Submitted:
05 June 2025
Posted:
06 June 2025
You are already at the latest version
Abstract
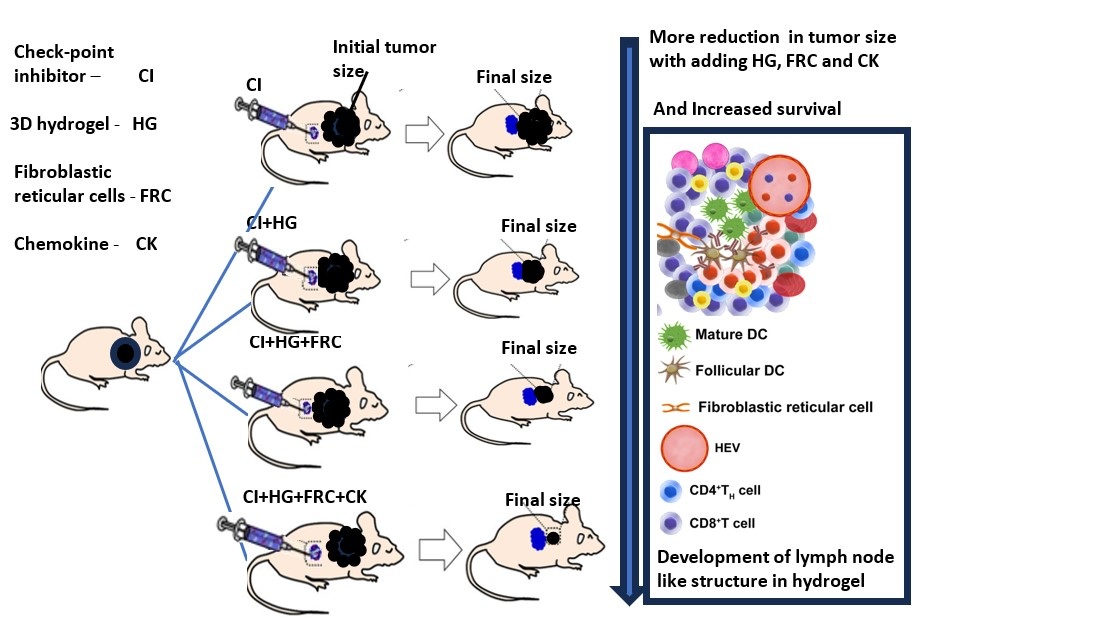
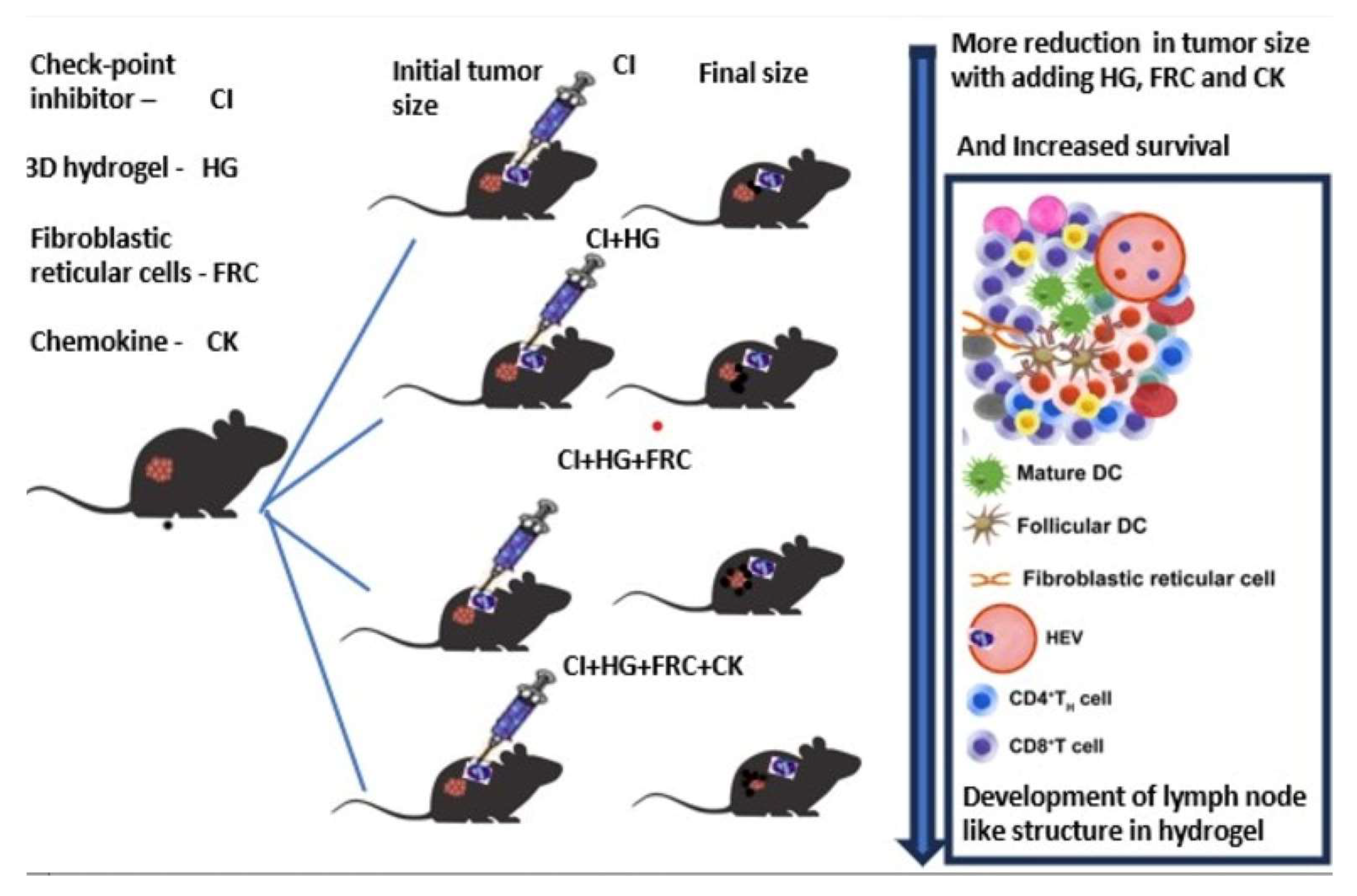
Keywords:
1. Introduction
2. Materials and Methods
2.1. Culture of Primary FRCs for Both Studies
2.2. Animals
2.2.1. Implantation of C57BL/6 Mouse with B16F10 Melanoma Cells
- Control Group (G1): Subcutaneous saline injections for baseline comparison.
- Checkpoint Inhibitor Groups (G2 & G3): Mice received immune checkpoint inhibitors targeting PD-1 and CTLA-4, with Group 2 receiving a standard dose of 100 µg of each antibody, and Group 3 receiving an escalated dose of 300 µg of each antibody.
