Submitted:
02 June 2025
Posted:
04 June 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Methods
2.1. Objectives
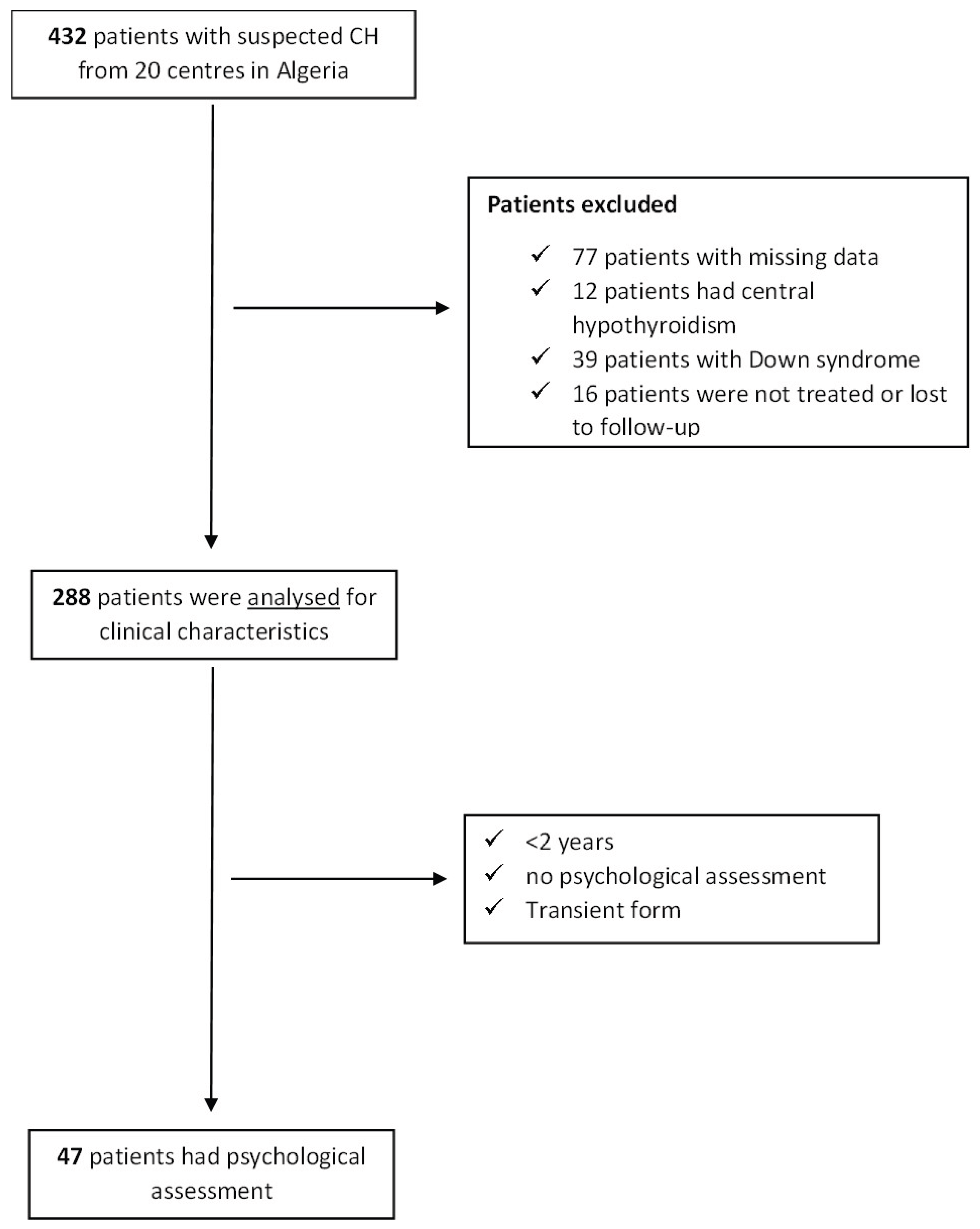
2.2. Study Design and Population
2.3. Data Collection
2.4. Definitions
2.5. Neurodevelopmental Assessment
2.6. Reassessment
2.7. Ethics
2.8. Statistical Analysis
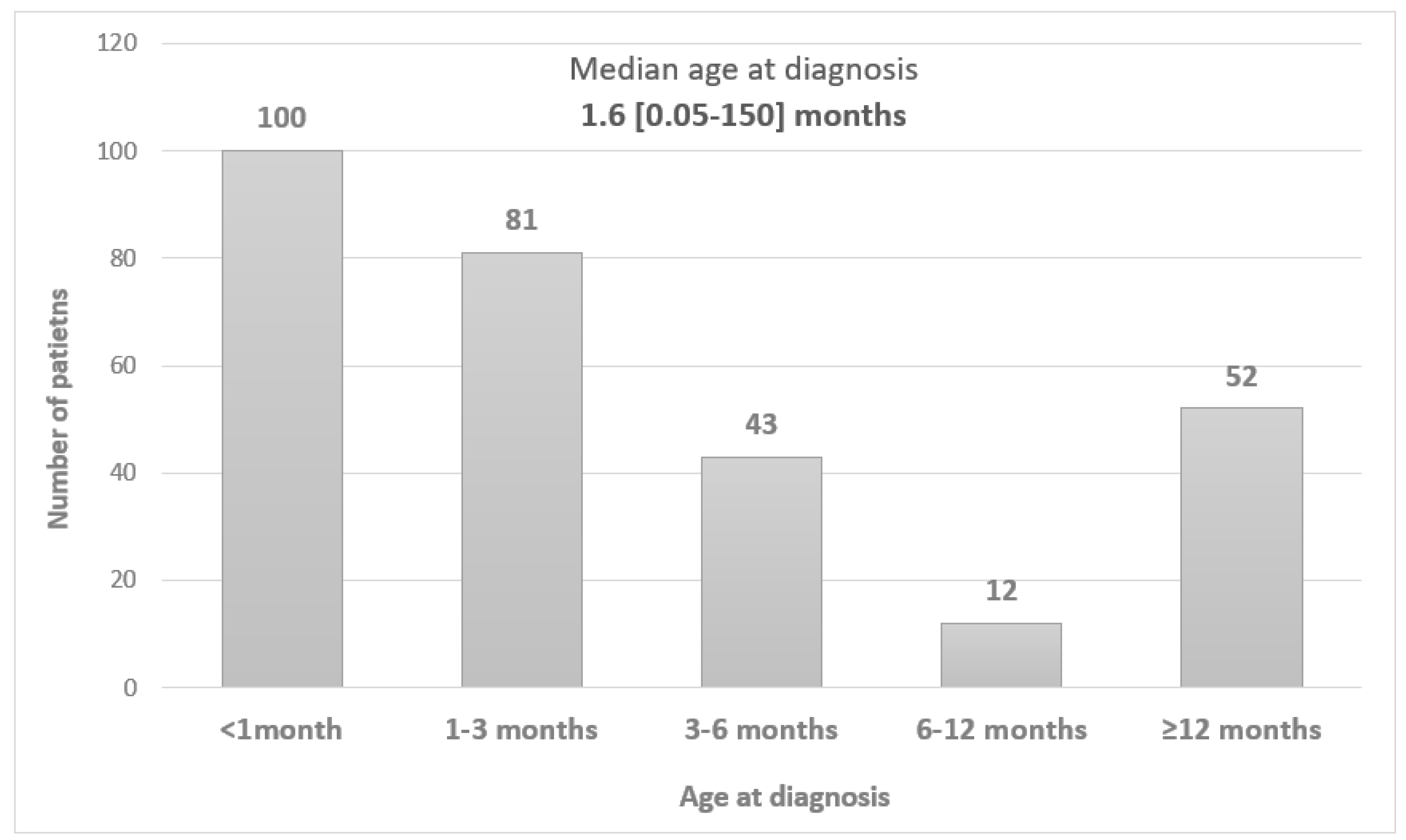
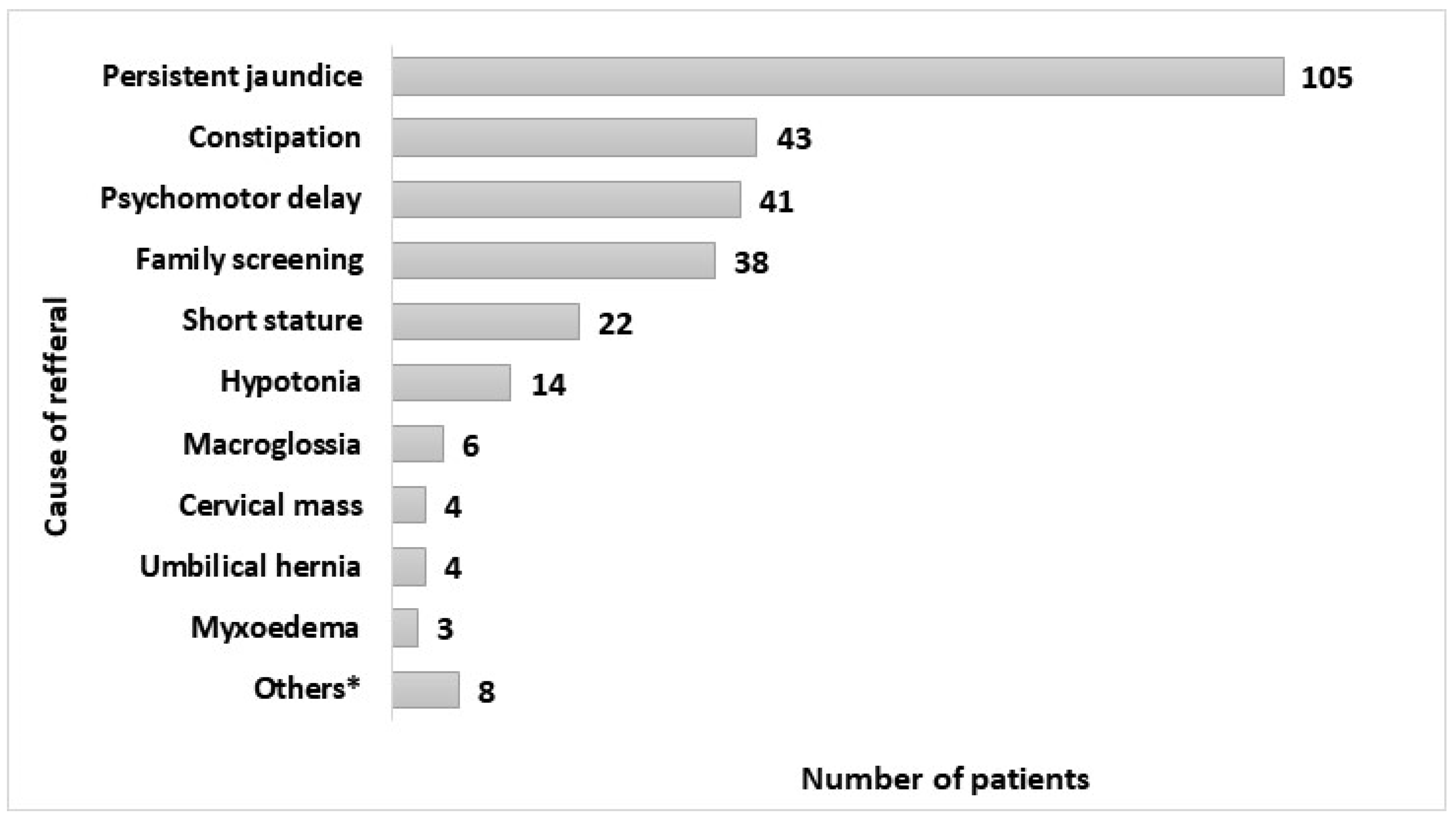
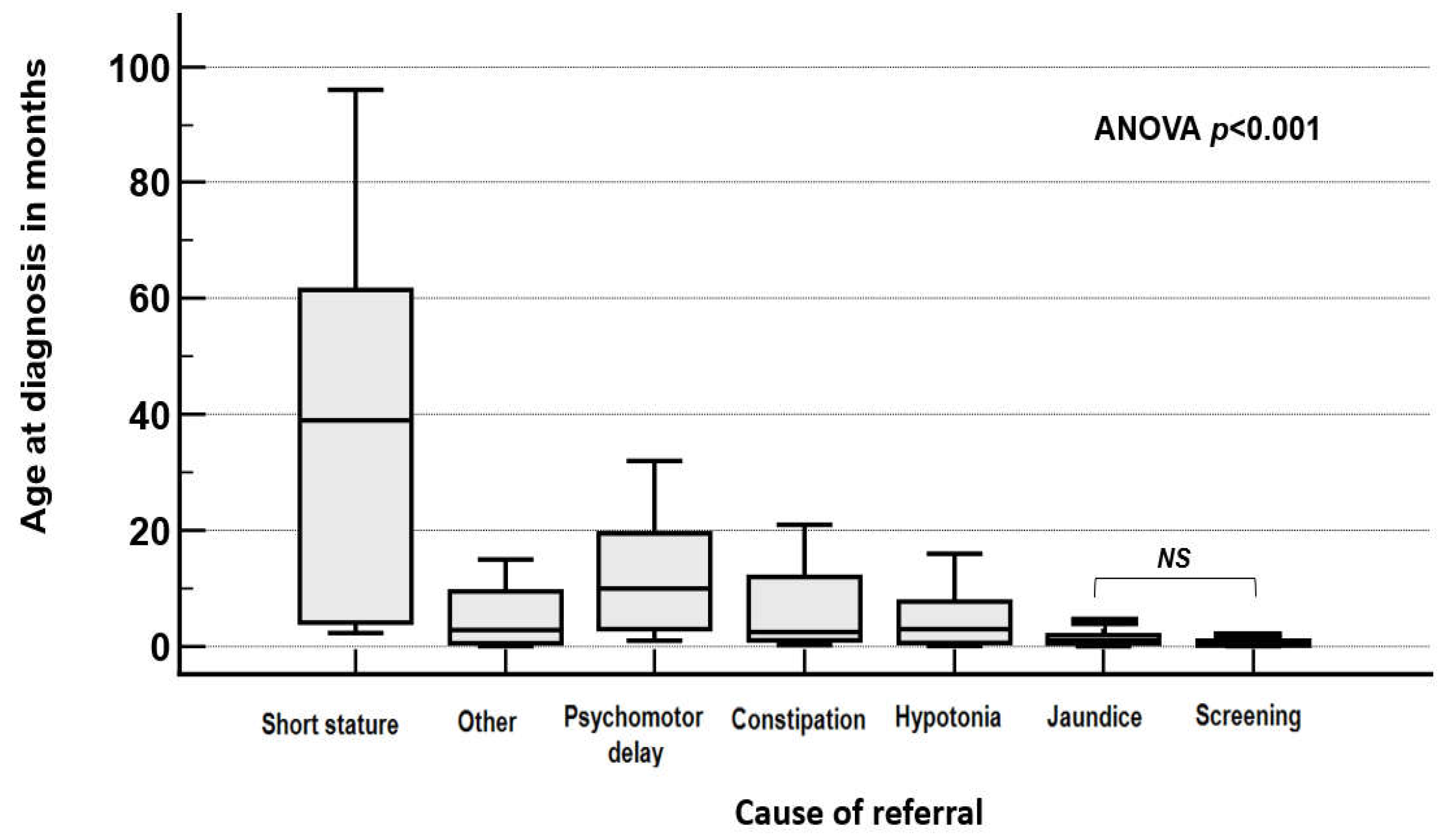
3. Results
3.1. Imaging Exams and Aetiological Groups
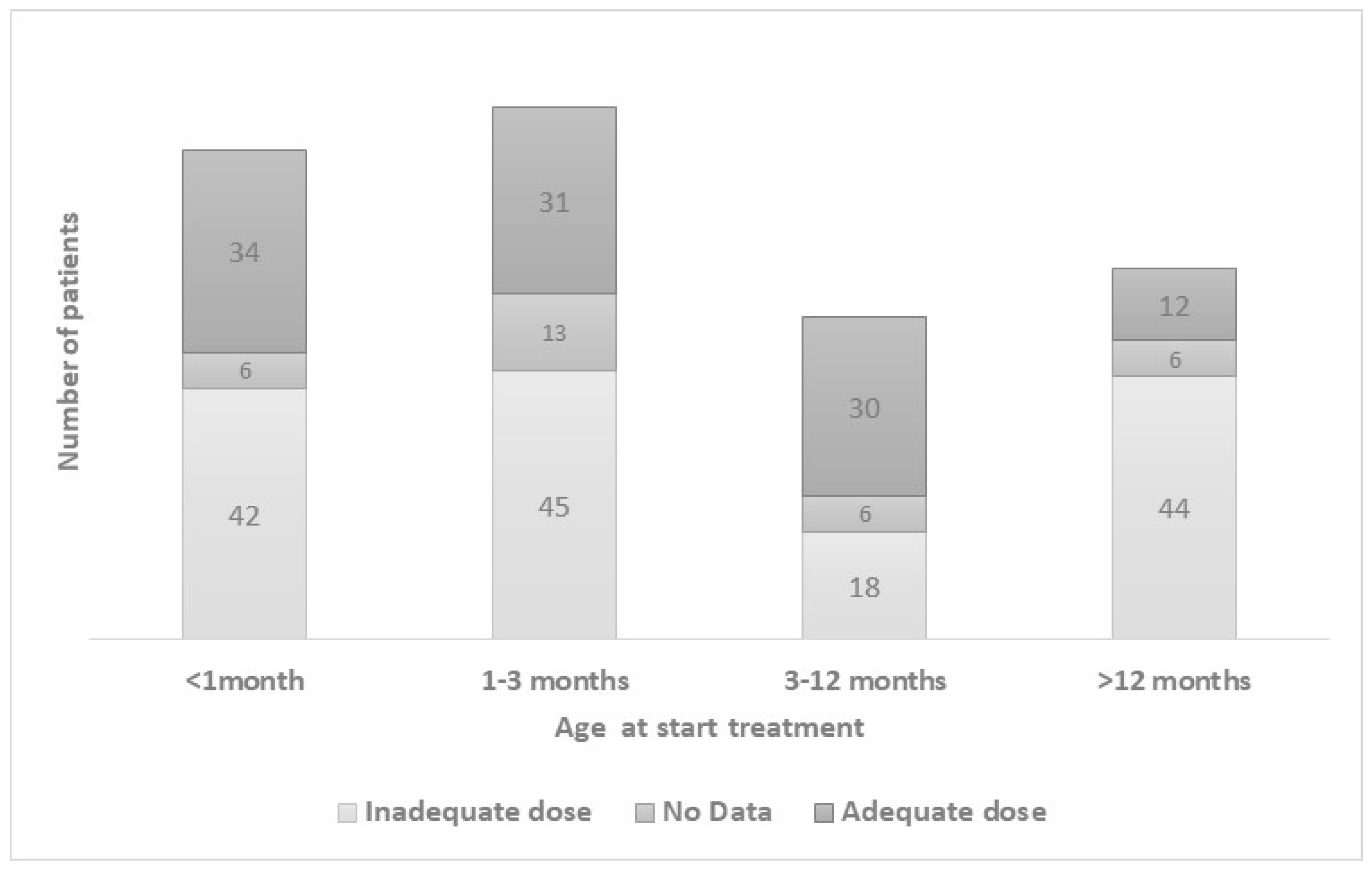
3.2. Treatment (Figure 5):
3.3. Outcomes
3.3.1. Transient vs permanent CH
3.3.2. Neurodevelopmental Assessment (Table 5)
| Neurodevelopmental Data | N(%) | Age at evaluation Mean± SD |
|---|---|---|
| School age | 206 (71.5%) | 10.1±3.3 [5.5-16.7] years |
| School age data | 88 (43%) | |
| School failure | 27/88 (31%) | 10.8±3.9 [5.1-21.7] years |
| Language Delay | 16(6%) | 18.4±18.1[0.95-48] months |
| Psychomotor Delay | 43(15%) | 23.3±25.1[1.9-93.9] months |
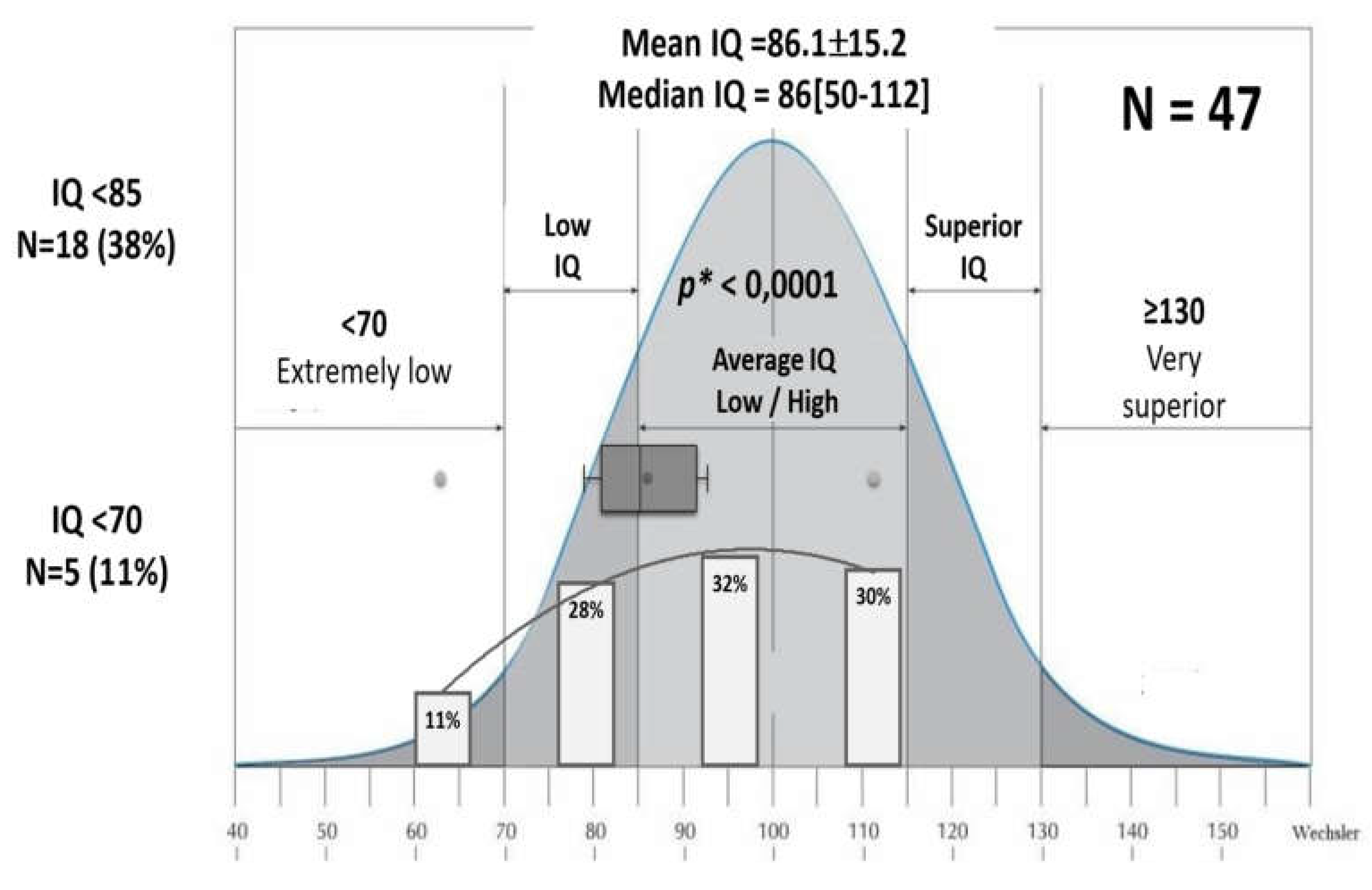
| IQ assessment N=47 | ||
| Normal IQ ≥85 | 29 (62%) | 5.1±1.7 [2.7-8] |
