Submitted:
23 May 2025
Posted:
26 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. Endoscopic Evaluation
2.3. Histopathological Evaluation
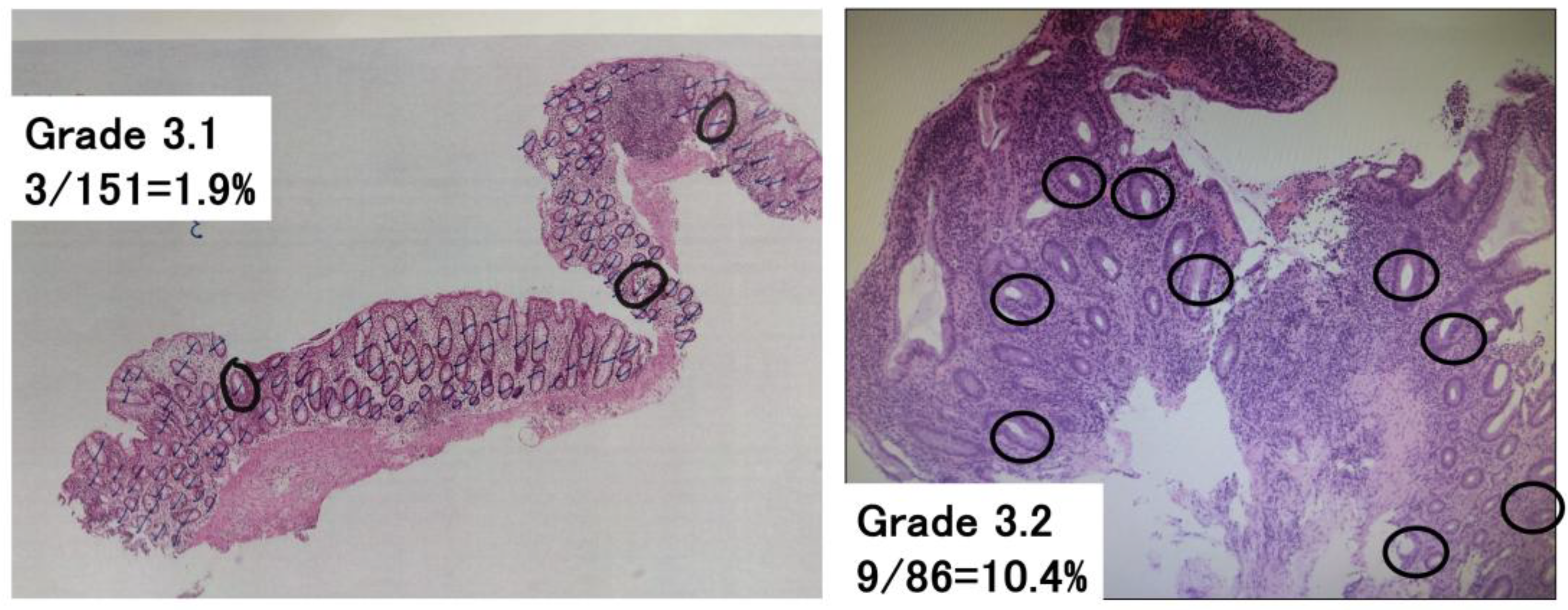
2.4. Komagane Evaluation Method of Geboes Score Grade 3
2.5. Raters
2.6. Reliability Testing
2.7. Statistical Analysis
3. Results
3.1. Patients’ Clinical Characteristics
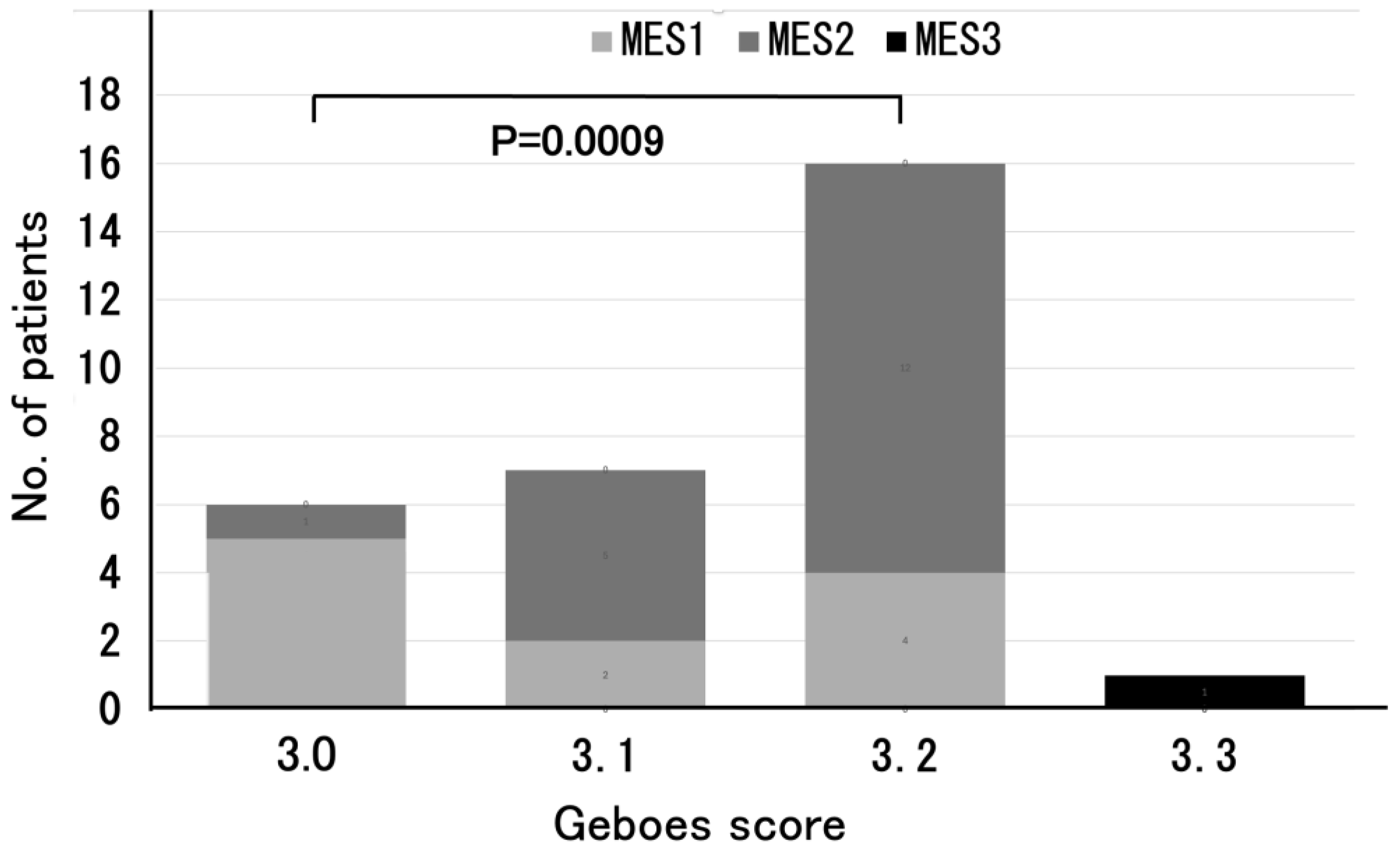
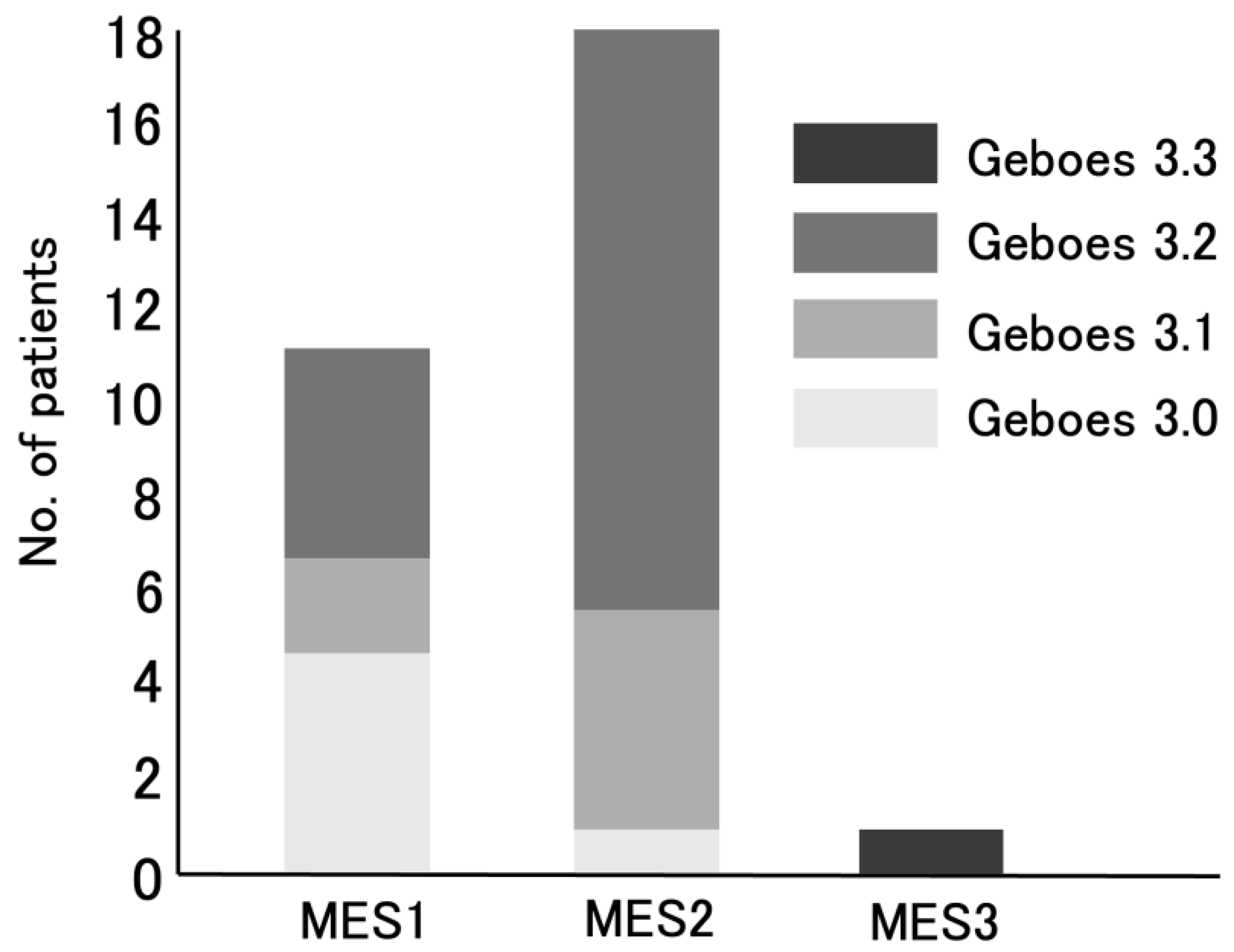
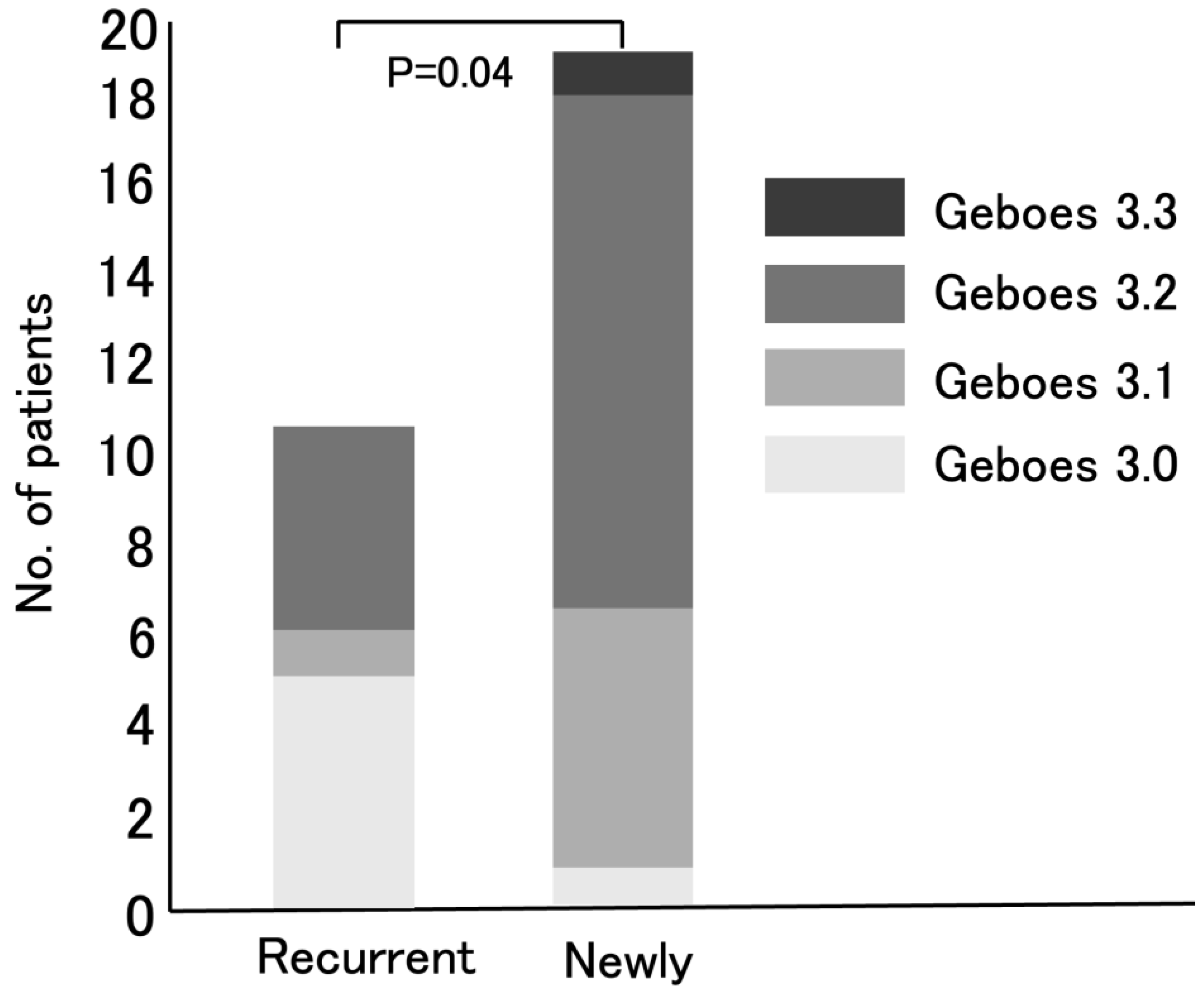
3.2. The Distribution of Geboes Score Grade 3
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Turner, D.; Ricciuto, A.; Lewis, A.; D’Amico, F.; Dhaliwal, J.; Griffiths, AM.; Bettenworth, D.; Sandborn, W.J.; Sands, B.E.; Reinisch, W.; et al. International Organization for the Study of IBD.STRIDE-II: an update on the selecting therapeutic targets in inflammatory bowel disease (STRIDE) initiative of the international organization for the study of IBD (IOIBD): determining therapeutic goals for treat-to-target strategies in IBD. Gastroenterology 2021, 160, 1570–1583. [Google Scholar] [PubMed]
