Submitted:
10 June 2025
Posted:
12 June 2025
You are already at the latest version
Abstract
Keywords:
1. Experimental Design and Sample Testing
1.1. Materials, Reagents and Instruments
1.2. Material Pre-Treatment
1.3. Experimental Design
1.4. Sample Testing and Data Processing Methods
2. Results and Analysis
2.1. Single Factor Experiment of Iron-Carbon Micro-Electrolysis
2.1.1. Effect of Different Reaction Times
2.1.2. Effect of Initial pH Value
2.1.3. Effect of Different Fe/C Mass Ratios
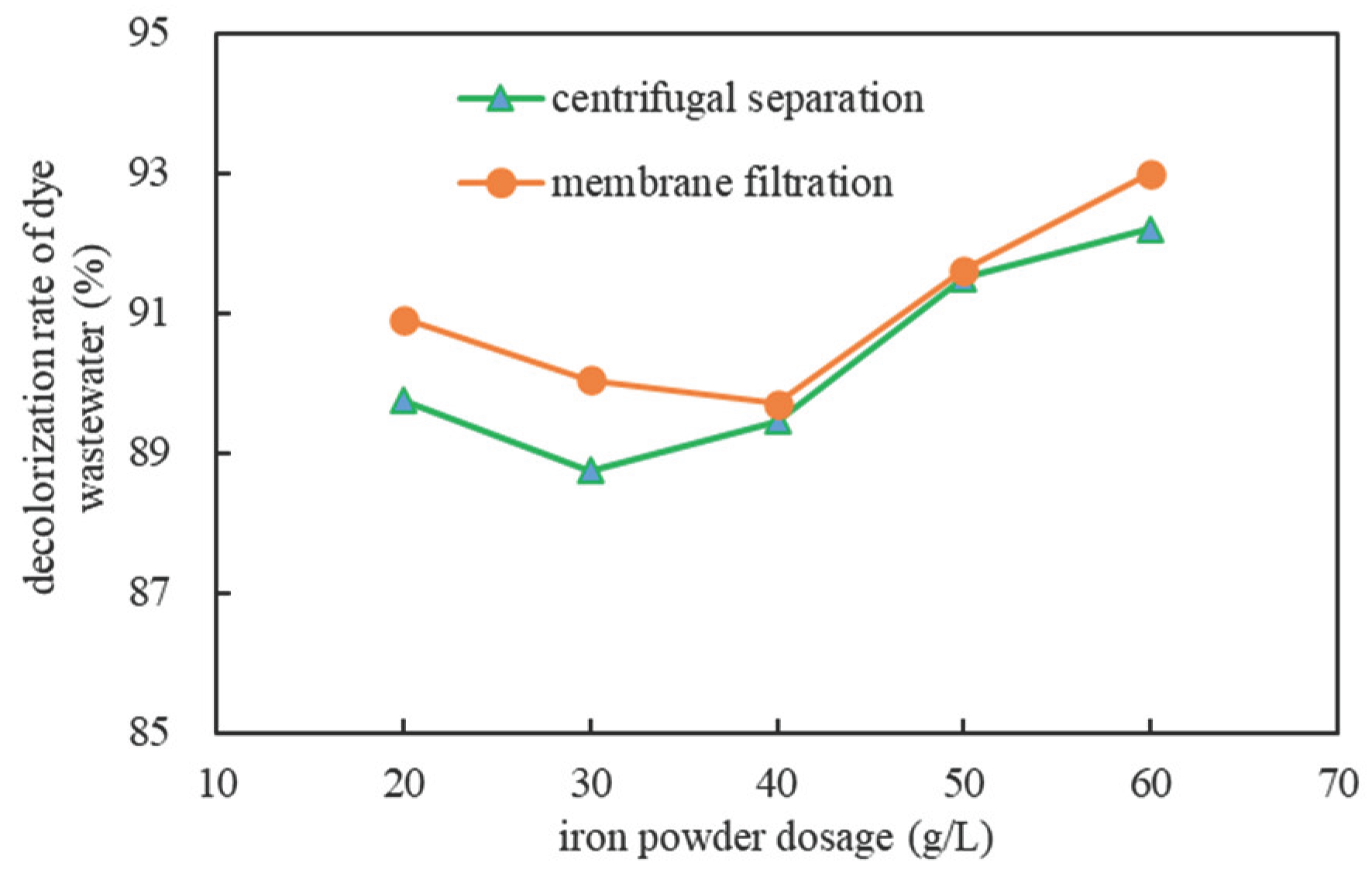
2.1.4. The Influence of Different Separation Methods
2.1.5. Iron-Carbon Micro-Electrolysis Experiments Under Optimal Conditions
2.2. Analysis of the Treatment Effect Of Iron-Carbon Micro-Electrolysis Combined Process
2.2.1. Iron-Carbon Micro-Electrolysis and Coagulation Sedimentation Combined Process
2.2.2. Iron Carbon Micro-Electrolysis and Fenton Oxidation Combined Process
3. Conclusion
References
- LEME D M, PRIMO F L, GOBO G G, et al. Genotoxicity assessment of reactive and disperse textile dyes using human dermal equivalent (3D cell culture system) [J]. J Toxicol Environ Heal A, 2015,78(7): 466-480. [CrossRef]
- Sun Xiaoxu, Xu Jin, Chu Zhiqiang. Experimental study of absorption of methylene blue with activated carbon prepared by modification pomelo peel in different methods [J]. Journal of Hohai University(Natural Sciences), 2021,49(06):536-542. [CrossRef]
- Li Zhuobing. Study on the preparation and adsorption characteristics of auricula chaff charcoal [D]. Northeast Electric Power University,2024.
- Zhang Changfeng. Study on preparation of almond shell-based activated carbon and its adsorption of methylene blue [J]. Industrial Microbiology, 2023, 53(03):59 -62.
- Luo Xinhao. Study on O3-coagulation enhanced pretreatment and O3-SBBR advanced treatment of wastewater from a printing and dyeing industrial park in Foshan [D]. South China University of Technology, 2022.
