Submitted:
02 June 2025
Posted:
04 June 2025
Read the latest preprint version here
Abstract
Keywords:
Key Points
- Ultra-high contrast MRI using bipolar filters (BLAIRs) can show abnormalities with very high contrast where little or no change from normal is seen with common conventional state-of-the-art sequences.
- The abnormalities shown with ultra-high contrast are due to small changes in tissue properties such as T1 and T2 in disease. This is complementary to existing sequences which show abnormalities due to larger changes in T1 and T2 in disease.
- At boundaries between tissues and fluids on ultra-high contrast images there is frequently an increase in contrast and an increase in the spatial resolution of that contrast.
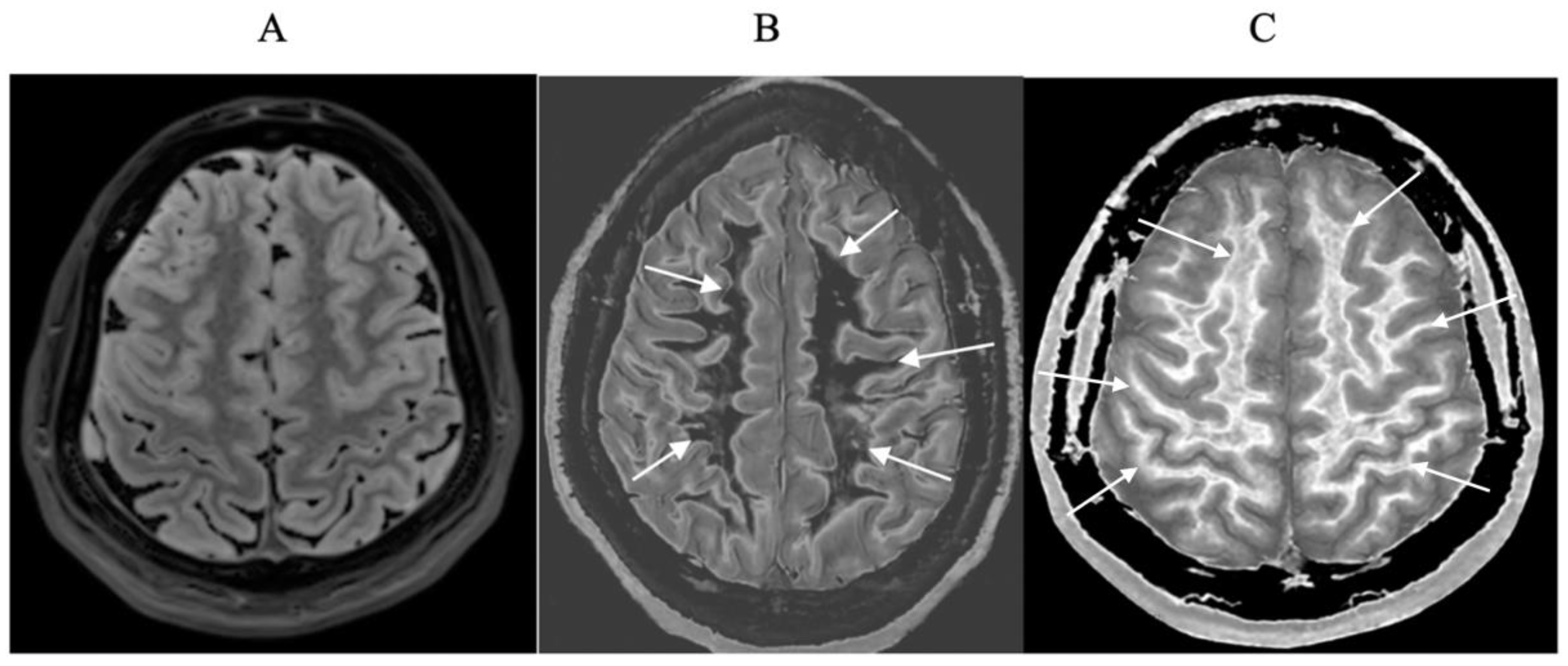
- Extensive abnormalities were seen in mild traumatic brain injury, multiple sclerosis and white matter associated with cerebral tumours in the absence of changes in T2-weighted spin echo or T2-FLAIR images.
- Implementation of BLAIR sequences does not require any increase in static or gradient magnetic field. They can be easily implemented on existing MR systems at very little cost.
