Submitted:
18 June 2025
Posted:
20 June 2025
You are already at the latest version
Abstract
Keywords:
Introduction
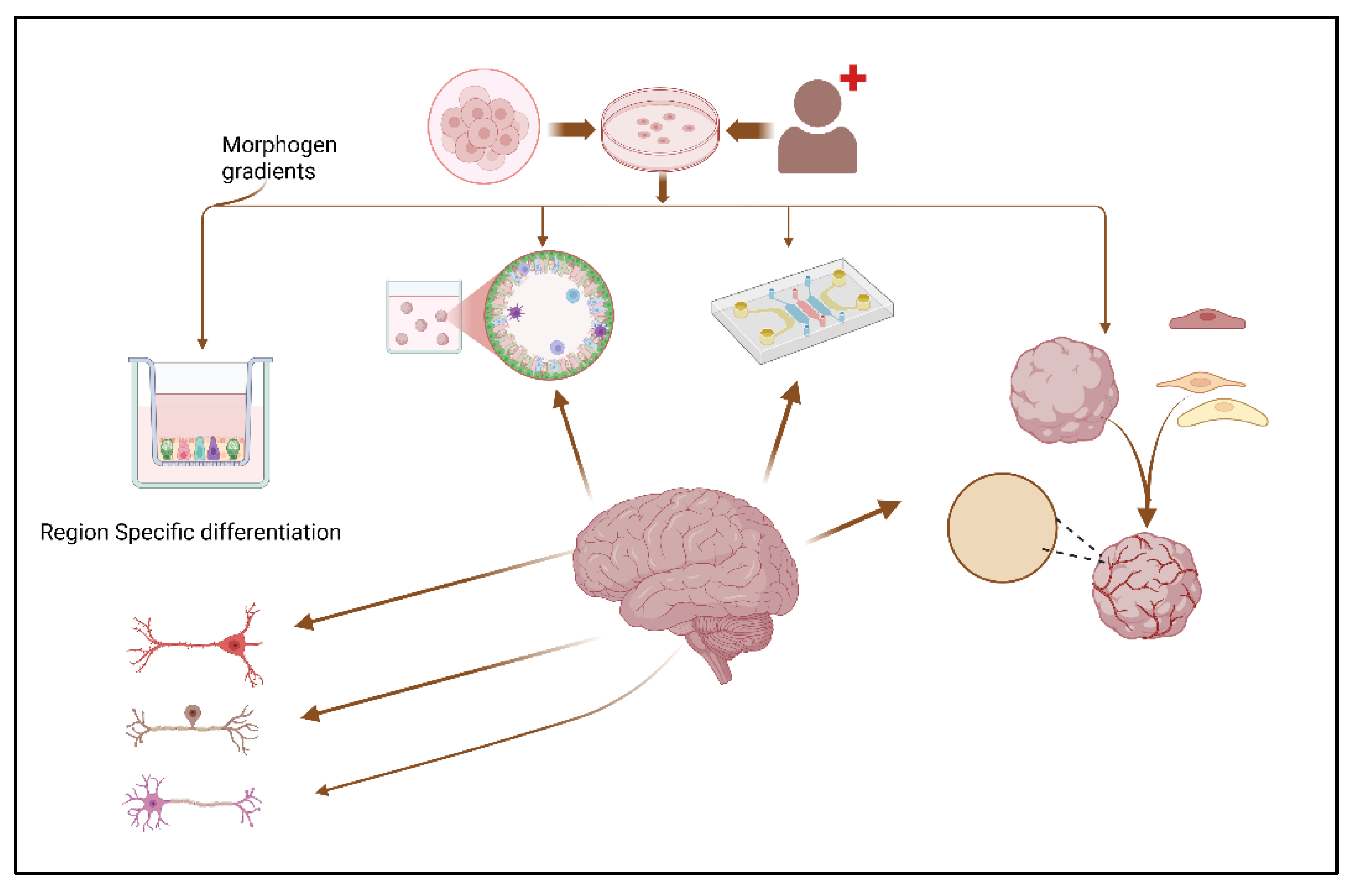
2. Advances in Brain Organoid Methodologies
3. Unravelling Alzheimer's and Parkinson's: Innovations Through Organoids and Organs-on-a-Chip
3.1. Parkinson’s Disease: Insights from Organoids and beyond
3.1.1. Parkinson's Disease Drug Discovery
3.2. Alzheimer’s Disease: A Closer Look Through Advanced Models
3.2.1. Alzheimer's Disease Drug Discovery
4. Technical Innovations in Organoid Systems
4.1. Incorporation of Neuronal Diversity and Complexity
4.2. Achievements in Vascularization
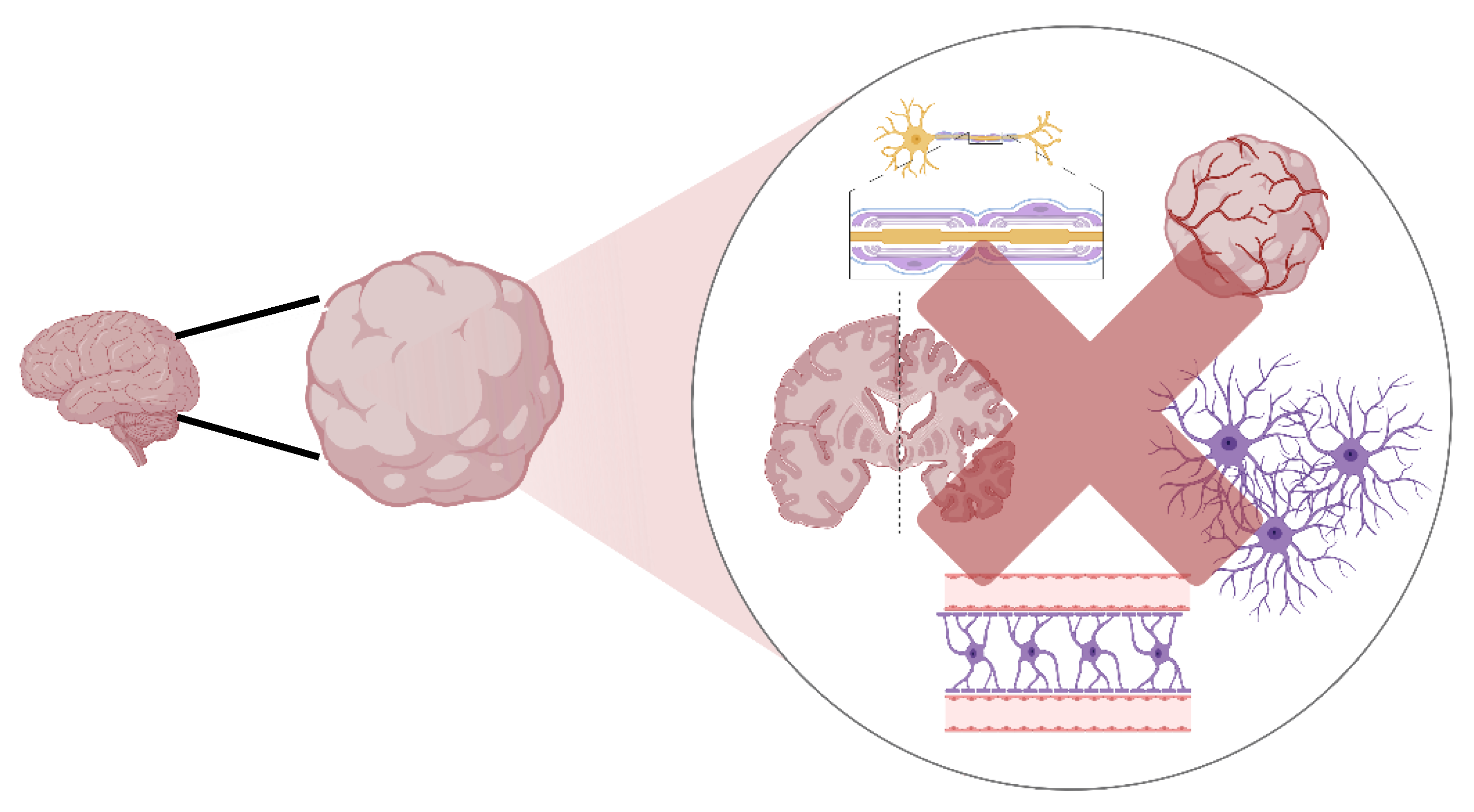
5. Limitations
6. Conclusion
Authorship Contribution Statement
Funding
Ethical Statements
Data Availability
Declaration of generative AI and AI-assisted technologies in the writing process
Declaration of Competing Interest
Clinical trial number
References
- Bassett DS, Sporns O (2017) Network neuroscience. Nat Neurosci 20:353–364. [CrossRef]
- Insel TR (2010) Rethinking schizophrenia. Nature 468:187–193. [CrossRef]
- Marín O (2016) Developmental timing and critical windows for the treatment of psychiatric disorders. Nat Med 22:1229–1238. [CrossRef]
- Takahashi K, Tanabe K, Ohnuki M, et al (2007) Induction of Pluripotent Stem Cells from Adult Human Fibroblasts by Defined Factors. Cell 131:861–872. [CrossRef]
- Lancaster MA, Renner M, Martin C-A, et al (2013) Cerebral organoids model human brain development and microcephaly. Nature 501:373–379. [CrossRef]
- Di Lullo E, Kriegstein AR (2017) The use of brain organoids to investigate neural development and disease. Nat Rev Neurosci 18:573–584. [CrossRef]
- Bassett AR (2017) Editing the genome of hiPSC with CRISPR/Cas9: disease models. Mamm Genome 28:348–364. [CrossRef]
- Amin ND, Paşca SP (2018) Building Models of Brain Disorders with Three-Dimensional Organoids. Neuron 100:389–405. [CrossRef]
- Quadrato G, Nguyen T, Macosko EZ, et al (2017) Cell diversity and network dynamics in photosensitive human brain organoids. Nature 545:48–53. [CrossRef]
