Submitted:
24 June 2025
Posted:
26 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
sFLC Biology
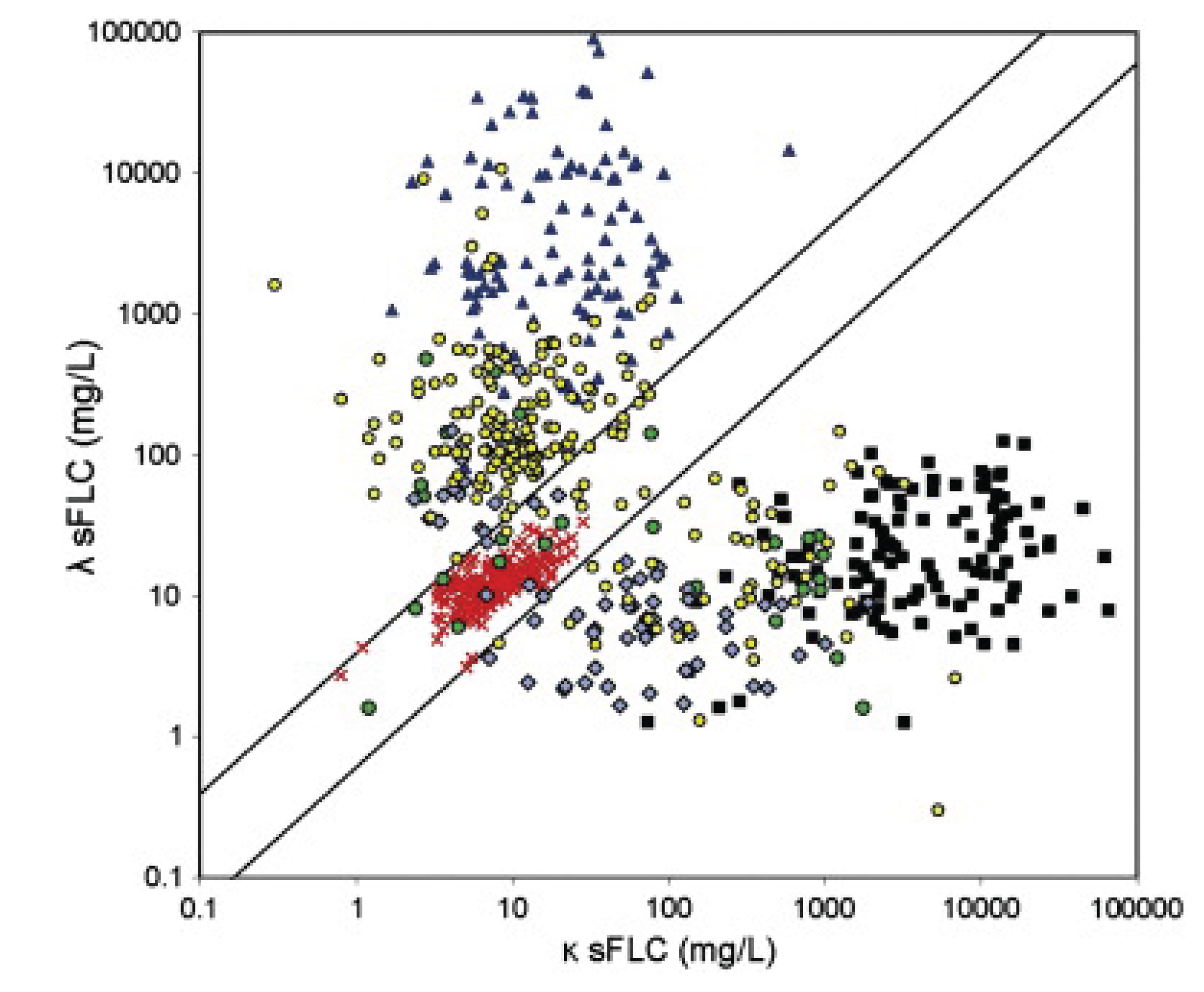
Clinical Relevance: Neoplasia Versus Inflammation
- Hegen, et al., reported on cerebrospinal fluid kappa free light chains as useful biomarker in multiple sclerosis from diagnosis to prediction of disease activity. (Hegen, 2022)
- Demortiere, et al. found FLC levels in patients with inaugural optic neuritis were useful to sort out multiple sclerosis (MS), myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) and neuromyelitis spectrum disorder (NMOSD). (Demortiere, 2025)
- Bracco et al. found the presence of FLC in cerebrospinal fluid of multiple sclerosis patients implicated a recent immunological stimulation leading to increased synthesis of FLC within the central nervous system. (Bracco, 1987)
- Matsumori et al. found that sFLCR was a “…more specific and sensitive for the diagnosis of T2D than HbA1c, and thus represents a potentially promising biomarker of inflammation.” (Matsumori, 2020)
- Basile, et al. found sFLCR > 0.63 was associated with left ventricular ejection fraction improvement in a small series of patients with NSTEMI, STEMI and stable angina at one year follow up. (Basile, 2019)
- Nakao, H. reported an increase in IgA nephropathy following COVID-19 mRNA vaccination. (Nakao, 2023)
- Park and Kwon pointed out the role of monoclonal FLC in producing kidney damage in Monoclonal Gammopathy of Renal Significance (MGRS) without multiple myeloma or other forms of neoplasia. (Park, 2023).
