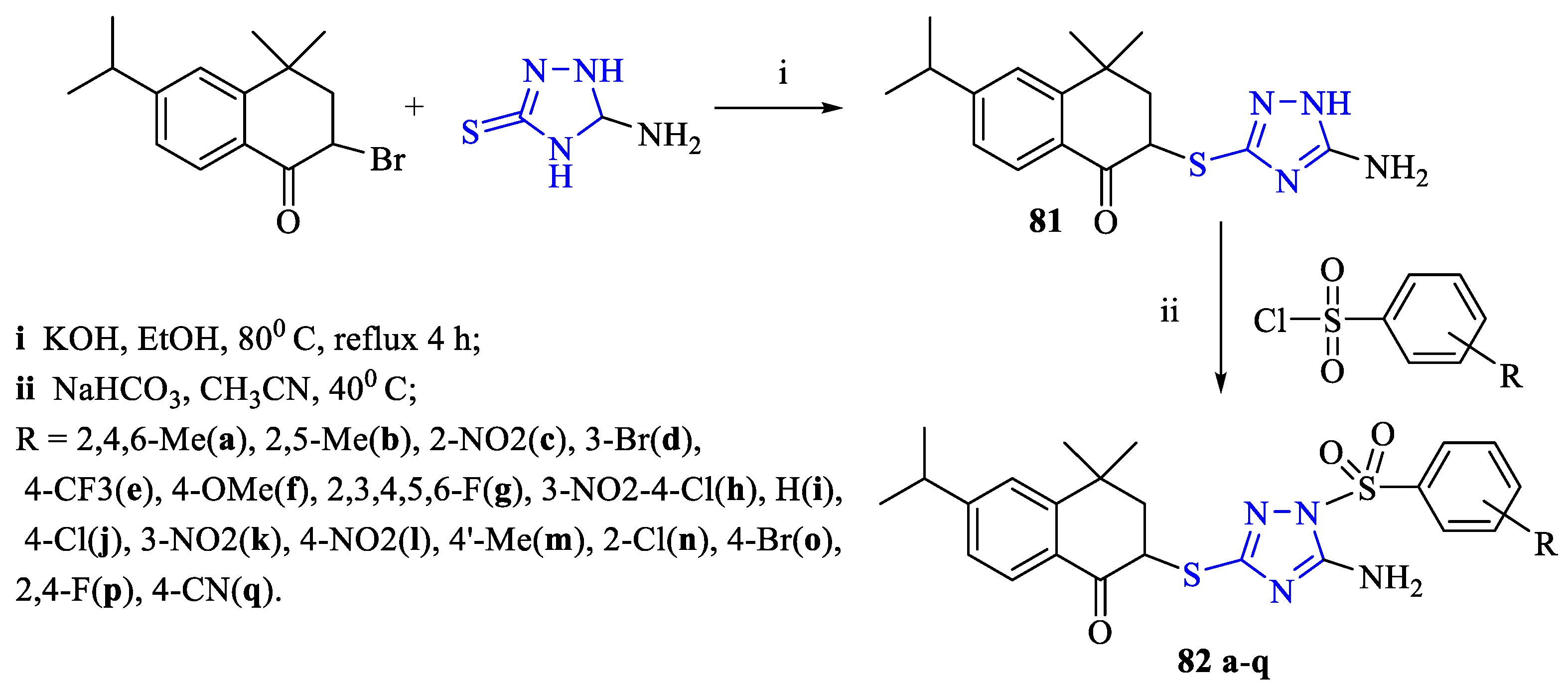
4. Cytotoxic Activity
5. Anti-Inflammatory and Analgesic Activities
6. Pesticidal Activity
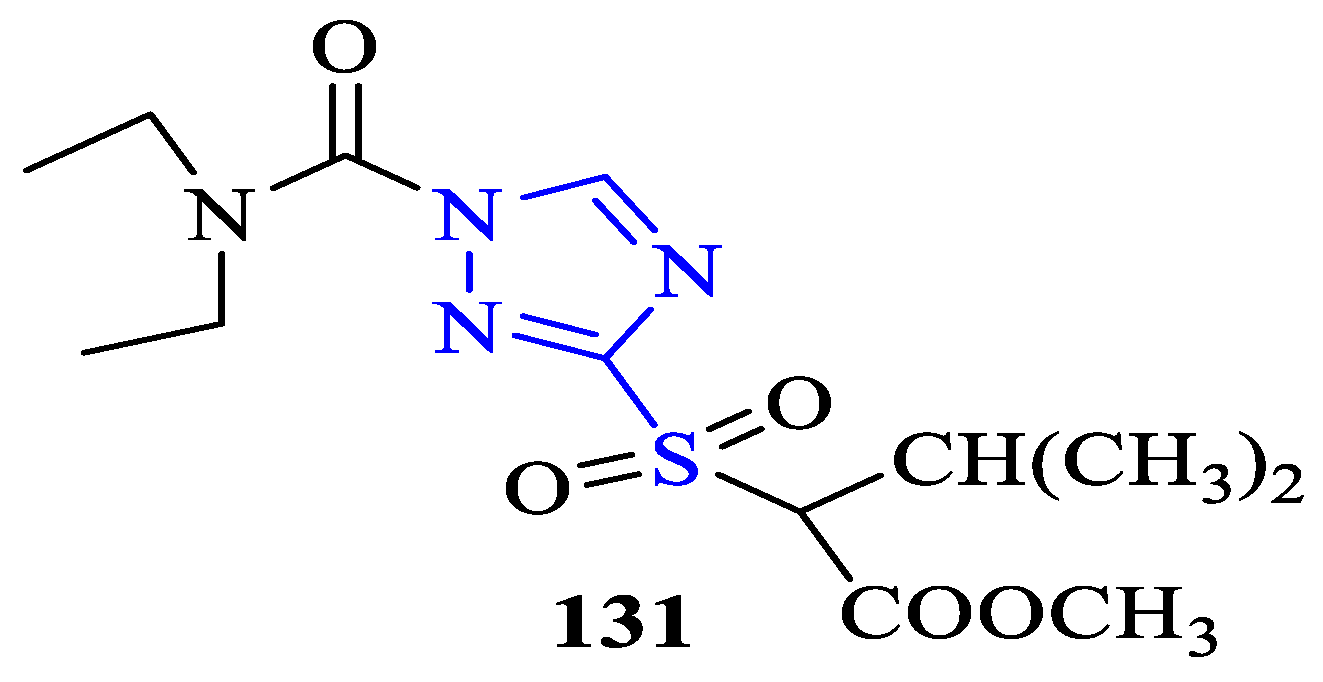
7. Other Types of Biological Activity
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Sharma, A.; Agrahari, A.K.; Rajkhowa, S.; Tiwari, V.K. Emerging Impact of Triazoles as Anti-Tubercular Agent. European Journal of Medicinal Chemistry 2022, 238, 114454. [Google Scholar] [CrossRef]
- Strzelecka, M.; Świątek, P. 1,2,4-Triazoles as Important Antibacterial Agents. Pharmaceuticals 2021, 14, 224. [Google Scholar] [CrossRef] [PubMed]
- Gao, F.; Wang, T.; Xiao, J.; Huang, G. Antibacterial Activity Study of 1,2,4-Triazole Derivatives. European Journal of Medicinal Chemistry 2019, 173, 274–281. [Google Scholar] [CrossRef]
- Abbas, S.; Zaib, S.; Ur Rahman, S.; Ali, S.; Hameed, S.; Tahir, M.N.; Munawar, K.S.; Shaheen, F.; Abbas, S.M.; Iqbal, J. Carbonic Anhydrase Inhibitory Potential of 1,2,4-Triazole-3-Thione Derivatives of Flurbiprofen, Ibuprofen and 4-Tert-Butylbenzoic Hydrazide: Design, Synthesis, Characterization, Biochemical Evaluation, Molecular Docking and Dynamic Simulation Studies. MC 2019, 15, 298–310. [Google Scholar] [CrossRef] [PubMed]
- Sicak, Y. Design and Antiproliferative and Antioxidant Activities of Furan-Based Thiosemicarbazides and 1,2,4-Triazoles: Their Structure-Activity Relationship and SwissADME Predictions. Med Chem Res 2021, 30, 1557–1568. [Google Scholar] [CrossRef]
- Zhang, Z.; Du, X.; Sheng, Q.; Shi, J.; Wang, J.; Chen, B. Method of Synthesizing 1,2,4-Triazole-3-Thione Compounds and Intermediates Thereof. U.S. Patent Application Pub. No: 2019/0127338 A1, 2 May 2019. [Google Scholar]
- Kaur, P.; Kaur, R.; Goswami, M. A REVIEW ON METHODS OF SYNTHESIS OF 1,2,4-TRIAZOLE DERIVATIVES. Int Res J Pharm 2018, 9, 1–35. [Google Scholar] [CrossRef]
- Ziyaev, A.; Terenteva, E.; Okmanov, R.; Sasmakov, S.; Toshmurodov, T.; Khamidova, U.; Umarova, M.; Azimova, S. Synthesis and Evaluation of Cytotoxic and Antimicrobial Activity of Some 3-Aryl-6-Phenyl-7H-[1,2,4]Triazolo [3,4-b][1,3,4]Thiadiazines. 10.5267/j.ccl 2024, 13, 549–556. [Google Scholar] [CrossRef]
- Sonawane, A.D.; Rode, N.D.; Nawale, L.; Joshi, R.R.; Joshi, R.A.; Likhite, A.P.; Sarkar, D. Synthesis and Biological Evaluation of 1,2,4-triazole-3-thione and 1,3,4-oxadiazole-2-thione as Antimycobacterial Agents. Chem Biol Drug Des 2017, 90, 200–209. [Google Scholar] [CrossRef]
- Othman, A.A.; Kihel, M.; Amara, S. 1,3,4-Oxadiazole, 1,3,4-Thiadiazole and 1,2,4-Triazole Derivatives as Potential Antibacterial Agents. Arabian Journal of Chemistry 2019, 12, 1660–1675. [Google Scholar] [CrossRef]
- Ghanaat, J.; Khalilzadeh, M.A.; Zareyee, D. Molecular Docking Studies, Biological Evaluation and Synthesis of Novel 3-Mercapto-1,2,4-Triazole Derivatives. Mol Divers 2021, 25, 223–232. [Google Scholar] [CrossRef]
- Ziyaev, A.; Sasmakov, S.; Okmanov, R.; Makhmudov, U.; Toshmurodov, T.; Ziyaeva, М.; Tosheva, N.; Azimova, S. Synthesis, Crystal Structure and Evaluation of the Cytotoxic, Antimicrobial Activity of Some S- and N-Derivatives of 5-Phenyl-1,2,4-Triazole-2,4-Dihydro-3-Thione. Chemical Data Collections 2025, 56, 101182. [Google Scholar] [CrossRef]
- Emami, L.; Sadeghian, S.; Mojaddami, A.; Khabnadideh, S.; Sakhteman, A.; Sadeghpour, H.; Faghih, Z.; Fereidoonnezhad, M.; Rezaei, Z. Design, Synthesis and Evaluation of Novel 1,2,4-Triazole Derivatives as Promising Anticancer Agents. BMC Chemistry 2022, 16, 91. [Google Scholar] [CrossRef] [PubMed]
- Glomb, T.; Minta, J.; Nowosadko, M.; Radzikowska, J.; Świątek, P. Search for New Compounds with Anti-Inflammatory Activity Among 1,2,4-Triazole Derivatives. Molecules 2024, 29, 6036. [Google Scholar] [CrossRef] [PubMed]
- El-Sebaey, S.A. Recent Advances in 1,2,4-Triazole Scaffolds as Antiviral Agents. ChemistrySelect 2020, 5, 11654–11680. [Google Scholar] [CrossRef]
- Elzoheiry, M.A.; Elmehankar, M.S.; Aboukamar, W.A.; El-Gamal, R.; Sheta, H.; Zenezan, D.; Nabih, N.; Elhenawy, A.A. Fluconazole as Schistosoma Mansoni Cytochrome P450 Inhibitor: In Vivo Murine Experimental Study. Experimental Parasitology 2022, 239, 108291. [Google Scholar] [CrossRef]
- Gao, D.X.; Song, S.; Kahn, J.S.; Cohen, S.R.; Fiumara, K.; Dumont, N.; Rosmarin, D. Treatment of Patients Experiencing Dupilumab Facial Redness with Itraconazole and Fluconazole: A Single-Institutional, Retrospective Medical Record Review. Journal of the American Academy of Dermatology 2022, 86, 938–940. [Google Scholar] [CrossRef]
- Shettar, A.; Shankar, V.K.; Ajjarapu, S.; Kulkarni, V.I.; Repka, M.A.; Murthy, S.N. Development and Characterization of Novel Topical Oil/PEG Creams of Voriconazole for the Treatment of Fungal Infections. Journal of Drug Delivery Science and Technology 2021, 66, 102928. [Google Scholar] [CrossRef]
- Navarro-Triviño, F.J. Leishmaniasis cutánea tratada con itraconazol oral. Piel 2021, 36, 563–565. [Google Scholar] [CrossRef]
- Abbas, A.A.; Dawood, K.M. Recent Developments in the Chemistry of 1H- and 4H-1,2,4-Triazoles. In Advances in Heterocyclic Chemistry; Elsevier, 2023; Volume 141, pp. 209–273. ISBN 978-0-443-19318-7. [Google Scholar]
- Burman, B.; Drutman, S.B.; Fury, M.G.; Wong, R.J.; Katabi, N.; Ho, A.L.; Pfister, D.G. Pharmacodynamic and Therapeutic Pilot Studies of Single-Agent Ribavirin in Patients with Human Papillomavirus–Related Malignancies. Oral Oncology 2022, 128, 105806. [Google Scholar] [CrossRef]
