Submitted:
29 May 2025
Posted:
30 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Cells.
2.2. Apoptosis and Cell Death Analysis.
2.3. Caspase-3 Activity Assay.
2.4. Preparation of Poly (lactide-co-glycolic acid) (PLGA) Nanoparticles (NPs).
2.5. Preparation of Doxorubicin Loaded NPs
2.6. In-Vivo DLBCL Subcutaneous Xenograft Model.
2.7. Statistical Analysis.
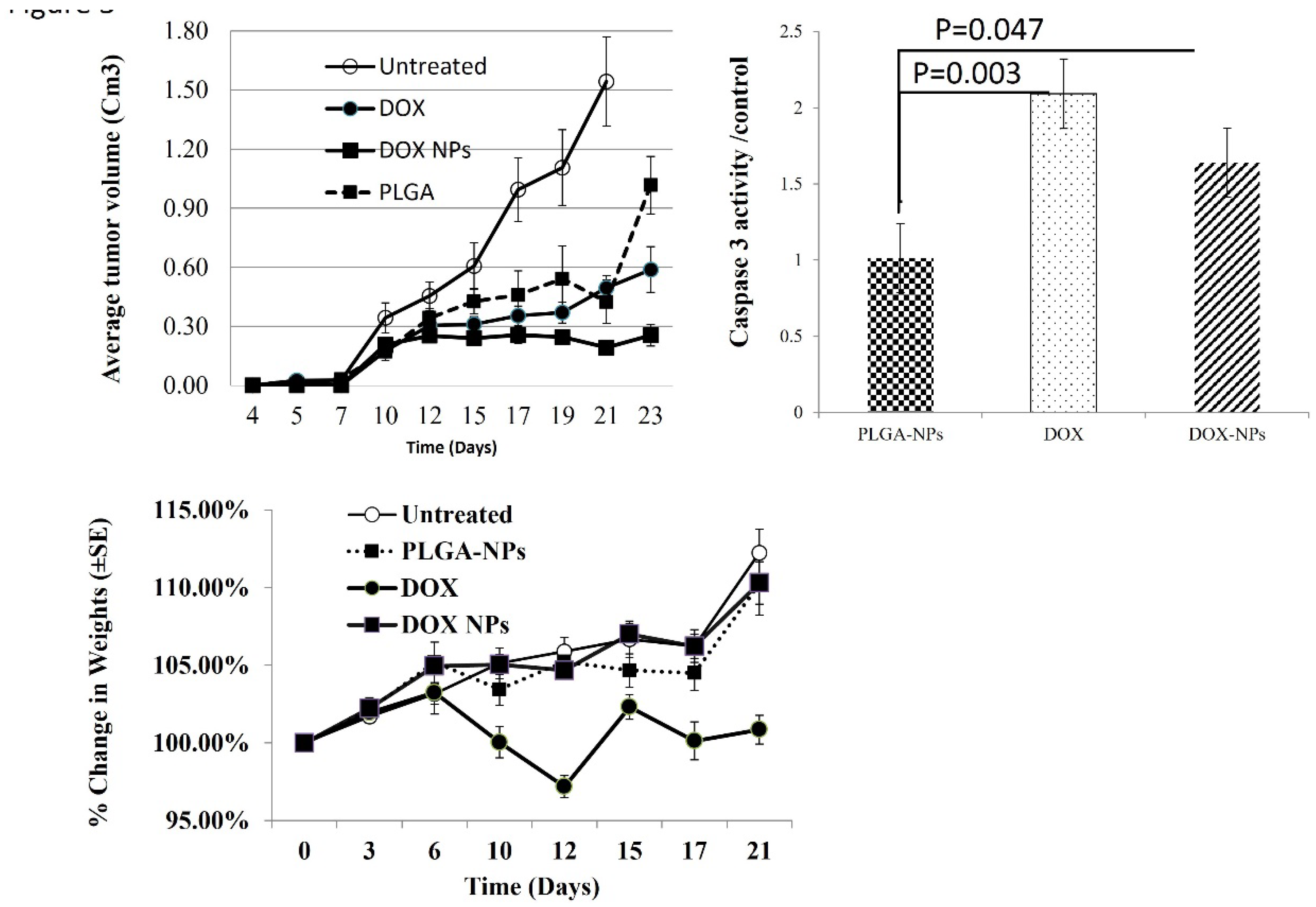
3. Results
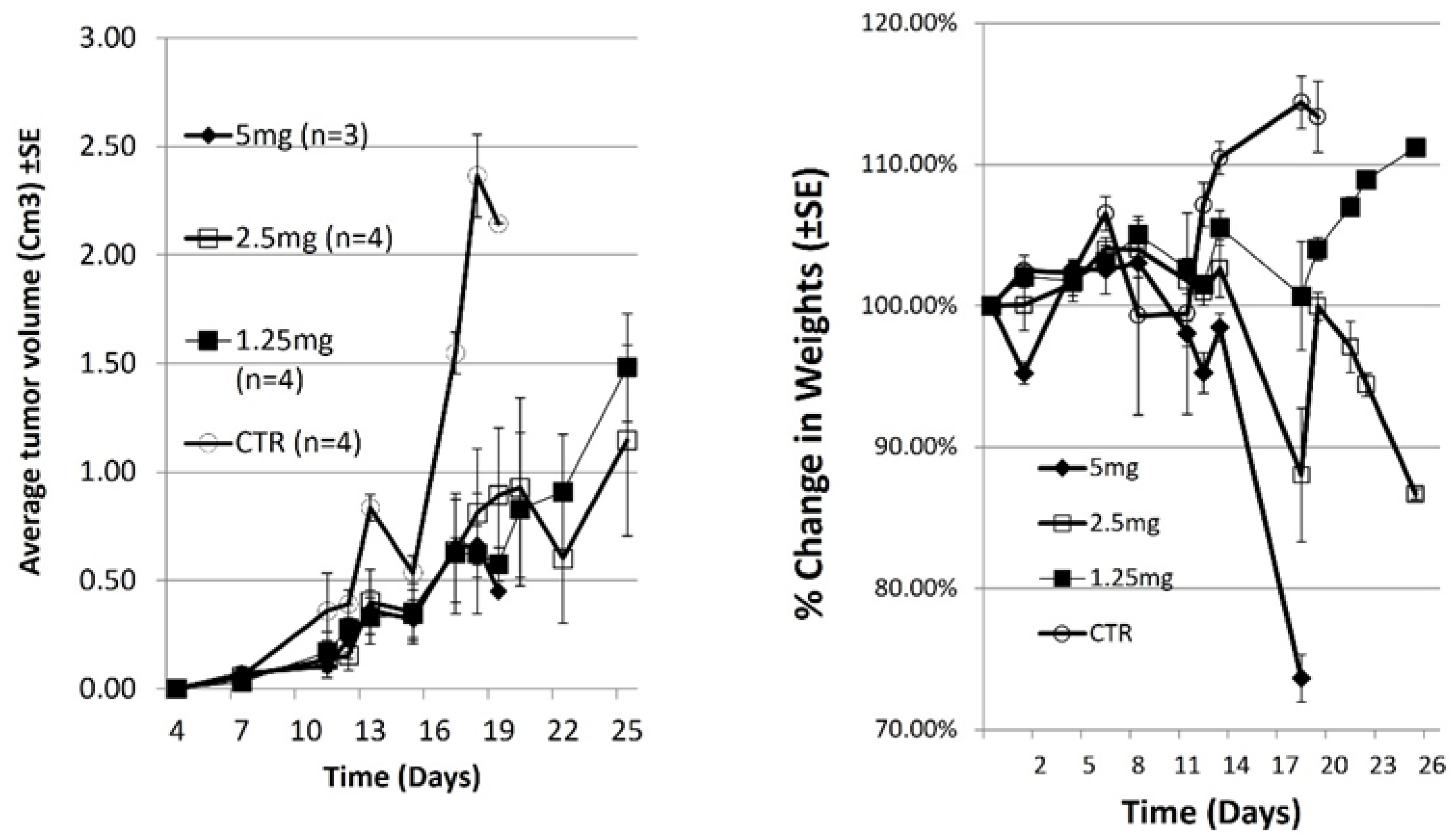
3.1. In Vivo Anticancer Activity and Cytotoxic Effect of Dox
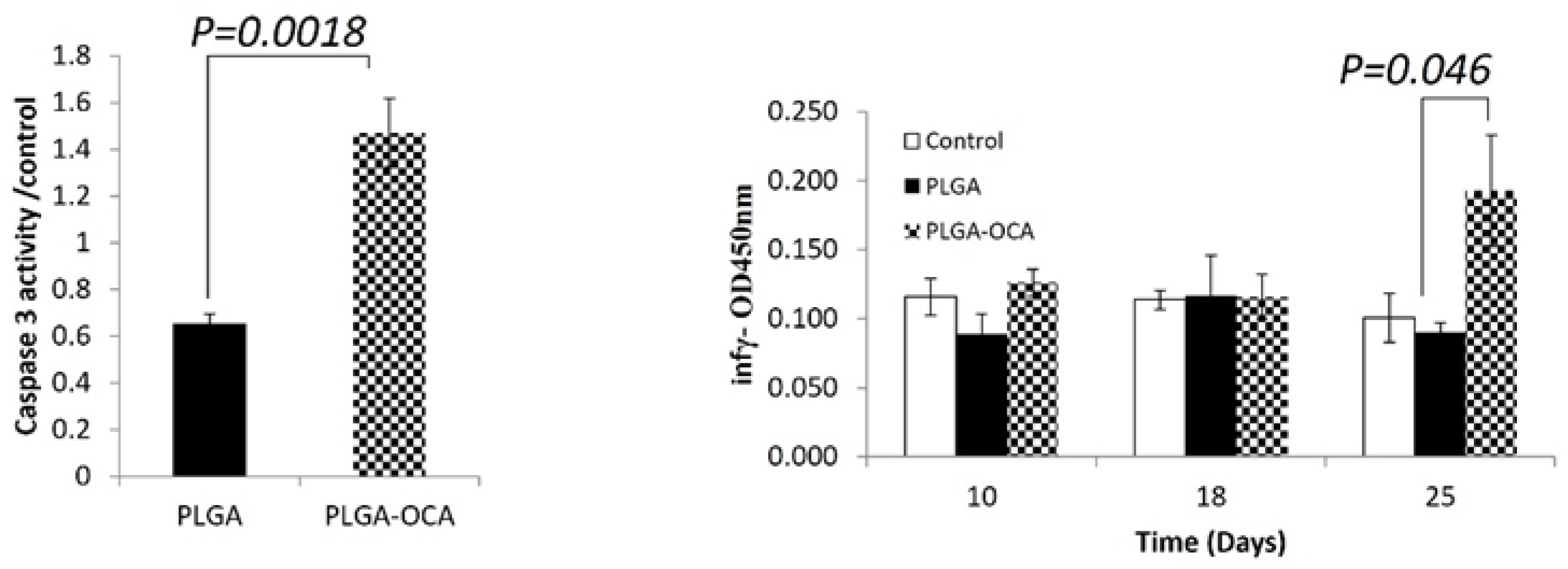
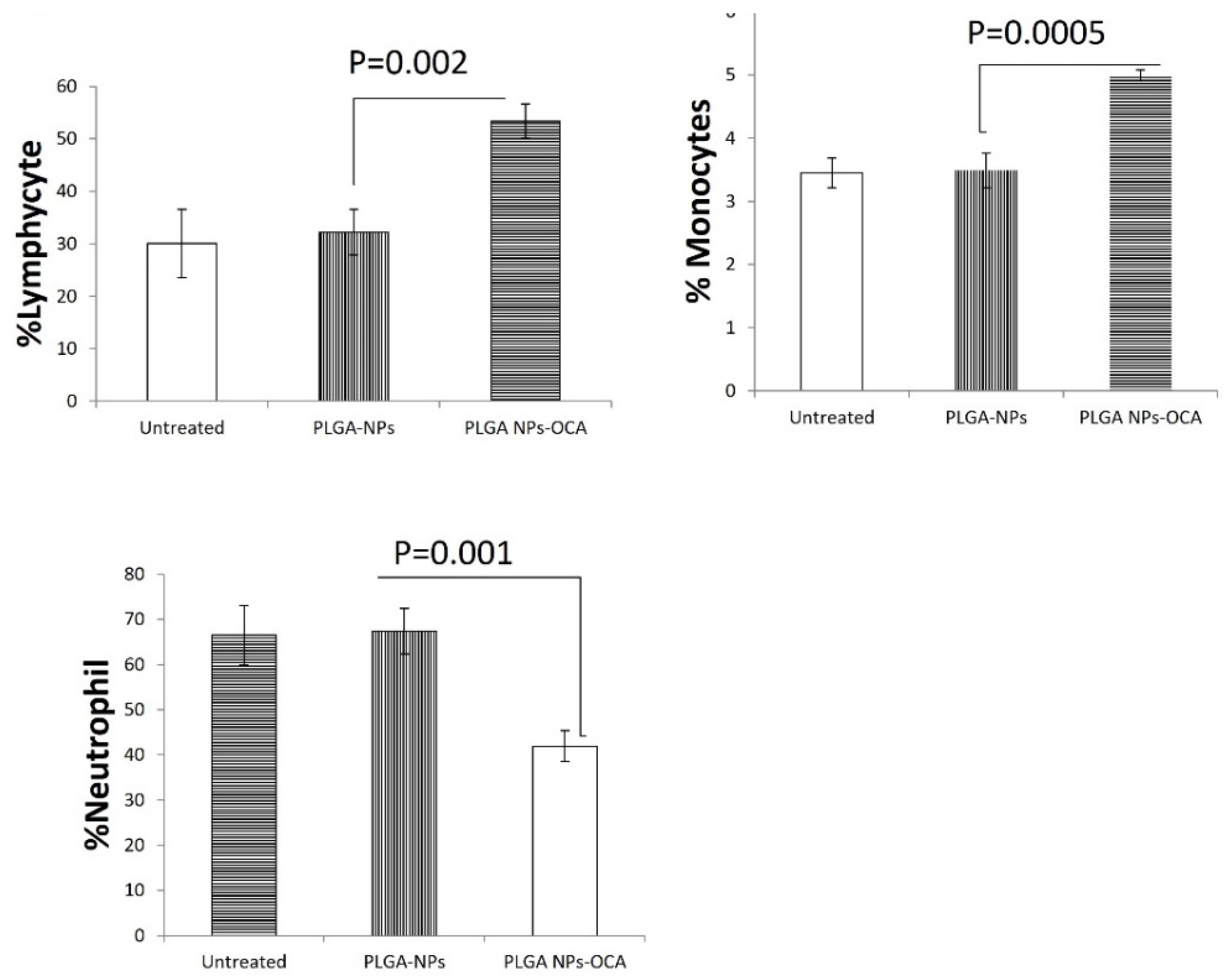
3.1. PLGA–OCA Enhance Immunity and Tumor Cell Death.
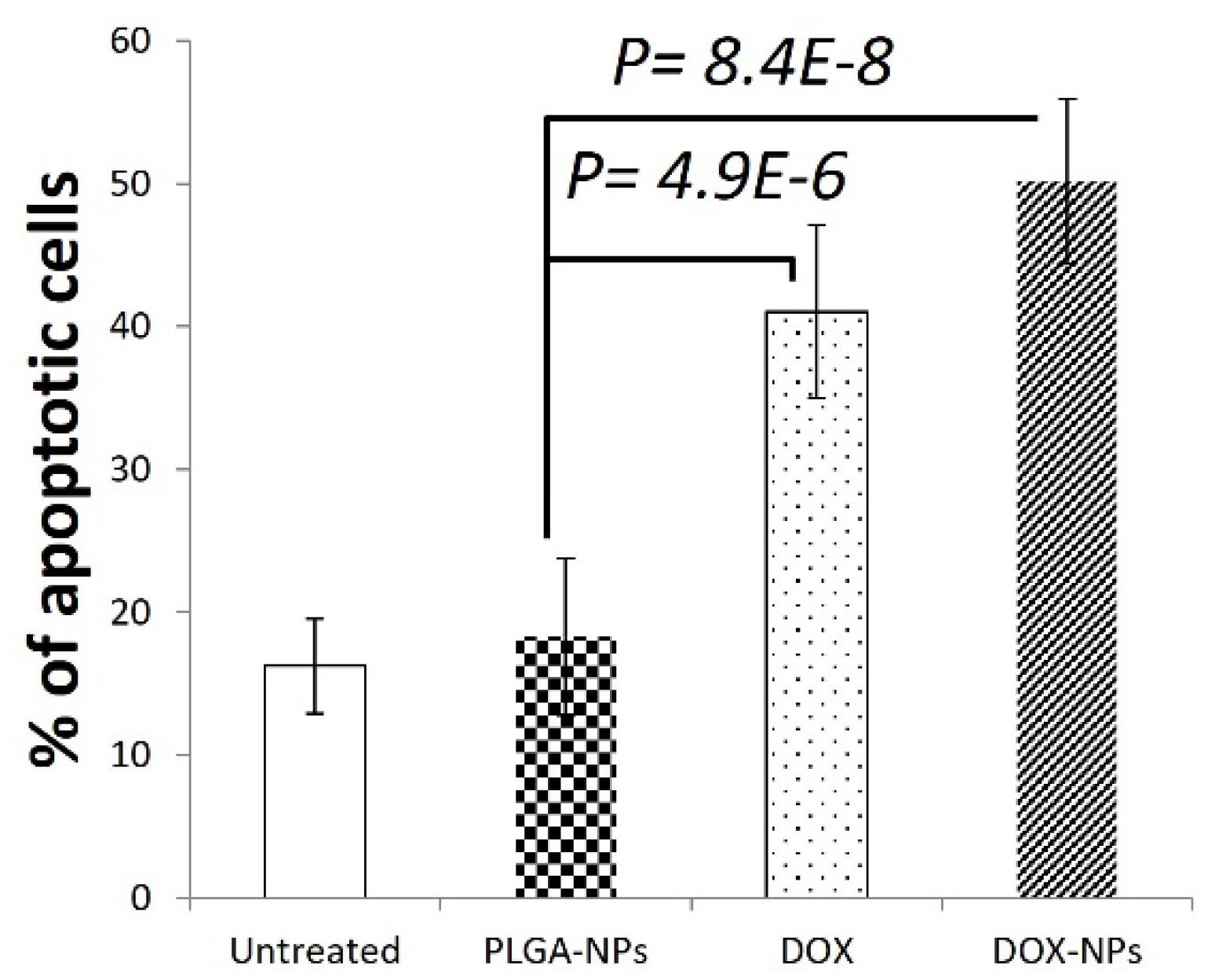
3.2. DOX-NPs Induce Cell Death In-Vitro and In-Vivo.
4. Discussion
Acknowledgments
Conflicts of Interest
References
- Foo, J.; Michor, F. Evolution of acquired resistance to anti-cancer therapy. Journal of theoretical biology 2014, 355, 10–20. [Google Scholar] [CrossRef] [PubMed]
- Girotti, M.R.; Marais, R. Deja Vu: EGF receptors drive resistance to BRAF inhibitors. Cancer discovery 2013, 3, 487–490. [Google Scholar] [CrossRef]
