Submitted:
20 May 2025
Posted:
21 May 2025
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
1.1. Global and European Surgical Error Rates
1.2. Detailed Analysis and Supporting Information
1.3. European Context and Specific Studies
| Region | Metric | Value |
|---|---|---|
| Global (WHO) | Annual surgical procedures | Over 300 million |
| Global (WHO) | Patients with disabling surgical adverse events | At least 7 million annually |
| Global (WHO) | Deaths from surgical adverse events | Over 1 million annually |
| Global (WHO) | Complication rate in industrialized countries | Up to 25% of inpatient ops |
| Europe (BMJ 2024) | Patients with adverse events | 38% (383/1,009 patients) |
| Europe (BMJ 2024) | Definitely preventable events | ~10% (103/1,009 patients) |
| Europe (EU) | Annual HAIs linked to surgery | 3.2 million patients |
| Europe (EU) | Deaths from HAIs | 37,000 annually |
1.4. Patient Safety and the Role of Machine Learning in Reducing Surgical Errors
2. Material and Methods
3. Results and Discussions
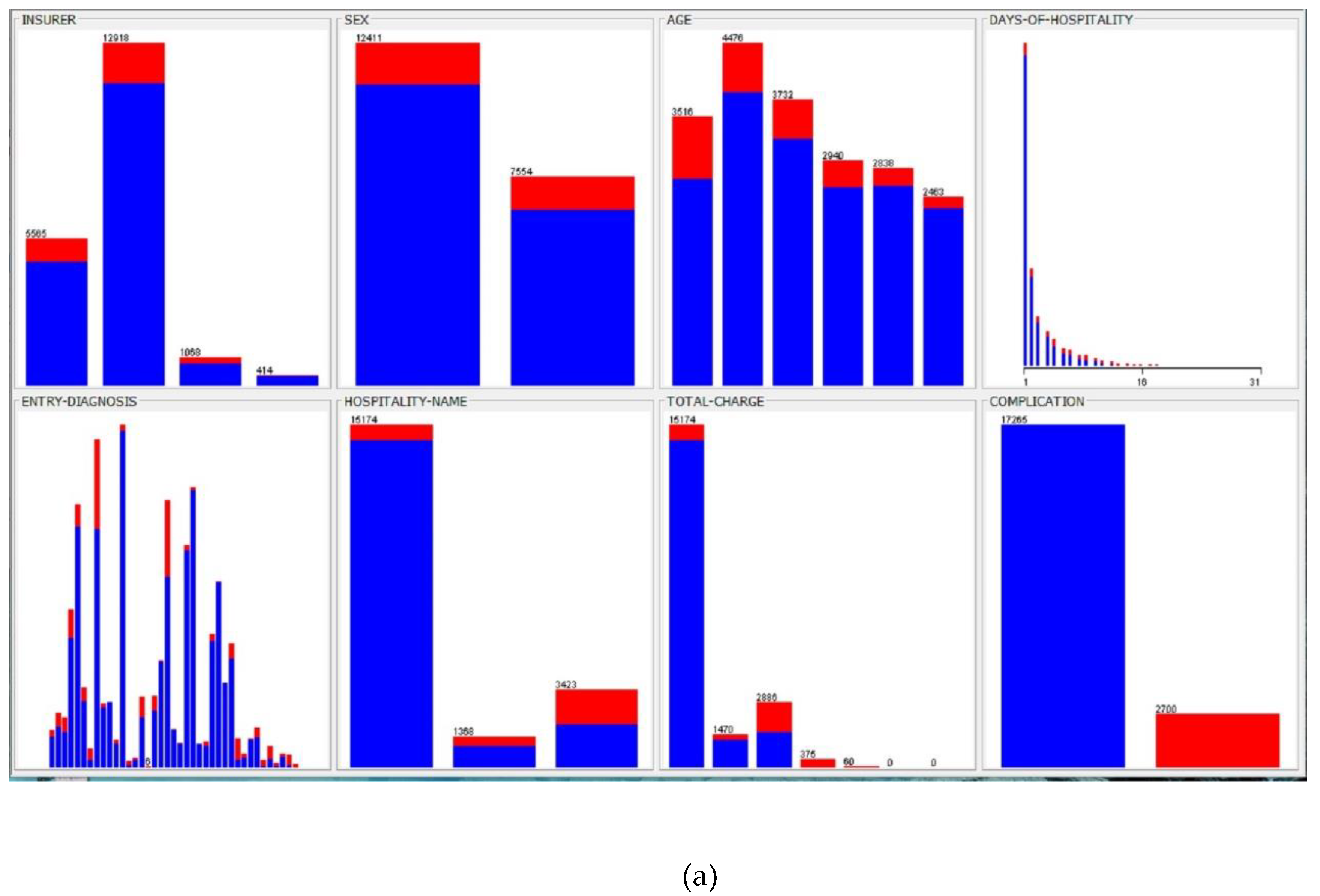

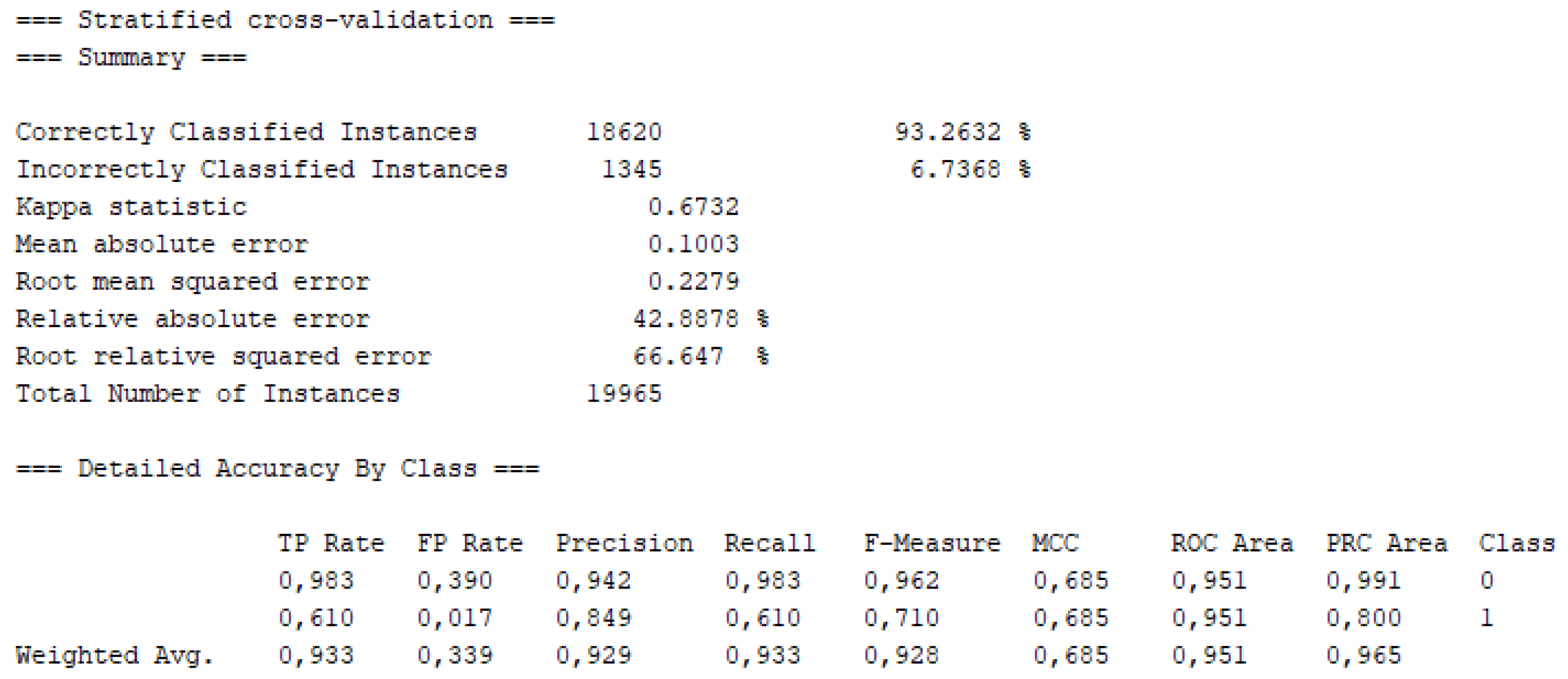
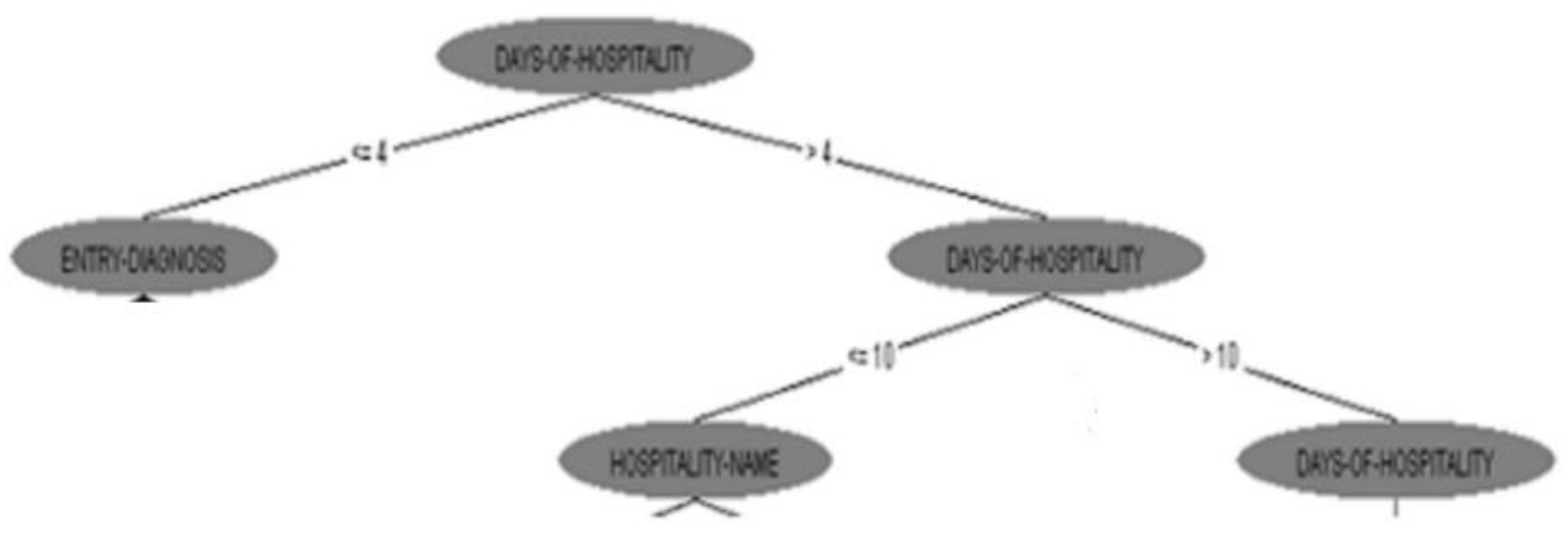
