Submitted:
23 June 2025
Posted:
24 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
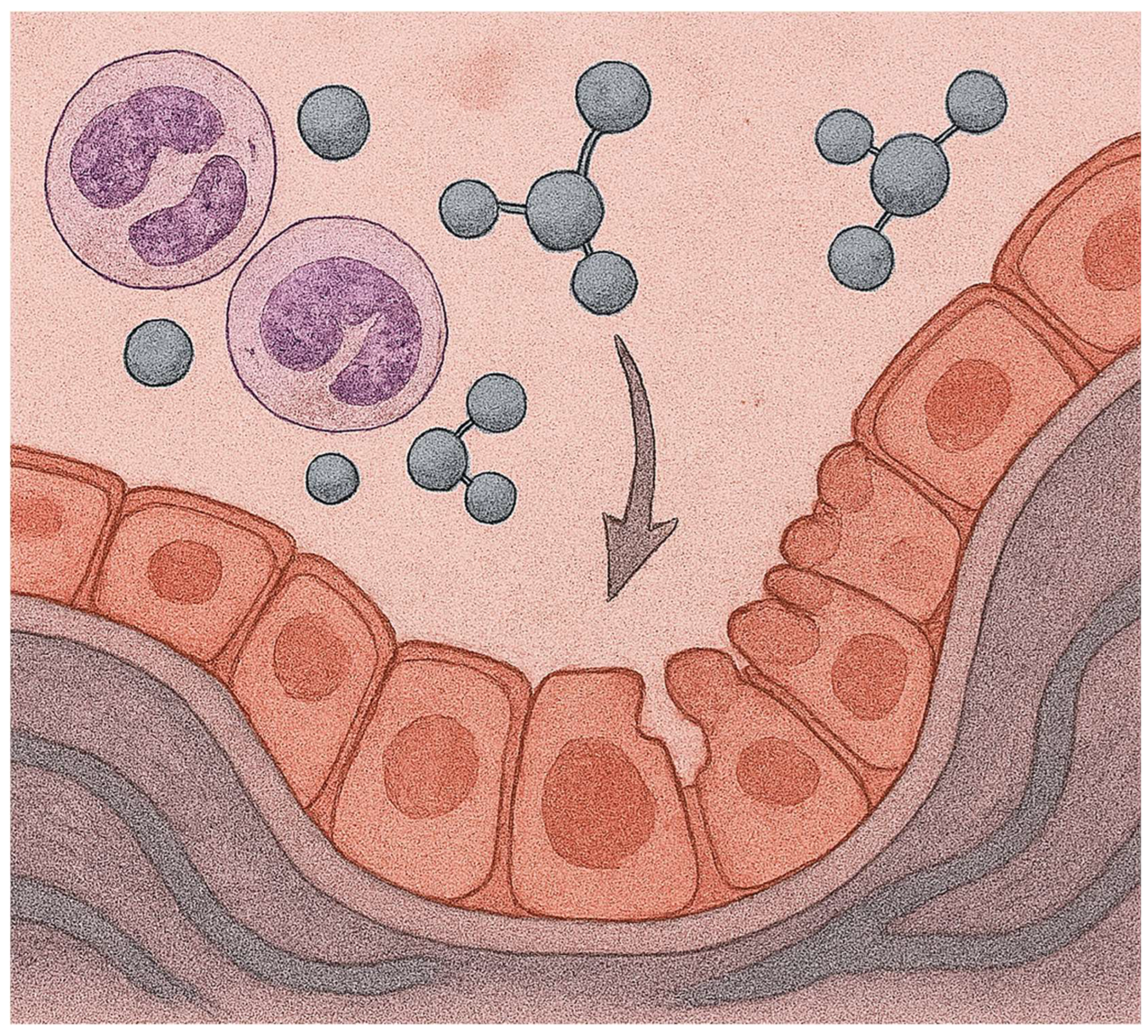
2. LPS Model of Acute Lung Injury
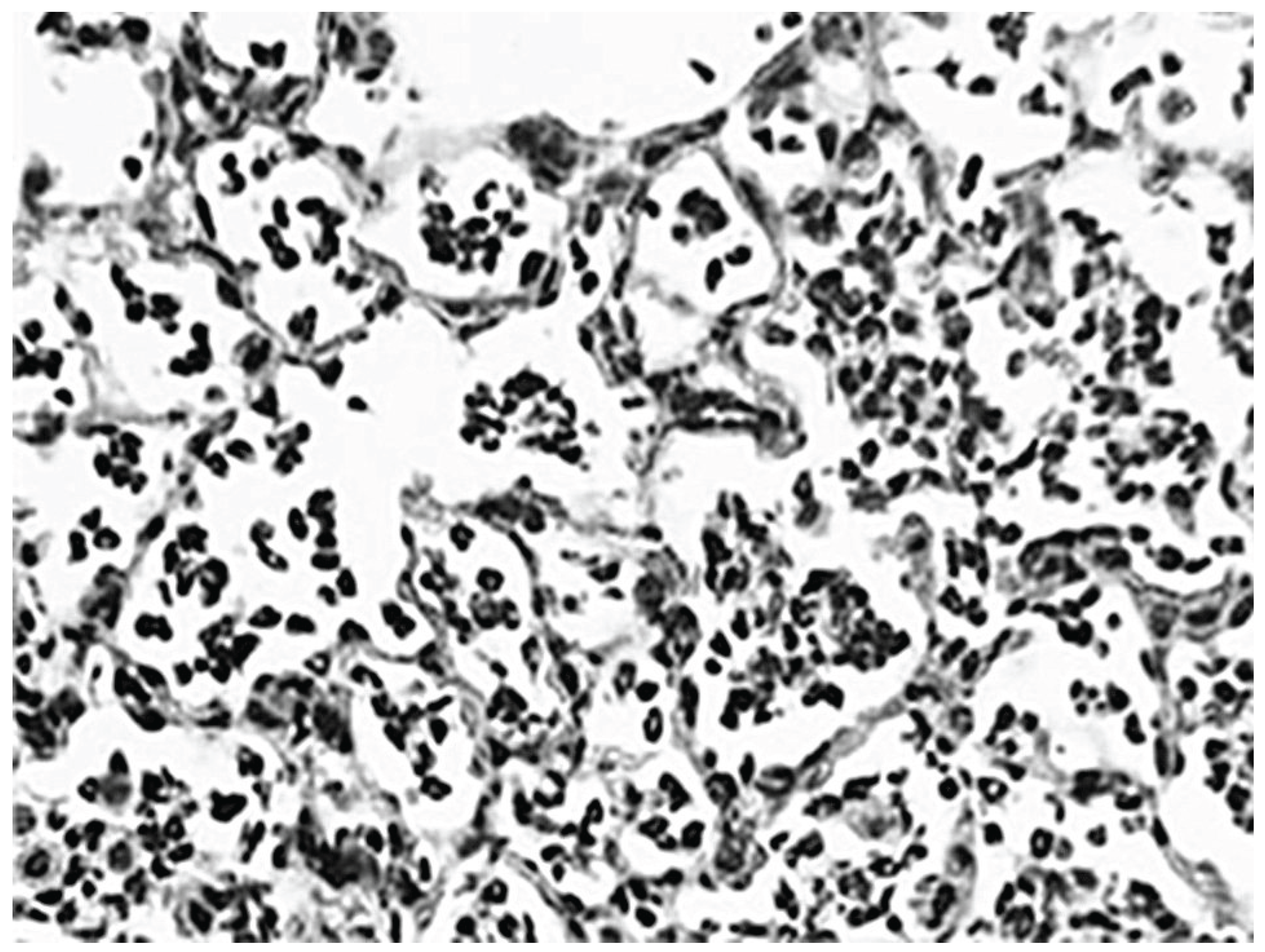
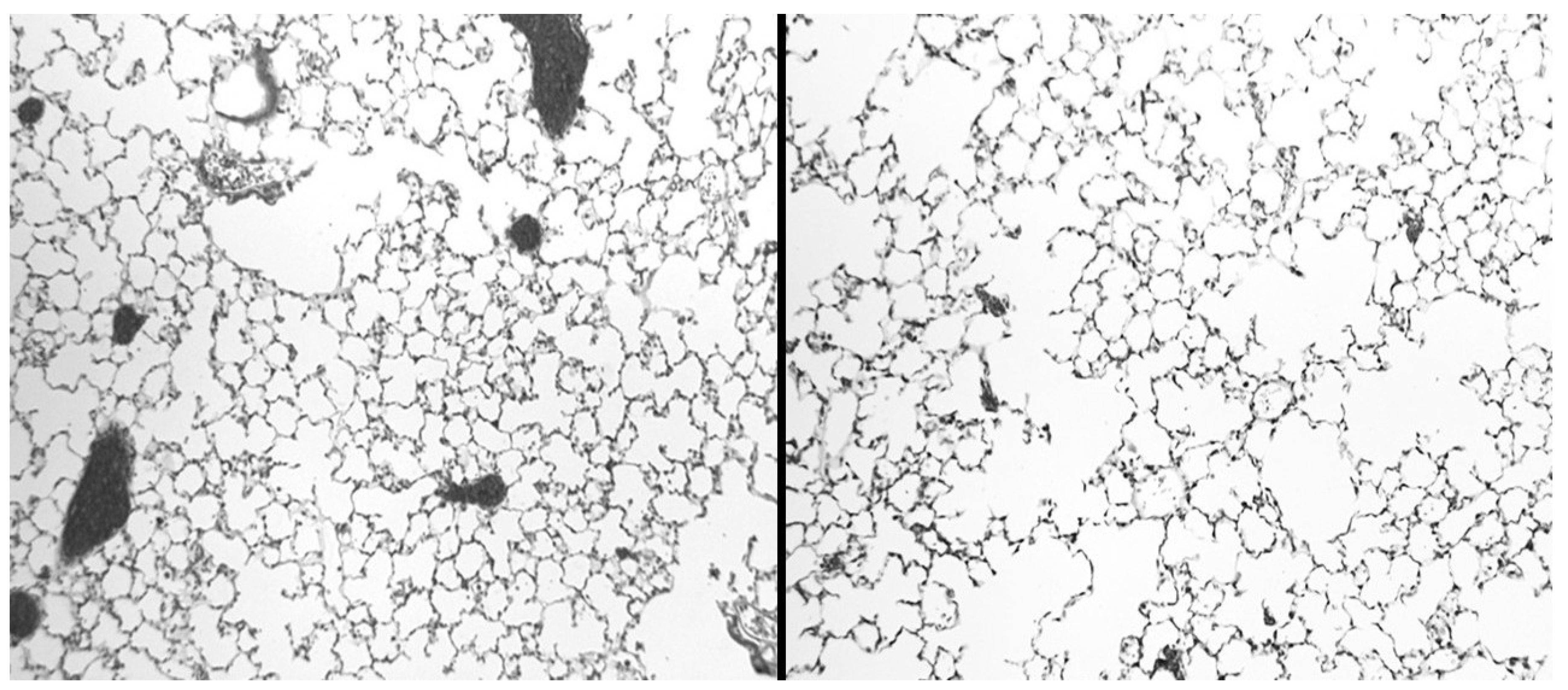
2.1. Biochemical and Morphological Features
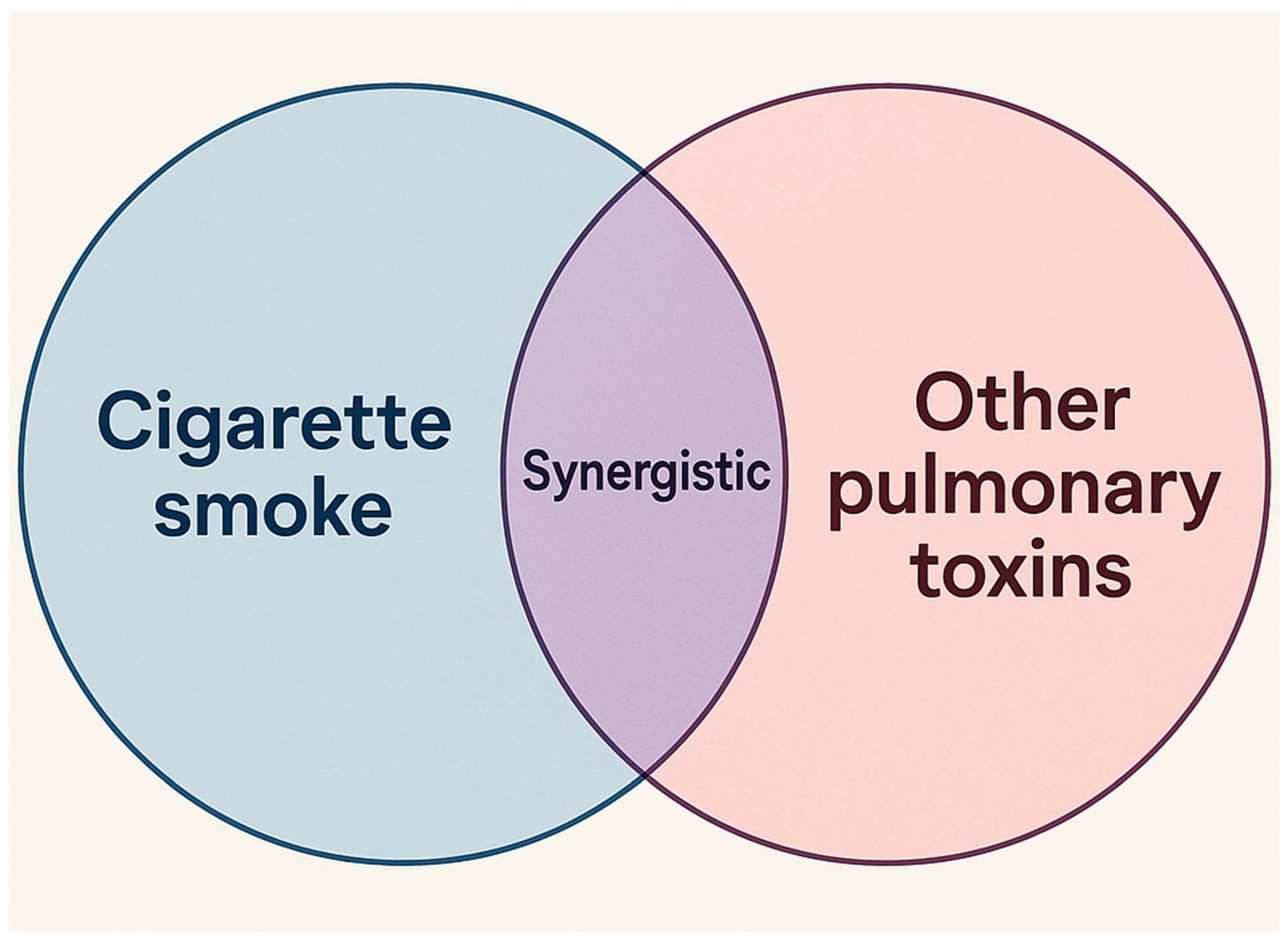
2.2. Synergistic Effect Between LPS and Cigarette Smoke
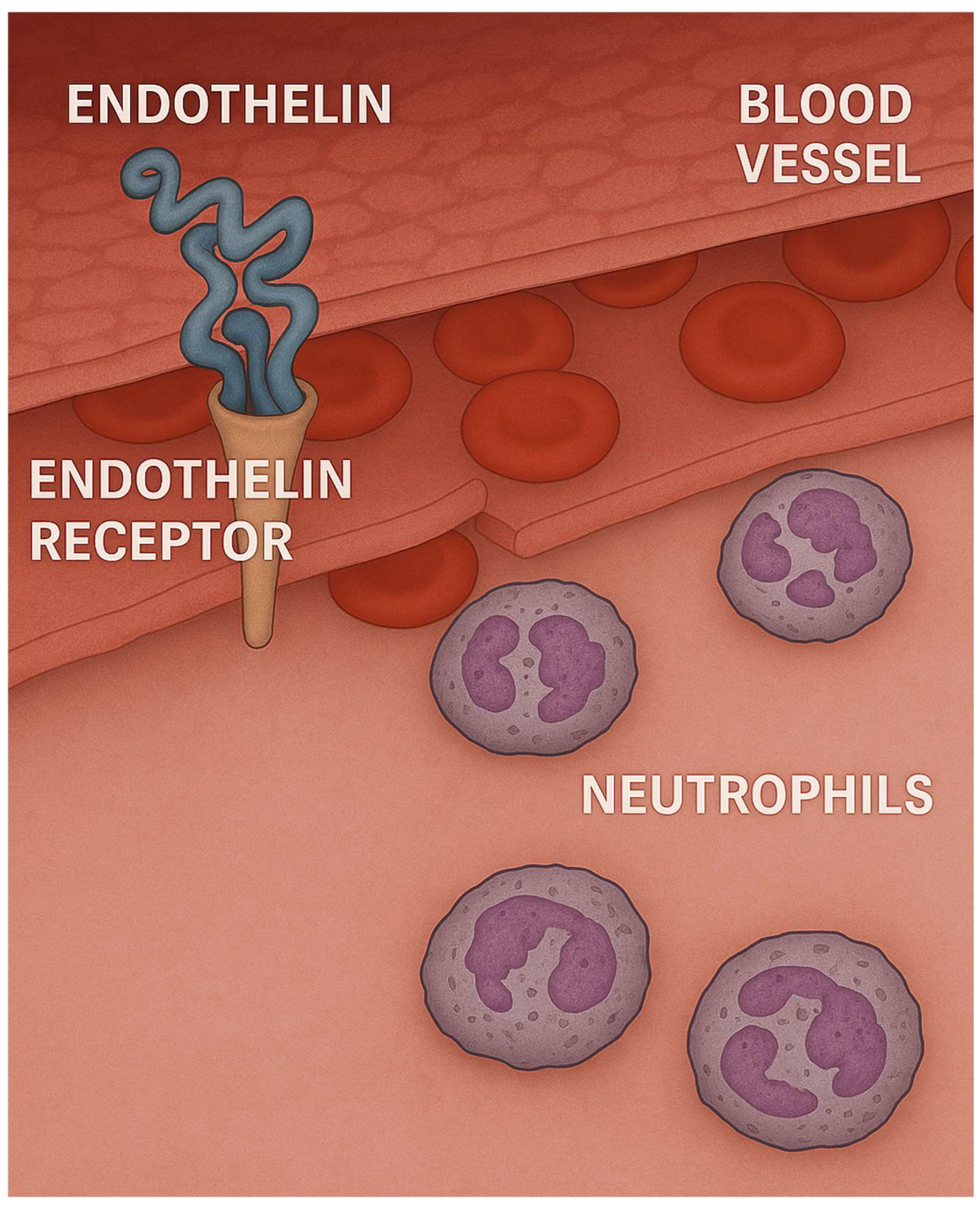
2.3. Role of Endothelin in LPS-Induced Lung Injury
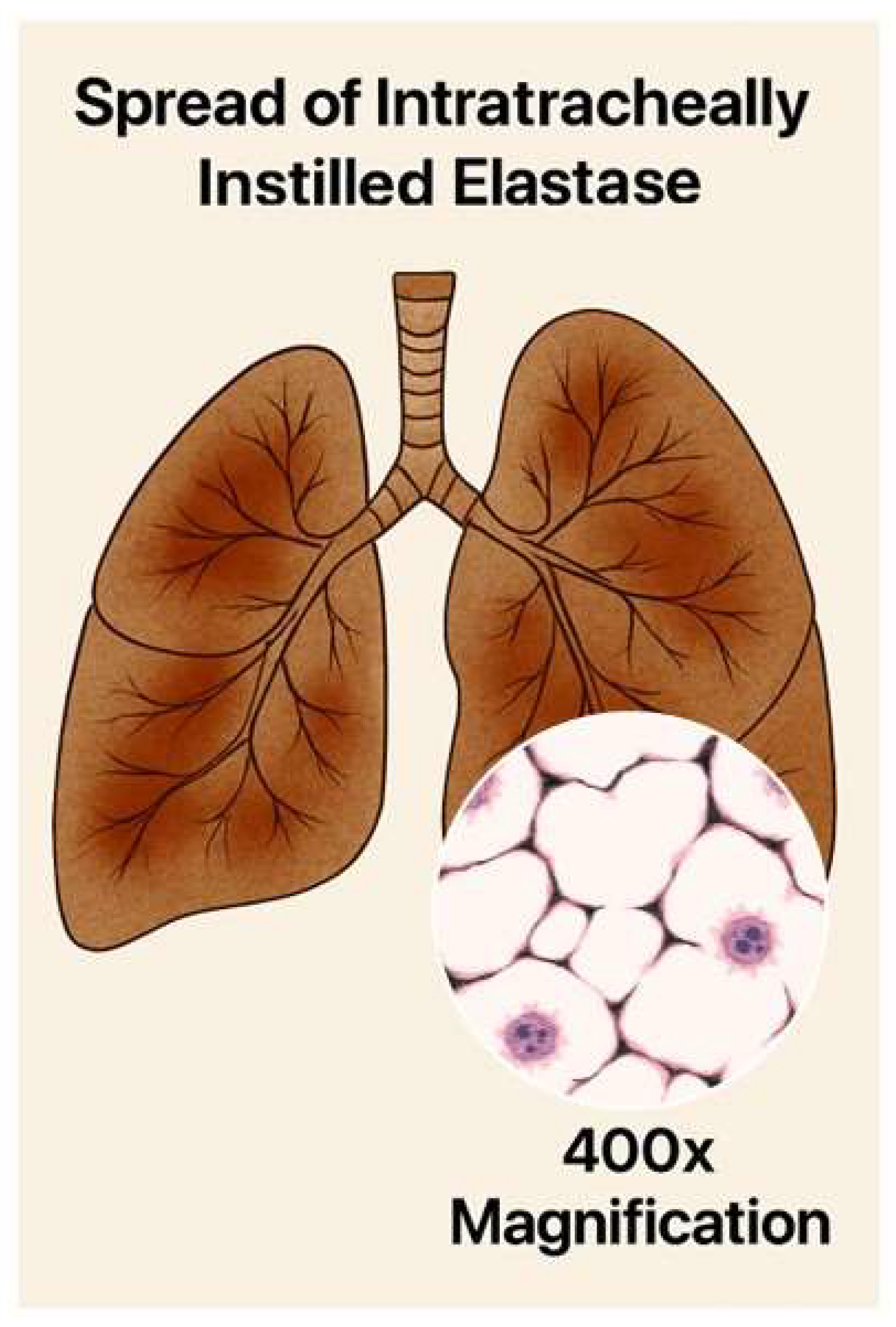
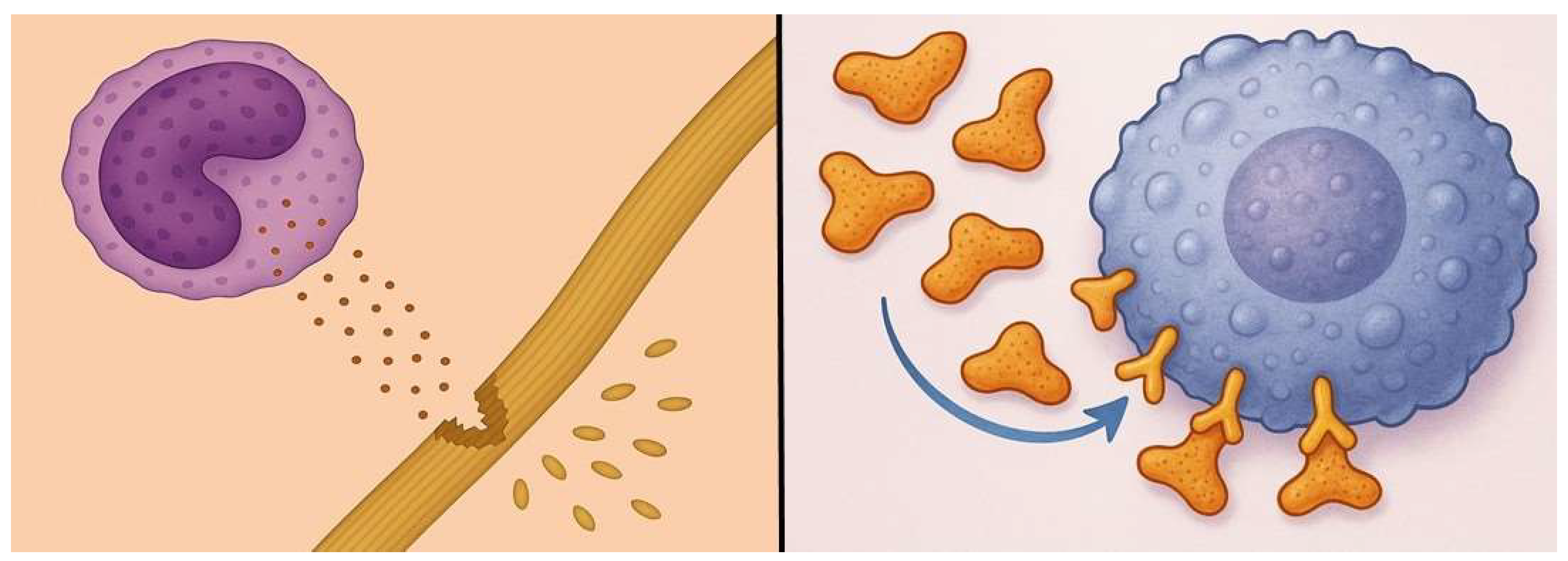
3. Elastase-Induced Pulmonary Emphysema
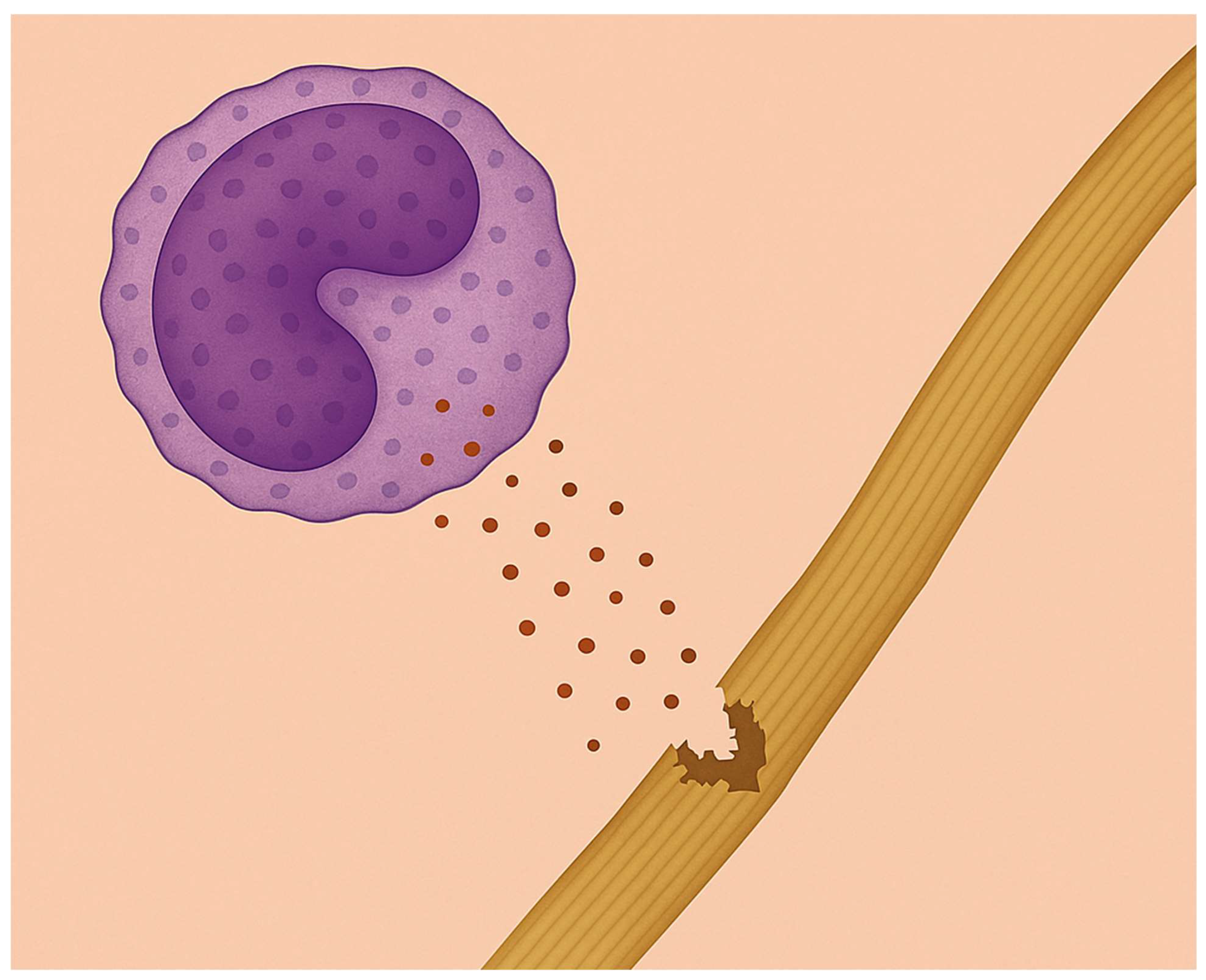
3.1. Biochemical and Morphological Features
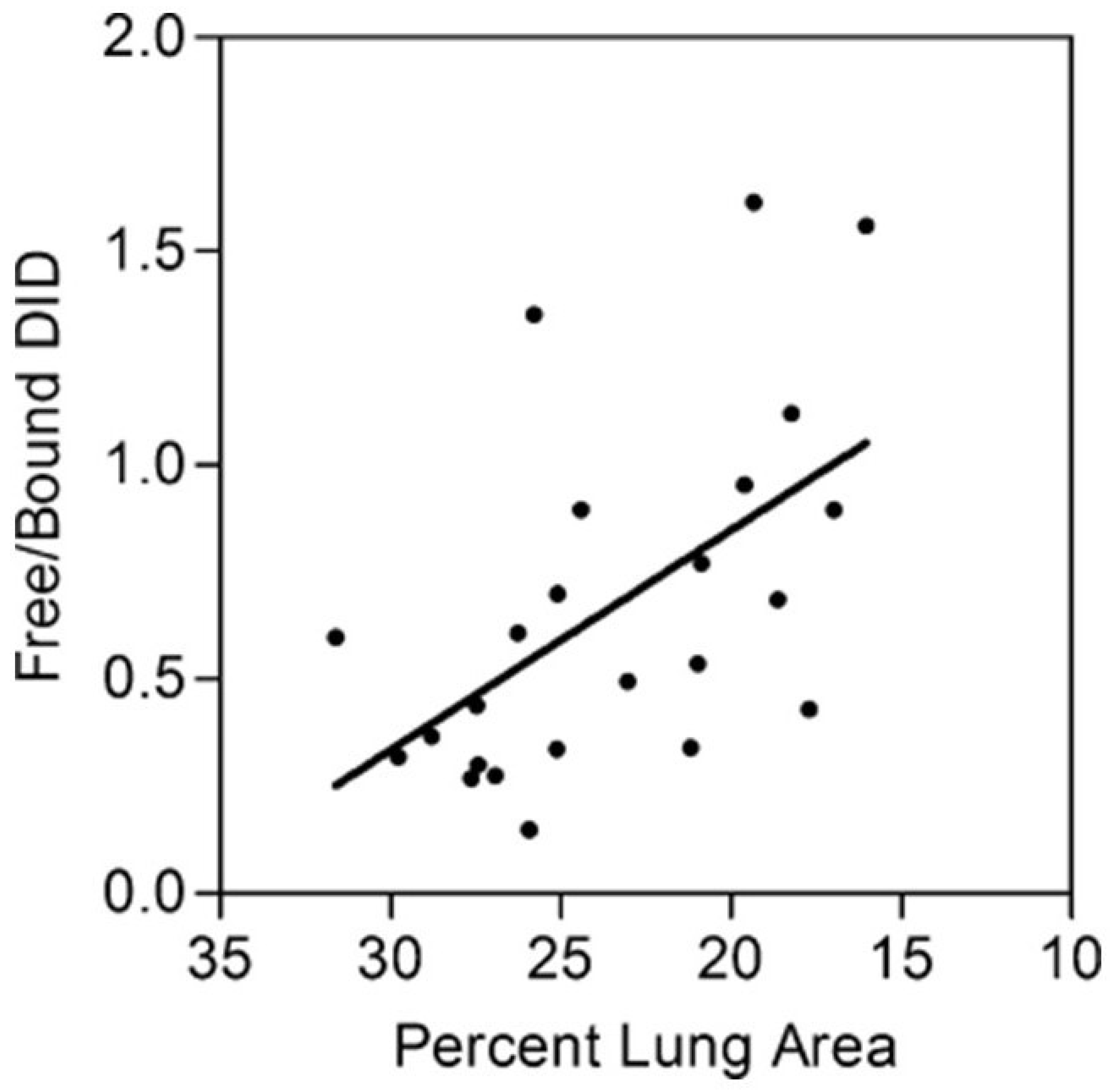
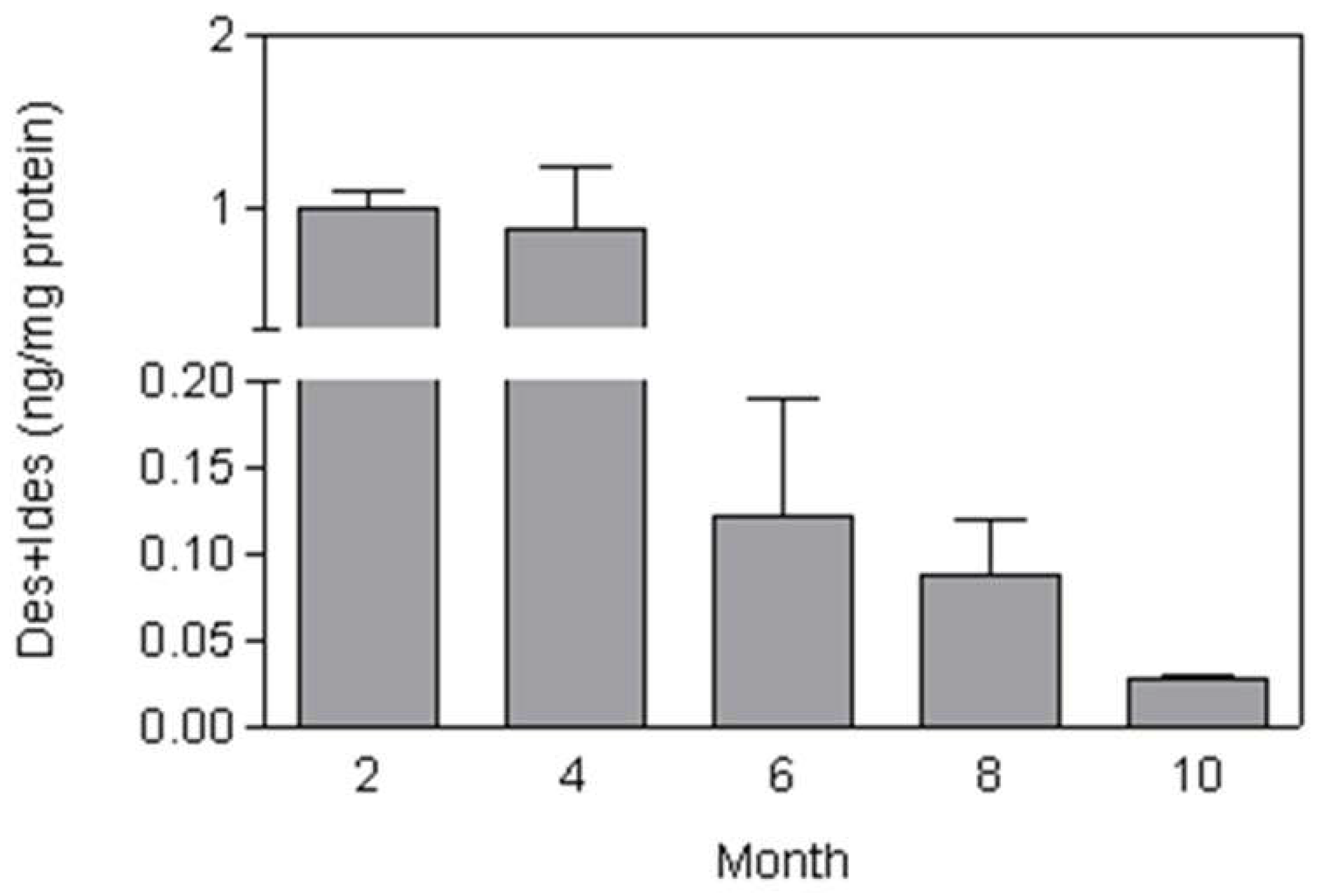
3.2. The Ratio of Free to Total Desmosine as a Biomarker for Pulmonary Emphysema
3.3. Combining Elastase-Induced Emphysema with LPS
4. Cigarette Smoke-Induced Pulmonary Emphysema
4.1. Biochemical and Morphological Features
4.2. The Effect of Aerosolized Hyaluronan on Airspace Enlargement
5. Bleomycin Model of Pulmonary Fibrosis
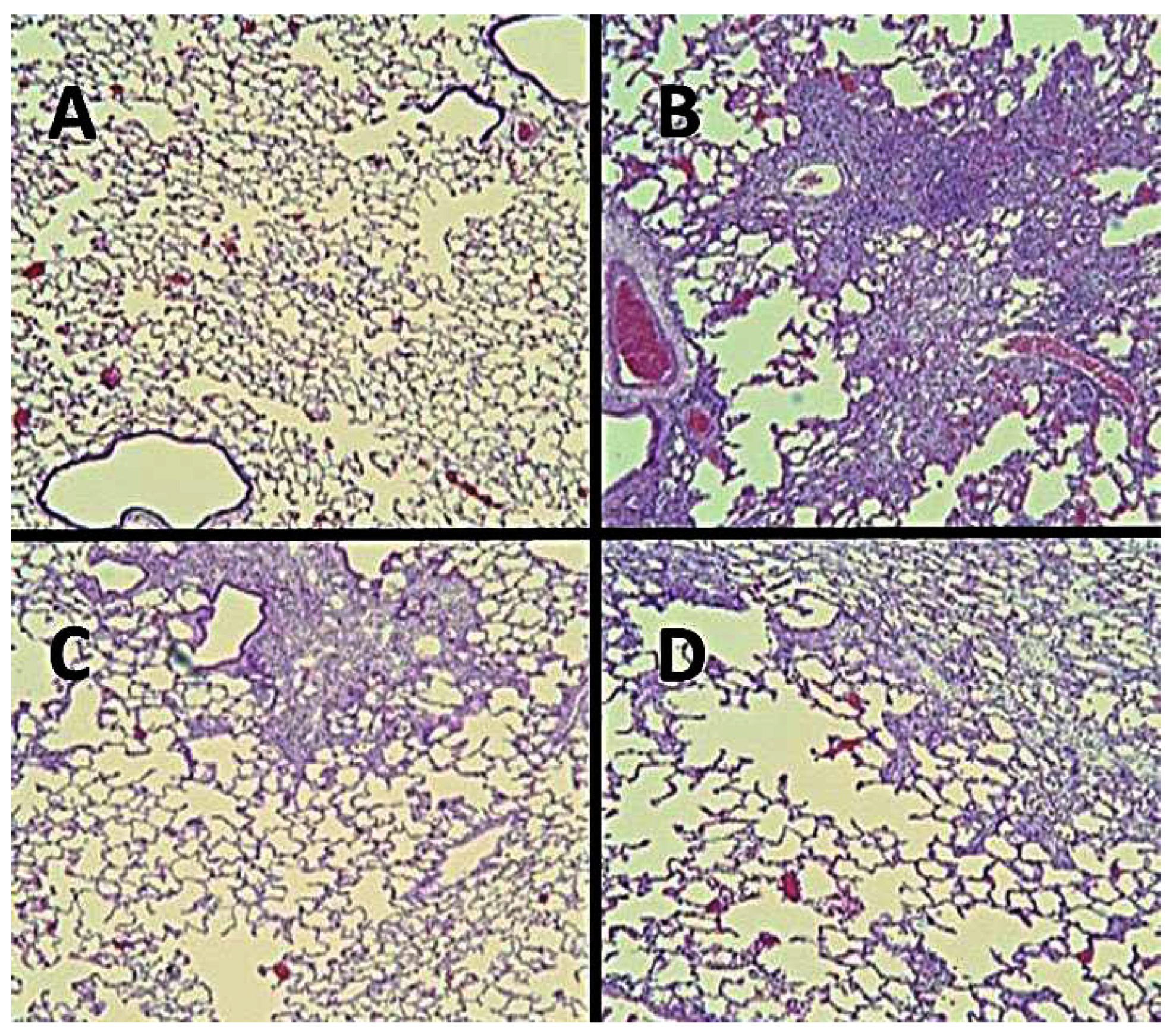
5.1. Biochemical and Morphological Features
5.2. The Effect of Endothelin Inhibition
6. Chemical Versus Genetic Models
7. Strategies for Addressing the Limitations of Animal Models
8. Conclusions
References
- Churg, A.; Cosio, M.; Wright, J.L. Mechanisms of cigarette smoke-induced COPD: insights from animal models. American Journal of Physiology-Lung Cellular and Molecular Physiology 2008, 294(4), L612–L631. [Google Scholar] [CrossRef] [PubMed]
- Upadhyay, P.; Wu, C.W.; Pham, A.; Zeki, A.A.; Royer, C.M.; Kodavanti, U.P.; Takeuchi, M.; Bayram, H.; Pinkerton, K.E. Animal models and mechanisms of tobacco smoke-induced chronic obstructive pulmonary disease (COPD). Journal of Toxicology and Environmental Health, Part B 2023, 26(5), 275–305. [Google Scholar] [CrossRef]
- Wright, J.L.; Churg, A. Animal models of cigarette smoke-induced chronic obstructive pulmonary disease. Expert Review of Respiratory Medicine 2010, 4(6), 723–734. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Xu, F.; Lin, Y. Cigarette smoke synergizes lipopolysaccharide-induced interieukin-1β and tumor necrosis factor–α secretion from macrophages via substance P–mediated nuclear factor–κB activation. American Journal of Respiratory Cell and Molecular Biology 2011, 44(3), 302–308. [Google Scholar] [CrossRef]
- Cantor, J. The Potential Role of Cigarette Smoke, Elastic Fibers, and Secondary Lung Injury in the Transition of Pulmonary Emphysema to Combined Pulmonary Fibrosis and Emphysema. International Journal of Molecular Sciences 2024, 25(21), 11793. [Google Scholar] [CrossRef] [PubMed]
