Submitted:
24 June 2025
Posted:
25 June 2025
You are already at the latest version
Abstract
Keywords:
| AAA | Aortic arch abnormalities |
| APVD | Anomalous pulmonary venous drainage |
| AV | Atrioventricular |
| AVSD | Atrioventricular septal defect |
| CA | Common atrium |
| CHB-c | Complete heart block (congenital) |
| CHB-ac | Complete heart block (acquired) |
| CHB-po | Complete heart block (post-op) |
| Dextro | Dextrocardia |
| D-hand | D-hand topology |
| DORV | Double outlet right ventricle |
| ECG | Electrocardiogram |
| HLHS | Hypoplastic left heart syndrome |
| IVC | Inferior vena cava |
| IRB | Institutional Review Board |
| JIR | Junctional intermittent rhythm |
| JPR | Junctional persistent rhythm |
| KACC | King Abdulaziz Cardiac Center |
| KAMC | King Abdulaziz Medical City |
| LVI | Left visceral isomerism |
| Levo | Levocardia |
| LIA | Left inferior axis (0 to +90) |
| LSA | Left superior axis (0 to -90) |
| L-hand | L-hand topology |
| LR | Left Right |
| Meso | Mesocardia |
| MRN | Medical record number |
| ODS | Online data supplements |
| P | P-value |
| PA | Pulmonary atresia |
| PAPVD | Partially anomalous pulmonary venous drainage |
| PPM | Permanent pacemaker |
| PS | Pulmonary stenosis |
| PV | Pulmonary valve |
| RVI | Right visceral isomerism |
| RIA | Right inferior axis (+90 to 180) |
| RSA | Right superior axis (-90 to 180) |
| SA | Sinoatrial |
| SLV | Single left ventricle defect |
| SRV | Single right ventricle defect |
| SVC | Superior vena cava |
| TAPVD | Totally anomalous pulmonary venous drainage |
| TGA | Transposition of the great arteries |
| V | Ventricle |
-
Clinical PerspectivesWhat Is New?This study presents several novel and clinically significant findings:
- ▪
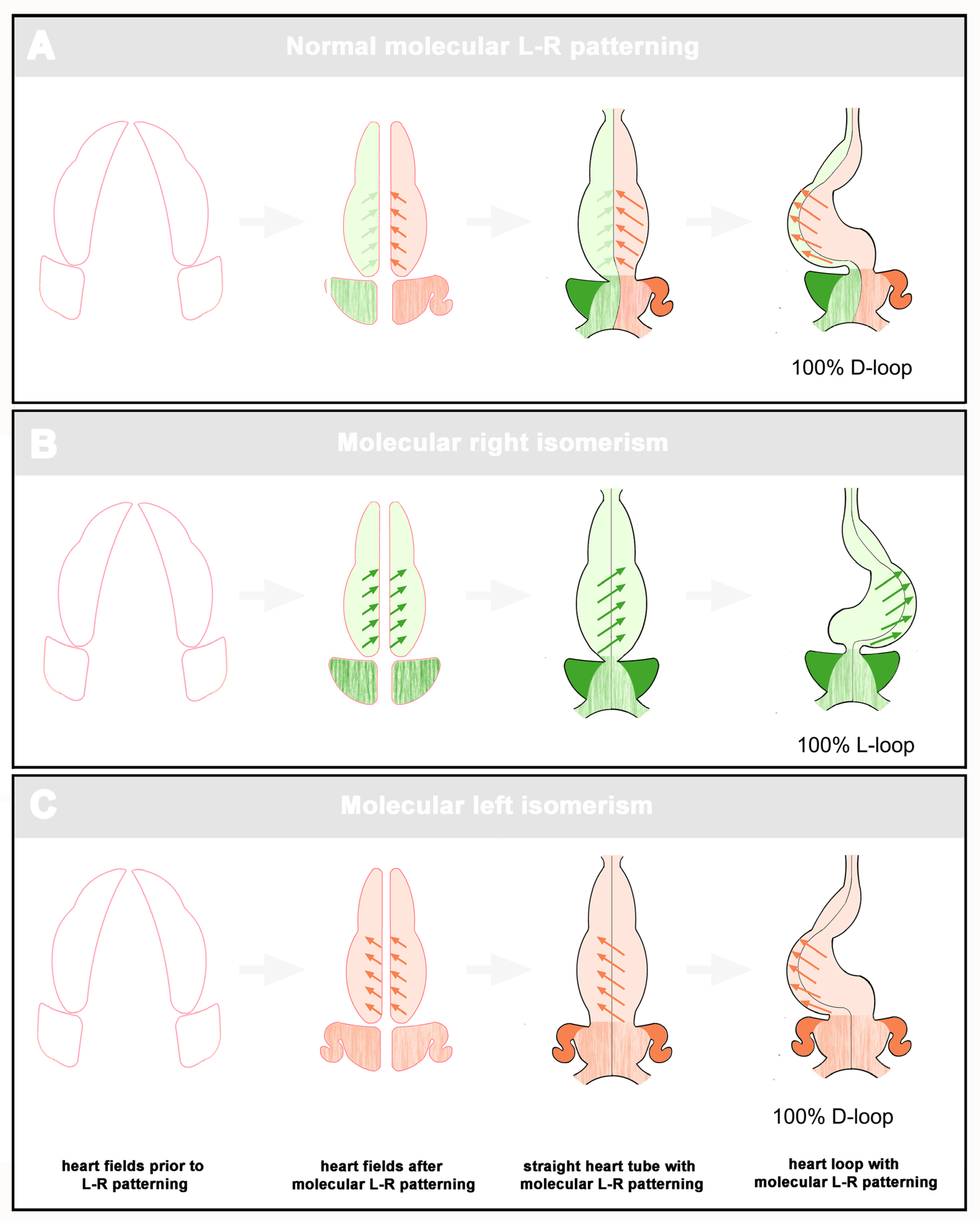
- We report a statistically significant difference in the distribution of ventricular topology between left visceral isomerism (LVI) and right visceral isomerism (RVI), revealing a strong bias towards D-hand topology in LVI that contrasts with a near-equal distribution in RVI. This observation challenges prevailing embryological theories of cardiac laterality and offer a more complex understanding of the developmental pathways involved in visceral isomerism.
- ▪
- Most notably, we have identified a novel and unexpected survival advantage for LVI patients with L-hand ventricular topology, demonstrating significantly lower mortality and improved long-term survival compared to those with D-hand topology. This finding has direct translational implications for risk stratification and management strategies in this high-risk patient group, warranting further investigation into the protective mechanisms associated with this specific cardiac configuration.