- Ding jing, Zhang jianliang, Ren ye, et al. Treatment of dyeing wastewater by a combined process of coagulation,A/O,flotation,ozone, and magnetic coagulation [J]. China water & wastewater, 2021, 37(20):112-115.
- Li Zhonghua, Chang Na, Xu Yinong et al. Fenton process treating small pore-sized ultrafiltration membrane concentrate of printing and dyeing wastewater [J]. Chinese Journal of Environmental Engineering,2024,18(01):41-50. [CrossRef]
- Feng Yabing, Sun Rong, Zhao Jing,et al. Engineering design of Fenton oxidation process in advanced treatment of printing and dyeing wastewater [J]. Water and wastewater Engineering, 2023,59(07):58 -63.
- He Yujie, Chen Wei, Zheng Xiaoying, et al. Pretreatment experiment of high-concentration aromatic tobacco flavor wastewater with Fenton reagent and PAM coagulation [J]. Journal of Hohai University(Natural Sciences),2012,40(05):525-529.
- Jing Xinjun, Cai Daniu, Li Bin, et al. Progress in the deep treatment technology of printing and dyeing wastewater [J]. Technology of Water Treatment, 2022,48(6):13 -19.
- Jing Xinjun. Study on the advanced treatment process of high concentration printing and dyeing wastewater [D]. Hebei University of Technology, 2023.
- Zhang Lianke, Wang Chang, Li Yumei et al. Mechanism of complex superior oxidation system Vis/LaFeO3 /PDS for methylene blue degradation [J]. Water Resources Protection,2021,37(06):142-149.
- Zhang Zhen. Study on the efficiency and mechanism of iron carbon microelectrolysis treatment of dyeing wastewater [D]. Northeast Electric Power University,2020.
- Zhen-Zhu Sun, Zhong-Hai Liu, Le Han, et al. Study on the treatment of simulated azo dye wastewater by a novel micro-electrolysis filler [J]. Water Science & Technology,2019,79(12): 2279 -2288. [CrossRef]
- LIU W W, TU X Y, WANG X P,et al. Pretreatment of coking wastewater by acid out micro-electrolysis process with in situ electrochemical peroxidation reaction [J]. Chem Eng J, 2012,200: 720 -728.