- Nair, A.; Morsy, M.A.; Jacob, S. Dose translation between laboratory animals and human in preclinical and clinical phases of drug development. Drug development research 2018, 79(8), 373–382. [Google Scholar] [CrossRef]
- Singh, G. Preclinical Drug Development. In Pharmaceutical Medicine and Translational Clinical Research; Academic Press, 2018; pp. 47–63. [Google Scholar]
- Iadarola, P.; Luisetti, M. The role of desmosines as biomarkers for chronic obstructive pulmonary disease. Expert Review of Respiratory Medicine 2013, 7(2), 137–144. [Google Scholar] [CrossRef]
- Cantor, J. Desmosine: The Rationale for Its Use as a Biomarker of Therapeutic Efficacy in the Treatment of Pulmonary Emphysema. Diagnostics 2025, 15(5), 578. [Google Scholar] [CrossRef]
- Dushianthan, A.; Grocott, M.P.W.; Postle, A.D.; Cusack, R. Acute respiratory distress syndrome and acute lung injury. Postgraduate medical journal 2011, 87(1031), 612–622. [Google Scholar] [CrossRef]
- Chen, H.; Bai, C.; Wang, X. The value of the lipopolysaccharide-induced acute lung injury model in respiratory medicine. Expert review of respiratory medicine 2010, 4(6), 773–783. [Google Scholar] [CrossRef]
- Marrocco, A.; Ortiz, L.A. Role of metabolic reprogramming in proinflammatory cytokine secretion from LPS or silica-activated macrophages. Frontiers in immunology 2022, 13, 936167. [Google Scholar] [CrossRef] [PubMed]
- Alhayaza, R.; Haque, E.; Karbasiafshar, C.; Sellke, F.W.; Abid, M.R. The relationship between reactive oxygen species and endothelial cell metabolism. Frontiers in chemistry 2020, 8, 592688. [Google Scholar] [CrossRef] [PubMed]
- Domscheit, H.; Hegeman, M.A.; Carvalho, N.; Spieth, P.M. Molecular dynamics of lipopolysaccharide-induced lung injury in rodents. Frontiers in physiology 2020, 11, 36. [Google Scholar] [CrossRef]
- Chimenti, L.; Morales-Quinteros, L.; Puig, F.; Camprubi-Rimblas, M.; Guillamat-Prats, R.; Gómez, M.N.; Tijero, J.; Blanch, L.; Matute-Bello, G.; Artigas, A. Comparison of direct and indirect models of early induced acute lung injury. Intensive Care Medicine Experimental 2020, 8, 1–13. [Google Scholar] [CrossRef]
- Matute-Bello, G.; Frevert, C.W.; Martin, T.R. Animal models of acute lung injury. American Journal of Physiology-Lung Cellular and Molecular Physiology 2008, 295(3), L379–L399. [Google Scholar] [CrossRef]
- Nguyen, N.; Xu, S.; Lam, T.Y.W.; Liao, W.; Wong, W.F.; Ge, R. ISM1 suppresses LPS-induced acute lung injury and post-injury lung fibrosis in mice. Molecular Medicine 2022, 28(1), 72. [Google Scholar] [CrossRef] [PubMed]