- ▪
- Furthermore, our study provides, to the best of our knowledge, the first evidence of a statistically significant association between ventricular topology and aortic arch sidedness across the entire cohort and within both LVI and RVI subgroups. This novel link suggests a functional relationship between early ventricular development and the subsequent formation of the aortic arch system, offering new insights into the integrated nature of cardiovascular development in visceral isomerism.
- What Are the Clinical Implications?
- ▪
- The novel association between ventricular topology and survival in LVI, in particular, has the potential to inform clinical practice and improve outcomes for these patients.
- ▪
- Moreover, the identified link between ventricular topology and aortic arch development contributes to a more holistic view of cardiac morphogenesis in this setting.
Introduction
Methods
Patient Population
- Inclusion criteria:
- ▪
- All patients with LVI or RVI seen at KACC from January 2000 until March 2023.
- Exclusion criteria:
- ▪
- Patients who did not have good quality echocardiograms due to poor imaging windows.
- ▪
- Patients who were erroneously labeled as visceral isomerism patients but found out to be non-isomeric during detailed data review process.
- Variables
- Each patient was evaluated for the following general variables:
- ▪
- Demographic data for the patient including sex and date of birth (DOB)
- ▪
- Mortality
- ▪
- Duration of follow-up (calculated from date of first echo, date of birth until the date of death or date of last follow-up)
- For any patient who had no documented follow-up after January 2021 until the end of our study (March 2023), we have contacted their families by phone using the documented contact information in the system to take additional information about patient’s current clinical condition and if he/she is still alive or not to determine mortality and survival period. Consent was taken from families by phone in accordance to our IRB committed guideline and a witness was present during the conducted phone calls. If we still were not able to contact the patients who had no follow-up documented in our system since January 2021, we considered them as missed follow-up patients.
- ▪
-
Main variables of cardio-vascular LR asymmetries:
- -
- Ventricular topology; D-hand or L-hand
- -
- Orientation of the cardiac apex; left-sided, right-sided, or midline
- -
- Aortic arch patterning; unilateral left, unilateral right, or bilateral (double aortic arch
- -
- Superior caval vein patterning; unilateral left, unilateral right, or bilateral
- -
- Inferior caval vein patterning; normal or interrupted
- ▪
-
Associated congenital heart defects:
- -
- Anomalies of pulmonary venous drainage; partial or total
- -
- Atrioventricular septal defect; balanced or unbalanced
- -
- Common atrium
- -
- Univentricular heart including the morphological identity of its ventricle
- -
- Hypoplastic left heart syndrome
- -
- Transposition of the great arteries
- -
- Double outlet right ventricle
- -
- Anatomy of pulmonary valve; stenosis, atresia, or normal
- -
- Aortic arch abnormalities; coarctation, interruption, or hypoplasia
- ▪
-
Associated congenital disorders of excitation and conduction:
- -
-
Generation of atrial activity (determined by the p-wave axis) divided into 4 groups:
- Left inferior axis (0 to +90)
- Left superior axis (0 to -90)
- Right inferior axis (+90 to 180)
- Right superior axis (-90 to 180)
- -
-
We further analyzed generation of atrial activity by combining the above 4 axis to another 4 groups
- Left axis (combining left inferior axis and left superior axis)
- Right axis (combining right inferior axis and right superior axis)
- Inferior axis (combining left inferior axis and right inferior axis)
- Superior axis (combining left superior axis and right superior axis)
- -
- Presence of documented atrial arrhythmias (SVT, atrial fibrillation, atrial flutter, atrial tachycardia)
- -
-
Conduction abnormalities (complete heart block, junctional rhythm whether intermittent or permanent).NOTE: Complete heart block was grouped into three categories:
-
- ▪
- Complete heart block (congenital)
- ▪
- Complete heart block (post-op)
- ▪
- Complete heart block (acquired)
-
- -
- Ventricular arrhythmias (ventricular fibrillation, ventricular tachycardia)
- -
- Permanent pacemaker insertion
- 1)
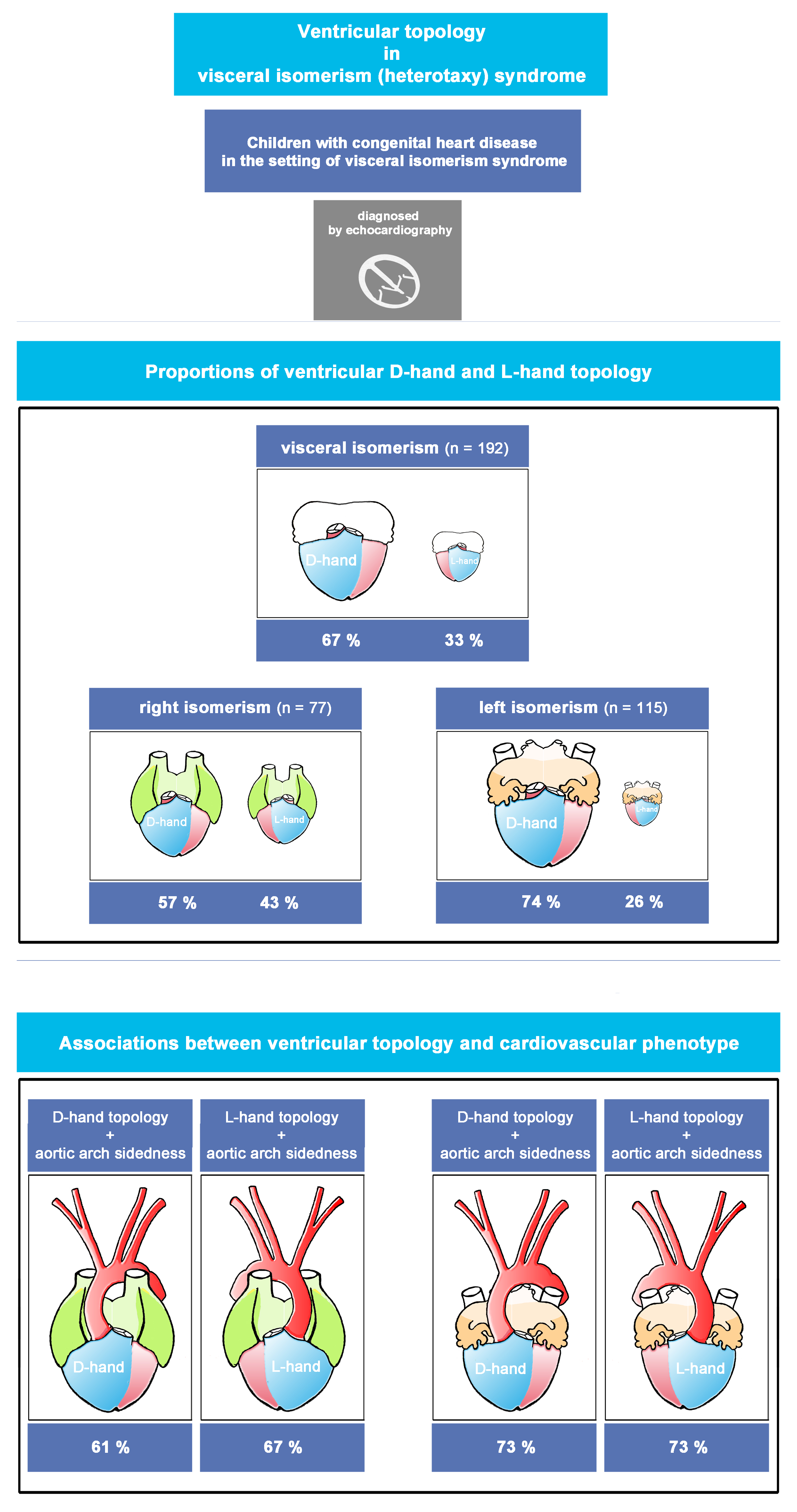
- the proportions of ventricular D- and L-hand topology in the whole study population.