- Chavez, J.C.; Bachmeier, C.; Kharfan-Dabaja, M.A. CAR T-cell therapy for B-cell lymphomas: clinical trial results of available products. Therapeutic advances in hematology 2019, 10, 2040620719841581. [Google Scholar] [CrossRef] [PubMed]
- Chatterjee, K.; Zhang, J.; Honbo, N.; Karliner, J.S. Doxorubicin cardiomyopathy. Cardiology 2010, 115, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Denard, B.; Lee, C.; Ye, J. Doxorubicin blocks proliferation of cancer cells through proteolytic activation of CREB3L1. eLife 2012, 1, e00090. [Google Scholar] [CrossRef]
- Denard, B.; Pavia-Jimenez, A.; Chen, W.; Williams, N.S.; Naina, H.; Collins, R.; Brugarolas, J.; Ye, J. Identification of CREB3L1 as a Biomarker Predicting Doxorubicin Treatment Outcome. PLoS One 2015, 10, e0129233. [Google Scholar] [CrossRef] [PubMed]
- Lüpertz, R.; Wätjen, W.; Kahl, R.; Chovolou, Y. Dose- and time-dependent effects of doxorubicin on cytotoxicity, cell cycle and apoptotic cell death in human colon cancer cells. Toxicology 2010, 271, 115–121. [Google Scholar] [CrossRef]
- Singal, P.K.; Iliskovic, N. Doxorubicin-induced cardiomyopathy. N Engl J Med 1998, 339, 900–905. [Google Scholar] [CrossRef]
- Carvalho, C.; Santos, R.X.; Cardoso, S.; Correia, S.; Oliveira, P.J.; Santos, M.S.; Moreira, P.I. Doxorubicin: the good, the bad and the ugly effect. Curr Med Chem 2009, 16, 3267–3285. [Google Scholar] [CrossRef]
