Submitted:
16 June 2025
Posted:
17 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Mechanisms of Neuroinflammation in Post-COVID-19 Syndrome
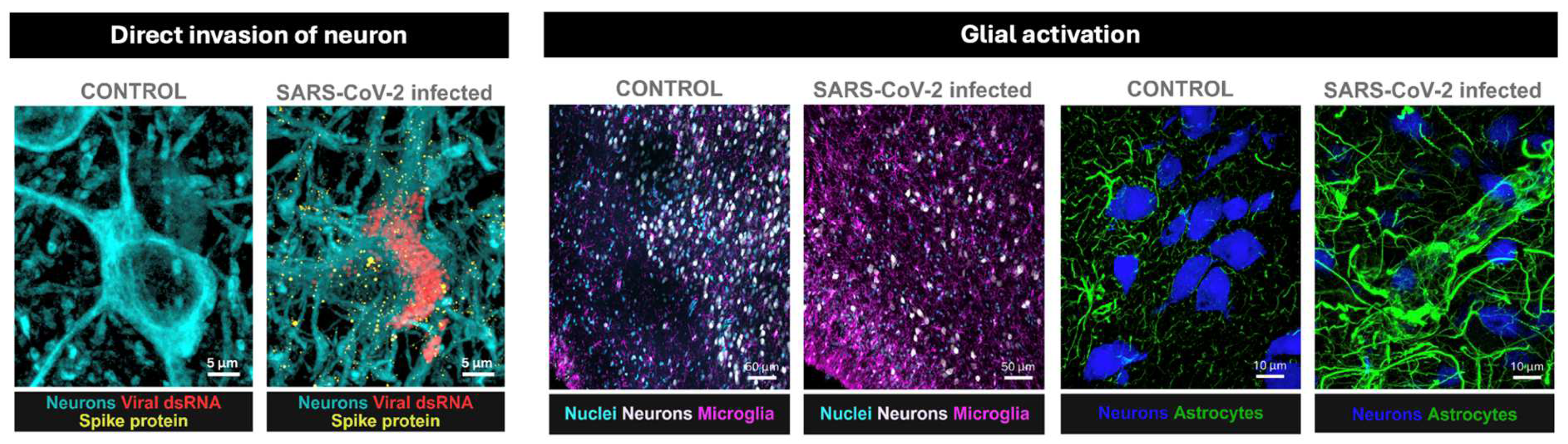
2.1. Direct Viral Invasion of Brain Parenchyma
2.2. Glial Reactivity
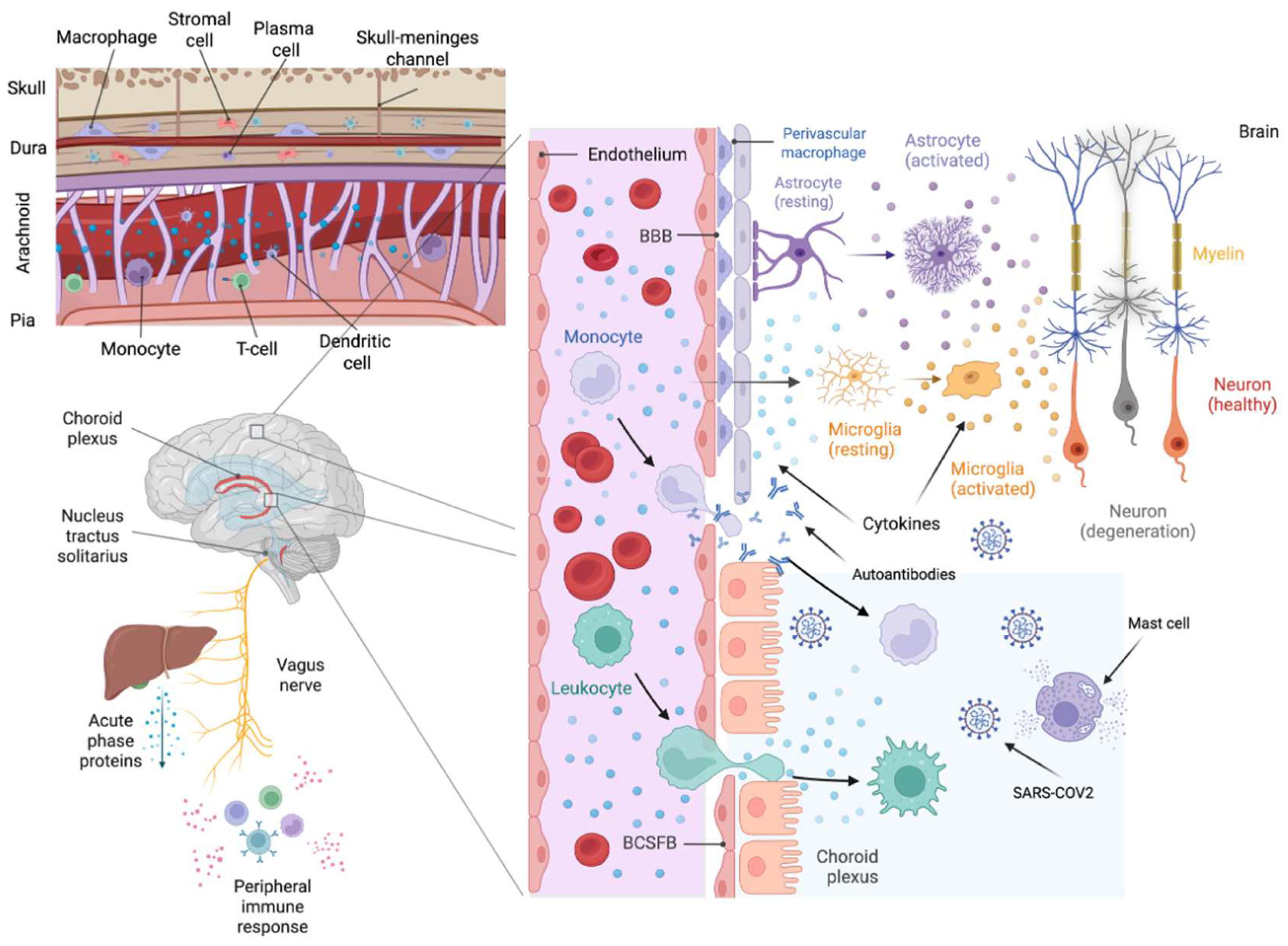
2.3. Blood Brain Barrier and Blood-CSF Barrier
2.4. Meningeal Immunity and Skull Bone Marrow
2.5. Autoimmunity
2.6. Reactivation of Endogenous Retroviruses and Latent Viral Elements
2.7. Neurovascular Unit and Oxygen Metabolism
2.8. Vagus Nerve Pathway
2.9. Mast Cell Activation and the Histamine System
3. Biomarker Evidence
3.1. Neuroimaging Biomarkers
3.2. Soluble Biomarkers
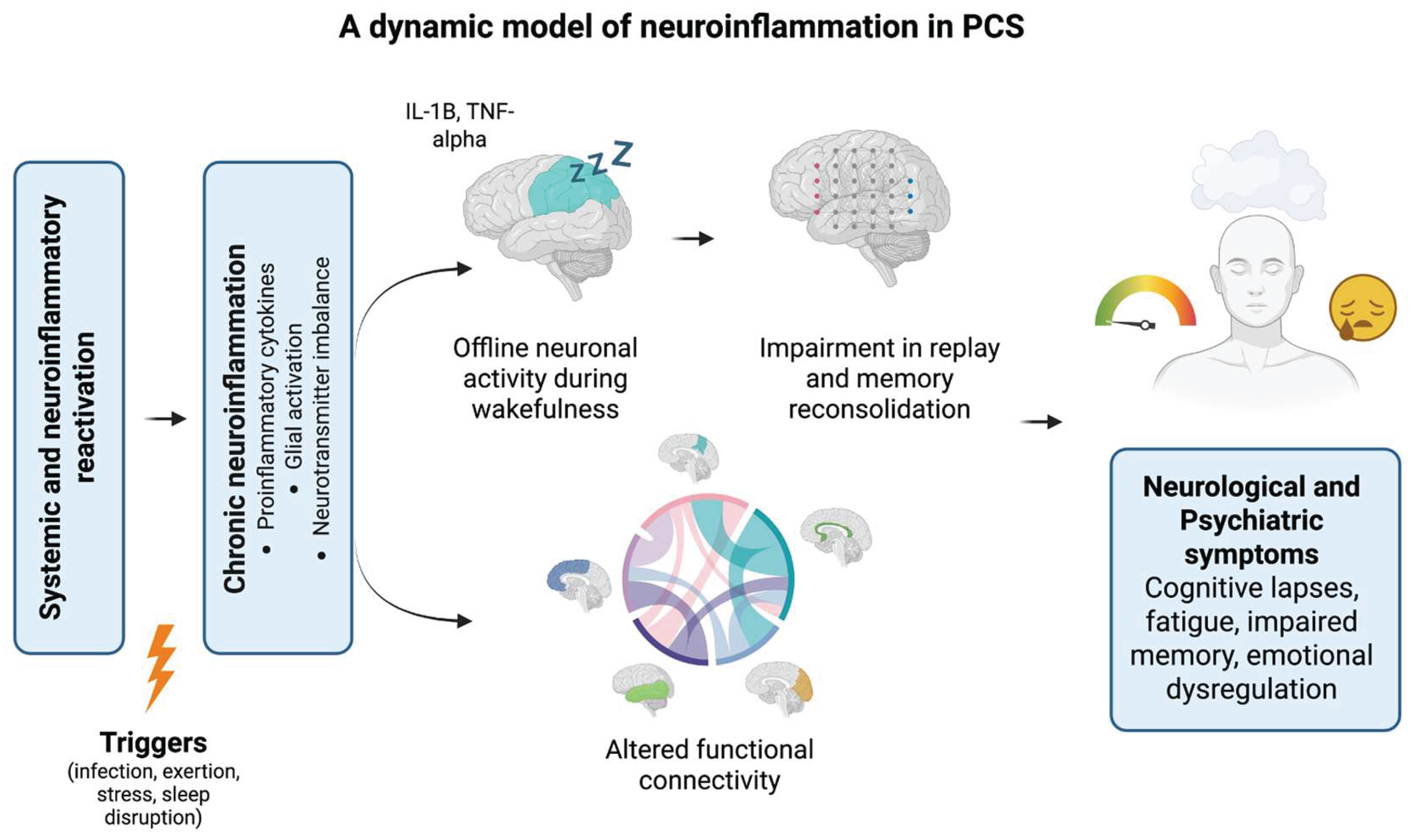
4. Neuroinflammation, Brain Dysfunction, and the Non-linear Relapse-Remitting Time-Course of Symptoms: A Speculative Biologically Grounded Mechanistic Model
5. Therapeutic Implications and Opportunities for Drug Development/Repurposing
6. Outstanding Questions and Directions for Further Research
|
Outstanding Questions on Neuroinflammation in PCS |
Suggested Research Approaches |
|---|---|
| 1. What specific neuroimmune pathways are activated during persistent neuroinflammation in post-COVID-19 syndrome, and do they induce local sleep in cortical microcircuits? | Multi-omics analyses; animal models; EEG-fMRI studies of local sleep under inflammation |
| 2. How do neuroinflammatory profiles differ based on initial COVID-19 severity? | Stratified longitudinal studies using biomarkers and neuroimaging |
| 3. What is the progression and resolution timeline of neuroinflammatory markers post-infection, and how does this relate to symptom fluctuation and flare cycles? | Longitudinal observational cohorts with time-resolved biomarker sampling and digital phenotyping |
| 4. Which neuroinflammatory biomarkers predict persistent neurological and cognitive symptoms, including those linked to reconsolidation failure or fatigue? | Prospective cohort studies and biomarker validation trials |
| 5. Are genetic or epigenetic markers associated with susceptibility to neuroinflammation, local sleep phenomena, or reconsolidation impairments in PCS? | Genome-wide association studies (GWAS); epigenetic analyses linked to cognitive profiles |
| 6. Can neuroimaging reliably differentiate neuroinflammatory patterns and local sleep signatures linked to specific symptom clusters? | Functional and structural neuroimaging studies (MRI, PET, mobile EEG) |
| 7. Which immunomodulatory therapies most effectively reduce neuroinflammatory markers, support metabolic resilience, and stabilize network function? | Randomized controlled clinical trials; mechanistically informed pharmacological studies |
| 8. Can targeting glial activation and adenosine signaling reduce sustained neuroinflammation and associated cognitive or emotional symptoms? | Preclinical experiments; clinical trials of astrocyte/microglia and purinergic-targeted therapies |
| 9. Which rehabilitation strategies most effectively support circuit reintegration, reconsolidation processes, and resilience to flare cycles? | Controlled trials of cognitive-behavioral and neuroplasticity-based rehabilitation approaches |
| 10. How can integrated multidisciplinary care models dynamically respond to fluctuating brain states and optimize long-term recovery in PCS? | Clinical pathway optimization involving neurology, psychiatry, immunology, and rehabilitation |
7. Conclusions
Conflict of Interests
Author Contributions
Acknowledgments
References
- Li, J.; Zhou, Y.; Ma, J.; Zhang, Q.; Shao, J.; Liang, S.; Yu, Y.; Li, W.; Wang, C. The long-term health outcomes, pathophysiological mechanisms and multidisciplinary management of long COVID. Signal Transduct Target Ther 2023, 8, 416. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Long COVID: major findings, mechanisms and recommendations. Nat Rev Microbiol 2023, 21, 133–146. [Google Scholar] [CrossRef]
- Su, S.; Zhao, Y.; Zeng, N.; Liu, X.; Zheng, Y.; Sun, J.; Zhong, Y.; Wu, S.; Ni, S.; Gong, Y. , et al. Epidemiology, clinical presentation, pathophysiology, and management of long COVID: an update. Mol Psychiatry 2023, 28, 4056–4069. [Google Scholar] [CrossRef]
- Shah, W.; Hillman, T.; Playford, E.D.; Hishmeh, L. Managing the long term effects of covid-19: summary of NICE, SIGN, and RCGP rapid guideline. BMJ 2021, 372, n136. [Google Scholar] [CrossRef] [PubMed]
- Soriano, J.B.; Murthy, S.; Marshall, J.C.; Relan, P.; Diaz, J.V.; Condition, W.H.O.C.C.D.W.G.o.P.-C.-. . A clinical case definition of post-COVID-19 condition by a Delphi consensus. Lancet Infect Dis 2022, 22, e102–e107. [Google Scholar] [CrossRef]
