Submitted:
28 May 2025
Posted:
29 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
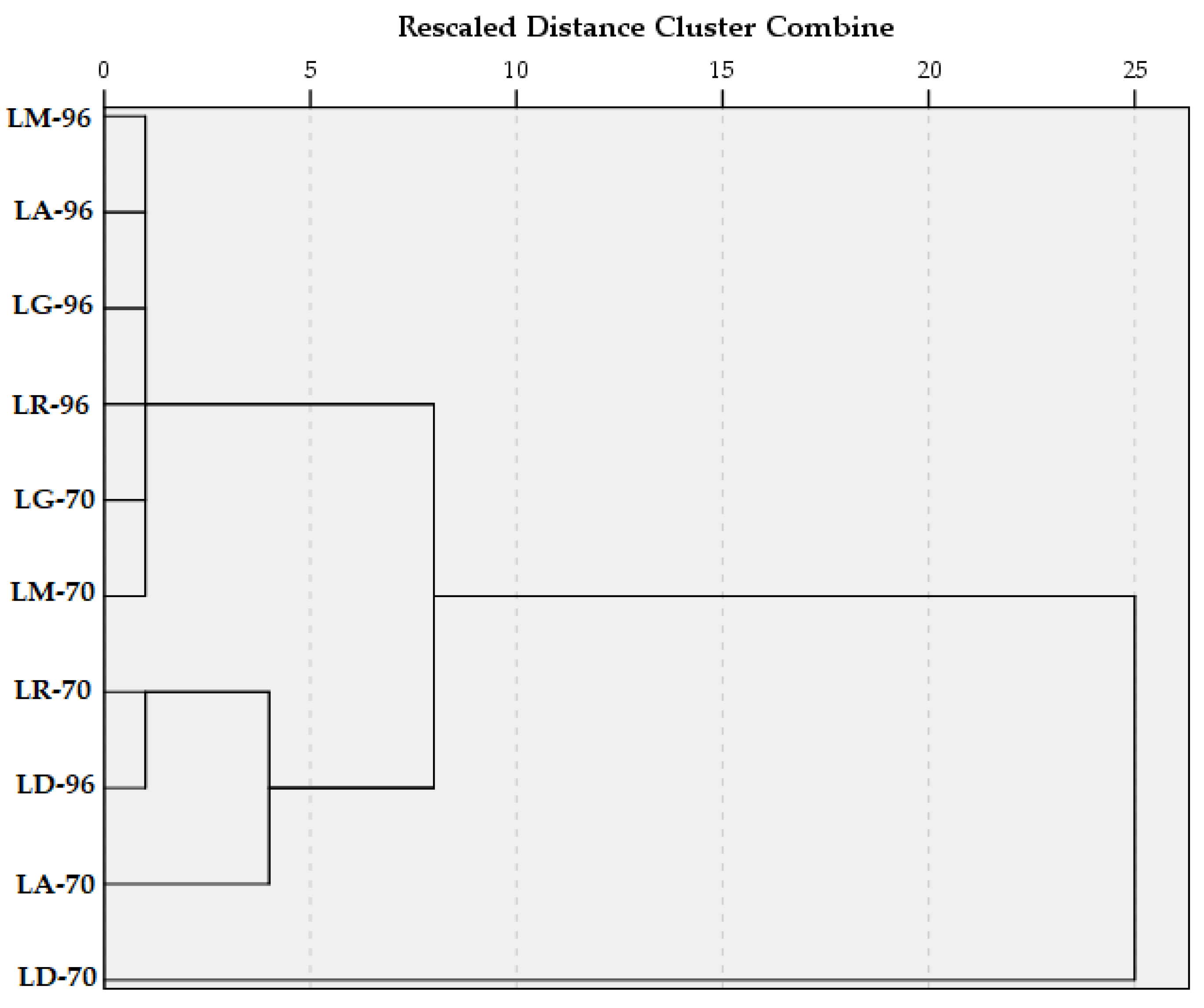
2. Results
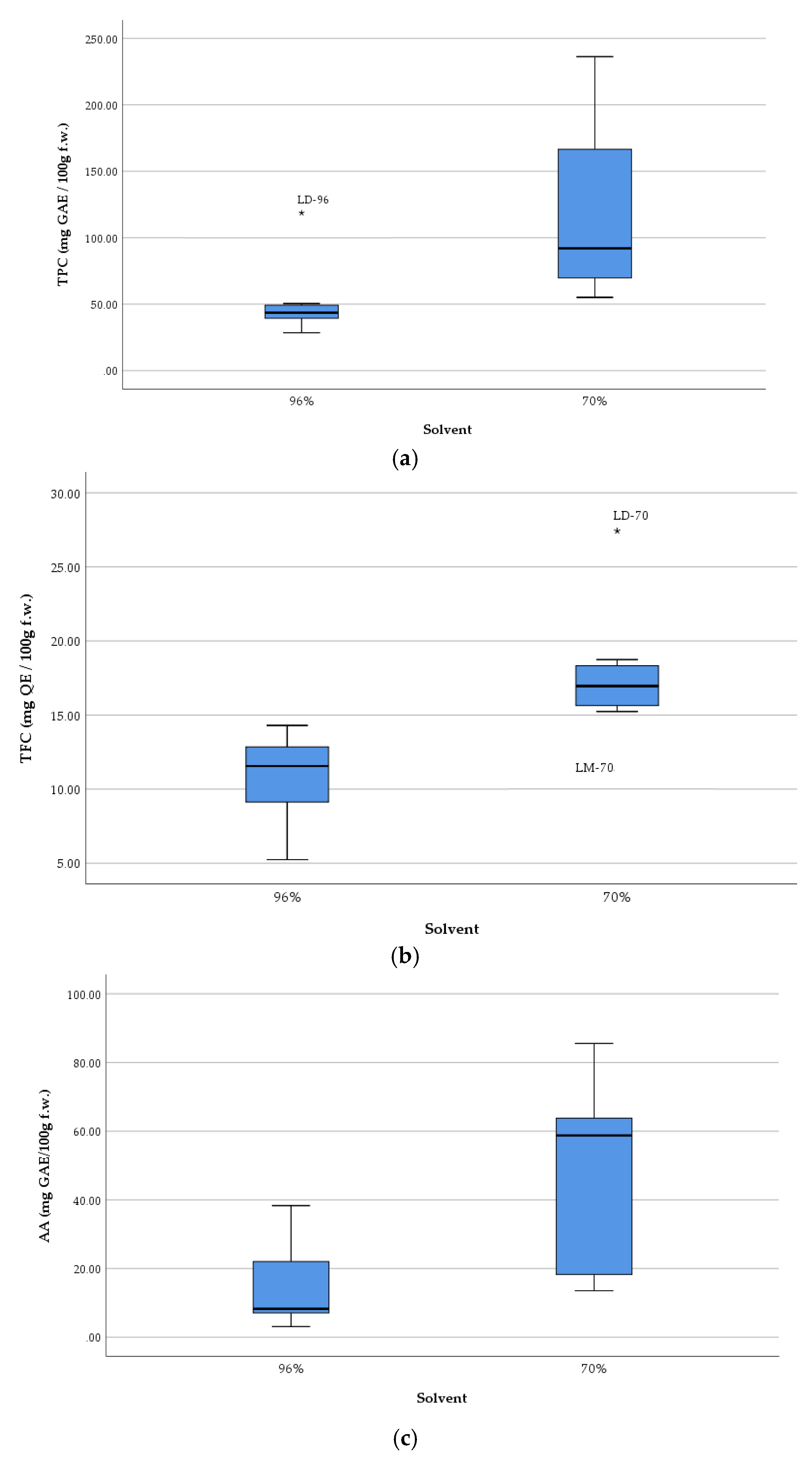
2.1. Total Phenolic Content (TPC)
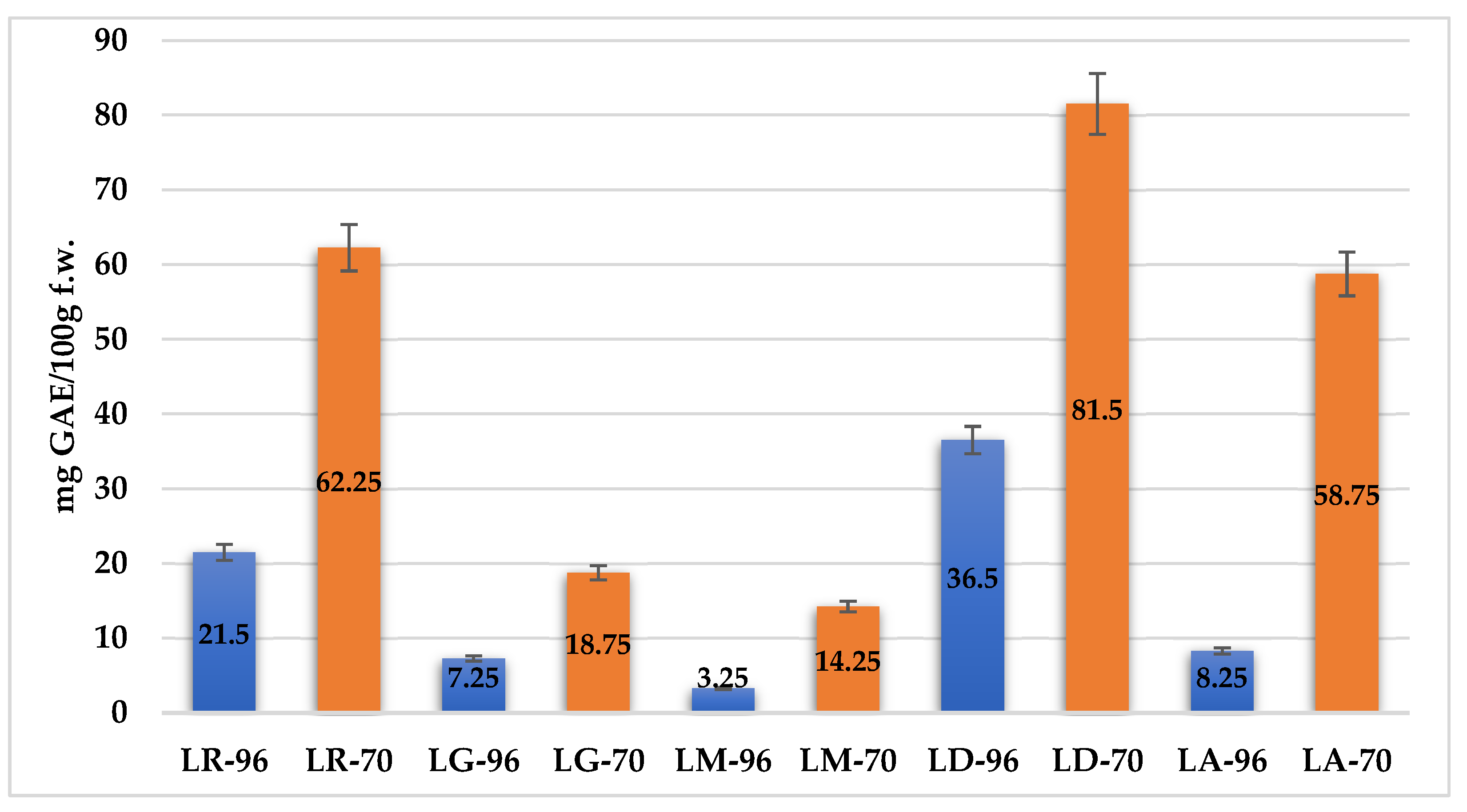
2.2. Antioxidant Activity (AA)
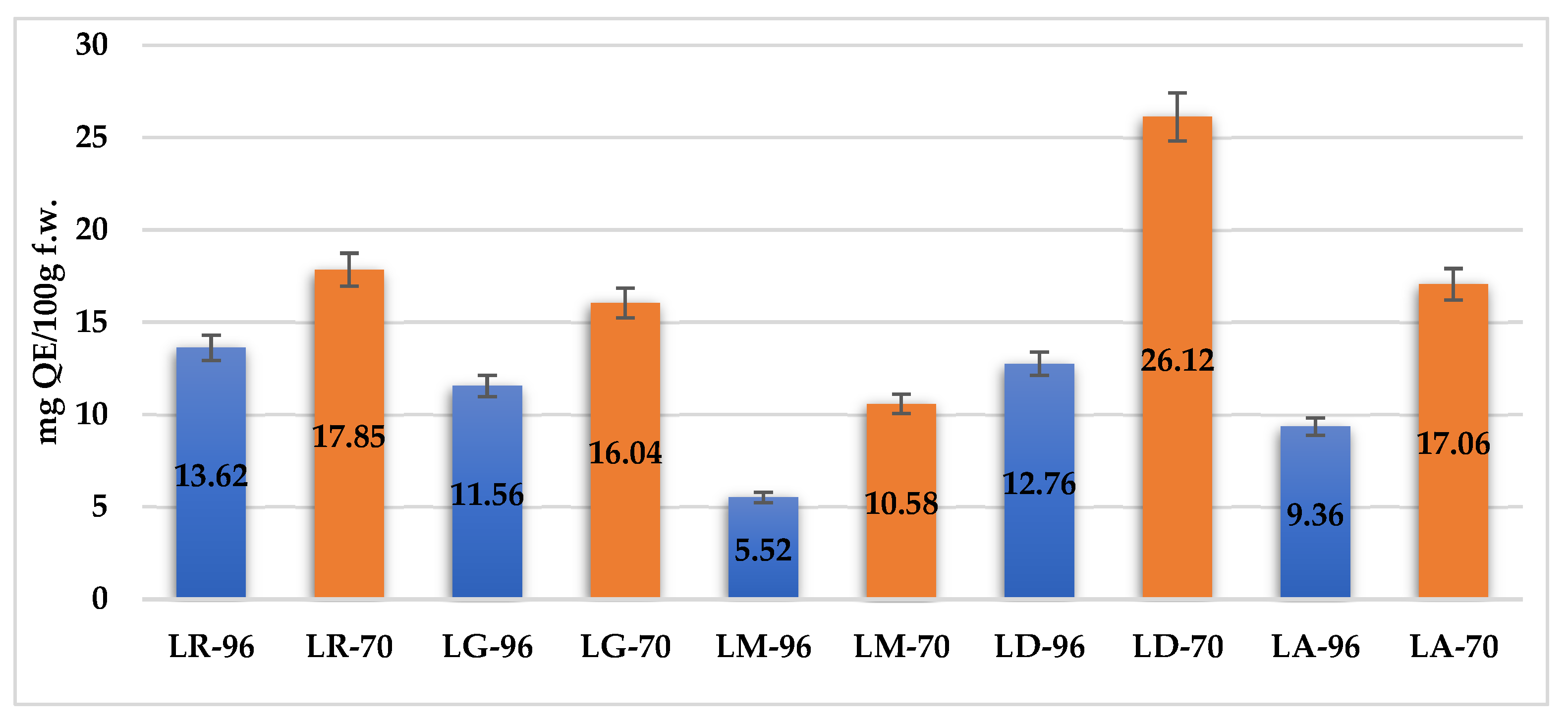
2.3. Total Flavonoid Content (TFC)
2.4. The Condensed Tannins Content (CTC)
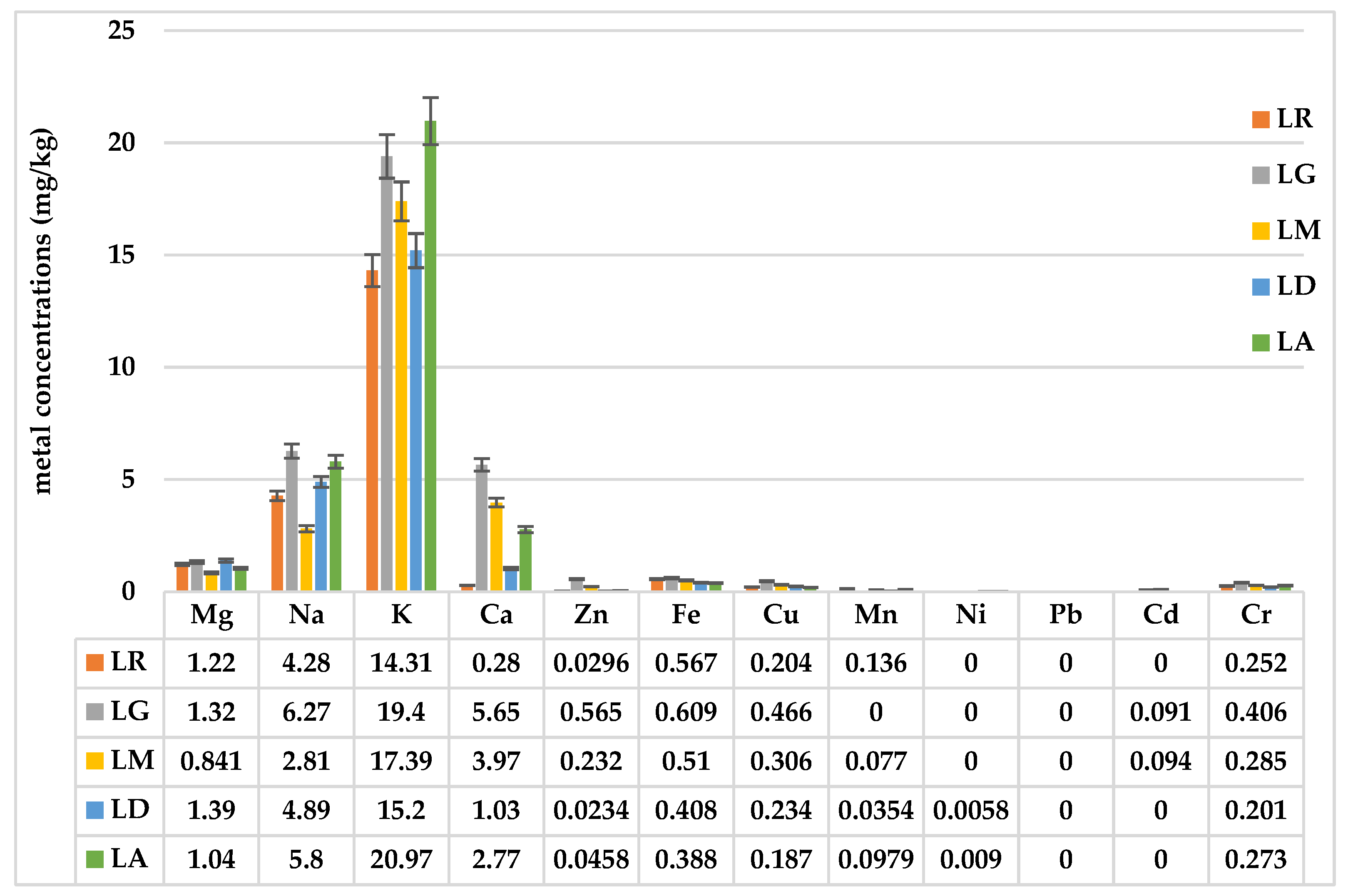
2.5. Metal Concentrations
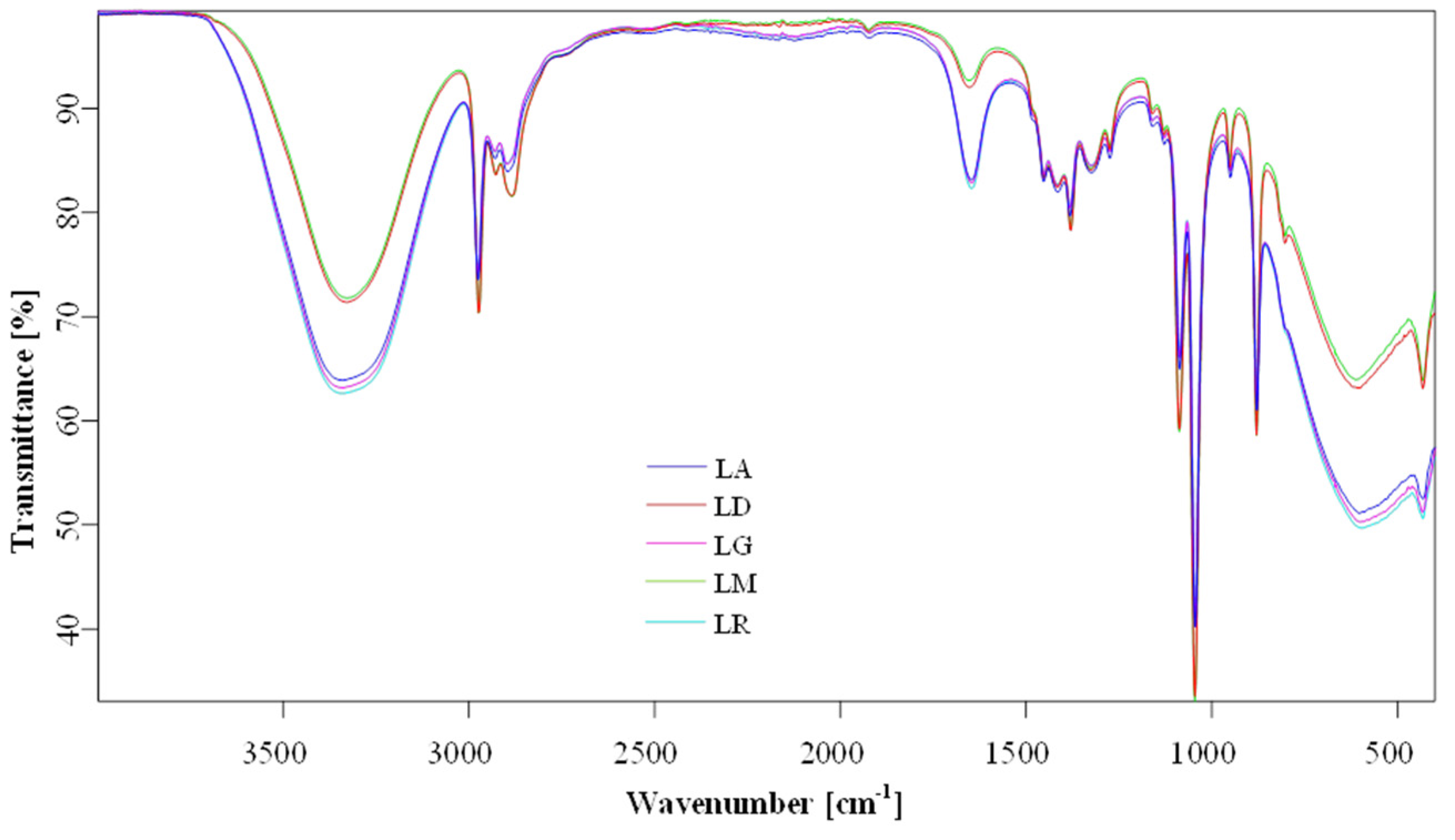
2.6. Fourier Transform Infrared (FTIR) Spectroscopy
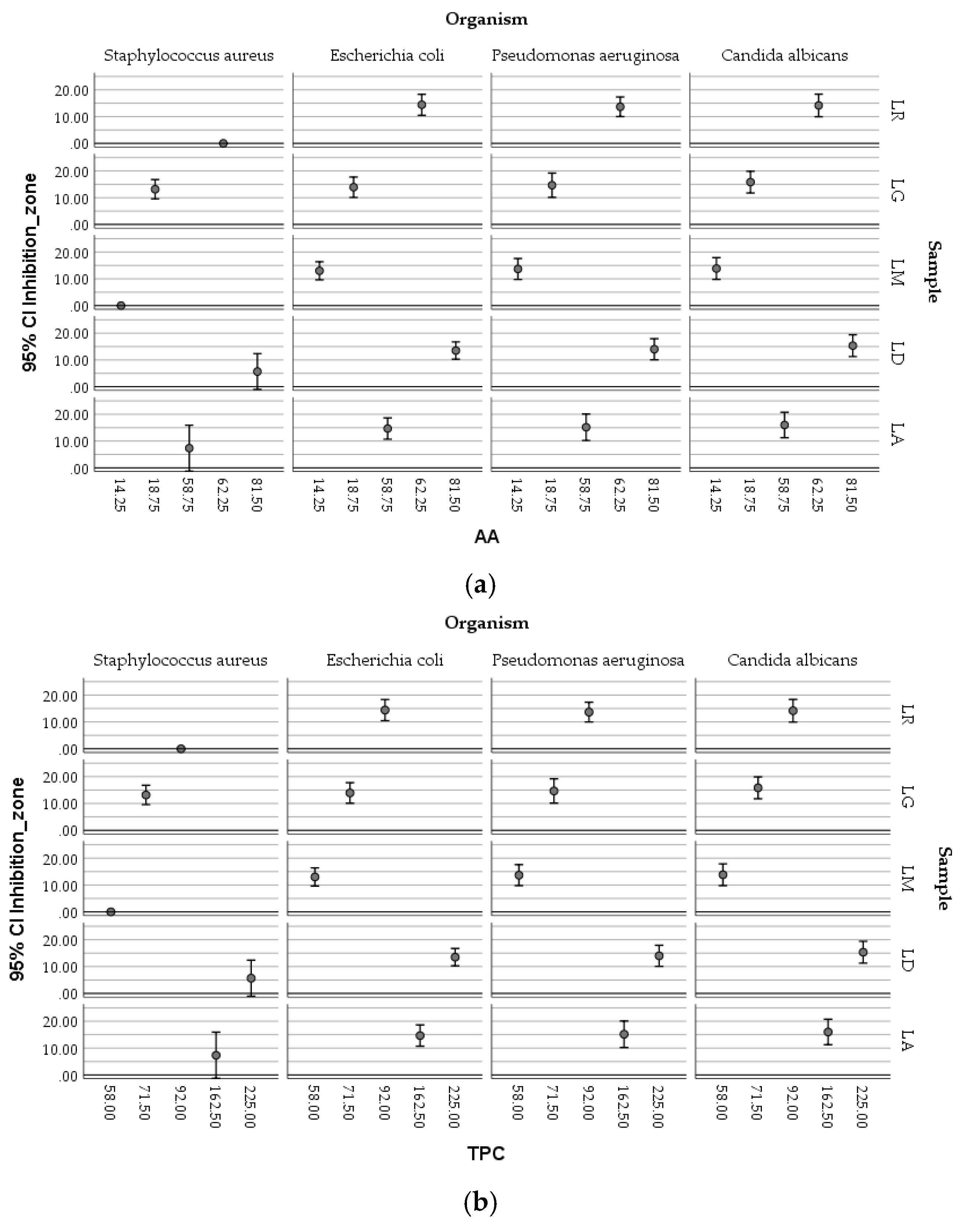
2.7. Antimicrobial Activity
2.7. Characteristics of Hydrogels Based on Lilium spp. Bulbs Macerates
2.8. Antioxidant Activity of Hydrogels HLD-70 and HLA-70
| No | Sample |
DPPH [μg/100g gel] |
| 1. | HLD-70 | 142.4 ±1.8% |
| 2. | HLA-70 | 86.8±6.3% |
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Sample Collection and Preparation
4.3. Total Polyphenol Content Analysis
4.4. Antioxidant Activity Analysis
4.5. Total Flavonoid Content Analysis
4.6. The Condensed Tannins Content (CTC)
4.7. Metal Concentrations Analysis
4.8. FTIR Spectroscopy Analysis
4.9. Antimicrobial Assay
4.10. Formulation of Dermatocosmetic Gels with Lilium spp. Bulbs Macerates
Preformulation Conditions
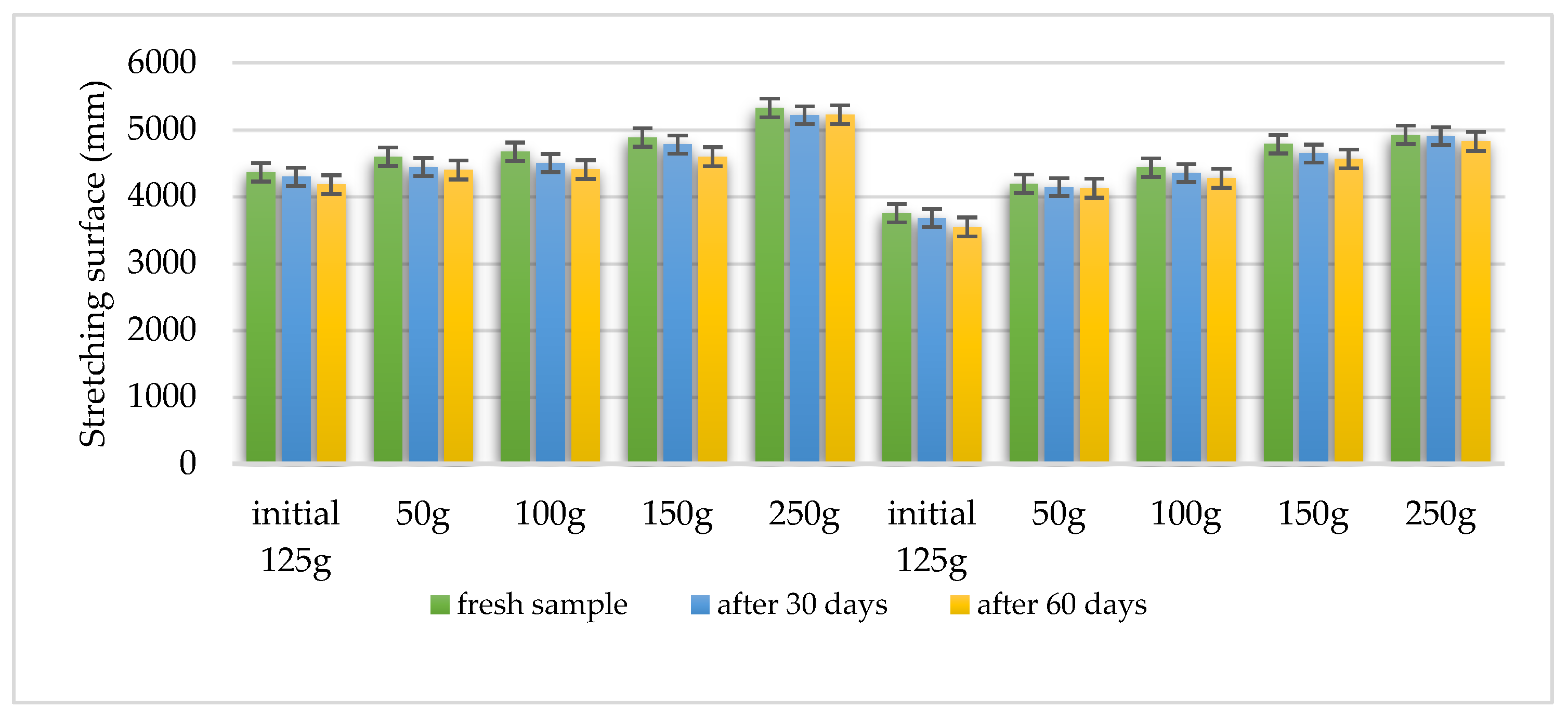
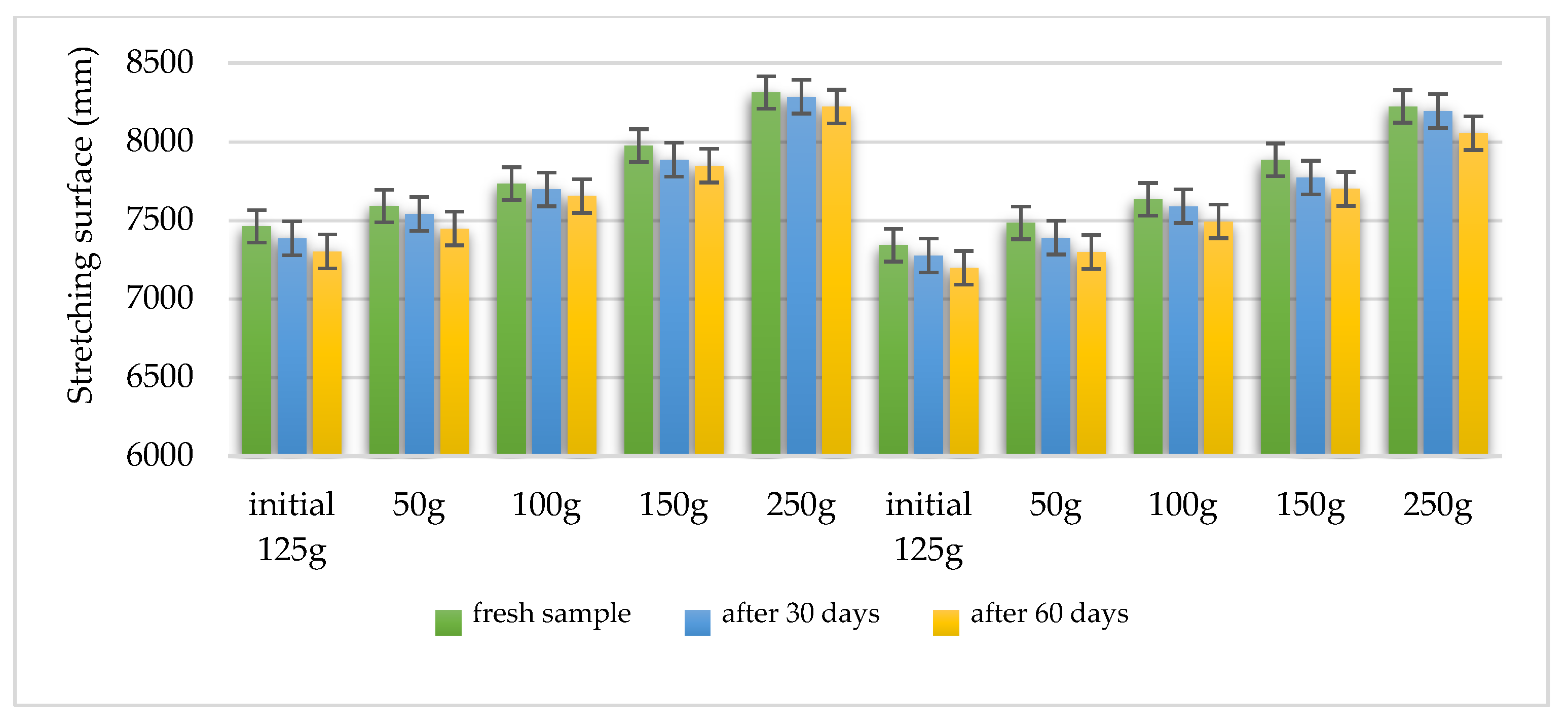
4.11. Characterization of Gel Preparations
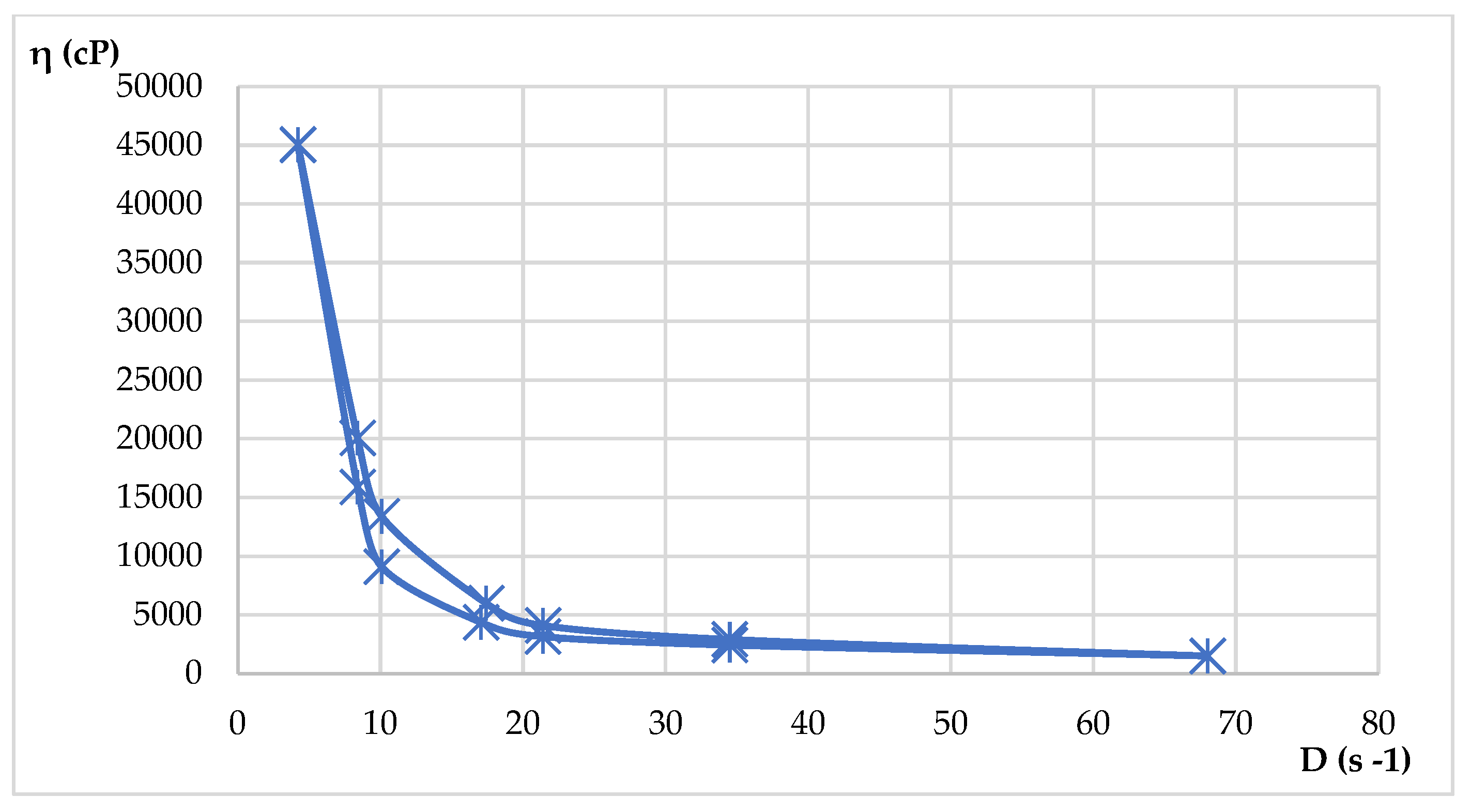
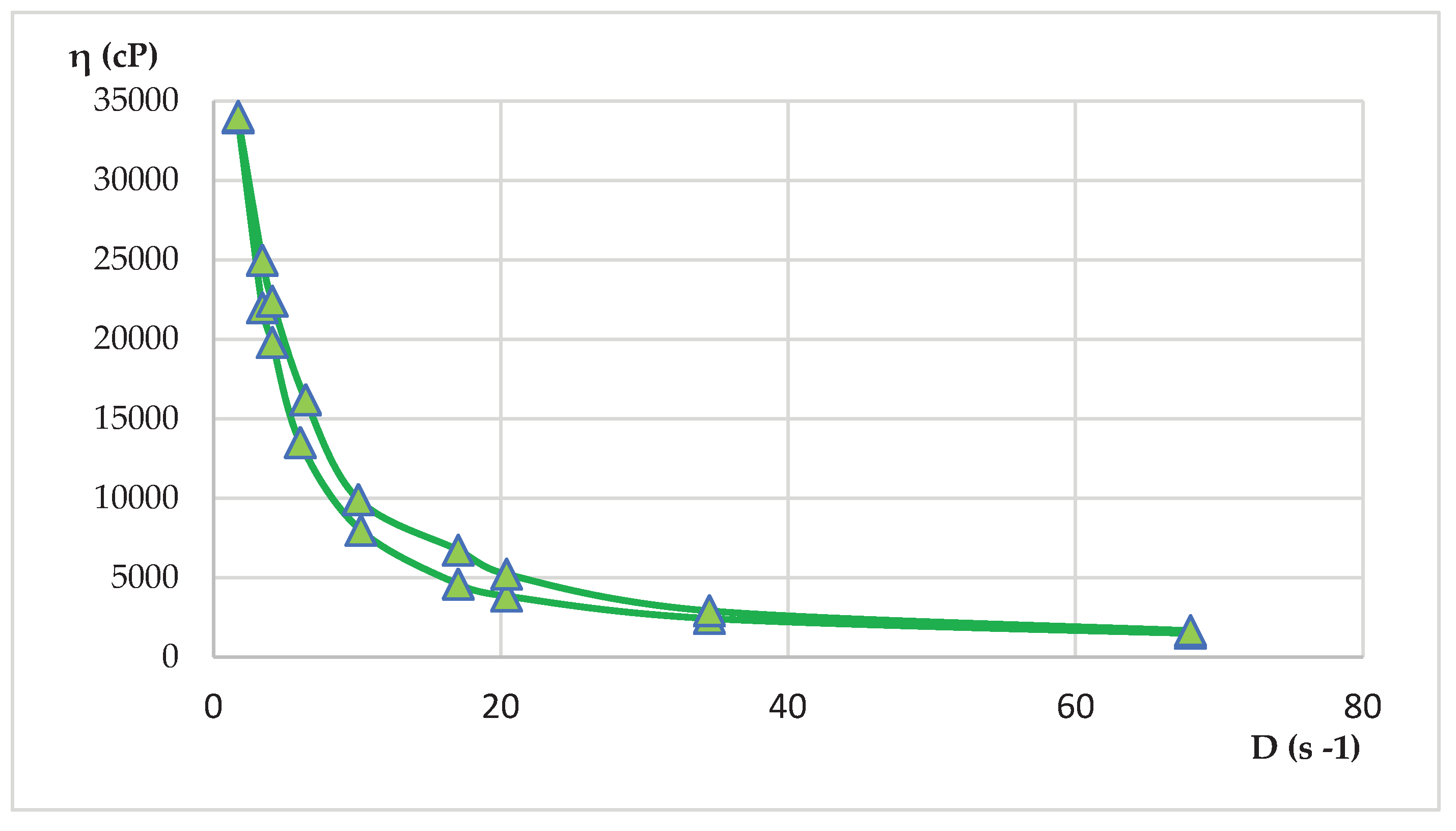
4.12. Rheological Measurements
4.13. Determination of Antioxidant Activity
4.14. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Zhou, Y.; An, R. Genus Lilium: A Review on Traditional Uses, Phytochemistry and Pharmacology. J. Ethnopharmacol. 2021, 270, 113851. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Zhang, Y.; Yan, L.; Guo, Y.; Niu, L. Phenolic Compounds and Antioxidant Activity of Bulb Extracts of Six Lilium Species Native to China. Molecules 2012, 17, 9361–9378. [Google Scholar] [CrossRef] [PubMed]
