Submitted:
09 June 2025
Posted:
09 June 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Presentation and Pathophysiology of NF1
3. MRI in NF1
4. Management
5. Targeted Genetic Treatment
6. Role of MEK Inhibitors in NF1 and Pediatric Populations
7. MEK Inhibitors Approved for Clinical Use
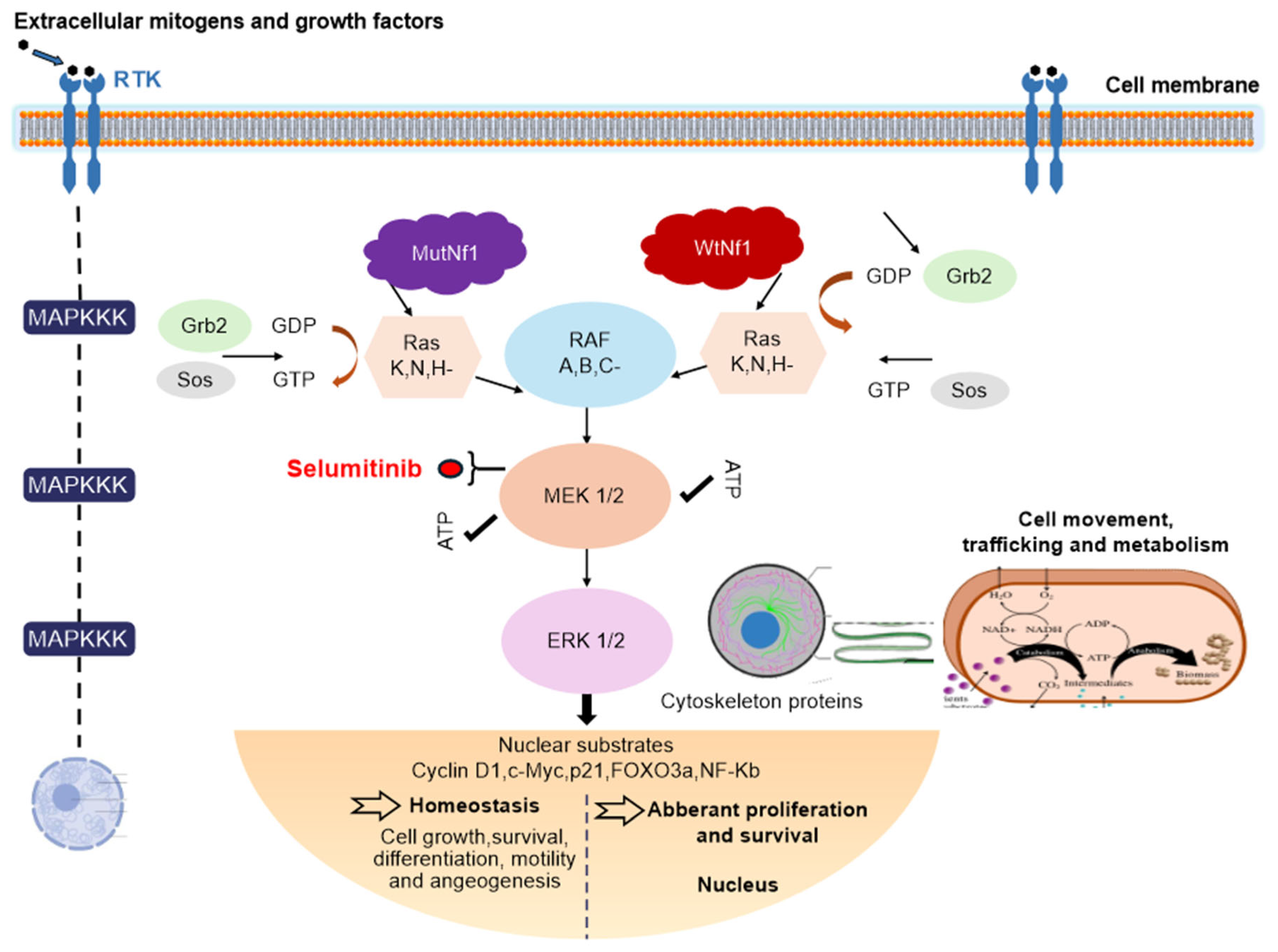
8. Pharmacological Overview of Selumetinib
9. Patents
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abdel-Aziz NN, El-Kamah GY, Khairat RA, Mohamed HR, Gad YZ, El-Ghor AM, et al. Mutational spectrum of NF1 gene in 24 unrelated Egyptian families with neurofibromatosis type 1. Mol Genet Genomic Med. (2021) 9:e1631. [CrossRef]
- Bergqvist C, Servy A, Valeyrie-Allanore L, Ferkal S, Combemale P, Wolkenstein P, et al. Neurofibromatosis 1 French national guidelines based on an extensive literature review since 1966. Orphanet J Rare Dis. (2020) 15:37. [CrossRef]
- Buchholzer, S., Verdeja, R., & Lombardi, T. (2021). Type I neurofibromatosis: Case report and review of the literature focused on oral and cutaneous lesions. Dermatopathology, 8(1), 17-24. [CrossRef]
- Kunc, V., Venkatramani, H., & Sabapathy, S. R. (2019). Neurofibromatosis 1 was diagnosed in mother only after a follow-up of her daughter. Indian Journal of Plastic Surgery, 52(2), 260. [CrossRef]
- Kallionpää RA, Uusitalo E, Leppävirta J, Pöyhönen M, Peltonen S, Peltonen J. Prevalence of neurofibromatosis type 1 in the Finnish population. Genet Med. (2018) 20:1082–6. [CrossRef]
- Campagne, O., Yeo, K. K., Fangusaro, J., & Stewart, C. F. (2021). Clinical pharmacokinetics and pharmacodynamics of selumetinib. Clinical Pharmacokinetics, 60(3), 283–303. [CrossRef]
- Ly, K. Ina, Jaishri O. Blakeley. The diagnosis and management of neurofibromatosis type 1. Medical Clinics 103.6 (2019): 1035-1054. [CrossRef]
