Submitted:
22 May 2025
Posted:
23 May 2025
You are already at the latest version
Abstract
Keywords:
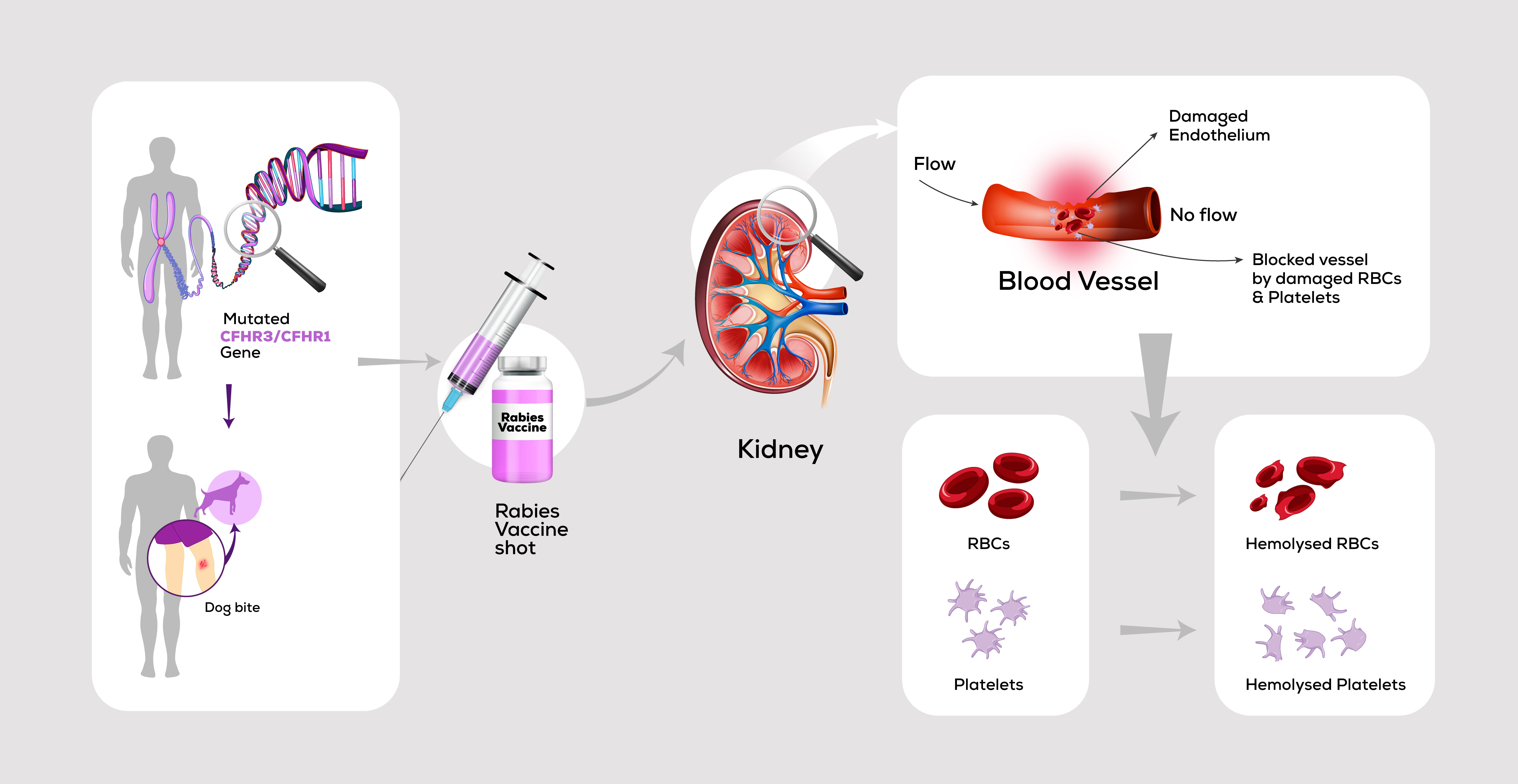
Background:
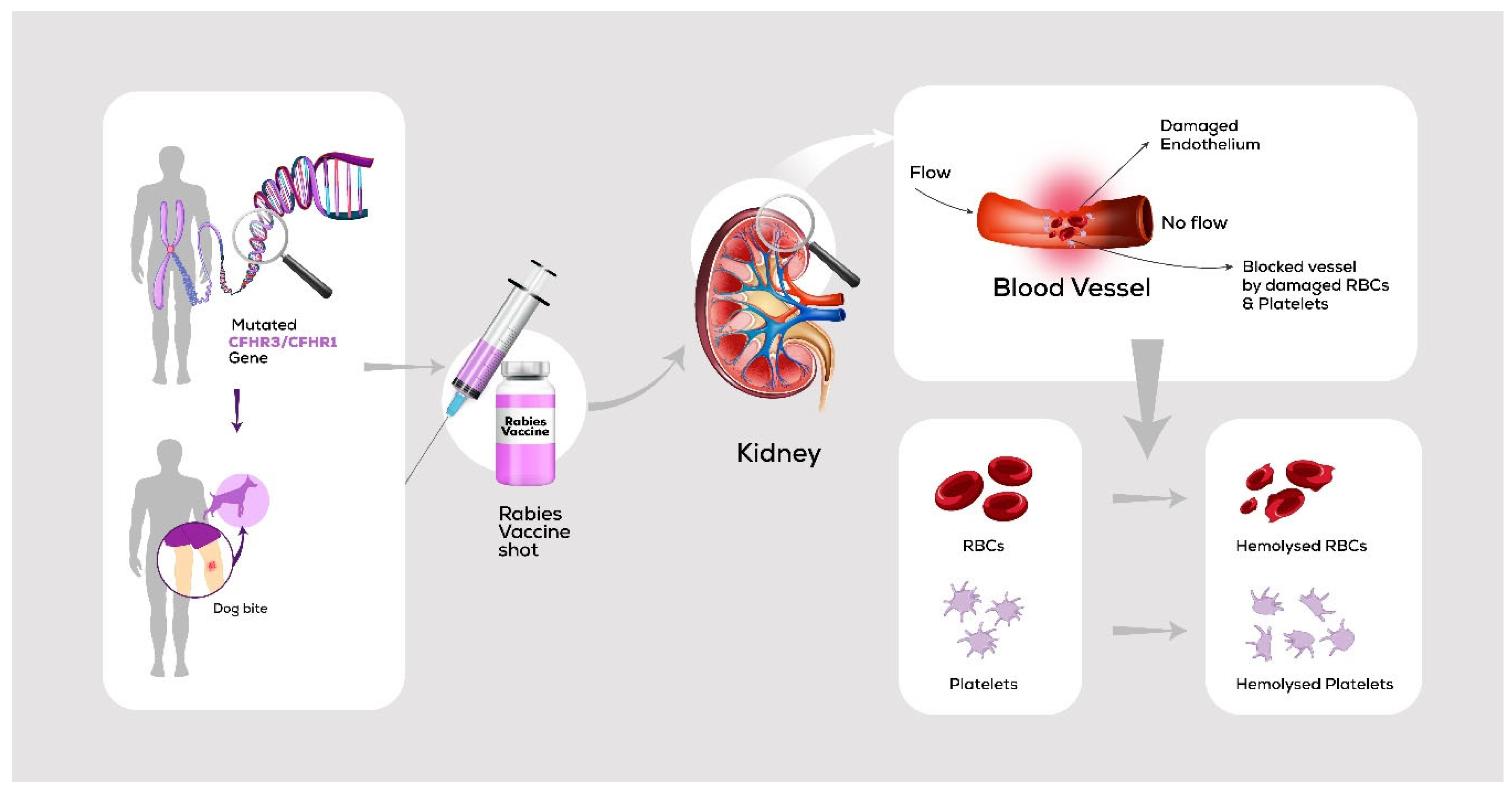
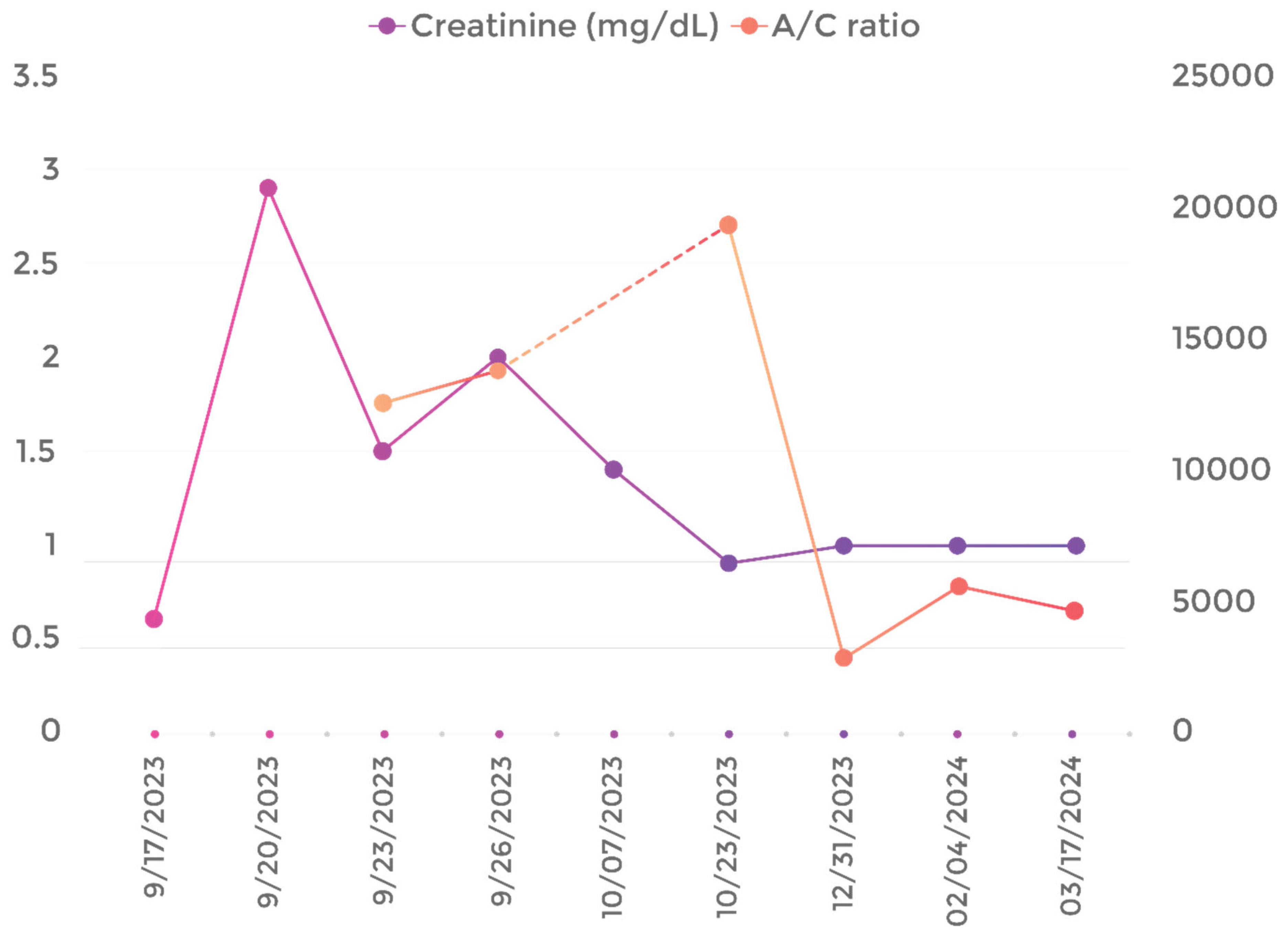
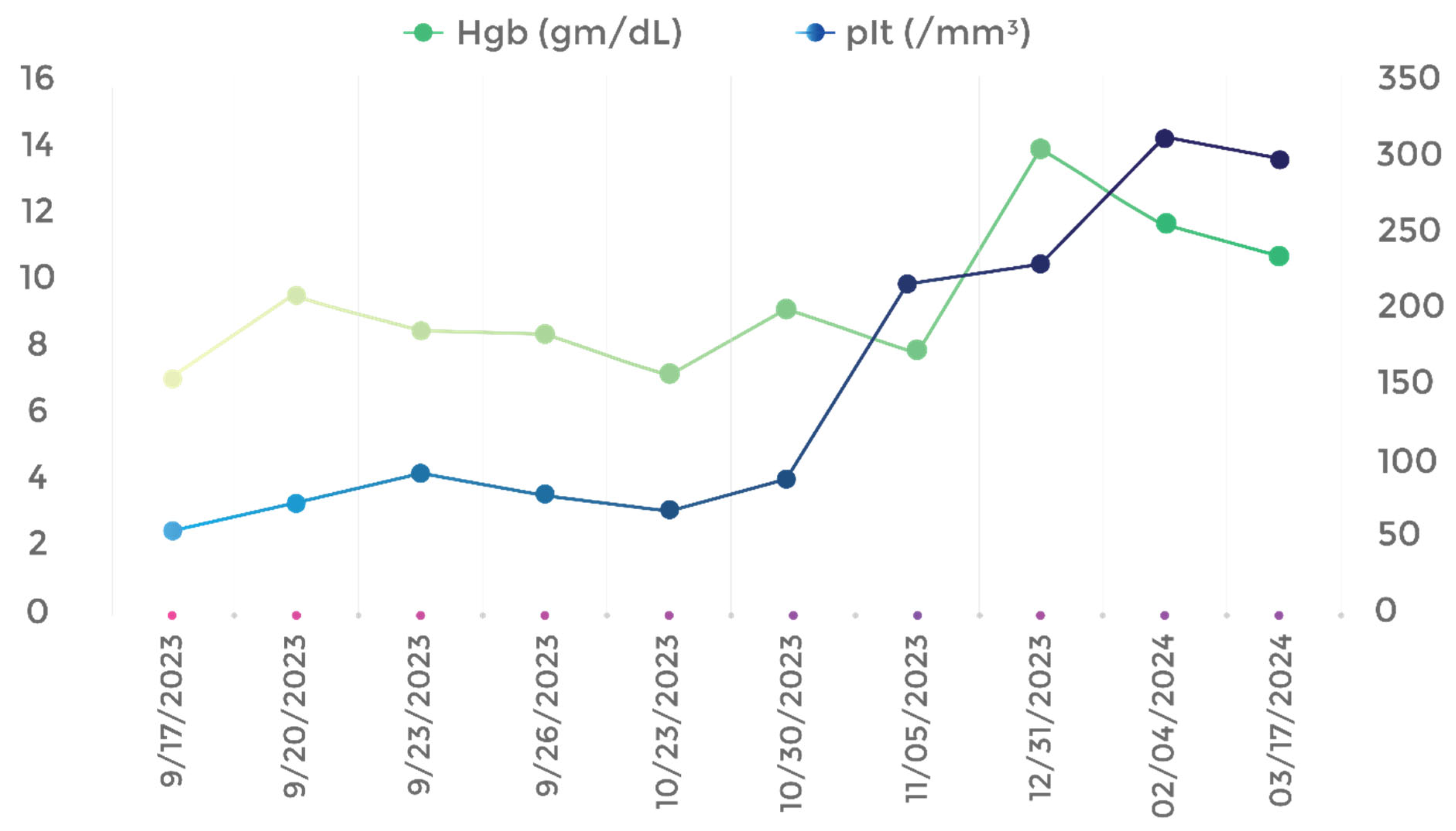
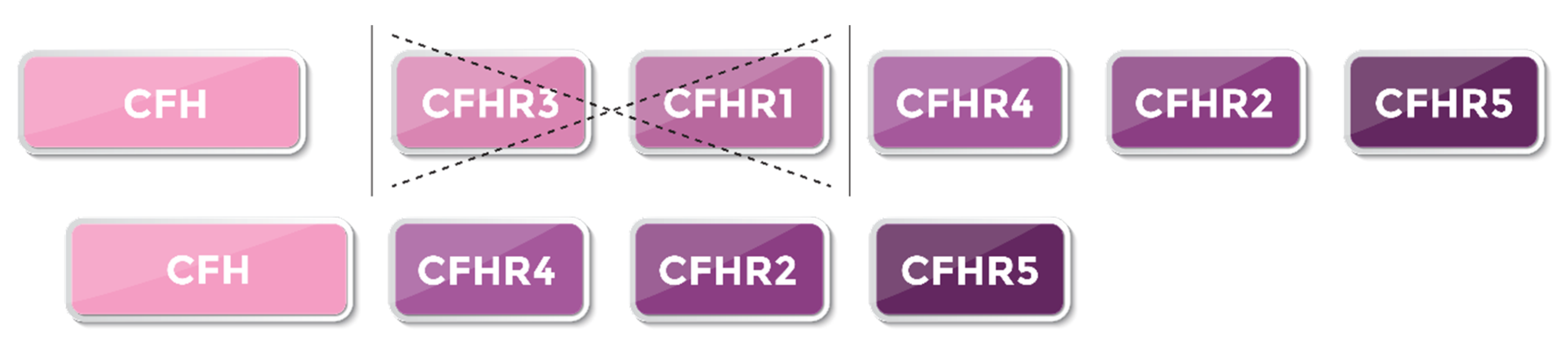
Discussion:
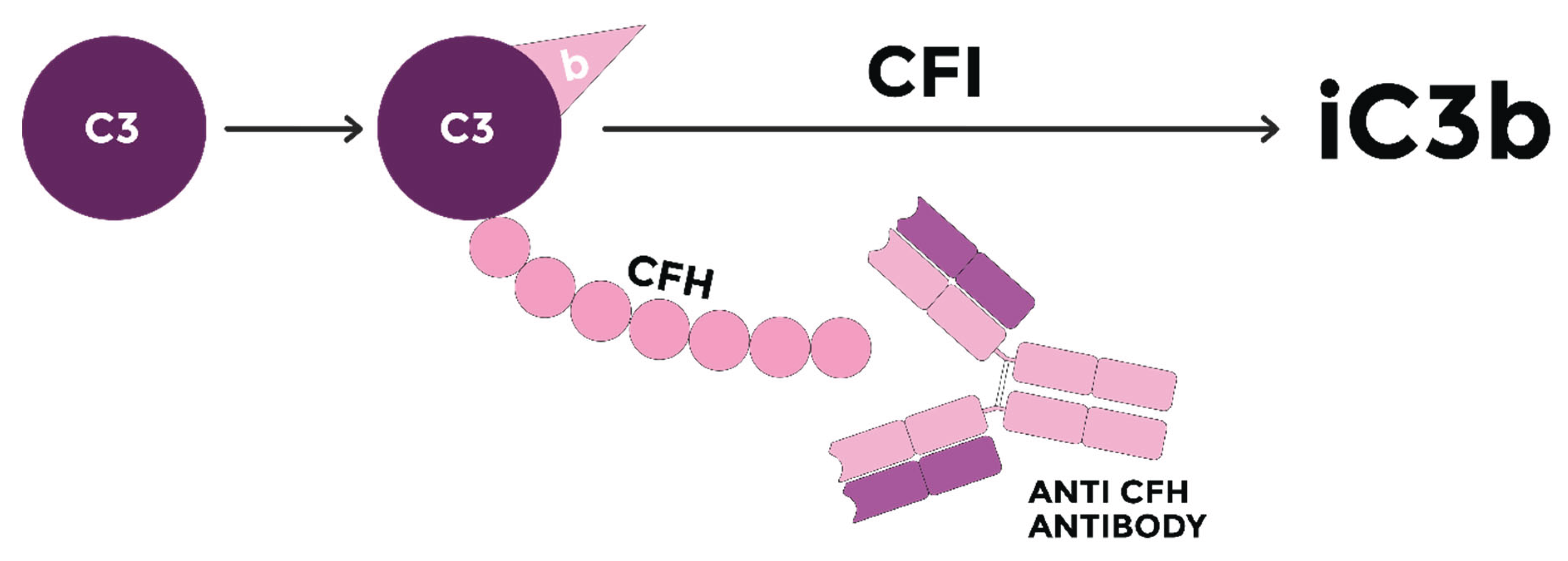
Acknowledgments
References
- Raina, R.; et al. Pediatric atypical hemolytic uremic syndrome advances. Cells, 2021; 1–21. [Google Scholar]
