Submitted:
09 June 2025
Posted:
10 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Methods
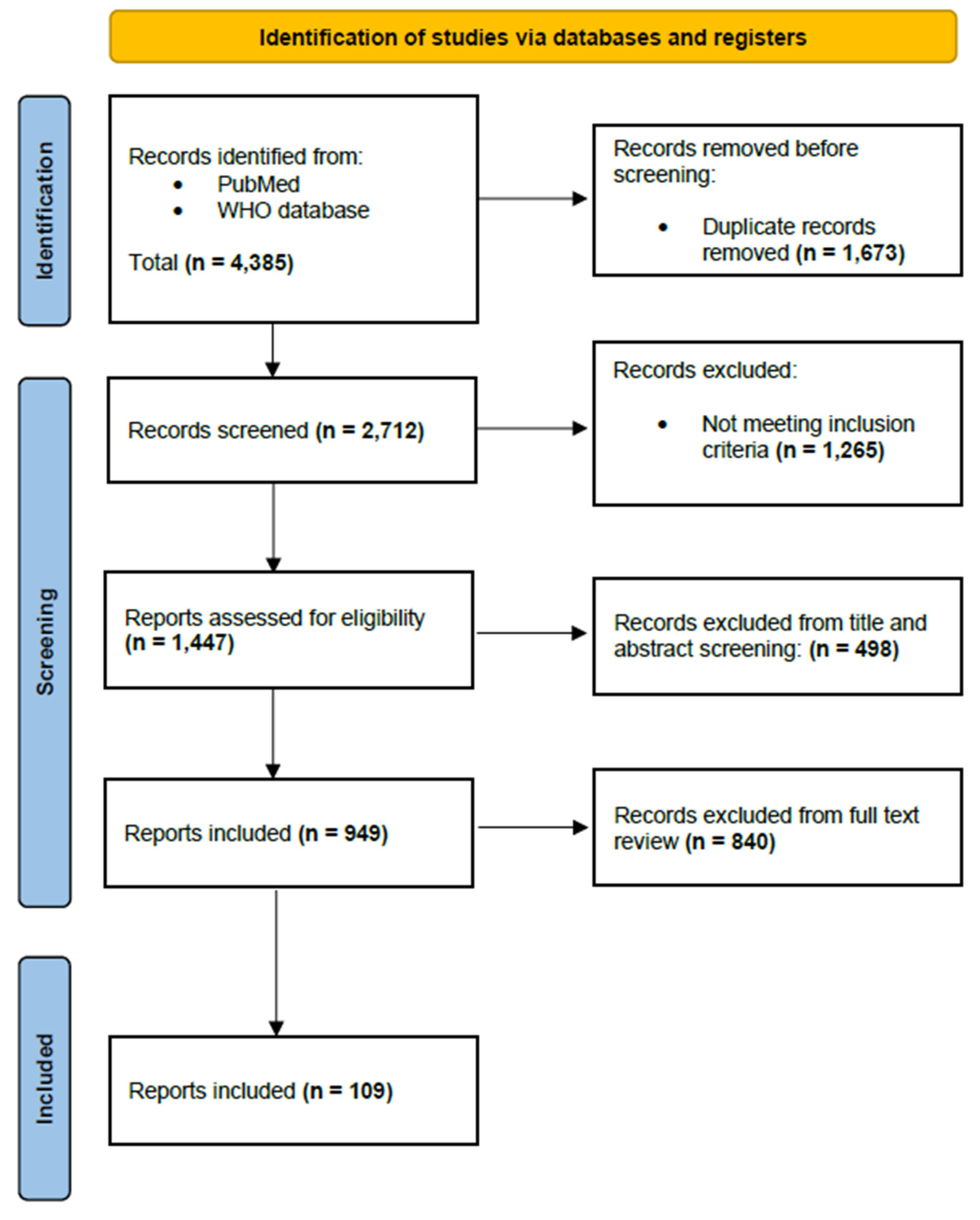
2.1. Study Design and Search Strategy
2.2. Eligibility Criteria
2.3. Search Methods for Identifying Data
2.4. Data Selection Process
2.5. Data Charting and Synthesis
3. Results
3.1. Article Characteristics
3.2. Relationship Between COVID-19 Vaccines and Autoimmune Disorders
3.3. Relapses or Flares in Individuals Already Experiencing Specific Autoimmune Disorders
3.3.1. Relapses or Flares in Graves’ Disease
3.3.2. Relapses or Flares in Hashimoto’s Thyroiditis
3.3.3. Relapses or Flares in Multiple Sclerosis
3.3.4. Relapses or Flares in Rheumatoid Arthritis
3.3.5. Relapses or Flares in Systemic Lupus Erythematosus
3.3.6. Relapses or Flares in Type 1 Diabetes Mellitus
3.4. Vaccination Associated with New Autoimmune Disease in Autoimmune Patients
3.5. Vaccination Associated with New Autoimmune Disorders in Persons Without Prior History of Autoimmunity
3.5.1. Graves’ Disease
3.5.2. Hashimoto’s Thyroiditis
3.5.3. Multiple Sclerosis
3.5.4. Rheumatoid Arthritis
3.5.5. Systemic Lupus Erythematosus
3.5.6. Type 1 Diabetes Mellitus
3.6. Adverse Events After COVID-19 Vaccination
3.7. Efficacy of COVID-19 Vaccination
3.8. Authors’ Perspectives on COVID-19 Vaccination for Patients with Autoimmune Disorders
4. Discussion
5. Conclusions
Author Contributions
Funding
Ethics
Informed Consent Statement
Conflicts of Interests
References
- Al-Midfai, Y. , Kujundzic, W., Uppal, S., Oakes, D., & Giezy, S. (2022). Acute Multiple Sclerosis Exacerbation After Vaccination With the Johnson & Johnson COVID-19 Vaccine: Novel Presentation and First Documented Case Report. Cureus. [CrossRef]
- Arksey, H., & O’Malley, L. (2005). Scoping studies: Towards a methodological framework. International Journal of Social Research Methodology, 8(1), 19–32. [CrossRef]
- Assawasaksakul, T., Lertussavavivat, T., Sathitratanacheewin, S., Oudomying, N., Vichaiwattana, P., Wanlapakorn, N., Poovorawan, Y., Avihingsanon, Y., Assawasaksakul, N., & Kittanamongkolchai, W. (2022). Comparison of immunogenicity and safety of inactivated, adenovirus-vectored and heterologous adenovirus-vectored/mRNA vaccines in patients with systemic lupus erythematosus and rheumatoid arthritis: A prospective cohort study (p. 2022.04.22.22274158). medRxiv. [CrossRef]
- Aydoğan, B. İ., Ünlütürk, U., & Cesur, M. (2022). Type 1 diabetes mellitus following SARS-CoV-2 mRNA vaccination. Endocrine, 78(1), 42–46. [CrossRef]
- Barbhaiya, M., Levine, J. M., Siegel, C. H., Bykerk, V. P., Jannat-Khah, D., & Mandl, L. A. (2022). Adverse events and disease flares after SARS-CoV-2 vaccination in patients with systemic lupus erythematosus. Clinical Rheumatology, 41(5), 1619–1622. [CrossRef]
- Bellinvia, A., Aprea, M. G., Portaccio, E., Pastò, L., Razzolini, L., Fonderico, M., Addazio, I., Betti, M., & Amato, M. P. (2022). Hypogammaglobulinemia is associated with reduced antibody response after anti-SARS-CoV-2 vaccination in MS patients treated with antiCD20 therapies. Neurological Sciences, 43(10), 5783–5794. [CrossRef]