-
Hydrogel-Based Therapies (G4, G5, G6): 3D hydrogels containing various therapeutic components were administered subcutaneously to evaluate synergistic effects:
- o
- G4: Combination of checkpoint inhibitors (300 µg each) and 2 million FRCs.
- o
- G5: 2 million FRCs and 90 ng of cytokines/chemokines, without checkpoint inhibitors.
- o
- G6: All components—checkpoint inhibitors (300 µg each), 2 million FRCs, and 90 ng cytokines (anti-mouse CTLA-4, LTα1β2, CXCL13, and CCL20)—to test maximal immune response.
2.2.2. Implantation of C57BL/6 Mouse with MC-38 Colorectal Carcinoma Cancer in Mice
- Control Group (G1): Subcutaneous saline injections as a baseline comparison.
-
Checkpoint Inhibitor Groups (G2, G3 & G4): Immune checkpoint inhibitors targeting PD-1 and CTLA-4, with varying doses:
- o
- G2: 50 µg of each antibody (anti-CTLA4 and anti-PD-1) per site, totaling 150 µg per mouse.
- o
- G3: 100 µg of each antibody per site, totaling 300 µg per mouse.
- o
- G4: 150 µg of each antibody per site, totaling 450 µg per mouse.
-
Hydrogel and Checkpoint Inhibitor-Based Therapy Groups (G5, G6 & G7): Mice received 3D hydrogels combined with checkpoint inhibitors to evaluate synergistic effects:
- o
- G5: Hydrogel plus 50 µg of each antibody (anti-CTLA4 and anti-PD-1) per site, totaling 150 µg per mouse.
- o
- G6: Hydrogel plus 100 µg of each antibody per site, totaling 300 µg per mouse.
- o
- G7: Hydrogel plus 150 µg of each antibody per site, totaling 450 µg per mouse.
-