- Liuba, P.; et al. Residual adverse changes in arterial endothelial function and LDL oxidation after a mild systemic inflammation induced by influenza vaccination. Ann. Med. 2007, 39, 392–399. [Google Scholar] [CrossRef] [PubMed]
- Abdelmassih, A. F. , Kamel, A., Mishriky, F. & Ismail, H. Is it infection or rather vascular inflammation ? Game-changer insights and recommendations from patterns of multi-organ involvement and affected subgroups in COVID-19. 2020. [Google Scholar] [CrossRef]
- AbdelMassih, A.; et al. Is the heparin-induced thrombocytopenia-like syndrome associated with ChAdOx vaccine related to the vaccine itself or to an autoimmune reaction to severe acute respiratory syndrome 2 coronavirus: insights and implications from previous reports in infected. New Microbes New Infect. 2021, 41, 100884. [Google Scholar] [CrossRef] [PubMed]
- Chang, J. C. & Hawley, H. B. Vaccine-associated thrombocytopenia and thrombosis: Venous endotheliopathy leading to venous combined micro- macrothrombosis. Med. 2021; 57. [Google Scholar]
- Kadikoylu, G. , Yavasoglu, I. & Bolaman, Z. Rabies vaccine-associated thrombotic thrombocytopenic purpura. Transfus. Med. 2014, 24, 428–429. [Google Scholar] [PubMed]
- Cui, Y. , Wei, J. & Peng, X. Case Report: Rabies Vaccine-Induced Thrombotic Thrombocytopenic Purpura in a Patient With Systemic Lupus Erythematosus. Front. Immunol. 2022, 13, 1–5. [Google Scholar]
- Gonzalez-Mancera, M. S. & Gonzalez, J. M. In reply to: Cd4+cd8+ double- positive t-lymphocytes: Pitfalls. Turkish J. Hematol. 2020, 37, 217–218. [Google Scholar]
- Rysava, R.; et al. Atypical hemolytic uremic syndrome triggered by mRNA vaccination against SARS-CoV-2: Case report. Front. Immunol. 2022, 13, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Spasiano, A.; et al. Underlying Genetics of aHUS: Which Connection with Outcome and Treatment Discontinuation? Int. J. Mol. Sci. 2023; 24. [Google Scholar]
- KAVUKÇU, S. , İRKEN, G., OLGUN, N. & GÜÇLÜ, A. Atypical hemolytic uremic syndrome. Pediatr. Int. 1995, 37, 638–641. [Google Scholar]
- Bagga, A.; et al. Hemolytic uremic syndrome in a developing country: Consensus guidelines. Pediatr. Nephrol. 2019, 34, 1465–1482. [Google Scholar] [CrossRef] [PubMed]
- Ayres, J. A. , Barraviera, B., Calvi, S. A., Carvalho, N. R. & Peraçoli, M. T. S. Antibody and cytokine serum levels in patients subjected to anti-rabies prophylaxis with serum-vaccination. J. Venom. Anim. Toxins Incl. Trop. Dis. 2006, 12, 435–455. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