1. Introduction
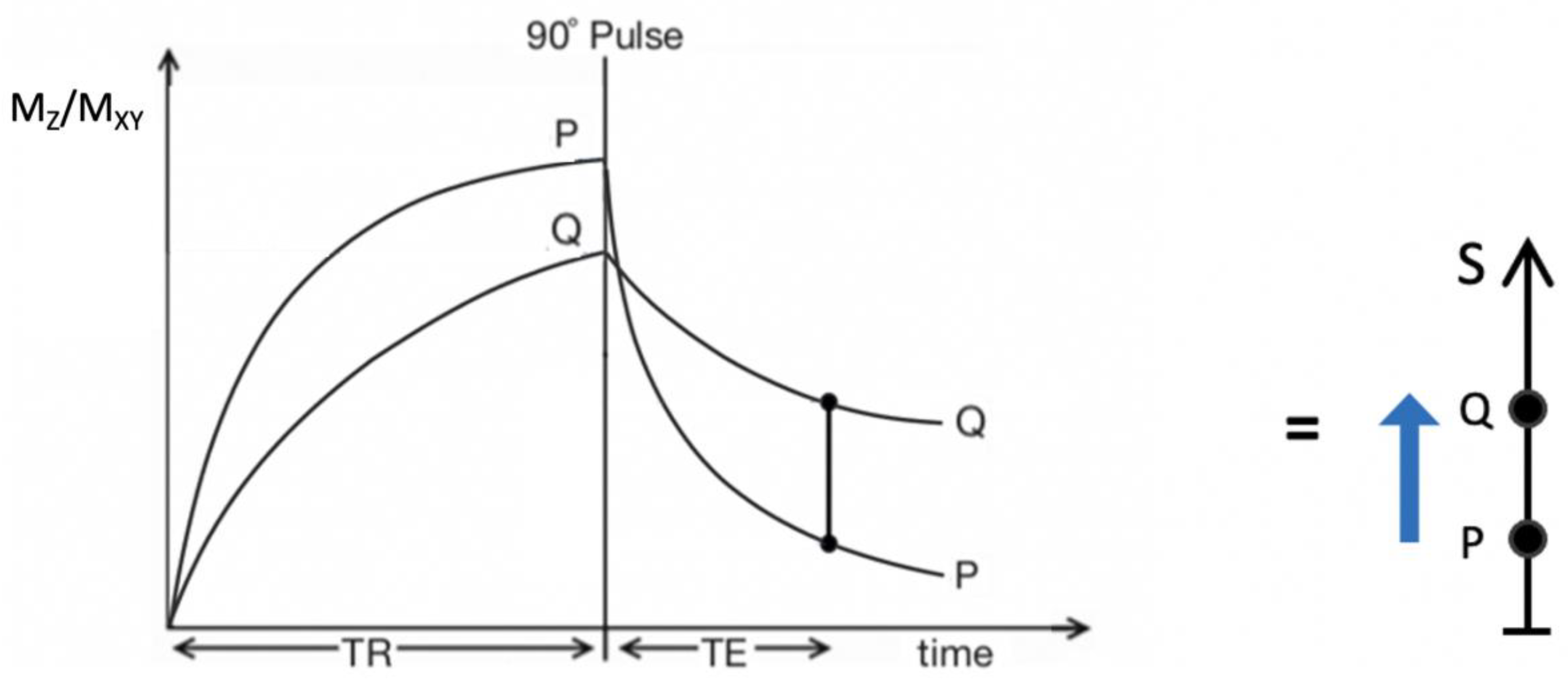
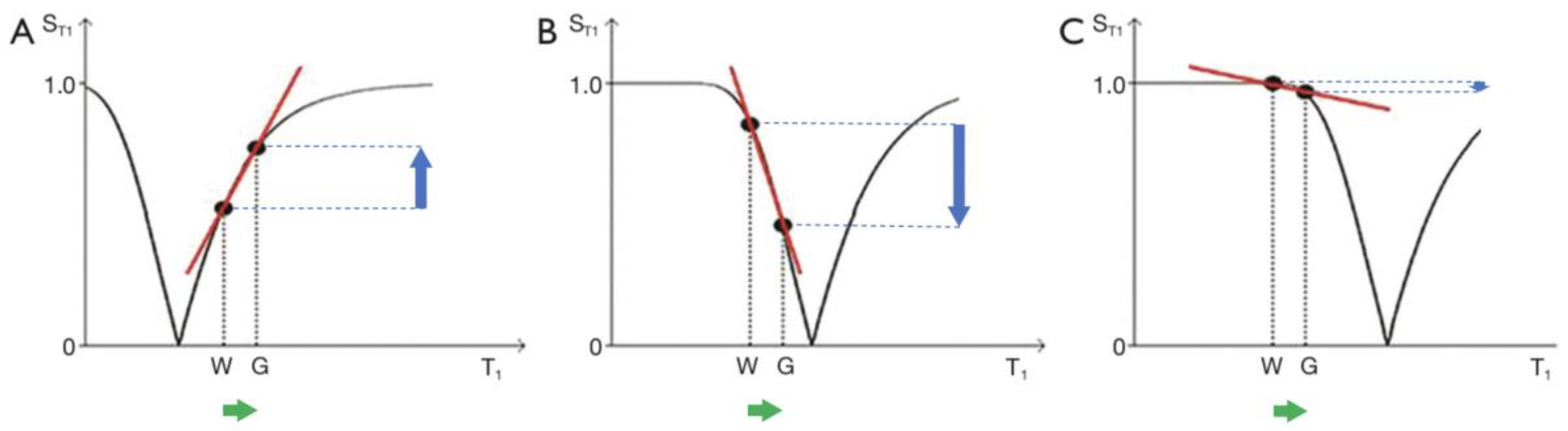
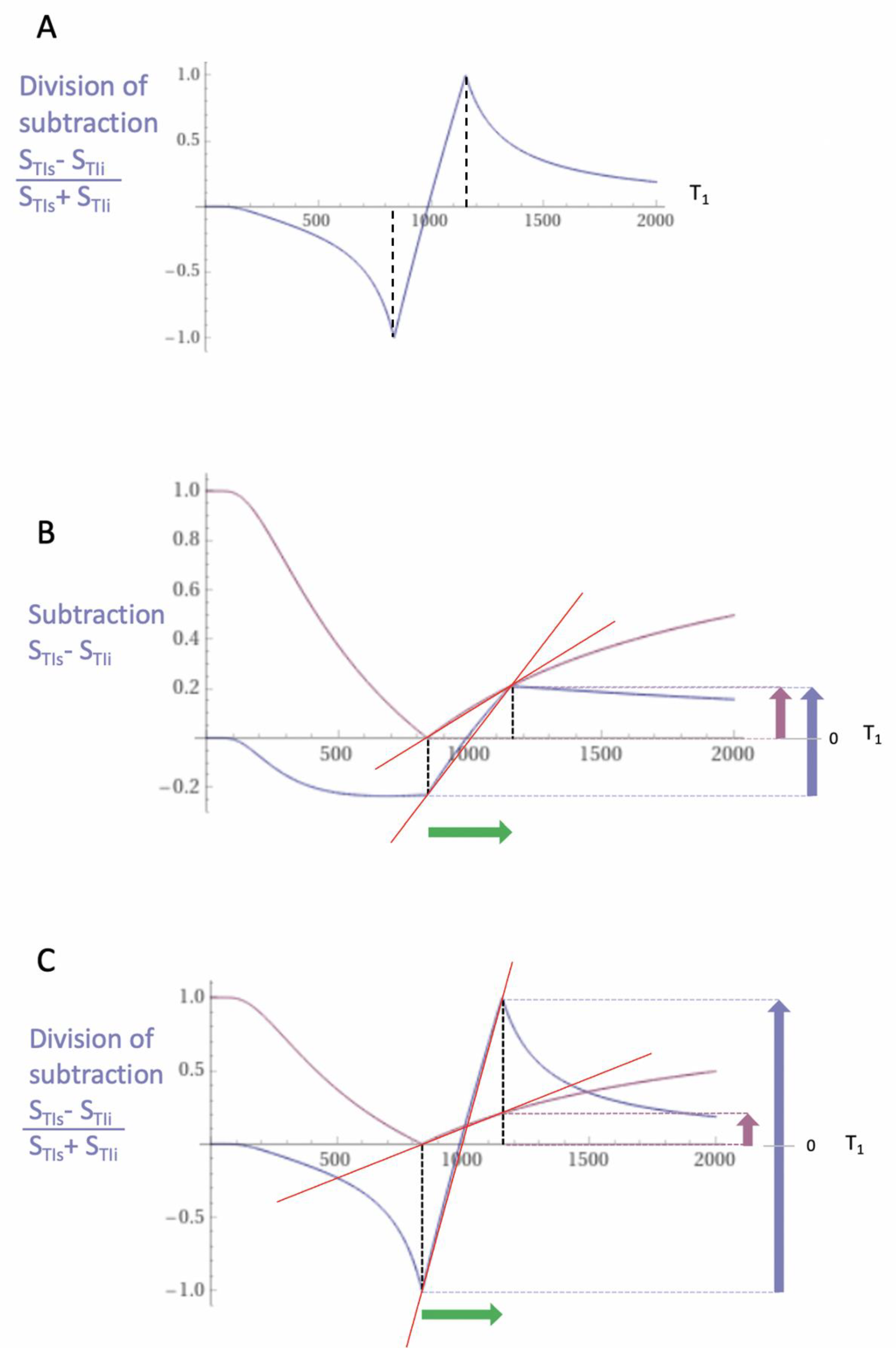
2. Basic Physics
3. Methods
4. Illustrative Cases
5. Discussion
Funding
Statement and Declarations
Ethical approval
Informed consent
Abbreviations
| BLAIR | BipoLAr fIlteR, bipolar filter |
| cdSIR | composite divided Subtracted Inversion Recovery |
| clSIR | composite logarithmic then Subtracted Inversion Recovery |
| DIR | Double Inversion Recovery |
| dSIR | divided Subtracted Inversion Recovery |
| hD | highest Domain |
| IR | Inversion Recovery |
| lD | lowest Domain |
| lSIR | logarithmic then Subtracted Inversion Recovery |
| mD | middle Domain |
| MP2RAGE | Magnetisation Prepared 2 Rapid Acquisition Gradient Echo |
| MT | Magnetisation Transfer |
| SIR | Subtracted Inversion Recovery |
| SOF | Signal from Other Filter |
| TP-bipolar filter | Tissue Property-bipolar filter |
| TP-filter | Tissue Property-filter |
| UHC | Ultra-High Contrast |
References
- Condron P, Cornfeld DM, Scadeng M, et al ( 2024) Ultra-high contrast MRI: the whiteout sign shown with divided subtracted inversion recovery (dSIR) sequences in post-insult leukoencephalopathy syndromes (PILS). Tomography 10(7):983-1013. [CrossRef]