Hydrogel, Checkpoint Inhibitor, and FRC-Based Therapy Groups (G8, G9 & G10): These groups were treated with 3D hydrogels, immune checkpoint inhibitors, and 5 million FRCs per mouse:
- o
- G8: Hydrogel + 100 µg of each antibody per site (300 µg per mouse) + 5 million FRCs.
- o
- G9: Hydrogel + 150 µg of each antibody per site (450 µg per mouse) + 5 million FRCs.
- o
- G10: Hydrogel + 200 µg of each antibody per site (600 µg per mouse) + 5 million FRCs.
2.3. Immunofluorescence Staining
2.4. Statistical Analysis
3. Results Summary
3.1. B16F10 Melanoma Cancer in Mice
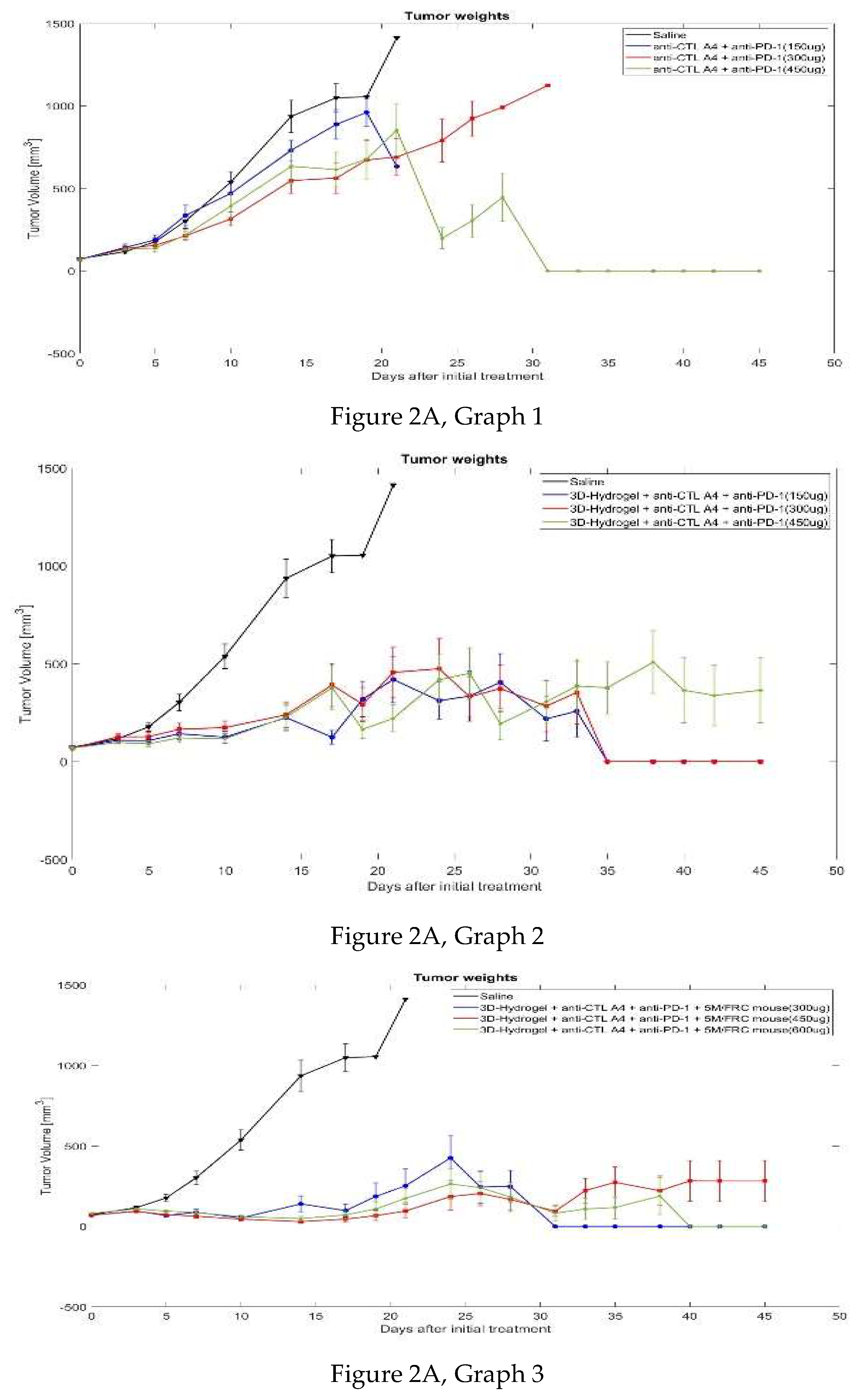
3.2. MC-38 Colorectal Cancer in Mice
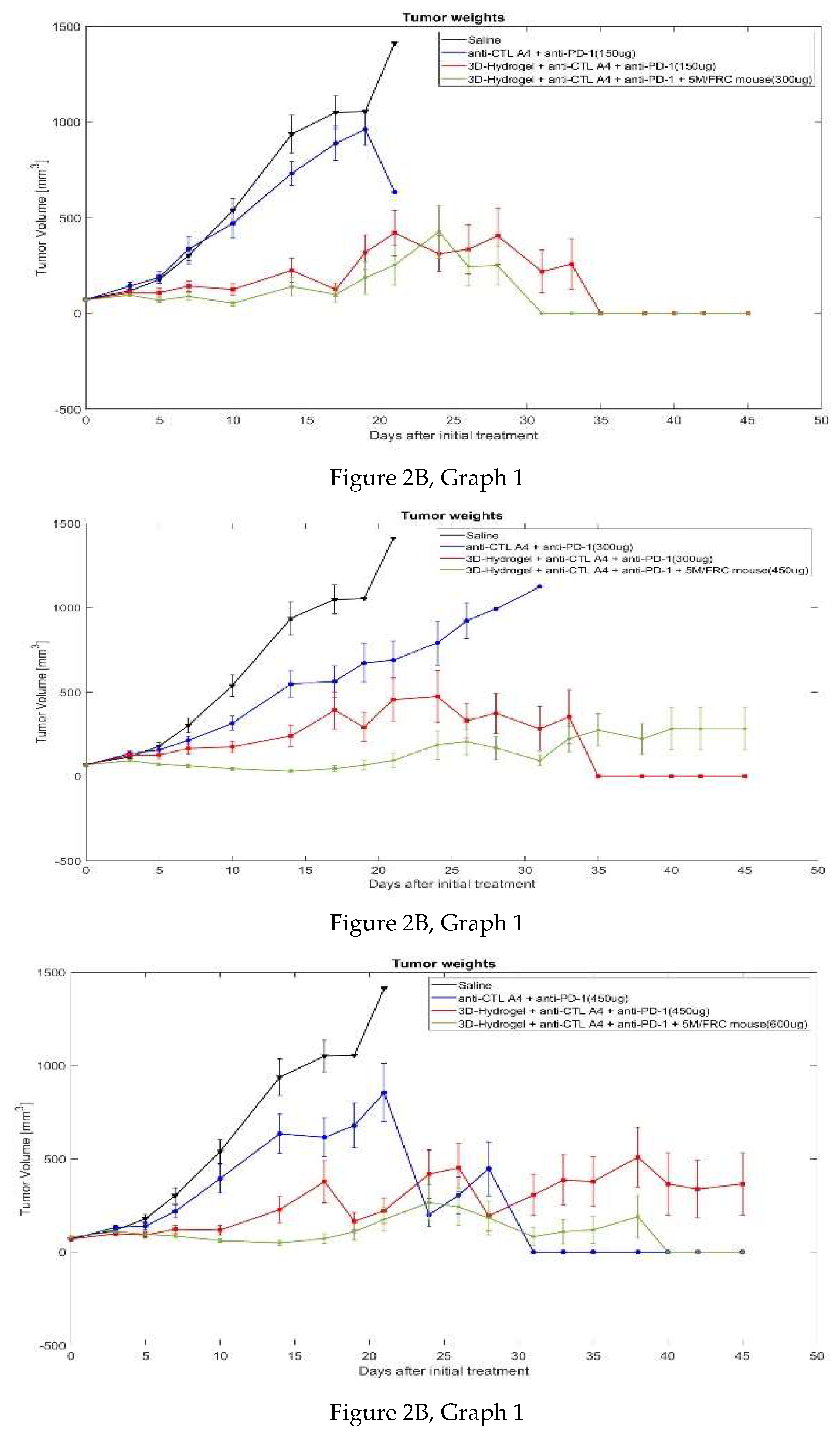
3.2.1. Tumor Volume
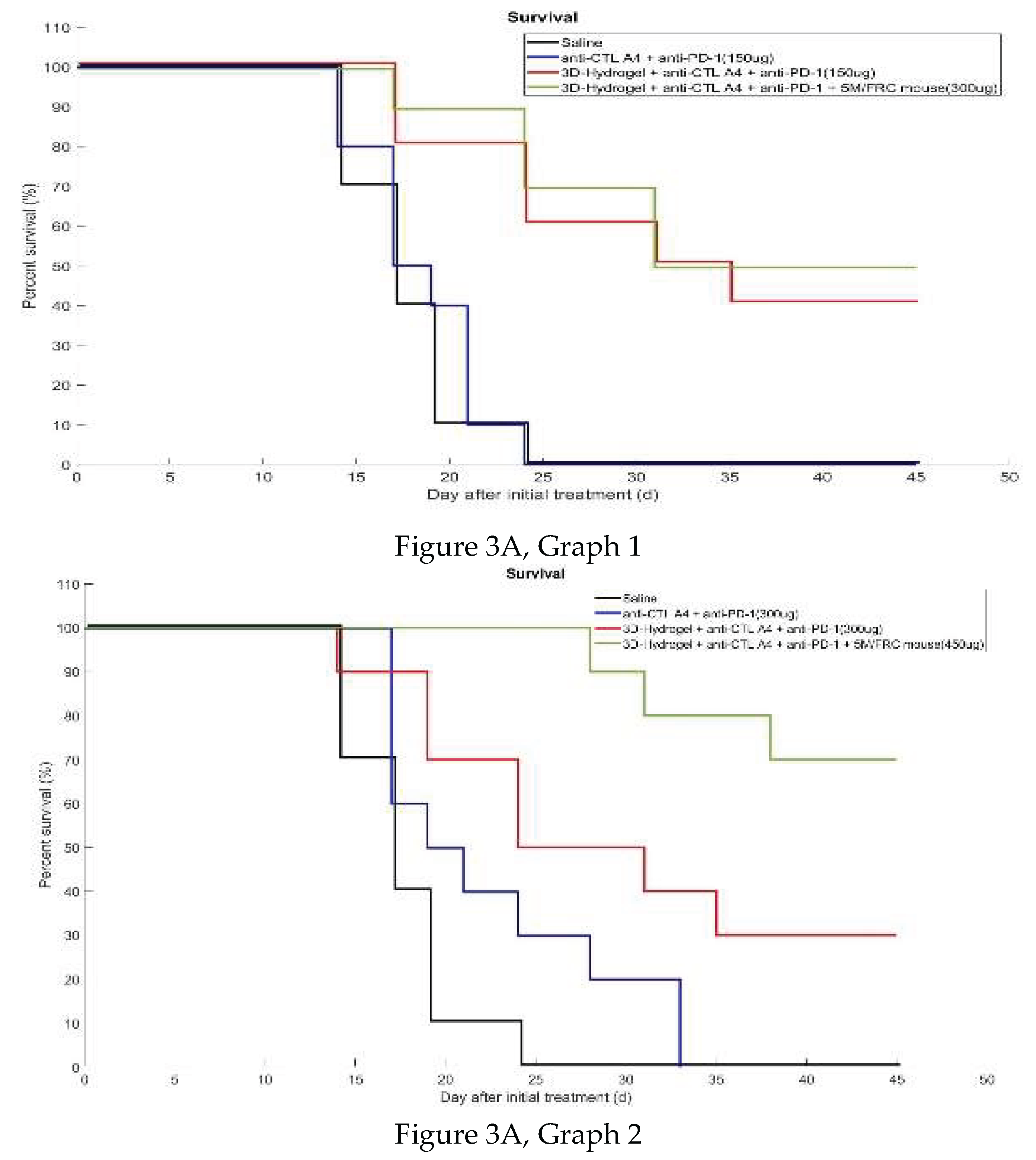
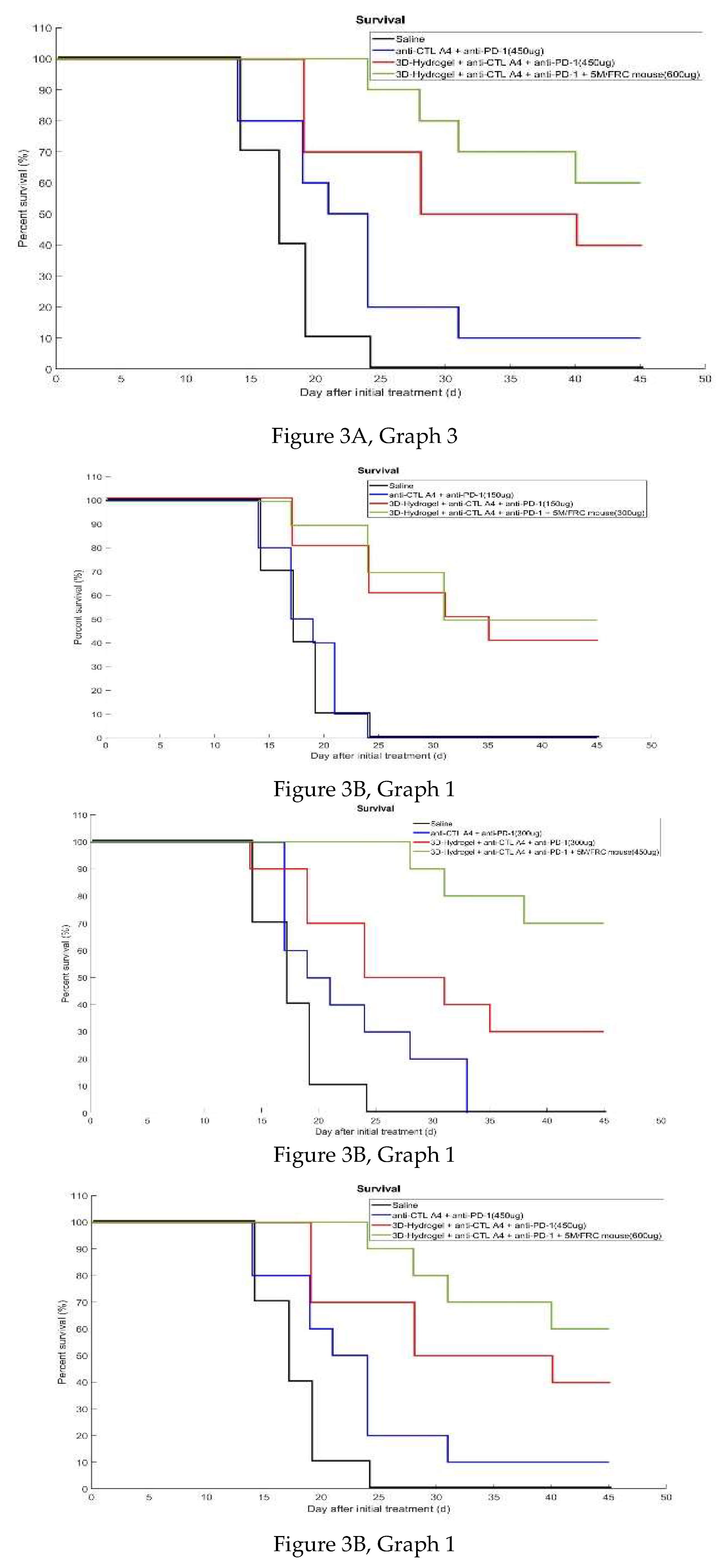
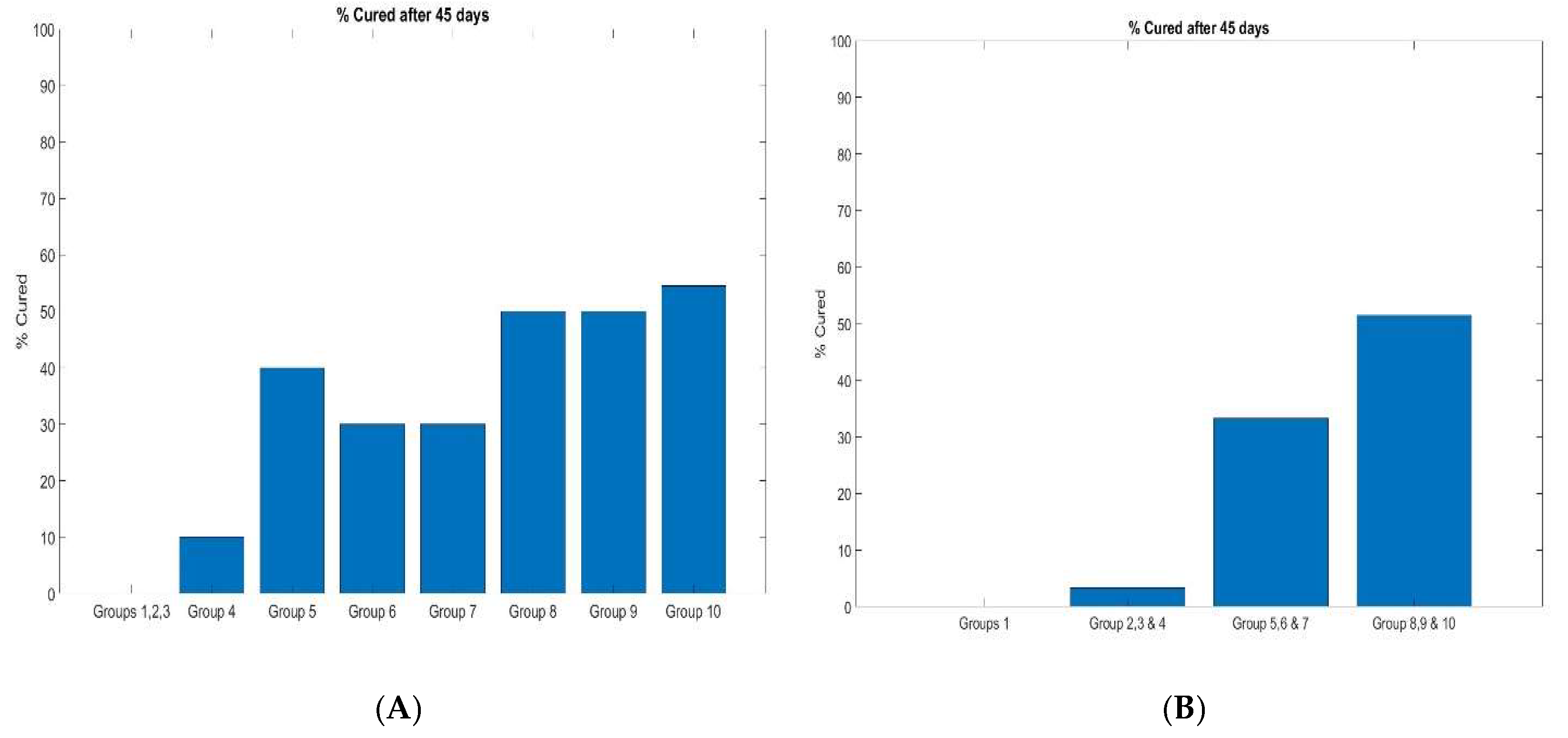
3.2.2. Percent Survival
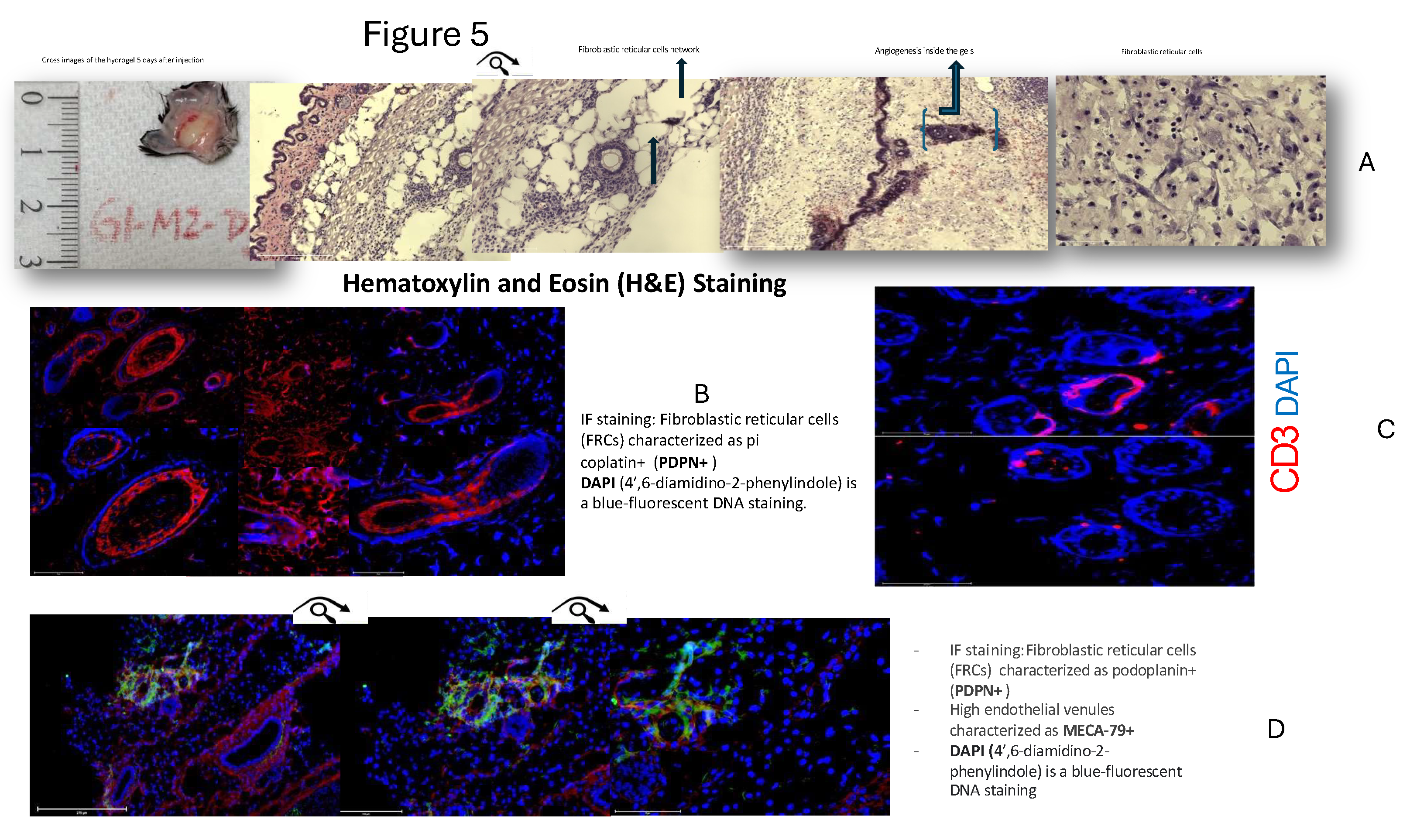
3.3. Staining Study
4. Discussion
5. Conclusion
Table of Contents
Author Contributions
Funding
Ethical Considerations
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kichloo, A.; Albosta, M.; Dahiya, D. S.; Guidi, J. C.; Aljadah, M.; Singh, J. P.; Shaka, H.; Wani, F.; Kumar, A.; Lekkala, M. R. Systemic Adverse Effects and Toxicities Associated with Immunotherapy: A Review. World journal of clinical oncology 2021, 12(3), 150–163. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zheng, L.; Chen, W.; Weng, W.; Song, J.; Ji, J. Delivery Strategies of Cancer Immunotherapy: Recent Advances and Future Perspectives. Journal of Hematology & Oncology 2019, 12 (1), 126. [CrossRef]