- VanElzakker, M.B.; Bues, H.F.; Brusaferri, L.; Kim, M.; Saadi, D.; Ratai, E.M.; Dougherty, D.D.; Loggia, M.L. Neuroinflammation in post-acute sequelae of COVID-19 (PASC) as assessed by [(11)C]PBR28 PET correlates with vascular disease measures. Brain Behav Immun 2024, 119, 713–723. [Google Scholar] [CrossRef]
- Dos Reis, R.S.; Selvam, S.; Ayyavoo, V. Neuroinflammation in Post COVID-19 Sequelae: Neuroinvasion and Neuroimmune Crosstalk. Rev Med Virol 2024, 34, e70009. [Google Scholar] [CrossRef]
- Bergmann, C.C.; Lane, T.E.; Stohlman, S.A. Coronavirus infection of the central nervous system: host-virus stand-off. Nat Rev Microbiol 2006, 4, 121–132. [Google Scholar] [CrossRef] [PubMed]
- Puelles, V.G.; Lutgehetmann, M.; Lindenmeyer, M.T.; Sperhake, J.P.; Wong, M.N.; Allweiss, L.; Chilla, S.; Heinemann, A.; Wanner, N.; Liu, S. , et al. Multiorgan and Renal Tropism of SARS-CoV-2. N Engl J Med 2020, 383, 590–592. [Google Scholar] [CrossRef]
- Meinhardt, J.; Radke, J.; Dittmayer, C.; Franz, J.; Thomas, C.; Mothes, R.; Laue, M.; Schneider, J.; Brunink, S.; Greuel, S. , et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci 2021, 24, 168–175. [Google Scholar] [CrossRef]
- Dube, M.; Le Coupanec, A.; Wong, A.H.M.; Rini, J.M.; Desforges, M.; Talbot, P.J. Axonal Transport Enables Neuron-to-Neuron Propagation of Human Coronavirus OC43. J Virol 2018, 92. [Google Scholar] [CrossRef]
- Awogbindin, I.O.; Ben-Azu, B.; Olusola, B.A.; Akinluyi, E.T.; Adeniyi, P.A.; Di Paolo, T.; Tremblay, M.E. Microglial Implications in SARS-CoV-2 Infection and COVID-19: Lessons From Viral RNA Neurotropism and Possible Relevance to Parkinson's Disease. Front Cell Neurosci 2021, 15, 670298. [Google Scholar] [CrossRef]
- Guerrero, J.I.; Barragan, L.A.; Martinez, J.D.; Montoya, J.P.; Pena, A.; Sobrino, F.E.; Tovar-Spinoza, Z.; Ghotme, K.A. Central and peripheral nervous system involvement by COVID-19: a systematic review of the pathophysiology, clinical manifestations, neuropathology, neuroimaging, electrophysiology, and cerebrospinal fluid findings. BMC Infect Dis 2021, 21, 515. [Google Scholar] [CrossRef] [PubMed]
- Meinhardt, J.; Streit, S.; Dittmayer, C.; Manitius, R.V.; Radbruch, H.; Heppner, F.L. The neurobiology of SARS-CoV-2 infection. Nat Rev Neurosci 2024, 25, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Rong, Z.; Mai, H.; Ebert, G.; Kapoor, S.; Puelles, V.G.; Czogalla, J.; Hu, S.; Su, J.; Prtvar, D.; Singh, I. , et al. Persistence of spike protein at the skull-meninges-brain axis may contribute to the neurological sequelae of COVID-19. Cell Host Microbe 2024, 32, 2112–2130. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhang, X.; Chai, Y.; Zhang, J.; Deng, Q.; Chen, X. Skull bone marrow and skull meninges channels: redefining the landscape of central nervous system immune surveillance. Cell Death Dis 2025, 16, 53. [Google Scholar] [CrossRef]
- Fodoulian, L.; Tuberosa, J.; Rossier, D.; Boillat, M.; Kan, C.; Pauli, V.; Egervari, K.; Lobrinus, J.A.; Landis, B.N.; Carleton, A. , et al. SARS-CoV-2 Receptors and Entry Genes Are Expressed in the Human Olfactory Neuroepithelium and Brain. iScience 2020, 23, 101839. [Google Scholar] [CrossRef]
- Zubair, A.S.; McAlpine, L.S.; Gardin, T.; Farhadian, S.; Kuruvilla, D.E.; Spudich, S. Neuropathogenesis and Neurologic Manifestations of the Coronaviruses in the Age of Coronavirus Disease 2019: A Review. JAMA Neurol 2020, 77, 1018–1027. [Google Scholar] [CrossRef]
- Beckman, D.; Bonillas, A.; Diniz, G.B.; Ott, S.; Roh, J.W.; Elizaldi, S.R.; Schmidt, B.A.; Sammak, R.L.; Van Rompay, K.K.A.; Iyer, S.S. , et al. SARS-CoV-2 infects neurons and induces neuroinflammation in a non-human primate model of COVID-19. Cell Rep 2022, 41, 111573. [Google Scholar] [CrossRef]
- Samudyata; Oliveira, A. O.; Malwade, S.; Rufino de Sousa, N.; Goparaju, S.K.; Gracias, J.; Orhan, F.; Steponaviciute, L.; Schalling, M.; Sheridan, S.D., et al. SARS-CoV-2 promotes microglial synapse elimination in human brain organoids. Mol Psychiatry 2022, 27, 3939–3950. [Google Scholar] [CrossRef]
- Jeong, G.U.; Lyu, J.; Kim, K.D.; Chung, Y.C.; Yoon, G.Y.; Lee, S.; Hwang, I.; Shin, W.H.; Ko, J.; Lee, J.Y. , et al. SARS-CoV-2 Infection of Microglia Elicits Proinflammatory Activation and Apoptotic Cell Death. Microbiol Spectr 2022, 10, e0109122. [Google Scholar] [CrossRef] [PubMed]
- Wijesinghe, S.S.; Rowe, J.B.; Mason, H.D.; Allinson, K.S.J.; Thomas, R.; Vontobel, D.S.; Fryer, T.D.; Hong, Y.T.; Bacioglu, M.; Spillantini, M.G. , et al. Post-mortem validation of in vivo TSPO PET as a microglial biomarker. Brain, 1093. [Google Scholar]