- Martins, et al. reported on 23 cases of non-myeloma light chain cast nephropathy (non MM-LCCN) pointing out that malignancy develops later in 43% of cases. (Martins, 2024).
- Lan, et al. presented a case of light chain proximal tubulopathy (LCPT) and light chain cast nephropathy (LCCN) in a 49 year-old patient with acute kidney injury associated with of lambda light chain multiple myeloma (LCMM). (Lan, 2024).
- Cassano, et al. reported on light chain deposition disease (LCDD) in which non-amyloid monoclonal light chains are deposited in different organs particularly kidney where monoclonal immunoglobulins are deposited in vascular basement membranes, glomerular basement membranes and tubular basement membranes in patients with plasma cell dyscrasias but also monoclonal gammopathy of unknown significance (MGUS). (Cassano, 2025).
- Gudowska-Sawczuk, et al. found COVID-19 vaccinated subjects had higher sFLC levels than COVID-19 patients and unvaccinated controls. (Gudowska-Sawczuk, 2022).
2. Materials and Methods
| Type | sFLCR | sFLC |
|---|---|---|
| Inflammation | 0.26–1.65 | k, λ, k & λ |
| Neoplasia | <0.26 or >1.65 | k, λ, k & λ |
3. Results
- Case LC #11 (VAERS ID# 2619645) received five doses of COVID-19 vaccines, three Moderna and two Pfizer, and was subsequently identified as having Monoclonal Gammopathy of Unknown Significance on the basis of elevation of sFLCk (2.75 mg/dl) with normal sFLCR (1.31). Serial sFLC determinations should be considered in such a case.
- Case# LC 63 (VAERS ID# 2509414 was a 60 year old male who presented 1 week after a second dose of BNT162b2 with weight gain, proteinuria, and hypoalbumenia. Urinary Bence Jones proteins were positive, both sFLCk (300 mg/L) and sFLCl (384 mg/L) were elevated but the ratio was normal at 0.78. Bone marrow biopsy demonstrated 10% plasma cell aggregates with amyloid deposits consistent with myeloma.
- Case LC# 24 (VAERS ID# 2516749) was a 41 year old woman with HLAB27+ arthritis in remission had onset of nephrotic syndrome 7-10 days after dose four of BNT162b2 with lower extremity edema, hematuria, proteinuria, hypoalbuminemia, and elevated sFLCk (37.74 mg/L) and sFLCl (37.95 mg/L) with sFLCR normal (0.99). Renal biopsy revealed focal segmental glomerulosclerosis (FSGS).
- LC# 42 (VAERS ID# 2267797) was a 47 year old woman received three doses of mRNA1273 6/4/2021, 7/2/2021 and the date of the third dose was not given. In November of 2021 she had onset of anemia, an IgG monoclonal band, and an elevated sFLCl (173 mg/L) with sFLCR of 0.09. Bone marrow biopsy showed 7-9% monoclonal plasma cell infiltration.