- Cunningham, N.; Lapointe, R.; Lerouge, S. Biomaterials for Enhanced Immunotherapy. APL bioengineering 2022, 6. [Google Scholar] [CrossRef] [PubMed]
- Liao, S.; Von Der Weid, P.-Y. Lymphatic System: An Active Pathway for Immune Protection. Seminars in Cell & Developmental Biology 2015, 38, 83–89. [Google Scholar] [CrossRef]
- Yang, F.; Shi, K.; Jia, Y.; Hao, Y.; Peng, J.; Qian, Z. Advanced Biomaterials for Cancer Immunotherapy. Acta Pharmacologica Sinica 2020, 41(7), 911–927. [Google Scholar] [CrossRef]
- Seidel, J. A.; Otsuka, A.; Kabashima, K.; Kabashima, K. Anti-PD-1 and Anti-CTLA-4 Therapies in Cancer: Mechanisms of Action, Efficacy, and Limitations. Frontiers in Oncology 2018, 8, 86. [Google Scholar] [CrossRef]
- Delclaux, I.; Ventre, K. S.; Jones, D.; Lund, A. W. The Tumor-Draining Lymph Node as a Reservoir for Systemic Immune Surveillance. Trends in cancer 10 (1), 28–37. [CrossRef]
- Notarbartolo, S.; Abrignani, S. Human T Lymphocytes at Tumor Sites. Seminars in Immunopathology 2022, 44(6), 883–901. [Google Scholar] [CrossRef] [PubMed]
- Nosenko, M. A.; Drutskaya, M. S.; Moisenovich, M. M.; Nedospasov, S. A. Bioengineering of Artificial Lymphoid Organs. Acta Naturae 2016, 8(2), 10–23. [Google Scholar] [CrossRef]
- Najibi, A. J.; Mooney, D. J.; Mooney, D. J. Cell and Tissue Engineering in Lymph Nodes for Cancer Immunotherapy. Advanced Drug Delivery Reviews 2020, 42–62. [Google Scholar] [CrossRef]