- van der Vaart, H.; Postma, D.S.; Timens, W.; Ten Hacken, N.H. Acute effects of cigarette smoke on inflammation and oxidative stress: a review. Thorax 2004, 59(8), 713–721. [Google Scholar] [CrossRef]
- Dauchet, L.; Hulo, S.; Cherot-Kornobis, N.; Matran, R.; Amouyel, P.; Edmé, J.L.; Giovannelli, J. Short-term exposure to air pollution: associations with lung function and inflammatory markers in non-smoking, healthy adults. Environment international 2018, 121, 610–619. [Google Scholar] [CrossRef]
- Alexander, L.E.C.; Shin, S.; Hwang, J.H. Inflammatory diseases of the lung induced by conventional cigarette smoke: a review. Chest 2015, 148(5), 1307–1322. [Google Scholar] [CrossRef]
- Lin, L.; Li, J.; Song, Q.; Cheng, W.; Chen, P. The role of HMGB1/RAGE/TLR4 signaling pathways in cigarette smoke-induced inflammation in chronic obstructive pulmonary disease. Immunity, inflammation and disease 2022, 10(11), e711. [Google Scholar] [CrossRef]
- Lugg, S.T.; Scott, A.; Parekh, D.; Naidu, B.; Thickett, D.R. Cigarette smoke exposure and alveolar macrophages: mechanisms for lung disease. Thorax 2022, 77(1), 94–101. [Google Scholar] [CrossRef] [PubMed]
- Seo, Y.S.; Park, J.M.; Kim, J.H.; Lee, M.Y. Cigarette smoke-induced reactive oxygen species formation: a concise review. Antioxidants 2023, 12(9), 1732. [Google Scholar] [CrossRef] [PubMed]
- Strzelak, A.; Ratajczak, A.; Adamiec, A.; Feleszko, W. Tobacco smoke induces and alters immune responses in the lung triggering inflammation, allergy, asthma and other lung diseases: a mechanistic review. International journal of environmental research and public health 2018, 15(5), 1033. [Google Scholar] [CrossRef]
- Kulkarni, G.S.; Nadkarni, P.P.; Cerreta, J.M.; Ma, S.; Cantor, J.O. Short-term cigarette smoke exposure potentiates endotoxin-induced pulmonary inflammation. Exp Lung Res. 2007, 33(1), 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bhalla, D.K.; Hirata, F.; Rishi, A.K.; Gairola, C.G. Cigarette smoke, inflammation, and lung injury: a mechanistic perspective. Journal of Toxicology and Environmental Health, Part B 2009, 12(1), 45–64. [Google Scholar] [CrossRef]
- Fabricio, A.S.; Rae, G.A.; D’Orléans-Juste, P.; Souza, G.E. Endothelin-1 as a central mediator of LPS-induced fever in rats. Brain research 2005, 1066(1–2), 92–100. [Google Scholar]
- Kowalczyk, A.; Kleniewska, P.; Kolodziejczyk, M.; Skibska, B.; Goraca, A. The role of endothelin-1 and endothelin receptor antagonists in inflammatory response and sepsis. Archivum immunologiae et therapiae experimentalis 2015, 63, 41–52. [Google Scholar] [CrossRef]