- Tavcar, P.; Potokar, M.; Kolenc, M.; Korva, M.; Avsic-Zupanc, T.; Zorec, R.; Jorgacevski, J. Neurotropic Viruses, Astrocytes, and COVID-19. Front Cell Neurosci 2021, 15, 662578. [Google Scholar] [CrossRef] [PubMed]
- Crunfli, F.; Carregari, V.C.; Veras, F.P.; Silva, L.S.; Nogueira, M.H.; Antunes, A.; Vendramini, P.H.; Valenca, A.G.F.; Brandao-Teles, C.; Zuccoli, G.D.S. , et al. Morphological, cellular, and molecular basis of brain infection in COVID-19 patients. Proc Natl Acad Sci U S A 2022, 119, e2200960119. [Google Scholar] [CrossRef]
- Andrews, M.G.; Mukhtar, T.; Eze, U.C.; Simoneau, C.R.; Ross, J.; Parikshak, N.; Wang, S.; Zhou, L.; Koontz, M.; Velmeshev, D. , et al. Tropism of SARS-CoV-2 for human cortical astrocytes. Proc Natl Acad Sci U S A 2022, 119, e2122236119. [Google Scholar] [CrossRef]
- Kong, W.; Montano, M.; Corley, M.J.; Helmy, E.; Kobayashi, H.; Kinisu, M.; Suryawanshi, R.; Luo, X.; Royer, L.A.; Roan, N.R. , et al. Neuropilin-1 Mediates SARS-CoV-2 Infection of Astrocytes in Brain Organoids, Inducing Inflammation Leading to Dysfunction and Death of Neurons. mBio 2022, 13, e0230822. [Google Scholar] [CrossRef] [PubMed]
- Schwabenland, M.; Salie, H.; Tanevski, J.; Killmer, S.; Lago, M.S.; Schlaak, A.E.; Mayer, L.; Matschke, J.; Puschel, K.; Fitzek, A. , et al. Deep spatial profiling of human COVID-19 brains reveals neuroinflammation with distinct microanatomical microglia-T-cell interactions. Immunity 2021, 54, 1594–1610. [Google Scholar] [CrossRef]
- Galea, I. The blood-brain barrier in systemic infection and inflammation. Cell Mol Immunol 2021, 18, 2489–2501. [Google Scholar] [CrossRef]
- Greene, C.; Connolly, R.; Brennan, D.; Laffan, A.; O'Keeffe, E.; Zaporojan, L.; O'Callaghan, J.; Thomson, B.; Connolly, E.; Argue, R. , et al. Blood-brain barrier disruption and sustained systemic inflammation in individuals with long COVID-associated cognitive impairment. Nat Neurosci 2024, 27, 421–432. [Google Scholar] [CrossRef]
- Zhang, L.; Zhou, L.; Bao, L.; Liu, J.; Zhu, H.; Lv, Q.; Liu, R.; Chen, W.; Tong, W.; Wei, Q. , et al. SARS-CoV-2 crosses the blood-brain barrier accompanied with basement membrane disruption without tight junctions alteration. Signal Transduct Target Ther 2021, 6, 337. [Google Scholar] [CrossRef]
- Wenzel, J.; Lampe, J.; Muller-Fielitz, H.; Schuster, R.; Zille, M.; Muller, K.; Krohn, M.; Korbelin, J.; Zhang, L.; Ozorhan, U. , et al. The SARS-CoV-2 main protease M(pro) causes microvascular brain pathology by cleaving NEMO in brain endothelial cells. Nat Neurosci 2021, 24, 1522–1533. [Google Scholar] [CrossRef]
- Lee, M.H.; Perl, D.P.; Steiner, J.; Pasternack, N.; Li, W.; Maric, D.; Safavi, F.; Horkayne-Szakaly, I.; Jones, R.; Stram, M.N. , et al. Neurovascular injury with complement activation and inflammation in COVID-19. Brain 2022, 145, 2555–2568. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Hehnly, C.; Lehtinen, M.K. The choroid plexus: a command center for brain-body communication during inflammation. Curr Opin Immunol 2025, 93, 102540. [Google Scholar] [CrossRef]
- Rau, A.; Gonzalez-Escamilla, G.; Schroeter, N.; Othman, A.; Dressing, A.; Weiller, C.; Urbach, H.; Reisert, M.; Groppa, S.; Hosp, J.A. Inflammation-Triggered Enlargement of Choroid Plexus in Subacute COVID-19 Patients with Neurological Symptoms. Ann Neurol 2024, 96, 715–725. [Google Scholar] [CrossRef]
- Diez-Cirarda, M.; Yus-Fuertes, M.; Delgado-Alonso, C.; Gil-Martinez, L.; Jimenez-Garcia, C.; Gil-Moreno, M.J.; Gomez-Ruiz, N.; Oliver-Mas, S.; Polidura, C.; Jorquera, M. , et al. Choroid plexus volume is enlarged in long COVID and associated with cognitive and brain changes. Mol Psychiatry, 1038. [Google Scholar]
- Mazzitelli, J.A.; Pulous, F.E.; Smyth, L.C.D.; Kaya, Z.; Rustenhoven, J.; Moskowitz, M.A.; Kipnis, J.; Nahrendorf, M. Skull bone marrow channels as immune gateways to the central nervous system. Nat Neurosci 2023, 26, 2052–2062. [Google Scholar] [CrossRef] [PubMed]
- Nunez-Castilla, J.; Stebliankin, V.; Baral, P.; Balbin, C.A.; Sobhan, M.; Cickovski, T.; Mondal, A.M.; Narasimhan, G.; Chapagain, P.; Mathee, K. , et al. Potential Autoimmunity Resulting from Molecular Mimicry between SARS-CoV-2 Spike and Human Proteins. Viruses 2022, 14. [Google Scholar] [CrossRef]
- Santos Guedes de Sa, K.; Silva, J.; Bayarri-Olmos, R.; Brinda, R.; Alec Rath Constable, R.; Colom Diaz, P.A.; Kwon, D.I.; Rodrigues, G.; Wenxue, L.; Baker, C. , et al. A causal link between autoantibodies and neurological symptoms in long COVID. medRxiv, 1101. [Google Scholar]
- Hansen, N. Psychiatric Symptoms in Acute and Persisting Forms of COVID-19 Associated with Neural Autoantibodies. Antibodies (Basel) 2023, 12. [Google Scholar] [CrossRef]