- Tian, Y.; Yang, W.; Yang, R.; Zhang, Q.; Hao, L.; Bian, E.; Yang, Y.; Huang, X.; Wu, Y.; Zhang, B. Ribavirin Inhibits the Growth and Ascites Formation of Hepatocellular Carcinoma through Downregulation of Type I CARM1 and Type II PRMT5. Toxicology and Applied Pharmacology 2022, 435, 115829. [Google Scholar] [CrossRef]
- Park, H.G.; Kim, J.H.; Dancer, A.N.; Kothapalli, K.S.; Brenna, J.T. The Aromatase Inhibitor Letrozole Restores FADS2 Function in ER+ MCF7 Human Breast Cancer Cells. Prostaglandins, Leukotrienes and Essential Fatty Acids 2021, 171, 102312. [Google Scholar] [CrossRef] [PubMed]
- Slomovitz, B.M.; Filiaci, V.L.; Walker, J.L.; Taub, M.C.; Finkelstein, K.A.; Moroney, J.W.; Fleury, A.C.; Muller, C.Y.; Holman, L.L.; Copeland, L.J.; et al. A Randomized Phase II Trial of Everolimus and Letrozole or Hormonal Therapy in Women with Advanced, Persistent or Recurrent Endometrial Carcinoma: A GOG Foundation Study. Gynecologic Oncology 2022, 164, 481–491. [Google Scholar] [CrossRef]
- Küçükgüzel, Ş.G.; Çıkla-Süzgün, P. Recent Advances Bioactive 1,2,4-Triazole-3-Thiones. European Journal of Medicinal Chemistry 2015, 97, 830–870. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Yamagishi, G.; Hiramatsu, T.; Hosoya, A.; Onoe, K.; Doi, H.; Nagata, H.; Wada, Y.; Onoe, H.; Watanabe, Y.; et al. Practical Synthesis of Precursor of [N-Methyl-11C]Vorozole, an Efficient PET Tracer Targeting Aromatase in the Brain. Bioorganic & Medicinal Chemistry 2011, 19, 1464–1470. [Google Scholar] [CrossRef]
- Chokshi, A.; Vaishya, R.; Inavolu, R.; Potta, T. Intranasal Spray Formulation Containing Rizatriptan Benzoate for the Treatment of Migraine. International Journal of Pharmaceutics 2019, 571, 118702. [Google Scholar] [CrossRef] [PubMed]
- Kastanayan, A.A.; Kartashova, E.A.; Zheleznyak, E.I. THE EFFECT OF THIOTRIAZOLINE ON ENERGY PRODUCTION IN CONDITIONS OF CHRONIC MYOCARDIAL ISCHEMIA. jour 2020, 1, 84–90. [Google Scholar] [CrossRef]
- Jeschke, P. Progress of Modern Agricultural Chemistry and Future Prospects: Progress of Modern Agricultural Chemistry and Future Prospects. Pest. Manag. Sci. 2016, 72, 433–455. [Google Scholar] [CrossRef]
- Sarkar, D.; Deshpande, S.R.; Maybhate, S.P.; Likhite, A.P.; Sarkar, S.; Khan, A.; Chaudhry, P.M.; Chavan, S.R. 1,2,4-Triazole derivatives and their antimycobacterial activity. World Intellectual Property Organization (WIPO) Patent. WO2011111077A1, 15 September 2011. [Google Scholar]
- Sadeghian, S.; Emami, L.; Mojaddami, A.; Khabnadideh, S.; Faghih, Z.; Zomorodian, K.; Rashidi, M.; Rezaei, Z. 1,2,4-Triazole Derivatives as Novel and Potent Antifungal Agents: Design, Synthesis and Biological Evaluation. Journal of Molecular Structure 2023, 1271, 134039. [Google Scholar] [CrossRef]
- Gupta, O.; Pradhan, T.; Chawla, G. An Updated Review on Diverse Range of Biological Activities of 1,2,4-Triazole Derivatives: Insight into Structure Activity Relationship. Journal of Molecular Structure 2023, 1274, 134487. [Google Scholar] [CrossRef]
- Wen, X.; Zhou, Y.; Zeng, J.; Liu, X. Recent Development of 1,2,4-Triazole-Containing Compounds as Anticancer Agents. CTMC 2020, 20, 1441–1460. [Google Scholar] [CrossRef]
- Vagish, C.B.; Sudeep, P.; Jayadevappa, H.P.; Ajay Kumar, K. 1,2,4-Triazoles: Synthetic and Medicinal Perspectives. Int. J. Curr. Res. 2020, 12, 12950–12960. [Google Scholar]
- Mostafa, M.S.; Radini, I.A.M.; El-Rahman, N.M.A.; Khidre, R.E. Synthetic Methods and Pharmacological Potentials of Triazolothiadiazines: A Review. Molecules 2024, 29, 1326. [Google Scholar] [CrossRef] [PubMed]
- Khramchikhin, A.V.; Skryl’nikova, M.A.; Esaulkova, I.L.; Sinegubova, E.O.; Zarubaev, V.V.; Gureev, M.A.; Puzyk, A.M.; Ostrovskii, V.A. Novel [1,2,4]Triazolo [3,4-b][1,3,4]Thiadiazine and [1,2,4]Triazolo [3,4-b][1,3,4]Thiadiazepine Derivatives: Synthesis, Anti-Viral In Vitro Study and Target Validation Activity. Molecules 2022, 27, 7940. [Google Scholar] [CrossRef]
- Bersani, M.; Failla, M.; Vascon, F.; Gianquinto, E.; Bertarini, L.; Baroni, M.; Cruciani, G.; Verdirosa, F.; Sannio, F.; Docquier, J.-D.; et al. Structure-Based Optimization of 1,2,4-Triazole-3-Thione Derivatives: Improving Inhibition of NDM-/VIM-Type Metallo-β-Lactamases and Synergistic Activity on Resistant Bacteria. Pharmaceuticals 2023, 16, 1682. [Google Scholar] [CrossRef]
- Elwahy, A.H.M.; Ginidi, A.R.S.; Shaaban, M.R.; Mohamed, A.H.; Gaber, H.M.; Ibrahim, L.I.; Farag, A.M.; Salem, M.E. Novel Bis([Triazolo [3,4-b]Thiadiazoles and Bis([Triazolo [3,4-b][Thiadiazines) with Antioxidant Activity. Arkivoc 2024, 2024. [Google Scholar] [CrossRef]
- Abdelli, A.; Azzouni, S.; Plais, R.; Gaucher, A.; Efrit, M.L.; Prim, D. Recent Advances in the Chemistry of 1,2,4-Triazoles: Synthesis, Reactivity and Biological Activities. Tetrahedron Letters 2021, 86, 153518. [Google Scholar] [CrossRef]
- Kumar, S.; Khokra, S.L.; Yadav, A. Triazole Analogues as Potential Pharmacological Agents: A Brief Review. Futur J Pharm Sci 2021, 7, 106. [Google Scholar] [CrossRef] [PubMed]