- Case LC #16 (VAERS ID# 2130959) was a 46 year old woman with past history of hypothyroidism and labile hypertension who had onset of severe bilateral knee pain and flu-like symptoms 31 days after her second dose of mRNA1273. A rheumatology consultation was obtained who obtained sFLCs showing sFLCk was 72.87 mg/dL with a ratio of 165.61.
4. Discussion
COVID-19, COVID-19 Vaccines and sFLC Patterns
| Pattern | N | Kappa | Lambda | Ratio |
|---|---|---|---|---|
| 1. Normal r, Kappa Elevated | 3 | 32.70 | 23.04 | 1.41 |
| 2. Normal r, Kappa & Lambda Elevated | 7 | 105.38 | 111.61 | 1.00 |
| 3. Elevated r, Kappa Elevated | 5 | 103.48 | 17.06 | 9.45 |
| 4. Elevated r, Kappa & Lambda Elevated | 5 | 158.64 | 74.69 | 34.74 |
| 5. Low r, Lambda Elevated | 3 | 64.48 | 231.67 | 0.09 |
| 6. Elevated r, Lambda Decreased8 | 2 | 7.05 | 3.49 | 2.07 |
| Avg. | 94.64 | 95.319 | 9.46 |
Limitations
5. Conclusions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Aklaghi, K., Maclachlan, K., Korde, N., Mailankody, S., Lesokhin, A., Hassoun, H., Lu, S., Patel, D., Shah, U., Tan, C., Hultcrantz, M., Iyengar, N., Shah, G. L., Scordo, M., Lahoud, O. B., Chung, D. J., & Landau, H. J. (2025). Evaluating serum free light chain ratio as a biomarker in multiple myeloma. Haematologica, 110(1), 326–338. [CrossRef]
- Basile, U., La Rosa, G., Napodano, C., Pocino, K., Cappannoli, L., Gulli, F., Cianfrocca, C., Di Stasio, E., & Biasucci, L. M. (2019). Free light chains: A novel biomarker of cardiovascular disease. A pilot study. European Review for Medical and Pharmacological Sciences, 23(6), 2563–2569. [CrossRef]
- Bird, J., Behrens, J., Westin, J., Turesson, I., Drayson, M., Beetham, R., D’Sa, S., Soutar, R., Waage, A., Gulbrandsen, N., Gregersen, H., Low, E. and (2009), UK Myeloma Forum (UKMF) and Nordic Myeloma Study Group (NMSG): guidelines for the investigation of newly detected M-proteins and the management of monoclonal gammopathy of undetermined significance (MGUS). British Journal of Haematology, 147: 22-42. [CrossRef]
- Bracco, F., Gallo, P., Menna, R., Battistin, L., & Tavolato, B. (1987). Free light chains in the CSF in multiple sclerosis. Journal of Neurology, 234(5), 303–307. [CrossRef]
- Cassano, R., Ferraro, S., Stella, A., Buda, G., Orciuolo, E., & Petrini, M. (2025). Light chain deposition disease: Pathogenesis, clinical characteristics, and treatment strategies. Annals of Hematology, 104(7), 2083–2093. [CrossRef]
- Davids, M. S., Murali, M. R., & Kuter, D. J. (2010). Serum free light chain analysis. American Journal of Hematology, 85(10), 787–790. [CrossRef]
- Demortiere, S., Marignier, R., Bertheaume, N., Vukusic, S., D’Hardivilliers, F., & Lebrun-Frenay, C. (2025). Diagnostic utility of kappa free light chain index in adults with inaugural optic neuritis. Neurology: Neuroimmunology & Neuroinflammation, 12(1), Article e200386. [CrossRef]
- Dispenzieri, A. (2019). POEMS syndrome: 2019 update on diagnosis, risk-stratification, and management. American Journal of Hematology, 94(7), 812–827. [CrossRef]
- Fend, F., Dogan, A., & Cook, J. R. (2023). Plasma cell neoplasms and related entities—evolution in diagnosis and classification. Virchows Archiv, 482(1), 163–177. [CrossRef]
- Gertz, M. A. (2024). Immunoglobulin light chain amyloidosis: 2024 update on diagnosis, prognosis, and treatment. American Journal of Hematology, 99(2), 309–324. [CrossRef]
- Gudowska-Sawczuk, M., & Mroczko, B. (2023). Free light chains k and λ as new biomarkers of selected diseases. International Journal of Molecular Sciences, 24(11), Article 9531. [CrossRef]