- 2)
- the proportions of ventricular D- and L-hand topology in cases of (a) LVI, and (b) RVI.
- 3)
- statistically significant differences in mortality between LVI and RVI patients.
- 4)
- statistically significant associations between the type of ventricular topology (D-hand, L-hand) and mortality, (a) in the whole study population, and (b, c) in the two study sub-populations (LVI, RVI).
- 5)
- statistically significant associations between the type of ventricular topology (D-hand, L-hand) and orientation of the cardiac apex, aortic arch patterning, and patterning of SVC and IVC; (a) in the whole study population, and (b, c) in the two study sub-populations (LVI, RVI).
- 6)
- statistically significant associations between the type of ventricular topology (D-hand, L-hand) and specific congenital heart defects (AVSD, single ventricle, common atrium, TGA, DORV, PV defects, anomalies in pulmonary and systemic venous drainage); (a) in the whole study population, and (b, c) in the two study sub-populations (LVI, RVAI).
- 7)
- statistically significant associations between the type of ventricular topology (D-hand, L-hand) and congenital disorders of excitation and conduction;(a) in the whole study population, and (b, c) in the two study sub-populations (LVI, RVI).
Study Design and Data Collection
Statistical Analysis
Ethical Considerations
Results

Sex Distribution
Size of Study Population and Sub-Populations (LVI, RVI), and Distribution of Ventricular Topologies
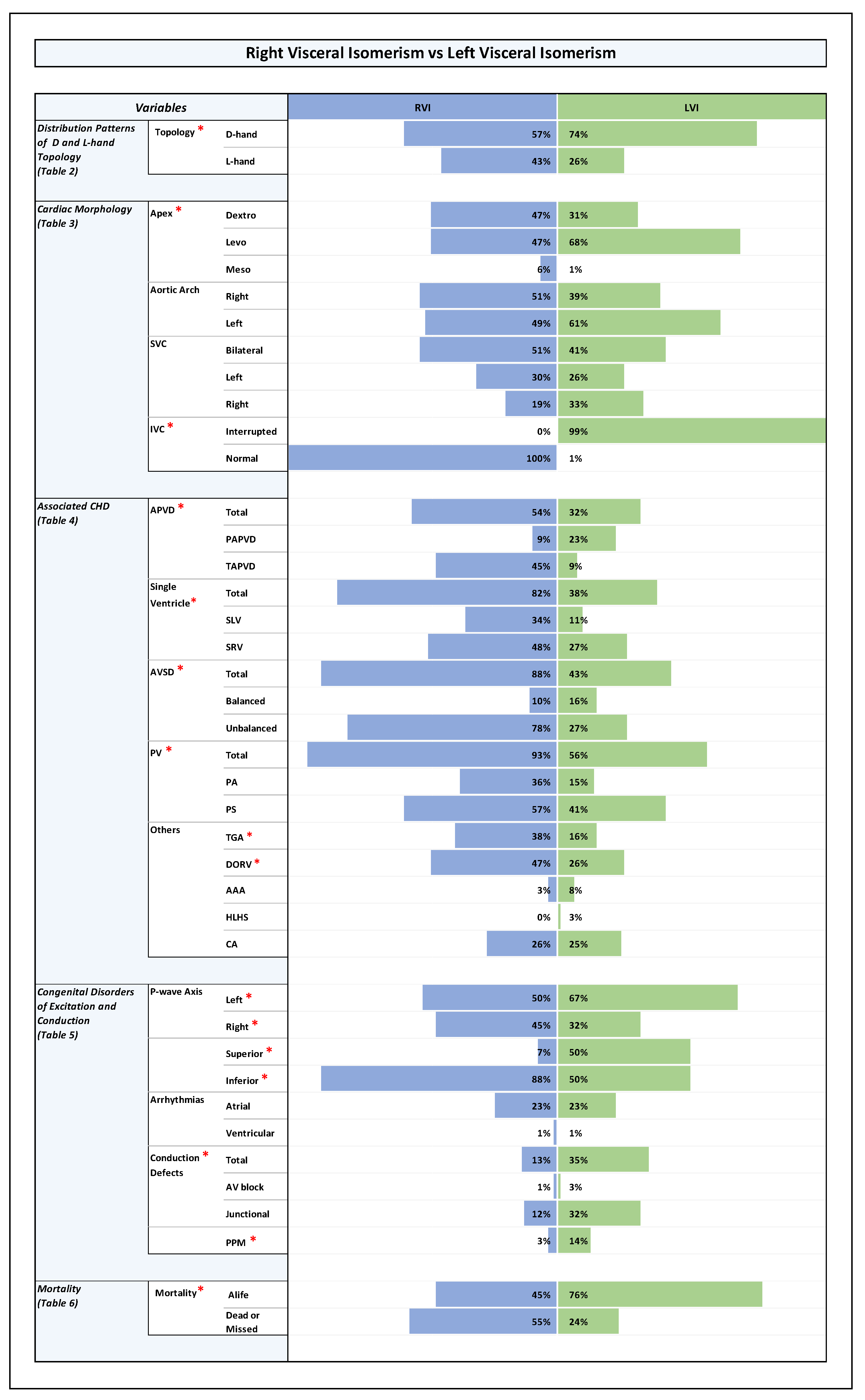
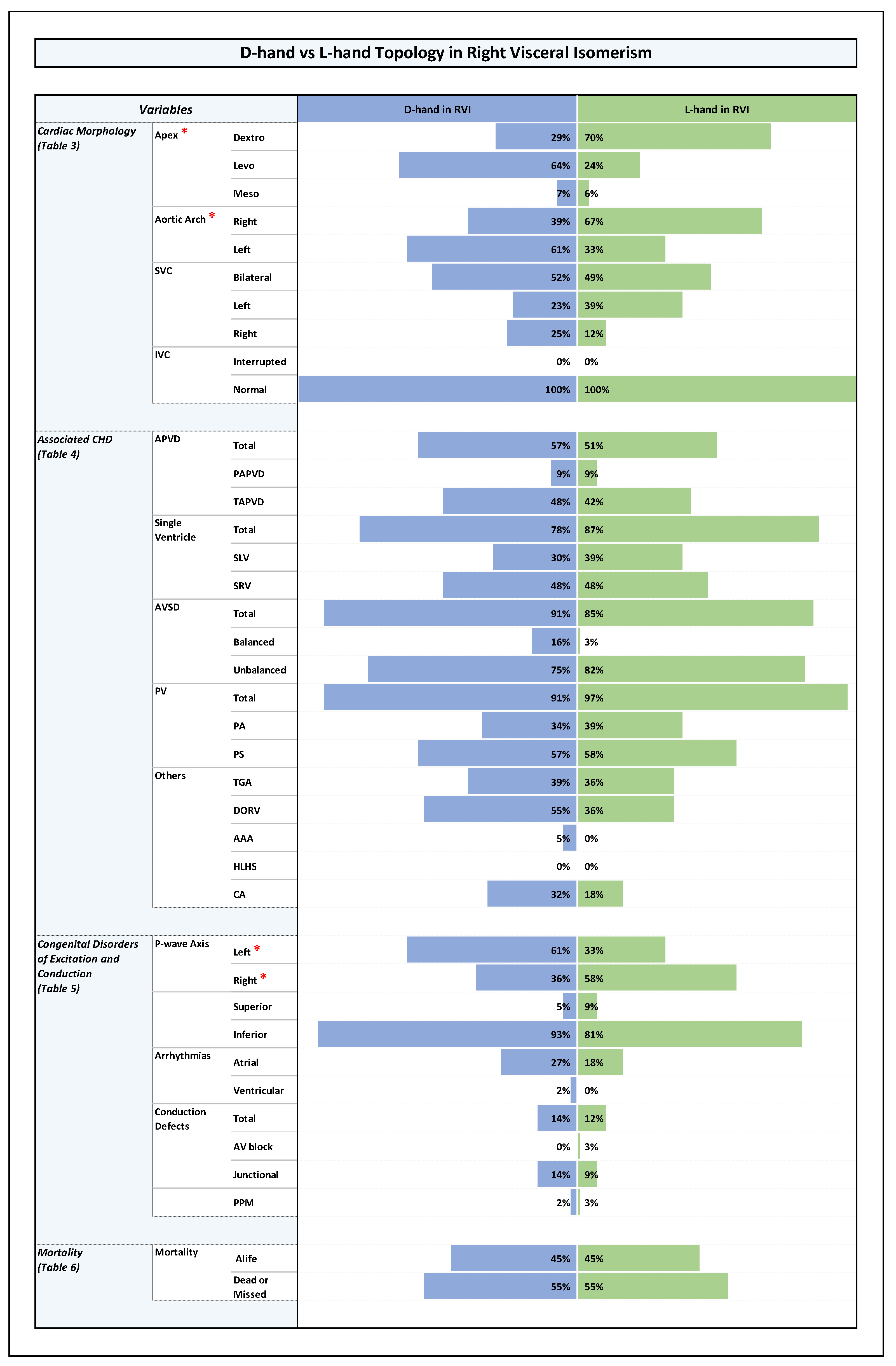
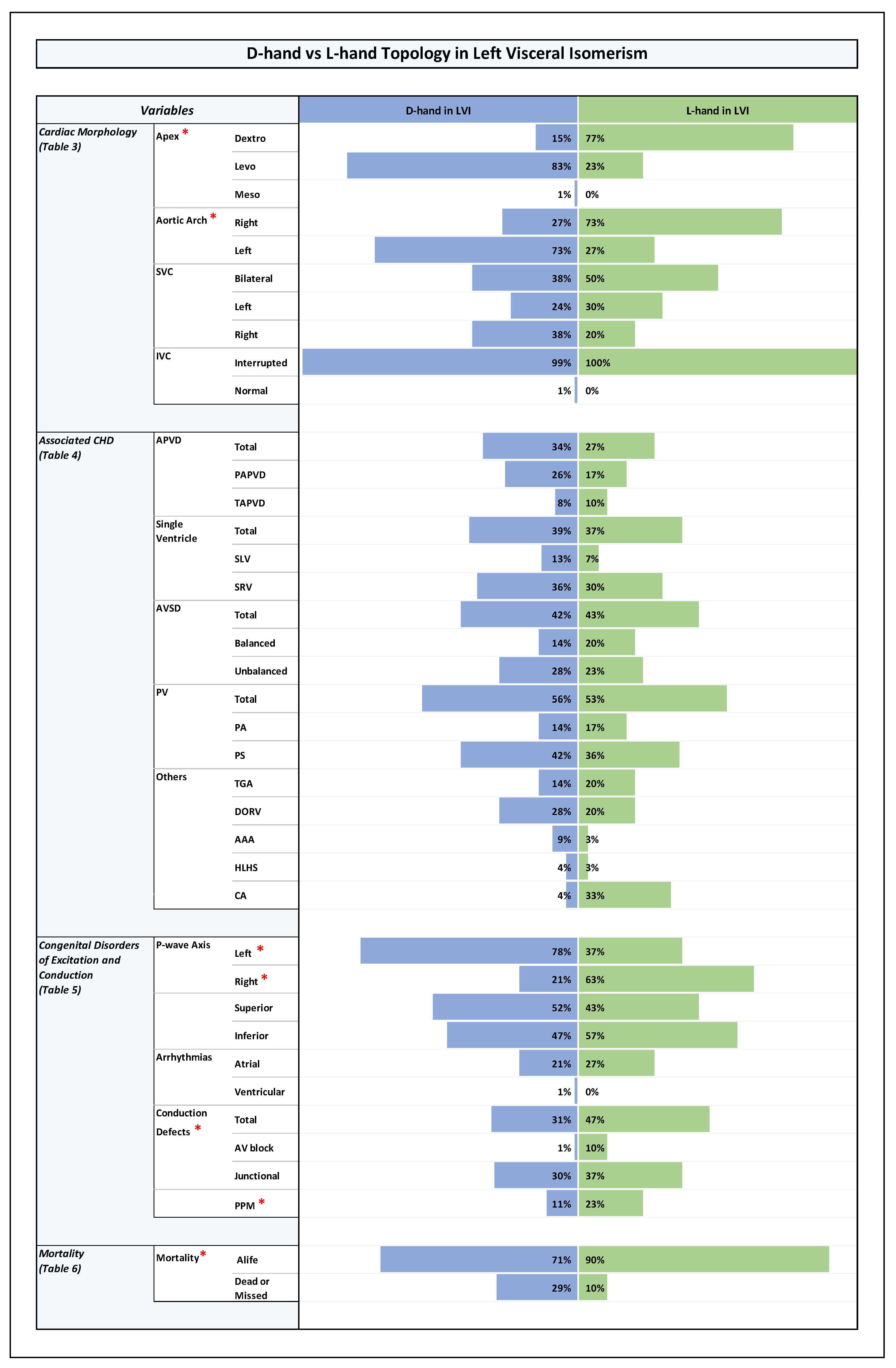
Orientation of the cardiac apex and patterning of aortic arch, SVC, and IVC (Plates 1-4 and Table 3 in ODS)
- Orientation of the cardiac apex:
- Aortic arch patterning (bilateral, unilateral right or unilateral left):
- Patterning of SVC (bilateral, unilateral right or unilateral left):
- Patterning of IVC (normal or interrupted):
Associated Congenital Heart Disease
- -
- Aortic arch abnormalities (coarctation, interruption, or hypoplasia)
- -
- Anomalies in pulmonary venous drainage (partially anomalous, totally anomalous)
- -
- Atrioventricular septal defect (balanced or unbalanced)
- -
- Univentricular heart (single ventricle) and in case of its presence, whether the existing single ventricle is of left or right ventricular morphology