- Kelava I, Lancaster MA (2016) Dishing out mini-brains: Current progress and future prospects in brain organoid research. Dev Biol 420:199–209. [CrossRef]
- Sloan SA, Andersen J, Pașca AM, et al (2018) Generation and assembly of human brain region–specific three-dimensional cultures. Nat Protoc 13:2062–2085. [CrossRef]
- Park SE, Georgescu A, Huh D (2019) Organoids-on-a-chip. Science 364:960–965. [CrossRef]
- Stiles J, Jernigan TL (2010) The Basics of Brain Development. Neuropsychol Rev 20:327–348. [CrossRef]
- Silbereis JC, Pochareddy S, Zhu Y, et al (2016) The Cellular and Molecular Landscapes of the Developing Human Central Nervous System. Neuron 89:248–268. [CrossRef]
- Dawson TM, Golde TE, Lagier-Tourenne C (2018) Animal models of neurodegenerative diseases. Nat Neurosci 21:1370–1379. [CrossRef]
- Eiraku M, Watanabe K, Matsuo-Takasaki M, et al (2008) Self-Organized Formation of Polarized Cortical Tissues from ESCs and Its Active Manipulation by Extrinsic Signals. Cell Stem Cell 3:519–532. [CrossRef]
- Qian X, Song H, Ming G (2019) Brain organoids: advances, applications and challenges. Development 146:dev166074. [CrossRef]
- Paşca SP (2019) Assembling human brain organoids. Science 363:126–127. [CrossRef]
- Das U, Chandramouli L, Uttarkar A, et al (2025) Discovery of natural compounds as novel FMS-like tyrosine kinase-3 (FLT3) therapeutic inhibitors for the treatment of acute myeloid leukemia: An in-silico approach. Asp Mol Med 5:100058. [CrossRef]
- Trujillo CA, Gao R, Negraes PD, et al (2019) Complex Oscillatory Waves Emerging from Cortical Organoids Model Early Human Brain Network Development. Cell Stem Cell 25:558-569.e7. [CrossRef]
- Miura Y, Li M-Y, Birey F, et al (2020) Generation of human striatal organoids and cortico-striatal assembloids from human pluripotent stem cells. Nat Biotechnol 38:1421–1430. [CrossRef]
- Bagley JA, Reumann D, Bian S, et al (2017) Fused cerebral organoids model interactions between brain regions. Nat Methods 14:743–751. [CrossRef]