- ZHU Q S, GUO S H,GUO C M,et al. Stability of Fe-C micro-electrolysis and biological process in treating ultra-high concentration organic wastewater [ J].Chem Eng J,2014,255:535 -540.
- Chen Kun, Gong Minghui, Li Rui, et al. Comprehensive Experimental Design of Fe-C Micro-electrolysis for Treating Dye Wastewate [J]. Research and Exploration in Laboratory,2023,42(04):183 -187+193.
- HE Wei, LI Guiju, SONG Jie et al. Research on a new type of micro-electrolysis material for the degradation of printing and dyeing wastewater [J]. Industrial Water Treatment,2018,38(10):71-74.
- JIA Yanping, ZHANG Zhen, TONG Zewei et al. Study on efficiency and mechanism of iron-carbon microelectrolysis treatment of dyeing wastewater [J]. CIESC Journal, 2020, 71(4): 1791-1801.
- SHE Shuaiqi, CHEN Hong, XUE Gang, et al. Mechanism for treatment of dyeing wastewater by Fe-C micro-electrolysis [J]. Environmental Protection of Chemical Industry,2021,41(6):699-704.
- Ren Qingqing. Removal of direct dyes and reactive dyes from printing and dyeing wastewater by microelectrolysis and the mechanism of decolorization [D]. Lanzhou Jiaotong University,2022.
- Chen G. Study on biological decolorization of azo dye wastewater and aerobic degradation performance of typical decolorization products [D].Donghua University,2012.
- Liu Xiaobo. Study on the Effect and Decolorization Mechanism of Azo Dye Wastewater Treatment by Micro Electrolysis [D]. Lanzhou Jiaotong University,2021.
- BAI Bo, CHEN Zhihong, WANG Liping. Treatment of direct scarlet 4BE dye wastewater by the hybrid method of Fe/C microelectrolysis and ultrasound radiation [J]. Applied Chemical Industry,2007, 36(02): 130-133.
- FAN Jinhui, ZHOU Weidong, YANG Xiedong, et al. Effect of Fe2+ and PMS synchronous coagulation and oxidation pretreatment on purification of sludge water by ultrafiltration [J].Water Resources Protection, 2021, 37(02):148-152.
- LI Chenlu, GUO Yani, ZHENG Libing et al. DOM removal characteristics and coagulation mechanism of silica sand loading coagulation process [J]. Water Resources Protection,2021,37(4):148-155.
- WEI Yuansong, ZHENG Libing, ZHANG Chun et al. Progress of application and research of advanced treatment technologies for reclaimed water reuse in thermal power generation plant in China [J]. Water Resources Protection,2018,34(6):1-11+16.
- Feng Yabing, Sun Rong, Zhao Jing et al. Engineering design of Fenton oxidation process in advanced treatment of printing and dyeing wastewater [J]. Water &Wastewater Engineering,2023,49(7): 58-63.
- Zhang Kunpu. Study on treatment of aromatic compounds in printing and dyeing wastewater by fenton oxidation [D]. Beijing University of Chemical Technology,2023.
- ZHAO Hazy, LI Mei, BAI Maomao et al. Research progress of advanced oxidation technology in dyeing wastewater treatment [J]. Applied Chemical Industry,2023,52(6):1884-1890.
- Qingwei, Zhao Tingyue, Liu Jian et al. Pretreatment of phosphoric ester flame-retardant wastewater with Fenton oxidation technique [J]. Water Resources Protection,2016,32(6):90-92.
- Yang Xingxia. Study on the performance of PAM-PAFC in degrading printing and dyeing wastewater [D]. Guizhou University,2022.
- Chen Xi, Wu Linfeng, Zhang Li et al. Advanced treatment of practical dyeing wastewater by electron beam coupled with coagulation technology [J]. Industrial Water Treatment, 2025,45(05):143-150.