- -
- Hypoplastic left heart syndrome
- -
- Common atrium
- -
- Transposition of the great arteries
- -
- Double outlet right ventricle
- -
- Anomalies of pulmonary valve (pulmonary stenosis, pulmonary atresia)
- (a)
- abnormal pulmonary venous drainage, mainly TAPVD (RVI: TAPVD (45%), PAPVD (9%); LVI: TAPVD (9%), PAPVD (23%); p-value 0.0001).
- (b)
- AVSD, mainly unbalanced AVSD (RVI: unbalanced AVSD (78%), balanced AVSD (10%); LVI: unbalanced AVSD (27%), balanced AVSD (16%); p-value 0.0001).
- (c)
- univentricular heart (single ventricle) pathologies (RVI: SLV (34%), SRV (48%); LVI: SLV (11%), SRV (27%); p-value 0.00001).
- (d)
- pulmonary stenosis or atresia (RVI: 93%; LVI:56%; p-value 0.0001).
- (e)
- TGA and DORV (RVI: TGA (38%), DORV (47%); LVI: TGA (16%), DORV (26%); p-value 0.001 and 0.003, respectively).
Congenital Disorders of Excitation and Conduction (Plates 1-4 and Table 5 in ODS)
- (1)
- RVI (23%) vs. LVI (23%), (p-value 0.17);
- (2)
- ventricular D-hand topology (23%) vs. L –hand topology (22 %) (p-value 0.42);
- (3)
- ventricular D-hand topology (27%) in RVI vs. L-hand topology in RVI (18%) (p-value 0.31);
- (4)
- ventricular D-hand topology (21%) in LVI vs. L-hand topology in LVI (27%) (p-value 0.71).
- Mortality and Long-Term Survival
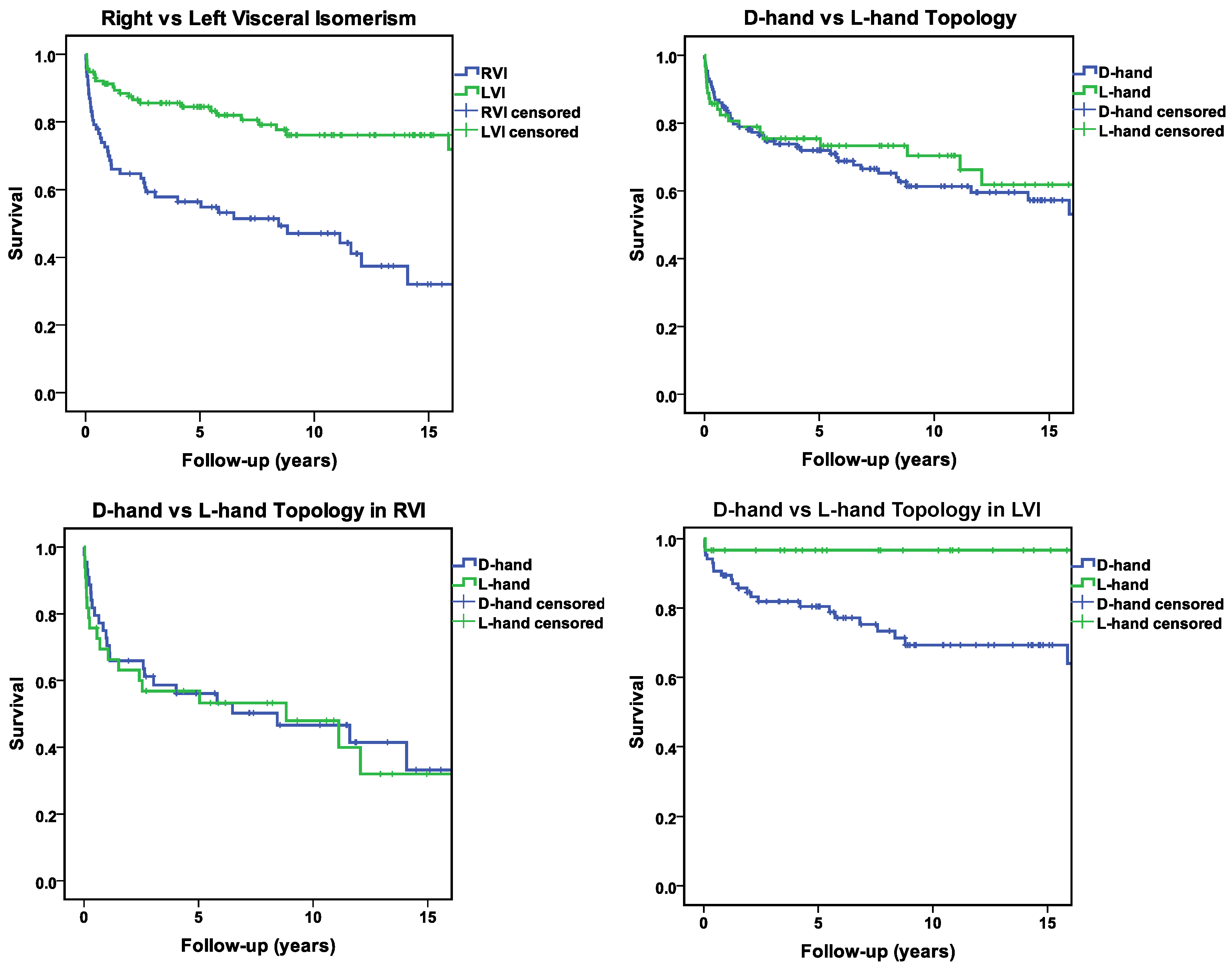
Discussion
Statistical Distribution Patterns of Ventricular D-Hand and L-Hand Topology
| RVI | LVI | |||||||
|---|---|---|---|---|---|---|---|---|
| Study | Case no. | D-hand | L-hand | Undetermined | Case no. | D-hand | L-hand | Undetermined |
| Stanger et al. [45] |
23 * | 34.8 % (8) | 65.2 % (15) | 17 * | 70.6 % (12) | 29.4 % (5) | ||
| Carvalho et al. [38] | 13 | 54 % (7) | 46 % (6) | 12 | 50 % (6) |
50 % (6) | ||
| Vairo et al. [39] | 28 | 53 % (15) | 47 % (13) | |||||
| Francalanci et al. [40] | 33 * | 60.6 % (20) | 39.4 % (13) | |||||
| Ho et al. [41] | 10 * | 60 % (6) | 40 % (4) | 20 * | 65 % (13) | 10 % (2) | 25 % (5) |
|
| Uemura et al. [8] | 125 * | 54 % (68) | 42 % (52) | 4 % (5) |
58 * | 79 % (46) | 16 % (9) | 5 % (3) |
| Smith et al. [11] | 10 * | 20 % (2) | 20 % (2) | 60 % (6) | 25 * | 60 % (15) | 32 % (8) | 8 % (2) |
| Yildirim et al. [66] | 43 | 53.5 % (23) | 18.6 % (8) | 27.9 % (12) | 88 | 59.1 % (52) | 22.7 % (20) | 18.2 % (16) |
| Loomba et al. [13] | 37 * | 50 % | 42 % | 8 % | 12 * | 47 % | 53 % | |