- Osaki T, Uzel SGM, Kamm RD (2020) On-chip 3D neuromuscular model for drug screening and precision medicine in neuromuscular disease. Nat Protoc 15:421–449. [CrossRef]
- Xiang Y, Tanaka Y, Patterson B, et al (2017) Fusion of Regionally Specified hPSC-Derived Organoids Models Human Brain Development and Interneuron Migration. Cell Stem Cell 21:383-398.e7. [CrossRef]
- Wang H, Ning X, Zhao F, et al (2024) Human organoids-on-chips for biomedical research and applications. Theranostics 14:788–818. [CrossRef]
- Huang D, Zhu X, Ye S, et al (2024) Tumour circular RNAs elicit anti-tumour immunity by encoding cryptic peptides. Nature 625:593–602. [CrossRef]
- Maoz BM, Herland A, FitzGerald EA, et al (2018) A linked organ-on-chip model of the human neurovascular unit reveals the metabolic coupling of endothelial and neuronal cells. Nat Biotechnol 36:865–874. [CrossRef]
- Vatine GD, Barrile R, Workman MJ, et al (2019) Human iPSC-Derived Blood-Brain Barrier Chips Enable Disease Modeling and Personalized Medicine Applications. Cell Stem Cell 24:995-1005.e6. [CrossRef]
- Nzou G, Wicks RT, Wicks EE, et al (2018) Human Cortex Spheroid with a Functional Blood Brain Barrier for High-Throughput Neurotoxicity Screening and Disease Modeling. Sci Rep 8:7413. [CrossRef]
- Skylar-Scott MA, Uzel SGM, Nam LL, et al (2019) Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci Adv 5:eaaw2459. [CrossRef]
- Corrò C, Novellasdemunt L, Li VSW (2020) A brief history of organoids. Am J Physiol-Cell Physiol 319:C151–C165. [CrossRef]
- Shin H, Jeong S, Lee J-H, et al (2021) 3D high-density microelectrode array with optical stimulation and drug delivery for investigating neural circuit dynamics. Nat Commun 12:492. [CrossRef]
- Das U, Banerjee S, Sarkar M, et al (2024) Circular RNA vaccines: Pioneering the next-gen cancer immunotherapy. Cancer Pathog Ther S2949713224000892. [CrossRef]
- Zhang X, Lopez-Berestein G, Sood AK, Calin GA (2016) Profiling Long Noncoding RNA Expression Using Custom-Designed Microarray. In: Feng Y, Zhang L (eds) Long Non-Coding RNAs. Springer New York, New York, NY, pp 33–41.