- Sznol, M.; Melero, I. Revisiting Anti-CTLA-4 Antibodies in Combination with PD-1 Blockade for Cancer Immunotherapy. Annals of Oncology 2021, 32(3), 295–297. [Google Scholar] [CrossRef]
- Savoia, P.; Astrua, C.; Fava, P. Ipilimumab (Anti-Ctla-4 Mab) in the Treatment of Metastatic Melanoma: Effectiveness and Toxicity Management. Human Vaccines & Immunotherapeutics 2016, 12 (5), 1092–1101. [CrossRef]
- FDA fast tracks nivolumab for advanced non-squamous non-small cell lung cancer. pharmaceutical-journal.com. https://pharmaceutical-journal.com/article/news/fda-fast-tracks-nivolumab-for-advanced-non-squamous-non-small-cell-lung-cancer.
- FDA Approval Summary: Pembrolizumab for Treatment of Metastatic Non-Small Cell Lung Cancer: First-Line Therapy and Beyond. Oncologist 2017, 22(11), 1392–1399. [CrossRef]
- Chae, Y. K.; Arya, A.; Iams, W. T.; Rocha Cruz, M.; Chandra, S.; Choi, J.; Giles, F. J. Current Landscape and Future of Dual Anti-CTLA4 and PD-1/PD-L1 Blockade Immunotherapy in Cancer; Lessons Learned from Clinical Trials with Melanoma and Non-Small Cell Lung Cancer (NSCLC). Journal for ImmunoTherapy of Cancer 2018, 6(1), 39. [Google Scholar] [CrossRef] [PubMed]
- Callahan, M. K.; Kluger, H. M.; Postow, M. A.; Segal, N. H.; Lesokhin, A. M.; Atkins, M. B.; Kirkwood, J. M.; Krishnan, S.; Bhore, R.; Horak, C.; Wolchok, J. D.; Sznol, M. Nivolumab Plus Ipilimumab in Patients With Advanced Melanoma: Updated Survival, Response, and Safety Data in a Phase I Dose-Escalation Study. Journal of Clinical Oncology 2017, 36(4), 391–398. [Google Scholar] [CrossRef] [PubMed]