- Freeman, B.D.; Machado, F.S.; Tanowitz, H.B.; Desruisseaux, M.S. Endothelin-1 and its role in the pathogenesis of infectious diseases. Life sciences 2014, 118(2), 110–119. [Google Scholar] [CrossRef]
- Zarpelon, A.C.; Pinto, L.G.; Cunha, T.M. Endothelin-1 induces neutrophil recruitment in adaptive inflammation via TNFα and CXCL1/CXCR2 in mice. Can. J. Physiol. Pharmacol. 2012. Retrieved from https://cdnsciencepub.com/doi/abs/10.1139/y11-116. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, T.; Liu, X.J.; Patel, H.; Stephani, R.; Cantor, J.O. Preferential recruitment of neutrophils by endothelin-1 in acute lung inflammation induced by lipopolysaccharide or cigarette smoke. Int J Chron Obstruct Pulmon Dis. 2008, 3(3), 477–81. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bhavsar, T.M.; Cerreta, J.M.; Liu, M.; Reznik, S.E.; Cantor, J.O. Phosphoramidon, an endothelin-converting enzyme inhibitor, attenuates lipopolysaccharide-induced acute lung injury. Experimental Lung Research 2008, 34(3), 141–154. [Google Scholar] [CrossRef]
- Cantor, J. The role of the extracellular matrix in the pathogenesis and treatment of pulmonary emphysema. International Journal of Molecular Sciences 2024, 25(19), 10613. [Google Scholar] [CrossRef]
- Voynow, J.A.; Shinbashi, M. Neutrophil elastase and chronic lung disease. Biomolecules 2021, 11(8), 1065. [Google Scholar] [CrossRef]
- Pandey, K.C.; De, S.; Mishra, P.K. Role of proteases in chronic obstructive pulmonary disease. Frontiers in pharmacology 2017, 8, 512. [Google Scholar] [CrossRef] [PubMed]
- Fagiola, M.; Reznik, S.; Riaz, M.; Qyang, Y.; Lee, S.; Avella, J.; Turino, G.; Cantor, J. The relationship between elastin cross linking and alveolar wall rupture in human pulmonary emphysema. American Journal of Physiology-Lung Cellular and Molecular Physiology 2023, 324(6), L747–L755. [Google Scholar] [CrossRef] [PubMed]
- Kozel, B.A.; Mecham, R.P. Elastic fiber ultrastructure and assembly. Matrix Biology 2019, 84, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Trębacz, H.; Barzycka, A. Mechanical properties and functions of elastin: an overview. Biomolecules 2023, 13(3), 574. [Google Scholar] [CrossRef]
- Roan, E.; Waters, C.M. What do we know about mechanical strain in lung alveoli? American Journal of Physiology-Lung Cellular and Molecular Physiology 2011, 301(5), L625–L635. [Google Scholar] [CrossRef]