| Tremblay et al. [42] | 131 * | 61 % (80) | 36 % (47) | 3 % (4) |
56 * | 66 % (37) | 34 % (19) | |
| Kiram et al. [43] | 184 | 57.6 % (106) | 41.9 % (77) | 0.5 % (1) |
118 | 66.1 % (78) | 33.1 % (39) | 0.8 % (1) |
| Oreto et al. [44] | 43 | 60.5 % (26) | 39.5 % (17) | 35 | 57 % (20) | 43 % (15) | ||
| Pooled data | 652 | 55.4 % (361) |
39.9 % (260) |
4.7 % (31) |
469 | 63.8 % (299) |
30.5 % (143) |
5.7 % (27) |
Cardiovascular Anomalies in the Setting of Visceral Isomerism, Their Distribution Among RVI and LVI Subsets, and Associations with Ventricular D-Hand and L-Hand Topology
| RVI | LVI | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Study | Case no. |
Left arch |
Right arch | Double arch | Undet. | Case no. | Left arch |
Right arch |
Double arch | Undet. |
| Ho et al. [41] | 10 * | 50 % (5) | 50 % (5) | 20 * | 75 % (15) | 25 % (5) | ||||
| Francalanci et al. [40] | 33 * | 60.6 % (20) | 39.4 % (13) | |||||||
| Smith et al. [11] | 10 * | 90 % (9) | 10 % (1) | 25 * | 88 % (22) | 12 % (3) | ||||
| Loomba et al. [13] | 37 * | 70 % (26) |
30 % (11) |
12 * | 95 % (11) |
5 % (1) |
||||
| Tremblay et al. [42] | 131 * | 63 % (77) | 37 % (46) | 8 | 57 * | 75 % (41) | 25 % (15) | 1 | ||
| Kiram et al. [43] | 184 | 46.7 % (86) | 53.3 % (98) | 118 | 60 % (71) | 40 % (47) | ||||
| Oreto et al. [44] | 43 | 70 % (30) | 30 % (13) | 35 | 51 % (18) | 49 % (17) | ||||
| Pooled data | 448 | 56.5 % (253) |
41.7 % (187) |
1.8 % (8) |
267 | 66.6 % (178) |
33 % (88) |
0.4 % (1) |
Limitations
Conclusions
Supplementary Materials
Acknowledgments
References
- Ramsdell AF. Left–right asymmetry and congenital cardiac defects: getting to the heart of the matter in vertebrate left–right axis determination. Developmental Biology. 2005;288:1–20. [CrossRef]
- Nakamura T, Hamada H. Left-right patterning: conserved and divergent mechanisms. Development. 2012;139:3257–3262. [CrossRef]
- Männer J. The functional significance of cardiac looping: comparative embryology, anatomy, and physiology of the looped design of vertebrate hearts. Journal of Cardiovascular Development and Disease. 2024;11:252. [CrossRef]
- Ivemark BI. Implications of agenesis of the spleen on the pathogenesis of cono-truncus anomalies in childhood. Acta Paediatrica. 1955;44(Suppl. 104):7–113. [CrossRef]