| Low IQ <85 | 13 (28%) | 5.2±1.8 [3-7.8] |
| Very Low IQ <70 | 5 (11%) | 4.8±2.5 [3-9.3] |
| Cognitive Disharmony | 7 (15%) | 5.6±1.3 [3-6.7] |
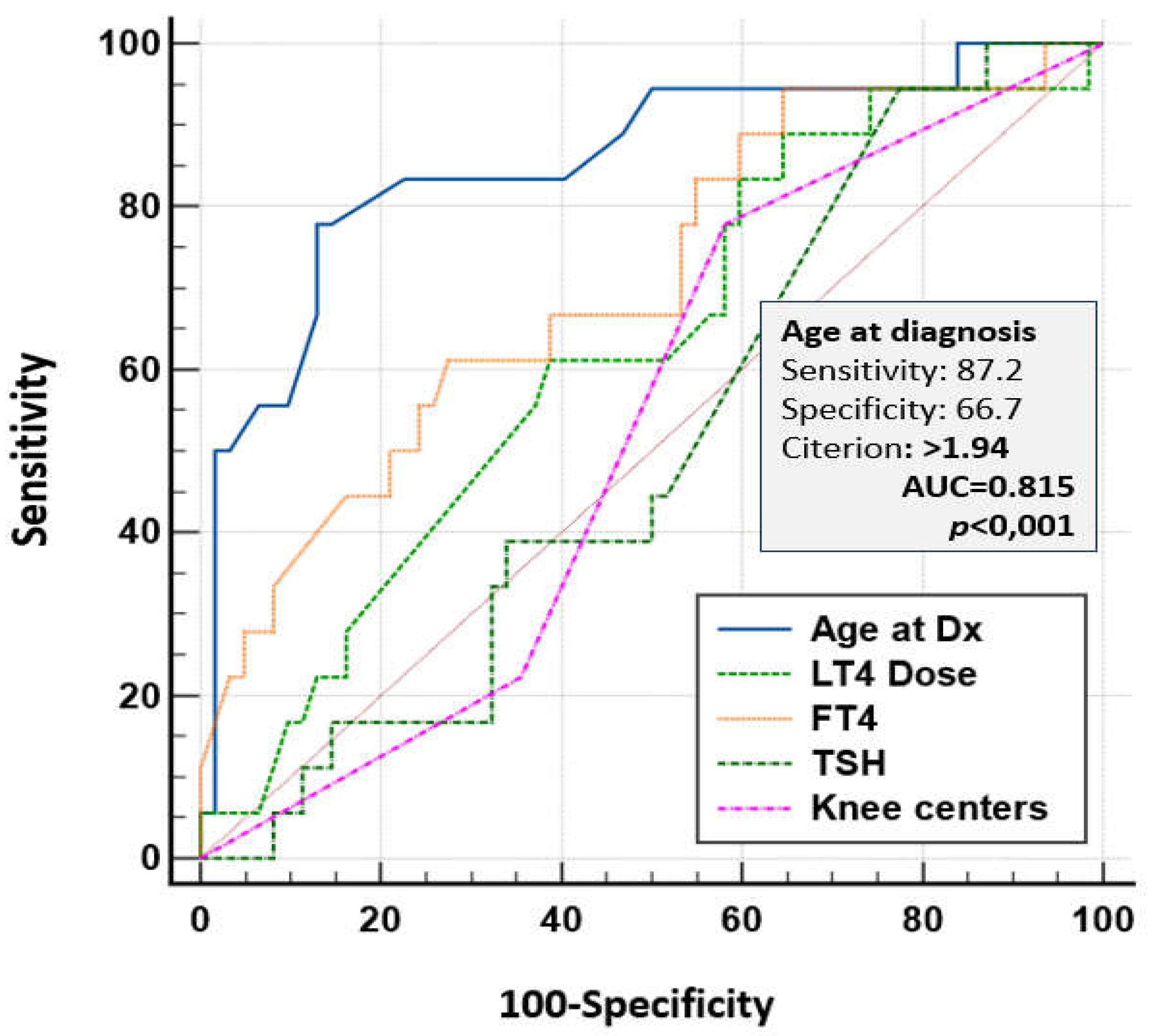
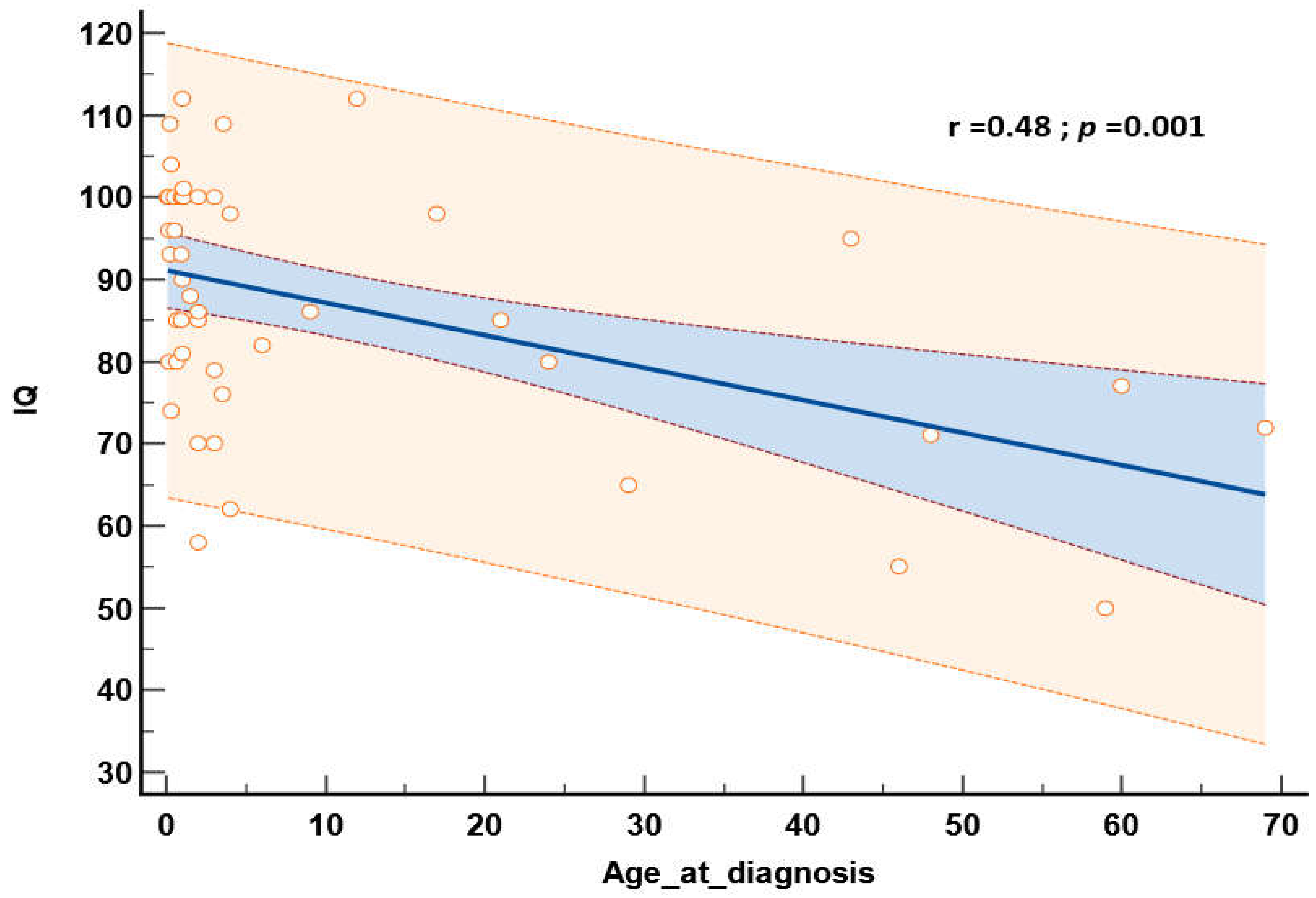
3.3.3. IQ Evaluation (Table 5 and 6, Figure 7 and Figure 8)
| Global N=47 |
IQ < 85 N=18 |
IQ≥85 N=29 |
p | ||
|---|---|---|---|---|---|
| Sexe M/F | 15/32 | 5/13 | 10/19 | 0.63 | |
| Mean Age IQ test years | 5.3±1.8 [2.5-9.3] | 5.5±2.1 [3-9.3] | 5.2±1.7 [2.5-8] | 0.68 | |
|
Age at treatment in month Median (range) |
2 (0.1-69) |
3.75 (0.2-69) |
1.1 (0.1-42.7) |
0.01 | |
| Age start L-T4 | ≤1 month (n, %) | 16 (25.5%) | 3 (17%) | 13 (45%) | 0.05 |
| >1 month (n, %) | 31 (74.5%) | 15 (83%) | 16 (55%) | ||
| Etiology | Dysgenesis | 25 (53%) | 13 (72%) | 12 (41%) | 0.05 |
| In-situ gland | 21 (45%) | 5 (28%) | 16 (55%) | ||
| L-T4 Dose µg/kg/j | <9 µg/kg/j | 27 (54%) | 12 (67%) | 15 (52%) | 0.26 |
| ≥9 µg/kg/j | 15 (32%) | 4 (22%) | 11 (38%) | ||
4. Discussion
4.1. The Consequences of Delayed Diagnosis and Treatment
4.2. Etiological Diagnosis and Genetic Factors in Congenital Hypothyroidism
4.3. Permanent and Transient Forms of CH