- Bleve, E., Venditti, V., Lenzi, A., Morano, S., & Filardi, T. (2022). COVID-19 vaccine and autoimmune diabetes in adults: Report of two cases. Journal of Endocrinological Investigation, 45(6), 1269–1270. [CrossRef]
- Brès, F., Joyeux, M.-A., Delemer, B., Vitellius, G., & Barraud, S. (2022). Three cases of thyroiditis after COVID-19 RNA-vaccine. Annales d’Endocrinologie, 83(4), 262–264. [CrossRef]
- Briggs, F. B. S., Mateen, F. J., Schmidt, H., Currie, K. M., Siefers, H. M., Crouthamel, S., Bebo, B. F., Fiol, J., Racke, M. K., O’Connor, K. C., Kolaczkowski, L. G., Klein, P., Loud, S., & McBurney, R. N. (2022). COVID-19 Vaccination Reactogenicity in Persons With Multiple Sclerosis. Neurology Neuroimmunology & Neuroinflammation, 9(1), e1104. [CrossRef]
- Chaudhary, S., Dogra, V., & Walia, R. (2022). Four cases of Graves’ disease following viral vector severe acute respiratory syndrome corona virus-2 (SARS-CoV-2) vaccine. Endocrine Journal, 69(12), 1431–1435. [CrossRef]
- Chaufan, C., Manwell, L., Heredia, C., McDonald, J., Chaufan, C., Manwell, L., Heredia, C., & McDonald, J. (2023). COVID-19 vaccines and autoimmune disorders: A scoping review protocol. AIMS Medical Science, 10(4), Article medsci-10-04-025. [CrossRef]
- Chee, Y. J., Liew, H., Hoi, W. H., Lee, Y., Lim, B., Chin, H. X., Lai, R. T. R., Koh, Y., Tham, M., Seow, C. J., Quek, Z. H., Chen, A. W., Quek, T. P. L., Tan, A. W. K., & Dalan, R. (2022). SARS-CoV-2 mRNA Vaccination and Graves’ Disease: A Report of 12 Cases and Review of the Literature. The Journal of Clinical Endocrinology & Metabolism, 107(6), e2324–e2330. [CrossRef]
- Chen, Y., Xu, Z., Wang, P., Li, X., Shuai, Z., Ye, D., & Pan, H. (2022). New-onset autoimmune phenomena post-COVID-19 vaccination. Immunology, 165(4), 386–401. [CrossRef]
- Cohen, A. D., & Shoenfeld, Y. (1996). Vaccine-induced autoimmunity. Journal of Autoimmunity, 9(6), 699–703. [CrossRef]
- Cooper, G. S., Bynum, M. L. K., & Somers, E. C. (2009). Recent insights in the epidemiology of autoimmune diseases: Improved prevalence estimates and understanding of clustering of diseases. Journal of Autoimmunity, 33(3–4), 197–207. [CrossRef]
- Cooper, G. S., & Stroehla, B. C. (2003). The epidemiology of autoimmune diseases. Autoimmunity Reviews, 2(3), 119–125. [CrossRef]
- Coyle, P. K., Gocke, A., Vignos, M., & Newsome, S. D. (2021). Vaccine Considerations for Multiple Sclerosis in the COVID-19 Era. Advances in Therapy, 38(7), 3550–3588. [CrossRef]
- di Filippo, L., Castellino, L., Allora, A., Frara, S., Lanzi, R., Perticone, F., Valsecchi, F., Vassallo, A., Giubbini, R., Rosen, C. J., & Giustina, A. (2023). Distinct Clinical Features of Post-COVID-19 Vaccination Early-onset Graves’ Disease. The Journal of Clinical Endocrinology & Metabolism, 108(1), 107–113. [CrossRef]