- Moller JH, Nakib A, Anderson RC, Edwards JE. Congenital cardiac disease associated with polysplenia. A developmental complex of bilateral „left-sidedness“. Circulation. 1967;36:789–799. [CrossRef]
- Stanger P, Benassi RC, Korns ME, Jue KL, Edwards JE. Diagrammatic portrayal of variations in cardiac structure. Reference to transposition, dextrocardia and the concept of four normal hearts. Circulation. 1968;37(Suppl. 4):1–16. [CrossRef]
- Bartram U, Wirbelauer J, Speer CP. Heterotaxy syndrome–asplenia and polysplenia as indicators of visceral malposition and complex congenital heart disease. Neonatology. 2005;88:278–290. [CrossRef]
- Uemura H, Ho SY, Devine WA, Kilpatrick LL, Anderson RH. Atrial appendages and venoatrial connections in hearts with patients with visceral heterotaxy. The Annals of Thoracic Surgery. 1995;60:561–569. [CrossRef]
- De Tommasi S, Daliento L, Ho SY, Macartney FJ, Anderson RH. Analysis of atrioventricular junction, ventricular mass, and ventriculoarterial junction in 43 specimens with atrial isomerism. British Heart Journal. 1981;45:236–247. [CrossRef]
- Ho SY, Seo JW, Brown NA, Cook AC, Fagg NL, Anderson RH. Morphology of the sinus node in human and mouse hearts with isomerism of the atrial appendages. British Heart Journal. 1995;74:437–442. [CrossRef]
- Smith A, Ho SY, Anderson RH, Connell MG, Arnold R, Wilkinson JL, Cook AC. The diverse cardiac morphology seen in hearts with isomerism of the atrial appendages with reference to the disposition of the specialised conduction system. Cardiology in the Young. 2006;16:437–454. [CrossRef]
- Anderson RH, Brown NA, Meno C, Spicer DE. The importance of being isomeric. Clinical Anatomy. 2015;28:477–486. [CrossRef]
- Loomba RS, Ahmed MM, Spicer DE, Backer CL, Anderson RH. Manifestations of bodily isomerism. Cardiovascular Pathology. 2016;25:173–180. [CrossRef]
- Anderson RH, Spicer DE, Loomba RS. Is an appreciation of isomerism the key to unlocking the mysteries of cardiac findings in heterotaxy? Journal of Cardiovascular Development and Disease. 2018;5:11. [CrossRef]
- Ticho BS, Goldstein AM, Van Praagh R. Extracardiac anomalies in the heterotaxy syndromes with focus on anomalies of midline-associated structures. The American Journal of Cardiology. 2000;85:729–734. [CrossRef]
- Goldstein AM, Ticho BS, Fishman MC. Patterning the heart’s left-right axis: From zebrafish to man. Developmental Genetics. 1998;22:278–287.