- Wang Y, Chang RYK, Britton WJ, Chan H-K (2022) Advances in the development of antimicrobial peptides and proteins for inhaled therapy. Adv Drug Deliv Rev 180:114066. [CrossRef]
- Benito-Kwiecinski S, Lancaster MA (2020) Brain Organoids: Human Neurodevelopment in a Dish. Cold Spring Harb Perspect Biol 12:a035709. [CrossRef]
- Prager BC, Xie Q, Bao S, Rich JN (2019) Cancer Stem Cells: The Architects of the Tumor Ecosystem. Cell Stem Cell 24:41–53. [CrossRef]
- Kang UJ, Boehme AK, Fairfoul G, et al (2019) Comparative study of cerebrospinal fluid α-synuclein seeding aggregation assays for diagnosis of Parkinson’s disease. Mov Disord 34:536–544. [CrossRef]
- Yemini E, Lin A, Nejatbakhsh A, et al (2021) NeuroPAL: A Multicolor Atlas for Whole-Brain Neuronal Identification in C. elegans. Cell 184:272-288.e11. [CrossRef]
- Chortos A (2022) High current hydrogels: Biocompatible electromechanical energy sources. Cell 185:2653–2654. [CrossRef]
- Wu S, Wagner G (2021) Deep computational analysis details dysregulation of eukaryotic translation initiation complex eIF4F in human cancers. Cell Syst 12:907-923.e6. [CrossRef]
- Tsuchiya M, Tachibana N, Nagao K, et al (2023) Organelle-selective click labeling coupled with flow cytometry allows pooled CRISPR screening of genes involved in phosphatidylcholine metabolism. Cell Metab 35:1072-1083.e9. [CrossRef]
- Shin AE, Giancotti FG, Rustgi AK (2023) Metastatic colorectal cancer: mechanisms and emerging therapeutics. Trends Pharmacol Sci 44:222–236. [CrossRef]
- Gleeson P, Cantarelli M, Marin B, et al (2019) Open Source Brain: A Collaborative Resource for Visualizing, Analyzing, Simulating, and Developing Standardized Models of Neurons and Circuits. Neuron 103:395-411.e5. [CrossRef]
- Ackerman SD, Perez-Catalan NA, Freeman MR, Doe CQ (2021) Astrocytes close a motor circuit critical period. Nature 592:414–420. [CrossRef]
- Ameen TB, Kashif SN, Abbas SMI, et al (2024) Unraveling Alzheimer’s: the promise of aducanumab, lecanemab, and donanemab. Egypt J Neurol Psychiatry Neurosurg 60:72. [CrossRef]
- Park J-C, Jang S-Y, Lee D, et al (2021) A logical network-based drug-screening platform for Alzheimer’s disease representing pathological features of human brain organoids. Nat Commun 12:280. [CrossRef]
- Jusop AS, Thanaskody K, Tye GJ, et al (2023) Development of brain organoid technology derived from iPSC for the neurodegenerative disease modelling: a glance through. Front Mol Neurosci 16:1173433. [CrossRef]
- Wray S (2021) Modelling neurodegenerative disease using brain organoids. Semin Cell Dev Biol 111:60–66. [CrossRef]
- Hofer M, Lutolf MP (2021) Engineering organoids. Nat Rev Mater 6:402–420. [CrossRef]
- Zheng L, Zhan Y, Wang C, et al (2024) Technological advances and challenges in constructing complex gut organoid systems. Front Cell Dev Biol 12:1432744. [CrossRef]
- Das U, Banerjee S, Sarkar M (2025) Bibliometric analysis of circular RNA cancer vaccines and their emerging impact. Vacunas 500391. [CrossRef]
- Huang S, Huang F, Zhang H, et al (2022) In vivo development and single-cell transcriptome profiling of human brain organoids. Cell Prolif 55:e13201. [CrossRef]
- Kathuria A, Lopez-Lengowski K, Vater M, et al (2020) Transcriptome analysis and functional characterization of cerebral organoids in bipolar disorder. Genome Med 12:34. [CrossRef]
- Brémond Martin C, Simon Chane C, Clouchoux C, Histace A (2021) Recent Trends and Perspectives in Cerebral Organoids Imaging and Analysis. Front Neurosci 15:629067. [CrossRef]
- Sandoval SO, Cappuccio G, Kruth K, et al (2024) Rigor and reproducibility in human brain organoid research: Where we are and where we need to go. Stem Cell Rep 19:796–816. [CrossRef]