- Dreyer-Alster, S., Menascu, S., Mandel, M., Shirbint, E., Magalashvili, D., Dolev, M., Flechter, S., Givon, U., Guber, D., Stern, Y., Miron, S., Polliack, M., Falb, R., Sonis, P., Gurevich, M., & Achiron, A. (2022). COVID-19 vaccination in patients with multiple sclerosis: Safety and humoral efficacy of the third booster dose. Journal of the Neurological Sciences, 434, 120155. [CrossRef]
- Etemadifar, M., Nouri, H., Salari, M., & Sedaghat, N. (2022). Detection of anti-NMDA receptor antibodies following BBIBP-CorV COVID-19 vaccination in a rituximab-treated person with multiple sclerosis presenting with manifestations of an acute relapse. Human Vaccines & Immunotherapeutics, 18(1), 2033540. [CrossRef]
- Faruk, T. Ö., Koseoglu, M., & Rabia, K. E. (2022). Varicella zoster virus infection due to Pfizer-BioNTech mRNA COVID-19 vaccine and sinovac vaccine in two relapsing-remitting multiple sclerosis patients during fingolimod therapy. Neuroimmunology Reports, 2, 100078. [CrossRef]
- Fitzpatrick, M. C., Moghadas, S. M., Pandey, A., & Galvani, A. P. (2022, December 13). Two Years of U.S. COVID-19 Vaccines Have Prevented Millions of Hospitalizations and Deaths. The Commonwealth Fund. [CrossRef]
- Flannery, P., Yang, I., Keyvani, M., & Sakoulas, G. (2021). Acute Psychosis Due to Anti-N-Methyl D-Aspartate Receptor Encephalitis Following COVID-19 Vaccination: A Case Report. Frontiers in Neurology, 12, 764197. [CrossRef]
- Frasca, L., Ocone, G., & Palazzo, R. (2023). Safety of COVID-19 Vaccines in Patients with Autoimmune Diseases, in Patients with Cardiac Issues, and in the Healthy Population. Pathogens, 12(2), 233. [CrossRef]
- Gad, A. H. E., Ahmed, S. M., Garadah, M. Y. A., & Dahshan, A. (2022). Multiple sclerosis patients’ response to COVID-19 pandemic and vaccination in Egypt. The Egyptian Journal of Neurology, Psychiatry and Neurosurgery, 58(1), 131. [CrossRef]
- Galanis, P., Vraka, I., Katsiroumpa, A., Siskou, O., Konstantakopoulou, O., Katsoulas, T., Mariolis-Sapsakos, T., & Kaitelidou, D. (2022). First COVID-19 Booster Dose in the General Population: A Systematic Review and Meta-Analysis of Willingness and Its Predictors. Vaccines, 10(7), 1097. [CrossRef]
- Gamonal, S. B. L., Marques, N. C. V., Pereira, H. M. B., & Gamonal, A. C. C. (2022). New-onset systemic lupus erythematosus after ChAdOX1 nCoV-19 and alopecia areata after BNT162b2 vaccination against SARS-CoV-2. Dermatologic Therapy, 35(9), e15677. [CrossRef]
- Ganakumar, V., Jethwani, P., Roy, A., Shukla, R., Mittal, M., & Garg, M. K. (2022). Diabetic ketoacidosis (DKA) in type 1 diabetes mellitus (T1DM) temporally related to COVID-19 vaccination. Diabetes & Metabolic Syndrome: Clinical Research & Reviews, 16(1), 102371. [CrossRef]
- Ghadiri, F., Sahraian, M. A., Azimi, A., & Moghadasi, A. N. (2022). The study of COVID-19 infection following vaccination in patients with multiple sclerosis. Multiple Sclerosis and Related Disorders, 57. [CrossRef]