4.4. Treatment of CH
4.5. Neurodevelopmental Data
4.5.1. Educational Outcomes and Cognitive Impairments
4.5.2. Preventing Delayed Diagnosis and Reducing Cognitive Risks in Congenital Hypothyroidism
4.5.3. Challenges in Timely Screening Implementation
4.5.4. Education and Public Awareness Initiatives
Healthcare Provider Education
Public Awareness Campaigns
5. Limitations
6. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- S. He, X. Ma, J. Yang, and L. Li, “Levothyroxine treatment for congenital hypothyroidism based on thyroid function: a 10-year clinical retrospective study,” BMC Endocr. Disord., vol. 22, no. 1, 2022. [CrossRef]
- M. Donaldson and J. Jones, “Optimising outcome in congenital hypothyroidism; current opinions on best practice in initial assessment and subsequent management,” J. Clin. Res. Pediatr. Endocrinol., vol. 4, no. 4, Nov. 2012. [CrossRef]
- R. J. McGill, T. J. Ward, and G. L. Canivez, “Use of translated and adapted versions of the WISC-V: Caveat emptor,” Sch. Psychol. Int., vol. 41, no. 3, pp. 276–294, Jun. 2020. [CrossRef]
- S. D. Grosse and G. Van Vliet, “Prevention of intellectual disability through screening for congenital hypothyroidism: How much and at what level?,” Arch. Dis. Child., vol. 96, no. 4, pp. 374–379, Apr. 2011. [CrossRef]
- F. Grob, S. Lain, and A. Olivieri, “Newborn screening for primary congenital hypothyroidism: past, present and future,” Eur. Thyroid J., vol. 14, no. 2, Feb. 2025. [CrossRef]
- C. I. Kaye et al., “Introduction to the newborn screening fact sheets,” Pediatrics, vol. 118, no. 3, pp. 1304–1312, Sep. 2006. [CrossRef]
- D. A. Fisher et al., “Screening for congenital hypothyroidism: Results of screening one million North American infants,” J. Pediatr., vol. 94, no. 5, pp. 700–705, May 197. [CrossRef]
- M. V Rastogi and S. H. LaFranchi, “Congenital hypothyroidism,” Orphanet J. Rare Dis., vol. 5, no. 1, p. 17, Dec. 2010. [CrossRef]
- L. Pezzuti, P. P. De Lima, and V. M. A. Dias, “Hipotireoidismo congĉnito: Perfil clínico dos recém-nascidos identificados pelo Programa de Triagem Neonatal de Minas Gerais,” J. Pediatr. (Rio. J)., vol. 85, no. 1, pp. 72–79, Feb. 2009. [CrossRef]