- Smirnova L, Caffo BS, Gracias DH, et al (2023) Organoid intelligence (OI): the new frontier in biocomputing and intelligence-in-a-dish. Front Sci 1:1017235. [CrossRef]
- Wadan A-HS (2025) Organoid intelligence and biocomputing advances: Current steps and future directions. Brain Organoid Syst Neurosci J 3:8–14. [CrossRef]
- Mansour AA, Schafer ST, Gage FH (2021) Cellular complexity in brain organoids: Current progress and unsolved issues. Semin Cell Dev Biol 111:32–39. [CrossRef]
- Acharya P, Choi NY, Shrestha S, et al (2024) Brain organoids: A revolutionary tool for modeling neurological disorders and development of therapeutics. Biotechnol Bioeng 121:489–506. [CrossRef]
- Rauth S, Karmakar S, Batra SK, Ponnusamy MP (2021) Recent advances in organoid development and applications in disease modeling. Biochim Biophys Acta BBA - Rev Cancer 1875:188527. [CrossRef]
- Das U, Chanda T, Kumar J, Peter A (2024) Discovery of Natural MCL1 Inhibitors using Pharmacophore modelling, QSAR, Docking, ADMET, Molecular Dynamics, and DFT Analysis.
- Atamian A, Birtele M, Hosseini N, et al (2024) Human cerebellar organoids with functional Purkinje cells. Cell Stem Cell 31:39-51.e6. [CrossRef]
- Uzquiano A, Kedaigle AJ, Pigoni M, et al (2022) Proper acquisition of cell class identity in organoids allows definition of fate specification programs of the human cerebral cortex. Cell 185:3770-3788.e27. [CrossRef]
- Sidhaye J, Knoblich JA (2021) Brain organoids: an ensemble of bioassays to investigate human neurodevelopment and disease. Cell Death Differ 28:52–67. [CrossRef]
- Molnár Z, Clowry GJ, Šestan N, et al (2019) New insights into the development of the human cerebral cortex. J Anat 235:432–451. [CrossRef]
- Das U, Uttarkar A, Kumar J, Niranjan V (2025) In silico exploration natural compounds for the discovery of novel dnmt3a inhibitors as potential therapeutic agents for acute myeloid leukaemia. Silico Res Biomed 100006. [CrossRef]
- Camp JG, Badsha F, Florio M, et al (2015) Human cerebral organoids recapitulate gene expression programs of fetal neocortex development. Proc Natl Acad Sci 112:15672–15677. [CrossRef]
- Lu Y, Shiau F, Yi W, et al (2020) Single-Cell Analysis of Human Retina Identifies Evolutionarily Conserved and Species-Specific Mechanisms Controlling Development. Dev Cell 53:473-491.e9. [CrossRef]
- Zou W, Lv Y, Zhang S, et al (2024) Lysosomal dynamics regulate mammalian cortical neurogenesis. Dev Cell 59:64-78.e5. [CrossRef]
- Ziffra RS, Kim CN, Ross JM, et al (2021) Single-cell epigenomics reveals mechanisms of human cortical development. Nature 598:205–213. [CrossRef]
- Lu T, Wang M, Zhou W, et al (2024) Decoding transcriptional identity in developing human sensory neurons and organoid modeling. Cell 187:7374-7393.e28. [CrossRef]
- Saglam-Metiner P, Devamoglu U, Filiz Y, et al (2023) Spatio-temporal dynamics enhance cellular diversity, neuronal function and further maturation of human cerebral organoids. Commun Biol 6:173. [CrossRef]
- Giandomenico SL, Mierau SB, Gibbons GM, et al (2019) Cerebral organoids at the air–liquid interface generate diverse nerve tracts with functional output. Nat Neurosci 22:669–679. [CrossRef]
- Xue W, Li H, Xu J, et al (2024) Effective cryopreservation of human brain tissue and neural organoids. Cell Rep Methods 4:100777. [CrossRef]
- Jacob F, Schnoll JG, Song H, Ming G (2021) Building the brain from scratch: Engineering region-specific brain organoids from human stem cells to study neural development and disease. In: Current Topics in Developmental Biology.