- Liang, Z.-X.; Zhang, J.-Z.; Sun, M.-Y.; Zhang, Y.-L.; Zhang, X.-H.; Li, H.; Shi, L. Variation of Phenolic Compounds and Antioxidant Capacities in Different Organs of Lilium pumilum. Nat. Prod. Commun. 2018, 13(6), 717–722. [Google Scholar] [CrossRef]
- Zaccai, M.; Yarmolinsky, L.; Khalfin, B.; Budovsky, A.; Gorelick, J.; Dahan, A.; Ben-Shabat, S. Medicinal Properties of Lilium candidum L. and Its Phytochemicals. Plants 2020, 9, 959. [Google Scholar] [CrossRef]
- Tang, Y.; Liu, Y.; Luo, K.; Xu, L.; Yang, P.; Ming, J. Potential Applications of Lilium Plants in Cosmetics: A Comprehensive Review Based on Research Papers and Patents. Antioxidants 2022, 11, 1458. [Google Scholar] [CrossRef] [PubMed]
- Shojaee-Aliabadi, S.; Abbasi, A.; Moradi, S.; Sanej, K.D.; Bazzaz, S.; Dehghan Nayeri, B.; Astanehe, N.; Mansourfar, R.; Kafsh, M.Z.; Ghorbanian, A.; Hosseini, H. Bioactive Compounds and Biological Activities of Madonna Lily (Lilium candidum L.). Springer Reference Series in Phytochemistry, 2023. [CrossRef]
- Patocka, J.; Navratilova, Z. Bioactivity of Lilium candidum L.: A Mini Review. Biomed. J. Sci. Tech. Res. 2019, 18(5), 13859-13860. [CrossRef]
- Tang, Y.-C.; Liu, Y.-J.; He, G.-R.; Cao, Y.-W.; Bi, M.-M.; Song, M.; Yang, P.-P.; Xu, L.-F.; Ming, J. Comprehensive Analysis of Secondary Metabolites in the Extracts from Different Lily Bulbs and Their Antioxidant Ability. Antioxidants 2021, 10, 1634. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Zhang, S.; Chen, Y.; Wang, G. Chemical Composition Analysis of Lilium brownii var. Viridulum Baker and the Effect of Postharvest Primary Processing on Its Quality. Appl. Sci. 2023, 13, 10795. [Google Scholar] [CrossRef]
- Al Fayad, A.; Othman, Y. Pre-Harvest Chemical Compounds Influence Lily (Lilium × elegans) Leaf and Flower Indigenous Phenols, Flavonoids and Gibberellic Acid Levels. Int. J. Plant Biol. 2024, 15, 551–560. [Google Scholar] [CrossRef]