- Sleiman, J.; Bassi, M.; Tsipotis, E.; Charabaty, A. Medical treatment options for ulcerative colitis. Clin. Colon. Rectal. Surg. 2022, 35, 428–436. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Loftus, E.V. Jr.; Limketkai, B.N.; Haydek, J.P.; Agrawal, M.; Scott, F.I.; Ananthakrishnan, A.N. AGA Clinical Guidelines Committee. Electronic address: clinicalpractice@gastro.org. AGA living clinical practice guideline on pharmacological management of moderate-to-severe ulcerative colitis. Gastroenterology, 2024; 167, 1307–1343. [Google Scholar]
- Nakase, H.; Sato, N.; Mizuno, N.; Ikawa, Y. The influence of cytokines on the complex pathology of ulcerative colitis. Autoimmun. Rev. 2022, 21, 103017. [Google Scholar] [CrossRef] [PubMed]
- Allocca, M.; Furfaro, F.; Fiorino, G.; Gilardi, D.; D’Alessio, S.; Danese, S. Can IL-23 be a good target for ulcerative colitis? Best Pract. Res. Clin. Gastroenterol. 2018, 32-33, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Geboes, K.; Riddell, R.; Ost, A.; Jensfelt, B.; Persson, T.; Löfberg, R. A reproducible grading scale for histological assessment of inflammation in ulcerative colitis. Gut 2000, 47, 404–409. [Google Scholar] [CrossRef] [PubMed]
- Bressenot, A.; Salleron, J.; Bastien, C.; Danese, S.; Boulagnon-Rombi, C.; Peyrin-Biroulet, L. Comparing histological activity indexes in UC. Gut 2015, 64, 1412–1418. [Google Scholar] [CrossRef] [PubMed]
- Rachmilewitz, D. Coated mesalazine (5-aminosalicylic acid) versus sulphasalazine in the treatment of active ulcerative colitis: a randomised trial. BMJ. 1989, 298, 82–86. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, K.W.; Tremaine, W.J.; Ilstrup, D.M. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N. Engl. J. Med. 1987, 317, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