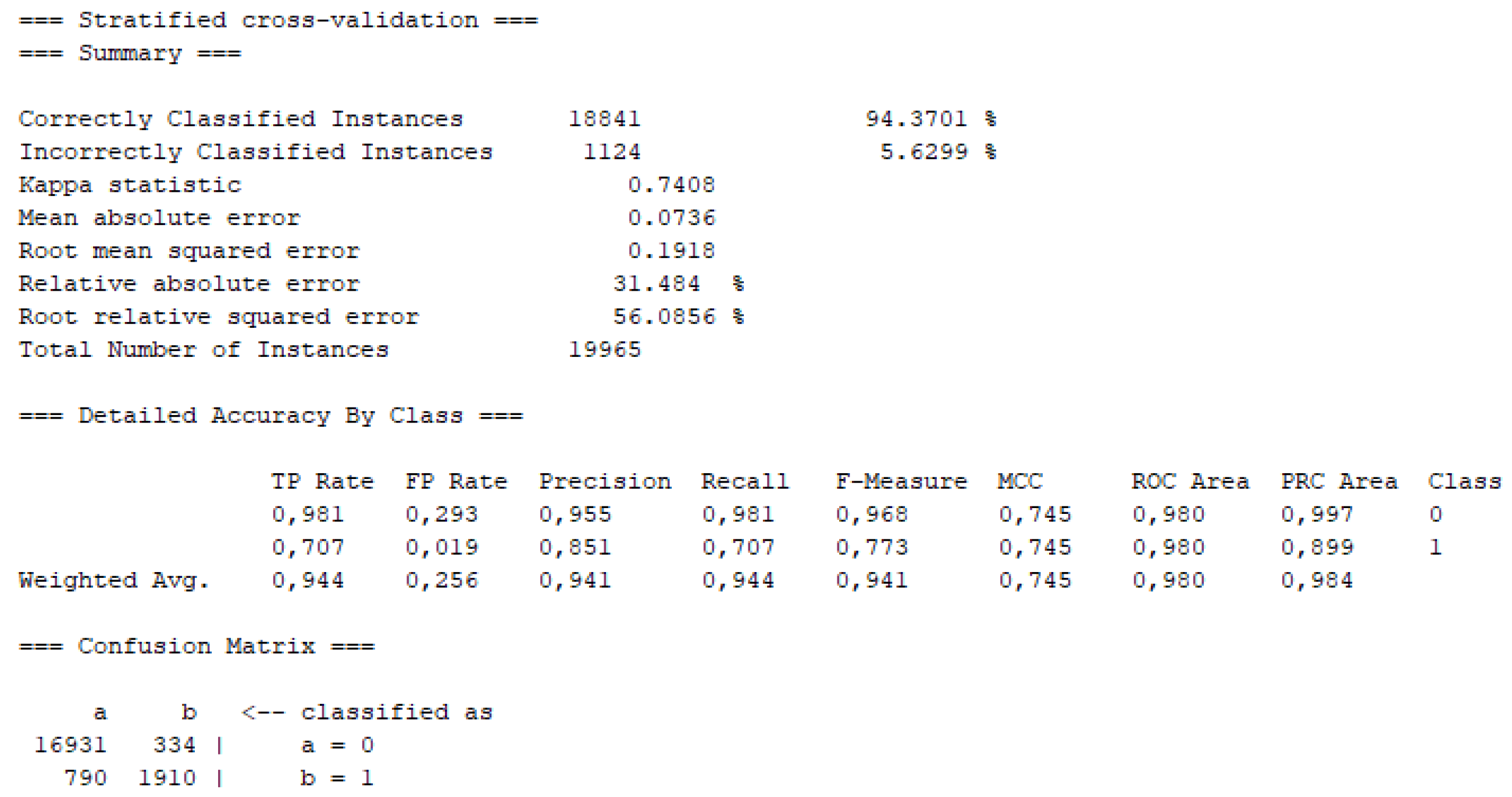
4. Conclusions
Ethical Approval
Funding Information
Author Contributions
Data Availability Statement
Acknowledgement
Conflict of Interest
References
- World Health Organization. (2019, September 13). WHO calls for urgent action to reduce patient harm in healthcare. https://www.who.
- A Makary, M.; Daniel, M. Medical error—the third leading cause of death in the US. BMJ 2016, 353, i2139. [Google Scholar] [CrossRef] [PubMed]
- Barach, P.; Small, S.D. Reporting and preventing medical mishaps: lessons from non-medical near miss reporting systems. BMJ 2000, 320, 759–763. [Google Scholar] [CrossRef] [PubMed]
- Topol, E.J. High-performance medicine: the convergence of human and artificial intelligence. Nat. Med. 2019, 25, 44–56. [Google Scholar] [CrossRef]
- Sarker, S.K.; Vincent, C. Errors in surgery. Int. J. Surg. 2005, 3, 75–81. [Google Scholar] [CrossRef]
- Havens, D.H.; Boroughs, L. (2000). “To err is human”: A report from the Institute of Medicine. Journal of Pediatric Health Care, 14(2), 77–80. [CrossRef]
- Flin, R. , & O’Connor, P. (2017). Safety at the sharp end: A guide to non-technical skills. CRC Press.