- Ma Y-J, Shao H, Fan S, et al (2020) New options for increasing the sensitivity, specificity and scope of synergistic contrast magnetic resonance imaging (scMRI) using Multiplied, Added, Subtracted and/or FiTted (MASTIR) pulse sequences. Quant Imaging Med Surg 10(10):2030-2065. [CrossRef]
- Ma Y-J, Moazamian D, Port JD, et al (2023) Targeted magnetic resonance imaging (tMRI) of small changes in the T1 and spatial properties of normal and near normal appearing white and gray matter in disease of the brain using divided subtracted inversion recovery (dSIR) and divided reverse subtracted inversion recovery (drSIR) sequences. Quant Imaging Med Surg 13(10):7304-7337. [CrossRef]
- Newburn G, McGeown JP, Kwon EE, et al (2023) Targeted MRI (tMRI) of small increases in the T1 of normal appearing white matter in mild traumatic brain injury (mTBI) using a divided subtracted inversion recovery (dSIR) sequence. OBM Neurobiology 7(4). [CrossRef]
- Ma Y-J, Moazamian D, Cornfeld DM, et al (2022) Improving the understanding and performance of clinical MRI using tissue property filters and the central contrast theorem, MASDIR pulse sequences and synergistic contrast MRI. Quant Imaging Med Surg 12(9):4658-4690.
- Cornfeld D, Condron P, Newburn G, et al (2024) Ultra-high contrast MRI: Using divided subtracted inversion recovery (dSIR) and divided echo subtraction (dES) sequences to study the brain and musculoskeletal system. Bioengineering 11:441. [CrossRef]
- Newburn G, Condron P, Kwon EE, et al (2024) Diagnosis of delayed post-hypoxic leukoencephalopathy (Grinker’s myelinopathy) with MRI using divided subtracted inversion recovery (dSIR) sequences: time for reappraisal of the syndrome? Diagnostics 14(4):418. [CrossRef]
- Yokoo T, Bae WC, Hamilton G, et al (2010) A quantitative approach to sequence and image weighting. J Comput Assist Tomogr 34(3):317-331.
- Young IR, Szeverenyi NM, Du J, Bydder GM (2020) Pulse sequences as tissue property filters (TP-filters): a way of understanding the signal, contrast and weighting of magnetic resonance images. Quant Imaging Med Surg 10(5):1080-1120.
- Bydder, M., Cornfeld, D.M., Melzer, T.R. et al (2025) Log subtracted inversion recovery. Magn Reson Imaging 117:110328.