- Knudsen, L.; Ochs, M. The micromechanics of lung alveoli: structure and function of surfactant and tissue components. Histochemistry and cell biology 2018, 150, 661–676. [Google Scholar] [CrossRef]
- Luisetti, M.; Ma, S.; Iadarola, P.; Stone, P.J.; Viglio, S.; Casado, B.; Lin, Y.Y.; Snider, G.L.; Turino, G.M. Desmosine as a biomarker of elastin degradation in COPD: current status and future directions. European Respiratory Journal 2008, 32(5), 1146–1157. [Google Scholar] [CrossRef]
- Huang, J.T.J.; Chaudhuri, R.; Albarbarawi, O.; Barton, A. Clinical validity of plasma and urinary desmosine as biomarkers for chronic obstructive pulmonary disease. Thorax 2012, 67(6), 502, https://thorax.bmj.com/content/67/6/502.short. [Google Scholar] [CrossRef]
- Moon, J.Y.; Leitao Filho, F.S.; Shahangian, K.; Takiguchi, H.; Sin, D.D. Blood and sputum protein biomarkers for chronic obstructive pulmonary disease (COPD). Expert review of proteomics 2018, 15(11), 923–935. [Google Scholar] [CrossRef] [PubMed]
- Ferrarotti, I.; Corsico, A.G.; Stolk, J.; Ottaviani, S.; Fumagalli, M.; Janciauskiene, S.; Iadarola, P. Advances in identifying urine/serum biomarkers in alpha-1 antitrypsin deficiency for more personalized future treatment strategies. COPD: Journal of Chronic Obstructive Pulmonary Disease 2017, 14(1), 56–65. [Google Scholar] [CrossRef]
- Liu, X.; Ma, S.; Liu, S.; Liu, M.; Turino, G.; Cantor, J. The ratio of free to bound desmosine and isodesmosine may reflect emphysematous changes in COPD. Lung 2015, 193, 329–334. [Google Scholar] [CrossRef] [PubMed]
- Hogea, S.P.; Tudorache, E.; Fildan, A.P.; Fira-Mladinescu, O.; Marc, M.; Oancea, C. Risk factors of chronic obstructive pulmonary disease exacerbations. The clinical respiratory journal 2020, 14(3), 183–197. [Google Scholar] [CrossRef]
- Mehraban, S.; Gu, G.; Ma, S.; Liu, X.; Turino, G.; Cantor, J. The proinflammatory activity of structurally altered elastic fibers. American journal of respiratory cell and molecular biology 2020, 63(5), 699–706. [Google Scholar] [CrossRef]
- de Oliveira, M.V.; Rocha, N.N.; Santos, R.S.; Rocco, M.R.M.; de Magalhães, R.F.; Silva, J.D.; et al. Endotoxin-Induced emphysema exacerbation: a novel model of chronic obstructive pulmonary disease exacerbations causing cardiopulmonary impairment and diaphragm dysfunction. Front Physiol 2019, 10, 664. [Google Scholar] [CrossRef]
- Kobayashi, S.; Fujinawa, R.; Ota, F.; Kobayashi, S.; Angata, T.; Ueno, M.; et al. A single dose of lipopolysaccharide into mice with emphysema mimics human chronic obstructive pulmonary disease exacerbation as assessed by micro-computed tomography. Am J Respir Cell Mol Biol 2013, 49, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Cantor, J.O.; Cerreta, J.M.; Ochoa, M.; Ma, S.; Liu, M.; Turino, G.M. Therapeutic effects of hyaluronan on smoke-induced elastic fiber injury: does delayed treatment affect efficacy? Lung 2011, 189, 51–56. [Google Scholar] [CrossRef]
