Submitted:
21 June 2025
Posted:
23 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.2. Study Selection
2.3. Data Extraction and Synthesis
2.4. Quality Assessment
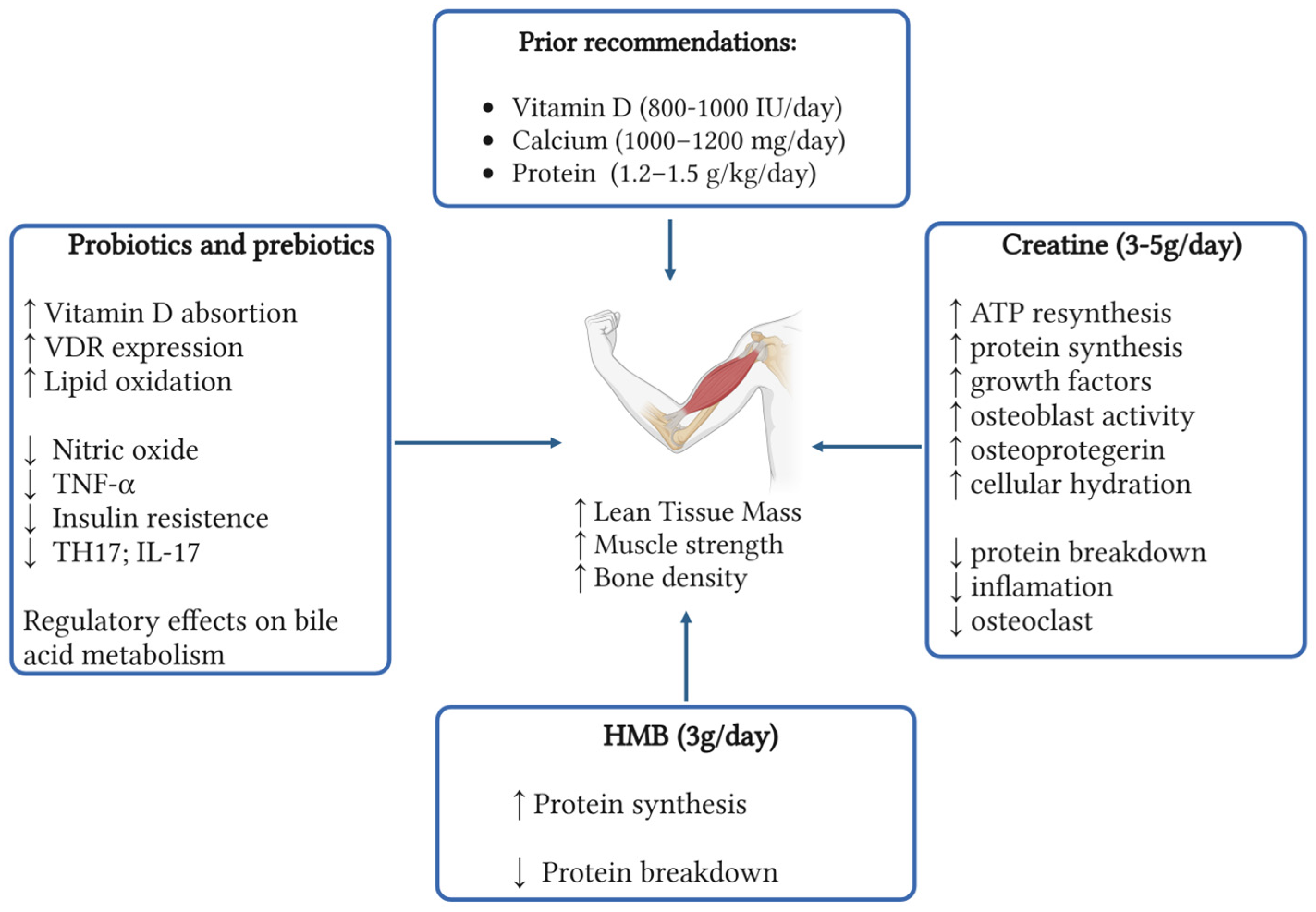
3. Creatine
3.1. Creatine: A Well-Known Supplement
3.1.1. Effects of Creatine in Aging Muscle
3.1.2. Effects on Aging Bone
3.1.3. Creatine Plus Other Supplements
3.1.4. Conclusions on Creatine
4. β-Hydroxy-β-Methylbutyrate
4.1. β-Hydroxy-β-Methylbutyrate: A Promising New Supplement
4.1.1. Effects on Aging Muscle
4.1.2. Effects on Aging Bone
4.1.3. HMB Plus Other Supplements
4.1.4. Conclusions on HMB
5. Prebiotics and Probiotics
5.1. Prebiotics and Probiotics: Targeting the Gut-Muscle-Bone Axis
5.1.1. Effects on Aging Muscle
5.1.2. Effects on Aging Bone
5.1.3. Gut Microbiota and Bile Acid Metabolism
5.1.5. Conclusions on Prebiotics and Probiotics
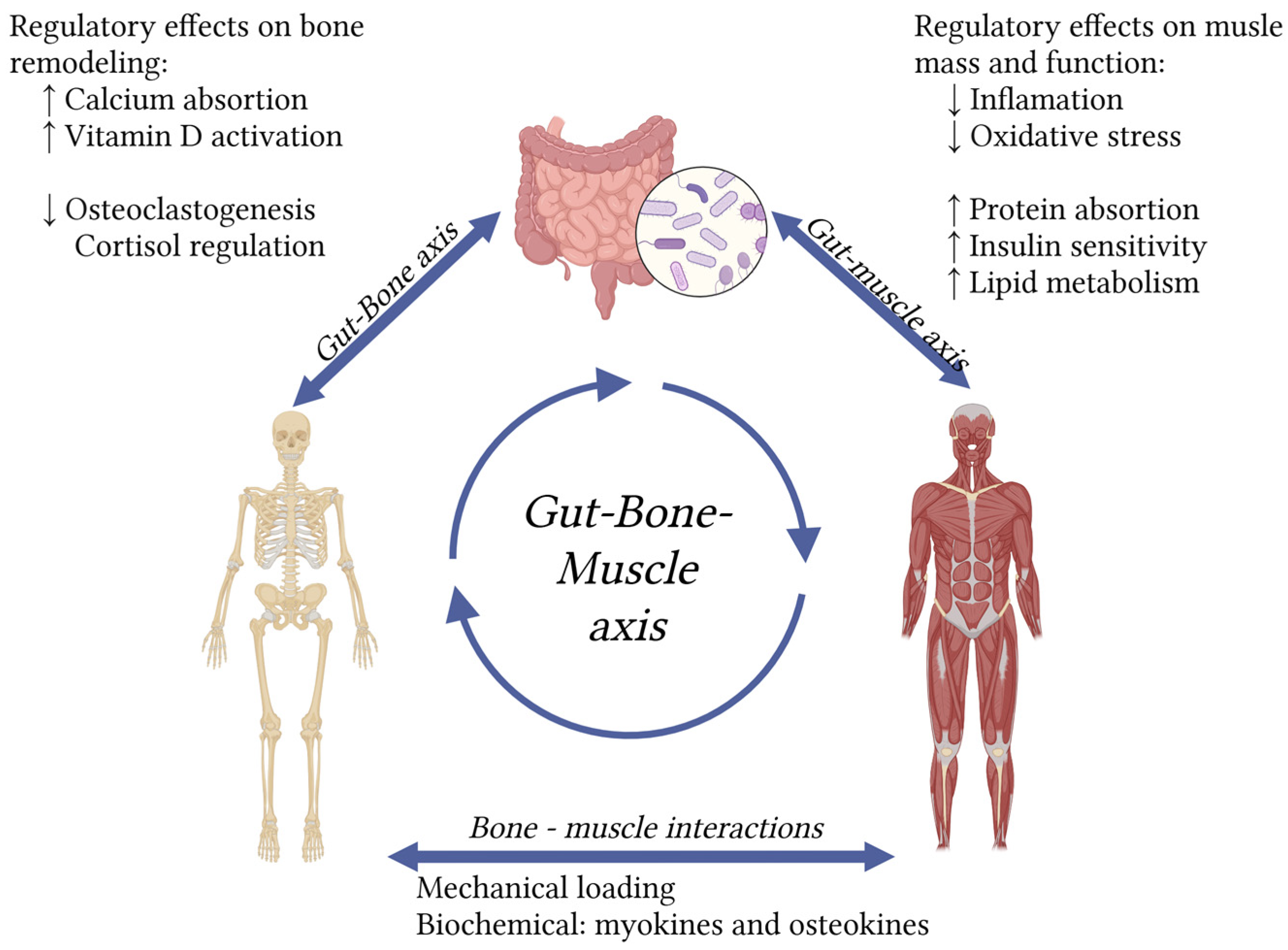
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Franulic F, Salech F, Rivas D, Duque G. Deciphering Osteosarcopenia through the hallmarks of aging. Mech Ageing Dev 2024;222:111997. [CrossRef]
- Kirk B, Zanker J, Duque G. Osteosarcopenia: epidemiology, diagnosis, and treatment—facts and numbers. J Cachexia Sarcopenia Muscle 2020;11:609–18. [CrossRef]
- US Preventive Services Task Force, Nicholson WK, Silverstein M, Wong JB, Chelmow D, Coker TR, et al. Screening for Osteoporosis to Prevent Fractures: US Preventive Services Task Force Recommendation Statement. JAMA 2025;333:498. [CrossRef]