- Gudowska-Sawczuk, M., Moniuszko-Malinowska, A., Pączek, S., Guziejko, K., Chorąży, M., & Mroczko, B. (2022). Evaluation of free light chains (FLCs) synthesis in response to exposure to SARS-CoV-2. International Journal of Molecular Sciences, 23(19), Article 11589. [CrossRef]
- Hegen, H., Arrambide, G., Gnanapavan, S., Kaplan, B., Khalil, M., Saadeh, R., Teunissen, C., Tumani, H., Villar, L. M., Willrich, M. A. V., & Zettl, U. K. (2022). Cerebrospinal fluid kappa free light chains for the diagnosis of multiple sclerosis: A consensus statement. Multiple Sclerosis Journal, 29(2), 182–195. [CrossRef]
- Jenner, E. (2014). Serum free light chains in clinical laboratory diagnostics. Clinica Chimica Acta, 427, 15–20. [CrossRef]
- Jones, H. B. (1848). On a new substance occurring in the urine of a patient with mollities ossium. Philosophical Transactions of the Royal Society, 138, 55–62. [CrossRef]
- Kaplan, B., Livneh, A., & Sela, B. A. (2011). Immunoglobulin free light chain dimers in human diseases. The Scientific World Journal, 11, 726–735. [CrossRef]
- Katzmann, J., Clark, R., Abraham, R., Bryant, Lymp, S., Bradwell, A., Kyle, R. Serum Reference Intervals and Diagnostic Ranges for Free κ and Free λ Immunoglobulin Light Chains: Relative Sensitivity for Detection of Monoclonal Light Chains, Clinical Chemistry, Volume 48, Issue 9, 1 September 2002, Pages 1437–1444. [CrossRef]
- Katzman, J. A., Kyle, R. A., Benson, J., Larson, D. R., Snyder, M. R., Lust, J. A., Rajkumar, S. V., & Dispenzieri, A. (2009). Screening panels for detection of monoclonal gammopathies. Clinical Chemistry, 55(8), 1517–1522. [CrossRef]
- Kyle, R. A., & Rajkumar, S. V. (2008). Multiple myeloma. Blood, 111(6), 2962–2972. [CrossRef]
- Kyle, R. A., Larson, D. R., Therneau, T. M., Dispenzieri, A., Kumar, S., Cerhan, J. R., & Rajkumar, S. V. (2018). Long-term follow-up of monoclonal gammopathy of undetermined significance. New England Journal of Medicine, 378(3), 241–249. [CrossRef]
- Lan, M., Guo, Y., Wang, C., Wang, X., Li, J., & Wang, Y. (2024). Lambda light chain–restricted non-crystalline proximal tubulopathy with cast nephropathy in multiple myeloma: A case report and literature review. BMC Nephrology, 25(1), Article 325. [CrossRef]
- Malecka-Gieldowska, M., Folta, M., Wisniewska, A., & Czyzewska, E. (2021). Cell population data and serum polyclonal immunoglobulin free light chains in the assessment of COVID-19 severity. Viruses, 13(7), Article 1381. [CrossRef]
- Martins, C., Gibier, J., Leroy, X., Bridoux, F., Touchard, G., Joly, D., Royal, V., Goujon, J. M., & Sirac, C. (2024). Non-myeloma light chain cast nephropathy: A multicenter retrospective study on clinicopathological characteristics. Haematologica, 109(8), 2557–2566. [CrossRef]
- Matsumori, A., Shimada, T., Shimada, M., & Drayson, M. T. (2020). Immunoglobulin free light chains: An inflammatory biomarker of diabetes. Inflammation Research, 69(7), 715–718. [CrossRef]
- Nakao, H., Koseki, T., Kato, K., Yamada, S., Tsuboi, N., Takahashi, K., & Mizuno, T. (2023). COVID-19 mRNA vaccination is associated with IgA nephropathy: An analysis of the Japanese adverse drug event report database. Journal of Pharmacy & Pharmaceutical Sciences, 26, Article 11453. [CrossRef]
- Park, K., & Kwon, S. (2024). Monoclonal gammopathy of renal significance from the perspective of nephrologists. Blood Research, 59(1), Article 28. [CrossRef]
- Salama, M., & Hoffman, R. (2023). Progress in the classification of hematopoietic and lymphoid neoplasms: Clinical implications. In R. Hoffman (Ed.), Hematology: Basic principles and practice (8th ed., pp. 800–812). Elsevier. [CrossRef]
- xAI. (n.d.). Grok 3 (Version 3) [Artificial intelligence language model]. xAI. https://x.ai.