- Wick, M.R. Pathologic features of smoking-related lung diseases, with emphasis on smoking-related interstitial fibrosis and a consideration of differential diagnoses. In Seminars in Diagnostic Pathology; WB Saunders, 2018; Volume 35, pp. 315–315. [Google Scholar]
- Morse, D.; Rosas, I.O. Tobacco smoke–induced lung fibrosis and emphysema. Annual review of physiology 2014, 76(1), 493–513. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Keller, S.; Turino, G.M. Modulation of Airspace Enlargement in Elastase-Induced Emphysema by Intratracheal Instalment of Hyaluronidase and Hyaluronic Acid. Experimental lung research 1995, 21(3), 423–436. [Google Scholar] [CrossRef]
- Cantor, J.O.; Cerreta, J.M.; Armand, G.; Turino, G.M. Further investigation of the use of intratracheally administered hyaluronic acid to ameliorate elastase-induced emphysema. Experimental lung research 1997, 23(3), 229–244. [Google Scholar] [CrossRef] [PubMed]
- Cantor, J.O.; Cerreta, J.M.; Ochoa, M.; Ma, S.; Chow, T.; Grunig, G.; Turino, G.M. Aerosolized hyaluronan limits airspace enlargement in a mouse model of cigarette smoke–induced pulmonary emphysema. Experimental lung research 2005, 31(4), 417–430. [Google Scholar] [CrossRef]
- Cantor, J.O.; Ma, S.; Liu, X.; Campos, M.A.; Strange, C.; et al. A 28-day clinical trial of aerosolized hyaluronan in alpha-1 antiprotease deficiency COPD using desmosine as a surrogate marker for drug efficacy. Respiratory Medicine 2021, 182, 106402. [Google Scholar] [CrossRef] [PubMed]
- Gul, A.; Yang, F.; Xie, C.; Du, W.; Mohammadtursun, N.; Wang, B.; Le, J.; Dong, J. Pulmonary fibrosis model of mice induced by different administration methods of bleomycin. BMC pulmonary medicine 2023, 23(1), 91. [Google Scholar] [CrossRef]
- Ishida, Y.; Kuninaka, Y.; Mukaida, N.; Kondo, T. Immune mechanisms of pulmonary fibrosis with bleomycin. International journal of molecular sciences 2023, 24(4), 3149. [Google Scholar] [CrossRef]
- Pei, Z.; Qin, Y.; Fu, X.; Yang, F.; Huo, F.; Liang, X.; Wang, S.; Cui, H.; Lin, P.; Zhou, G.; Yan, J. Inhibition of ferroptosis and iron accumulation alleviates pulmonary fibrosis in a bleomycin model. Redox biology 2022, 57, 102509. [Google Scholar] [CrossRef] [PubMed]