- Marsh, K. M. , Turrentine, F. E., Knight, K., Attridge, E., Chen, X., Vittitow, S., & Jones, R. S. (2022). Defining and studying errors in surgical care: A systematic review. Annals of Surgery, 275(6), 1067–1073. [CrossRef]
- Smith, J. P. , Brown, A. L., & Taylor, C. R. (2018). Advances in patient safety: Addressing medical errors in modern healthcare. Patient Safety Journal, 10(3), 25–33.
- Patel, S. (2020). Advances in patient safety: Addressing medical errors in modern healthcare. Patient Safety Journal, 10(3), 25–33.
- Duclos, A.; Frits, M.L.; Iannaccone, C.; Lipsitz, S.R.; Cooper, Z.; Weissman, J.S.; Bates, D.W. Safety of inpatient care in surgical settings: cohort study. BMJ 2024, 387, e080480. [Google Scholar] [CrossRef]
- Cohen, A.J.; Lui, H.; Zheng, M.; Cheema, B.; Patino, G.; Kohn, M.A.; Enriquez, A.; Breyer, B.N. Rates of Serious Surgical Errors in California and Plans to Prevent Recurrence. JAMA Netw. Open 2021, 4, e217058–e217058. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.; Magalhães, C.; Rocha, M.; Osório, N.S. Machine Learning-Enhanced T Cell Neoepitope Discovery for Immunotherapy Design. Cancer Informatics 2019, 18. [Google Scholar] [CrossRef]
- Stahel, P.F.; Holland, K.; Nanz, R. Machine learning approaches for improvement of patient safety in surgery. Patient Saf. Surg. 2024, 18, 37. [Google Scholar] [CrossRef]
- Elfanagely, O. , Toyoda, Y., Othman, S., Mellia, J. A., Basta, M., Liu, T., & Fischer, J. P. (2021). Machine learning and surgical outcomes prediction: A systematic review. Journal of Surgical Research, 264, 346–361. [CrossRef]
- Locke, S. , Bashall, A., Al-Adely, S., Moore, J., Wilson, A., & Kitchen, G. B. (2021). Natural language processing in medicine: A review. Trends in Anaesthesia and Critical Care, 38, 4–9. [CrossRef]
- Michou, I.; Maroulis, I.; Koutsojannis, C. Machine learning for medical error prevention in departments of surgery: A review of challenges and biases. World J. Biol. Pharm. Heal. Sci. 2025, 22, 383–389. [Google Scholar] [CrossRef]
- Norori, N.; Hu, Q.; Aellen, F.M.; Faraci, F.D.; Tzovara, A. Addressing bias in big data and AI for health care: A call for open science. Patterns 2021, 2, 100347. [Google Scholar] [CrossRef]
- Baker, A. Crossing the Quality Chasm: A New Health System for the 21st Century. BMJ 2001, 323, 1192. [Google Scholar] [CrossRef]
- Shanafelt, T.D.; Balch, C.M.; Bechamps, G.; Russell, T.; Dyrbye, L.; Satele, D.; Collicott, P.; Novotny, P.J.; Sloan, J.; Freischlag, J. Burnout and Medical Errors Among American Surgeons. Ann. Surg. 2010, 251, 995–1000. [Google Scholar] [CrossRef]
- Al-Ghunaim, T.A.; Johnson, J.; Biyani, C.S.; Alshahrani, K.M.; Dunning, A.; O'Connor, D.B. Surgeon burnout, impact on patient safety and professionalism: A systematic review and meta-analysis. Am. J. Surg. 2022, 224, 228–238. [Google Scholar] [CrossRef] [PubMed]
- Carter, S.D. The surgeon as a risk factor. . 2002, 36, 141–65. [Google Scholar] [CrossRef] [PubMed]
- Challen, R.; Denny, J.; Pitt, M.; Gompels, L.; Edwards, T.; Tsaneva-Atanasova, K. Artificial intelligence, bias and clinical safety. BMJ Qual. Saf. 2019, 28, 231–237. [Google Scholar] [CrossRef]
- Balch, J. A. , & Loftus, T. J. (2023). Actionable artificial intelligence: Overcoming barriers to adoption of prediction tools. Surgery, 174(3), 730–732. [CrossRef]
- Al Mamlook, R. E. , Wells, L. J., & Sawyer, R. (2023). Machine-learning models for predicting surgical site infections using patient pre-operative risk and surgical procedure factors. American Journal of Infection Control, 51(5), 544–550. [CrossRef]
- Zhou, Z.-H. Ensemble Methods: Foundations and algorithms; CRC Press. [CrossRef]
- Zhou, Z. H. (2019). Machine learning. Springer.