- Ugurbil K (2014) Magnetic resonance imaging at ultrahigh fields. IEE Trans Biomed Eng 61:1364-1379.
- Feinberg DA, Beckett AJS, Vu AT, et al (2023) Next-generation MRI scanner designed for ultra-high-resolution human brain imaging at 7 Tesla. Nat Methods 20:2048-2057. [CrossRef]
- Bydder M, Condron P, Cornfeld DM, et al (2024) Validation of an ultra-high contrast divided subtracted inversion recovery technique using a standard T1 phantom. NMR Biomed 2024;e5269.
- Losa L, Peruzzo D, Galbiati S, Locatelli F, Agarwal N (2024) Enhancing T1 signal of normal-appearing white matter with divided subtracted inversion recovery: a pilot study in mild traumatic brain injury. NMR Biomed 37(10):e5175. [CrossRef]
- Hajnal JV, Baudouin CJ, Oatridge A, Young IR, Bydder GM (1992) Design and implementation of magnetisation transfer pulse sequences for clinical use. J Comput Assist Tomogr 16(1):7-18.
- Nürnberger L, Gracien R-M, Hok P, et al (2017) Longitudinal changes of cortical microstructure in Parkinson’s disease assessed with T1 relaxometry. NeuroImage Clinical 13:405-414.
- Roeben B, Zeltner L, Hagberg GE, Scheffler K, Schols L, Bender B (2023) Susceptibility-weighted imaging reveals subcortical iron deposition in PLA2G6-associated neurodegeneration: the “Double Cortex Sign”. Mov Disord 38(5):904-906.
- Mueller SG (2024) 7T MP2RAGE for cortical myelin segmentation: impact of aging. PLoS One 19(4):e0299670.
- Ma Y-J, Fan S, Shao H, et al (2020) Use of multiplied, added, subtracted and/or fitted inversion recovery (MASTIR) pulse sequences. Quant Imaging Med Surg 10(6):1334-1369. [CrossRef]
- Bydder GM, Young IR (1985) MR imaging: clinical use of the inversion recovery sequence. J Comput Assist Tomogr 9(4):659-675.
- Redpath TW, Smith FW (1994) Technical note: use of a double inversion recovery pulse sequence to image selectively grey or white brain matter. Br J Radiol 67(804):1258-1263.
- Costagli M, Lapucci C, Zacà D, et al (2022) Improved detection of multiple sclerosis lesions with T2-prepared double inversion recovery at 3T. J Neuroimaging 32:902-909. [CrossRef]
- Tillema J-M, Weigand SD, Dayan M, et al (2018) Dark rims: novel sequence enhances diagnostic specificity in multiple sclerosis. AJNR Am J Neuroradiol 39(6):1052-1058. [CrossRef]
- Marques JP, Kober T, Krueger G, van der Zwaag W, Van de Moortele P-F, Gruetter R (2010) MP2RAGE, a self bias-field corrected sequence for improved segmentation and T1-mapping at high field. Neuroimage 49(2):1271-1281. [CrossRef]
- Beaumont J, Saint-Jalmes H, Acosta O, et al (2019) Multi T1-weighted contrast MRI with fluid and white matter suppression at 1.5T. Magn Reson Imaging 63:217-225.
- Beaumont J, Gambarota G, Saint-Jalmes H, et al (2021) High-resolution multi-T1-weighted contrast and T1 mapping with low B1 sensitivity using the fluid and white matter suppression (FLAWS) sequence at 7T. Magn Reson Med 85(3):1364-1378.
- Müller J, La Rosa F, Beaumont J, et al (2022) Fluid and white matter suppression. New sensitive 3T magnetic resonance imaging contrasts for cortical lesion detection in multiple sclerosis. Invest Radiol 57(9):592-600.
- Durozard P, Maarouf A, Zaaraoui W, et al (2024) Cortical lesions as an early hallmark of Multiple Sclerosis: visualization by 7T MRI. Invest Radiol 59(11):747-753. [CrossRef]
- Saranathan M, Tourdias T, Bayram E, Ghanouni P, Rutt BK (2015) Optimization of white-matter-nulled magnetization prepared rapid gradient echo (MP-RAGE) imaging. Magn Reson Med 73(5):1786-1794. [CrossRef]
- Grewal SS, Middlebrooks EH, Kaufmann TJ, et al (2018) Fast gray matter acquisition T1 inversion recovery MRI to delineate the mammillothalamic tract for preoperative direct targeting of the anterior nucleus of the thalamus for deep brain stimulation in epilepsy. Neurosurg Focus 45(2):E6. [CrossRef]
- Middlebrooks EH, Lin C, Westerhold E, et al (2020) Improved detection of focal cortical dysplasia using a novel 3D imaging sequence: Edge-Enhancing Gradient Echo (3D-EDGE) MRI. NeuroImage 28:102449. [CrossRef]
- Tao S, Zhou E, Greco V, et al (2023) Edge-Enhancing Gradient-Echo MP2RAGE for clinical epilepsy imaging at 7T. AJNR Am J Neuroradiol 44(3):268-270.