- Kirk B, Cawthon PM, Arai H, Ávila-Funes JA, Barazzoni R, Bhasin S, et al. The Conceptual Definition of Sarcopenia: Delphi Consensus from the Global Leadership Initiative in Sarcopenia (GLIS). Age Ageing 2024;53:afae052. [CrossRef]
- Smith C, Sim M, Dalla Via J, Levinger I, Duque G. The Interconnection Between Muscle and Bone: A Common Clinical Management Pathway. Calcif Tissue Int 2023;114:24–37. [CrossRef]
- Kirk B, Prokopidis K, Duque G. Nutrients to mitigate osteosarcopenia: the role of protein, vitamin D and calcium. Curr Opin Clin Nutr Metab Care 2021;24:25–32. [CrossRef]
- Candow DG, Forbes SC, Chilibeck PD, Cornish SM, Antonio J, Kreider RB. Effectiveness of Creatine Supplementation on Aging Muscle and Bone: Focus on Falls Prevention and Inflammation. J Clin Med 2019;8:488. [CrossRef]
- Brosnan JT, Brosnan ME. Creatine: Endogenous Metabolite, Dietary, and Therapeutic Supplement. Annu Rev Nutr 2007;27:241–61. [CrossRef]
- Barcelos RP, Stefanello ST, Mauriz JL, Gonzalez-Gallego J, Soares FAA. Creatine and the Liver: Metabolism and Possible Interactions. Mini-Rev Med Chem 2015;16:12–8. [CrossRef]
- Nedeljkovic D, Ostojic SM. Dietary exposure to creatine-precursor amino acids in the general population. Amino Acids 2025;57:29. [CrossRef]
- Kreider RB, Kalman DS, Antonio J, Ziegenfuss TN, Wildman R, Collins R, et al. International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine. J Int Soc Sports Nutr 2017;14:18. [CrossRef]