- Giossi, R., Consonni, A., Clerici, V. T., Zito, A., Rigoni, E., Antozzi, C., Brambilla, L., Crisafulli, S. G., Bellino, A., Frangiamore, R., Bonanno, S., Vanoli, F., Ciusani, E., Corsini, E., Andreetta, F., Baggi, F., Tramacere, I., Mantegazza, R., Conte, A., … Confalonieri, P. (2022). Anti-Spike IgG in multiple sclerosis patients after BNT162b2 vaccine: An exploratory case-control study in Italy. Multiple Sclerosis and Related Disorders, 58. [CrossRef]
- Guimarães, L. E., Baker, B., Perricone, C., & Shoenfeld, Y. (2015). Vaccines, adjuvants and autoimmunity. Pharmacological Research, 100, 190–209. [CrossRef]
- Guo, M., Liu, X., Chen, X., & Li, Q. (2023). Insights into new-onset autoimmune diseases after COVID-19 vaccination. Autoimmunity Reviews, 22(7), 103340. [CrossRef]
- Havla, J., Schultz, Y., Zimmermann, H., Hohlfeld, R., Danek, A., & Kümpfel, T. (2022). First manifestation of multiple sclerosis after immunization with the Pfizer-BioNTech COVID-19 vaccine. Journal of Neurology, 269(1), 55–58. [CrossRef]
- Hromić-Jahjefendić, A., Barh, D., Uversky, V., Aljabali, A. A., Tambuwala, M. M., Alzahrani, K. J., Alzahrani, F. M., Alshammeri, S., & Lundstrom, K. (2023). Can COVID-19 Vaccines Induce Premature Non-Communicable Diseases: Where Are We Heading to? Vaccines, 11(2), 208. [CrossRef]
- Jacobson, D. L., Gange, S. J., Rose, N. R., & Graham, N. M. (1997). Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clinical Immunology and Immunopathology, 84(3), 223–243. [CrossRef]
- Jara, L. J., Vera-Lastra, O., Mahroum, N., Pineda, C., & Shoenfeld, Y. (2022). Autoimmune post-COVID vaccine syndromes: Does the spectrum of autoimmune/inflammatory syndrome expand? Clinical Rheumatology, 41(5), 1603–1609. [CrossRef]
- Kataria, S., Rogers, S., Bilal, U., Baktashi, H., & Singh, R. (2022). Multiple Sclerosis Relapse Following COVID-19 Vaccination: A Case Report and Literature Review. Cureus. [CrossRef]
- Kim, H. J., Jung, M., Lim, B. J., & Han, S. H. (2022). New-onset class III lupus nephritis with multi-organ involvement after COVID-19 vaccination. Kidney International, 101(4), 826–828. [CrossRef]
- Kirwan, P. D., Charlett, A., Birrell, P., Elgohari, S., Hope, R., Mandal, S., De Angelis, D., & Presanis, A. M. (2022). Trends in COVID-19 hospital outcomes in England before and after vaccine introduction, a cohort study. Nature Communications, 13(1), 4834. [CrossRef]
- Kreuter, A., Licciardi-Fernandez, M. J., Burmann, S. -N., Burkert, B., Oellig, F., & Michalowitz, A. -L. (2022). Induction and exacerbation of subacute cutaneous lupus erythematosus following mRNA-based or adenoviral vector-based SARS-CoV-2 vaccination. Clinical and Experimental Dermatology, 47(1), 161–163. [CrossRef]
- Lemoine, C., Padilla, C., Krampe, N., Doerfler, S., Morgenlander, A., Thiel, B., & Aggarwal, R. (2022). Systemic lupus erythematous after Pfizer COVID-19 vaccine: A case report. Clinical Rheumatology, 41(5), 1597–1601. [CrossRef]
- Levac, D., Colquhoun, H., & O’Brien, K. K. (2010). Scoping studies: Advancing the methodology. Implementation Science: IS, 5, 69. [CrossRef]
- Lim, E., Kim, Y. H., Jeong, N.-Y., Kim, S.-H., Won, H., Bae, J.-S., Choi, N.-K., & COVID-19 Vaccine Safety Committee (CoVaSC). (2025). The association between acute transverse myelitis and COVID-19 vaccination in Korea: Self-controlled case series study. European Journal of Neurology, 32(1), e70020. [CrossRef]