- Middlebrooks EH, Tao S, Zhou X, et al (2023) Synthetic inversion image generation using MP2RAGE T1 mapping for surgical targeting in deep brain stimulation and lesioning. Stereotact Funct Neurosurg 101:326-331. [CrossRef]











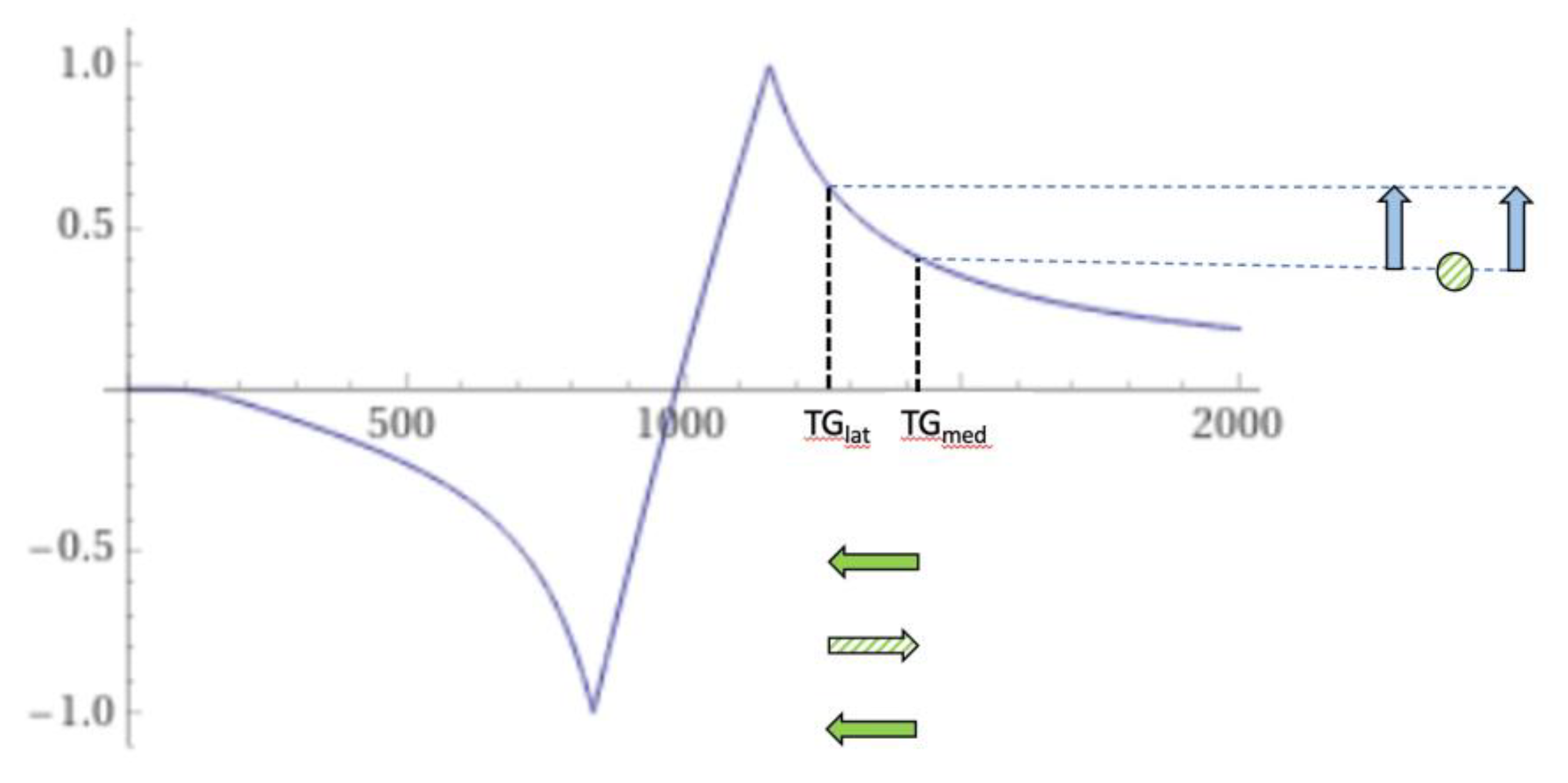
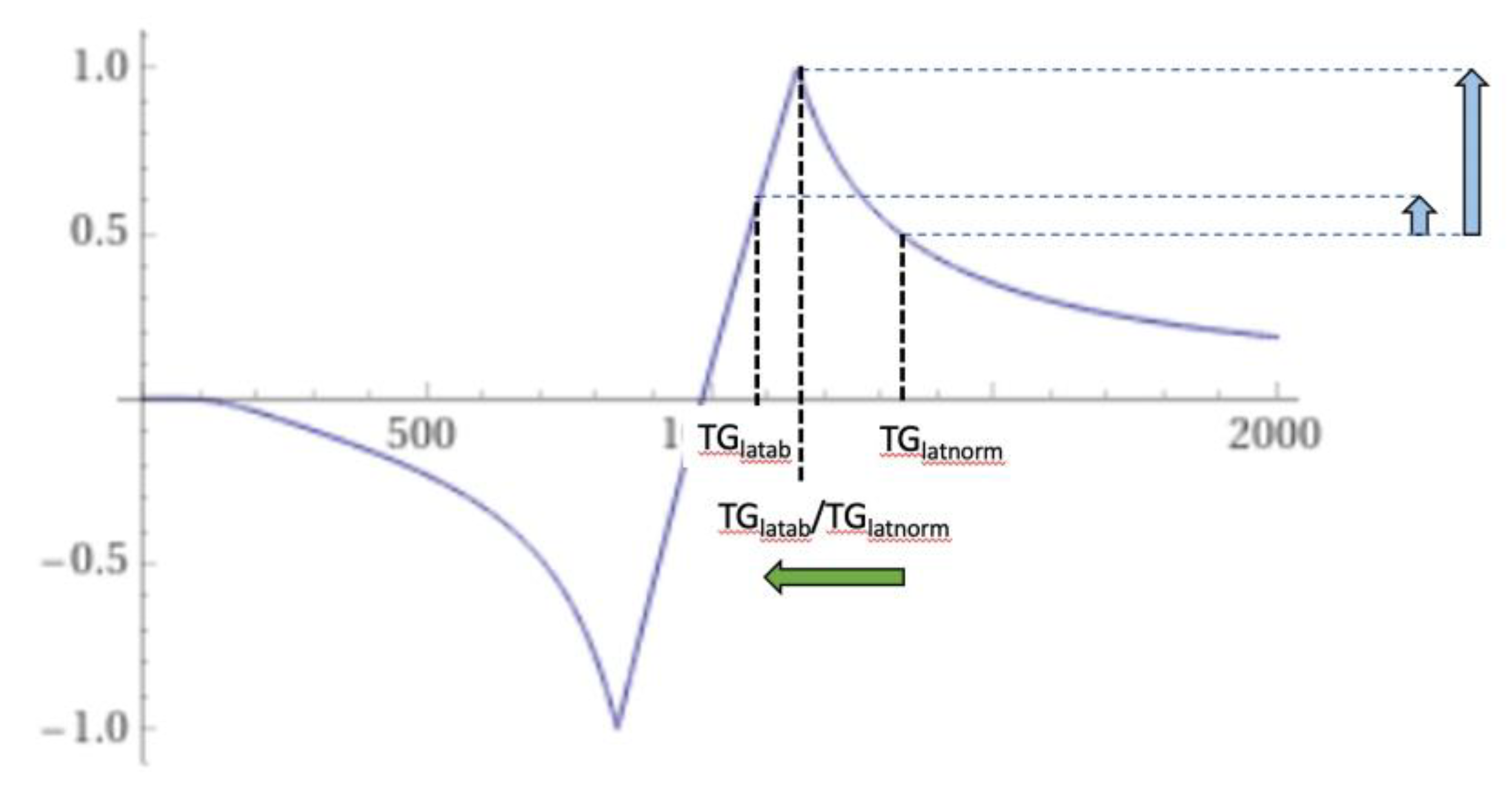
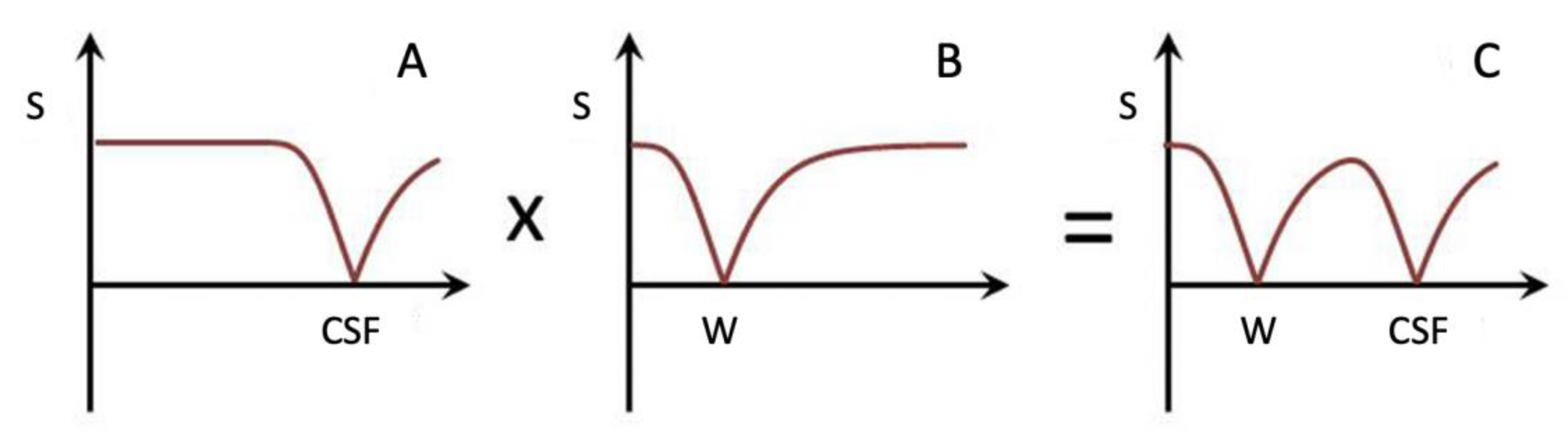

| # | Sequence | TR (ms) | TI (ms) | TE (ms) | Matrix Size Voxel Size (mm) |
Number of Slices | Slice Thickness (mm) |
|---|---|---|---|---|---|---|---|
| 1* | 2D FSE IR (for white nulling matter) | 5,796 | 350 | 7 | 256 x 224 0.9 x 1.0 Z512 0.4 x 0.4 |