- Seibert, F.S.; Stervbo, U.; Wiemers, L.; Skrzypczyk, S.; Hogeweg, M.; Bertram, S.; Kurek, J.; Anft, M.; Westhoff, T.H.; Babel, N. Severity of neurological Long-COVID symptoms correlates with increased level of autoantibodies targeting vasoregulatory and autonomic nervous system receptors. Autoimmun Rev 2023, 22, 103445. [Google Scholar] [CrossRef] [PubMed]
- Kitsou, K.; Kotanidou, A.; Paraskevis, D.; Karamitros, T.; Katzourakis, A.; Tedder, R.; Hurst, T.; Sapounas, S.; Kotsinas, A.; Gorgoulis, V. , et al. Upregulation of Human Endogenous Retroviruses in Bronchoalveolar Lavage Fluid of COVID-19 Patients. Microbiol Spectr 2021, 9, e0126021. [Google Scholar] [CrossRef]
- Gimenez-Orenga, K.; Pierquin, J.; Brunel, J.; Charvet, B.; Martin-Martinez, E.; Perron, H.; Oltra, E. HERV-W ENV antigenemia and correlation of increased anti-SARS-CoV-2 immunoglobulin levels with post-COVID-19 symptoms. Front Immunol 2022, 13, 1020064. [Google Scholar] [CrossRef]
- Douville, R.N.; Nath, A. Human endogenous retroviruses and the nervous system. Handb Clin Neurol 2014, 123, 465–485. [Google Scholar]
- Brunel, J.; Paganini, J.; Galloux, M.; Charvet, B.; Perron, H. HERV-W ENV transcription in B cells predicting symptomatic COVID-19 and risk for long COVID can express a full-length protein despite stop codon in mRNA from chromosome X via a ribosome readthrough. Microbes Infect, 1016. [Google Scholar]
- Gruchot, J.; Reiche, L.; Werner, L.; Herrero, F.; Schira-Heinen, J.; Meyer, U.; Kury, P. Molecular dissection of HERV-W dependent microglial- and astroglial cell polarization. Microbes Infect, 1016. [Google Scholar]
- Gruchot, J.; Lewen, I.; Dietrich, M.; Reiche, L.; Sindi, M.; Hecker, C.; Herrero, F.; Charvet, B.; Weber-Stadlbauer, U.; Hartung, H.P. , et al. Transgenic expression of the HERV-W envelope protein leads to polarized glial cell populations and a neurodegenerative environment. Proc Natl Acad Sci U S A 2023, 120, e2308187120. [Google Scholar] [CrossRef] [PubMed]
- Charvet, B.; Brunel, J.; Pierquin, J.; Iampietro, M.; Decimo, D.; Queruel, N.; Lucas, A.; Encabo-Berzosa, M.D.M.; Arenaz, I.; Marmolejo, T.P. , et al. SARS-CoV-2 awakens ancient retroviral genes and the expression of proinflammatory HERV-W envelope protein in COVID-19 patients. iScience 2023, 26, 106604. [Google Scholar] [CrossRef] [PubMed]
- Josephs, S.F.; Henry, B.; Balachandran, N.; Strayer, D.; Peterson, D.; Komaroff, A.L.; Ablashi, D.V. HHV-6 reactivation in chronic fatigue syndrome. Lancet 1991, 337, 1346–1347. [Google Scholar] [CrossRef]
- Rousseau, B.A.; Bhaduri-McIntosh, S. Inflammation and Epstein-Barr Virus at the Crossroads of Multiple Sclerosis and Post-Acute Sequelae of COVID-19 Infection. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Schaeffer, S.; Iadecola, C. Revisiting the neurovascular unit. Nat Neurosci 2021, 24, 1198–1209. [Google Scholar] [CrossRef]
- Pretorius, E.; Venter, C.; Laubscher, G.J.; Kotze, M.J.; Oladejo, S.O.; Watson, L.R.; Rajaratnam, K.; Watson, B.W.; Kell, D.B. Prevalence of symptoms, comorbidities, fibrin amyloid microclots and platelet pathology in individuals with Long COVID/Post-Acute Sequelae of COVID-19 (PASC). Cardiovasc Diabetol 2022, 21, 148. [Google Scholar] [CrossRef]
- Fekete, M.; Lehoczki, A.; Szappanos, A.; Toth, A.; Mahdi, M.; Sotonyi, P.; Benyo, Z.; Yabluchanskiy, A.; Tarantini, S.; Ungvari, Z. Cerebromicrovascular mechanisms contributing to long COVID: implications for neurocognitive health. Geroscience 2025, 47, 745–779. [Google Scholar] [CrossRef] [PubMed]
- Libby, P. Endothelial inflammation in COVID-19. Science 2024, 386, 972–973. [Google Scholar] [CrossRef]
- Ikemizu, Y.; Oda, Y.; Hirose, Y.; Sasaki, T.; Iyo, M. Cerebellar and Occipital Alterations in Brain Perfusion in a Patient With Post-acute COVID-19 Encephalopathy Misdiagnosed As Primary Psychotic Disorder. Cureus 2024, 16, e52953. [Google Scholar] [CrossRef]
- Chien, C.; Heine, J.; Khalil, A.; Schlenker, L.; Hartung, T.J.; Boesl, F.; Schwichtenberg, K.; Rust, R.; Bellmann-Strobl, J.; Franke, C. , et al. Altered brain perfusion and oxygen levels relate to sleepiness and attention in post-COVID syndrome. Ann Clin Transl Neurol 2024, 11, 2016–2029. [Google Scholar] [CrossRef]
- Adingupu, D.D.; Soroush, A.; Hansen, A.; Twomey, R.; Dunn, J.F. Brain hypoxia, neurocognitive impairment, and quality of life in people post-COVID-19. J Neurol 2023, 270, 3303–3314. [Google Scholar] [CrossRef] [PubMed]