- Legru, A.; Verdirosa, F.; Vo-Hoang, Y.; Tassone, G.; Vascon, F.; Thomas, C.A.; Sannio, F.; Corsica, G.; Benvenuti, M.; Feller, G.; et al. Optimization of 1,2,4-Triazole-3-Thiones toward Broad-Spectrum Metallo-β-Lactamase Inhibitors Showing Potent Synergistic Activity on VIM- and NDM-1-Producing Clinical Isolates. J. Med. Chem. 2022, 65, 16392–16419. [Google Scholar] [CrossRef]
- Jat, L.R.; Sharma, Dr.V.; Agarwal, R. A Review on Synthesis and Biological Activity of 1,2,4-Triazole Derivatives. Int J Pharm Sci Rev Res 2023, 79. [Google Scholar] [CrossRef]
- Raman, A.P.S.; Aslam, M.; Awasthi, A.; Ansari, A.; Jain, P.; Lal, K.; Bahadur, I.; Singh, P.; Kumari, K. An Updated Review on 1,2,3-/1,2,4-Triazoles: Synthesis and Diverse Range of Biological Potential. Mol Divers 2025, 29, 899–964. [Google Scholar] [CrossRef]
- Raafat, C.; Ali, T.; Abuo-Rahma, G. Recent Advances in Pharmacologically Important 1,2,4-Triazoles as Promising Antifungal Agents against Candida Albicans. Octahedron Drug Research 2023, 0, 11–33. [Google Scholar] [CrossRef]
- Shaker, R.M. The Chemistry of Mercapto- and Thione-Substituted 1,2,4-Triazoles and Their Utility in Heterocyclic Synthesis. ChemInform 2007, 38, chin.200721253. [Google Scholar] [CrossRef]
- Korol, N.I.; Slivka, M.V. Recent Progress in the Synthesis of Thiazolo [3,2-b][1,2,4]Triazoles (Microreview). Chem Heterocycl Comp 2017, 53, 852–854. [Google Scholar] [CrossRef]
- Aly, A.A.; A. Hassan, A.; Makhlouf, M.M.; Bräse, S. Chemistry and Biological Activities of 1,2,4-Triazolethiones—Antiviral and Anti-Infective Drugs. Molecules 2020, 25, 3036. [Google Scholar] [CrossRef] [PubMed]
- Dai, J.; Tian, S.; Yang, X.; Liu, Z. Synthesis Methods of 1,2,3-/1,2,4-Triazoles: A Review. Front. Chem. 2022, 10, 891484. [Google Scholar] [CrossRef]
- Shi, Y.-J.; Song, X.-J.; Li, X.; Ye, T.-H.; Xiong, Y.; Yu, L.-T. Synthesis and Biological Evaluation of 1,2,4-Triazole and 1,3,4-Thiadiazole Derivatives as Potential Cytotoxic Agents. Chem. Pharm. Bull. 2013, 61, 1099–1104. [Google Scholar] [CrossRef]
- Hoggarth, E. 251. Compounds Related to Thiosemicarbazide. Part II. 1-Benzoylthiosemicarbazides. J. Chem. Soc. 1949, 1163. [Google Scholar] [CrossRef]
- Mane, M.M.; Pore, D.M. A Novel One Pot Multi-Component Strategy for Facile Synthesis of 5-Aryl-[1,2,4]Triazolidine-3-Thiones. Tetrahedron Letters 2014, 55, 6601–6604. [Google Scholar] [CrossRef]
- Ramesh, R.; Lalitha, A. PEG-Assisted Two-Component Approach for the Facile Synthesis of 5-Aryl-1,2,4-Triazolidine-3-Thiones under Catalyst-Free Conditions. RSC Adv. 2015, 5, 51188–51192. [Google Scholar] [CrossRef]
- Ramesh, R.; Lalitha, A. Facile and Green Chemistry Access to 5-aryl-1,2,4-Triazolidine-3-thiones in Aqueous Medium. ChemistrySelect 2016, 1, 2085–2089. [Google Scholar] [CrossRef]
- Pore, D.M.; Hegade, P.G.; Mane, M.M.; Patil, J.D. The Unprecedented Synthesis of Novel Spiro-1,2,4-Triazolidinones. RSC Adv. 2013, 3, 25723. [Google Scholar] [CrossRef]
- Patil, J.D.; Pore, D.M. [C16MPy]AlCl3Br: An Efficient Novel Ionic Liquid for Synthesis of Novel 1,2,4-Triazolidine-3-Thiones in Water. RSC Adv. 2014, 4, 14314. [Google Scholar] [CrossRef]
- Masram, L.B.; Salim, S.S.; Barkule, A.B.; Gadkari, Y.U.; Telvekar, V.N. An Efficient and Expeditious Synthesis of 1,2,4-Triazolidine-3-Thiones Using Meglumine as a Reusable Catalyst in Water. J Chem Sci 2022, 134, 94. [Google Scholar] [CrossRef]
- Rezaei, I.; Nobarzad, R.S.; Shahri, F.; Nazeriyeh, I. A Novel, Effective, Green and Recyclable α-Fe2O3@MoS2@Ni Magnetic Nanocatalyst in Preparation of a Series of 1,2,4-Triazolidine-3-Thiones and Spiro-Triazole Hybrids. 10.5267/j.ccl 2024, 13, 725–736. [Google Scholar] [CrossRef]
- Godhani, D.R.; Jogel, A.A.; Sanghani, A.M.; Mehta, J.P. ChemInform Abstract: Synthesis and Biological Screening of 1,2,4-Triazole Derivatives. ChemInform 2015, 46, chin.201545146. [Google Scholar] [CrossRef]
- Dayama, D.S.; Khatale, P.N.; Khedkar, S.A.; Nazarkar, S.R.; Vedpathak, P.A. Synthesis and Biological Evaluation of Some Novel 1,2,4-Triazole Derivatives. Der Pharma Chem. 2014, 6, 123–127. [Google Scholar]
- Seelam, N.; Shrivastava, S.P.; S., P.; Gupta, S. Synthesis and in Vitro Study of Some Fused 1,2,4-Triazole Derivatives as Antimycobacterial Agents. Journal of Saudi Chemical Society 2016, 20, 411–418. [Google Scholar] [CrossRef]
- Agrawal, R.; Pancholi, S.S. Synthesis, Characterization and Evaluation of Antimicrobial Activity of a Series of 1,2,4-Triazoles. Der Pharma Chem. 2011, 3, 32–40. [Google Scholar]
- Belkadi, M.; Othman, A.A. Regioselective Glycosylation: Synthesis, Characterization and Biological Evaluation of New Acyclo C-Nucleosides Bearing 5-(Substituted)-1,3,4-Oxadiazole-2-Thione, 5-(Substituted)-4-Amino-1,2,4-Triazole-3-Thiol and 5-(Substituted)-1,2,4-Triazole-3-Thiones Moieties. Trends Appl. Sci. Res. 2011, 6, 19–33. [Google Scholar]
- Datoussaid, Y.; Othman, A.A.; Kirsch, G. Synthesis and Antibacterial Activity of Some 5,5’-(1,4-Phenylene)-bis-1,3,4-Oxadiazole and bis-1,2,4-Triazole Derivatives as Precursors of New S-Nucleosides. S. Afr. J. Chem. 2012, 65, 30–35. [Google Scholar]
- Andrews, B.; Ahmed, M. Synthesis and Characterization of Pyrimidine Bearing 1,2,4-Triazole Derivatives and Their Potential Antibacterial Action. Der Pharma Chem. 2014, 6, 162–169. [Google Scholar]
- Andrews, B.; Ahmed, M. Synthesis and Characterization of Pyrimidine Bearing 1,2,4-Triazole Derivatives and Their Potential Antifungal Action. Int. J. ChemTech Res. 2014, 6, 1013–1021. [Google Scholar]