- Song, Z.; Zhang, Y.; Luo, Y.; Ti, Y.; Wang, W.; Ban, Y.; Tang, Y.; Hou, Y.; Xu, L.; Ming, J.; Yang, P. Systematic evaluation on the physicochemical characteristics of a series polysaccharides extracted from different edible lilies by ultrasound and subcritical water. Front. Nutr. 2022, 9, 998942. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Zhang, Y.-L.; Liang, Z.-X.; Zhang, X.-X.; Si, G.-C.; Niu, L.-X. Phenolic Compounds and Antioxidant Properties of Bulb Extracts of Lilium leucanthum (Baker) Baker Native to China. Qual. Assur. Saf. Crops Foods 2015, 7(2), 141–151. [Google Scholar] [CrossRef]
- Loy, C.J.; Mahmood, K.; Saliou, C. Compositions Comprising Lilium candidum Extracts and Uses Thereof. U.S. Patent 2013, US 8,481,093 B2.
- Kaushik, N.; Kim, J.-H.; Nguyen, L.N.; Kaushik, N.K.; Choi, K.-A. Characterization of Bioactive Compounds Having Antioxidant and Anti-Inflammatory Effects of Liliaceae Family Flower Petal Extracts. J. Funct. Biomater. 2022, 13, 284. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A., Zielonka-Brzezicka, J., Pechaiko, D., Tkacz, M., Klimowicz, A. The evaluation of the antioxidant properties of Ginkgo biloba L. leaves after the end of the growing season. Pomeranian J. Life Sci., 2017, 63, 24-30.
- Biernacka, P., Adamska, I., Felisiak, K. The Potential of Ginkgo biloba as a Source of Biologically Active Compounds - A Review of the Recent Literature and Patents. Molecules, 2023, 28, 3993.
- Serrano, J., Puupponen-Pimiä, R., Dauer, A., Aura, A.M., Saura-Calixto, F. Tannins: Current knowledge of food sources, intake, bioavailability and biological effects. Molecular Nutrition and Food Research, 2009, 53, 2, 310-329.
- Wei, X.; Ding, H.; Wei, Q.; Liu, X.; Liu, H.; Li, H.; Yang, Y. Nutrient Uptake and Partitioning in Oriental Lily (Lilium spp. ‘Sorbonne’). Horticulturae 2023, 9(4), 473. [CrossRef]
