Submitted:
30 May 2025
Posted:
30 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Overview of Monocytes
2.1. Monocyte Development and Differentiation
2.2. Monocyte Heterogeneity and Its Homeostasis
2.3. Phenotypic Features of Monocytes
2.4. Functional Features of Monocytes
2.4.1. Cytokine Secretion
2.4.2. Differentiation Potential
2.4.3. Antigen Presentation
2.4.4. Migration and Extravasation
2.5. Transcriptional Features of Monocytes
3. Monocyte Characteristics in MPNs
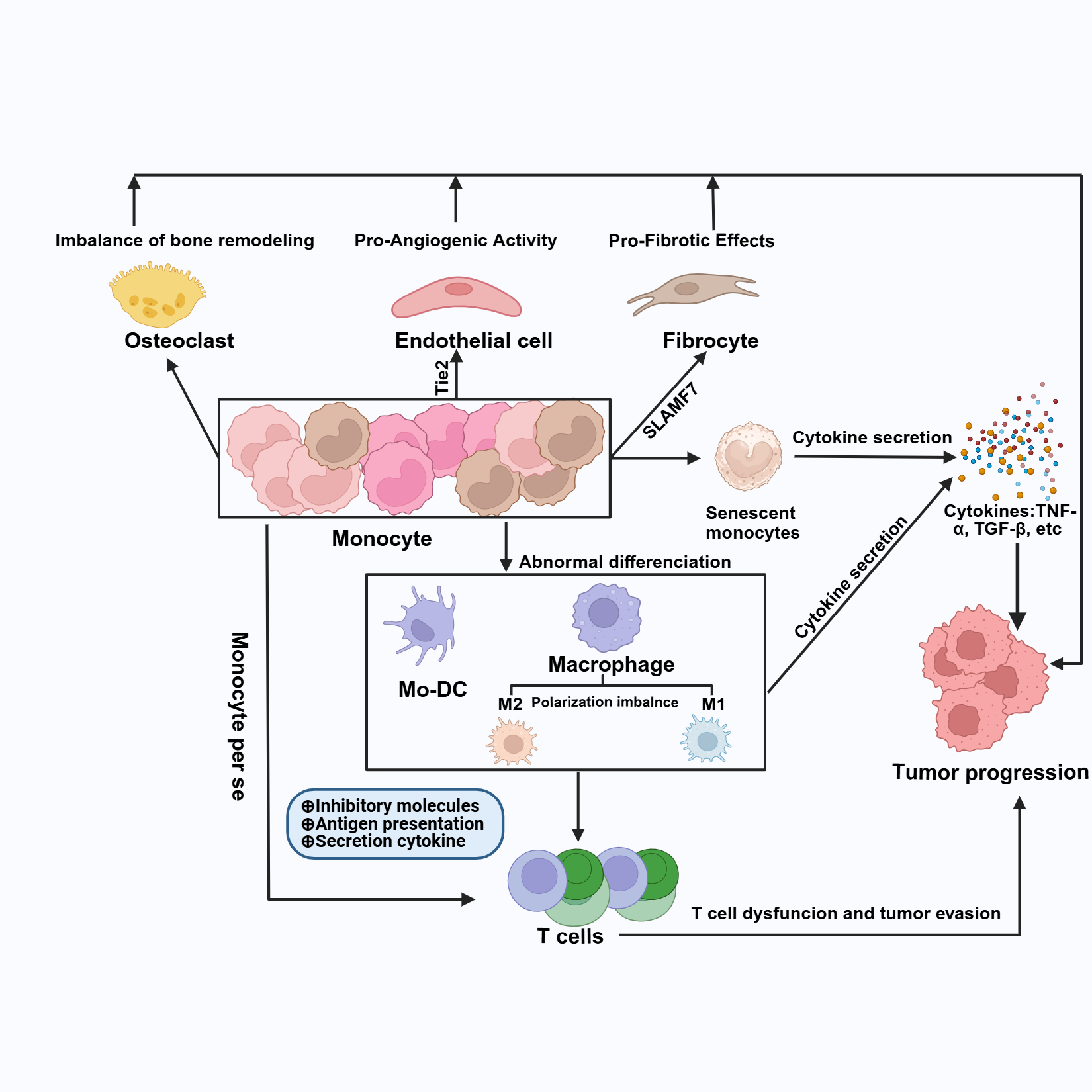
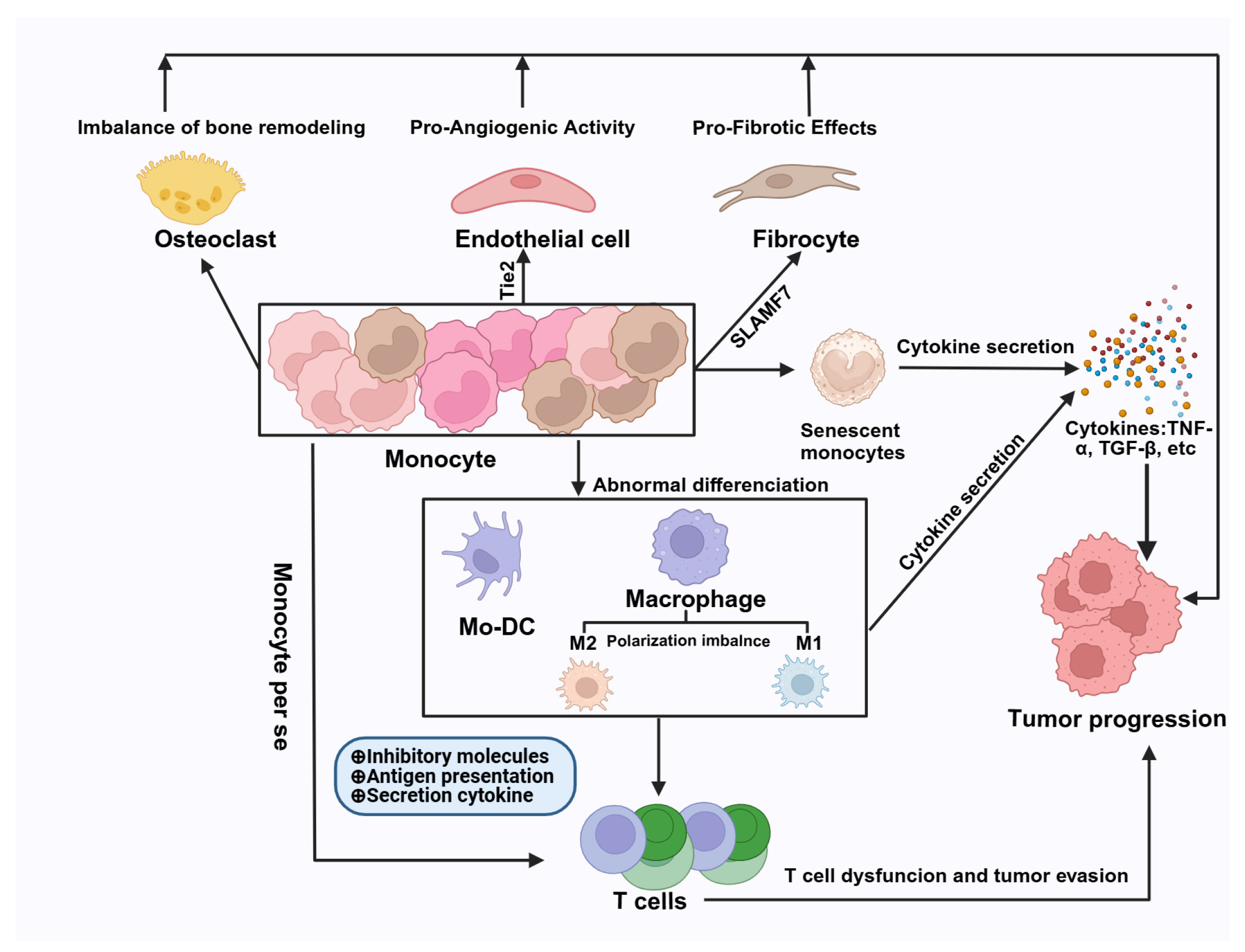
4. Mechanism of Monocyte Involvement in MPN Progression
- Pro-Aiogenic Activity: Monocytes may exhibit pro-angiogenic properties by expressing Tie2 or even differentiating into endothelial cells, thereby promoting vascularization and tumor progression [52,53].
- Pro-Fibrotic Effects: Through the expression of SLAMF7 and differentiation into fibrocytes, monocytes may contribute to fibrotic processes within the bone marrow microenvironment [54].
- Pro-Tumoral Effects: Monocytes can express programmed death-ligand 1 (PD-L1), which directly inhibits T cell function, thereby facilitating tumor progression. This immune evasion mechanism allows tumors to escape immune surveillance [55,56].
- Cytokine Secretion: An imbalance in monocyte subsets can lead to abnormal cytokine secretion, particularly an excess of TNF-α. This cytokine is known to promote MPN progression. Additionally, intrinsic disturbances in monocyte function may result in abnormal interactions with T cells, further contributing to tumor growth [57].
- Differentiation Potential: Monocytes present the ability to differentiate into various cell types, including DCs, macrophages. These differentiated cells can interact with T cells, potentially inhibiting their functions and facilitating tumor evasion [48].
- Osteoclast Differentiation: Monocytes can also differentiate into osteoclasts, which may play a significant role in the progression of MPNs by affecting the bone marrow microenvironment [58].
- Senescence and Dysfunction: Monocytes may enter a senescent state, leading to mitochondrial dysfunction. This impairment can result in abnormal migration, antigen presentation, and overall immune response, further complicating their role in MPNs [59,60].
4.1. Pro-Angiogenesis via Expressing Tie2
4.2. Pro-Fibrosis via Expressing SLAMF7
4.3. Protumoral Effect via Expressing PD-L1
4.4. Tumorigenic with Abnormal Cytokine Secretion
4.5. Over-Differentiation Towards Osteoclasts
4.6. Abnormal Differentiation Towards Macrophages
4.7. Abnormal Differentiation Towards DCs
4.8. Effect of Monocytes Per Se
4.9. Potential Treatments Targeting Monocytes for MPNs
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Arber DA, Orazi A, Hasserjian RP, Borowitz MJ, Calvo KR, Kvasnicka HM, Wang SA, Bagg A, Barbui T, Branford S, Bueso-Ramos CE, Cortes JE, Dal Cin P, DiNardo CD, Dombret H, Duncavage EJ, Ebert BL, Estey EH, Facchetti F, Foucar K, Gangat N, Gianelli U, Godley LA, Gökbuget N, Gotlib J, Hellström-Lindberg E, Hobbs GS, Hoffman R, Jabbour EJ, Kiladjian JJ, Larson RA, Le Beau MM, Loh ML, Löwenberg B, Macintyre E, Malcovati L, Mullighan CG, Niemeyer C, Odenike OM, Ogawa S, Orfao A, Papaemmanuil E, Passamonti F, Porkka K, Pui CH, Radich JP, Reiter A, Rozman M, Rudelius M, Savona MR, Schiffer CA, Schmitt-Graeff A, Shimamura A, Sierra J, Stock WA, Stone RM, Tallman MS, Thiele J, Tien HF, Tzankov A, Vannucchi AM, Vyas P, Wei AH, Weinberg OK, Wierzbowska A, Cazzola M, Döhner H, Tefferi A. International Consensus Classification of Myeloid Neoplasms and Acute Leukemias: integrating morphologic, clinical, and genomic data. Blood. 2022 Sep 15;140(11):1200-1228. [CrossRef]
