Submitted:
19 June 2025
Posted:
24 June 2025
You are already at the latest version
Abstract
Keywords:
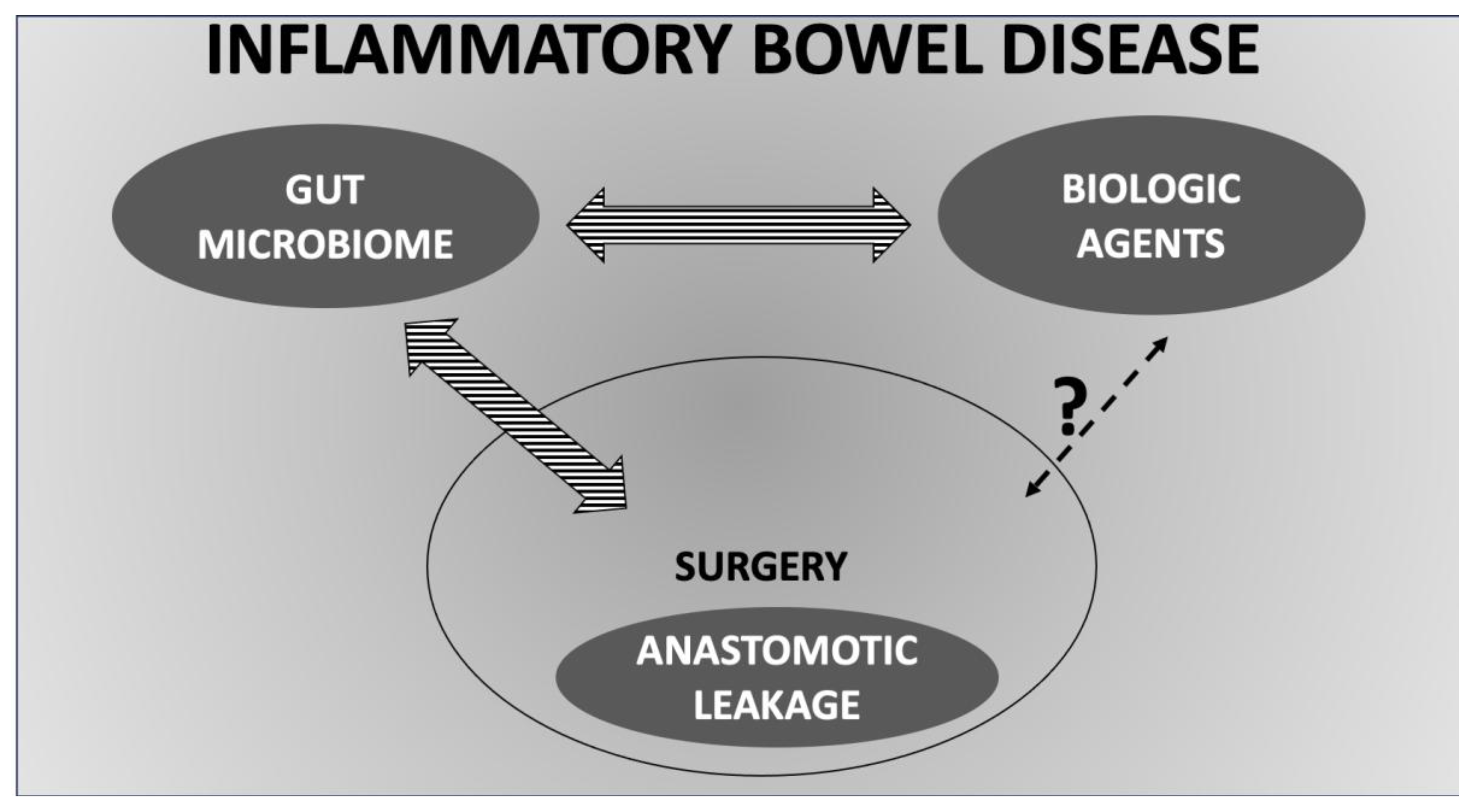
1. Introduction
2. Materials and Methods
3. Results and Discussion
3.1. The Gut Microbiota in Inflammatory Bowel Disease
3.1.1. Dysbiosis
3.1.2. Faecalibacterium prausnitzii
3.2. Fluctuation of Gut Microbiota and Implications for Treatment Response Prediction
3.3. Longitudinal Changes of Gut Microbiota in IBD Patients Treated with Biologic Agents
3.3.1. Adalimumab
3.3.2. Infliximab
3.3.3. Ustekinumab
3.3.4. Vedolizumab
3.4. Surgery in Inflammatory Bowel Disease, Surgical Complications and Gut Microbiota
3.4.1. Surgery and the Role of Gut Microbiota
3.4.2. Anastomotic Leakage and the Role of Gut Microbiota
3.4.3. Anastomotic Leakage and the Role of Biologic Agents
4. Conclusions
Author Contributions
Funding
Informed Consent Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| IBD | Inflammatory Bowel Disease |
| SCFA | short-chain fatty acids |
| CD | Crohn’s Disease |
| UC | Ulcerative Colitis |
References
- Dolinger M, Torres J, Vermeire S. Crohn's disease. Lancet. 2024; 403: 1177-1191.