- Lander A, King T, Brown NA. Left–right development: mammalian phenotypes and conceptual models. Seminars in Cell & Developmental Biology. 1998;9:35–41. [CrossRef]
- Anderson RH, Webb S, Brown NA. Defective lateralisation in children with congenitally malformed hearts. Cardiology in the Young. 1998;8:512–531. [CrossRef]
- Männer J. The anatomy of cardiac looping: a step towards the understanding of the morphogenesis of several forms of congenital malformations. Clinical Anatomy. 2009;22:21–35. [CrossRef]
- Moorman AFM, Soufan AT, Hagoort J, De Boer PAJ, Christoffels VM. Development of the building plan of the heart. Annals of the New York Academy of Sciences. 2004;1015:171–181. [CrossRef]
- Palmer AR. Symmetry breaking and the evolution of development. Science. 2004;306:828–833. [CrossRef]
- Inaki M, Liu J, Matsuno K. Cell chirality: its origin and roles in left–right asymmetric development. Philosophical Transactions of the Royal Society B: Biological Sciences. 2016;371(1710):20150403. [CrossRef]
- Rahman T, Zhang H, Fan J, Wan LQ. Cell chirality in cardiovascular development and disease. APL Bioengineering. 2020;4:031503. [CrossRef]
- Ekman G. Experimentelle Beiträge zur Herzentwicklung der Amphibien. Wilhelm Roux’s Archives of Developmental Biology. 1925;106:320–352. [CrossRef]
- Copenhaver WM. Experiments on the development of the heart of Amblystoma punctatum. Journal of Experimental Zoology. 1926;43:321–371. [CrossRef]
- Goerttler K. Die Bedeutung der ventrolateralen Mesodermbezirke für die Herzanlage der Amphibienkeime. Anatomischer Anzeiger. 1928;66(Suppl.):132–139.
- DeHaan R. Cardia bifida and the development of pacemaker function in the early chick heart. Developmental Biology. 1959;1:586–602. [CrossRef]
- Zwirner R, Kuhlo B. Die prospektive Potenz der rechten und der linken Herzanlage (ein experimenteller Beitrag zur Asymmetrie des Herzens). Wilhelm Roux’s Archives of Developmental Biology. 1964;155:511–524. [CrossRef]
- Nadal-Ginard B, Paz Garcia M. The morphologic expression of each cardiac primordium in the chick embryo. Journal of Embryology and Experimental Morphology. 1972;28:141–152. [CrossRef]
- Weiss PA. Entwicklungsphysiologie der Tiere. Steinkopff; 1930. [CrossRef]
- Taber, LA. Taber LA. Biophysical mechanisms of cardiac looping. International Journal of Developmental Biology. 2006;50:323–332. [CrossRef]
- Taber LA, Voronov DA, Ramasubramanian A. The role of mechanical forces in the torsional component of cardiac looping. Annals of the New York Academy of Sciences. 2010;1188:103–110. [CrossRef]
- Shi Y, Yao J, Young JM, Fee JA, Perucchio R, Taber LA. Bending and twisting the embryonic heart: a computational model for c-looping based on realistic geometry. Frontiers in Physiology. 2014;5:297. [CrossRef]
- Bayraktar M, Männer J. Cardiac looping may be driven by compressive loads resulting from unequal growth of the heart and pericardial cavity. Observations on a physical simulation model. Frontiers in Physiology. 2014;5:112. [CrossRef]
- Huhta JC, Smallhorn JF, MacCartney FJ. Two dimensional echocardiographic diagnosis of situs. British Heart Journal. 1982;48:97–108. [CrossRef]
- Jacobs JP, Anderson RH, Weinberg PM, Walters III HL, Tchervenkov CI, Del Duca D, Franklin RCG, Aiello VD, Beland MJ, Colan SD et al. The nomenclature, definition and classification of cardiac structures in the setting of heterotaxy. Cardiology in the Young. 2007;17(S4):1–28. [CrossRef]
- Al-Zahrani RS, Alharbi SH, Tuwaijri RM, Alzomaili BT, Althubaiti A, Yelbuz TM. Transposition of the great arteries: A laterality defect in the group of heterotaxy syndromes or an outflow tract malformation? Annals of Pediatric Cardiology. 2018;11:237–249. [CrossRef]
- Carvalho JS, Rigby ML, Shinebourne EA, Anderson RH. Cross sectional echocardiography for recognition of ventricular topology in atrioventricular septal defect. Heart. 1989;61:285–288. [CrossRef]
- Vairo U, Marino B, Parretti di Iulio D, Guccione P, Carotti A, Formigari R et al. Ventricular-infundibular morphology in visceral heterotaxia with left isomerism. An echocardiographic-angiocardiographic study. Giornale Italiano di Cardiologia. 1991;21:969–974.