- Suttantapidok S, Owattanapanich W. Clinical Characteristics, Prognostic Factors, and Thrombotic and Bleeding Outcomes in Philadelphia Chromosome-Negative Myeloproliferative Neoplasms: A Single-Center Cohort Study in Thailand. Cureus. 2025 Apr 12;17(4):e82141. [CrossRef]
- Fisher DAC, Fowles JS, Zhou A, Oh ST. Inflammatory Pathophysiology as a Contributor to Myeloproliferative Neoplasms. Front Immunol. 2021 Jun 1;12:683401. [CrossRef]
- Hermouet, S. Mutations, inflammation and phenotype of myeloproliferative neoplasms. Front Oncol. 2023 May 22;13:1196817. [CrossRef]
- Luque Paz D, Kralovics R, Skoda RC. Genetic basis and molecular profiling in myeloproliferative neoplasms. Blood. 2023 Apr 20;141(16):1909-1921. [CrossRef]
- Li B, Gale RP, Xu Z, Qin T, Song Z, Zhang P, Bai J, Zhang L, Zhang Y, Liu J, Huang G, Xiao Z. Non-driver mutations in myeloproliferative neoplasm-associated myelofibrosis. J Hematol Oncol. 2017 May 2;10(1):99. [CrossRef]
- Aguirre LE, Jain A, Ball S, Ali NA, Volpe VO, Tinsley-Vance S, Sallman D, Sweet K, Lancet J, Padron E, Yun S, Kuykendall A, Komrokji R. Triple-Negative Myelofibrosis: Disease Features, Response to Treatment and Outcomes. Clin Lymphoma Myeloma Leuk. 2024 Jul;24(7):459-467. [CrossRef]
- Gleitz HFE, Benabid A, Schneider RK. Still a burning question: the interplay between inflammation and fibrosis in myeloproliferative neoplasms. Curr Opin Hematol. 2021 Sep 1;28(5):364-371. [CrossRef]
- Cominal JG, Cacemiro MDC, Berzoti-Coelho MG, Pereira IEG, Frantz FG, Souto EX, Covas DT, de Figueiredo-Pontes LL, Oliveira MC, Malmegrim KCR, de Castro FA. Bone Marrow Soluble Mediator Signatures of Patients With Philadelphia Chromosome-Negative Myeloproliferative Neoplasms. Front Oncol. 2021 May 18;11:665037. [CrossRef]
- Cacemiro MDC, Cominal JG, Tognon R, Nunes NS, Simões BP, Lôbo de Figueiredo-Pontes L, Bazzo Catto LF, Traina F, Xisto Souto E, Albani Zambuzi F, Frantz FG, de Castro FA. Philadelphia-negative myeloproliferative neoplasms as disorders marked by cytokine modulation. Hematol Transfus Cell Ther. 2018 Apr-Jun;40(2):120-131. [CrossRef]
- Koschmieder S, Chatain N. Role of inflammation in the biology of myeloproliferative neoplasms. Blood Rev. 2020 Jul;42:100711. [CrossRef]
- Strickland M, Quek L, Psaila B. The immune landscape in BCR-ABL negative myeloproliferative neoplasms: inflammation, infections and opportunities for immunotherapy. Br J Haematol. 2022 Mar;196(5):1149-1158. [CrossRef]
- Jung SH, Lee SE, Yun S, Min DE, Shin Y, Chung YJ, Lee SH. Different inflammatory, fibrotic, and immunologic signatures between pre-fibrotic and overt primary myelofibrosis. Haematologica. 2025 Apr 1;110(4):938-951. [CrossRef]
- Morsia E, Gangat N. Myeloproliferative Neoplasms with Monocytosis. Curr Hematol Malig Rep. 2022 Feb;17(1):46-51. [CrossRef]
- Bassan VL, Barretto GD, de Almeida FC, Palma PVB, Binelli LS, da Silva JPL, Fontanari C, Castro RC, de Figueiredo Pontes LL, Frantz FG, de Castro FA. Philadelphia-negative myeloproliferative neoplasms display alterations in monocyte subpopulations frequency and immunophenotype. Med Oncol. 2022 Sep 29;39(12):223. [CrossRef]
- CE, Newberry KJ, Prijic S, Knez L, Bozinovic K, Harris DM, Spaeth EL, Post SM, Multani AS, Rampal RK, Ahn J, Levine RL, Creighton CJ, Kantarjian HM, Estrov Z. Role of neoplastic monocyte-derived fibrocytes in primary myelofibrosis. J Exp Med. 2016 Aug 22;213(9):1723-40. [CrossRef]
- Spanoudakis E, Papoutselis M, Bazdiara I, Lamprianidi E, Kordella X, Tilkeridis C, Tsatalas C, Kotsianidis I. The JAK2V617F Point Mutation Increases the Osteoclast Forming Ability of Monocytes in Patients with Chronic Myeloproliferative Neoplasms and Makes their Osteoclasts more Susceptible to JAK2 Inhibition. Mediterr J Hematol Infect Dis. 2018 Nov 1;10(1):e2018058. [CrossRef]