- Di Pietro S, Reali L, Tona E, Belfiore G, Praticò AD, Ruggieri M, David E, Foti PV, Santonocito OG, Basile A, Palmucci S. "Magnetic Resonance Imaging of Central Nervous System Manifestations of Type 1 Neurofibromatosis: Pictorial Review and Retrospective Study of Their Frequency in a Cohort of Patients." Journal of Clinical Medicine 13.11 (2024): 3311. [CrossRef]
- Wilson BN, John AM, Handler MZ, Schwartz RA. Neurofibromatosis type 1: New developments in genetics and treatment. Journal of the American Academy of Dermatology 84.6 (2021): 1667-1676. [CrossRef]
- Ahlawat, S.; Blakeley, J.O.; Langmead, S.; Belzberg, A.J.; Fayad, L.M. Current status and recommendations for imaging in neurofibromatosis type 1, neurofibromatosis type 2, and schwannomatosis. Skelet. Radiol. 2020, 49, 199–219. [CrossRef]
- Thakur U, Ramachandran S, Mazal AT, Cheng J, Le L, Chhabra A. Multiparametric whole-body MRI of patients with neurofibromatosis type I: spectrum of imaging findings. Skeletal radiology (2024): 1-16. [CrossRef]
- Chiang YZ, Al-Niaimi F, Ferguson J, August PJ, Madan V. Carbon dioxide laser treatment of cutaneous neurofibromas. Dermatol Ther (Heidelb) 2012: 2(1):7. [CrossRef]
- Weiss B, Widemann BC, Wolters P, Dombi E, Vinks A, Cantor A et al. Sirolimus for progressive neurofibromatosis type 1-associated plexiform neurofibromas: a neurofibromatosis Clinical Trials Consortium phase II study. Neuro Oncol 2015; 17:596-603. [CrossRef]
- Allouche J, Bellon N, Saidani M, Stanchina-Chatrousse L, Masson Y, Patwardhan A et al. In vitro modeling of hyperpigmentation associated to neurofibromatosis type 1 using melanocytes derived from human embryonic stem cells. Proc Natl Acad Sci U S A 2015; 112:9034-9. [CrossRef]
- Nakayama J, Imafuku S, Mori T, Sato C. Narrowband ultraviolet B irradiation increases the serum level of vitamin D(3) in patients with neurofibromatosis 1. J Dermatol 2013;40:829-31. [CrossRef]