- Cordingley DM, Cornish SM, Candow DG. Anti-Inflammatory and Anti-Catabolic Effects of Creatine Supplementation: A Brief Review. Nutrients 2022;14:544. [CrossRef]
- Candow DG, Chilibeck PD, Forbes SC, Fairman CM, Gualano B, Roschel H. Creatine supplementation for older adults: Focus on sarcopenia, osteoporosis, frailty and Cachexia. Bone 2022;162:116467. [CrossRef]
- Candow DG, Kirk B, Chilibeck PD, Duque G. The potential of creatine monohydrate supplementation in the management of osteosarcopenia. Curr Opin Clin Nutr Metab Care 2025;28:235–9. [CrossRef]
- Candow DG, Chilibeck PD, Gordon J, Vogt E, Landeryou T, Kaviani M, et al. Effect of 12 months of creatine supplementation and whole-body resistance training on measures of bone, muscle and strength in older males. Nutr Health 2021;27:151–9. [CrossRef]
- Fragala MS, Cadore EL, Dorgo S, Izquierdo M, Kraemer WJ, Peterson MD, et al. Resistance Training for Older Adults: Position Statement From the National Strength and Conditioning Association. J Strength Cond Res 2019;33:2019–52. [CrossRef]
- Wallimann T, Hemmer W. Creatine kinase in non-muscle tissues and cells. Mol Cell Biochem 1994;133–134:193–220. [CrossRef]
- Ch’ng JL, Ibrahim B. Transcriptional and posttranscriptional mechanisms modulate creatine kinase expression during differentiation of osteoblastic cells. J Biol Chem 1994;269:2336–41.