- Williams H, Mack C, Baraz R, Marimuthu R, Naralashetty S, Li S, Medbury H. Monocyte Differentiation and Heterogeneity: Inter-Subset and Interindividual Differences. Int J Mol Sci. 2023 May 15;24(10):8757. [CrossRef]
- Kiss M, Caro AA, Raes G, Laoui D. Systemic Reprogramming of Monocytes in Cancer. Front Oncol. 2020 Sep 17;10:1399. [CrossRef]
- Ng LG, Liu Z, Kwok I, Ginhoux F. Origin and Heterogeneity of Tissue Myeloid Cells: A Focus on GMP-Derived Monocytes and Neutrophils. Annu Rev Immunol. 2023 Apr 26;41:375-404. [CrossRef]
- Guilliams M, Mildner A, Yona S. Developmental and Functional Heterogeneity of Monocytes. Immunity. 2018 Oct 16;49(4):595-613. [CrossRef]
- Ginhoux F, Mildner A, Gautier EL, Schlitzer A, Jakubzick C, Varol C, Bain C, Guermonprez P. Editorial: Monocyte Heterogeneity and Function. Front Immunol. 2021 Jan 7;11:626725. [CrossRef]
- Canè S, Ugel S, Trovato R, Marigo I, De Sanctis F, Sartoris S, Bronte V. The Endless Saga of Monocyte Diversity. Front Immunol. 2019 Aug 6;10:1786. [CrossRef]
- Patel AA, Zhang Y, Fullerton JN, Boelen L, Rongvaux A, Maini AA, Bigley V, Flavell RA, Gilroy DW, Asquith B, Macallan D, Yona S. The fate and lifespan of human monocyte subsets in steady state and systemic inflammation. J Exp Med. 2017 Jul 3;214(7):1913-1923. [CrossRef]
- Tak T, Drylewicz J, Conemans L, de Boer RJ, Koenderman L, Borghans JAM, Tesselaar K. Circulatory and maturation kinetics of human monocyte subsets in vivo. Blood. 2017 Sep 21;130(12):1474-1477. [CrossRef]
- Bianchini M, Duchêne J, Santovito D, Schloss MJ, Evrard M, Winkels H, Aslani M, Mohanta SK, Horckmans M, Blanchet X, Lacy M, von Hundelshausen P, Atzler D, Habenicht A, Gerdes N, Pelisek J, Ng LG, Steffens S, Weber C, Megens RTA. PD-L1 expression on nonclassical monocytes reveals their origin and immunoregulatory function. Sci Immunol. 2019 Jun 21;4(36):eaar3054. [CrossRef]
- Kapellos TS, Bonaguro L, Gemünd I, Reusch N, Saglam A, Hinkley ER, Schultze JL. Human Monocyte Subsets and Phenotypes in Major Chronic Inflammatory Diseases. Front Immunol. 2019 Aug 30;10:2035. [CrossRef]
- Robinson A, Han CZ, Glass CK, Pollard JW. Monocyte Regulation in Homeostasis and Malignancy. Trends Immunol. 2021 Feb;42(2):104-119. [CrossRef]
- Guglietta S, Krieg C. Phenotypic and functional heterogeneity of monocytes in health and cancer in the era of high dimensional technologies. Blood Rev. 2023 Mar;58:101012. [CrossRef]
- Li C, Xiao M, Geng S, Wang Y, Zeng L, Lai P, Gong Y, Chen X. Comprehensive analysis of human monocyte subsets using full-spectrum flow cytometry and hierarchical marker clustering. Front Immunol. 2024 Apr 29;15:1405249.
- Hoffmann J, Fišer K, Liebetrau C, Staubach N, Kost D, Voss S, Heiden AZ, Dörr O, Lipps C, Nef HM, Möllmann H, Hamm CW, Keller T, Troidl C. High-Content Immunophenotyping and Hierarchical Clustering Reveal Sources of Heterogeneity and New Surface Markers of Human Blood Monocyte Subsets. Thromb Haemost. 2020 Jan;120(1):141-155. [CrossRef]
- Boyette LB, Macedo C, Hadi K, Elinoff BD, Walters JT, Ramaswami B, Chalasani G, Taboas JM, Lakkis FG, Metes DM. Phenotype, function, and differentiation potential of human monocyte subsets. PLoS One. 2017 Apr 26;12(4):e0176460. [CrossRef]
- Sánchez-Torres C, García-Romo GS, Cornejo-Cortés MA, Rivas-Carvalho A, Sánchez-Schmitz G. CD16+ and CD16- human blood monocyte subsets differentiate in vitro to dendritic cells with different abilities to stimulate CD4+ T cells. Int Immunol. 2001 Dec;13(12):1571-81. [CrossRef]