- El-Feky, S.M.; Abou-Zeid, L.A.; Massoud, M.A.; George, S.K.; Eisa, H.M. Computational Design, Molecular Modeling and Synthesis of New 1,2,4-Triazole Analogs with Potential Antifungal Activities. SMU Med. J. 2014, 1, 224–242. [Google Scholar]
- Mali, R.K.; Somani, R.R.; Toraskar, M.P.; Mali, K.K.; Naik, P.P.; Shirodkar, P.Y. Synthesis of Some Antifungal and Anti-Tubercular 1,2,4-Triazole Analogues. Int. J. ChemTech Res. 2009, 1, 168–173. [Google Scholar]
- El Ashry, E.S.H.; El Tamany, E.S.H.; El Fattah, M.E.D.A.; Boraei, A.T.A.; Abd El-Nabi, H.M. Regioselective Synthesis, Characterization and Antimicrobial Evaluation of S-Glycosides and S,N-Diglycosides of 1,2-Dihydro-5-(1H-Indol-2-Yl)-1,2,4-Triazole-3-Thione. European Journal of Medicinal Chemistry 2013, 66, 106–113. [Google Scholar] [CrossRef]
- Amara, S.; Othman, A.A. A Convenient New Synthesis, Characterization and Antibacterial Activity of Double Headed Acyclo-C-Nucleosides from Unprotected d-Glucose. Arabian Journal of Chemistry 2016, 9, S1840–S1846. [Google Scholar] [CrossRef]
- Ledeţi, I.; Bercean, V.; Alexa, A.; Şoica, C.; Şuta, L.-M.; Dehelean, C.; Trandafirescu, C.; Muntean, D.; Licker, M.; Fuliaş, A. Preparation and Antibacterial Properties of Substituted 1,2,4-Triazoles. Journal of Chemistry 2015, 2015, 1–5. [Google Scholar] [CrossRef]
- Manikrao, A.M.; Fursule, R.A.; Sable, P.M.; Kunjwani, H.K. Accelerated Synthesis of 3-(N-Substituted Carboxamidomethylthio)-4(H)-1,2,4-Triazoles Under Microwave Irradiation. Int. J. ChemTech Res. 2009, 1, 1268–1272. [Google Scholar]
- Farhan, M.; Assy, M. Heterocyclization of Isoniazid: Synthesis and Antimicrobial Activity of Some New Pyrimidine 1, 3-Thiazole, 1, 2, 4-Thiadiazole, and 1, 2, 4-Triazole, Derivatives Derived from Isoniazid. Egypt. J. Chem. 2018, 0, 0–0. [Google Scholar] [CrossRef]
- Barbuceanu, S.-F.; Draghici, C.; Barbuceanu, F.; Bancescu, G.; Saramet, G. Design, Synthesis, Characterization and Antimicrobial Evaluation of Some Heterocyclic Condensed Systems with Bridgehead Nitrogen from Thiazolotriazole Class. Chem. Pharm. Bull. 2015, 63, 694–700. [Google Scholar] [CrossRef]
- Tratrat, C.; Haroun, M.; Paparisva, A.; Geronikaki, A.; Kamoutsis, Ch.; Ćirić, A.; Glamočlija, J.; Soković, M.; Fotakis, Ch.; Zoumpoulakis, P.; et al. Design, Synthesis and Biological Evaluation of New Substituted 5-Benzylideno-2-Adamantylthiazol [3,2-b][1,2,4]Triazol-6(5 H )Ones. Pharmacophore Models for Antifungal Activity. Arabian Journal of Chemistry 2018, 11, 573–590. [Google Scholar] [CrossRef]
- Venkatachalam, T.; Sasi, P.; Senthilkumar, N.; Muthukrishnan, M.; Asrar Ahamed, A.; Premkumar, R. Design, Synthesis, and In-Vitro Anti-Tuberculosis Activity of 2-Substituted-1,5-Diphenyl-1,2-Dihydro-3H-1,2,4-Triazole-3-Thione Derivatives. J. Phys.: Conf. Ser. 2024, 2801, 012016. [Google Scholar] [CrossRef]
- Hozien, Z.A.; EL-Mahdy, A.F.M.; Abo Markeb, A.; Ali, L.S.A.; El-Sherief, H.A.H. Synthesis of Schiff and Mannich Bases of New s -Triazole Derivatives and Their Potential Applications for Removal of Heavy Metals from Aqueous Solution and as Antimicrobial Agents. RSC Adv. 2020, 10, 20184–20194. [Google Scholar] [CrossRef]
- Cui, J.; Jin, J.; Chaudhary, A.S.; Hsieh, Y.; Zhang, H.; Dai, C.; Damera, K.; Chen, W.; Tai, P.C.; Wang, B. Design, Synthesis and Evaluation of Triazole-Pyrimidine Analogues as SecA Inhibitors. ChemMedChem 2016, 11, 43–56. [Google Scholar] [CrossRef]
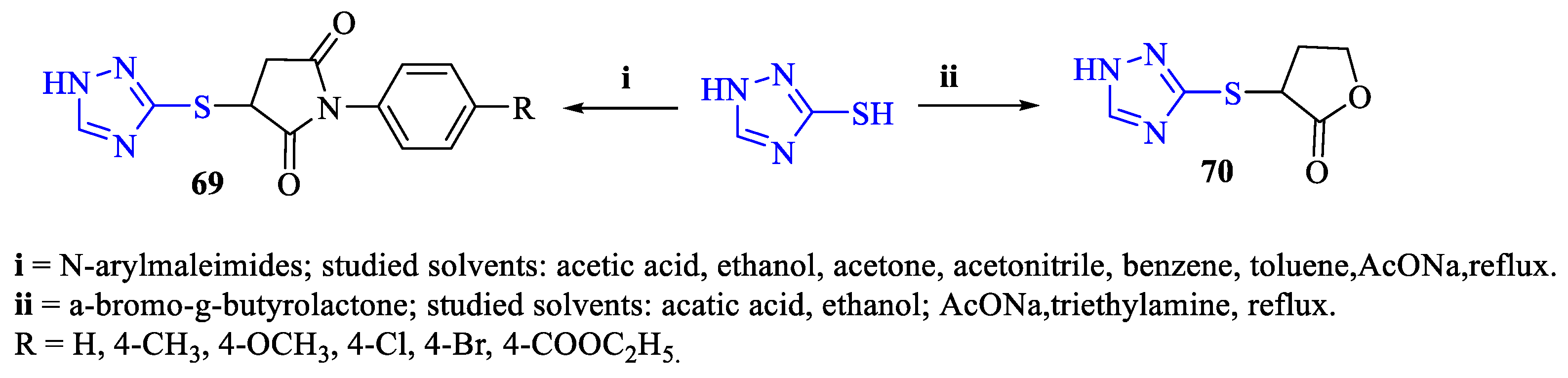
- Holota, S.; Derkach, H.; Antoniv, O.; Slyvka, N.; Kutsyk, R.; Gzella, A.; Lesyk, R. Study of 1,2,4-triazole-3(5)-thiol Behavior in Reactions with 1-phenyl-1H-pyrrole-2,5-dione Derivatives and 3-bromodihydrofuran-2(3H)-one and Antimicrobial Activity of Products. Chem. Proc. 2021, 3, 68. [Google Scholar] [CrossRef]
- Mioc, M.; Avram, S.; Tomescu, A.B.; Chiriac, D.V.; Heghes, A.; Voicu, M.; Voicu, A.; Citu, C.; Kurunczi, L. Docking Study of 3-Mercapto-1,2,4-Triazole Derivatives as Inhibitors for VEGFR and EGFR. Rev. Chim. 2017, 68, 500–503. [Google Scholar] [CrossRef]
- Mioc, M.; Avram, S.; Bercean, V.; Balan Porcarasu, M.; Soica, C.; Susan, R.; Kurunczi, L. Synthesis, Characterization and Antiproliferative Activity Assessment of a Novel 1H-5-Mercapto-1,2,4 Triazole Derivative. Rev. Chim. 2017, 68, 745–747. [Google Scholar] [CrossRef]
- Mioc, M.; Soica, C.; Bercean, V.; Avram, S.; Balan-Porcarasu, M.; Coricovac, D.; Ghiulai, R.; Muntean, D.; Andrica, F.; Dehelean, C.; et al. Design, Synthesis and Pharmaco-Toxicological Assessment of 5-Mercapto-1,2,4-Triazole Derivatives with Antibacterial and Antiproliferative Activity. International Journal of Oncology 2017, 50, 1175–1183. [Google Scholar] [CrossRef]
- Mioc, M.; Avram, S.; Bercean, V.; Kurunczi, L.; Ghiulai, R.M.; Oprean, C.; Coricovac, D.E.; Dehelean, C.; Mioc, A.; Balan-Porcarasu, M.; et al. Design, Synthesis and Biological Activity Evaluation of S-Substituted 1H-5-Mercapto-1,2,4-Triazole Derivatives as Antiproliferative Agents in Colorectal Cancer. Front. Chem. 2018, 6, 373. [Google Scholar] [CrossRef]
- Aliabadi, A.; Mohammadi-Frarni, A.; Azizi, M.; Ahmadi, F. Design, Synthesis and Cytotoxicity Evaluation of N-(5-Benzylthio)-4H-1,2,4-Triazol-3-YL)-4-Fluorobenzamide Derivatives as Potential Anticancer Agents. Pharm Chem J 2016, 49, 694–699. [Google Scholar] [CrossRef]