- Lebbé, C.; Meyer, N.; Mortier, L.; Marquez-Rodas, I.; Robert, C.; Rutkowski, P.; Menzies, A. M.; Eigentler, T.; Ascierto, P. A.; Smylie, M.; Schadendorf, D.; Ajaz, M.; Svane, I. M.; Gonzalez, R.; Rollin, L.; Lord-Bessen, J.; Saci, A.; Grigoryeva, E.; Pigozzo, J. Evaluation of Two Dosing Regimens for Nivolumab in Combination With Ipilimumab in Patients With Advanced Melanoma: Results From the Phase IIIb/IV CheckMate 511 Trial. Journal of Clinical Oncology 2019, 37(11), 867–875. [Google Scholar] [CrossRef] [PubMed]
- Five-Year Survival Outcomes with Nivolumab (NIVO) plus Ipilimumab (IPI) versus Chemotherapy (Chemo) as First-Line (1L) Treatment for Metastatic Non–Small Cell Lung Cancer (NSCLC): Results from CheckMate 227. Journal of Clinical Oncology 2022, 40 (17_suppl), LBA9025. [CrossRef]
- Kim, D.; Cha, B.-H.; Ahn, J.; Arai, Y.; Choi, B.; Lee, S.-H. Physicochemical Properties in 3D Hydrogel Modulate Cellular Reprogramming into Induced Pluripotent Stem Cells. Advanced Functional Materials 2021, 31(7), 2007041. [Google Scholar] [CrossRef]
- Lavrentev, F.V.; Shilovskikh, V.V.; Alabusheva, V.S.; Yurova, V.Y.; Nikitina, A.A.; Ulasevich, S.A.; Skorb, E.V. Diffusion-Limited Processes in Hydrogels with Chosen Applications from Drug Delivery to Electronic Components. Molecules 2023, 28, 5931. [Google Scholar] [CrossRef]
- C. Wu, L. Z. Teng, C. Wang, T. Qian, Z. Hu, Z. Zeng, Gels. 2023, 9, 116.