- Huang MN, Nicholson LT, Batich KA, Swartz AM, Kopin D, Wellford S, Prabhakar VK, Woroniecka K, Nair SK, Fecci PE, Sampson JH, Gunn MD. Antigen-loaded monocyte administration induces potent therapeutic antitumor T cell responses. J Clin Invest. 2020 Feb 3;130(2):774-788. [CrossRef]
- Tacke F, Ginhoux F, Jakubzick C, van Rooijen N, Merad M, Randolph GJ. Immature monocytes acquire antigens from other cells in the bone marrow and present them to T cells after maturing in the periphery. J Exp Med. 2006 Mar 20;203(3):583-97. [CrossRef]
- Jakubzick C, Gautier EL, Gibbings SL, Sojka DK, Schlitzer A, Johnson TE, Ivanov S, Duan Q, Bala S, Condon T, van Rooijen N, Grainger JR, Belkaid Y, Ma’ayan A, Riches DW, Yokoyama WM, Ginhoux F, Henson PM, Randolph GJ. Minimal differentiation of classical monocytes as they survey steady-state tissues and transport antigen to lymph nodes. Immunity. 2013 Sep 19;39(3):599-610. [CrossRef]
- Jakubzick CV, Randolph GJ, Henson PM. Monocyte differentiation and antigen-presenting functions. Nat Rev Immunol. 2017 Jun;17(6):349-362. [CrossRef]
- Shi C, Pamer EG. Monocyte recruitment during infection and inflammation. Nat Rev Immunol. 2011 Oct 10;11(11):762-74. [CrossRef]
- Tacke F, Alvarez D, Kaplan TJ, Jakubzick C, Spanbroek R, Llodra J, Garin A, Liu J, Mack M, van Rooijen N, Lira SA, Habenicht AJ, Randolph GJ. Monocyte subsets differentially employ CCR2, CCR5, and CX3CR1 to accumulate within atherosclerotic plaques. J Clin Invest. 2007 Jan;117(1):185-94. [CrossRef]
- Ancuta P, Rao R, Moses A, Mehle A, Shaw SK, Luscinskas FW, Gabuzda D. Fractalkine preferentially mediates arrest and migration of CD16+ monocytes. J Exp Med. 2003 Jun 16;197(12):1701-7. [CrossRef]
- Cormican S, Griffin MD. Human Monocyte Subset Distinctions and Function: Insights From Gene Expression Analysis. Front Immunol. 2020 Jun 4;11:1070. [CrossRef]
- Anbazhagan K, Duroux-Richard I, Jorgensen C, Apparailly F. Transcriptomic network support distinct roles of classical and non-classical monocytes in human. Int Rev Immunol. 2014 Nov-Dec;33(6):470-89. [CrossRef]
- Schmidl C, Renner K, Peter K, Eder R, Lassmann T, Balwierz PJ, Itoh M, Nagao-Sato S, Kawaji H, Carninci P, Suzuki H, Hayashizaki Y, Andreesen R, Hume DA, Hoffmann P, Forrest AR, Kreutz MP, Edinger M, Rehli M; FANTOM consortium. Transcription and enhancer profiling in human monocyte subsets. Blood. 2014 Apr 24;123(17):e90-9. [CrossRef]
- Wong KL, Tai JJ, Wong WC, Han H, Sem X, Yeap WH, Kourilsky P, Wong SC. Gene expression profiling reveals the defining features of the classical, intermediate, and nonclassical human monocyte subsets. Blood. 2011 Aug 4;118(5):e16-31. [CrossRef]
- Gren ST, Rasmussen TB, Janciauskiene S, Håkansson K, Gerwien JG, Grip O. A Single-Cell Gene-Expression Profile Reveals Inter-Cellular Heterogeneity within Human Monocyte Subsets. PLoS One. 2015 Dec 9;10(12):e0144351. [CrossRef]
- Barone M, Catani L, Ricci F, Romano M, Forte D, Auteri G, Bartoletti D, Ottaviani E, Tazzari PL, Vianelli N, Cavo M, Palandri F. The role of circulating monocytes and JAK inhibition in the infectious-driven inflammatory response of myelofibrosis. Oncoimmunology. 2020 Jun 23;9(1):1782575. [CrossRef]
- de la Guardia RD, Correa JG, López-Millán B, Juan M, Bueno C, Cervantes F, Menéndez P. Detection of inflammatory monocytes but not mesenchymal stem/stromal cells in peripheral blood of patients with myelofibrosis. Br J Haematol. 2018 Apr;181(1):133-137. [CrossRef]
- Romano M, Sollazzo D, Trabanelli S, Barone M, Polverelli N, Perricone M, Forte D, Luatti S, Cavo M, Vianelli N, Jandus C, Palandri F, Catani L. Mutations in JAK2 and Calreticulin genes are associated with specific alterations of the immune system in myelofibrosis. Oncoimmunology. 2017 Jul 5;6(10):e1345402. [CrossRef]