- Susaimanickam PJ, Kiral FR, Park I-H (2022) Region Specific Brain Organoids to Study Neurodevelopmental Disorders. Int J Stem Cells 15:26–40. [CrossRef]
- Castiglione H, Vigneron P-A, Baquerre C, et al (2022) Human Brain Organoids-on-Chip: Advances, Challenges, and Perspectives for Preclinical Applications. Pharmaceutics 14:2301. [CrossRef]
- Saglam-Metiner P, Yildirim E, Dincer C, et al (2024) Humanized brain organoids-on-chip integrated with sensors for screening neuronal activity and neurotoxicity. Microchim Acta 191:71. [CrossRef]
- Zlokovic BV, Zlokovic BV, Apuzzo MLJ (1998) Strategies to Circumvent Vascular Barriers of the Central Nervous System. Neurosurgery 43:877–878. [CrossRef]
- Dunn KM, Nelson MT (2014) Neurovascular signaling in the brain and the pathological consequences of hypertension. Am J Physiol-Heart Circ Physiol 306:H1–H14. [CrossRef]
- Sweeney MD, Kisler K, Montagne A, et al (2018) The role of brain vasculature in neurodegenerative disorders. Nat Neurosci 21:1318–1331. [CrossRef]
- Mansour AA, Gonçalves JT, Bloyd CW, et al (2018) An in vivo model of functional and vascularized human brain organoids. Nat Biotechnol 36:432–441. [CrossRef]
- Vodyanik MA, Yu J, Zhang X, et al (2010) A Mesoderm-Derived Precursor for Mesenchymal Stem and Endothelial Cells. Cell Stem Cell 7:718–729. [CrossRef]
- Sun X-Y, Luo Z-G (2022) Vascularizing the brain organoids. J Mol Cell Biol 14:mjac040. [CrossRef]
- Shi Y, Sun L, Wang M, et al (2020) Vascularized human cortical organoids (vOrganoids) model cortical development in vivo. PLOS Biol 18:e3000705. [CrossRef]
- Kong D, Park KH, Kim D-H, et al (2023) Cortical-blood vessel assembloids exhibit Alzheimer’s disease phenotypes by activating glia after SARS-CoV-2 infection. Cell Death Discov 9:32. [CrossRef]
- Sun X-Y, Ju X-C, Li Y, et al (2022) Generation of vascularized brain organoids to study neurovascular interactions. eLife 11:e76707. [CrossRef]
- Patsch C, Challet-Meylan L, Thoma EC, et al (2015) Generation of vascular endothelial and smooth muscle cells from human pluripotent stem cells. Nat Cell Biol 17:994–1003. [CrossRef]
- Morita R, Suzuki M, Kasahara H, et al (2015) ETS transcription factor ETV2 directly converts human fibroblasts into functional endothelial cells. Proc Natl Acad Sci 112:160–165. [CrossRef]
- Cakir B, Xiang Y, Tanaka Y, et al (2019) Engineering of human brain organoids with a functional vascular-like system. Nat Methods 16:1169–1175. [CrossRef]
- Kofman S, Sun X, Ogbolu VC, et al (2023) Vascularized Brain Assembloids with Enhanced Cellular Complexity Provide Insights into The Cellular Deficits of Tauopathy.
- Das U (2025) Emerging trends and research landscape of the tumor microenvironment in head-and-neck cancer: A comprehensive bibliometric analysis. Cancer Plus 0:025060008. [CrossRef]
- Bagchi S, Chhibber T, Lahooti B, et al (2019) In-vitro blood-brain barrier models for drug screening and permeation studies: an overview. Drug Des Devel Ther Volume 13:3591–3605. [CrossRef]
- Van Der Helm MW, Van Der Meer AD, Eijkel JCT, et al (2016) Microfluidic organ-on-chip technology for blood-brain barrier research. Tissue Barriers 4:e1142493. [CrossRef]