- Le Berre C, Honap S, Peyrin-Biroulet L. Ulcerative colitis. Lancet. 2023; 402: 571-584.
- Eckburg PB, Bik EM, Bernstein CN, Purdom E, Dethlefsen L, Sargent M, et al. Diversity of the human intestinal microbial flora. Science. 2005; 308: 1635-8.
- Frank DN, St Amand AL, Feldman RA, Boedeker EC, Harpaz N, Pace NR. Molecular-phylogenetic characterization of microbial community imbalances in human inflammatory bowel diseases. Proc Natl Acad Sci U S A. 2007 ;104: 13780-5. [CrossRef]
- Pisani A, Rausch P, Bang C, Ellul S, Tabone T, Marantidis Cordina C, et al. Dysbiosis in the Gut Microbiota in Patients with Inflammatory Bowel Disease during Remission. Microbiol Spectr. 2022; 10: e0061622. [CrossRef]
- Machiels K, Joossens M, Sabino J, De Preter V, Arijs I, Eeckhaut V, et al. A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut. 2014; 63: 1275-83.
- Schulthess J, Pandey S, Capitani M, Rue-Albrecht KC, Arnold I, Franchini F, et al. The Short Chain Fatty Acid Butyrate Imprints an Antimicrobial Program in Macrophages. Immunity. 2019; 50: 432-445.e7.
- Khalaf R, Sciberras M, Ellul P. The role of the fecal microbiota in inflammatory bowel disease. Eur J Gastroenterol Hepatol. 2024; 36: 1249-1258. [CrossRef]
- Martín R, Rios-Covian D, Huillet E, Auger S, Khazaal S, Bermúdez-Humarán LG, et al. Faecalibacterium: a bacterial genus with promising human health applications. FEMS Microbiol Rev. 2023; 47: fuad039.
- Lopez-Siles M, Martinez-Medina M, Abellà C, Busquets D, Sabat-Mir M, Duncan SH, et al. Mucosa-associated Faecalibacterium prausnitzii phylotype richness is reduced in patients with inflammatory bowel disease. Appl Environ Microbiol. 2015; 81: 7582-92. [CrossRef]
- Touch S, Godefroy E, Rolhion N, Danne C, Oeuvray C, Straube M, et al. Human CD4+CD8α+ Tregs induced by Faecalibacterium prausnitzii protect against intestinal inflammation. JCI Insight. 2022; 7: e154722.
- Zhou L, Zhang M, Wang Y, Dorfman RG, Liu H, Yu T, et al. Faecalibacterium prausnitzii Produces Butyrate to Maintain Th17/Treg Balance and to Ameliorate Colorectal Colitis by Inhibiting Histone Deacetylase 1. Inflamm Bowel Dis. 2018; 24: 1926-1940.
- Lavelle A, Sokol H. Gut microbiota-derived metabolites as key actors in inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2020 Apr;17[4]:223-237. [CrossRef]
- Varela E, Manichanh C, Gallart M, Torrejón A, Borruel N, Casellas F, et al. Colonisation by Faecalibacterium prausnitzii and maintenance of clinical remission in patients with ulcerative colitis. Aliment Pharmacol Ther. 2013; 38: 151-61.
- Magnusson MK, Strid H, Sapnara M, Lasson A, Bajor A, Ung KA, et al. Anti-TNF Therapy Response in Patients with Ulcerative Colitis Is Associated with Colonic Antimicrobial Peptide Expression and Microbiota Composition. J Crohns Colitis. 2016; 10: 943-52. [CrossRef]
- Alexandrescu L, Nicoara AD, Tofolean DE, Herlo A, Nelson Twakor A, et al. Healing from Within: How Gut Microbiota Predicts IBD Treatment Success-A Systematic Review. Int J Mol Sci. 2024; 25: 8451.