- Fan W, Cao W, Shi J, Gao F, Wang M, Xu L, Wang F, Li Y, Guo R, Bian Z, Li W, Jiang Z, Ma W. Contributions of bone marrow monocytes/macrophages in myeloproliferative neoplasms with JAK2V617F mutation. Ann Hematol. 2023 Jul;102(7):1745-1759. [CrossRef]
- Kong T, Laranjeira ABA, Letson CT, Yu L, Lin S, Fowles JS, Fisher DAC, Ng S, Yang W, He F, Youn M, Mark K, Jose AS, Liu J, Kim AB, Cox MJ, Fulbright MC, Jayanthan A, Los G, Rentschler SL, Ding L, Sakamoto KM, Dunn SE, Challen GA, Oh ST. RSK1 is an exploitable dependency in myeloproliferative neoplasms and secondary acute myeloid leukemia. Nat Commun. 2025 Jan 16;16(1):492. [CrossRef]
- Bassan VL, de Freitas Martins Felício R, Ribeiro Malmegrim KC, Attié de Castro F. Myeloproliferative Neoplasms Transcriptome Reveals Pro-Inflammatory Signature and Enrichment in Peripheral Blood Monocyte-Related Genes. Cancer Invest. 2024 Aug;42(7):605-618. [CrossRef]
- Campanelli R, Rosti V, Fois G, Bonetti E, Barosi G, Massa M. CD14(bright)CD16(low) intermediate monocytes expressing Tie2 are increased in the peripheral blood of patients with primary myelofibrosis. Exp Hematol. 2014 Apr;42(4):244-6. [CrossRef]
- Campanelli R, Fois G, Catarsi P, Poletto V, Villani L, Erba BG, Maddaluno L, Jemos B, Salmoiraghi S, Guglielmelli P, Abbonante V, Di Buduo CA, Balduini A, Iurlo A, Barosi G, Rosti V, Massa M; AGIMM Investigators. Tie2 Expressing Monocytes in the Spleen of Patients with Primary Myelofibrosis. PLoS One. 2016 Jun 9;11(6):e0156990. [CrossRef]
- Maekawa T, Kato S, Kawamura T, Takada K, Sone T, Ogata H, Saito K, Izumi T, Nagao S, Takano K, Okada Y, Tachi N, Teramoto M, Horiuchi T, Hikota-Saga R, Endo-Umeda K, Uno S, Osawa Y, Kobayashi A, Kobayashi S, Sato K, Hashimoto M, Suzu S, Usuki K, Morishita S, Araki M, Makishima M, Komatsu N, Kimura F. Increased SLAMF7high monocytes in myelofibrosis patients harboring JAK2V617F provide a therapeutic target of elotuzumab. Blood. 2019 Sep 5;134(10):814-825. [CrossRef]
- Cimen Bozkus C, Roudko V, Finnigan JP, Mascarenhas J, Hoffman R, Iancu-Rubin C, Bhardwaj N. Immune Checkpoint Blockade Enhances Shared Neoantigen-Induced T-cell Immunity Directed against Mutated Calreticulin in Myeloproliferative Neoplasms. Cancer Discov. 2019 Sep;9(9):1192-1207. [CrossRef]
- Shin DS, Zaretsky JM, Escuin-Ordinas H, Garcia-Diaz A, Hu-Lieskovan S, Kalbasi A, Grasso CS, Hugo W, Sandoval S, Torrejon DY, Palaskas N, Rodriguez GA, Parisi G, Azhdam A, Chmielowski B, Cherry G, Seja E, Berent-Maoz B, Shintaku IP, Le DT, Pardoll DM, Diaz LA Jr, Tumeh PC, Graeber TG, Lo RS, Comin-Anduix B, Ribas A. Primary Resistance to PD-1 Blockade Mediated by JAK1/2 Mutations. Cancer Discov. 2017 Feb;7(2):188-201. [CrossRef]
- Fleischman AG, Aichberger KJ, Luty SB, Bumm TG, Petersen CL, Doratotaj S, Vasudevan KB, LaTocha DH, Yang F, Press RD, Loriaux MM, Pahl HL, Silver RT, Agarwal A, O’Hare T, Druker BJ, Bagby GC, Deininger MW. TNFα facilitates clonal expansion of JAK2V617F positive cells in myeloproliferative neoplasms. Blood. 2011 Dec 8;118(24):6392-8. [CrossRef]
- Veletic I, Manshouri T, Multani AS, Yin CC, Chen L, Verstovsek S, Estrov Z. Myelofibrosis osteoclasts are clonal and functionally impaired. Blood. 2019 May 23;133(21):2320-2324. [CrossRef]
- De Maeyer RPH, Chambers ES. The impact of ageing on monocytes and macrophages. Immunol Lett. 2021 Feb;230:1-10. [CrossRef]
- Gibellini L, De Biasi S, Paolini A, Borella R, Boraldi F, Mattioli M, Lo Tartaro D, Fidanza L, Caro-Maldonado A, Meschiari M, Iadisernia V, Bacca E, Riva G, Cicchetti L, Quaglino D, Guaraldi G, Busani S, Girardis M, Mussini C, Cossarizza A. Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia. EMBO Mol Med. 2020 Dec 7;12(12):e13001. [CrossRef]