- Zhu, L., Hu, Q., Zhang, L., & Li, A. (2024). The role of minimal residual disease and serum free light chain ratio in the management of multiple myeloma. Discover Oncology, 15(1), Article 229. [CrossRef]
| 1 | Neoplastic and pre or para neoplastic conditions are combined for this analysis. |
| 2 | Outlier removed LC #29 l = 5975. |
| 3 | Reference values: sFLCk 3.3-19.4 mg/L, sFLCl 5.7-26.6 mg/L, sFLCR= 0.26-1.65 |
| 4 | Elevated sFLC values with abnormal ratios are in red. |
| 5 | Outlier removed LC # 29 l = 5975 |
| 6 | Malecka-Gieldowska, 2021 |
| 7 | Gudowska-Sawczuk, 2022 |
| 8 | Possible suppression of l producing plasma cells |
| 9 | Outlier removed LC # 29 l = 5975 |
| Classification of Plasma Cell Neoplasms and Paraneoplastic Conditions (Katzman, 2009; Davids, 2010; Kaplan, 2011; Kyle, 2018; Dispenzieri, 2019; Zhu, 2024; Gertz, 2024; Aklaghi, 2025) |
| 1. Non-IgM MGUS |
| 2. Smoldering myeloma |
| 3. Multiple myeloma |
| 4. Solitary bone plasmacytoma |
| 5. Solitary extraosseous plasmacytoma |
| 6. Immunoglobulin light chain amyloidosis |
| 7. Localized AL amyloidosis |
| 8. Waldenström macroglobulinemia |
| 9. Light chain deposition disease (LCDD) |
| 10. POEMS syndrome (polyneuropathy, organomegaly, endocrinopathy, M-protein, skin changes) |
| Monoclonal Gammopathies | Diabetes |
| Multiple sclerosis | Cardiovascular disease |
| SARS-CoV-2 infection | Rheumatoid arthritis |
| HCV | Sjogren’s syndrome |
| HBV | SLE |
| HIV | Lung cancer |
| Lyme Disease | Breast cancer |
| Tick-born encephalitis | Bowel Disease |
| sFLCk | sFLCl | sFLCR | |
|---|---|---|---|
| Avg | 96.13 | 95.32 | 9.863 |
| Min | 4.27 | 0.44 | 0.01 |
| Max | 338 | 5975 | 165.61 |
| Group | kFLC (mg/L) | λFLC (mg/L) | k/λ Ratio |
|---|---|---|---|
| Reported AEs after COVID-19 Vaccine n = 25 |
94.6 (7.05–300) | 95.31 (3.49–384)5 | 9.46 (0.09–165.61) |
| COVID-19 ICU6 n = 45 |
47.03 (43.52–64.76) | 34.71 (30.66–47.23) | 1.34 (1.20–1.52) |
| COVID-19 non-ICU2 n = 43 |
24.62 (21.22–36.45) | 25.83 (19.26–28.38) | 1.27 (1.06–1.35) |
| Vaccinated Controls7 n = 20 |
17.83 ± 3.03 (12.10–23.70) | 13.22 ± 3.87 (9.24–22.00) | 1.40 ± 0.24 (0.88–1.77) |
| Mild COVID-193 n = 80, (67 vaccinated) |
16.76 ± 5.51 (5.25-42.50) | 16.38 ± 6.17 (6.32-36.50) | 1.10 ± 0.28 (0.44-1.94) |
| Non-Vaccinated Controls2 n = 20 |
10.25 ± 2.13 (6.28–15.04) | 10.26 ± 2.76 (6.84–18.89) | 1.03 ± 0.22 (0.51–1.41) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