- Della Latta, V.; Cecchettini, A.; Del Ry, S.; Morales, M.A. Bleomycin in the setting of lung fibrosis induction: from biological mechanisms to counteractions. Pharmacological research 2015, 97, 122–130. [Google Scholar] [CrossRef]
- Scotton, C.J.; Chambers, R.C. Bleomycin revisited: towards a more representative model of IPF? American Journal of Physiology-Lung Cellular and Molecular Physiology 2010, 299(4), L439–L441. [Google Scholar] [CrossRef]
- Liu, X.; Khadtare, N.; Patel, H.; Stephani, R.; Cantor, J. Time-dependent effects of HJP272, an endothelin receptor antagonist, in bleomycin-induced pulmonary fibrosis. Pulmonary pharmacology & therapeutics 2017, 45, 164–169. [Google Scholar]
- Raghu, G.; Behr, J.; Brown, K.K.; Egan, J.J.; Kawut, S.M.; Flaherty, K.R.; Martinez, F.J.; Nathan, S.D.; Wells, A.U.; Collard, H.R.; Costabel, U. Treatment of idiopathic pulmonary fibrosis with ambrisentan: a parallel, randomized trial. Annals of internal medicine 2013, 158(9), 641–649. [Google Scholar] [CrossRef]
- Li, H.F.; Wang, J.X.; Xie, Z.F.; Li, L.H.; Li, B.; Huang, F.F.; Li, J.; Zhou, X.L. Bosentan and ambrisentan in the treatment of idiopathic pulmonary fibrosis: a meta-analysis. European Review for Medical & Pharmacological Sciences 2024, 28(3). [Google Scholar]
- Wu, X.; Li, W.; Luo, Z.; Chen, Y. A comprehensive comparison of the safety and efficacy of drugs in the treatment of idiopathic pulmonary fibrosis: a network meta-analysis based on randomized controlled trials. BMC Pulmonary Medicine 2024, 24(1), 58. [Google Scholar] [CrossRef] [PubMed]
- Suki, B.; Bates, J.H. Lung tissue mechanics as an emergent phenomenon. Journal of applied physiology 2011, 110(4), 1111–1118. [Google Scholar] [CrossRef] [PubMed]
- Leonard-Duke, J.; Agro, S.M.; Csordas, D.J.; Bruce, A.C.; Eggertsen, T.G.; Tavakol, T.N.; Comlekoglu, T.; Barker, T.H.; Bonham, C.A.; Saucerman, J.J.; Taite, L.J. Multiscale computational model predicts how environmental changes and treatments affect microvascular remodeling in fibrotic disease. PNAS nexus 2025, 4(1), pgae551. [Google Scholar] [CrossRef]
- Philippe, P.; Mansi, O. Nonlinearity in the epidemiology of complex health and disease processes. Theoretical medicine and bioethics 1998, 19, 591–607. [Google Scholar] [CrossRef] [PubMed]
- Hunt, A.G.; Sahimi, M. Flow, transport, and reaction in porous media: Percolation scaling, critical-path analysis, and effective medium approximation. Reviews of Geophysics 2017, 55(4), 993–1078. [Google Scholar] [CrossRef]
- Júnior, C.; Ulldemolins, A.; Narciso, M.; Almendros, I.; Farré, R.; Navajas, D.; López, J.; Eroles, M.; Rico, F.; Gavara, N. Multi-step extracellular matrix remodelling and stiffening in the development of idiopathic pulmonary fibrosis. International Journal of Molecular Sciences 2023, 24(2), 1708. [Google Scholar] [CrossRef]