- Jones N, Iljin K, Dumont DJ, Alitalo K. Tie receptors: new modulators of angiogenic and lymphangiogenic responses. Nat Rev Mol Cell Biol. 2001 Apr;2(4):257-67. [CrossRef]
- Venneri MA, De Palma M, Ponzoni M, Pucci F, Scielzo C, Zonari E, Mazzieri R, Doglioni C, Naldini L. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood. 2007 Jun 15;109(12):5276-85. [CrossRef]
- De Palma M, Murdoch C, Venneri MA, Naldini L, Lewis CE. Tie2-expressing monocytes: regulation of tumor angiogenesis and therapeutic implications. Trends Immunol. 2007 Dec;28(12):519-24. [CrossRef]
- Cortes J, Talpaz M, Smith HP, Snyder DS, Khoury J, Bhalla KN, Pinilla-Ibarz J, Larson R, Mitchell D, Wise SC, Rutkoski TJ, Smith BD, Flynn DL, Kantarjian HM, Rosen O, Van Etten RA. Phase 1 dose-finding study of rebastinib (DCC-2036) in patients with relapsed chronic myeloid leukemia and acute myeloid leukemia. Haematologica. 2017 Mar;102(3):519-528. [CrossRef]
- Anampa JD, Flynn DL, Leary C, Oh S, Xue X, Oktay MH, Condeelis JS, Sparano JA. Phase Ib Clinical and Pharmacodynamic Study of the TIE2 Kinase Inhibitor Rebastinib with Paclitaxel or Eribulin in HER2-Negative Metastatic Breast Cancer. Clin Cancer Res. 2025 Jan 17;31(2):266-277. [CrossRef]
- Zhang Y, Brekken RA. Are TEMs Canceled? Questioning the Functional Relevance of Tie2-Expressing Macrophages. Cancer Res. 2022 Apr 1;82(7):1172-1173. [CrossRef]
- Altalbawy FMA, Babamuradova Z, Baldaniya L, Singh A, Singh KU, Ballal S, Sabarivani A, Sead FF, Alam R, Alshahrani MY. The multifaceted role of CS1 (SLAMF7) in immunoregulation: Implications for cancer therapy and autoimmune disorders. Exp Cell Res. 2025 Apr 1;447(1):114516. [CrossRef]
- Zhang Z, Zhang Y, Chen Z, Xia L. Emerging roles of SLAMF7 in immune cells and related diseases. Innate Immun. 2025 Jan-Dec;31:17534259251326700. [CrossRef]
- Van Egeren D, Kamaz B, Liu S, Nguyen M, Reilly CR, Kalyva M, DeAngelo DJ, Galinsky I, Wadleigh M, Winer ES, Luskin MR, Stone RM, Garcia JS, Hobbs GS, Michor F, Cortes-Ciriano I, Mullally A, Hormoz S. Transcriptional differences between JAK2-V617F and wild-type bone marrow cells in patients with myeloproliferative neoplasms. Exp Hematol. 2022 Mar;107:14-19. [CrossRef]
- Martino EA, Palmieri S, Galli M, Derudas D, Mina R, Della Pepa R, Zambello R, Vigna E, Bruzzese A, Mangiacavalli S, Zamagni E, Califano C, Musso M, Conticello C, Cerchione C, Mele G, Di Renzo N, Offidani M, Tarantini G, Casaluci GM, Rago A, Ria R, Uccello G, Barilà G, Palumbo G, Pettine L, De Magistris C, Vincelli ID, Brunori M, Accardi F, Amico V, Amendola A, Fontana R, Bongarzoni V, Rossini B, Cotzia E, Gozzetti A, Rizzi R, Sgherza N, Curci P, Mancuso K, Reddiconto G, Maroccia A, Franceschini L, Bertuglia G, Nappi D, Barbieri E, Quaresima M, Petrucci MT, Di Raimondo F, Neri A, Tripepi G, Musto P, Morabito F, Gentile M. Outcomes and prognostic indicators in daratumumab-refractory multiple myeloma: a multicenter real-world study of elotuzumab, pomalidomide, and dexamethasone in 247 patients. ESMO Open. 2025 Jan 7;10(2):104084. [CrossRef]
- Liu R, Li HF, Li S. PD-1-mediated inhibition of T cell activation: Mechanisms and strategies for cancer combination immunotherapy. Cell Insight. 2024 Feb 23;3(2):100146. [CrossRef]
- Oh SA, Wu DC, Cheung J, Navarro A, Xiong H, Cubas R, Totpal K, Chiu H, Wu Y, Comps-Agrar L, Leader AM, Merad M, Roose-Germa M, Warming S, Yan M, Kim JM, Rutz S, Mellman I. PD-L1 expression by dendritic cells is a key regulator of T-cell immunity in cancer. Nat Cancer. 2020 Jul;1(7):681-691. [CrossRef]