- Baumann D, Hagele T, Mochayedi J, et al. Proimmunogenic impact of MEK inhibition synergizes with agonist anti-CD40 immunostimulatory antibodies in tumor therapy. Nat Commun. 2020;11(1):2176. [CrossRef]
- Erkes DA, Cai W, Sanchez IM, et al. Mutant BRAF and MEK inhibitors regulate the tumor immune microenvironment via pyroptosis. Cancer Discov. 2020;10(2):254–269. [CrossRef]
- Poon E, Mullins S, Watkins A, et al. The MEK inhibitor selumetinib complements CTLA-4 blockade by reprogramming the tumor immune microenvironment. J Immunother Cancer. 2017;5(1):63. [CrossRef]
- Dombi E, Baldwin A, Marcus LJ, Fisher MJ, Weiss B, Kim A, Whitcomb P, Martin S, Aschbacher-Smith LE, Rizvi TA, et al. Activity of selumetinib in neurofibromatosis type 1-related plexiform neurofibromas. N Engl J Med. 2016;375(26):2550–60. [CrossRef]
- Gross AM, Wolters PL, Dombi E, Baldwin A, Whitcomb P, Fisher MJ, Weiss B, Kim A, Bornhorst M, Shah AC, Martin S, Roderick MC, Pichard DC, Carbonell A, Paul SM, Therrien J, Kapustina O, Heisey K, Clapp DW, Zhang C, Peer CJ, Figg WD, Smith M, Glod J, Blakeley JO, Steinberg SM, Venzon DJ, Doyle LA, Widemann BC. Selumetinib in children with inoperable plexiform neurofibromas. N Engl J Med. 2020;382(15):1430–42. [CrossRef]
- Gross AM, Glassberg B, Wolters PL, Dombi E, Baldwin A, Fisher MJ, Kim A, Bornhorst M, Weiss BD, Blakeley JO, Whitcomb P, Paul SM, Steinberg SM, Venzon DJ, Martin S, Carbonell A, Heisey K, Therrien J, Kapustina O, Dufek A, Derdak J, Smith MA, Widemann BC. Selumetinib in children with neurofibromatosis type 1 and asymptomatic inoperable plexiform neurofibroma at risk for developing tumor-related morbidity. Neuro Oncol. 2022;24(11):1978–88. [CrossRef]
- Koselugo (selumetinib) Japanese prescribing information; 2022.