- Bhaduri A, Andrews MG, Mancia Leon W, et al (2020) Cell stress in cortical organoids impairs molecular subtype specification. Nature 578:142–148. [CrossRef]
- Short PJ, McRae JF, Gallone G, et al (2018) De novo mutations in regulatory elements in neurodevelopmental disorders. Nature 555:611–616. [CrossRef]
- Wang Y, Wang L, Guo Y, et al (2018) Engineering stem cell-derived 3D brain organoids in a perfusable organ-on-a-chip system. RSC Adv 8:1677–1685. [CrossRef]
- Marton RM, Miura Y, Sloan SA, et al (2019) Differentiation and maturation of oligodendrocytes in human three-dimensional neural cultures. Nat Neurosci 22:484–491. [CrossRef]
- Ormel PR, Vieira De Sá R, Van Bodegraven EJ, et al (2018) Microglia innately develop within cerebral organoids. Nat Commun 9:4167. [CrossRef]
- Lin Y-T, Seo J, Gao F, et al (2018) APOE4 Causes Widespread Molecular and Cellular Alterations Associated with Alzheimer’s Disease Phenotypes in Human iPSC-Derived Brain Cell Types. Neuron 98:1141-1154.e7. [CrossRef]
- Kim J, Koo B-K, Knoblich JA (2020) Human organoids: model systems for human biology and medicine. Nat Rev Mol Cell Biol 21:571–584. [CrossRef]
- Ene HM, Karry R, Farfara D, Ben-Shachar D (2023) Mitochondria play an essential role in the trajectory of adolescent neurodevelopment and behavior in adulthood: evidence from a schizophrenia rat model. Mol Psychiatry 28:1170–1181. [CrossRef]
- Sung JH, Wang YI, Narasimhan Sriram N, et al (2019) Recent Advances in Body-on-a-Chip Systems. Anal Chem 91:330–351. [CrossRef]
- Wang F, Liu Y, Fu C, et al (2021) Paper-Based Bipolar Electrode Electrochemiluminescence Platform for Detection of Multiple miRNAs. Anal Chem 93:1702–1708. [CrossRef]
- Lu X, Stepanov P, Yang W, et al (2019) Superconductors, orbital magnets and correlated states in magic-angle bilayer graphene. Nature 574:653–657. [CrossRef]
- Birey F, Andersen J, Makinson CD, et al (2017) Assembly of functionally integrated human forebrain spheroids. Nature 545:54–59. [CrossRef]
- Berg J, Sorensen SA, Ting JT, et al (2021) Human neocortical expansion involves glutamatergic neuron diversification. Nature 598:151–158. [CrossRef]
- Abud EM, Ramirez RN, Martinez ES, et al (2017) iPSC-Derived Human Microglia-like Cells to Study Neurological Diseases. Neuron 94:278-293.e9. [CrossRef]
- Wang YI, Abaci HE, Shuler ML (2017) Microfluidic blood–brain barrier model provides in vivo-like barrier properties for drug permeability screening. Biotechnol Bioeng 114:184–194. [CrossRef]
- Choi YJ, Heo K, Park HS, et al (2016) The resveratrol analog HS-1793 enhances radiosensitivity of mouse-derived breast cancer cells under hypoxic conditions. Int J Oncol 49:1479–1488. [CrossRef]
- Studer L, Vera E, Cornacchia D (2015) Programming and Reprogramming Cellular Age in the Era of Induced Pluripotency. Cell Stem Cell 16:591–600. [CrossRef]
- Lancaster MA, Knoblich JA (2014) Organogenesis in a dish: Modeling development and disease using organoid technologies. Science 345:1247125. [CrossRef]
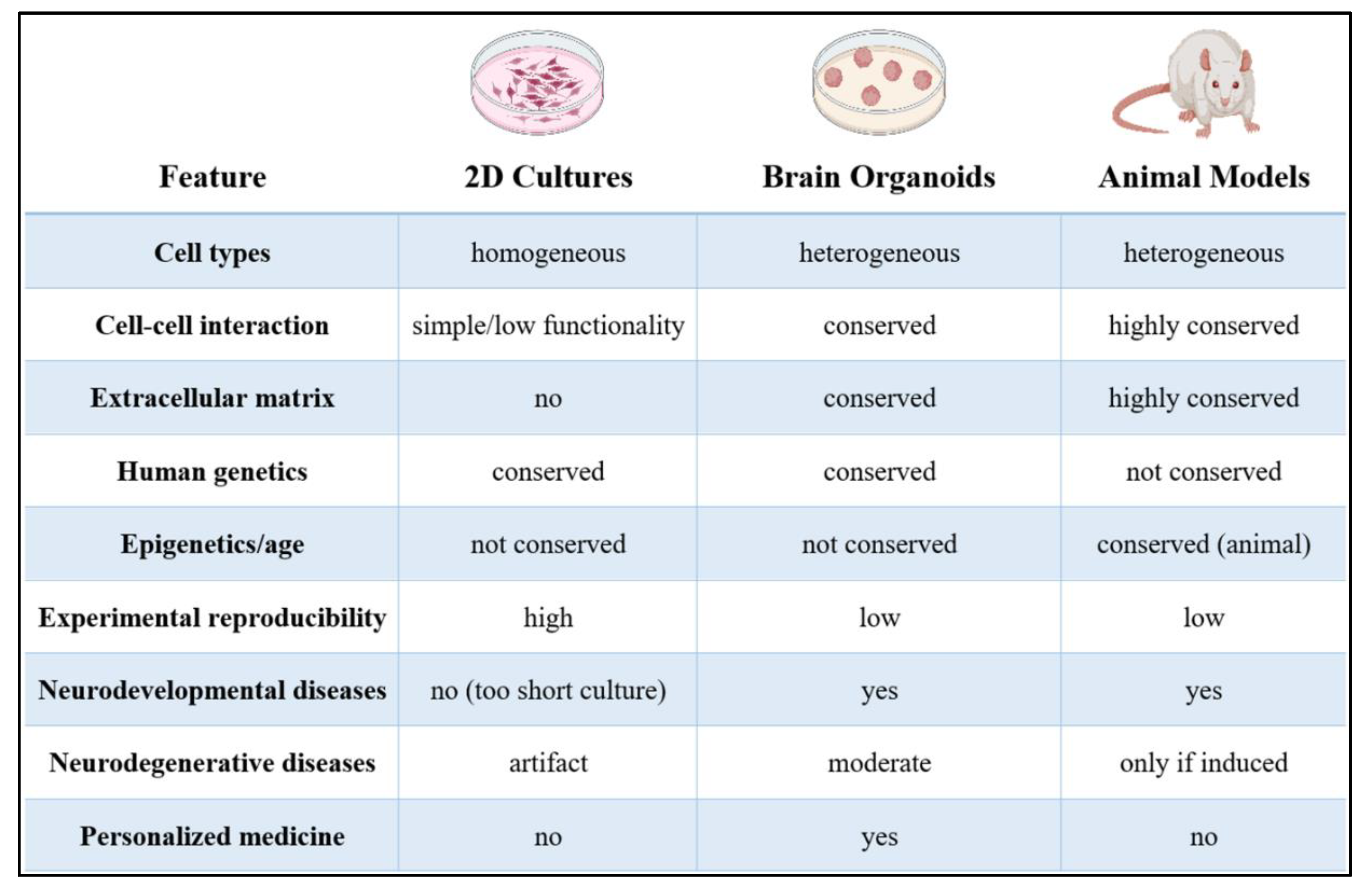
| Strategy | Description | Advantages | Limitations | Key References |