- SUN Shaozhe, SHEN Yaxue, LI Jun et al. Analysis on Flocculation Decolorization and COD Removal of Dye Wastewater by Aluminum Salt Coagulant [J]. Contemporary Chemical Industry, 2025,54(04):972-977.
- PANG Yue, LOU Honghai, GAO Rui et al. Study on Advanced Treatment of Dyeing Wastewater by Fenton Oxidation [J]. Technology of Water Treatment,2023,49(05):40-44.
- QIAO Tianyu, YANG Kailin, FENG Mingming et al. Research Progress on Application of Micro Electrolysis-Fenton Oxidation Combined Technology in Industrial Wastewater Treatment [J]. China Resources Comprehensive Utilization,2023,41(7):89-92.
- Kong Zhangliang. Treatment of Azo Dye Wastewater by Iron-Carbon Microelectrolysis Combined with Fenton Reagent [D]. Jiangxi University of Science and Technology,2019.
- Yan Wang, Xianwei Wu, Ju Yi, et al. Pretreatment of printing and dyeing wastewater by Fe/C micro-electrolysis combined with H2O2 process [J]. Water Science & Technology,2018,244:1-11. [CrossRef]
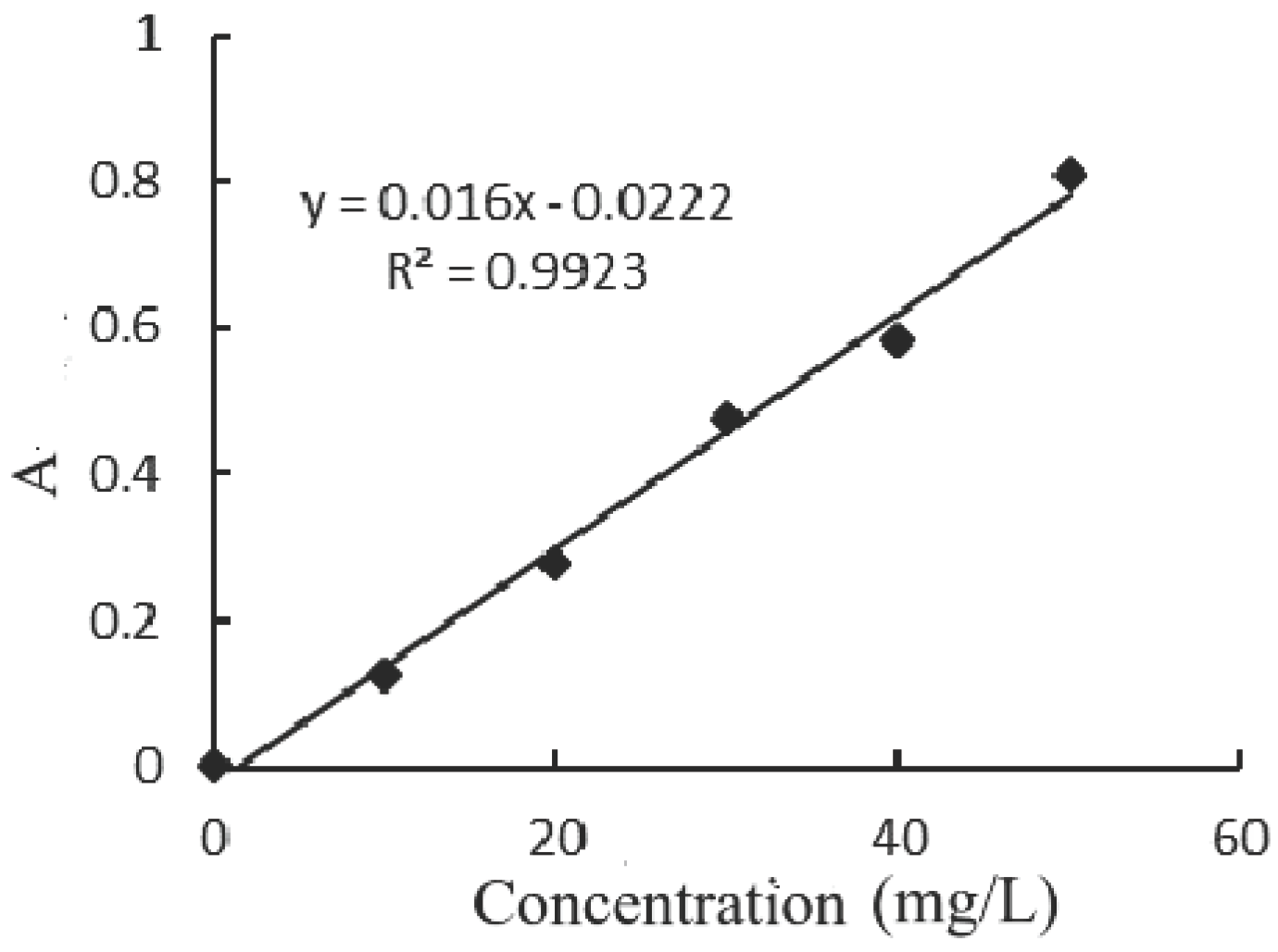


| Reaction time (min) | Absorbance after treatment | Concentration after treatment (mg/L) |
Decolorization rate (%) |
| 50 | 0.309 | 20.7 ×25 | 65.50 |
| 70 | 0.211 | 14.58 × 25 | 75.70 |
| 90 | 0.213 | 14.70 ×10 | 90.20 |
| 110 | 0.797 | 51.20 | 96.59 |
| 130 | 0.184 | 12.89 | 99.14 |
| Initial pH | Absorbance after treatment | Concentration after treatment (mg/L) | Decolorization rate (%) |
| 3 | 0.031 | 3.33 | 99.78 |
| 4 | 0.065 | 5.45 | 99.64 |
| 5 | 0.157 | 11.20 | 99.25 |
| 6 | 0.111 | 8.325×10 | 94.45 |
| 7 | 0.357 | 23.70×10 | 84.20 |