- D. B. Grant, I. Smith, P. W. Fuggle, S. Tokar, and J. Chapple, “Congenital hypothyroidism detected by neonatal screening: Relationship between biochemical severity and early clinical features,” Arch. Dis. Child., vol. 67, no. 1, pp. 87–90, Jan. 1992. [CrossRef]
- S. Raiti and G. H. Newns, “Cretinism: Early diagnosis its relation to mental prognosis,” Arch. Dis. Child., vol. 46, no. 249, pp. 692–694, Oct. 1971. [CrossRef]
- J. Alm, A. Larsson, and R. Zetterström, “Congenital Hypothyroidism in Sweden Incidence and Age at Diagnosis,” Acta Pædiatrica, vol. 67, no. 1, pp. 1–3, Jan. 1978. [CrossRef]
- S. LaFranchi, “Congenital hypothyroidism: Etiologies, diagnosis, and management,” Thyroid, vol. 9, no. 7, pp. 735–740, Jul. 1999. [CrossRef]
- K. A. Yunis, M. R. Nasr, G. Lepejian, S. Najjar, and R. Daher, “Short Report: False-negative primary neonatal thyroid screening: the need for clinical vigilance and secondary screening,” J. Med. Screen., vol. 10, no. 1, pp. 2–4, Mar. 2003. [CrossRef]
- L. A. Pramono and A. Yuwono, “Late Diagnosis of Congenital Hypothyroidism in Young Adult.,” Acta Med. Indones., vol. 51, no. 3, pp. 272–274, Jul. 2019, [Online]. Available: http://www.ncbi.nlm.nih.gov/pubmed/31699952.