- Szogi, T.; Borsos, B.N.; Masic, D.; Radics, B.; Bella, Z.; Banfi, A.; Ordog, N.; Zsiros, C.; Kiricsi, A.; Pankotai-Bodo, G. , et al. Novel biomarkers of mitochondrial dysfunction in Long COVID patients. Geroscience 2025, 47, 2245–2261. [Google Scholar] [CrossRef] [PubMed]
- Yamada, S.; Hashita, T.; Yanagida, S.; Sato, H.; Yasuhiko, Y.; Okabe, K.; Noda, T.; Nishida, M.; Matsunaga, T.; Kanda, Y. SARS-CoV-2 causes dysfunction in human iPSC-derived brain microvascular endothelial cells potentially by modulating the Wnt signaling pathway. Fluids Barriers CNS 2024, 21, 32. [Google Scholar] [CrossRef] [PubMed]
- Pavlov, V.A.; Wang, H.; Czura, C.J.; Friedman, S.G.; Tracey, K.J. The cholinergic anti-inflammatory pathway: a missing link in neuroimmunomodulation. Mol Med 2003, 9, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Gallowitsch-Puerta, M.; Pavlov, V.A. Neuro-immune interactions via the cholinergic anti-inflammatory pathway. Life Sci 2007, 80, 2325–2329. [Google Scholar] [CrossRef]
- Bonaz, B.; Sinniger, V.; Pellissier, S. Targeting the cholinergic anti-inflammatory pathway with vagus nerve stimulation in patients with Covid-19? Bioelectron Med 2020, 6, 15. [Google Scholar] [CrossRef]
- Llados, G.; Massanella, M.; Coll-Fernandez, R.; Rodriguez, R.; Hernandez, E.; Lucente, G.; Lopez, C.; Loste, C.; Santos, J.R.; Espana-Cueto, S. , et al. Vagus nerve dysfunction in the post-COVID-19 condition: a pilot cross-sectional study. Clin Microbiol Infect 2024, 30, 515–521. [Google Scholar] [CrossRef]
- Komegae, E.N.; Farmer, D.G.S.; Brooks, V.L.; McKinley, M.J.; McAllen, R.M.; Martelli, D. Vagal afferent activation suppresses systemic inflammation via the splanchnic anti-inflammatory pathway. Brain Behav Immun 2018, 73, 441–449. [Google Scholar] [CrossRef]
- Pavlov, V.A.; Tracey, K.J. The vagus nerve and the inflammatory reflex--linking immunity and metabolism. Nat Rev Endocrinol 2012, 8, 743–754. [Google Scholar] [CrossRef]
- Wu, M.L.; Xie, C.; Li, X.; Sun, J.; Zhao, J.; Wang, J.H. Mast cell activation triggered by SARS-CoV-2 causes inflammation in brain microvascular endothelial cells and microglia. Front Cell Infect Microbiol 2024, 14, 1358873. [Google Scholar] [CrossRef]
- Sumantri, S.; Rengganis, I. Immunological dysfunction and mast cell activation syndrome in long COVID. Asia Pac Allergy 2023, 13, 50–53. [Google Scholar] [CrossRef] [PubMed]
- Weinstock, L.B.; Brook, J.B.; Walters, A.S.; Goris, A.; Afrin, L.B.; Molderings, G.J. Mast cell activation symptoms are prevalent in Long-COVID. Int J Infect Dis 2021, 112, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Z.; An, Q.; Zhang, W.; Li, Y.; Zhang, Q.; Yan, H. Histamine and receptors in neuroinflammation: Their roles on neurodegenerative diseases. Behav Brain Res 2024, 465, 114964. [Google Scholar] [CrossRef] [PubMed]
- Raghavan, S.; Leo, M.D. Histamine Potentiates SARS-CoV-2 Spike Protein Entry Into Endothelial Cells. Front Pharmacol 2022, 13, 872736. [Google Scholar] [CrossRef]
- de Esch, I.J.; Thurmond, R.L.; Jongejan, A.; Leurs, R. The histamine H4 receptor as a new therapeutic target for inflammation. Trends Pharmacol Sci 2005, 26, 462–469. [Google Scholar]
- Yu, F.; Liu, X.; Ou, H.; Li, X.; Liu, R.; Lv, X.; Xiao, S.; Hu, M.; Liang, T.; Chen, T. , et al. The histamine receptor H1 acts as an alternative receptor for SARS-CoV-2. mBio 2024, 15, e0108824. [Google Scholar] [CrossRef]
- Haas, H.L.; Sergeeva, O.A.; Selbach, O. Histamine in the nervous system. Physiol Rev 2008, 88, 1183–1241. [Google Scholar] [CrossRef]
- Yamada, Y.; Yoshikawa, T.; Naganuma, F.; Kikkawa, T.; Osumi, N.; Yanai, K. Chronic brain histamine depletion in adult mice induced depression-like behaviours and impaired sleep-wake cycle. Neuropharmacology 2020, 175, 108179. [Google Scholar] [CrossRef]
- Flik, G.; Folgering, J.H.; Cremers, T.I.; Westerink, B.H.; Dremencov, E. Interaction Between Brain Histamine and Serotonin, Norepinephrine, and Dopamine Systems: In Vivo Microdialysis and Electrophysiology Study. J Mol Neurosci 2015, 56, 320–328. [Google Scholar] [CrossRef]
- Salvucci, F.; Codella, R.; Coppola, A.; Zacchei, I.; Grassi, G.; Anti, M.L.; Nitisoara, N.; Luzi, L.; Gazzaruso, C. Antihistamines improve cardiovascular manifestations and other symptoms of long-COVID attributed to mast cell activation. Front Cardiovasc Med 2023, 10, 1202696. [Google Scholar] [CrossRef]
- Braga, J.; Lepra, M.; Kish, S.J.; Rusjan, P.M.; Nasser, Z.; Verhoeff, N.; Vasdev, N.; Bagby, M.; Boileau, I.; Husain, M.I. , et al. Neuroinflammation After COVID-19 With Persistent Depressive and Cognitive Symptoms. JAMA Psychiatry 2023, 80, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Clouston, S.A.P.; Vaska, P.; Babalola, T.; Gardus, J., 3rd; Huang, C.; Soriolo, N.; Fontana, A.; DeLorenzo, C.; Parsey, R.; Luft, B.J. Glial activation among individuals with neurological post-acute sequelae of coronavirus disease 2019: A positron emission tomography study of brain fog using [(18)F]-FEPPA. Brain Behav Immun Health 2025, 44, 100945. [Google Scholar] [CrossRef]