- Rafiyath, S.M.; Rasul, M.; Lee, B.; Wei, G.; Lamba, G.; Liu, D. Comparison of safety and toxicity of liposomal doxorubicin vs. conventional anthracyclines: a meta-analysis. Experimental hematology & oncology 2012, 1, 10. [Google Scholar]
- Lu, Y.; Chen, S.C. Micro and nano-fabrication of biodegradable polymers for drug delivery. Advanced drug delivery reviews 2004, 56, 1621–1633. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR effect: Unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Advanced drug delivery reviews 2011, 63, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. The enhanced permeability and retention (EPR) effect in tumor vasculature: the key role of tumor-selective macromolecular drug targeting. Advances in enzyme regulation 2001, 41, 189–207. [Google Scholar] [CrossRef] [PubMed]
- Alizadeh, D.; Trad, M.; Hanke, N.T.; Larmonier, C.B.; Janikashvili, N.; Bonnotte, B.; Katsanis, E.; Larmonier, N. Doxorubicin eliminates myeloid-derived suppressor cells and enhances the efficacy of adoptive T-cell transfer in breast cancer. Cancer Res 2014, 74, 104–118. [Google Scholar] [CrossRef]
- Abd-Elrahman, I.; Nassar, T.; Khairi, N.; Perlman, R.; Benita, S.; Ben Yehuda, D. Novel targeted mtLivin nanoparticles treatment for disseminated diffuse large B-cell lymphoma. Oncogene 2021, 40, 334–344. [Google Scholar] [CrossRef]
- Harush-Frenkel, O.; Bivas-Benita, M.; Nassar, T.; Springer, C.; Sherman, Y.; Avital, A.; Altschuler, Y.; Borlak, J.; Benita, S. A safety and tolerability study of differently-charged nanoparticles for local pulmonary drug delivery. Toxicology and applied pharmacology 2010, 246, 83–90. [Google Scholar] [CrossRef]
- Karra, N.; Nassar, T.; Ripin, A.N.; Schwob, O.; Borlak, J.; Benita, S. Antibody conjugated PLGA nanoparticles for targeted delivery of paclitaxel palmitate: efficacy and biofate in a lung cancer mouse model. Small 2013, 9, 4221–4236. [Google Scholar] [CrossRef]
- Carreno, B.M.; Garbow, J.R.; Kolar, G.R.; Jackson, E.N.; Engelbach, J.A.; Becker-Hapak, M.; Carayannopoulos, L.N.; Piwnica-Worms, D.; Linette, G.P. Immunodeficient mouse strains display marked variability in growth of human melanoma lung metastases. Clin Cancer Res 2009, 15, 3277–3286. [Google Scholar] [CrossRef]