- L. Nagendra et al., “Etiological Profile, Targeted Levothyroxine Dosing and Impact of Partial Newborn Screening in Congenital Hypothyroidism—A Single Centre Experience,” Indian J. Endocrinol. Metab., vol. 27, no. 5, pp. 445–449, Sep. 2023. [CrossRef]
- G. A. De Jonge, “Congenital Hypothyroidism in the Netherlands,” Lancet, vol. 308, no. 7977, p. 143, Jul. 1976. [CrossRef]
- R. Wolter et al., “Neuropsychological study in treated thyroid dysgenesis,” Acta Paediatr., vol. 68, no. S277, pp. 41–46, Nov. 1979. [CrossRef]
- B. B. Jacobsen and N. J. Brandt, “Congenital hypothyroidism in Denmark.,” Arch. Dis. Child., vol. 56, no. 2, pp. 134–6, Feb. 1981. [CrossRef]
- O. F. Tarim and N. Yordam, “Congenital hypothyroidism in Turkey: a retrospective evaluation of 1000 cases.,” Turk. J. Pediatr., vol. 34, no. 4, pp. 197–202, 1992, doi:1306338.
- Nasheiti NA, “Childhood Hypothyroidism in Iraq: A Retrospective Study,” Int J Endocrinol Metab, no. 3, pp. 136–139, 2005, doi: 68232424.
- C. Y. Chen, K. T. Lee, C. T. C. Lee, W. Ter Lai, and Y. Bin Huang, “Epidemiology and clinical characteristics of congenital hypothyroidism in an Asian population: A nationwide population-based study,” J. Epidemiol., vol. 23, no. 2, pp. 85–94, 2013. [CrossRef]
- M. Deliana, J. R. Batubara, B. Tridjaja, and A. B. Pulungan, “Hipotiroidisme kongenital di Bagian Ilmu Kesehatan Anak RS Ciptomangunkusumo Jakarta, tahun 1992-2002,” Sari Pediatr., vol. 5, no. 2, p. 79, Dec. 2016. [CrossRef]
- M. Saoud, S. Al-Fahoum, and Y. Kabalan, “Congenital hypothyroidism: a five-year retrospective study at Children’s University Hospital, Damascus, Syria,” Qatar Med. J., vol. 2019, no. 1, Sep. 2019. [CrossRef]
- J. Fuyong, “An Analytical Study for Early Diagnosis and Treatment of Infants & Children Suffering From Congenital Hypothyroidism in China,” Int. J. Clin. Endocrinol. Metab., pp. 017–021, Jul. 2015. [CrossRef]
- G. Van Vliet and J. Deladoëy, “Diagnosis, Treatment and Outcome of Congenital Hypothyroidism,” Endocr. Dev., vol. 26, pp. 50–59, 2014. [CrossRef]
- H. Klein, S. Meltzer, and F. M. Kenny, “Improved prognosis in congenital bypothyroidism treated before age three months,” J. Pediatr., vol. 81, no. 5, pp. 912–915, Nov. 1972. [CrossRef]
- Gulshan, B. Tahmina, M. Fouzia, and R. Mizanur, “Neurodevelopmental outcome of congenital hypothyroidism in children between 1-5 years of age,” Bangladesh J. Med. Sci., vol. 10, no. 4, pp. 245–251, 2011. [CrossRef]
- T. John and D. Anirudhan, “Physical growth and intellectual function of children with congenital hypothyroidism: An observational study,” Sri Lanka J. Child Heal., vol. 52, no. 2, pp. 148–154, Jun. 2023. [CrossRef]
- B. Pulungan, M. E. Oldenkamp, A. S. P. Van Trotsenburg, W. Windarti, and H. Gunardi, “Effect of delayed diagnosis and treatment of congenital hypothyroidism on intelligence and quality of life: an observational study,” Med. J. Indones., vol. 28, no. 4, pp. 396–401, Dec. 2019. [CrossRef]
- J. Léger, B. Larroque, and J. Norton, “Influence of severity of congenital hypothyroidism and adequacy of treatment on school achievement in young adolescents: A population-based cohort study,” Acta Paediatr. Int. J. Paediatr., vol. 90, no. 11, pp. 1249–1256, 2001. [CrossRef]
- E. Kreisner, L. Schermann, E. Camargo-Neto, and J. L. Gross, “Predictors of intellectual outcome in a cohort of brazilian children with congenital hypothyroidism,” Clin. Endocrinol. (Oxf)., vol. 60, no. 2, pp. 250–255, Feb. 2004. [CrossRef]
- K. A. Selva, A. Harper, A. Downs, P. A. Blasco, and S. H. LaFranchi, “Neurodevelopmental Outcomes in Congenital Hypothyroidism: Comparison of Initial T4 Dose and Time to Reach Target T4 and TSH,” J. Pediatr., vol. 147, no. 6, pp. 775–780, Dec. 2005. [CrossRef]
- J. Léger, E. Ecosse, M. Roussey, J. L. Lanoë, and B. Larroque, “Subtle Health Impairment and Socioeducational Attainment in Young Adult Patients with Congenital Hypothyroidism Diagnosed by Neonatal Screening: A Longitudinal Population-Based Cohort Study,” J. Clin. Endocrinol. Metab., vol. 96, no. 6, pp. 1771–1782, Jun. 2011. [CrossRef]
- P. van Trotsenburg et al., “Congenital Hypothyroidism: A 2020–2021 Consensus Guidelines Update—An ENDO-European Reference Network Initiative Endorsed by the European Society for Pediatric Endocrinology and the European Society for Endocrinology,” Thyroid, vol. 31, no. 3, pp. 387–419, Mar. 2021. [CrossRef]
- N. Makretskaya et al., “High frequency of mutations in’dyshormonogenesis genes’ in severe congenital hypothyroidism,” PLoS One, vol. 13, no. 9, 2018. [CrossRef]
- J. H. Shin et al., “Genetic Evaluation of Congenital Hypothyroidism with Gland in situ Using Targeted Exome Sequencing,” Ann. Clin. Lab. Sci., vol. 51, no. 1, pp. 73–81, 2021, doi:33653783.