- Thi N.N., Song H.S., Oh E.-J., Lee Y.-G., Ko J.-H., Kwon J. E., Kang S.-C., Lee D.-Y., Jung I.H., Baek N.-I., Phenylpropanoids from Lilium Asiatic hybrid flowers and their anti-inflammatory activities, Applied Biological Chemistry 60(5):1-7, 2017. [CrossRef]
- Munafo, J. P., Jr.; Gianfagna, T. J. Chemistry and Biological Activity of Steroidal Glycosides from the Lilium Genus. Nat. Prod. Rep. 2015, 32(3), 454–477. [Google Scholar] [CrossRef] [PubMed]
- Kendel A., Zimmermann B., Chemical Analysis of Pollen by FT-Raman and FTIR Spectroscopies, Front. Plant Sci., 11, 2020. [CrossRef]
- Shahzadi, T.; Anwar, M. R.; Shah, M. M.; Al-Sadi, A. M.; Zaman, Q.; Asif, M.; Ashraf, S.; Munawar, A.; Abid, M. Lilium philadelphicum Flower as a Novel Source of Antimicrobial Agents: Characterization, Bioactive Fractions and Mode of Action. Sustainability 2021, 13(15), 8471. [Google Scholar] [CrossRef]
- Stanciu, G.; Aonofriesei, F.; Lupșor, S.; Oancea, E.; Mititelu, M. Chemical Composition, Antioxidant Activity, and Antibacterial Activity of Black Poplar Buds’ Hydroalcoholic Extracts from Dobrogea Area. Molecules 2023, 28, 4920. [Google Scholar] [CrossRef] [PubMed]
- Stanciu, G.; Aonofriesei, F.; Lupșor, S.; Popescu, A.; Sîrbu, R. Study of Phenolic Compounds and Antimicrobial Activity of Lavandula angustifolia L. Flowers Extracts. Rev. Chim. (Bucharest) 2019, 70(5), 1800–1805. [Google Scholar] [CrossRef]
- Stanciu, G.; Rotariu, R.; Popescu, A.; Tomescu, A. Phenolic and Mineral Composition of Wild Chicory Grown in Romania. Rev. Chim. (Bucharest) 2019, 70(4), 1173–1177. [Google Scholar] [CrossRef]
- Stanciu, G.; Lupsor, S.; Tomescu, A.; Sîrbu, R. Evaluation of Antioxidant Capacities and Phenols Composition of Wild and Cultivated Berries. Rev. Chim. (Bucharest) 2019, 70(2), 373–377. [Google Scholar] [CrossRef]
