Submitted:
17 June 2025
Posted:
19 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Case Description
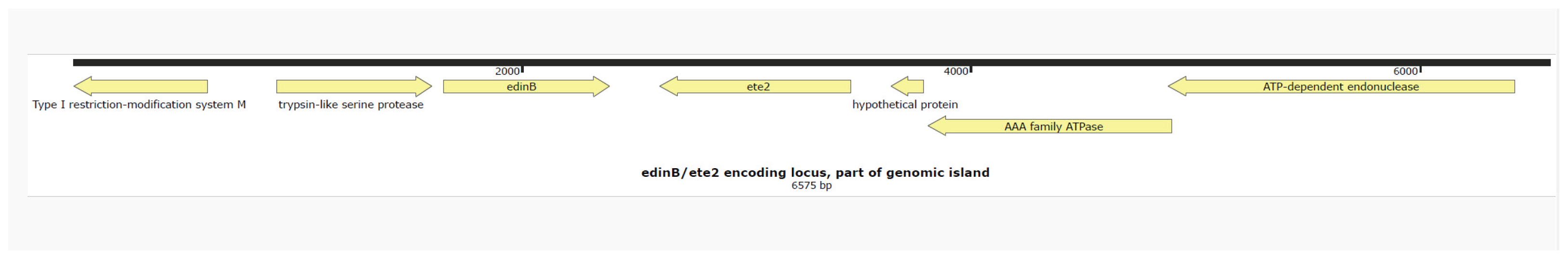
2.1. Isolate Characterisation
3. Discussion
Authors’ contributions
Funding statement
Institutional Review Board Statement
Informed Consent Statement
Data availability
Conflicts of Interest
References
- Sabat AJ, Wouthuyzen-Bakker M, Rondags A, Hughes L, Akkerboom V, Koutsopetra O, et al. Case Report: Necrotizing fasciitis caused by Staphylococcus aureus positive for a new sequence variant of exfoliative toxin E. Front. Genet. 2002;13:964358. [CrossRef]
- Lisotto, P., Raangs, E. C., Couto, N., Rosema, S., Lokate, M., Zhou, X., Friedrich, et al. Long-read sequencing-based in silico phage typing of vancomycin-resistant Enterococcus faecium. BMC Genomics. 2021, 22(1), Article 758. [CrossRef]
- Strauß L, Ruffing U, Abdulla S, Alabi A, Akulenko R, Garrine M, et al. Detecting Staphylococcus aureus Virulence and Resistance Genes: a Comparison of Whole-Genome Sequencing and DNA Microarray Technology. J Clin Microbiol. 2016 Apr;54(4):1008-16. [CrossRef]
- Baig S, Rhod Larsen A, Martins Simões P, Laurent F, Johannesen TB, Lilje B, Tristan A, et al. Evolution and Population Dynamics of Clonal Complex 152 Community-Associated Methicillin-Resistant Staphylococcus aureus. mSphere. 2020 Jul 1;5(4):e00226-20. [CrossRef]
- Imanishi I, Nicolas A, Caetano AB, Castro TLP, Tartaglia NR, Mariutti R, et al. Exfoliative toxin E, a new Staphylococcus aureus virulence factor with host-specific activity. Sci Rep. 2019 Nov 8;9(1):16336.
- Bukowski M, Wladyka B, Dubin G. Exfoliative toxins of Staphylococcus aureus. Toxins (Basel). 2010 May;2(5):1148-65. [CrossRef]
- Wiegers W, Just I, Müller H, Hellwig A, Traub P, Aktories K. Alteration of the cytoskeleton of mammalian cells cultured in vitro by Clostridium botulinum C2 toxin and C3 ADP-ribosyltransferase. Eur J Cell Biol. 1991 Apr;54(2):237-45.
- Boyer L, Doye A, Rolando M, Flatau G, Munro P, Gounon P, et al. Induction of transient macroapertures in endothelial cells through RhoA inhibition by Staphylococcus aureus factors. J Cell Biol. 2006 Jun 5;173(5):809-19.