- Najibi, A. J.; Mooney, D. J.; Mooney, D. J. Cell and Tissue Engineering in Lymph Nodes for Cancer Immunotherapy. Advanced Drug Delivery Reviews 2020, 42–62. [Google Scholar] [CrossRef]
- Perez-Shibayama, C.; Gil-Cruz, C.; Ludewig, B. Fibroblastic Reticular Cells at the Nexus of Innate and Adaptive Immune Responses. Immunological Reviews 2019, 289(1), 31–41. [Google Scholar] [CrossRef] [PubMed]
- Vella, G.; Guelfi, S.; Bergers, G.; Bergers, G. High Endothelial Venules: A Vascular Perspective on Tertiary Lymphoid Structures in Cancer. Frontiers in Immunology 2021, 12, 736670. [Google Scholar] [CrossRef]
- Milutinovic, S.; Abe, J.; Godkin, A. J.; Stein, J. V.; Gallimore, A. The Dual Role of High Endothelial Venules in Cancer Progression versus Immunity. Trends in cancer 2021, 7(3), 214–225. [Google Scholar] [CrossRef]
- Lu, T. T.; Lu, T. T.; Browning, J. L. Role of the Lymphotoxin/LIGHT System in the Development and Maintenance of Reticular Networks and Vasculature in Lymphoid Tissues. Frontiers in Immunology 2014, 5, 47. [Google Scholar] [CrossRef] [PubMed]
- Sardi, M.; Lubitz, A.; Giese, C. Modeling Human Immunity In Vitro: Improving Artificial Lymph Node Physiology by Stromal Cells. 2016, 2 (3), 143–150. [CrossRef]
- Suematsu, S.; Watanabe, T. Generation of a Synthetic Lymphoid Tissue–like Organoid in Mice. Nature Biotechnology 2004, 22(12), 1539–1545. [Google Scholar] [CrossRef] [PubMed]
- Maarouf, O. H.; Uehara, M.; Kasinath, V.; Solhjou, Z.; Banouni, N.; Bahmani, B.; Jiang, L.; Yilmam, O. A.; Guleria, I.; Lovitch, S. B.; Grogan, J. L.; Fiorina, P.; Sage, P. T.; Bromberg, J. S.; McGrath, M. M.; Abdi, R. Repetitive Ischemic Injuries to the Kidneys Result in Lymph Node Fibrosis and Impaired Healing. JCI insight 2018, 3 (13). [CrossRef]

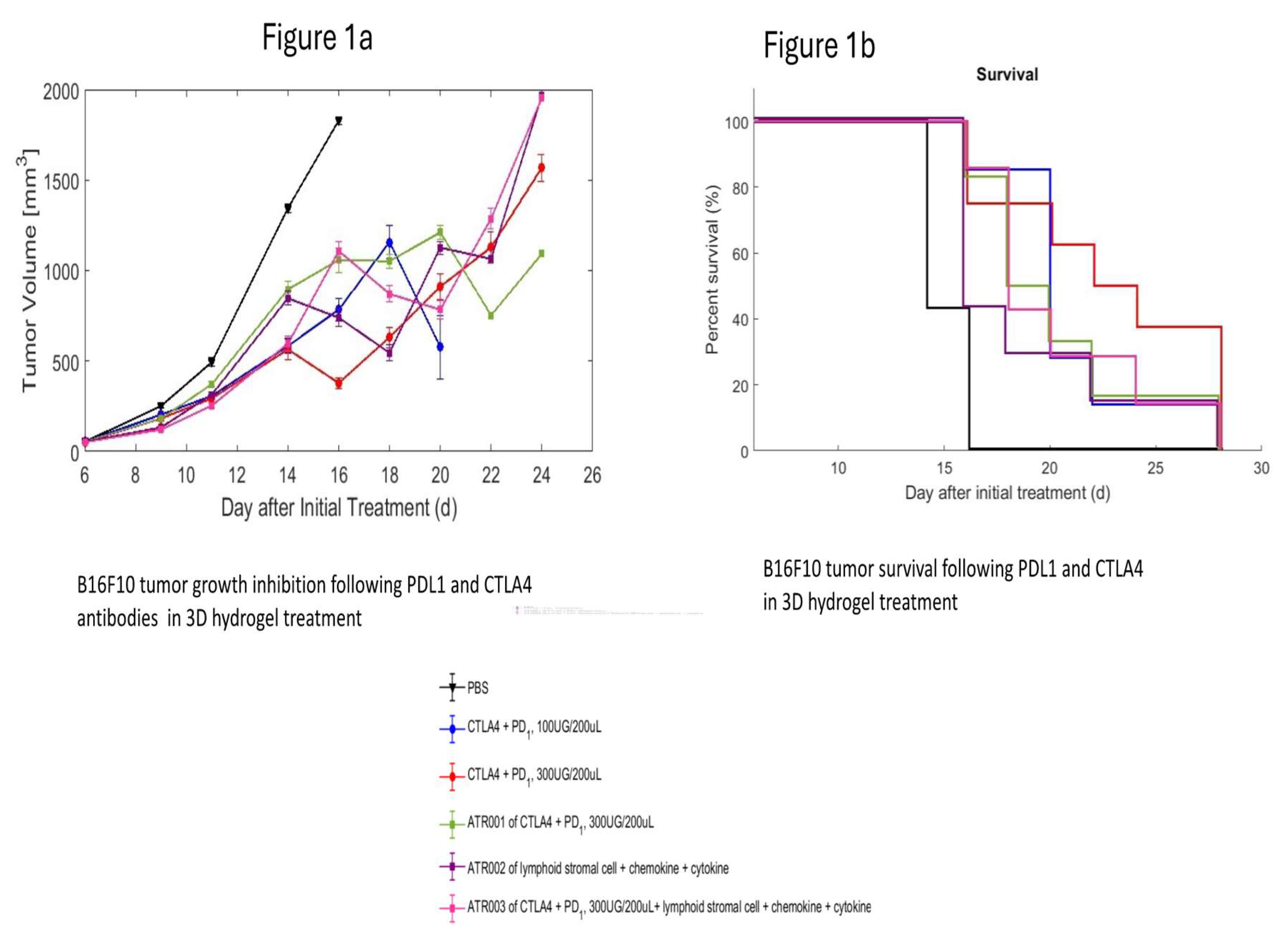
| Group | No. of mice | Treatment | DOSE, Route (mg/kg) | % MAX TGI on (Day) | MST* (Day) |
| G1 | 7 | Vehicle | ----, SC | ---- | 16.0 |
| G2 | 7 | Anti-mouse PD-1Ab + Anti-mouse CTLA4 Ab | 100μg + 100μg, IP | 56.7% (14) | 20.2 |
| G3 | 8 | 3D hydrogel + PD-1Ab + Anti-mouse CTLA4 Ab | 300μg + 300μg, IP | 58.8% (16) | 23.4 |
| G4 | 6 | 3D hydrogel + PD-1Ab + Anti-mouse CTLA4 Ab + FRC | 200μL+ 300μg + 300μg +2m FRC, SC | 40.5% (16) | 19.6 |
| G5 | 7 | 3D hydrogel + FRC +chemokines/cytokine | 2m FRC, 90ng Chemokine/cytokine, SC | 47.1% (9) | 16.8 |
| G6 | 7 | 3D hydrogel + PD-1Ab + Anti-mouse CTLA4 Ab + FRC +chemokines/cytokine | 200μL+ 300μg + 300μg +2m FRC, Chemokine/cytokine, SC | 55.7% (14) | 17 |
| Group | No. of mice | Treatment | Dose | Treatment Schedule(Est)** |
| G1 | 10 | Saline | Q3D, Days 7, 10, 13, 16, and 19** (injection in three sites every 3 days from day 7 until day 20) | |
| G2 | 10 | anti- CTLA4 + anti-PD-1 | 50ug per mAB Per site (3 sites total 150ug per mouse) |
Q3D, Days 7, 10, 13, 16, and 19**(Injection in three sites every 3 days from day 7 until day 19) |
| G3 | 10 | anti- CTLA4 + anti-PD-1 |
100ug per mAB Per site (3 sites total 300ug mouse) | Q3D, Days 7, 10, 13, 16, and 20** (Injection in three sites every three days from day 7 until day 20) |
| G4 | 10 | anti- CTLA4 + anti-PD-1 |
150ug per mAB Per site (3 sites total 450ug mouse) | Q3D, Days 7, 10, 13, 16, and 20** (Injection in three sites every 3 days from day 7 until day 19) |
| G5 | 10 | 3D hydrogel + anti- CTLA4 + anti-PD-1 |
50ug per mAB Per site (3 sites total 150ug mouse)) |
Q3D, Days 7, 10, 13, 16, and 20** (Injection in three sites every 3 days from day 7 until day 20) |
| G6 | 10 | 3D hydrogel + anti- CTLA4 + anti-PD-1 |
100ug per mAB Per site (3 sites total 300ug mouse) | Q3D, Days 7, 10, 13, 16, and 20** (Injection in three sites every 3 days from day 7 until day 20) |