- L. Li et al., “Mutation screening of eight genes and comparison of the clinical data in a Chinese cohort with congenital hypothyroidism.” May 04, 2022. [CrossRef]
- R. J. Bruellman et al., “Increased prevalence of TG and TPO mutations in Sudanese children with congenital hypothyroidism,” J. Clin. Endocrinol. Metab., vol. 105, no. 5, 2020. [CrossRef]
- B. Albert et al., “Etiology of Increasing Incidence of Congenital Hypothyroidism in New Zealand from 1993–2010,” J. Clin. Endocrinol. Metab., vol. 97, no. 9, pp. 3155–3160, Sep. 2012. [CrossRef]
- S. Hall, A. Hutchesson, and J. Kirk, “Congenital hypothyroidism, seasonality and consanguinity in the West Midlands, England,” Acta Paediatr., vol. 88, no. 2, pp. 212–215, Feb. 1999. [CrossRef]
- S. Bundey and H. Alam, “A Five-Year Prospective Study of the Health of Children in Different Ethnic Groups, with Particular Reference to the Effect of Inbreeding,” Eur. J. Hum. Genet., vol. 1, no. 3, pp. 206–219, 1993. [CrossRef]
- J. Kumar, R. Gordillo, F. J. Kaskel, C. M. Druschel, and R. P. Woroniecki, “Increased Prevalence of Renal and Urinary Tract Anomalies in Children with Congenital Hypothyroidism,” J. Pediatr., vol. 154, no. 2, pp. 263–266, Feb. 2009. [CrossRef]
- E. Danner et al., “Comorbidity in Congenital Hypothyroidism—A Nationwide, Population-based Cohort Study,” J. Clin. Endocrinol. Metab., vol. 108, no. 12, pp. e1695–e1701, Nov. 2023. [CrossRef]
- J. Golbahar, H. Al-Khayyat, B. Hassan, W. Agab, E. Hassan, and A. Darwish, “Neonatal Screening for Congenital Hypothyroidism: A Retrospective Hospital Based Study from Bahrain,” J. Pediatr. Endocrinol. Metab., vol. 23, no. 1–2, Jan. 2010. [CrossRef]
- Benallègue and F Kedji, “Consanguinité et santé publique. Une étude algérienne,” Arch. Fr. Pediatr., vol. 41, no. 6, pp. 435–40, 1984, [Online]. Available: http://www.ncbi.nlm.nih.gov/pubmed/6487047.
- K. Nagasaki et al., “Re-Evaluation of the Prevalence of Permanent Congenital Hypothyroidism in Niigata, Japan: A Retrospective Study,” Int. J. Neonatal Screen., vol. 7, no. 2, p. 27, May 2021. [CrossRef]
- N. Matejek et al., “Predictors of transient congenital primary hypothyroidism: data from the German registry for congenital hypothyroidism (AQUAPE ‘HypoDok’),” Eur. J. Pediatr., vol. 180, no. 8, pp. 2401–2408, Aug. 2021. [CrossRef]
- N. Kanike, A. Davis, and P. S. Shekhawat, “Transient hypothyroidism in the newborn: To treat or not to treat,” Transl. Pediatr., vol. 6, no. 4, pp. 349–350, Oct. 2017. [CrossRef]
- E. J. Schoen, W. Clapp, T. T. To, and B. H. Fireman, “The key role of newborn thyroid scintigraphy with isotopic iodide ( 123I) in defining and managing congenital hypothyroidism,” Pediatrics, vol. 114, no. 6, pp. e683–e688, Dec. 2004. [CrossRef]
- J. Simoneau-Roy, S. Marti, C. Deal, C. Huot, P. Robaey, and G. Van Vliet, “Cognition and behavior at school entry in children with congenital hypothyroidism treated early with high-dose levothyroxine,” J. Pediatr., vol. 144, no. 6, pp. 747–752, Jun. 2004. [CrossRef]
- K. Rahmani et al., “Congenital Hypothyroidism: Optimal Initial Dosage and Time of Initiation of Treatment: A Systematic Review,” Int. J. Endocrinol. Metab., vol. 14, no. 3, Jun. 2016. [CrossRef]
- P. E. Aleksander et al., “Mean High-Dose l-Thyroxine Treatment Is Efficient and Safe to Achieve a Normal IQ in Young Adult Patients With Congenital Hypothyroidism,” J. Clin. Endocrinol. Metab., vol. 103, no. 4, pp. 1459–1469, Apr. 2018. [CrossRef]
- S. R. Rose et al., “Congenital Hypothyroidism: Screening and Management,” Pediatrics, vol. 151, no. 1, Jan. 2023. [CrossRef]
- Esposito et al., “Effect of initial levothyroxine dose on neurodevelopmental and growth outcomes in children with congenital hypothyroidism,” Front. Endocrinol. (Lausanne)., vol. 13, Sep. 2022. [CrossRef]
- M. C. Vigone et al., “Treatment of congenital hypothyroidism: comparison between L-thyroxine oral solution and tablet formulations up to 3 years of age,” Eur. J. Endocrinol., vol. 186, no. 1, pp. 45–52, Jan. 2022. [CrossRef]
- T. Vulsma, M. H. Gons, and J. J. M. de Vijlder, “Maternal-Fetal Transfer of Thyroxine in Congenital Hypothyroidism Due to a Total Organification Defect or Thyroid Agenesis,” N. Engl. J. Med., vol. 321, no. 1, pp. 13–16, Jul. 1989. [CrossRef]
- S. LaFranchi, “Congenital hypothyroidism: A newborn screening success story?,” Endocrinologist, vol. 4, no. 6, pp. 477–486, 1994. [CrossRef]
- N. E. C. H. Collaborative, “Neonatal hypothyroidism screening: Status of patients at 6 years of age,” J. Pediatr., vol. 107, no. 6, pp. 915–919, Dec. 1985. [CrossRef]
- S. Bargagna et al., “School attainments in children with congenital hypothyroidism detected by neonatal screening and treated early in life,” Eur. J. Endocrinol., vol. 140, no. 5, pp. 407–413, 1999. [CrossRef]
- J. A. Hulse, “Outcome for congenital hypothyroidism.,” Arch. Dis. Child., vol. 59, no. 1, pp. 23–29, Jan. 1984. [CrossRef]
- J. Rovet, R. Ehrlich, and D. Sorbara, “Intellectual outcome in children with fetal hypothyroidism,” J. Pediatr., vol. 110, no. 5, pp. 700–704, 1987. [CrossRef]
- G. H. Murphy, J. A. Hulse, I. Smith, and D. B. Grant, “Congenital Hypothyroidism: Physiological and Psychological Factors in Early Development,” J. Child Psychol. Psychiatry, vol. 31, no. 5, pp. 711–725, 1990. [CrossRef]
- P. W. Fuggle, D. B. Grant, I. Smith, and G. Murphy, “Intelligence, motor skills and behaviour at 5 years in early-treated congenital hypothyroidism,” Eur. J. Pediatr., vol. 150, no. 8, pp. 570–574, 1991. [CrossRef]
- J. Glorieux, J. Dussault, and G. Van Vliet, “Intellectual development at age 12 years of children with congenital hypothyroidism diagnosed by neonatal screening,” J. Pediatr., vol. 121, no. 4, pp. 581–584, 1992. [CrossRef]
- S. Bargagna et al., “Neuropsychological development in a child with early-treated congenital hypothyroidism as compared with her unaffected identical twin,” Eur. J. Endocrinol., vol. 136, no. 1, pp. 100–104, 1997. [CrossRef]
- W. F. Simons, P. W. Fuggle, D. B. Grant, and I. Smith, “Intellectual development at 10 years in early treated congenital hypothyroidism.,” Arch. Dis. Child., vol. 71, no. 3, pp. 232–234, Sep. 1994. [CrossRef]
- Hauri-Hohl, ; et al., “Impaired Neuromotor Outcome in School-Age Children With Congenital Hypothyroidism Receiving Early High-Dose Substitution Treatment,” Pediatr. Res., vol. 70, no. 6, pp. 614–618, Dec. 2011. [CrossRef]
- W. F. Simons, P. W. Fuggle, D. B. Grant, and I. Smith, “Educational progress, behaviour, and motor skills at 10 years in early treated congenital hypothyroidism,” Arch. Dis. Child., vol. 77, no. 3, pp. 219–222, 1997. [CrossRef]
- M. A. Romero et al., “Analysis of motor, cognitive and language performance of infants undergoing treatment for congenital hypothyroidism,” J. Pediatr. (Rio. J)., vol. 101, no. 2, pp. 172–178, Mar. 2025. [CrossRef]
- M. Habib, H. Abou faddan, K. Metwalley, and T. Ismail, “Growth and Developmental Milestones in children with Congenital Hypothyroidism Attending Assiut Health Insurance Clinic.,” Egypt. J. Community Med., pp. 0–0, Mar. 2022. [CrossRef]
- J. F. Rovet, “Congenital hypothyroidism: Long-term outcome,” Thyroid, vol. 9, no. 7, pp. 741–748, Jul. 1999. [CrossRef]
- J. Léger, “Congenital hypothyroidism: A clinical update of long-term outcome in young adults,” Eur. J. Endocrinol., vol. 172, no. 2, pp. R67–R77, Feb. 2015. [CrossRef]
- W. Smith, A. M. Klein, J. R. Henderson, and N. C. Myrianthopoulos, “Congenital hypothyroidism-signs and symptoms in the newborn period,” J. Pediatr., vol. 87, no. 6 PART 1, pp. 958–962, 1975. [CrossRef]
- N. Ahmad, A. Irfan, and S. Al Saedi, “Congenital hypothyroidism: Screening, diagnosis, management, and outcome,” J. Clin. Neonatol., vol. 6, no. 2, p. 64, 2017. [CrossRef]
- H. Hannon, B. Therrell, and W. H. O. H. D. Programme, “Guidelines on the prevention and control of congenital hypothyroidism / prepared by Harry Hannon, Brad Therrell.” World Health Organization, p. ONLINE, 1990.