- Gross AM, Wolters PL, Dombi E, Baldwin A, Whitcomb P, Fisher MJ, Weiss B, Kim A, Bornhorst M, Shah AC, Martin S, Roderick MC, Pichard DC, Carbonell A, Paul SM, Therrien J, Kapustina O, Heisey K, Clapp DW, Zhang C, Peer CJ, Figg WD, Smith M, Glod J, Blakeley JO, Steinberg SM, Venzon DJ, Doyle LA, Widemann BC. Selumetinib in children with inoperable plexiform neurofibromas. N Engl J Med. 2020 Apr 9;382(15):1430-1442. [CrossRef]
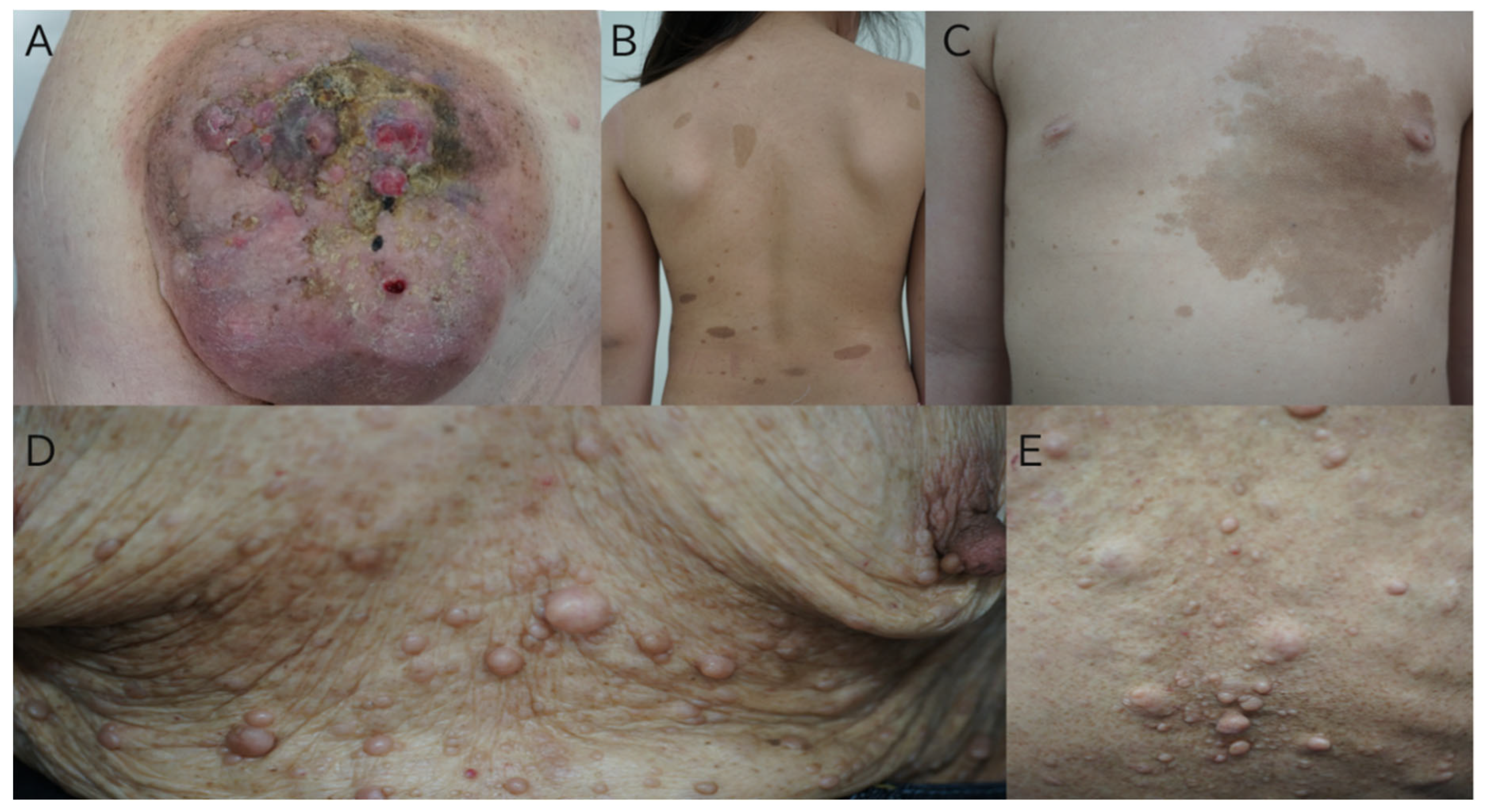
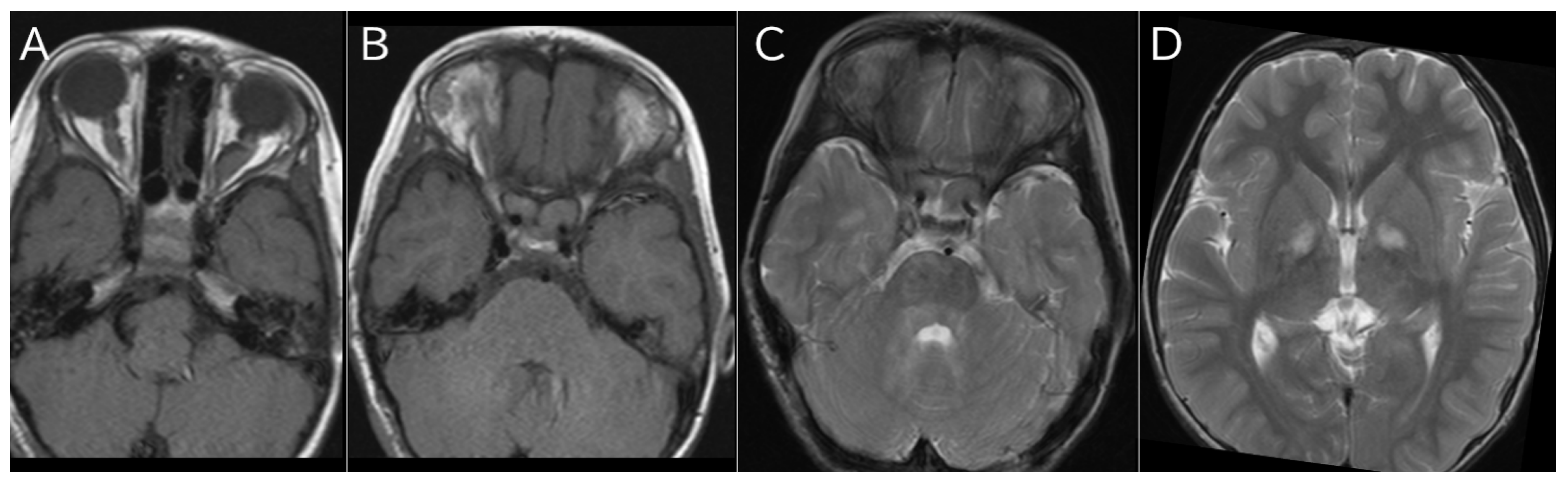
| Diagnostic Criteria | Clinical Context |
|---|---|
| Two or more of the following: | |
| 1. Café-au-lait macules (CALMs) | Presence of six or more CALMs, at least 5 mm in diameter in prepubertal individuals or 15 mm in post pubertal individuals, suggests NF1. |
| Having only two or three CALMs is generally normal. | |
| CALMs usually appear in infancy and have smooth, well-defined edges. | |
| 2. Neurofibromas | Diagnosis includes two or more neurofibromas of any kind or one plexiform neurofibroma. |
| Dermal and subcutaneous neurofibromas typically develop later in childhood. | |
| Plexiform neurofibromas may caus changes in skin texture or color. | |
| 3. Axillary or inguinal freckling (Crowe sign) | These freckles typically develop after age 5. |
| Freckles in non-sun-exposed areas are unusual in individuals without NF1 | |
| 4. Optic glioma (OPG) | Often invisible without specialized eye exams but may be present in infancy. |
| Early detection is vital to protect vision. | |
| 5. Iris hamartomas (Lisch nodules) | Age-dependent, these nodules are uncommon in infants and toddlers but appear in most teenagers. |
| They require a slit-lamp examination for detection and do not affect vision. | |
| 6. Osseous abnormalities | Typical findings include tibial dysplasia, such as bowing, cortical thickening, or pseudoarthrosis. |
| Infants or toddlers with bowing of the tibia should undergo radiography and be referred to an orthopedic specialist. | |
| 7. Family history | A first-degree relative (parent, sibling, or child) with NF1 based on diagnostic criteria confirms familial inheritance. |