| G7 | 10 | 3D hydrogel + anti- CTLA4 +anti-PD-1 | 150ug per mAB Per site (3 sites total 450ug mouse) | Q3D, Days 7, 10, 13, 16, and 20** (Injection in three sites every 3 days from day 7 until day 20) |
| G8 | 10 | 3D hydrogel + anti- CTLA4 + anti-PD-1+ 5M FRC/mouse |
100ug per mAB Per site (3 sites total 300ug mouse) | QD, Days 7 **and 13 |
| G9 | 10 | 3D hydrogel + anti- CTLA4 + anti-PD-1+ 5M FRC/mouse |
150ug per mAB Per site (3 sites total 450ug mouse) | QD, Days 7 **and 13 |
| G10 | 10 | 3D hydrogel + anti- CTLA4 + anti-PD-1+ 5M FRC/mouse |
200ug per mAB Per site (3 sites total 600ug per mouse) | QD, Days 7 **and 13 |
| Day | 0 | 3 | 5 | 7 | 10 | 14 | 17 | 19 | 21 | 24 | 26 | 28 | 31 | 33 | 35 | 38 | 40 | 42 | 45 | ||||||||||||||||||
| Group 1 | 72.9 | 116 | 177 | 302 | 538 | 936 | 1050 | 1054 | 1412 | ||||||||||||||||||||||||||||
| Group 2 | 70.6 | 142 | 188 | 336 | 470 | 731 | 888 | 961 | 633 | ||||||||||||||||||||||||||||
| Group 3 | 68.6 | 136 | 155 | 213 | 315 | 547 | 563 | 672 | 690 | 790 | 923 | 992 | 1124 | ||||||||||||||||||||||||
| Group 4 | 68.7 | 131 | 138 | 217 | 393 | 634 | 614 | 677 | 853 | 198 | 304 | 446 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 5 | 69.2 | 108 | 107 | 142 | 124 | 224 | 124 | 319 | 419 | 311 | 335 | 404 | 218 | 257 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 6 | 67.9 | 126 | 127 | 164 | 173 | 239 | 391 | 292 | 455 | 474 | 330 | 373 | 283 | 353 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 7 | 69.6 | 97.8 | 91.3 | 121 | 117 | 227 | 376 | 164 | 221 | 417 | 451 | 193 | 305 | 385 | 377 | 508 | 364 | 338 | 364 | ||||||||||||||||||
| Group 8 | 69.7 | 95.0 | 68.2 | 88.3 | 52.9 | 139 | 98.4 | 186 | 253 | 425 | 245 | 249 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 9 | 69.6 | 93.8 | 72.9 | 63.6 | 45.5 | 30.1 | 45.9 | 67.0 | 96.0 | 186 | 204 | 167 | 94.6 | 223 | 274 | 222 | 283 | 282 | 282 | ||||||||||||||||||
| Group 10 | 78.1 | 111 | 98.1 | 89.5 | 61.0 | 49.3 | 71.8 | 108 | 174 | 264 | 241 | 182 | 82.7 | 108 | 118 | 188 | 0 | 0 | 0 | ||||||||||||||||||
| (a) | |||||||||||||||||||||||||||||||||||||
| Day | 0 | 3 | 5 | 7 | 10 | 14 | 17 | 19 | 21 | 24 | 26 | 28 | 31 | 33 | 35 | 38 | 40 | 42 | 45 | ||||||||||||||||||
| Group 1 | 4.7 | 10.3 | 21.8 | 42.5 | 63.7 | 98.6 | 85.2 | 0 | 0 | ||||||||||||||||||||||||||||
| Group 2 | 4.6 | 21.7 | 29.7 | 62.4 | 77.5 | 61.4 | 90.3 | 83.5 | 0 | ||||||||||||||||||||||||||||
| Group 3 | 4.1 | 16.7 | 19.5 | 23.6 | 42.0 | 77.7 | 93.2 | 116 | 109 | 130 | 106 | 5.1 | 2.7 | ||||||||||||||||||||||||
| Group 4 | 4.7 | 12.1 | 21.5 | 34.9 | 77.4 | 105 | 103.9 | 119 | 157 | 64.5 | 98.8 | 144 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 5 | 4.6 | 13.0 | 23.1 | 27.9 | 29.6 | 61.7 | 34.9 | 91 | 118 | 93.3 | 127 | 146 | 111 | 132 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 6 | 4.3 | 15.6 | 21.9 | 31.2 | 33.3 | 66.1 | 109.7 | 87.5 | 127 | 153 | 103 | 118 | 129 | 162 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 7 | 4.6 | 5.6 | 14.7 | 23.5 | 26.4 | 71.0 | 113 | 47.7 | 68.2 | 130 | 129 | 78.9 | 110 | 134 | 132 | 159 | 167 | 155 | 167 | ||||||||||||||||||
| Group 8 | 4.0 | 6.7 | 10.8 | 21.0 | 13.3 | 49.0 | 40.3 | 84.2 | 105 | 137 | 99.5 | 100 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | ||||||||||||||||||
| Group 9 | 4.5 | 7.2 | 7.9 | 10.3 | 10.0 | 10.0 | 19.6 | 30.9 | 41.1 | 84.5 | 75.8 | 66.8 | 30.6 | 76.3 | 96.3 | 91 | 127 | 127 | 127 | ||||||||||||||||||
| Group 10 | 5.4 | 4.3 | 6.3 | 10.7 | 8.3 | 13.4 | 25.4 | 43.6 | 62.7 | 95 | 99.8 | 90.8 | 50.2 | 65.9 | 71.7 | 114 | 0 | 0 | 0 | ||||||||||||||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