- Hall, J.K.; Bates, J.H.; Casey, D.T.; Bartolák-Suki, E.; Lutchen, K.R.; Suki, B. Predicting alveolar ventilation heterogeneity in pulmonary fibrosis using a non-uniform polyhedral spring network model. Frontiers in Network Physiology 2023, 3, 1124223. [Google Scholar] [CrossRef]
- Casey, D.T.; Bou Jawde, S.; Herrmann, J.; Mori, V.; Mahoney, J.M.; Suki, B.; Bates, J.H. Percolation of collagen stress in a random network model of the alveolar wall. Scientific reports 2021, 11(1), 16654. [Google Scholar] [CrossRef]
- Caicedo, H.H.; Darrow, J.J.; Caicedo, J.C.; Pentland, A. Prioritizing Early Disease Intervention. Therapeutic Innovation & Regulatory Science 2023, 57(6), 1148–1152. [Google Scholar]
- Fan, H.; Wang, Y.; Zhao, K.; Su, L.; Deng, C.; Huang, J.; Chen, G. Incomplete knockdown of MyD88 inhibits LPS-induced lung injury and lung fibrosis in a mouse model. Inflammation 2023, 46(6), 2276–2288. [Google Scholar] [CrossRef]
- Hung, Y.H.; Hsieh, W.Y.; Hsieh, J.S.; Liu, F.C.; Tsai, C.H.; Lu, L.C.; Huang, C.Y.; Wu, C.L.; Lin, C.S. Alternative roles of STAT3 and MAPK signaling pathways in the MMPs activation and progression of lung injury induced by cigarette smoke exposure in ACE2 knockout mice. International journal of biological sciences 2016, 12(4), 454. [Google Scholar] [CrossRef] [PubMed]
- Doyle, A.; McGarry, M.P.; Lee, N.A.; Lee, J.J. The construction of transgenic and gene knockout/knockin mouse models of human disease. Transgenic research 2012, 21, 327–349. [Google Scholar] [CrossRef] [PubMed]
- Gawenis, L.R.; Hodges, C.A.; McHugh, D.R.; Valerio, D.M.; Miron, A.; Cotton, C.U.; Liu, J.; Walker, N.M.; Strubberg, A.M.; Gillen, A.E.; Mutolo, M.J. A BAC transgene expressing human CFTR under control of its regulatory elements rescues Cftr knockout mice. Scientific Reports 2019, 9(1), 11828. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, E. Replacement strategies for animal studies in inhalation testing. Sci 2021, 3(4), 45. [Google Scholar] [CrossRef]
- Tanner, L.; Single, A.B. Animal models reflecting chronic obstructive pulmonary disease and related respiratory disorders: translating preclinical data into clinical relevance. Journal of innate immunity 2020, 12(3), 203–225. [Google Scholar] [CrossRef]
- McGonigle, P.; Ruggeri, B. Animal models of human disease: challenges in enabling translation. Biochemical pharmacology 2014, 87(1), 162–171. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