| NF1 is fully penetrant, meaning symptoms will always be present, even if mild, and it does not skip generations. |
| Tumor Type | Description | Key Features |
|---|---|---|
| Plexiform Neurofibromas (PN) | Occur in ~60% of NF1 patients; often congenital and arise from neural crest cells. Grow concentrically within large nerves, causing potential disfigurement and nerve dysfunction. | Unlimited growth; can lead to pain, neuropathy, or ambulation interference. Risk of malignant transformation (~15%) to Malignant Peripheral Nerve Sheath Tumors (MPNST). |
| Malignant Peripheral Nerve Sheath Tumors (MPNST) | Aggressive form arising from pre-existing PN, primarily affecting individuals aged 20–40 years. Poor prognosis with a high risk of recurrence and metastasis. | Radio- and chemo-insensitive; gross total resection required for potential cure. 5-year survival ~50%; median survival for inoperable/metastatic cases reduced to 11–12 months. |
| Cutaneous Neurofibromas (CN) | Skin tumors originating from cutaneous nerves or hair bulge cells. Form small, rubbery nodules, usually amelanotic or erythematous. Commonly appear in adolescence. | Limited growth potential; minimal risk of malignant transformation. Removal options include surgical resection, CO2 laser ablation, and advanced photocoagulation methods. |
| Subcutaneous Neurofibromas (SCN) | Exhibit indistinct borders and appear violaceous. Typically develop during late childhood or adolescence, mostly on the trunk, arms, and face. | Cause discomfort, itching, and visible disfigurement; rarely grow larger than 2 cm. Physical removal or destruction is the only effective treatment. |
| Gliomas | Predominantly Optic Pathway Gliomas (OPG), with a smaller subset of high-grade gliomas. Often occur in NF1 patients within the first 5 years of life. | OPG is benign in most cases (~75–80% 5-year survival), but can lead to vision loss or endocrine issues. High-grade gliomas have a 50-fold increased risk in NF1. |
| Optic Pathway Gliomas (OPG) | WHO grade I astrocytic tumors often associated with NF1. Frequently observed in 15–20% of NF1 patients, manifesting early in life and sometimes spontaneously regressing. | Vision impairment and/or endocrine disturbances may occur. Treatment options include chemotherapy (e.g., carboplatin and vincristine) or MEK inhibitors; surgery is rarely used. |