- Coiffier, B.; Thieblemont, C.; Van Den Neste, E.; Lepeu, G.; Plantier, I.; Castaigne, S.; Lefort, S.; Marit, G.; Macro, M.; Sebban, C.; :, *!!! REPLACE !!!*; et al. : Long-term outcome of patients in the LNH-98.5 trial, the first randomized study comparing rituximab-CHOP to standard CHOP chemotherapy in DLBCL patients: a study by the Groupe d'Etudes des Lymphomes de l'Adulte. Blood 2010, 116, 2040–2045. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Locke, F.L.; Bartlett, N.L.; Lekakis, L.J.; Miklos, D.B.; Jacobson, C.A.; Braunschweig, I.; Oluwole, O.O.; Siddiqi, T.; Lin, Y.; et al. Axicabtagene Ciloleucel CAR T-Cell Therapy in Refractory Large B-Cell Lymphoma. N Engl J Med 2017, 377, 2531–2544. [Google Scholar] [CrossRef]
- Schuster, S.J.; Svoboda, J.; Chong, E.A.; Nasta, S.D.; Mato, A.R.; Anak, Ö.; Brogdon, J.L.; Pruteanu-Malinici, I.; Bhoj, V.; Landsburg, D.; et al. Chimeric Antigen Receptor T Cells in Refractory B-Cell Lymphomas. N Engl J Med 2017, 377, 2545–2554. [Google Scholar] [CrossRef] [PubMed]
- Sehn, L.H.; Berry, B.; Chhanabhai, M.; Fitzgerald, C.; Gill, K.; Hoskins, P.; Klasa, R.; Savage, K.J.; Shenkier, T.; Sutherland, J.; et al. The revised International Prognostic Index (R-IPI) is a better predictor of outcome than the standard IPI for patients with diffuse large B-cell lymphoma treated with R-CHOP. Blood 2007, 109, 1857–1861. [Google Scholar] [CrossRef]