| Assembloids | Fusion of multiple region-specific organoids to model inter-regional interactions | - Models circuit formation between distinct brain regions - Enables study of migration patterns - Allows investigation of region-specific pathologies |
- Complex to generate reproducibly - Limited by a lack of defined axon guidance cues - Challenging to analyze interactions quantitatively |
[24,106,107] |
| Brain-on-a-chip | Integration of organoids with microfluidic platforms to provide dynamic flow and environmental control | - Improves nutrient/oxygen delivery - Enables controlled exposure to factors - Allows mechanical stimulation |
- Technical complexity - Higher cost - Requires specialized expertise |
[12,105] |
| Extended culture periods | Maintaining organoids in culture for prolonged periods (>1 year) | - Allows development of mature features - Models later developmental processes - Enables aging-related studies |
- Resource-intensive - Core necrosis in larger organoids - Increased variability over time |
[11,108] |
| Co-culture with vascular cells | Addition of endothelial cells to promote vascularization | - Reduces necrotic core formation - Improves nutrient delivery - Models the blood-brain barrier |
- Difficult to achieve full vascularization - Usually affects only the periphery - Integration challenges |
[83,91,108] |
| Addition of microglia | Incorporation of iPSC-derived microglia or primitive microglia | - Models neuroinflammatory responses - Enables study of immune-neural interactions - Critical for modeling neurodegenerative diseases |
- Difficult to achieve physiological ratios - Requires complex protocols - Microglia maturation challenges |
[17,100,109] |
| Air-liquid interface culture | Growing organoids at an air-liquid interface to improve oxygen diffusion | - Reduces necrosis - Promotes growth and maturation - Enhances neural network formation |
- Alters 3D structure - Technical challenges - Different from in vivo environment |
[74,86,96] |
| Transcription factor overexpression | Forced expression of maturation-promoting factors | - Accelerates maturation - Enhances specific cell type development - Allows modeling of age-related features |
- May produce artificial phenotypes - Potentially disrupts developmental timing - Technical complexity |
[88,110] |
| Chemical aging induction | Addition of compounds that induce aging-like phenotypes (e.g., progerin, oxidative stressors) | - Rapidly induces aging phenotypes - Enables study of late-onset disorders - Reduces experimental timeline |
- May not recapitulate natural aging processes - Potential off-target effects - Limited validation against true aging |
[108,111,112] |
| Growth factor supplementation | Strategic addition of growth factors and morphogens | - Promotes cell-type-specific development - Enhances maturation - Directs regional identity |
- May skew developmental programs - Batch-to-batch variability - Cost implications |
[9,64,113] |
| Genetic integration of reporters | Incorporation of fluorescent reporters for real-time monitoring | - Allows live tracking of development - Enables targeted analysis - Facilitates cell isolation |
- Technical complexity - Potential disruption of normal function - Limited to specific markers |
[5,6] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