| Condition | % of Patients Affected | MRI Characteristics |
|---|---|---|
| Brain tumor | Varies by tumor site and grade | In pediatric patients, low-grade gliomas are common, primarily affecting the cerebellum and brainstem; in adults, high-grade gliomas are more frequent, mainly in the cerebral hemispheres. On T2-weighted scans: hyperintense areas; On T1-weighted scans: isointense or slightly hypointense, with enhancement following gadolinium. |
| Brain abnormalities (Unidentified Bright Objects; UBO or FASI) | 43-93% | Regions with increased signal on T2-weighted MRI, absent on T1-weighted scans, with no mass effect or enhancement; these anomalies are seldom found in individuals over 20 years old. The cerebellum, brainstem, and basal ganglia are frequently affected areas. |
| Plexiform neurofibromas | Up to 30% | Most frequently located in the craniomaxillofacial area. On T2-weighted scans: heterogeneous masses with high signal and a low signal center; On T1-weighted scans: slightly hyperintense compared to muscle tissue, with varying degrees of contrast enhancement. |
| Spinal Tumors (Peripheral Nerve Sheath Tumors and Intramedullary Tumors) | 40-96% | Primarily benign neurofibromas, while intramedullary tumors are uncommon. Neurofibromas show a high signal on T2-weighted scans with a central low signal target and low signal on T1-weighted images, displaying varied contrast enhancement. Indicators of malignant transformation in nerve sheath tumors include increasing size, tumor diameter >5 cm, unclear margins, absence of central low signal target on T2, and central necrosis. |
| Lesions in the Visual Pathway (Optic Pathway Gliomas) | 5-15% | Enlargement of optic nerves or optic chiasm (diameter over 3.9 mm); On T2-weighted scans: core with low signal surrounded by a higher intensity ring; On T1-weighted scans: isointense signal, showing enhancement after gadolinium administration. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