26 | 4 |
| 2* | 2D FSE IR (used with #1 for narrow mD dSIR) | 5,796 | 500 | 7 | 256 x 224 0.9 x 1.0 Z512 0.4 x 0.4 |
26 | 4 |
| 3* | 2D FSE IR (for longer T1 nulling) | 5,796 | 600 | 7 | 256 x 224 0.9 x 1.0 Z512 0.4 x 0.4 |
26 | 4 |
| 4* | 2D FSE IR (used with #1 for wide mD dSIR) | 5,796 | 800 | 7 | 256 x 224 0.9 x 1.0 Z512 0.4 x 0.4 |
26 | 4 |
| 5 | 3D BRAVO (for white matter nulling) | 2,000 | 400 | 6 | 256 x 256 0.8 x 0.8 Z512 |
220 | 0.8 |
| 6 | 3D BRAVO (used with #5 for wide mD dSIR) | 2,000 | 800 | 6 | 256 x 256 0.8 x 0.8 Z512 |
220 | 0.8 |
| 7* | 2D T2-FLAIR | 6,300 | 1,851 | 102 | 320 x 240 0.7 x 0.7 Z512 0.4 x 0.4 |
26 | 4 |
| 8 | 3D T2-FLAIR | 6,300 | 1,850 | 102 | 256 x 256 0.8 x 0.8 Z512 0.6 x 0.6 |
252 | 0.8 |
| 9 | 3D susceptibility weighted | 40 | - | 32 | 300 x 300 0.8 x 0.8 Z512 |
110 | 2 |
| Bipolar Filter | Reverse Bipolar Filter | Tissue Properties and Options |
|---|---|---|
| DIR | - | rm, T1, T2, ±MT |
| SIR | rSIR | rm, T1, T2, ±MT |
| dSIR cdSIR |
drSIR cdrSIR |
T1, ±MT T1, ±T2, ±T2*, ±D*, ±c, ±MT |
| lSIR clSIR |
lrSIR clrSIR |
T1, ±MT T1, ±T2, ±T2*, ±D*, ±c, ±MT |


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