- Pachuta-Stec, A.; Rzymowska, J.; Mazur, L.; Mendyk, E.; Pitucha, M.; Rzączyńska, Z. Synthesis, Structure Elucidation and Antitumour Activity of N-Substituted Amides of 3-(3-Ethylthio-1,2,4-Triazol-5-Yl)Propenoic Acid. European Journal of Medicinal Chemistry 2009, 44, 3788–3793. [Google Scholar] [CrossRef]
- Murty, M.S.R.; Ram, K.R.; Rao, B.R.; Rao, R.V.; Katiki, M.R.; Rao, J.V.; Pamanji, R.; Velatooru, L.R. Synthesis, Characterization, and Anticancer Studies of S and N Alkyl Piperazine-Substituted Positional Isomers of 1,2,4-Triazole Derivatives. Med Chem Res 2014, 23, 1661–1671. [Google Scholar] [CrossRef]
- Zhu, X.-P.; Lin, G.-S.; Duan, W.-G.; Li, Q.-M.; Li, F.-Y.; Lu, S.-Z. Synthesis and Antiproliferative Evaluation of Novel Longifolene-Derived Tetralone Derivatives Bearing 1,2,4-Triazole Moiety. Molecules 2020, 25, 986. [Google Scholar] [CrossRef] [PubMed]
- Shahzad, S.A.; Yar, M.; Khan, Z.A.; Shahzadi, L.; Naqvi, S.A.R.; Mahmood, A.; Ullah, S.; Shaikh, A.J.; Sherazi, T.A.; Bale, A.T.; et al. Identification of 1,2,4-Triazoles as New Thymidine Phosphorylase Inhibitors: Future Anti-Tumor Drugs. Bioorganic Chemistry 2019, 85, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Mruthyunjaya, J.H.; Gopalakrishna, B. Synthesis and Antitumor Activity of New 2-(Pyridin-4-yl)thiazolo [3,2-b][1,2,4]triazol-6(5H)-one Derivatives. UJPBS 2013, 1, 46–51. [Google Scholar]
- El-Sherief, H.A.M.; Youssif, B.G.M.; Abbas Bukhari, S.N.; Abdelazeem, A.H.; Abdel-Aziz, M.; Abdel-Rahman, H.M. Synthesis, Anticancer Activity and Molecular Modeling Studies of 1,2,4-Triazole Derivatives as EGFR Inhibitors. European Journal of Medicinal Chemistry 2018, 156, 774–789. [Google Scholar] [CrossRef]
- Aouad, M.R.; Al-Mohammadi, H.M.; Al-blewi, F.F.; Ihmaid, S.; Elbadawy, H.M.; Althagfan, S.S.; Rezki, N. Introducing of Acyclonucleoside Analogues Tethered 1,2,4-Triazole as Anticancer Agents with Dual Epidermal Growth Factor Receptor Kinase and Microtubule Inhibitors. Bioorganic Chemistry 2020, 94, 103446. [Google Scholar] [CrossRef]
- Holota, S.; Komykhov, S.; Sysak, S.; Gzella, A.; Cherkas, A.; Lesyk, R. Synthesis, Characterization and In Vitro Evaluation of Novel 5-Ene-Thiazolo [3,2-b][1,2,4]Triazole-6(5H)-Ones as Possible Anticancer Agents. Molecules 2021, 26, 1162. [Google Scholar] [CrossRef]
- Zhou, W.; Xu, C.; Dong, G.; Qiao, H.; Yang, J.; Liu, H.; Ding, L.; Sun, K.; Zhao, W. Development of Phenyltriazole Thiol-Based Derivatives as Highly Potent Inhibitors of DCN1-UBC12 Interaction. European Journal of Medicinal Chemistry 2021, 217, 113326. [Google Scholar] [CrossRef]
- El-Wahab, H.A.A.A.; Ali, A.M.; Abdel-Rahman, H.M.; Qayed, W.S. Synthesis, Biological Evaluation, and Molecular Modeling Studies of Acetophenones-tethered 1,2,4-triazoles and Their Oximes as Epidermal Growth Factor Receptor Inhibitors. Chem Biol Drug Des 2022, 100, 981–993. [Google Scholar] [CrossRef]
- Boraei, A.T.A.; Gomaa, M.S.; El Ashry, E.S.H.; Duerkop, A. Design, Selective Alkylation and X-Ray Crystal Structure Determination of Dihydro-Indolyl-1,2,4-Triazole-3-Thione and Its 3-Benzylsulfanyl Analogue as Potent Anticancer Agents. European Journal of Medicinal Chemistry 2017, 125, 360–371. [Google Scholar] [CrossRef] [PubMed]
- Manikrao, A.M.; Fursule, R.A.; Rajesh, K.S.; Kunjwani, H.K.; Sabale, P.M. Synthesis and Biological Screening of Novel Derivatives of 3-(N-Substituted Carboxamidoethylthio)-(4H)-1,2,4-triazoles. Indian J. Chem. B 2010, 49, 1642–1647.1. [Google Scholar] [CrossRef]
- Manikrao, A.M.; Chaple, D.R.; Khatale, P.N.; Sable, P.M.; Jawarkar, R.D. Impact of Tautomery of 3-(4H-1,2,4-Triazol-3-Ylthio)-N-Phenylpropanamide on the COX-1 Inhibitory Mechanism. Journal of Enzyme Inhibition and Medicinal Chemistry 2013, 28, 523–529. [Google Scholar] [CrossRef]
- Uzgören-Baran, A.; Tel, B.C.; Sarıgöl, D.; Öztürk, E.İ.; Kazkayası, İ.; Okay, G.; Ertan, M.; Tozkoparan, B. Thiazolo [3,2-b]-1,2,4-Triazole-5(6H)-One Substituted with Ibuprofen: Novel Non-Steroidal Anti-Inflammatory Agents with Favorable Gastrointestinal Tolerance. European Journal of Medicinal Chemistry 2012, 57, 398–406. [Google Scholar] [CrossRef]
- Cetin, A.; Gecibesler, I. Evaluation as Antioxidant Agents of 1,2,4-Triazole Derivatives: Effects of Essential Functional Groups. J App Pharm Sci 2015, 120–126. [Google Scholar] [CrossRef]
- Tozkoparan, B.; Küpeli, E.; Yeşilada, E.; Ertan, M. Preparation of 5-Aryl-3-Alkylthio-l,2,4-Triazoles and Corresponding Sulfones with Antiinflammatory–Analgesic Activity. Bioorganic & Medicinal Chemistry 2007, 15, 1808–1814. [Google Scholar] [CrossRef]
- Muneer, C.P.; Begum, T.S.; Shafi, P.M. Synthesis, Characterization and Antioxidant Study of a Few 3-Substituted 1,2,4-Triazole-5-thiones and Their Derivatives. Int. J. Chem. Sci. 2014, 12, 129–135. [Google Scholar]
- Shiradkar, M.R.; Ghodake, M.; Bothara, K.G.; Bhandari, S.V.; Nikalje, A.; Akula, K.C.; Desai, N.C.; Burange, P.J. Synthesis and Anticonvulsant Activity of Clubbed Thiazolidinone–Barbituric Acid and Thiazolidinone-Triazole Derivatives. ARKIVOC 2007, xiv, 58–74. [Google Scholar] [CrossRef]
- Makovik, Y.V. Synthesis, Physical-Chemical and Biological Properties of S-Derivatives 5-(3-Pyridyl)- and 5-(3-Pyridyl)-4-Phenyl-1,2,4-Triazol-3-Thione [Dissertation]. National Medical Academy of Postgraduate Education named after P.L. Shupik; 2008.