- Courjon J, Munro P, Benito Y, Visvikis O, Bouchiat C, Boyer L, et al. EDIN-B Promotes the Translocation of Staphylococcus aureus to the Bloodstream in the Course of Pneumonia. Toxins. 2015; 7(10):4131-4142. [CrossRef]
- Czech A, Yamaguchi T, Bader L, Linder S, Kaminski K, Sugai M, Aepfelbacher M. Prevalence of Rho-inactivating epidermal cell differentiation inhibitor toxins in clinical Staphylococcus aureus isolates. J Infect Dis. 2001 Sep 15;184(6):785-8. [CrossRef]
- Yamaguchi T, Nishifuji K, Sasaki M, Fudaba Y, Aepfelbacher M, Takata T, et al. Identification of the Staphylococcus aureus etd pathogenicity island which encodes a novel exfoliative toxin, ETD, and EDIN-B. Infect Immun. 2002 Oct;70(10):5835-45.
- Egyir B, Owusu-Nyantakyi C, Bortey A, Rabbi Amuasi G, Owusu FA, et al. . Whole genome sequencing revealed high proportions of ST152 MRSA among clinical Staphylococcus aureus isolates from ten hospitals in Ghana. mSphere 9:e00446-24. [CrossRef]
- Shittu AO, Okon K, Adesida S, Oyedara O, Witte W, Strommenger B, et al. Antibiotic resistance and molecular epidemiology of Staphylococcus aureus in Nigeria. BMC Microbiol. 2011 May 5;11:92. [CrossRef]
- Gillet Y, Issartel B, Vanhems P, Fournet JC, Lina Get al. Association between Staphylococcus aureus strains carrying gene for Panton-Valentine leukocidin and highly lethal necrotising pneumonia in young immunocompetent patients. Lancet. 2002 Mar 2;359(9308):753-9. [CrossRef]
- Allou N, Allyn J, Traversier N, Baron M, Blondé R, Dupieux C, et al. SARS-CoV-2 with Panton-Valentine leukocidin-producing Staphylococcus aureus healthcare-associated pneumonia in the Indian Ocean. Heliyon. 2022 Sep;8(9):e10422. [CrossRef]
- Larsen SAH, Kyhl K, Baig S, Petersen A, Av Steinum MR, Clemmensen S, et al. Life-Threatening Necrotizing Pneumonia with Panton-Valentine Leukocidin-Producing, Methicillin-Sensitive Staphylococcus aureus in a Healthy Male Co-Infected with Influenza B. Infect Dis Rep. 2021 Dec 26;14(1):12-19.
- Löffler B, Niemann S, Ehrhardt C, Horn D, Lanckohr C, Lina G, et al. Pathogenesis of Staphylococcus aureus necrotizing pneumonia: the role of PVL and an influenza coinfection. Expert Rev Anti Infect Ther. 2013 Oct;11(10):1041-51.
- Otto, M. Staphylococcus aureus toxins. Current Opinion in Microbiology. 2014, 17:32–37.

| Gene | Virulence factor |
| aur | ACME |
| cap5H | capsule |
| cap5J | capsule |
| cap5K | capsule |
| ebpS | adhesion |
| edinB | exotoxin |
| etE2 | toxin |
| eno | adhesion |
| hl | hemolysin |
| hla | hemolysin |
| hlb-intact | hemolysin |
| hlgA | hemolysin |
| hlgB | hemolysin |
| hlIII | hemolysin |
| icaA | adhesion |
| icaD | biofilm |
| isaB | antigen |
| isdA | MSCRAMM |
| lukF-PV | leukotoxin |
| lukS-PV | PVL |
| sak | immune-evasion |
| scn | immune-evasion |
| setB1 | superantigen-like |
| setB3 | superantigen-like |
| ssl01 | superantigen-like |
| ssl02 | superantigen-like |
| ssl10 | superantigen-like |
| sspA | protease |
| sspB | superantigen-like |
| sspP | superantigen-like |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