- Nutma, E.; Fancy, N.; Weinert, M.; Tsartsalis, S.; Marzin, M.C.; Muirhead, R.C.J.; Falk, I.; Breur, M.; de Bruin, J.; Hollaus, D. , et al. Translocator protein is a marker of activated microglia in rodent models but not human neurodegenerative diseases. Nat Commun 2023, 14, 5247. [Google Scholar] [CrossRef] [PubMed]
- Braga, J.; Kuik, E.J.Y.; Lepra, M.; Rusjan, P.M.; Kish, S.J.; Vieira, E.L.; Nasser, Z.; Verhoeff, N.; Vasdev, N.; Chao, T. , et al. Astrogliosis Marker [(11)C]SL25.1188 After COVID-19 With Ongoing Depressive and Cognitive Symptoms. Biol Psychiatry 2025, 97, 816–824. [Google Scholar] [CrossRef]
- Chaganti, J.R.; Talekar, T.K.; Brew, B.J. Asymmetrical glymphatic dysfunction in patients with long Covid associated neurocognitive impairment- correlation with BBB disruption. BMC Neurol 2025, 25, 112. [Google Scholar] [CrossRef]
- Ernst, T.; Ryan, M.C.; Liang, H.J.; Wang, J.P.; Cunningham, E.; Saleh, M.G.; Kottilil, S.; Chang, L. Neuronal and Glial Metabolite Abnormalities in Participants With Persistent Neuropsychiatric Symptoms After COVID-19: A Brain Proton Magnetic Resonance Spectroscopy Study. J Infect Dis 2023, 228, 1559–1570. [Google Scholar] [CrossRef] [PubMed]
- Pajuelo, D.; Dezortova, M.; Hajek, M.; Ibrahimova, M.; Ibrahim, I. Metabolic changes assessed by 1H MR spectroscopy in the corpus callosum of post-COVID patients. MAGMA 2024, 37, 937–946. [Google Scholar] [CrossRef]
- Chaganti, J.; Poudel, G.; Cysique, L.A.; Dore, G.J.; Kelleher, A.; Matthews, G.; Darley, D.; Byrne, A.; Jakabek, D.; Zhang, X. , et al. Blood brain barrier disruption and glutamatergic excitotoxicity in post-acute sequelae of SARS COV-2 infection cognitive impairment: potential biomarkers and a window into pathogenesis. Front Neurol 2024, 15, 1350848. [Google Scholar] [CrossRef]
- Sharma, A.A.; Nenert, R.; Goodman, A.M.; Szaflarski, J.P. Brain temperature and free water increases after mild COVID-19 infection. Sci Rep 2024, 14, 7450. [Google Scholar] [CrossRef]
- Douaud, G.; Lee, S.; Alfaro-Almagro, F.; Arthofer, C.; Wang, C.; McCarthy, P.; Lange, F.; Andersson, J.L.R.; Griffanti, L.; Duff, E. , et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature 2022, 604, 697–707. [Google Scholar] [CrossRef]
- Hosp, J.A.; Reisert, M.; Dressing, A.; Gotz, V.; Kellner, E.; Mast, H.; Arndt, S.; Waller, C.F.; Wagner, D.; Rieg, S. , et al. Cerebral microstructural alterations in Post-COVID-condition are related to cognitive impairment, olfactory dysfunction and fatigue. Nat Commun 2024, 15, 4256. [Google Scholar] [CrossRef] [PubMed]
- Kenji Sudo, F.; Pinto, T.P.; F, G.Q.B.-A.; Bramati, I.; Marins, T.F.; Monteiro, M.; Meireles, F.; Soares, R.; Erthal, P.; Calil, V. , et al. Cognitive, behavioral, neuroimaging and inflammatory biomarkers after hospitalization for covid-19 in Brazil. Brain Behav Immun, 1016. [Google Scholar]
- Serrano Del Pueblo, V.M.; Serrano-Heras, G.; Romero Sanchez, C.M.; Landete, P.P.; Rojas-Bartolome, L.; Feria, I.; Morris, R.G.M.; Strange, B.; Mansilla, F.; Zhang, L. , et al. Brain and cognitive changes in patients with long COVID compared with infection-recovered control subjects. Brain 2024, 147, 3611–3623. [Google Scholar] [CrossRef]
- Besteher, B.; Rocktaschel, T.; Garza, A.P.; Machnik, M.; Ballez, J.; Helbing, D.L.; Finke, K.; Reuken, P.; Gullmar, D.; Gaser, C. , et al. Cortical thickness alterations and systemic inflammation define long-COVID patients with cognitive impairment. Brain Behav Immun 2024, 116, 175–184. [Google Scholar] [CrossRef] [PubMed]
- Peluso, M.J.; Deitchman, A.N.; Torres, L.; Iyer, N.S.; Munter, S.E.; Nixon, C.C.; Donatelli, J.; Thanh, C.; Takahashi, S.; Hakim, J. , et al. Long-term SARS-CoV-2-specific immune and inflammatory responses in individuals recovering from COVID-19 with and without post-acute symptoms. Cell Rep 2021, 36, 109518. [Google Scholar] [CrossRef]
- Phetsouphanh, C.; Darley, D.R.; Wilson, D.B.; Howe, A.; Munier, C.M.L.; Patel, S.K.; Juno, J.A.; Burrell, L.M.; Kent, S.J.; Dore, G.J. , et al. Immunological dysfunction persists for 8 months following initial mild-to-moderate SARS-CoV-2 infection. Nat Immunol 2022, 23, 210–216. [Google Scholar] [CrossRef] [PubMed]
- Espin, E.; Yang, C.; Shannon, C.P.; Assadian, S.; He, D.; Tebbutt, S.J. Cellular and molecular biomarkers of long COVID: a scoping review. EBioMedicine 2023, 91, 104552. [Google Scholar] [CrossRef]