- Lioulios, G., Tsouchnikas, I., Dimitriadis, C., Giamalis, P., Pella, E., Christodoulou, M., Stangou, M., & Papagianni, A. (2022). Two Cases of Autoimmune Thyroid Disorders after COVID Vaccination in Dialysis Patients. International Journal of Molecular Sciences, 23(19), Article 19. [CrossRef]
- Lohmann, L., Glaser, F., Möddel, G., Lünemann, J. D., Wiendl, H., & Klotz, L. (2022). Severe disease exacerbation after mRNA COVID-19 vaccination unmasks suspected multiple sclerosis as neuromyelitis optica spectrum disorder: A case report. BMC Neurology, 22(1), 185. [CrossRef]
- Mahroum, N., Lavine, N., Ohayon, A., Seida, R., Alwani, A., Alrais, M., Zoubi, M., & Bragazzi, N. L. (2022). COVID-19 Vaccination and the Rate of Immune and Autoimmune Adverse Events Following Immunization: Insights From a Narrative Literature Review. Frontiers in Immunology, 13, 872683. [CrossRef]
- Mahroum, N., & Shoenfeld, Y. (2022). COVID-19 vaccination can occasionally trigger autoimmune phenomena, probably via inducing age-associated B cells. International Journal of Rheumatic Diseases, 25(1), 5–6. [CrossRef]
- Manta, R., Martin, C., Muls, V., & Poppe, K. G. (2022). New-onset Graves’ disease following SARS-CoV-2 vaccination: A case report. [CrossRef]
- McDonald, I., Murray, S. M., Reynolds, C. J., Altmann, D. M., & Boyton, R. J. (2021). Comparative systematic review and meta-analysis of reactogenicity, immunogenicity and efficacy of vaccines against SARS-CoV-2. Npj Vaccines, 6(1), 1–14. [CrossRef]
- Mele, A. A., Ogbuagu, H., Parag, S., Pierce, B., Mele, A. A., Ogbuagu, H., Parag, S., & Pierce, B. H. (2022). A Case of Multiple Sclerosis Uncovered Following Moderna SARS-CoV-2 Vaccination. Cureus, 14. [CrossRef]
- Molina-Rios, S., Rojas-Martinez, R., Estévez-Ramirez, G. M., & Medina, Y. F. (2023). Systemic lupus erythematosus and antiphospholipid syndrome after COVID-19 vaccination. A case report. Modern Rheumatology Case Reports, 7(1), 43–46. [CrossRef]
- Munn, Z., Stern, C., Aromataris, E., Lockwood, C., & Jordan, Z. (2018). What kind of systematic review should I conduct? A proposed typology and guidance for systematic reviewers in the medical and health sciences. BMC Medical Research Methodology, 18(1), 5. [CrossRef]
- Nakamura, H., Nagasawa, Y., Kobayashi, H., Tsukamoto, M., Takayama, T., & Kitamura, N. (2022). Successful Treatment of SARS-CoV-2 Vaccination-related Activation of Rheumatoid Arthritis with Positive Findings for Epstein-Barr Virus. Internal Medicine, 61(13), 2073–2076. [CrossRef]
- Nelson, M. C., Rytting, H., Greenbaum, L. A., & Goldberg, B. (2022). Presentation of SLE after COVID vaccination in a pediatric patient. BMC Rheumatology, 6(1), 81. [CrossRef]
- Patrizio, A., Ferrari, S. M., Antonelli, A., & Fallahi, P. (2022). Worsening of Graves’ ophthalmopathy after SARS-CoV-2 mRNA vaccination. Autoimmunity Reviews, 21(7), 103096. [CrossRef]