- Rădulescu, C., Olteanu, R.L., Buruleanu, L.C., Nechifor (Tudorache), M., Dulamă, I.D., Știrbescu, R.M., Bucurică, I.A., Stănescu, S.G., Bănică, A.L. Polyphenolic Screening and the Antioxidant Activity of Grape Pomace Extracts of Romanian White and Red Grape Varieties. Antioxidants, 2024, 13(9):1133. [CrossRef]
- John, P.M.J, Thomas J.G., Chemistry and biological activity of steroidal glycosides from the Lilium genus, Nat Prod Rep 32, 454–477, 2015.
- Panus, E.; Miron, A.; Aprotosoaie, A. C.; Trifan, A.; Luca, S. V.; Vasincu, A.; Cioancă, O. Phytochemical Characterization, Antioxidant and Antimicrobial Activity of Vegetative Buds of Romanian Spruce, Picea abies (L.) H. Karst. Molecules 2024, 29(9), 2128. [CrossRef]
- Neculai, A.-M.; Stanciu, G.; Lepădatu, A.C.; Cima, L.-M.; Mititelu, M.; Neacsu, S.M. Development of New Dermato-Cosmetic Therapeutic Formulas with Extracts of Vinca minor L. Plants from the Dobrogea Region. Int. J. Mol. Sci. 2023, 24, 16234. [Google Scholar] [CrossRef]
- Foster, L.; Foppiani, J.A.; Xun, H.; Lee, D.; Utz, B.; Hernandez Alvarez, A.; Domingo-Escobar, M.J.; Taritsa, I.C.; Gavlasova, D.; Lee, T.C.; et al. Zinc in Dermatology: Exploring Its Emerging Role in Enhancing Botulinum Toxin Formulations and Clinical Efficacy. Curr. Issues Mol. Biol. 2024, 46, 12088–12098. [Google Scholar] [CrossRef] [PubMed]
- Mimaki, Y.; Satou, T.; Kuroda, M.; Sashida, Y;. Hatakeyama, Y. Steroidal saponins from the bulbs of Lilium candidum. Phytochemistry, 1999, 51(4), 567-573. [CrossRef]
- Zaccai, M., Yarmolinsky L., Khalfin B., Budovsky A., Gorelich J., Dahan A., Ben-Shabat S. Medicinal Properties of Lilium candidum L. and Its Phytochemicals, Plants (Basel). 2020, 9(8), 959. [CrossRef]
- Jiri Patocka, Zdenka Navratilova. Bioactivity of Lilium candidum L: A Mini Review. Biomed J Sci & Tech Res. 2019, 18(5), 13859-13862. [CrossRef]
- Al-Bayati N., Antiproliferative activity of Lilium candidum Alkaloid extract on human breast cancer cell line. Journal of Pharmaceutical Sciences and Research. 2018, 10(8), 2014-2016.