- Holmes, G.; Donkin, A.; Witten, I. WEKA: A machine learning workbench. Proceedings of ANZIIS ‘94—Australian New Zealnd Intelligent Information Systems Conference2002, Brisbane, Australia, 29 November–2 December 1994; pp. 357–361. [Google Scholar] [CrossRef]
- Yan, H.; Jiang, Y.; Zheng, J.; Peng, C.; Li, Q. A multilayer perceptron-based medical decision support system for heart disease diagnosis. Expert Syst. Appl. 2006, 30, 272–281. [Google Scholar] [CrossRef]
- Hall, M. A. , L. A. (1998). Practical feature subset selection for machine learning. In Computer Science ’98, Proceedings of the 21st Australasian Computer Science Conference ACSC’98 (pp.
- Bilimoria, K.Y.; Liu, Y.; Paruch, J.L.; Zhou, L.; Kmiecik, T.E.; Ko, C.Y.; Cohen, M.E. Development and Evaluation of the Universal ACS NSQIP Surgical Risk Calculator: A Decision Aid and Informed Consent Tool for Patients and Surgeons. J. Am. Coll. Surg. 2013, 217, 833–842.e3. [Google Scholar] [CrossRef] [PubMed]
- Bertsimas, D. , Dunn, J. M. ( 153(11), 1050–1059. [CrossRef]
- Rajkomar, A.; Dean, J.; Kohane, I. Machine Learning in Medicine. New Engl. J. Med. 2019, 380, 1347–1358. [Google Scholar] [CrossRef]
- Shilo, S. , Cohen, R. ( 3(2), 45–53. [CrossRef]
- De Vries, E.N.; Ramrattan, M.A.; Smorenburg, S.M.; Gouma, D.J.; Boermeester, M.A. The incidence and nature of in-hospital adverse events: a systematic review. Quality & Safety in Health Care 2020, 19, 216–223. [Google Scholar]
- Lundberg, S. M. , & Lee, S.-I. (2017). A unified approach to interpreting model predictions. In Advances in Neural Information Processing Systems (Vol. 30, pp. 4765–4774).
- Hashimoto, D.A.; Rosman, G.; Rus, D.; Meireles, O.R.M. Artificial Intelligence in Surgery: Promises and Perils. Ann. Surg. 2018, 268, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Cross, J.L.; Choma, M.A.; Onofrey, J.A. Bias in medical AI: Implications for clinical decision-making. PLOS Digit. Health 2024, 3, e0000651. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
| Risk Accuracy (%)/ML algorithms | All Together | Hospitalization | Clinical Assessment |
|---|---|---|---|
| Ensemble Algorithm (Vote) | 93.6 | 85.0 | 87.7 |
| Random Forest | 94.2 | 89.9 | 90.5 |
| Decision Tree (J48) | 93.1 | 89.0 | 90.6 |
| Neural Network (Multilayer Perceptron) | 93.8 | 90.0 | 91.9 |
| Bayes (Naive Bayes) | 87.7 | 85.2 | 86.4 |
| Model | Accuracy (%) | Sensitivity (%) | AUC-ROC | Source |
|---|---|---|---|---|
| Decision Tree (J48) | 93.3 | 92.0 | 0.95 | This study |
| Random Forest | 94.3 | 94.4 | 0.98 | This study |
| Neural Network | 93.8 | 91.8 | 0.94 | This study |
| ACS NSQIP Calculator | 90.0 | 82.0 | 0.88 | Bilimoria et al. |
| Bertsimas et al. (2018) | 92.0 | 89.0 | 0.93 | Bertsimas et al. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