| iron-carbon ratio | Absorbance after treatment | Concentration after treatment (mg/L) |
Decolorization rate (%) |
| 2:1 | 0.165 | 11.70×10 | 92.20 |
| 1:1 | 0.213 | 14.70×10 | 90.20 |
| 1:2 | 0.305 | 20.45 | 98.64 |
| 1:3 | 0.055 | 4.83 | 99.68 |
| Separation | Iron powder dosage (g/L) | Absorbance after treatment (Dilute 10 times) |
Concentration after Treatment(mg/L) |
Decolorization rate (%) |
| centrifugal separation |
20 | 0.244 | 16.638×10 | 88.91 |
| 30 | 0.268 | 18.137×10 | 87.91 | |
| 40 | 0.251 | 17.075×10 | 88.62 | |
| 50 | 0.202 | 14.013×10 | 90.66 | |
| 60 | 0.185 | 12.950×10 | 91.37 | |
| membrane filtration |
20 | 0.216 | 14.887×10 | 90.08 |
| 30 | 0.237 | 16.200×10 | 89.20 | |
| 40 | 0.245 | 16.700×10 | 88.87 | |
| 50 | 0.199 | 13.825×10 | 90.78 | |
| 60 | 0.166 | 11.763×10 | 92.16 |
| Wastewater | color | Dye concentration (mg/L) | Chromaticity (times) |
| pretreatment | reddish-black | 1500 | 80000 |
| after treatment | light orange | 14.51 | 3000 |
| Wastewater | Color | Dye concentration (mg/L) |
Chromaticity (times) |
| Unpurified water | Reddish-black | 1500 | 80000 |
| After Iron-carbon micro-electrolysis treatment |
Orange-red | 14.51 | 3000 |
| After coagulation and sedimentation treatment | Light yellow | 3.51 | 60 |
| Waster water | Color | Dye concentration (mg/L) |
Chromaticity (times) |
| Unpurified water | Reddish-black | 1500 | 80000 |
| After Iron-carbon micro-electrolysis treatment |
Orange-red | 14.51 | 3000 |
| After Fenton oxidation | Light yellow | 3.14 | 40 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