- Institute of Cell Biology, ETH Hoenggerberg, CH-8093 Zurich, Switzerland, Gerber I, Ap Gwynn I, Alini M, Wallimann T. Stimulatory effects of creatine on metabolic activity, differentiation and mineralization of primary osteoblast-like cells in monolayer and micromass cell cultures. Eur Cell Mater 2005;10:8–22. [CrossRef]
- Kearns AE, Khosla S, Kostenuik PJ. Receptor activator of nuclear factor kappaB ligand and osteoprotegerin regulation of bone remodeling in health and disease. Endocr Rev 2008;29:155–92. [CrossRef]
- Hong M, Wang J, Jin L, Ling K. The impact of creatine levels on musculoskeletal health in the elderly: a mendelian randomization analysis. BMC Musculoskelet Disord 2024;25:1004. [CrossRef]
- Forbes SC, Candow DG. Creatine and strength training in older adults: an update. Transl Exerc Biomed 2024;1:212–22. [CrossRef]
- Candow DG, Little JP, Chilibeck PD, Abeysekara S, Zello GA, Kazachkov M, et al. Low-Dose Creatine Combined with Protein during Resistance Training in Older Men. Med Sci Sports Exerc 2008;40:1645–52. [CrossRef]
- Cochet C, Belloni G, Buondonno I, Chiara F, D’Amelio P. The Role of Nutrition in the Treatment of Sarcopenia in Old Patients: From Restoration of Mitochondrial Activity to Improvement of Muscle Performance, a Systematic Review. Nutrients 2023;15:3703. [CrossRef]
- Fernández-Landa J, Calleja-González J, León-Guereño P, Caballero-García A, Córdova A, Mielgo-Ayuso J. Effect of the Combination of Creatine Monohydrate Plus HMB Supplementation on Sports Performance, Body Composition, Markers of Muscle Damage and Hormone Status: A Systematic Review. Nutrients 2019;11:2528. [CrossRef]
- Phillips SM, Lau KJ, D’Souza AC, Nunes EA. An umbrella review of systematic reviews of β-hydroxy-β-methyl butyrate supplementation in ageing and clinical practice. J Cachexia Sarcopenia Muscle 2022;13:2265–75. [CrossRef]
- Kuriyan R, Lokesh DP, Selvam S, Jayakumar J, Philip MG, Shreeram S, et al. The relationship of endogenous plasma concentrations of β-Hydroxy β-Methyl Butyrate (HMB) to age and total appendicular lean mass in humans. Exp Gerontol 2016;81:13–8. [CrossRef]
- Yang C, Song Y, Li T, Chen X, Zhou J, Pan Q, et al. Effects of Beta-Hydroxy-Beta-Methylbutyrate Supplementation on Older Adults with Sarcopenia: A Randomized, Double-Blind, Placebo-Controlled Study. J Nutr Health Aging 2023;27:329–39. [CrossRef]
- Rathmacher JA, Pitchford LM, Stout JR, Townsend JR, Jäger R, Kreider RB, et al. International society of sports nutrition position stand: β-hydroxy-β-methylbutyrate (HMB). J Int Soc Sports Nutr 2025;22:2434734. [CrossRef]
- Bear DE, Langan A, Dimidi E, Wandrag L, Harridge SDR, Hart N, et al. β-Hydroxy-β-methylbutyrate and its impact on skeletal muscle mass and physical function in clinical practice: a systematic review and meta-analysis. Am J Clin Nutr 2019;109:1119–32. [CrossRef]
- Mayhew AJ, Sohel N, Beauchamp MK, Phillips S, Raina P. Sarcopenia Definition and Outcomes Consortium 2020 Definition: Association and Discriminatory Accuracy of Sarcopenia With Disability in the Canadian Longitudinal Study on Aging. J Gerontol Ser A 2023;78:1597–603. [CrossRef]