- Naseer, M.A.; Husain, A. Studies on Chromene Based 2, 6-Disubstituted-Thiazolo [3,2-B] [1,2,4] Triazole Derivatives: Synthesis and Biological Evaluation. J. Drug Delivery Ther. 2019, 9, 236–242. [Google Scholar] [CrossRef]
- Krutov, I.A.; Gavrilova, E.L.; Burangulova, R.N.; Kornilov, S.S.; Valieva, A.A.; Samigullina, A.I.; Gubaidullin, A.T.; Sinyashin, O.G.; Semina, I.I.; Nikitin, D.O.; et al. Modification of Diphenylphosphorylacetic Hydrazide with Thiosemicarbazide and Triazole Units. Russ J Gen Chem 2017, 87, 2794–2800. [Google Scholar] [CrossRef]
- Navidpour, L.; Shabani, S.; Heidari, A.; Bashiri, M.; Ebrahim-Habibi, A.; Shahhosseini, S.; Shafaroodi, H.; Abbas Tabatabai, S.; Toolabi, M. 5-[Aryloxypyridyl (or Nitrophenyl)]-4H-1,2,4-Triazoles as Novel Flexible Benzodiazepine Analogues: Synthesis, Receptor Binding Affinity and Lipophilicity-Dependent Anti-Seizure Onset of Action. Bioorganic Chemistry 2021, 106, 104504. [Google Scholar] [CrossRef] [PubMed]
- Sert-Ozgur, S.; Tel, B.C.; Somuncuoglu, E.I.; Kazkayasi, I.; Ertan, M.; Tozkoparan, B. Design and Synthesis of 1,2,4-Triazolo [3,2- b ]-1,3,5-thiadiazine Derivatives as a Novel Template for Analgesic/Anti-Inflammatory Activity. Archiv der Pharmazie 2017, 350, e1700052. [Google Scholar] [CrossRef]
- Toma, A.; Mogoşan, C.; Vlase, L.; Leonte, D.; Zaharia, V. Heterocycles 39. Synthesis, Characterization and Evaluation of the Anti-Inflammatory Activity of Thiazolo [3,2-b][1,2,4]Triazole Derivatives Bearing Pyridin-3/4-Yl Moiety. Med Chem Res 2017, 26, 2602–2613. [Google Scholar] [CrossRef]
- Cristina, A. HETEROCYCLES 46. SYNTHESIS, CHARACTERIZATION AND BIOLOGICAL EVALUATION OF THIAZOLO [3,2-b][1,2,4]TRIAZOLES BEARING BENZENESULFONAMIDE MOIETY. FARMACIA 2018, 66, 883–893. [Google Scholar] [CrossRef]
- Mauler-Machnik, A.; Rosslenbroich, H.J.; Dutzmann, S.; Applegate, J.; Jautelat, M. JAU 6476 - A New Dimension DMI Fungicide. In Proceedings of the Brighton Crop Protection Conference - Pests and Diseases; BCPC: Farnham, Surrey, UK, 2002; pp. 389–394. [Google Scholar]