- Kundel, H.L.; Polansky, M. Measurement of observer agreement. Radiology 2003, 228, 303–308. [Google Scholar] [CrossRef] [PubMed]
- Lemmens, B.; Arijs, I.; Van Assche, G.; Sagaert, X.; Geboes, K.; Ferrante, M.; Rutgeerts, P.; Vermeire, S.; De Hertogh, G. Correlation between the endoscopic and histologic score in assessing the activity of ulcerative colitis. Inflamm. Bowel Dis. 2013, 19, 1194–1201. [Google Scholar] [CrossRef] [PubMed]
- Pan, Y.; Yang, W.; Tang, B.; Wang, X.; Zhang, Q.; Li, W.; Li, L. The protective and pathogenic role of Th17 cell plasticity and function in the tumor microenvironment. Front Immunol. 2023, 14, 1192303. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.; Tian, J.; Wang, S. Insight into non-pathogenic Th17 cells in autoimmune diseases. Front Immunol. 2018, 9, 1112. [Google Scholar] [CrossRef]
| Males/females, n (%) | 20 (67) /10 (33) |
| Age, yrs; median (IQR) | 46 (37–57) |
| Disease duration, yrs; median (IQR) | 0 (0–2) |
| Disease extension, n (%) | |
| Proctitis | 5 (17) |
| Left-sided colitis | 7 (23) |
| Total colitis | 18 (60) |
| Disease course, n (%) | |
| First-episode | 19 (63) |
| Recurrent | |
| 11(37) | |
| C-reactive protein, mg/dL; median (IQR) | 0.04 (0.02-0.81) |
| Serum LRG, mg/mL; median (IQR) | 17 (16-19) |
| CAI, median (IQR) | 8 (6-13) |
| NRS, median (IQR) | 6 (4-9) |
| MES0, n (%) | 0 (0) |
| MES1, n (%) | 11 (37) |
| MES2, n (%) | 18 (60) |
| MES3, n (%) | 1 (3) |
| Medication before entry, n (%): | |
| None | 22 (73) |
| 5-ASA | 5 (17) |
| Sulfasalazine | 1 (3) |
| Prednisolone and 5-ASA | 1 (3) |
| Janus kinase inhibitor and 5-ASA | 1 (3) |
| Grade 3.0 | No infiltration | 6 (20%) |
| Grade 3.1 | <5% of crypts involved | 7 (23%) |
| Grade 3.2 | <50% of crypts involved | 16 (53%) |
| Grade 3.3 | >50% of crypts involved | 1 (3%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