- Schierova D, Roubalova R, Kolar M, Stehlikova Z, Rob F, Jackova Z, et al. Fecal Microbiota Changes and Specific Anti-Bacterial Response in Patients with IBD during Anti-TNF Therapy. Cells. 2021; 10: 3188. [CrossRef]
- Halfvarson J, Brislawn CJ, Lamendella R, Vázquez-Baeza Y, Walters WA, Bramer LM, et al. Dynamics of the human gut microbiota in inflammatory bowel disease. Nat Microbiol. 2017; 2: 17004. [CrossRef]
- Eckenberger J, Butler JC, Bernstein CN, Shanahan F, Claesson MJ. Interactions between Medications and the Gut Microbiota in Inflammatory Bowel Disease. Microorganisms. 2022; 10: 1963.
- Meade S, Liu Chen Kiow J, Massaro C, Kaur G, Squirell E, Bressler B, et al. Gut microbiota-associated predictors as biomarkers of response to advanced therapies in inflammatory bowel disease: a systematic review. Gut Microbes. 2023; 15: 2287073. [CrossRef]
- Rajca S, Grondin V, Louis E, Vernier-Massouille G, Grimaud JC, Bouhnik Y, et al. Alterations in the intestinal microbiota [dysbiosis] as a predictor of relapse after infliximab withdrawal in Crohn's disease. Inflamm Bowel Dis. 2014; 20: 978-86.
- Caenepeel C, Falony G, Machiels K, Verstockt B, Goncalves PJ, Ferrante M, et al. Dysbiosis and Associated Stool Features Improve Prediction of Response to Biological Therapy in Inflammatory Bowel Disease. Gastroenterology. 2024; 166: 483-495.
- Zhou Y, Xu ZZ, He Y, Yang Y, Liu L, Lin Q, et al. Gut Microbiota Offers Universal Biomarkers across Ethnicity in Inflammatory Bowel Disease Diagnosis and Infliximab Response Prediction. mSystems. 2018; 3: e00188-17. [CrossRef]
- Ding NS, McDonald JAK, Perdones-Montero A, Rees DN, Adegbola SO, Misra R, et al. Metabonomics and the Gut Microbiota Associated with Primary Response to Anti-TNF Therapy in Crohn's Disease. J Crohns Colitis. 2020; 14: 1090-1102.
- Busquets D, Mas-de-Xaxars T, López-Siles M, Martínez-Medina M, Bahí A, Sàbat M, et al. Anti-tumour Necrosis Factor Treatment with Adalimumab Induces Changes in the Microbiota of Crohn's Disease. J Crohns Colitis. 2015; 9:899-906. [CrossRef]
- Ribaldone DG, Caviglia GP, Abdulle A, Pellicano R, Ditto MC, Morino M, et al. Adalimumab Therapy Improves Intestinal Dysbiosis in Crohn's Disease. J Clin Med. 2019; 8: 1646.
- Chen L, Lu Z, Kang D, Feng Z, Li G, Sun M, et al. Distinct alterations of fecal microbiota refer to the efficacy of adalimumab in Crohn's disease. Front Pharmacol. 2022; 13: 913720. [CrossRef]
- Oh HN, Shin SY, Kim JH, Baek J, Kim HJ, Lee KM, et al. Dynamic changes in the gut microbiota composition during adalimumab therapy in patients with ulcerative colitis: implications for treatment response prediction and therapeutic targets. Gut Pathog. 2024; 16: 44. [CrossRef]
- Sakurai T, Nishiyama H, Sakai K, De Velasco MA, Nagai T, Komeda Y, et al. Mucosal microbiota and gene expression are associated with long-term remission after discontinuation of adalimumab in ulcerative colitis. Sci Rep. 2020; 10: 19186.
- Aden K, Rehman A, Waschina S, Pan WH, Walker A, Lucio M, et al. Metabolic Functions of Gut Microbes Associate with Efficacy of Tumor Necrosis Factor Antagonists in Patients With Inflammatory Bowel Diseases. Gastroenterology. 2019; 157: 1279-1292.e11. [CrossRef]
- Ditto MC, Parisi S, Landolfi G, Borrelli R, Realmuto C, Finucci A, et al. Intestinal microbiota changes induced by TNF-inhibitors in IBD-related spondyloarthritis. RMD Open. 2021; 7: e001755.