- Schipani, M.; Rivolta, G.M.; Margiotta-Casaluci, G.; Mahmoud, A.M.; Al Essa, W.; Gaidano, G.; Bruna, R. New Frontiers in Monoclonal Antibodies for Relapsed/Refractory Diffuse Large B-Cell Lymphoma. Cancers (Basel) 2023, 16. [Google Scholar] [CrossRef] [PubMed]
- Morton, L.M.; Wang, S.S.; Devesa, S.S.; Hartge, P.; Weisenburger, D.D.; Linet, M.S. Lymphoma incidence patterns by WHO subtype in the United States, 1992-2001. Blood 2006, 107, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Dorostkar, H.; Haghiralsadat, B.F.; Hemati, M.; Safari, F.; Hassanpour, A.; Naghib, S.M.; Roozbahani, M.H.; Mozafari, M.R.; Moradi, A. Reduction of Doxorubicin-Induced Cardiotoxicity by Co-Administration of Smart Liposomal Doxorubicin and Free Quercetin: In Vitro and In Vivo Studies. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef]
- Chehelgerdi, M.; Chehelgerdi, M.; Allela, O.Q.B.; Pecho, R.D.C.; Jayasankar, N.; Rao, D.P.; Thamaraikani, T.; Vasanthan, M.; Viktor, P.; Lakshmaiya, N.; et al. Progressing nanotechnology to improve targeted cancer treatment: overcoming hurdles in its clinical implementation. Mol Cancer 2023, 22, 169. [Google Scholar] [CrossRef]