- Su H, Zhou H, Gong Y, Xiang S, Shao W, Zhao X, et al. The effects of β-hydroxy-β-methylbutyrate or HMB-rich nutritional supplements on sarcopenia patients: a systematic review and meta-analysis. Front Med 2024;11:1348212. [CrossRef]
- Gu W-T, Zhang L-W, Wu F-H, Wang S. The effects of β-hydroxy-β-methylbutyrate supplementation in patients with sarcopenia: A systematic review and meta-analysis. Maturitas 2025;195:108219. [CrossRef]
- Tomaszewska E, Muszyński S, Donaldson J, Dobrowolski P, Chand DKP, Tomczyk-Warunek A, et al. Femoral µCT Analysis, Mechanical Testing and Immunolocalization of Bone Proteins in β-Hydroxy β-Methylbutyrate (HMB) Supplemented Spiny Mouse in a Model of Pregnancy and Lactation-Associated Osteoporosis. J Clin Med 2021;10:4808. [CrossRef]
- Tomaszewska E, Donaldson J, Kosiński J, Dobrowolski P, Tomczyk-Warunek A, Hułas-Stasiak M, et al. β-Hydroxy-β-Methylbutyrate (HMB) Supplementation Prevents Bone Loss during Pregnancy—Novel Evidence from a Spiny Mouse (Acomys cahirinus) Model. Int J Mol Sci 2021;22:3047. [CrossRef]
- Rathmacher J, Pitchford L, Khoo P, Sharp R. Probiotic Bacillus coagulans GBI-30, 6086 Supplementation Improves β-Hydroxy-β-methylbutyrate Bioavailability. FASEB J 2021;35:fasebj.2021.35.S1.01949. [CrossRef]
- Gepner Y, Hoffman JR, Shemesh E, Stout JR, Church DD, Varanoske AN, et al. Combined effect of Bacillus coagulans GBI-30, 6086 and HMB supplementation on muscle integrity and cytokine response during intense military training. J Appl Physiol 2017;123:11–8. [CrossRef]
- Rathmacher JA, Pitchford LM, Khoo P, Angus H, Lang J, Lowry K, et al. Long-term Effects of Calcium β-Hydroxy-β-Methylbutyrate and Vitamin D3 Supplementation on Muscular Function in Older Adults With and Without Resistance Training: A Randomized, Double-blind, Controlled Study. J Gerontol Ser A 2020;75:2089–97. [CrossRef]
- Nasimi N, Sohrabi Z, Dabbaghmanesh MH, Eskandari MH, Bedeltavana A, Famouri M, et al. A Novel Fortified Dairy Product and Sarcopenia Measures in Sarcopenic Older Adults: A Double-Blind Randomized Controlled Trial. J Am Med Dir Assoc 2021;22:809–15. [CrossRef]
- Rittig N, Bach E, Thomsen HH, Møller AB, Hansen J, Johannsen M, et al. Anabolic effects of leucine-rich whey protein, carbohydrate, and soy protein with and without β-hydroxy-β-methylbutyrate (HMB) during fasting-induced catabolism: A human randomized crossover trial. Clin Nutr 2017;36:697–705. [CrossRef]
- Martín R, Langella P. Emerging Health Concepts in the Probiotics Field: Streamlining the Definitions. Front Microbiol 2019;10:1047. [CrossRef]
- Zaib S, Hayat A, Khan I. Probiotics and their Beneficial Health Effects. Mini-Rev Med Chem 2024;24:110–25. [CrossRef]
- Davani-Davari D, Negahdaripour M, Karimzadeh I, Seifan M, Mohkam M, Masoumi S, et al. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods 2019;8:92. [CrossRef]
- Yoo S, Jung S-C, Kwak K, Kim J-S. The Role of Prebiotics in Modulating Gut Microbiota: Implications for Human Health. Int J Mol Sci 2024;25:4834. [CrossRef]
- Locantore P, Del Gatto V, Gelli S, Paragliola RM, Pontecorvi A. The Interplay between Immune System and Microbiota in Osteoporosis. Mediators Inflamm 2020;2020:1–8. [CrossRef]