- Zhuang X, Tian Z, Feng R, Li M, Li T, Zhou G, et al. Fecal Microbiota Alterations Associated With Clinical and Endoscopic Response to Infliximab Therapy in Crohn's Disease. Inflamm Bowel Dis. 2020; 26: 1636-1647. [CrossRef]
- Sanchis-Artero L, Martínez-Blanch JF, Manresa-Vera S, Cortés-Castell E, Valls-Gandia M, Iborra M, et al. Evaluation of changes in intestinal microbiota in Crohn's disease patients after anti-TNF alpha treatment. Sci Rep. 2021; 11: 10016. [CrossRef]
- Seong G, Kim N, Joung JG, Kim ER, Chang DK, Chun Jet al. Changes in the Intestinal Microbiota of Patients with Inflammatory Bowel Disease with Clinical Remission during an 8-Week Infliximab Infusion Cycle. Microorganisms. 2020; 8: 874.
- Tamburini FB, Tripathi A, Gold MP, Yang JC, Biancalani T, McBride JM, et al. Gut Microbial Species and Endotypes Associate with Remission in Ulcerative Colitis Patients Treated with Anti-TNF or Anti-integrin Therapy. J Crohns Colitis. 2024; 18: 1819-1831. [CrossRef]
- Rob F, Schierova D, Stehlikova Z, Kreisinger J, Roubalova R, Coufal S, et al. Association between ustekinumab therapy and changes in specific anti-microbial response, serum biomarkers, and microbiota composition in patients with IBD: A pilot study. PLoS One. 2022; 17: e0277576.
- Doherty MK, Ding T, Koumpouras C, Telesco SE, Monast C, Das A, et al. Fecal Microbiota Signatures Are Associated with Response to Ustekinumab Therapy among Crohn's Disease Patients. mBio. 2018; 9: e02120-17. [CrossRef]
- Xu F, Xie R, He L, Wang H, Zhu Y, Yang X, Yu H. Oral Microbiota Associated with Clinical Efficacy of Ustekinumab in Crohn's Disease. Endocr Metab Immune Disord Drug Targets. 2025 Jan 13. [CrossRef]
- Liu J, Fang H, Hong N, Lv C, Zhu Q, Feng Y, et al. Gut Microbiota and Metabonomic Profile Predict Early Remission to Anti-Integrin Therapy in Patients with Moderate to Severe Ulcerative Colitis. Microbiol Spectr. 2023; 11: e0145723.
- Ananthakrishnan AN, Luo C, Yajnik V, Khalili H, Garber JJ, Stevens BW, et al. Gut Microbiota Function Predicts Response to Anti-integrin Biologic Therapy in Inflammatory Bowel Diseases. Cell Host Microbe. 2017; 21: 603-610.e3. [CrossRef]
- Hu Y, Wu Z, Yang X, Ding J, Wang Q, Fang H, et al. Reduced gut microbiota diversity in ulcerative colitis patients with latent tuberculosis infection during vedolizumab therapy: insights on prophylactic anti-tuberculosis effects. BMC Microbiol. 2024; 24: 543.
- Tsai L, Ma C, Dulai PS, Prokop LJ, Eisenstein S, Ramamoorthy SL, et al. Contemporary Risk of Surgery in Patients With Ulcerative Colitis and Crohn's Disease: A Meta-Analysis of Population-Based Cohorts. Clin Gastroenterol Hepatol. 2021; 19: 2031-2045.e11.
- Lewis JD, Daniel SG, Li H, Hao F, Patterson AD, Hecht AL, et al. Surgery for Crohn's Disease Is Associated With a Dysbiotic Microbiota and Metabolome: Results From Two Prospective Cohorts. Cell Mol Gastroenterol Hepatol. 2024; 18: 101357. [CrossRef]
- Fang X, Vázquez-Baeza Y, Elijah E, Vargas F, Ackermann G, Humphrey G, et al. Gastrointestinal Surgery for Inflammatory Bowel Disease Persistently Lowers Microbiota and Metabolome Diversity. Inflamm Bowel Dis. 2021; 27: 603-616.
- Murphy PB, Khot Z, Vogt KN, Ott M, Dubois L. Quality of Life After Total Proctocolectomy With Ileostomy or IPAA: A Systematic Review. Dis Colon Rectum. 2015; 58: 899-908.