- Eisenreichova, E.; Haladova, M.; Mucaji, P.; Budesinsky, M.; Ubik, K. A new steroidal saponin from the bulbs of Lilium candidum L. Pharmazie, 2000, 55(7), 549–550.
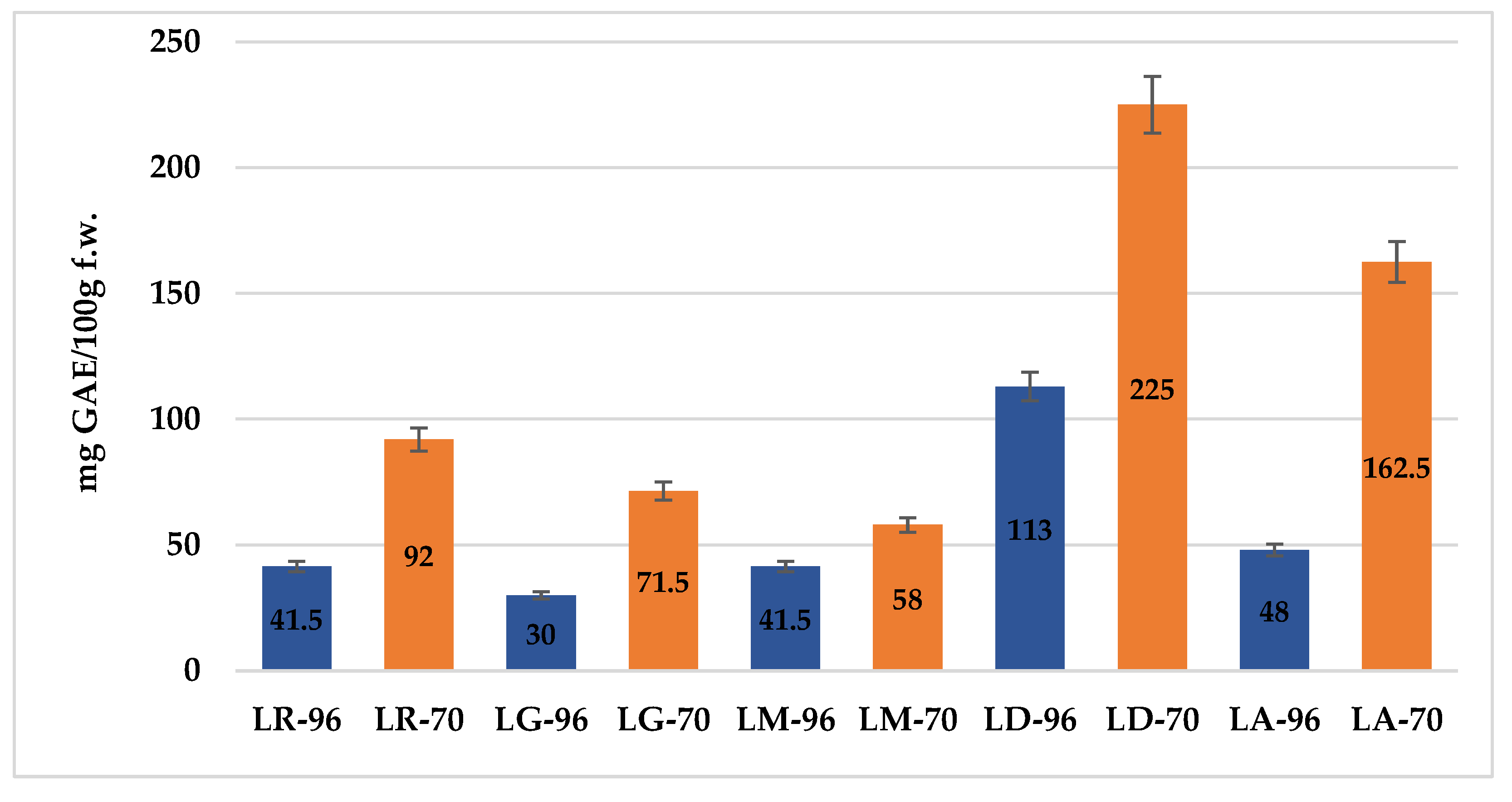
| Sample code | CTC* [mg CE/100g f.w.] |
| LR-70 | n.a. |
| LG-70 | n.a. |
| LA-70 | n.a. |
| LM-70 | 34.97 ± 17.48 |
| LD-70 | 46.62 ± 10.09 |
| Variables | TPC | AA | TFC |
| TPC | 1 | 0.907** | 0.839** |
| AA | 0.907** | 1 | 0.895** |
| TFC | 0.839** | 0.895** | 1 |
| Wavenumber [cm−1]/ Relative intensity* | Tentative assignments | ||||
| LR | LD | LG | LM | LA | |
| 878m | 879m | 878m | 879m | 878m | C-H bending of substituted aromatic ring |
| 949w | 952w | 950w | 952w | 950w | C-H bending of substituted aromatic ring |
| 1044s | 1045s | 1044s | 1045s | 1044s | O-H of carboxyl group |
| 1086m | 1087m | 1086m | 1087m | 1086m | C-O stretching of single bond of alcohol |
| 1128w | 1129w | 1128w | 1129w | 1128w | C-C stretching of substituted ring |
| 1159w | 1160w | 1160w | 1160w | 1160w | C-O stretching vibration combined with aromatic ring |
| 1274w | 1274w | 1274w | 1274w | 1274w | C-C in-phase (symmetric motion) ring stretching |
| 1324w | 1325w | 1325w | 1326w | 1324w | C-O stretching vibration combined with aromatic ring |
| 1382w | 1380w | 1382w | 1380w | 1382w | bending vibrations of CH2 |
| 1414w | 1416w | 1415w | 1416w | 1415w | bending vibration of the O-H of alcohol |
| 1453w | 1453w | 1453w | 1453w | 1453w | bending vibrations of CH2 |
| 1647w | 1652w | 1647w | 1654w | 1647w | C=C stretching vibration of double bond |
| 2897w | 2885w | 2897w | 2884w | 2896w | bending vibration of the O-H of phenol |
| 2929w | 2927w | 2930w | 2928w | 2929w | C-O/C=O of carbonyl/carboxyl groups |
| 2975m | 2973m | 2975w | 2973m | 2975m | bending vibration of the O-H of phenol |
| 3335s | 3328m | 3342m | 3329m | 3341s | bending vibration of the O-H of alcohol |
| *w-weak; m-medium; s-strong | |||||
| Organism | Volume [µL] | Sample –Inhibition Zone [mm] | |||||
|
LA-70 Sensitivity (S)/ Resistance (R) |
LD-70 Sensitivity (S)/ Resistance (R) |
LG-70Sensitivity (S)/ Resistance (R) |
LM-70Sensitivity (S)/ Resistance (R) |
LR-70Sensitivity (S)/ Resistance (R) |
ControlSensitivity (S)/ Resistance (R) |
||
|
Staphylococcus aureus ATCC 25922 |
5 | 0 (R) | 0 (R) | 8 (S) | 0 (R) | 0 (R) | 0 (R) |
| 10 | 0 (R) | 0 (R) | 11 (S) | 0 (R) | 0 (R) | 0 (R) | |
| 15 | 0 (R) | 0 (R) | 13 (S) | 0 (R) | 0 (R) | 0 (R) | |
| 20 | 12 (S) | 10 (S) | 14 (S) | 0 (R) | 0 (R) | 0 (R) | |
| 25 | 15 (S) | 10 (S) | 15 (S) | 0 (R) | 0 (R) | 0 (R) | |