- Polack, F. P., Thomas, S. J., Kitchin, N., Absalon, J., Gurtman, A., Lockhart, S., Perez, J. L., Marc, G. P., Moreira, E. D., Zerbini, C., Bailey, R., Swanson, K. A., Roychoudhury, S., Koury, K., Li, P., Kalina, W. V., Cooper, D., Frenck, R. W., Hammitt, L. L., … Gruber, W. C. (2020). Safety and Efficacy of the BNT162b2 mRNA Covid-19 Vaccine. New England Journal of Medicine, 383(27), 2603–2615. [CrossRef]
- Prof Peter Hotez MD PhD [@PeterHotez]. (2025, February 4). Here’s where the 3.2 million lives saved comes from https://t.co/A7Z1Vc2VL2 [Tweet]. Twitter. https://x.com/PeterHotez/status/1886573301977284968.
- Raviv, Y., Betesh-Abay, B., Valdman-Grinshpoun, Y., Boehm-Cohen, L., Kassirer, M., & Sagy, I. (2022). First Presentation of Systemic Lupus Erythematosus in a 24-Year-Old Male following mRNA COVID-19 Vaccine. Case Reports in Rheumatology, 2022(1), 9698138. [CrossRef]
- Ruggeri, R. M., Giovanellla, L., & Campennì, A. (2022). SARS-CoV-2 vaccine may trigger thyroid autoimmunity: Real-life experience and review of the literature. Journal of Endocrinological Investigation, 45(12), 2283–2289. [CrossRef]
- Sagy, I., Zeller, L., Raviv, Y., Porges, T., Bieber, A., & Abu-Shakra, M. (2022). New-onset systemic lupus erythematosus following BNT162b2 mRNA COVID-19 vaccine: A case series and literature review. Rheumatology International, 42(12), 2261–2266. [CrossRef]
- Sakurai, K., Narita, D., Saito, N., Ueno, T., Sato, R., Niitsuma, S., Takahashi, K., & Arihara, Z. (2022). Type 1 diabetes mellitus following COVID-19 RNA-based vaccine. Journal of Diabetes Investigation, 13(7), 1290–1292. [CrossRef]
- Sasaki, H., Itoh, A., Watanabe, Y., Nakajima, Y., Saisho, Y., Irie, J., Meguro, S., & Itoh, H. (2022). Newly developed type 1 diabetes after coronavirus disease 2019 vaccination: A case report. Journal of Diabetes Investigation, 13(6), 1105–1108. [CrossRef]
- Sasaki, K., Morioka, T., Okada, N., Natsuki, Y., Kakutani, Y., Ochi, A., Yamazaki, Y., Shoji, T., Ohmura, T., & Emoto, M. (2022). New-onset fulminant type 1 diabetes after severe acute respiratory syndrome coronavirus 2 vaccination: A case report. Journal of Diabetes Investigation, 13(7), 1286–1289. [CrossRef]
- Sato, T., Kodama, S., Kaneko, K., Imai, J., & Katagiri, H. (2022). Type 1 Diabetes Mellitus Associated with Nivolumab after Second SARS-CoV-2 Vaccination, Japan. Emerging Infectious Diseases, 28(7), 1518–1520. [CrossRef]
- Scaramuzza, A. E., Cherubini, V., Schiaffini, R., Rabbone, I., The Diabetes Study Group of the Italian Society for Pediatric Endocrinology and Diabetes, Gallo, F., Fichera, G., Arnaldi, C., Bonfanti, R., Lombardo, F., De Marco, R., Pascarella, F., Tornese, G., Bobbio, A., Suprani, T., Minuto, N., Franceschi, R., Piccinno, E., Mozzillo, E., … Cavalli, C. (2022). A nationwide survey of Italian pediatric diabetologists about COVID-19 vaccination in children and adolescents with type 1 diabetes. Acta Diabetologica, 59(8), 1109–1111. [CrossRef]