- Asai A, Yasuoka H, Matsui M, Tsuchimoto Y, Fukunishi S, Higuchi K. Programmed Death 1 Ligand Expression in the Monocytes of Patients with Hepatocellular Carcinoma Depends on Tumor Progression. Cancers (Basel). 2020 Aug 14;12(8):2286. [CrossRef]
- Prestipino A, Emhardt AJ, Aumann K, O’Sullivan D, Gorantla SP, Duquesne S, Melchinger W, Braun L, Vuckovic S, Boerries M, Busch H, Halbach S, Pennisi S, Poggio T, Apostolova P, Veratti P, Hettich M, Niedermann G, Bartholomä M, Shoumariyeh K, Jutzi JS, Wehrle J, Dierks C, Becker H, Schmitt-Graeff A, Follo M, Pfeifer D, Rohr J, Fuchs S, Ehl S, Hartl FA, Minguet S, Miething C, Heidel FH, Kröger N, Triviai I, Brummer T, Finke J, Illert AL, Ruggiero E, Bonini C, Duyster J, Pahl HL, Lane SW, Hill GR, Blazar BR, von Bubnoff N, Pearce EL, Zeiser R. Oncogenic JAK2V617F causes PD-L1 expression, mediating immune escape in myeloproliferative neoplasms. Sci Transl Med. 2018 Feb 21;10(429):eaam7729. [CrossRef]
- Zaretsky JM, Garcia-Diaz A, Shin DS, Escuin-Ordinas H, Hugo W, Hu-Lieskovan S, Torrejon DY, Abril-Rodriguez G, Sandoval S, Barthly L, Saco J, Homet Moreno B, Mezzadra R, Chmielowski B, Ruchalski K, Shintaku IP, Sanchez PJ, Puig-Saus C, Cherry G, Seja E, Kong X, Pang J, Berent-Maoz B, Comin-Anduix B, Graeber TG, Tumeh PC, Schumacher TN, Lo RS, Ribas A. Mutations Associated with Acquired Resistance to PD-1 Blockade in Melanoma. N Engl J Med. 2016 Sep 1;375(9):819-29. [CrossRef]
- Wang JC, Chen C, Kundra A, Kodali S, Pandey A, Wong C, Cheung T, Gotlieb V, Joseph G, Tribie S. Programmed Cell Death Receptor (PD-1) Ligand (PD-L1) expression in Philadelphia chromosome-negative myeloproliferative neoplasms. Leuk Res. 2019 Apr;79:52-59. [CrossRef]
- Hobbs G, Cimen Bozkus C, Moshier E, Dougherty M, Bar-Natan M, Sandy L, Johnson K, Foster JE, Som T, Macrae M, Marble H, Salama M, El Jamal SM, Zubizarreta N, Wadleigh M, Stone R, Bhardwaj N, Iancu-Rubin C, Mascarenhas J. PD-1 inhibition in advanced myeloproliferative neoplasms. Blood Adv. 2021 Dec 14;5(23):5086-5097. [CrossRef]
- Isfort S, von Bubnoff N, Al-Ali HK, Becker H, Götze T, le Coutre P, Griesshammer M, Moskwa C, Wohn L, Riedel J, Palandri F, Manz K, Hochhaus A, Döhner K, Heidel FH. FRACTION: protocol of a phase II study of Fedratinib and Nivolumab combination in patients with myelofibrosis and resistance or suboptimal response to JAK-inhibitor treatment of the German MPN study group (GSG-MPN). Ann Hematol. 2024 Aug;103(8):2775-2785. [CrossRef]
- Fisher DAC, Miner CA, Engle EK, Hu H, Collins TB, Zhou A, Allen MJ, Malkova ON, Oh ST. Cytokine production in myelofibrosis exhibits differential responsiveness to JAK-STAT, MAP kinase, and NFκB signaling. Leukemia. 2019 Aug;33(8):1978-1995. [CrossRef]
- Lai HY, Brooks SA, Craver BM, Morse SJ, Nguyen TK, Haghighi N, Garbati MR, Fleischman AG. Defective negative regulation of Toll-like receptor signaling leads to excessive TNF-α in myeloproliferative neoplasm. Blood Adv. 2019 Jan 22;3(2):122-131. [CrossRef]
- Heaton WL, Senina AV, Pomicter AD, Salama ME, Clair PM, Yan D, Bell RN, Gililland JM, Prchal JT, O’Hare T, Deininger MW. Autocrine Tnf signaling favors malignant cells in myelofibrosis in a Tnfr2-dependent fashion. Leukemia. 2018 Nov;32(11):2399-2411. [CrossRef]
- Müller P, Baldauf CK, Haage TR, Waldleben AM, Richter F, Pfizenmaier K, Fischer T. Anti-inflammatory treatment in MPN: targeting TNFR1 and TNFR2 in JAK2-V617F-induced disease. Blood Adv. 2021 Dec 14;5(23):5349-5359. [CrossRef]
- Andrew Dunbar, Min Lu, Mirko Farina, et al. Increased Interleukin-8 (IL8)-CXCR2 Signaling Promotes Progression of Bone Marrow Fibrosis in Myeloproliferative Neoplasms. Blood 2020; 136 (Supplement 1): 6–7.