| 30 | 17 (S) | 14 (S) | 18 (S) | 0 (R) | 0 (R) | 0 (R) | |
|
Escherichia coli ATCC 25923 |
5 | 9 (S) | 9 (S) | 8 (S) | 8 (S) | 8 (S) | 8 (S) |
| 10 | 12 (S) | 11(S) | 12(S) | 12 (S) | 12 (S) | 10 (S) | |
| 15 | 14(S) | 13 (S) | 13-14(S) | 12 (S) | 15-16 (S) | 12 (S) | |
| 20 | 16 (S) | 15 (S) | 15(S) | 13 (S) | 16 (S) | 14 (S) | |
| 25 | 18 (S) | 16 (S) | 17(S) | 16 (S) | 17 (S) | 15 (S) | |
| 30 | 19 (S) | 17 (S) | 18(S) | 17 (S) | 18 (S) | 16 (S) | |
|
Pseudomonas aeruginosa ATCC 27853 |
5 | 8 (S) | 9 (S) | 8 (S) | 8 (S) | 8 (S) | 7 (S) |
| 10 | 12 (S) | 11 (S) | 12 (S) | 11(S) | 12 (S) | 9 (S) | |
| 15 | 15 (S) | 13 (S) | 14 (S) | 13 (S) | 13 (S) | 10 (S) | |
| 20 | 16 (S) | 15 (S) | 16 (S) | 16 (S) | 15 (S) | 14 (S) | |
| 25 | 19 (S) | 17 (S) | 18 (S) | 16 (S) | 16 (S) | 15 (S) | |
| 30 | 21(S) | 19(S) | 20 (S) | 18 (S) | 18 (S) | 16 (S) | |
|
Candida albicans ATCC 10231 |
5 | 10 (S) | 10 (S) | 10 (S) | 9 (S) | 9 (S) | 9 (S) |
| 10 | 13 (S) | 12 (S) | 13 (S) | 11(S) | 11(S) | 11(S) | |
| 15 | 14 (S) | 15 (S) | 15 (S) | 13(S) | 13 (S) | 13 (S) | |
| 20 | 17 (S) | 16 (S) | 18(S) | 14(S) | 15 (S) | 15 (S) | |
| 25 | 20 (S) | 19 (S) | 19 (S) | 16 (S) | 17 (S) | 17 (S) | |
| 30 | 22 (S) | 20 (S) | 20 (S) | 20(S) | 20 (S) | 20 (S) | |
| Variables | TPC | AA | TFC | CTC | Inhibition zone |
| TPC | 1 | 0.861** | 0.876** | 0.404** | 0.056 |
| AA | 0.861** | 1 | 0.865** | 0.171 | -0.011 |
| TFC | 0.876** | 0.865** | 1 | 0.310** | 0.063 |
| CTC | 0.404** | 0.171 | 0.310** | 1 | -0.110 |
| Inhibition zone | 0.056 | -0.011 | 0.063 | 0.110 | 1 |
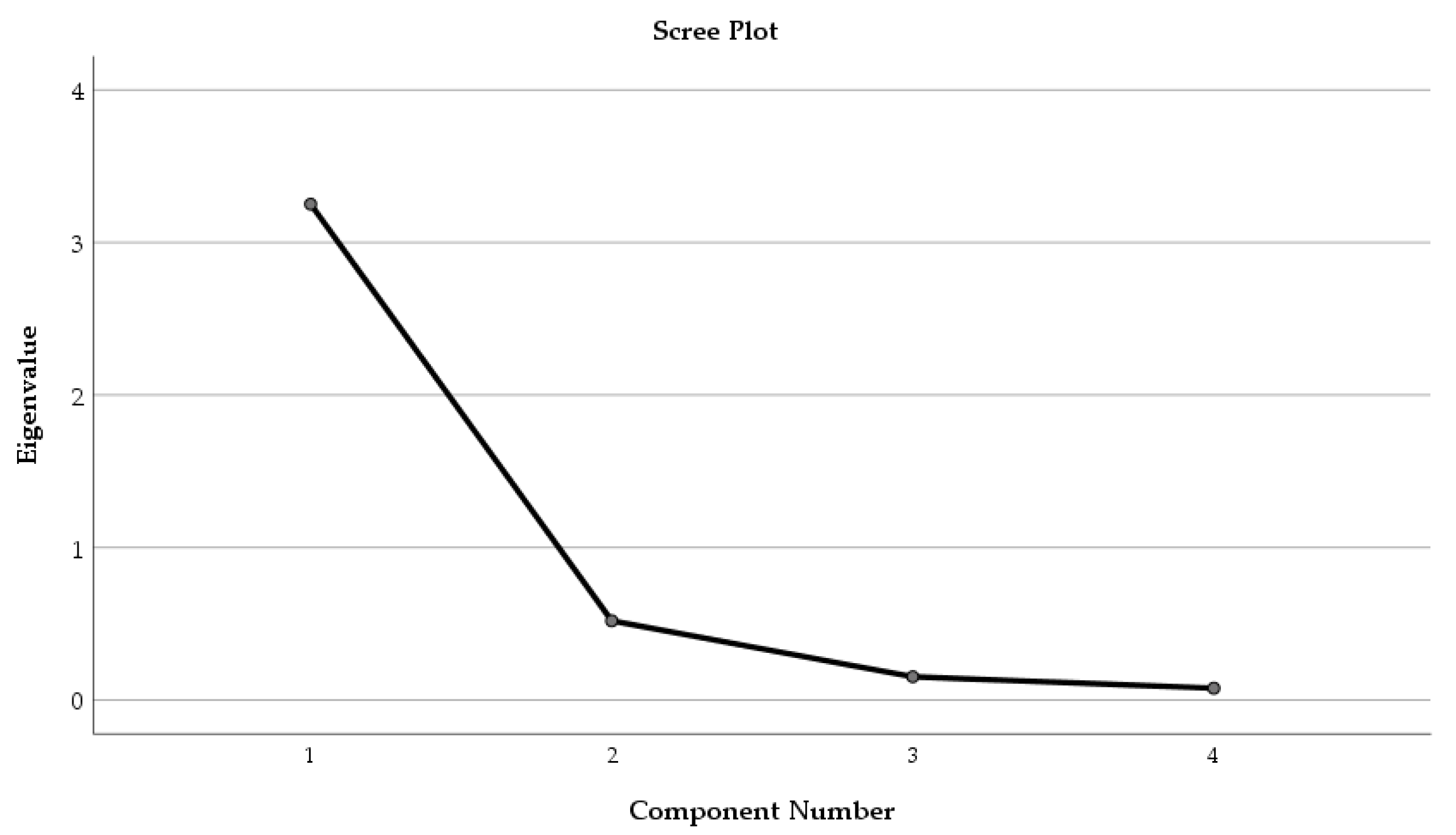
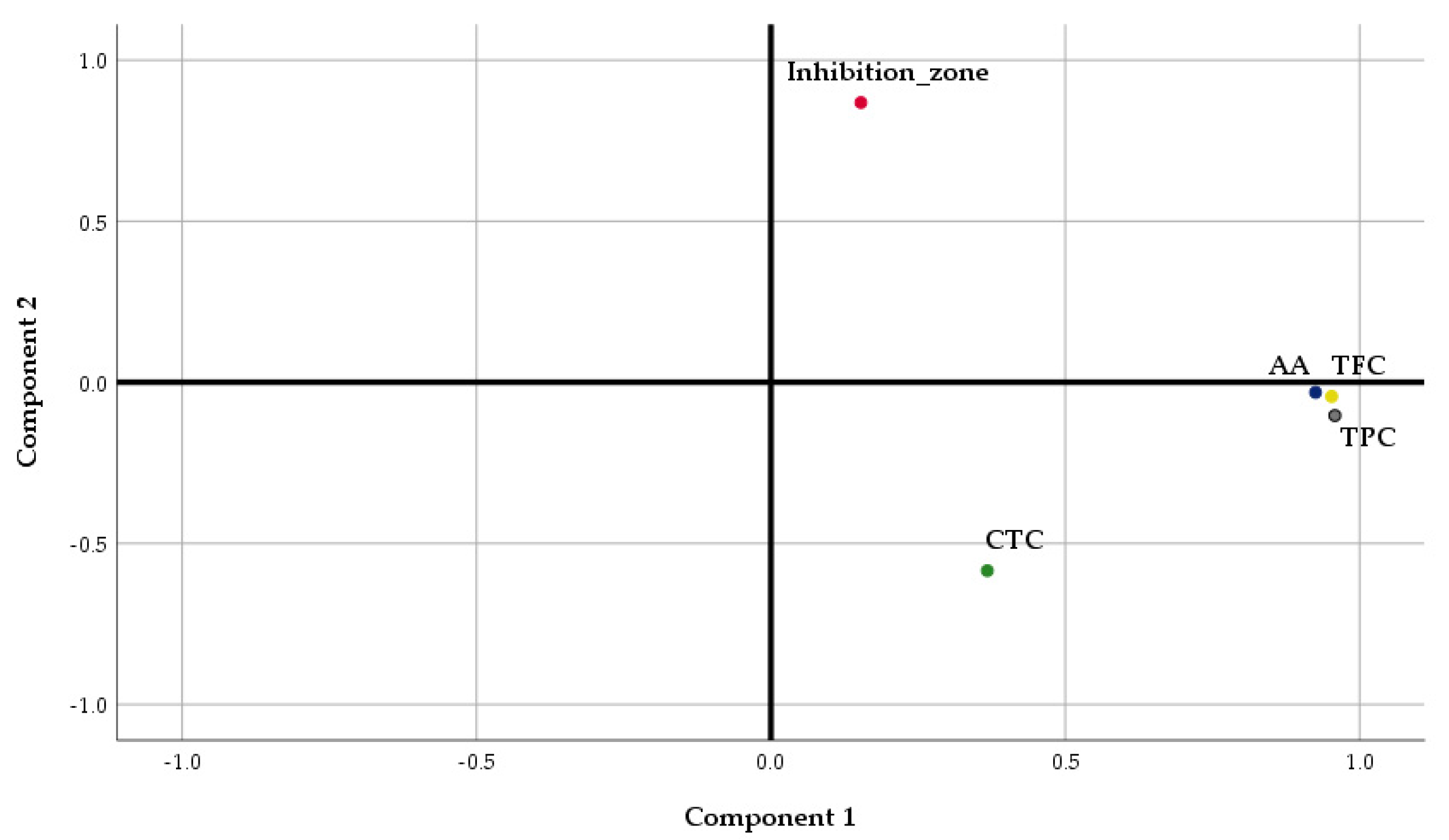
| Factor | Component 1 | Component 2 |
| Eigenvalue | 2.875 | 1.073 |
| Cumulative variance (%) | 56.78 | 78.96 |
| TPC | 0.958 | -0.103 |
| AA | 0.925 | -0.031 |
| TFC | 0.952 | -0.044 |
| CTC | 0.368 | -0.585 |
| Inhibition zone | 0.153 | 0.868 |
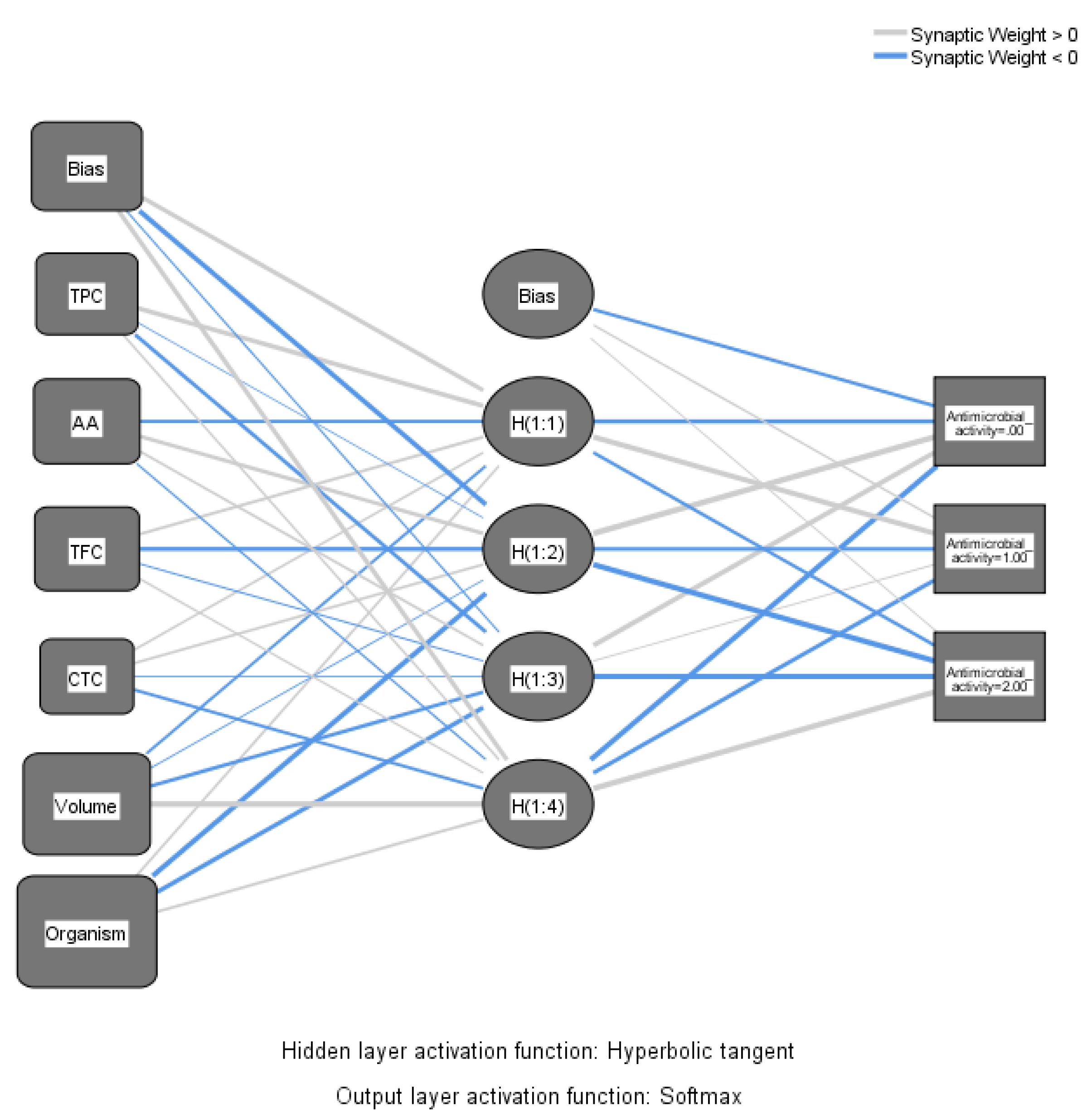
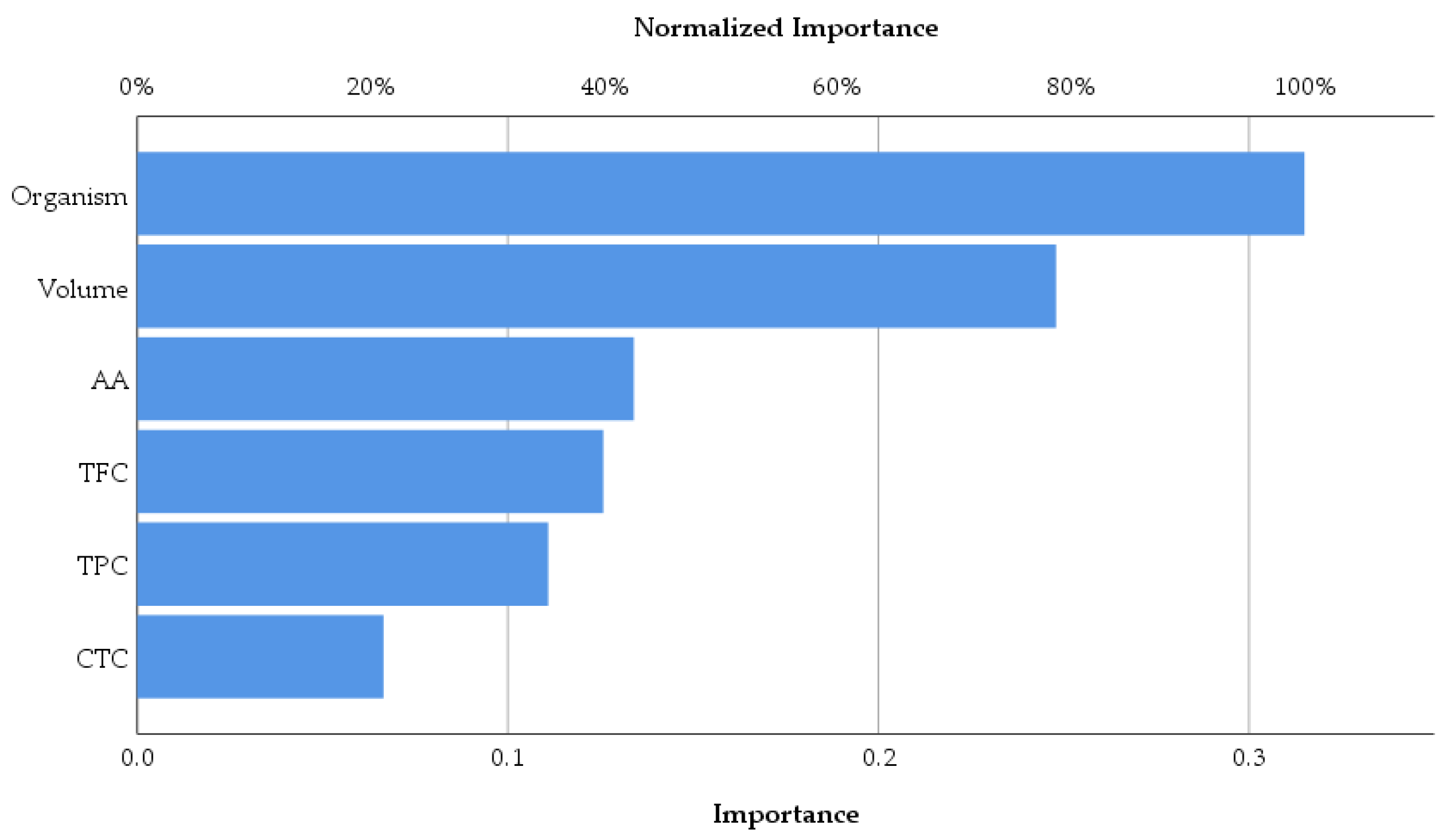
| Predictor | Hidden Layer 1 | Output Layer (strengthens of antimicrobial activity) | ||||||
| H(1:1) | H(1:2) | H(1:3) | H(1:4) | 0 | 1 | 2 | ||
| Input Layer | (Bias) | 1.762 | -2.251 | -0.280 | 2.043 | |||
| TPC | 2.062 | -0.004 | -1.059 | 0.394 | ||||
| AA | -0.758 | 1.270 | 0.618 | -0.281 | ||||
| TFC | 0.687 | -1.309 | -0.145 | 0.329 | ||||
| CTC | 0.498 | 0.623 | -0.063 | -0.704 | ||||
| Volume | -0.626 | -0.086 | -0.947 | 4.049 | ||||
| Organism | 0.561 | -2.297 | -2.023 | 0.634 | ||||
| Hidden Layer 1 | (Bias) | -0.940 | 0.345 | 0.104 | ||||
| H(1:1) | -1.633 | 2.544 | -0.764 | |||||