- Pandey KavitaR, Naik SureshR, Vakil BabuV. Probiotics, prebiotics and synbiotics- a review. J Food Sci Technol 2015;52:7577–87. [CrossRef]
- Whisner CM, Castillo LF. Prebiotics, Bone and Mineral Metabolism. Calcif Tissue Int 2018;102:443–79. [CrossRef]
- Yatsunenko T, Rey FE, Manary MJ, Trehan I, Dominguez-Bello MG, Contreras M, et al. Human gut microbiome viewed across age and geography. Nature 2012;486:222–7. [CrossRef]
- Yadav D, Ghosh TS, Mande SS. Global investigation of composition and interaction networks in gut microbiomes of individuals belonging to diverse geographies and age-groups. Gut Pathog 2016;8:17. [CrossRef]
- Saponaro F, Bertolini A, Baragatti R, Galfo L, Chiellini G, Saba A, et al. Myokines and Microbiota: New Perspectives in the Endocrine Muscle–Gut Axis. Nutrients 2024;16:4032. [CrossRef]
- Chenhuichen C, Cabello-Olmo M, Barajas M, Izquierdo M, Ramírez-Vélez R, Zambom-Ferraresi F, et al. Impact of probiotics and prebiotics in the modulation of the major events of the aging process: A systematic review of randomized controlled trials. Exp Gerontol 2022;164:111809. [CrossRef]
- Sotoudegan F, Daniali M, Hassani S, Nikfar S, Abdollahi M. Reappraisal of probiotics’ safety in human. Food Chem Toxicol 2019;129:22–9. [CrossRef]
- Papadopoulou S. Sarcopenia: A Contemporary Health Problem among Older Adult Populations. Nutrients 2020;12:1293. [CrossRef]
- He Y, Chen Y. The potential mechanism of the microbiota-gut-bone axis in osteoporosis: a review. Osteoporos Int 2022;33:2495–506. [CrossRef]
- Liao X, Wu M, Hao Y, Deng H. Exploring the Preventive Effect and Mechanism of Senile Sarcopenia Based on “Gut–Muscle Axis.” Front Bioeng Biotechnol 2020;8:590869. [CrossRef]
- Zhu J, Peng F, Yang H, Luo J, Zhang L, Chen X, et al. Probiotics and muscle health: the impact of Lactobacillus on sarcopenia through the gut-muscle axis. Front Microbiol 2025;16:1559119. [CrossRef]
- Ticinesi A, Lauretani F, Milani C, Nouvenne A, Tana C, Del Rio D, et al. Aging Gut Microbiota at the Cross-Road between Nutrition, Physical Frailty, and Sarcopenia: Is There a Gut–Muscle Axis? Nutrients 2017;9:1303. [CrossRef]
- Wang G, Li Y, Liu H, Yu X. Gut microbiota in patients with sarcopenia: a systematic review and meta-analysis. Front Microbiol 2025;16:1513253. [CrossRef]
- Lou J, Wang Q, Wan X, Cheng J. Changes and correlation analysis of intestinal microflora composition, inflammatory index, and skeletal muscle mass in elderly patients with sarcopenia. Geriatr Gerontol Int 2024;24:140–6. [CrossRef]
- Picca A, Ponziani FR, Calvani R, Marini F, Biancolillo A, Coelho-Júnior HJ, et al. Gut Microbial, Inflammatory and Metabolic Signatures in Older People with Physical Frailty and Sarcopenia: Results from the BIOSPHERE Study. Nutrients 2019;12:65. [CrossRef]
- Yi R, Feng M, Chen Q, Long X, Park K-Y, Zhao X. The Effect of Lactobacillus plantarum CQPC02 on Fatigue and Biochemical Oxidation Levels in a Mouse Model of Physical Exhaustion. Front Nutr 2021;8:641544. [CrossRef]
- Herman MA. Glucose transport and sensing in the maintenance of glucose homeostasis and metabolic harmony. J Clin Invest 2006;116:1767–75. [CrossRef]