- Häuser-Hahn, I.; Baur, P.; Schmitt, W. Prothioconazole – A New Dimension DMI. Biochemistry, Mode of Action, Systemic Effects. Pflanzenschutz-Nachrichten Bayer 2004, 57, 237–248. [Google Scholar]
- Dutzmann, S.; Suty-Heinze, A. Prothioconazole: A Broad Spectrum Demethylation-Inhibitor (DMI) for Arable Crops. Pflanzenschutz-Nachrichten Bayer 2004, 57, 249–264. [Google Scholar]
- Davies, P.; Muncey, M. Prothioconazole for control of Sclerotinia sclerotiorum in oilseed rape/canola. Pflanzenschutz-Nachrichten Bayer 2004, 57, 283–293. [Google Scholar]
- Kuck, K.H.; Mehl, A. Prothioconazole: Sensitivity profile and anti-resistance strategy. Pflanzenschutz-Nachrichten Bayer 2004, 57, 225–236. [Google Scholar]
- Yano, T.; Yoshii, T.; Ueda, T.; Hori, M.; Hirai, K. Synthesis and Herbicidal Activity of New 2-(1-Carbamoyl-1,2,4-triazol-3-ylsulfonyl)alkanoates. J. Pestic. Sci. 2002, 27, 97–105. [Google Scholar] [CrossRef]
- Knyazyan, A.M. Synthesis of 5-(2-Thioxo-3H-thiazol-5-yl)-[1,2,4]triazole-3-thione Derivatives Exhibiting Fungicidal and Growth-Stimulating Activity. Khimičeskij žurnal Armenii 2012, 65, 94–104. [Google Scholar]
- Chai, B.; Qian, X.; Cao, S.; Liu, H.; Song, G. Synthesis and Insecticidal Activity of 1,2,4-Triazole Derivatives. ARKIVOC 2003, ii, 141–145. [Google Scholar] [CrossRef]
- Othman, M.S.; Naz, H.; Rahim, F.; Ullah, H.; Hussain, R.; Taha, M.; Khan, S.; Fareid, M.A.; Aboelnaga, S.M.; Altaleb, A.T.; et al. New Cholinesterase Inhibitors Based on 1,2,4-Triazole Bearing Benzenesulfonohydrazide Skeleton: Synthesis, in Vitro and in Silico Studies. Results in Chemistry 2024, 10, 101717. [Google Scholar] [CrossRef]
- Mahajan, P.G.; Dige, N.C.; Vanjare, B.D.; Raza, H.; Hassan, M.; Seo, S.-Y.; Kim, C.-H.; Lee, K.H. Synthesis and Biological Evaluation of 1,2,4-Triazolidine-3-Thiones as Potent Acetylcholinesterase Inhibitors: In Vitro and in Silico Analysis through Kinetics, Chemoinformatics and Computational Approaches. Mol Divers 2020, 24, 1185–1203. [Google Scholar] [CrossRef]
- Salerno, L.; Siracusa, M.; Guerrera, F.; Romeo, G.; Pittalà, V.; Modica, M.; Mennini, T.; Russo, F. Synthesis of New 5-Phenyl [1,2,4]Triazole Derivatives as Ligands for the 5-HT1A Serotonin Receptor. Arkivoc 2004, 2004, 312–324. [Google Scholar] [CrossRef]