- Francalanci P, Marino B, Boldrini R, Abella R, Iorio F, Bosman C. Morphology of the atrioventricular valve in asplenia syndrome: a peculiar type of atrioventricular canal defect. Cardiovascular Pathology. 1996;5:145–151. [CrossRef]
- Ho SY, Cook A, Anderson RH, Allan LD, Fagg N. Isomerism of the atrial appendages in the fetus. Pediatric Pathology. 1991;11:589–608. [CrossRef]
- Tremblay C, Loomba RS, Frommelt PC, Perrin D, Spicer DE, Backer C, Anderson RH. Segregating bodily isomerism or heterotaxy: potential echocardiographic correlations of morphological findings. Cardiology in the Young. 2017;27:1470–1480. [CrossRef]
- Kiram VS, Choudhary S, Shaik A, Gadabanahalli K, Raj V, Bhat V. Spectrum of cardiac anomalies associated with heterotaxy: a single-center study of a large series based on computed tomography. Pediatric Cardiology. 2020;41:1414–1424. [CrossRef]
- Oreto L, Mandraffino G, Ciliberti P, Santangelo TP, Romeo P, Celona A, Gitto P, Galetti L, Iorio FS, Di Pino A et al. Classifying anomalies in right and left isomerism: concordant and discordant patterns. Congenital Heart Disease. 2023;18:97–111. [CrossRef]
- Stanger P, Abraham MR, Edwards JE. Cardiac malpositions. An overview based on sixty-five necropsy specimens. Circulation. 1977;56:159–172.
- Berg C, Geipel A, Kamil D, Knüppel M, Breuer J, Krapp M, Baschat A, Germer U, Hansmann M, Gembruch U et al. The syndrome of left atrial isomerism. Sonographic findings and outcome in prenatally diagnosed cases. Journal of Ultrasound in Medicine. 2005;24:921–931. [CrossRef]
- Buca DIP, Khalil A, Rizzo G, Familiari A, Di Giovanni S, Liberati M, Murgano D, Ricciardulli A, Fanfani F, Scambia G et al. Outcome of prenatally diagnosed fetal heterotaxy: systematic review and meta-analysis. Ultrasound in Obstetrics & Gynecology. 2018;51:323–330. [CrossRef]
- Deng H, Xia H, Deng S. Genetic basis of human left-right asymmetry disorders. Expert Reviews in Molecular Medicine. 2014;16:e19. [CrossRef]
- Icardo JM, Sanchez de Vega MJ. Spectrum of heart malformations in mice with situs solitus, situs inversus, and associated visceral heterotaxy. Circulation. 1991;84:2547–2558. [CrossRef]
- Weininger WJ, Lopes Floro K, Bennett MB, Withington SL, Preis JI, Barbera JP, Mohun TJ, Dunwoodie SL. Cited2 is required both for heart morphogenesis and establishment of the left-right axis in mouse development. Development. 2005;132:1337–1348. [CrossRef]
- Bamforth SD, Braganca J, Farthing CR, Schneider JE, Broadbent C, Michell AC, Clarke K, Neubauer S, Norris D, Brown N et al. Cited2 controls left-right patterning and heart development through a Nodal-Pitx2c pathway. Nature Genetics. 2004;11:1189–1196. [CrossRef]
- Meno C, Shimono A, Saijoh Y, Yashiro K, Mochida K, Ohishi S, Noji S, Kondho H, Hamada H. Lefty-1 is required for left-right determination as a regulator of lefty-2 and nodal. Cell. 1998;94:287–297. [CrossRef]
- Tsukui T, Capdevila J, Tamura K, Ruiz-Lozano P, Rodriguez-Esteban C, Yonei-Tamura S, Magallon J, Chandraratna RA, Chien K, Blumberg B et al. Multiple left-right asymmetry defects in Shh (-/-) mice unveil a convergence of the Shh and retinoic acid pathways in the control of Lefty-1. Proceedings of the National Academy of Sciences of the United States of America. 1999;96:11356–11381.
- Hildreth V, Webb S, Chaudry B, Peat JD, Phillips HM, Brown N, Anderson RH, Henderson DJ. Left cardiac isomerism in the sonic hedgehog null mouse. Journal of Anatomy. 2009;214:894–904. [CrossRef]
- Lin CR, Kioussi C, O’Cannell S, Briata P, Szeto D, Liu F, Izpisua-Belmonte JC, Rosenfeld MG. Pitx2 regulated lung asymmetry, cardiac positioning and pituitary and tooth morphogenesis. Nature. 1999;401:279–282. [CrossRef]
- Green JM, Chiaramida AJ. 12-lead EKG Confidence: A Step-by-step Guide. Springer Publishing Company; 2014. 115.
- Congdon ED. Transformation of the aortic arch system during development of the human embryo. Contributions to Embryology. 1922;68:49–110.
- Rana MS, Sizarov A, Christoffels VM, Moorman AFM. Development of the human aortic arch system captured in an interactive three-dimensional reference model. American Journal of Medical Genetics Part A. 2014;164:1372–1383. [CrossRef]
- Anderson RH, Bamforth SD. Morphogenesis of the mammalian aortic arch arteries. Frontiers in Cell and Developmental Biology. 2022;10:892900. [CrossRef]
- Loomba RS, Hlavacek AM, Spicer DE, Anderson RH. Isomerism or heterotaxy: which term leads to better understanding. Cardiology in the Young. 2015;25:1037–1043. [CrossRef]
- Yashihiro K, Shiratori H, Hamada H. Haemodynamics determined by a genetic program govern asymmetric development of the aortic arch. Nature. 2007;450:285–288. [CrossRef]
- Hastreiter AR, D’Cruz IA, Cantez T. Right-sided aortic arch. Part I: Occurrence of right aortic arch in various types of congenital heart disease. British Heart Journal. 1966;28:722–725. [CrossRef]
- Rose V, Izukawa T, Moes CAF. Syndromes of asplenia and polysplenia. A review of cardiac and non-cardiac malformations in 60 cases with special reference to diagnosis and prognosis. British Heart Journal. 1975;37:840–852. [CrossRef]
- Peoples WM, Moller JH, Edwards JE. Polysplenia: a review of 146 cases. Pediatric Cardiology. 1983;4:129–137. [CrossRef]
- Kowalski WJ, Teslovich NC, Dur O, Keller BB, Pekkan K. Computational hemodynamics optimization predicts dominant aortic arch selection is driven by embryonic outflow tract orientation in the chick embryo. Biomechanics and Modeling in Mechanobiology. 2012;11:1057–1073. [CrossRef]
- Yildirim SV, Tokel K, Varan B, Aslamaci S, Ekici E. Clinical investigations over 13 years to establish the nature of the cardiac defects in patients having abnormalities of lateralization. Cardiology in the Young. 2007;17:275–282. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