| H(1:2) | 4.430 | -1.155 | -3.076 | |||||
| H(1:3) | 2.251 | 0.121 | -2.558 | |||||
| H(1:4) | -2.416 | -1.490 | 3.636 | |||||
| Characteristics | Formula HLD-70 | Formula HLA-70 |
| Organoleptic evaluation – after 24 h |
appearance: homogeneous, translucent; color: dark yellow; smell: specific |
appearance: homogeneous, translucent; color: yellow-light; smell: specific |
| Organoleptic evaluation – after 30 days | constant initial characteristics | constant initial characteristics |
| Organoleptic evaluation – after 60 days | constant initial characteristics | constant initial characteristics |
| pH – after 24 h | 4.6 – 4.9 | 4.8 – 5.0 |
| pH – after 30 days | 5.0 – 5.2 | 5.1 – 5.4 |
| pH – after 60 days | 5.4 | 5.6 |
| Viscosity – after 24 h | 690 ± 0.88 mPa·s | 642 ± 0.88 mPa·s |
| Viscosity – after 30 days | 670 ± 0.25 mPa·s | 628 ± 0.22 mPa·s |
| Viscosity – after 60 days | 628 ± 0.76 mPa·s | 608 ± 0.46 mPa·s |
| Sample |
Shear spead D [s-1] The interval |
Viscosity ƞ [cP] The interval |
Shear stress τ [mPa] The interval |
| HLD-70 | 6.10 – 78 | 2600 – 34200 | 682500 – 140380 |
| HLA-70 | 5.8- 62 | 820 - 48812 | 48400 – 146820 |
| Sample |
K Consistency coefficient |
n Flow coefficient |
R Correlation coefficient Ostwald de Waele |
| HLD-70 | 14.624 | 0.5221 | 0.9973 |
| HLA-70 | 12.164 | 0.4632 | 0.9982 |
| Metal | Concentration domain (mg/L) | R2 | Calibration curve equation |
| Cadmium | 0.0004 – 0.004 | 0.9989 | y = 0.0122973x + 0.0422208 |
| Calcium | 40.00 – 200.00 | 0.9996 | y = 0.000138x + 0.0000701 |
| Chromium | 0.002 – 0.02 | 0.9913 | y = 0.0138310x + 0.0125257 |
| Copper | 0.003 – 0.03 | 0.9958 | y = 0.1937482x + 0.0141176 |
| Iron | 0.05 – 2.0 | 0.9929 | y = 0.0007637x + 0.0308354 |
| Lead | 0.01 – 0.1 | 0.9976 | y = 0.0145906x+ 0.0042433 |
| Magnesium | 1.0 – 5.0 | 0.9932 | y = 0.0062880x +0.0571131 |
| Manganese | 0.0015 – 0.015 | 0.9950 | y = 0.0129916x + 0.0202151 |
| Nickel | 0.007 – 0.07 | 0.9926 | y = 0.0107338 x + 0.0041630 |
| Potassium | 1.0 – 5.0 | 0.9975 | y = 0.0013933x + 0.0024000 |
| Sodium | 5.0 – 25.0 | 0.9966 | y = 0.0000076x + 0.0034889 |
| Zinc | 0.0005 – 0.005 | 0.9925 | y = 0.3079303x + 0.0533993 |
| Components | Mass [g] | |
| HLD-70 | HLA-70 | |
| Carbopol 940 | 2.0 | 2.0 |
| Glycerine | 5 | 5 |
| Triethanolamine | q.s** | q.s** |
| Hydroalcoholic macerateof Lilium spp. bulbs | 5.0 | 5.0 |
| Purified water | until 100 | until 100 |
| η = f(D) (2) |
D = f(τ) (3) |
D = ωxR (4) |
τ = ηxD (5) |
τ = k x Dn (6) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