| Family history | ||
| Consanguinity | N(%) | 75 (26%) |
| Family history of CH in a sibling | N(%) | 27 (9%) |
| Dysgenesis | ||
| Athyreosis Ectopy Hypoplasia Total dysgenesis |
N(%) N(%) N(%) N(%) |
52 (18%) 17 (6%) 40 (14%) 109 (38%) |
| All Gland in situ | N(%) | 125 (43%) |
| Goitre | N(%) | 25 (9%) |
| Undetermined | N(%) | 29 (10%) |
| Associated abnormalities | N(%) | 44 (15%) |
| Heart defects | N(%) | 23 (8%) |
| Renal defects | N(%) | 9 (3%) |
| Biological data | ||
| TSH mU/l | Median [range] | 65.35 [8.12 -7100] |
| FT4 pmol/l | Mean ± SDS [range] | 7.6±6.2[0.01-27] |
| TSH >100 mU/l | N,% | 125 (43.4%) |
| FT4 <5 pmol/l | N,% | 102 (35.4%) |
| Global CH cohort N=288 |
In situ gland/Goitre N=150 |
Undetermined N=29 |
Ectopy/Hypoplasia N=57 |
Athyreosis N=52 |
p | |
|---|---|---|---|---|---|---|
| Age at diagnosis (months) Median (range) |
2 (0,1-375) |
1.5 (0,05-51) |
0.86 (0.13-32) |
4 (0,07-155) |
2 (0,06-150) |
0.001* |
| M/F Sex ratio |
129/159 0.81 |
80/69 1.2 |
15/15 1 |
24/33 0.7 |
10/42 0.24 |
0.0003 |
| Consanguinity %(N) | 26% (75) | 31% (47) | 27% (8) | 23% (13) | 13.5% (7) | 0.078 |
| TSH mU/ml Median (range) |
65.3 (8.1-7100) |
50 (8.1-4344) |
47.5 (10-1170) |
96.3 (10.1-572.6) |
100 (17.9-7100) |
0.034* |
| TSH>100 mU/L %(N) | 43% (125) | 35% (53) | 41% (12) | 39% (22) | 73% (38) | <0.0001 |
| FT4 (pmol/L) Mean ±SD [range] |
7.6±6.4 [0.01-25.4] |
8.8±6.1 [0.01-25.7] |
8.8±2.3 [0-25.4] |
7.2±5.5 [0.02-27] |
2.6±2.7 [0.01-10.6] |
0.003 |
| FT4< 5 pmol/L %,(N) | 35% (102) | 26% (39) | 38% (11) | 37% (21) | 60% (31) | 0.0002 |
| Transient CH N=24 | Permanent CH N=23 | p | |
|---|---|---|---|
| Sex ratio M/F | 12/12 | 8/15 | 0.297 |
| Age at diagnosis in Months Median (range) | 4,3 (0,16-46) | 2.4 (0,1-12) | 0.440 |
| TSH mU/l Mean (range) | 222,7 (4,3-100) | 239,1 (7,6-1000) | 0.947 |
| TSH<10 mU/l, N(%) | 4(17%) | 0(0%) | 0.037 |
| FT4 pmol/L Mean (range) | 13,4 (0,1-17,2) | 7,5 (0,1-18,1) | 0.003 |
| FT4<10 pmol/L N, (%) | 5(21%) | 14(61%) | 0.011 |
| In Situ gland, N (%) | 23 (96%) | 10(43%) | 0.001 |
| Dysgenesis, N(%) | 1(5%) | 13(57%) |
| Reference (Original study) |
Country |
Period | Age at Diagnosis | % Diagnosed in Neonatal Period | Late Diagnosis Rates |
|---|---|---|---|---|---|
| Raiti 1971[11] | UK | – | – | 6%<1month 22%<3months |
16% <6months 55% by 2 year |
| De Jonge 1976[17] | Netherland | 1972-1974 | – | 10% <1month | 50% at 3months |
| Alm 1978 [12] | Sweden | 1969–1975 | – | 20% | 52% after 3 months |
| Wolter 1979[18] | Belgium | – | 3-6 months | 7%<1month 46% <3months |
21% >1year |
| Jacobsen 1981 [19] | Denmark | 1970-1975 | – | 10% (1st month) | 70% by 1 year |
| Tarim 1992 [20] | Turkey | 1964-1989 | 49 months | 3.1% | 55.4% after 2 years |
| Nasheiti 2005 [21] | Iraq | 1993-2003 | 2.3 years | 25% | 75% beyond neonatal period |
| Chen 2013 [22] | South Asian countries | 1997–2008 | 12 months | Taiwan 56% < 3 months | 22% > 1years Pakistan, India 70% > 1 year |
| Deliana 2017[23] | Indonesia | 1992-2002 | – | Minimal | 53% at 1–5 years 6.7% after 12 years |
| Saoud 2019[24] | Syria | 2008-2012 | 8 months | >25% (1st month) | 75% beyond neonatal period |
| Fuyong 2015[25] | China | - | 18-24 months | - | 55.2 %<6months 7.9 % 6 months- 2 yr 36.9 %, 3 -7 yr |
|
Van Vliet With NBS [26] |
With NBS | - | Within 2-4 weeks | >95% | Rare |
| Our study | Algeria | 2005-2023 | 1.6 months | 35% (<1 month) | 65% ≥1 month |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