- Shea, B. J., Reeves, B. C., Wells, G., Thuku, M., Hamel, C., Moran, J., Moher, D., Tugwell, P., Welch, V., Kristjansson, E., & Henry, D. A. (2017). AMSTAR 2: A critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ, 358, j400. [CrossRef]
- Shih, S.-R., & Wang, C.-Y. (2022). SARS-CoV-2 vaccination related hyperthyroidism of Graves’ disease. Journal of the Formosan Medical Association, 121(9), 1881–1882. [CrossRef]
- Shoenfeld, Y. (2009). Infections, vaccines and autoimmunity. Lupus, 18(13), 1127–1128. [CrossRef]
- Shoenfeld, Y., & Agmon-Levin, N. (2011). ’ASIA’—Autoimmune/inflammatory syndrome induced by adjuvants. Journal of Autoimmunity, 36(1), 4–8. [CrossRef]
- Shoenfeld, Y., & Aron-Maor, A. (2000). Vaccination and Autoimmunity—‘vaccinosis’: A Dangerous Liaison? Journal of Autoimmunity, 14(1), 1–10. [CrossRef]
- Singh, R., Kaur, U., Singh, A., & Chakrabarti, S. S. (2022). New onset rheumatoid arthritis with refractory hyper-eosinophilia associated with inactivated COVID-19 vaccin. [CrossRef]
- So, H., Li, T., Chan, V., Tam, L.-S., & Chan, P. K. (2022). Immunogenicity and safety of inactivated and mRNA COVID-19 vaccines in patients with systemic lupus erythematosus. Therapeutic Advances in Musculoskeletal Disease, 14, 1759720X221089586. [CrossRef]
- Sogbe, M., Blanco-Di Matteo, A., Di Frisco, I. M., Bastidas, J. F., Salterain, N., & Gavira, J. J. (2023). Systemic lupus erythematosus myocarditis after COVID-19 vaccination. Reumatología Clínica, 19(2), 114–116. [CrossRef]
- Sugimoto, T., Yorishima, A., Oka, N., Masuda, S., Yoshida, Y., & Hirata, S. (2022). Exacerbation of systemic lupus erythematosus after receiving mRNA-1273-based coronavirus disease 2019 vaccine. The Journal of Dermatology, 49(6), e199–e200. [CrossRef]
- Taieb, A., Sawsen, N., Asma, B. A., Ghada, S., Hamza, E., Yosra, H., Amel, M., Molka, C., Maha, K., & Koussay, A. (2022). A rare case of grave’s disease after SARS-CoV-2 vaccine: Is it an adjuvant effect? European Review for Medical and Pharmacological Sciences, 26(7), 2627–2630. [CrossRef]
- Tang, Q., Li, F., Tian, J., Kang, J., & He, J. (2022). Attitudes towards and safety of the SARS-CoV-2 inactivated vaccines in 188 patients with systemic lupus erythematosus: A post-vaccination cross-sectional survey. Clinical and Experimental Medicine, 23(2), 457–463. [CrossRef]
- Tang, X., He, B., Liu, Z., Zhou, Z., & Li, X. (2022). Fulminant type 1 diabetes after COVID-19 vaccination. Diabetes & Metabolism, 48(2), 101324. [CrossRef]
- Toljan, K., Amin, M., Kunchok, A., & Ontaneda, D. (2022). New diagnosis of multiple sclerosis in the setting of mRNA COVID-19 vaccine exposure. Journal of Neuroimmunology, 362, 577785. [CrossRef]
- Tricco, A. C., Lillie, E., Zarin, W., O’Brien, K. K., Colquhoun, H., Levac, D., Moher, D., Peters, M. D. J., Horsley, T., Weeks, L., Hempel, S., Akl, E. A., Chang, C., McGowan, J., Stewart, L., Hartling, L., Aldcroft, A., Wilson, M. G., Garritty, C., … Straus, S. E. (2018). PRISMA Extension for Scoping Reviews (PRISMA-ScR): Checklist and Explanation. Annals of Internal Medicine, 169(7), 467–473. [CrossRef]