- Samelyuk, Y.G.; Kaplaushenko, A.G.; Pruglo, Y.S. Synthesis and Actoprotective Activity of Salts of 2-(5-(4-Methoxyphenyl(3,4,5-trimethoxyphenyl))-1,2,4-Triazole-3-ylthio)acetic Acids. Zaporozh. Med. J. 2014, 83, 107–111. [Google Scholar]
- Dovbnia, D.; Frolova, Y.; Kaplaushenko, A. A Study of Hypoglycemic Activity of Acids and Salts Containing 1,2,4-Triazole. Ceska Slov. Farm. 2023, 72, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Fateev, I.V.; Sasmakov, S.A.; Abdurakhmanov, J.M.; Ziyaev, A.A.; Khasanov, S.Sh.; Eshboev, F.B.; Ashirov, O.N.; Frolova, V.D.; Eletskaya, B.Z.; Smirnova, O.S.; et al. Synthesis of Substituted 1,2,4-Triazole-3-Thione Nucleosides Using E. Coli Purine Nucleoside Phosphorylase. Biomolecules 2024, 14, 745. [Google Scholar] [CrossRef]
- Hadjadj, H.; Kahloula, K.; Meddah, B.; Slimani, M. 5-Phenyl-1, 2, 4-Triazole- 3- Thiol Subchronic Exposure Induce Neuro-Comportemental Desorder in Wistar Rats. SAJEB 2019, 8, 178–186. [Google Scholar] [CrossRef]
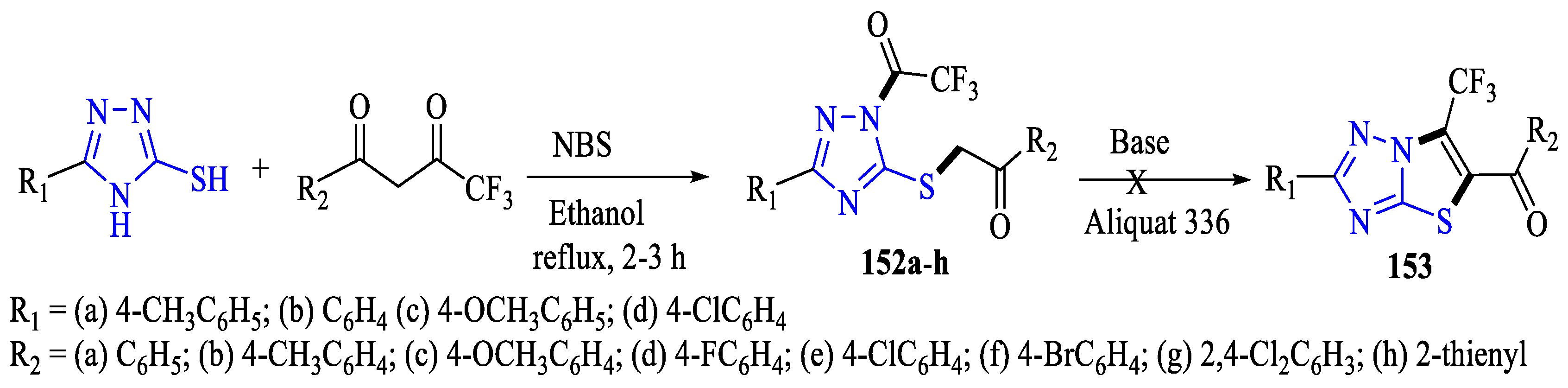
- Aggarwal, R.; Kumar, P.; Hooda, M.; Kumar, S. Serendipitous N, S -Difunctionalization of Triazoles with Trifluoromethyl-β-Diketones: Access to Regioisomeric 1-Trifluoroacetyl-3-Aryl-5-(2-Oxo-2-Arylethylthio)-1,2,4-Triazoles as DNA-Groove Binders. RSC Adv. 2024, 14, 6738–6751. [Google Scholar] [CrossRef]
- Ebdrup, S.; Sørensen, L.G.; Olsen, O.H.; Jacobsen, P. Synthesis and Structure−Activity Relationship for a Novel Class of Potent and Selective Carbamoyl-Triazole Based Inhibitors of Hormone Sensitive Lipase. J. Med. Chem. 2004, 47, 400–410. [Google Scholar] [CrossRef]
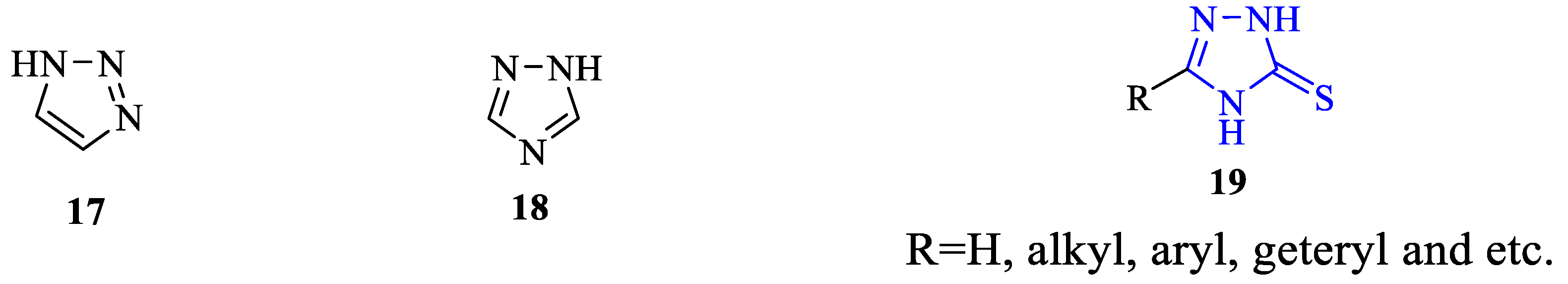
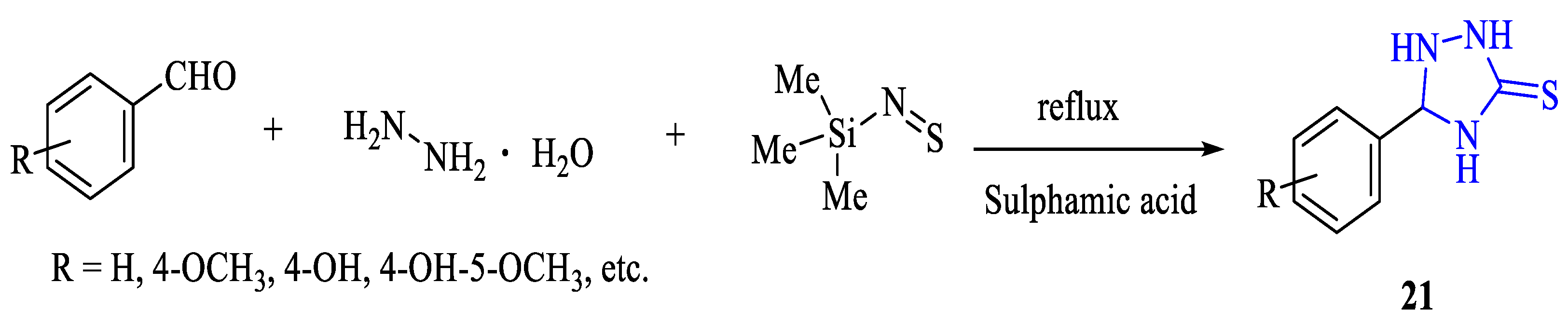
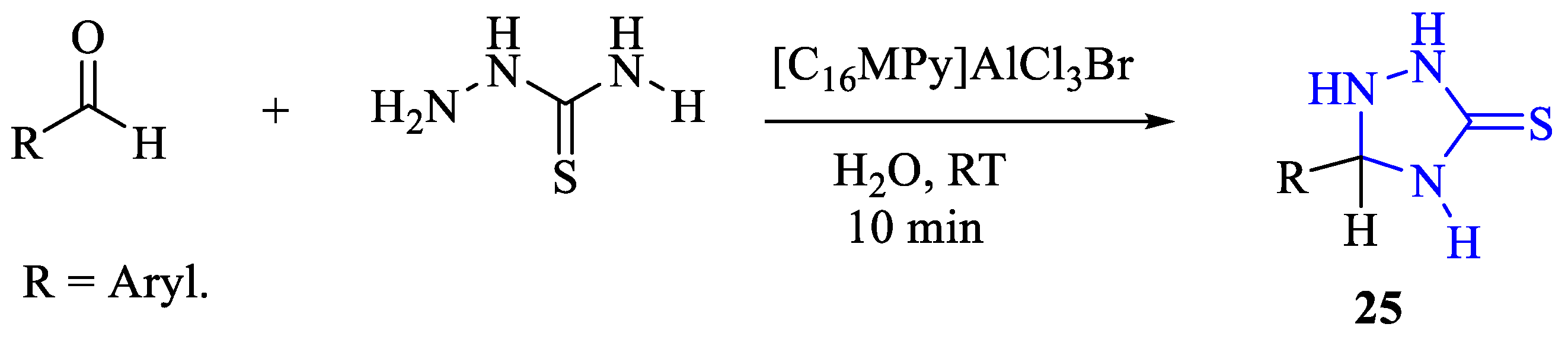
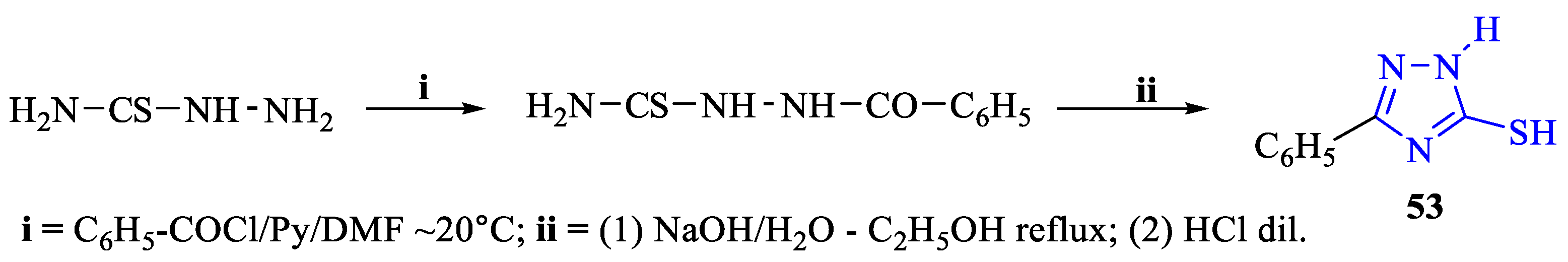
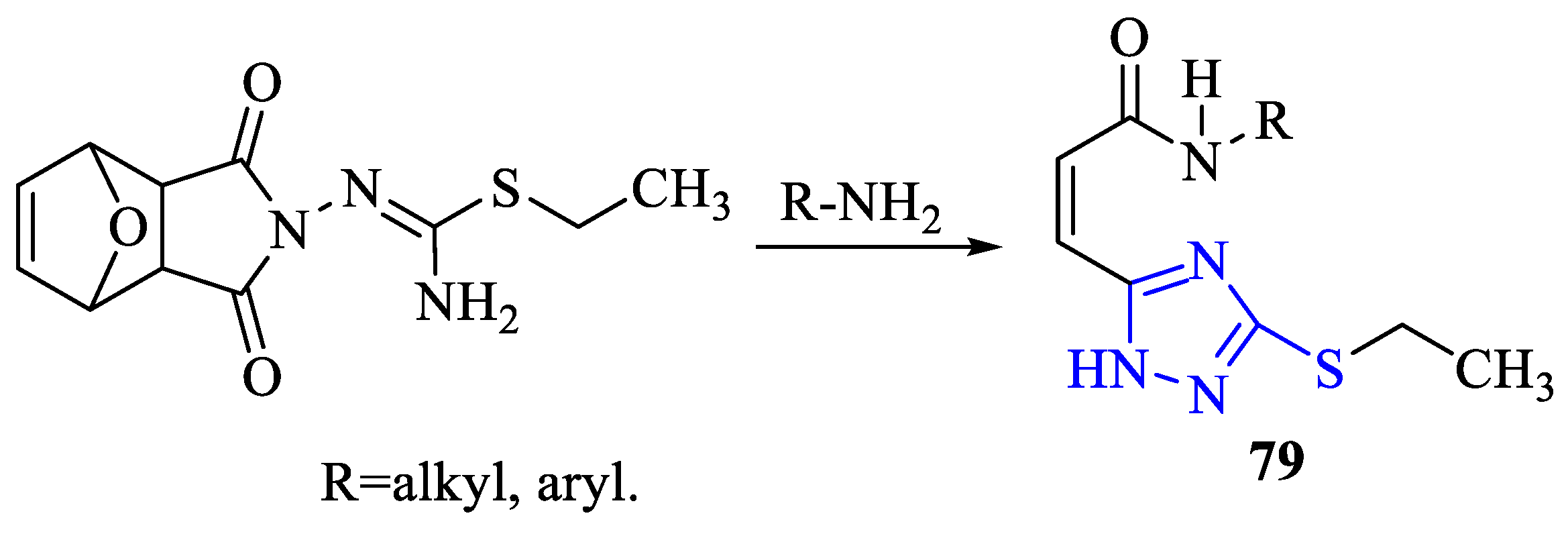
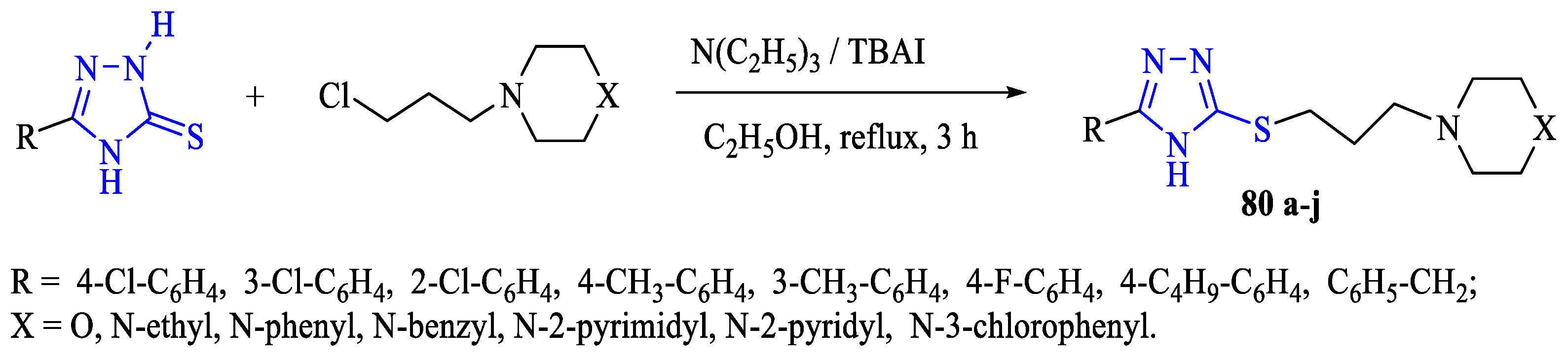
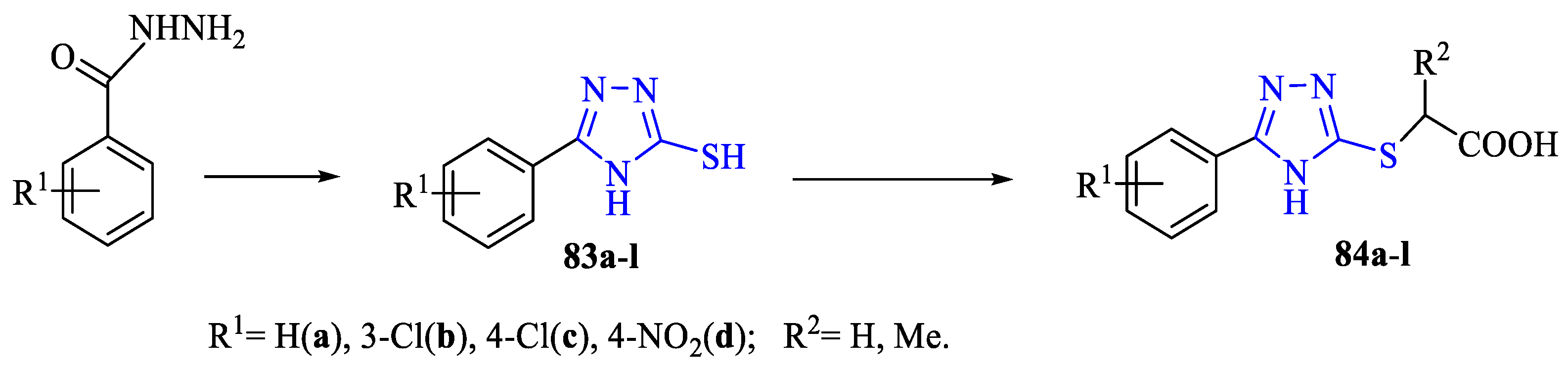
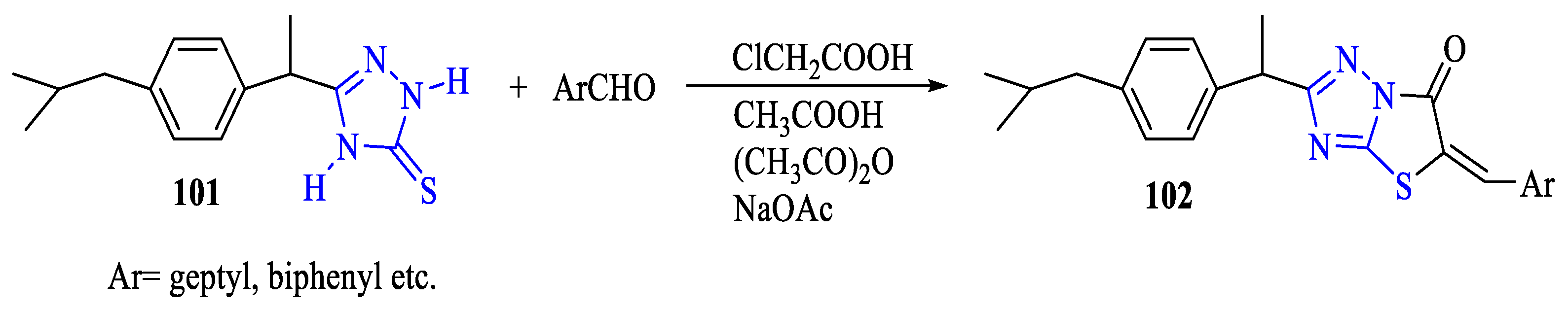
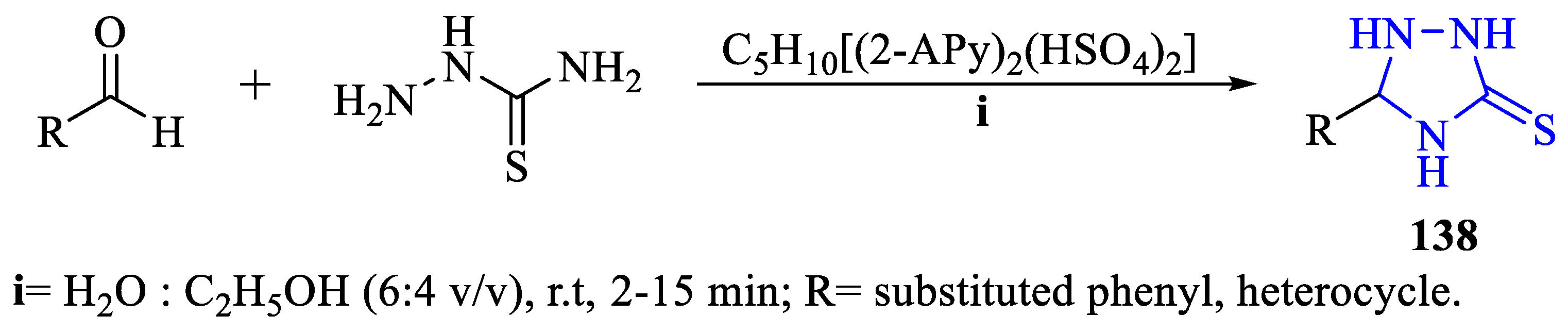

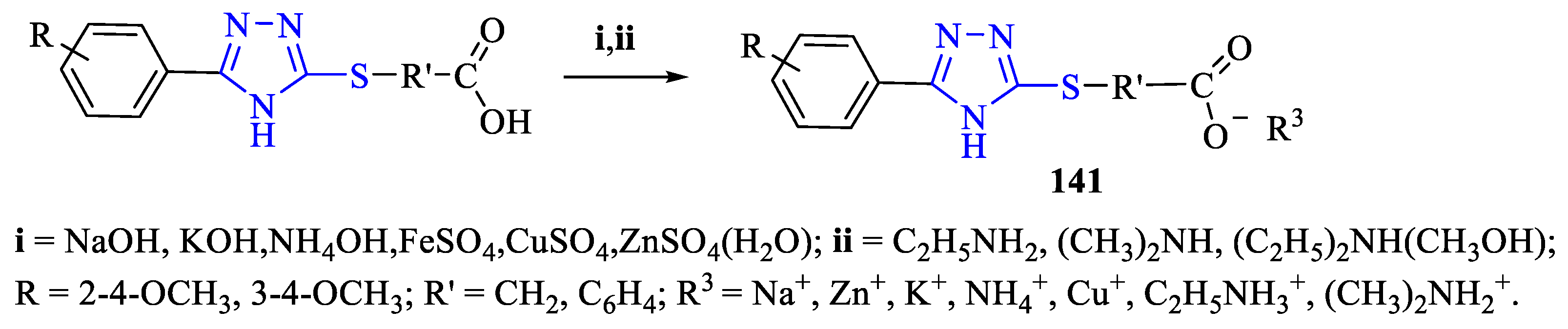
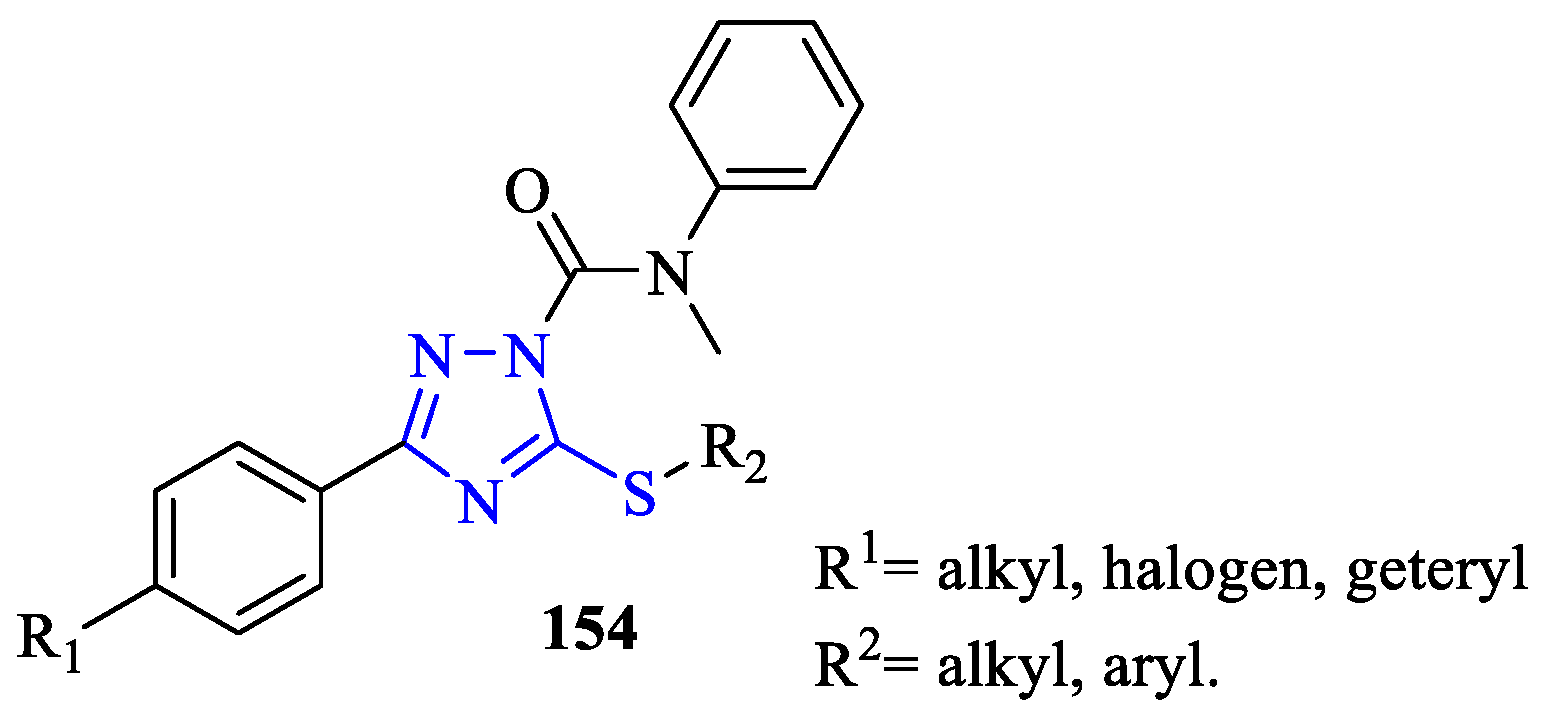
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