- McDermott FD, Heeney A, Kelly ME, Steele RJ, Carlson GL, Winter DC. Systematic review of preoperative, intraoperative and postoperative risk factors for colorectal anastomotic leaks. Br J Surg. 2015; 102: 462-79. [CrossRef]
- Hansen RB, Balachandran R, Valsamidis TN, Iversen LH. The role of preoperative mechanical bowel preparation and oral antibiotics in prevention of anastomotic leakage following restorative resection for primary rectal cancer - a systematic review and meta-analysis. Int J Colorectal Dis. 2023; 38: 129.
- Liu Y, Li B, Wei Y. New understanding of gut microbiota and colorectal anastomosis leak: A collaborative review of the current concepts. Front Cell Infect Microbiol. 2022; 12: 1022603. [CrossRef]
- Shi Y, Cui H, Wang F, Zhang Y, Xu Q, Liu D, et al. Role of gut microbiota in postoperative complications and prognosis of gastrointestinal surgery: A narrative review. Medicine [Baltimore]. 2022; 101: e29826.
- Olivas AD, Shogan BD, Valuckaite V, Zaborin A, Belogortseva N, Musch M, et al. Intestinal tissues induce an SNP mutation in Pseudomonas aeruginosa that enhances its virulence: possible role in anastomotic leak. PLoS One. 2012; 7: e44326. [CrossRef]
- Shogan BD, Belogortseva N, Luong PM, Zaborin A, Lax S, Bethel C, et al. Collagen degradation and MMP9 activation by Enterococcus faecalis contribute to intestinal anastomotic leak. Sci Transl Med. 2015; 7: 286ra68.
- Schardey HM, Kamps T, Rau HG, Gatermann S, Baretton G, Schildberg FW. Bacteria: a major pathogenic factor for anastomotic insufficiency. Antimicrob Agents Chemother. 1994; 38: 2564-7. [CrossRef]
- Hajjar R, Gonzalez E, Fragoso G, Oliero M, Alaoui AA, Calvé A, et al. Gut microbiota influence anastomotic healing in colorectal cancer surgery through modulation of mucosal proinflammatory cytokines. Gut. 2023; 72: 1143-1154.
- Hernández-González PI, Barquín J, Ortega-Ferrete A, Patón V, Ponce-Alonso M, Romero-Hernández B, et al. Anastomotic leak in colorectal cancer surgery: Contribution of gut microbiota and prediction approaches. Colorectal Dis. 2023; 25: 2187-2197. [CrossRef]
- Palmisano S, Campisciano G, Iacuzzo C, Bonadio L, Zucca A, Cosola D, et al. Role of preoperative gut microbiota on colorectal anastomotic leakage: preliminary results. Updates Surg. 2020; 72: 1013-1022.
- Lehr K, Lange UG, Hipler NM, Vilchez-Vargas R, Hoffmeister A, Feisthammel J, et al. Prediction of anastomotic insufficiency based on the mucosal microbiota prior to colorectal surgery: a proof-of-principle study. Sci Rep. 2024; 14: 15335. [CrossRef]
- Lianos GD, Frountzas M, Kyrochristou ID, Sakarellos P, Tatsis V, Kyrochristou GD, et al. What Is the Role of the Gut Microbiota in Anastomotic Leakage After Colorectal Resection? A Scoping Review of Clinical and Experimental Studies. J Clin Med. 2024; 13: 6634.
- El-Hussuna A, Qvist N, Zangenberg MS, Langkilde A, Siersma V, Hjort S, et al. No effect of anti-TNF-α agents on the surgical stress response in patients with inflammatory bowel disease undergoing bowel resections: a prospective multi-center pilot study. BMC Surg. 2018; 18: 91.
- Shwaartz C, Fields AC, Sobrero M, Cohen BD, Divino CM. Effect of Anti-TNF Agents on Postoperative Outcomes in Inflammatory Bowel Disease Patients: A Single Institution Experience. J Gastrointest Surg. 2016; 20: 1636-42. [CrossRef]
- El-Hussuna A, Andersen J, Bisgaard T, Jess P, Henriksen M, Oehlenschlager J, et al. Biologic treatment or immunomodulation is not associated with postoperative anastomotic complications in abdominal surgery for Crohn's disease. Scand J Gastroenterol. 2012; 47: 662-8.