- Almulla, A.F.; Thipakorn, Y.; Zhou, B.; Vojdani, A.; Maes, M. Immune activation and immune-associated neurotoxicity in Long-COVID: A systematic review and meta-analysis of 103 studies comprising 58 cytokines/chemokines/growth factors. Brain Behav Immun 2024, 122, 75–94. [Google Scholar] [CrossRef] [PubMed]
- Vrettou, C.S.; Vassiliou, A.G.; Keskinidou, C.; Mourelatos, P.; Asimakos, A.; Spetsioti, S.; Diamantopoulos, A.; Jahaj, E.; Antonoglou, A.; Katsaounou, P. , et al. A Prospective Study on Neural Biomarkers in Patients with Long-COVID Symptoms. J Pers Med 2024, 14. [Google Scholar] [CrossRef]
- Pacheco-Jaime, L.; Garcia-Vicente, C.; Ariza, M.; Cano, N.; Garolera, M.; Carreras-Vidal, L.; Roura, I.; Capdevila-Lacasa, C.; Oltra, J.; Pardo, J. , et al. Structural brain changes in post-COVID condition and its relationship with cognitive impairment. Brain Commun 2025, 7, fcaf070. [Google Scholar] [CrossRef]
- Telser, J.; Grossmann, K.; Weideli, O.C.; Hillmann, D.; Aeschbacher, S.; Wohlwend, N.; Velez, L.; Kuhle, J.; Maleska, A.; Benkert, P. , et al. Concentrations of Serum Brain Injury Biomarkers Following SARS-CoV-2 Infection in Individuals with and without Long-COVID-Results from the Prospective Population-Based COVI-GAPP Study. Diagnostics (Basel) 2023, 13. [Google Scholar]
- Bark, L.; Larsson, I.M.; Wallin, E.; Simren, J.; Zetterberg, H.; Lipcsey, M.; Frithiof, R.; Rostami, E.; Hultstrom, M. Central nervous system biomarkers GFAp and NfL associate with post-acute cognitive impairment and fatigue following critical COVID-19. Sci Rep 2023, 13, 13144. [Google Scholar] [CrossRef] [PubMed]
- Lempriere, S. Single-cell transcriptomics reveals neuroinflammation in severe COVID-19. Nat Rev Neurol 2021, 17, 461. [Google Scholar] [CrossRef] [PubMed]
- Almutairi, M.M.; Sivandzade, F.; Albekairi, T.H.; Alqahtani, F.; Cucullo, L. Neuroinflammation and Its Impact on the Pathogenesis of COVID-19. Front Med (Lausanne) 2021, 8, 745789. [Google Scholar] [CrossRef]
- Fekete, R.; Simats, A.; Biro, E.; Posfai, B.; Cserep, C.; Schwarcz, A.D.; Szabadits, E.; Kornyei, Z.; Toth, K.; Ficho, E. , et al. Microglia dysfunction, neurovascular inflammation and focal neuropathologies are linked to IL-1- and IL-6-related systemic inflammation in COVID-19. Nat Neurosci 2025, 28, 558–576. [Google Scholar] [CrossRef]
- Vitkovic, L.; Bockaert, J.; Jacque, C. "Inflammatory" cytokines: neuromodulators in normal brain? J Neurochem 2000, 74, 457–471. [Google Scholar] [CrossRef]
- Siclari, F.; Tononi, G. Local aspects of sleep and wakefulness. Curr Opin Neurobiol 2017, 44, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Krueger, J.M.; Nguyen, J.T.; Dykstra-Aiello, C.J.; Taishi, P. Local sleep. Sleep Med Rev 2019, 43, 14–21. [Google Scholar] [CrossRef]
- Pietrobon, A.J.; Andrejew, R.; Custodio, R.W.A.; Oliveira, L.M.; Scholl, J.N.; Teixeira, F.M.E.; de Brito, C.A.; Glaser, T.; Kazmierski, J.; Goffinet, C. , et al. Dysfunctional purinergic signaling correlates with disease severity in COVID-19 patients. Front Immunol 2022, 13, 1012027. [Google Scholar] [CrossRef]
- Olafsdottir, H.F.; Bush, D.; Barry, C. The Role of Hippocampal Replay in Memory and Planning. Curr Biol 2018, 28, R37–R50. [Google Scholar] [CrossRef]
- Zhang, Y.; Ye, G.; Zeng, W.; Zhu, R.; Li, C.; Zhu, Y.; Li, D.; Liu, J.; Wang, W.; Li, P. , et al. Segregation and integration of resting-state brain networks in a longitudinal long COVID cohort. iScience 2025, 28, 112237. [Google Scholar] [CrossRef]
- Verger, A.; Doyen, M.; Heyer, S.; Goehringer, F.; Bruyere, A.; Kaphan, E.; Chine, M.; Menard, A.; Horowitz, T.; Guedj, E. Reorganization of brain connectivity in post-COVID condition: a (18)F-FDG PET study. EJNMMI Res 2025, 15, 28. [Google Scholar] [CrossRef] [PubMed]
- Smallwood, J.; Bernhardt, B.C.; Leech, R.; Bzdok, D.; Jefferies, E.; Margulies, D.S. The default mode network in cognition: a topographical perspective. Nat Rev Neurosci 2021, 22, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Madden, D.; Stephens, T.M.; Scott, J.; O'Neal Swann, C.; Prather, K.; Hoffmeister, J.; Ding, L.; Dunn, I.F.; Conner, A.K.; Yuan, H. Functional connectivity of default mode network in non-hospitalized patients with post-COVID cognitive complaints. Front Neurosci 2025, 19, 1576393. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.; Zhou, C.; Deng, J.; Zhou, J. JAK inhibition as a new treatment strategy for patients with COVID-19. Biochem Pharmacol 2022, 202, 115162. [Google Scholar] [CrossRef] [PubMed]