- Sun, L.; Liu, H.; Ye, Y.; Lei, Y.; Islam, R.; Tan, S.; Tong, R.; Miao, Y.B.; Cai, L. Smart nanoparticles for cancer therapy. Signal transduction and targeted therapy 2023, 8, 418. [Google Scholar] [CrossRef]
- Aryal, S.; Park, S.; Park, H.; Park, C.; Kim, W.C.; Thakur, D.; Won, Y.J.; Key, J. Clinical Trials for Oral, Inhaled and Intravenous Drug Delivery System for Lung Cancer and Emerging Nanomedicine-Based Approaches. International journal of nanomedicine 2023, 18, 7865–7888. [Google Scholar] [CrossRef]
- Liu, W.Y.; Hsieh, Y.S.; Wu, Y.T. Poly (Lactic-Co-Glycolic) Acid-Poly (Vinyl Pyrrolidone) Hybrid Nanoparticles to Improve the Efficiency of Oral Delivery of β-Carotene. Pharmaceutics 2022, 14. [Google Scholar] [CrossRef]
- Jia, J.; Xiong, Z.A.; Qin, Q.; Yao, C.G.; Zhao, X.Z. Picosecond pulsed electric fields induce apoptosis in a cervical cancer xenograft. Molecular medicine reports 2015, 11, 1623–1628. [Google Scholar] [CrossRef]
- Fessi, H.; Puisieux, F.; Devissaguet, J.P.; Ammoury, N.; Benita, S. NANOCAPSULE FORMATION BY INTERFACIAL POLYMER DEPOSITION FOLLOWING SOLVENT DISPLACEMENT. International Journal of Pharmaceutics 1989, 55, R1–R4. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