- Kunitake H, Hodin R, Shellito PC, Sands BE, Korzenik J, Bordeianou L. Perioperative treatment with infliximab in patients with Crohn's disease and ulcerative colitis is not associated with an increased rate of postoperative complications. J Gastrointest Surg. 2008; 12: 1730-6; discussion 1736-7. [CrossRef]
- Rosenfeld G, Qian H, Bressler B. The risks of post-operative complications following pre-operative infliximab therapy for Crohn's disease in patients undergoing abdominal surgery: a systematic review and meta-analysis. J Crohns Colitis. 2013; 7: 868-77.
- Alsaleh A, Gaidos JK, Kang L, Kuemmerle JF. Timing of Last Preoperative Dose of Infliximab Does Not Increase Postoperative Complications in Inflammatory Bowel Disease Patients. Dig Dis Sci. 2016; 61: 2602-7. [CrossRef]
- Gaines S, Hyoju S, Williamson AJ, van Praagh JB, Zaborina O, Rubin DT, et al. Infliximab Does Not Promote the Presence of Collagenolytic Bacteria in a Mouse Model of Colorectal Anastomosis. J Gastrointest Surg. 2020; 24: 2637-2642. [CrossRef]

| Ref | Disease | Biologic agent | Patients | Type of sample | Main outcomes | F. prausnitzii |
|---|---|---|---|---|---|---|
| 15 | UC | TNF inhibitors | 56 | Faecal, sigmoid biopsies | Not reported | ↑ especially in responders |
| 25 | CD | Adalimumab | 15 | Rectal mucosal biopsy | ↑ Firmicutes, ↑ Bacteroides, ↑ Actinobacteria / ↓ E. coli | Non-significant changes |
| 26 | CD | Adalimumab | 20 | Faecal | ↓ Proteobacteria in responders (from 15.8% to 6.8%; p: 0.049) |
Non-significant changes |
| 27 | CD | Adalimumab | 8 | Faecal | No differences in α- and β-diversity ↑ Firmicutes and ↓ Proteobacteria in responders |
Not reported |
| 28 | UC | Adalimumab | 131 | Faecal | ↓ Burkholderia-Caballeronia-Paraburkholderia, ↓ Staphylococcus, ↑ Bifidobacterium, ↑ Dorea in responders |
Not reported |
| 29 | UC | Adalimumab | 9 | Biopsy | ↑ Dorea, ↑ Lachnospira in responders | Not reported |
| 30 | IBD | Infliximab, vedolizumab | 35 | Faecal | ↑ α-diversity | Not reported |
| 31 | IBD + EA | TNF inhibitors | 20 | Faecal | No differences in α- and β-diversity ↑ Lachnospiraceae and ↑ Coprococcus |
Not reported |
| 32 | CD | Infliximab | 49 | Faecal | ↑ α-diversity ↑ Bacteroidetes, ↑ Firmicutes, ↓ Enterobacterales ↑ SCFA-producing taxa (Lachnospira, Blautia) |
Not reported |
| 33 | IBD | TNF inhibitors | 27 | Faecal | ↑ α-diversity only in responders ↑ Firmicutes, ↑ Lachnospiraceae in responders |
↑ F. prausnitzii/E. coli (F/E) ratio in responders |
| 34 | IBD | Infliximab | 40 | Faecal | ↑ Shannon in patients with mucosal healing ↑ Blautia, ↑ Bacteroides, ↓ Prevotella in patients with mucosal healing |
↑ in patients with mucosal healing |
| 35 | UC | Infliximab, etrolizumab | 287 | Faecal | Shannon diversity and species richness ↑ in remitters Bifidobacterium breve ↓ |
More abundant in non-remitters |
| 36 | IBD | Ustekinumab | 11 | Faecal | No differences in α- and β-diversity No differences in abundances of phyla or genera |
Not reported |
| 37 | CD | Ustekinumab | 306 | Faecal | α-diversity of responders changed over time (↑ from baseline to 4 weeks, ↓ from 4 to 6, and ↑ than baseline at 22 weeks) | More abundant at baseline in responders |
| 39 | UC | Vedolizumab | 29 | Faecal | ↑ Verrucomicrobiota in responders | Not reported |
| 40 | IBD | Vedolizumab | 85 | Faecal | In CD: Bifidobacterium longum, Eggerthella, Ruminococcus gnavus, Roseburia inulinivorans, Veillonella parvula ↓ in responders. In UC: Strepotococcus salivarium ↑ in non-responders. |
Not reported |
| 41 | UC | Vedolizumab | 45 | Faecal | ↑ Firmicutes | Not reported |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