- Wee, L.E.; Lim, J.T.; Tay, A.T.; Chiew, C.J.; Young, B.E.; Wong, B.; Lim, R.; Lee, C.L.; Tan, J.; Vasoo, S. , et al. Nirmatrelvir/ritonavir treatment and risk for post-acute sequelae of COVID-19 in older Singaporeans: author's response. Clin Microbiol Infect 2025, 31, 139–140. [Google Scholar] [CrossRef]
- Al-Aly, Z. SARS-CoV-2 antivirals and post-COVID-19 condition. Lancet Infect Dis 2025, 25, 6–8. [Google Scholar] [CrossRef]
- AminJafari, A.; Ghasemi, S. The possible of immunotherapy for COVID-19: A systematic review. Int Immunopharmacol 2020, 83, 106455. [Google Scholar] [CrossRef]
- Zhang, Q.; Niu, Y.; Li, Y.; Xia, C.; Chen, Z.; Chen, Y.; Feng, H. Meningeal lymphatic drainage: novel insights into central nervous system disease. Signal Transduct Target Ther 2025, 10, 142. [Google Scholar] [CrossRef]
- Nam, M.H.; Sa, M.; Ju, Y.H.; Park, M.G.; Lee, C.J. Revisiting the Role of Astrocytic MAOB in Parkinson's Disease. Int J Mol Sci 2022, 23. [Google Scholar] [CrossRef]
- Lee, D.H.; Seubert, S.; Huhn, K.; Brecht, L.; Rotger, C.; Waschbisch, A.; Schlachetzki, J.; Klausmeyer, A.; Melms, A.; Wiese, S. , et al. Fingolimod effects in neuroinflammation: Regulation of astroglial glutamate transporters? PLoS One 2017, 12, e0171552. [Google Scholar]
- Oliveros, G.; Wallace, C.H.; Chaudry, O.; Liu, Q.; Qiu, Y.; Xie, L.; Rockwell, P.; Figueiredo-Pereira, M.E.; Serrano, P.A. Repurposing ibudilast to mitigate Alzheimer's disease by targeting inflammation. Brain 2023, 146, 898–911. [Google Scholar] [CrossRef] [PubMed]
- Toti, A.; Micheli, L.; Lucarini, E.; Ferrara, V.; Ciampi, C.; Margiotta, F.; Failli, P.; Gomiero, C.; Pallecchi, M.; Bartolucci, G. , et al. Ultramicronized N-Palmitoylethanolamine Regulates Mast Cell-Astrocyte Crosstalk: A New Potential Mechanism Underlying the Inhibition of Morphine Tolerance. Biomolecules 2023, 13. [Google Scholar] [CrossRef]
- Dong, H.; Wang, Y.; Zhang, X.; Zhang, X.; Qian, Y.; Ding, H.; Zhang, S. Stabilization of Brain Mast Cells Alleviates LPS-Induced Neuroinflammation by Inhibiting Microglia Activation. Front Cell Neurosci 2019, 13, 191. [Google Scholar] [CrossRef]
- Ramirez-Ponce, M.P.; Flores, J.A.; Barrella, L.; Ales, E. Ketotifen is a microglial stabilizer by inhibiting secretory vesicle acidification. Life Sci 2023, 319, 121537. [Google Scholar] [CrossRef]
- Thangam, E.B.; Jemima, E.A.; Singh, H.; Baig, M.S.; Khan, M.; Mathias, C.B.; Church, M.K.; Saluja, R. The Role of Histamine and Histamine Receptors in Mast Cell-Mediated Allergy and Inflammation: The Hunt for New Therapeutic Targets. Front Immunol 2018, 9, 1873. [Google Scholar] [CrossRef]
- Aldossari, A.A.; Assiri, M.A.; Ansari, M.A.; Nadeem, A.; Attia, S.M.; Bakheet, S.A.; Albekairi, T.H.; Alomar, H.A.; Al-Mazroua, H.A.; Almanaa, T.N. , et al. Histamine H4 Receptor Antagonist Ameliorates the Progression of Experimental Autoimmune Encephalomyelitis via Regulation of T-Cell Imbalance. Int J Mol Sci 2023, 24. [Google Scholar] [CrossRef] [PubMed]
- Mehranfard, D.; Speth, R.C. Cholinergic anti-inflammatory pathway and COVID-19. Bioimpacts 2022, 12, 171–174. [Google Scholar]
- Khan, M.W.Z.; Ahmad, M.; Qudrat, S.; Afridi, F.; Khan, N.A.; Afridi, Z.; Fahad; Azeem, T. ; Ikram, J. Vagal nerve stimulation for the management of long COVID symptoms. Infect Med (Beijing) 2024, 3, 100149. [Google Scholar] [CrossRef] [PubMed]
- Wood, G.K.; Sargent, B.F.; Ahmad, Z.U.; Tharmaratnam, K.; Dunai, C.; Egbe, F.N.; Martin, N.H.; Facer, B.; Pendered, S.L.; Rogers, H.C. , et al. Posthospitalization COVID-19 cognitive deficits at 1 year are global and associated with elevated brain injury markers and gray matter volume reduction. Nat Med 2025, 31, 245–257. [Google Scholar] [CrossRef]
- Calzari, L.; Dragani, D.F.; Zanotti, L.; Inglese, E.; Danesi, R.; Cavagnola, R.; Brusati, A.; Ranucci, F.; Di Blasio, A.M.; Persani, L. , et al. Epigenetic patterns, accelerated biological aging, and enhanced epigenetic drift detected 6 months following COVID-19 infection: insights from a genome-wide DNA methylation study. Clin Epigenetics 2024, 16, 112. [Google Scholar] [CrossRef]
- Saunders, E.G.; Pouliopoulou, D.V.; Miller, E.; Billias, N.; MacDermid, J.C.; Brunton, L.; Pereira, T.V.; Quinn, K.L.; Bobos, P. Rehabilitation interventions and outcomes for post-COVID condition: a scoping review. BMJ Public Health 2025, 3, e001827. [Google Scholar] [CrossRef] [PubMed]
- Bailey, J.; Lavelle, B.; Miller, J.; Jimenez, M.; Lim, P.H.; Orban, Z.S.; Clark, J.R.; Tomar, R.; Ludwig, A.; Ali, S.T. , et al. Multidisciplinary Center Care for Long COVID Syndrome-A Retrospective Cohort Study. Am J Med 2025, 138, 108–120. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