- Trougakos, I. P., Terpos, E., Alexopoulos, H., Politou, M., Paraskevis, D., Scorilas, A., Kastritis, E., Andreakos, E., & Dimopoulos, M. A. (2022). Adverse effects of COVID-19 mRNA vaccines: The spike hypothesis. Trends in Molecular Medicine, 28(7), 542–554. [CrossRef]
- Vera-Lastra, O., Ordinola Navarro, A., Cruz Domiguez, M. P., Medina, G., Sánchez Valadez, T. I., & Jara, L. J. (2021). Two cases of Graves’ disease following SARS-CoV-2 vaccination: An autoimmune/inflammatory syndrome induced by adjuvants. Thyroid®, 31(9), 1436–1439. [CrossRef]
- Vojdani, A., & Kharrazian, D. (2020). Potential antigenic cross-reactivity between SARS-CoV-2 and human tissue with a possible link to an increase in autoimmune diseases. Clinical Immunology (Orlando, Fla.), 217, 108480. [CrossRef]
- Wang, H., Sun, Y., & Lan, C. E. (2022a). Systemic lupus erythematosus with acrocyanosis after AstraZeneca COVID-19 vaccination. The Kaohsiung Journal of Medical Sciences, 38(12), 1230–1231. [CrossRef]
- Wang, H., Sun, Y., & Lan, C. E. (2022b). Systemic lupus erythematosus with acrocyanosis after AstraZeneca COVID-19 vaccination. The Kaohsiung Journal of Medical Sciences, 38(12), 1230–1231. [CrossRef]
- Watad, A., Sharif, K., & Shoenfeld, Y. (2017). The ASIA syndrome: Basic concepts. Mediterranean Journal of Rheumatology, 28(2), 64–69. [CrossRef]
- Watanabe, T., Minaga, K., Hara, A., Yoshikawa, T., Kamata, K., & Kudo, M. (2022). Case Report: New-Onset Rheumatoid Arthritis Following COVID-19 Vaccination. Frontiers in Immunology, 13. [CrossRef]
- Widhani, A., Hasibuan, A. S., Rismawati, R., Maria, S., Koesnoe, S., Hermanadi, M. I., Ophinni, Y., Yamada, C., Harimurti, K., Sari, A. N. L., Yunihastuti, E., & Djauzi, S. (2023). Efficacy, Immunogenicity, and Safety of COVID-19 Vaccines in Patients with Autoimmune Diseases: A Systematic Review and Meta-Analysis. Vaccines, 11(9), 1456. [CrossRef]
- Yakou, F., Saburi, M., Hirose, A., Akaoka, H., Hirota, Y., Kobayashi, T., Awane, N., Asahi, N., Amagawa, T., Ozawa, S., Ohno, A., & Matsushita, T. (2022). A Case Series of Ketoacidosis After Coronavirus Disease 2019 Vaccination in Patients With Type 1 Diabetes. Frontiers in Endocrinology, 13, 840580. [CrossRef]
- Yano, M., Morioka, T., Natsuki, Y., Sasaki, K., Kakutani, Y., Ochi, A., Yamazaki, Y., Shoji, T., & Emoto, M. (2022). New-onset Type 1 Diabetes after COVID-19 mRNA Vaccination. Internal Medicine, 61(8), 1197–1200. [CrossRef]
- Yonezawa, H., Ohmura, S., Ohkubo, Y., & Miyamoto, T. (2022). New-onset Seropositive Rheumatoid Arthritis Following COVID-19 Vaccination in a Patient with Seronegative Status. Internal Medicine, 61(22), 3449–3452. [CrossRef]
- Zhao, T., Shen, J., Zhu, Y., Tian, X., Wen, G., Wei, Y., Xu, B., Fu, C., Xie, Z., Xi, Y., Li, Z., Peng, J., Wu, Y., Tang, X., Wan, C., Pan, L., Li, Z., & Qin, D. (2022). Immunogenicity of Inactivated SARS-CoV-2 Vaccines in Patients With Rheumatoid Arthritis: A Case Series. Frontiers in Public Health, 10, 875558. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