- Vermeersch G, Proost P, Struyf S, Gouwy M, Devos T. CXCL8 and its cognate receptors CXCR1/CXCR2 in primary myelofibrosis. Haematologica. 2024 Jul 1;109(7):2060-2072. [CrossRef]
- Yahara Y, Nguyen T, Ishikawa K, Kamei K, Alman BA. The origins and roles of osteoclasts in bone development, homeostasis and repair. Development. 2022 Apr 15;149(8):dev199908. [CrossRef]
- Curto-Garcia N, Harrison C, McLornan DP. Bone marrow niche dysregulation in myeloproliferative neoplasms. Haematologica. 2020 May;105(5):1189-1200. [CrossRef]
- Thiele J, Braeckel C, Wagner S, Falini B, Dienemann D, Stein H, Fischer R. Macrophages in normal human bone marrow and in chronic myeloproliferative disorders: an immunohistochemical and morphometric study by a new monoclonal antibody (PG-M1) on trephine biopsies. Virchows Arch A Pathol Anat Histopathol. 1992;421(1):33-9. [CrossRef]
- Molitor DCA, Boor P, Buness A, Schneider RK, Teichmann LL, Körber RM, Horvath GL, Koschmieder S, Gütgemann I. Macrophage frequency in the bone marrow correlates with morphologic subtype of myeloproliferative neoplasm. Ann Hematol. 2021 Jan;100(1):97-104. [CrossRef]
- Wakahashi K, Minagawa K, Kawano Y, Kawano H, Suzuki T, Ishii S, Sada A, Asada N, Sato M, Kato S, Shide K, Shimoda K, Matsui T, Katayama Y. Vitamin D receptor-mediated skewed differentiation of macrophages initiates myelofibrosis and subsequent osteosclerosis. Blood. 2019 Apr 11;133(15):1619-1629. [CrossRef]
- Shi Z, Liu J, Zhao Y, Yang L, Cai Y, Zhang P, Xu Z, Qin T, Qu S, Pan L, Wu J, Yan X, Li Z, Zhang W, Yan Y, Huang H, Huang G, Li B, Wu X, Xiao Z. ASXL1 mutations accelerate bone marrow fibrosis via EGR1-TNFA axis-mediated neoplastic fibrocyte generation in myeloproliferative neoplasms. Haematologica. 2023 May 1;108(5):1359-1373. [CrossRef]
- Heine A, Held SA, Daecke SN, Wallner S, Yajnanarayana SP, Kurts C, Wolf D, Brossart P. The JAK-inhibitor ruxolitinib impairs dendritic cell function in vitro and in vivo. Blood. 2013 Aug 15;122(7):1192-202. [CrossRef]
- Humblet-Baron S, Barber JS, Roca CP, Lenaerts A, Koni PA, Liston A. Murine myeloproliferative disorder as a consequence of impaired collaboration between dendritic cells and CD4 T cells. Blood. 2019 Jan 24;133(4):319-330. [CrossRef]
- Sachetto ATA, Mackman N. Monocyte Tissue Factor Expression: Lipopolysaccharide Induction and Roles in Pathological Activation of Coagulation. Thromb Haemost. 2023 Nov;123(11):1017-1033. [CrossRef]
- He F, Laranjeira AB, Kong T, Lin S, Ashworth KJ, Liu A, Lasky NM, Fisher DA, Cox MJ, Fulbright MC, Antunes-Heck L, Yu L, Brakhane M, Gao B, Sykes SM, D’Alessandro A, Di Paola J, Oh ST. Multiomic profiling reveals metabolic alterations mediating aberrant platelet activity and inflammation in myeloproliferative neoplasms. J Clin Invest. 2024 Feb 1;134(3):e172256. [CrossRef]
- Wang C, Cheng Y, Li B, Qiu X, Hu H, Zhang X, Lu Z, Zheng F. Transcriptional characteristics and functional validation of three monocyte subsets during aging. Immun Ageing. 2023 Sep 27;20(1):50. [CrossRef]
- Cortés M, Brischetto A, Martinez-Campanario MC, Ninfali C, Domínguez V, Fernández S, Celis R, Esteve-Codina A, Lozano JJ, Sidorova J, Garrabou G, Siegert AM, Enrich C, Pintado B, Morales-Ruiz M, Castro P, Cañete JD, Postigo A. Inflammatory macrophages reprogram to immunosuppression by reducing mitochondrial translation. Nat Commun. 2023 Nov 17;14(1):7471. [CrossRef]
- Sardenberg C, Suassuna P, Watanabe R, Cruz Andreoli MC, Aparecida Dalboni M, Faria Seabra V, Draibe SA, Cendoroglo Neto M, Jaber B. Balance between cytokine production by peripheral blood mononuclear cells and reactive oxygen species production by monocytes in patients with chronic kidney disease. Ren Fail. 2004 Nov;26(6):673-81. [CrossRef]
- Ravi S, Mitchell T, Kramer P, Chacko B, Darley-Usmar VM. Mitochondria in monocytes and macrophages-implications for translational and basic research. Int J Biochem Cell Biol. 2014 Aug;53:202-207. [CrossRef]
- Shallis RM, Zeidan AM, Wang R, Podoltsev NA. Epidemiology of the Philadelphia Chromosome-Negative Classical Myeloproliferative Neoplasms. Hematol Oncol Clin North Am. 2021 Apr;35(2):177-189. [CrossRef]
| Monocyte disturbance | Human monocytes | Mouse monocytes | |
|---|---|---|---|
| Pro-angiogenesis (Tie2) | 52,53 | ||
| Pro-fibrosis (SLAMF7) | 54,69 | 54 | |
| Abnormal cytokine secretion (TNF-α, IL-1β, CXCL8) | 46,47,79,80 | ||
| Inhibiting T cell function (PDL1) | 74,76 | 74 | |
| Abnormal Differentiation | mo-DC | 48 | 92 |
| Macrophages | 49,90 | 90 | |
| Osteoclast | 17,58 | ||
| NCT Number | Targets | Study Title | Study Status |
|---|---|---|---|
| NCT05393674 | PD1/PDL1 | Fedratinib in Combination with Nivolumab | ACTIVE_NOT_RECRUITING |
| NCT01822509 | PD1/PDL1 | Ipilimumab or Nivolumab in Treating Patients With Relapsed Hematologic Malignancies After Donor Stem Cell Transplant | COMPLETED |
| NCT02421354 | PD1/PDL1 | Nivolumab in Treating Patients With Primary Myelofibrosis, Post-Essential Thrombocythemia Myelofibrosis, or Post-Polycythemia Vera Myelofibrosis | TERMINATED |
| NCT04517851 | SLAMF7 | Elotuzumab for the Treatment of JAK2-Mutated Myelofibrosis | ACTIVE_NOT_RECRUITING |
| NCT02805868 | IL-6 | Siltuximab in Treating Patients With Primary, Post-Polycythemia Vera, or Post-Essential Thrombocythemia Myelofibrosis | WITHDRAWN |
| NCT05835466 | CXCL8/CXCR | Reparixin in Patients with Myelofibrosis Myeloproliferative Neoplasms Research Consortium (MPN-RC 120) | RECRUITING |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