- Vial G, Coudy-Gandilhon C, Pinel A, Wauquier F, Chevenet C, Béchet D, et al. Lipid accumulation and mitochondrial abnormalities are associated with fiber atrophy in the skeletal muscle of rats with collagen-induced arthritis. Biochim Biophys Acta BBA - Mol Cell Biol Lipids 2020;1865:158574. [CrossRef]
- Wang Y, Lei P. Efficacy of probiotic supplements in the treatment of sarcopenia: A systematic review and meta-analysis. PLOS ONE 2025;20:e0317699. [CrossRef]
- Lee M-C, Tu Y-T, Lee C-C, Tsai S-C, Hsu H-Y, Tsai T-Y, et al. Lactobacillus plantarum TWK10 Improves Muscle Mass and Functional Performance in Frail Older Adults: A Randomized, Double-Blind Clinical Trial. Microorganisms 2021;9:1466. [CrossRef]
- Artoni De Carvalho JA, Magalhães LR, Polastri LM, Batista IET, De Castro Bremer S, Caetano HRDS, et al. Prebiotics improve osteoporosis indicators in a preclinical model: systematic review with meta-analysis. Nutr Rev 2023;81:891–903. [CrossRef]
- Sun J. Dietary vitamin D, vitamin D receptor, and microbiome. Curr Opin Clin Nutr Metab Care 2018;21:471–4. [CrossRef]
- Jones ML, Martoni CJ, Prakash S. Oral Supplementation With Probiotic L. reuteri NCIMB 30242 Increases Mean Circulating 25-Hydroxyvitamin D: A Post Hoc Analysis of a Randomized Controlled Trial. J Clin Endocrinol Metab 2013;98:2944–51. [CrossRef]
- Nilsson AG, Sundh D, Bäckhed F, Lorentzon M. Lactobacillus reuteri reduces bone loss in older women with low bone mineral density: a randomized, placebo-controlled, double-blind, clinical trial. J Intern Med 2018;284:307–17. [CrossRef]
- Wu S, Yoon S, Zhang Y-G, Lu R, Xia Y, Wan J, et al. Vitamin D receptor pathway is required for probiotic protection in colitis. Am J Physiol-Gastrointest Liver Physiol 2015;309:G341–9. [CrossRef]
- Sato K, Suematsu A, Okamoto K, Yamaguchi A, Morishita Y, Kadono Y, et al. Th17 functions as an osteoclastogenic helper T cell subset that links T cell activation and bone destruction. J Exp Med 2006;203:2673–82. [CrossRef]
- Dar HY, Pal S, Shukla P, Mishra PK, Tomar GB, Chattopadhyay N, et al. Bacillus clausii inhibits bone loss by skewing Treg-Th17 cell equilibrium in postmenopausal osteoporotic mice model. Nutrition 2018;54:118–28. [CrossRef]
- Dar HY, Shukla P, Mishra PK, Anupam R, Mondal RK, Tomar GB, et al. Lactobacillus acidophilus inhibits bone loss and increases bone heterogeneity in osteoporotic mice via modulating Treg-Th17 cell balance. Bone Rep 2018;8:46–56. [CrossRef]
- Sapra L, Dar HY, Bhardwaj A, Pandey A, Kumari S, Azam Z, et al. Lactobacillus rhamnosus attenuates bone loss and maintains bone health by skewing Treg-Th17 cell balance in Ovx mice. Sci Rep 2021;11:1807. [CrossRef]
- Kassem A, Henning P, Kindlund B, Lindholm C, Lerner UH. TLR5, a novel mediator of innate immunity-induced osteoclastogenesis and bone loss. FASEB J 2015;29:4449–60. [CrossRef]
- Ohlsson C, Nigro G, Boneca IG, Bäckhed F, Sansonetti P, Sjögren K. Regulation of bone mass by the gut microbiota is dependent on NOD1 and NOD2 signaling. Cell Immunol 2017;317:55–8. [CrossRef]
- Zhang M, Sun J, Zhao H, Liu Y, Tang Z, Wen Y, et al. Alginate oligosaccharides relieve estrogen-deprived osteosarcopenia by affecting intestinal Th17 differentiation and systemic inflammation through the manipulation of bile acid metabolism. Int J Biol Macromol 2025;295:139581. [CrossRef]
- Schepper JD, Collins F, Rios-Arce ND, Kang HJ, Schaefer L, Gardinier JD, et al. Involvement of the Gut Microbiota and Barrier Function in Glucocorticoid-Induced Osteoporosis. J Bone Miner Res 2020;35:801–20. [CrossRef]
- Zupan J, Komadina R, Marc J. The relationship between osteoclastogenic and anti-osteoclastogenic pro-inflammatory cytokines differs in human osteoporotic and osteoarthritic bone tissues. J Biomed Sci 2012;19:28. [CrossRef]
- Deng B, Wu J, Li X, Zhang C, Men X, Xu Z. Effects of Bacillus subtilis on growth performance, serum parameters, digestive enzyme, intestinal morphology, and colonic microbiota in piglets. AMB Express 2020;10:212. [CrossRef]
- Yang J, Zhan K, Zhang M. Effects of the Use of a Combination of Two Bacillus Species on Performance, Egg Quality, Small Intestinal Mucosal Morphology, and Cecal Microbiota Profile in Aging Laying Hens. Probiotics Antimicrob Proteins 2020;12:204–13. [CrossRef]
- Wallimann A, Magrath W, Thompson K, Moriarty T, Richards RG, Akdis CA, et al. Gut microbial-derived short-chain fatty acids and bone: a potential role in fracture healing. Eur Cell Mater 2021;41:454–70. [CrossRef]
- Zhang Y-W, Cao M-M, Li Y-J, Dai G-C, Lu P-P, Zhang M, et al. The regulative effect and repercussion of probiotics and prebiotics on osteoporosis: involvement of brain-gut-bone axis. Crit Rev Food Sci Nutr 2023;63:7510–28. [CrossRef]
- Lin H, Liu T, Li X, Gao X, Wu T, Li P. The role of gut microbiota metabolite trimethylamine N-oxide in functional impairment of bone marrow mesenchymal stem cells in osteoporosis disease. Ann Transl Med 2020;8:1009–1009. [CrossRef]
- Gehart H, Van Es JH, Hamer K, Beumer J, Kretzschmar K, Dekkers JF, et al. Identification of Enteroendocrine Regulators by Real-Time Single-Cell Differentiation Mapping. Cell 2019;176:1158-1173.e16. [CrossRef]
- Larabi AB, Masson HLP, Bäumler AJ. Bile acids as modulators of gut microbiota composition and function. Gut Microbes 2023;15:2172671. [CrossRef]
- Li X-J, Fang C, Zhao R-H, Zou L, Miao H, Zhao Y-Y. Bile acid metabolism in health and ageing-related diseases. Biochem Pharmacol 2024;225:116313. [CrossRef]
- Mancin L, Wu GD, Paoli A. Gut microbiota–bile acid–skeletal muscle axis. Trends Microbiol 2023;31:254–69. [CrossRef]
- Hernández-Gómez JG, López-Bonilla A, Trejo-Tapia G, Ávila-Reyes SV, Jiménez-Aparicio AR, Hernández-Sánchez H. In Vitro Bile Salt Hydrolase (BSH) Activity Screening of Different Probiotic Microorganisms. Foods 2021;10:674. [CrossRef]
- Joyce SA, MacSharry J, Casey PG, Kinsella M, Murphy EF, Shanahan F, et al. Regulation of host weight gain and lipid metabolism by bacterial bile acid modification in the gut. Proc Natl Acad Sci 2014;111:7421–6. [CrossRef]
- Sato Y, Atarashi K, Plichta DR, Arai Y, Sasajima S, Kearney SM, et al. Novel bile acid biosynthetic pathways are enriched in the microbiome of centenarians. Nature 2021;599:458–64. [CrossRef]
- Zhao Y-X, Song Y-W, Zhang L, Zheng F-J, Wang X-M, Zhuang X-H, et al. Association between bile acid metabolism and bone mineral density in postmenopausal women. Clinics 2020;75:e1486. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



