Submitted:
12 May 2025
Posted:
15 May 2025
You are already at the latest version
Abstract
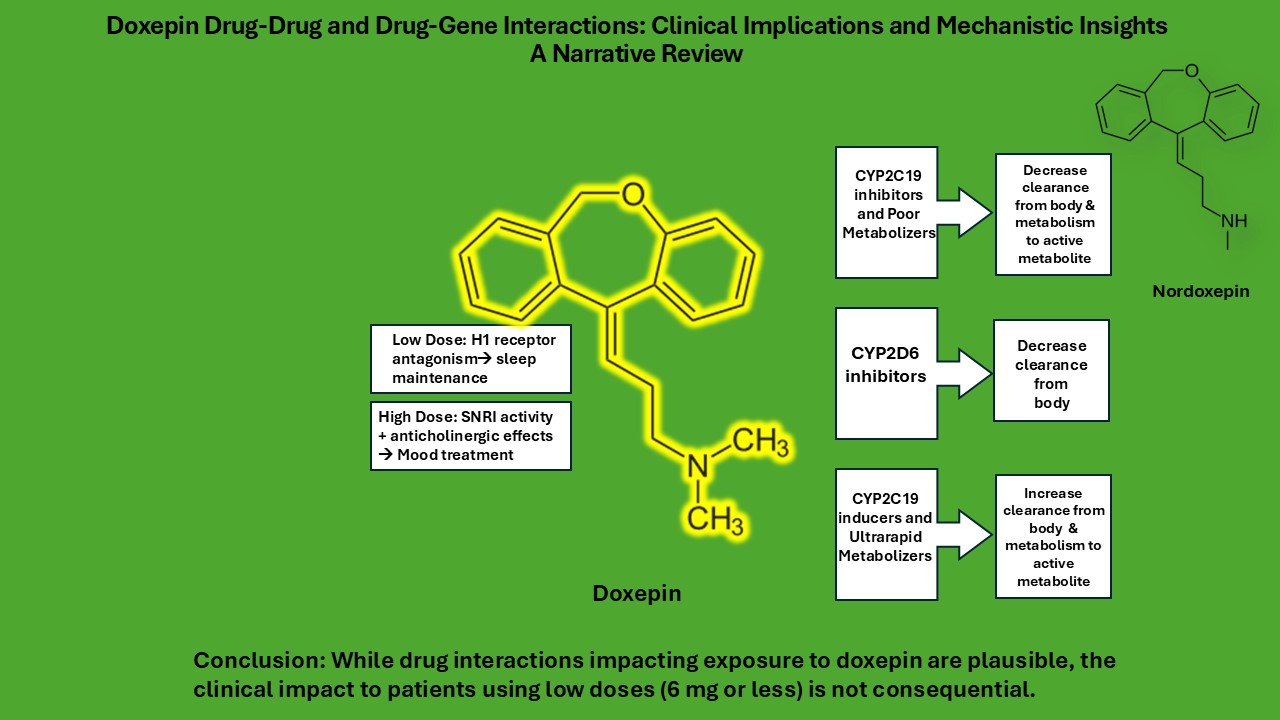
Keywords:
1. Introduction
2. Materials and Methods
3. Results
3.1. Doxepin CYP450 Enzyme Inhibition
3.2. CYP2D6/2C19 Inhibitors and Inducers
3.2.1. Fluoxetine
3.2.2. Sertraline
3.2.3. Cimetidine
3.2.4. Fluvoxamine
3.2.5. Rifampin
3.3. Other Drug Interactions
3.3.1. Tolazamide
3.3. Pharmacogenomics
4. Discussion
5. Conclusions
References
- Almasi, A.; Patel, P.; Meza, C. E. Doxepin. StatPearls - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK542306/.
- Kirchheiner, J.; Meineke, I.; Muller, G.; Roots, I.; Brockmoller, J. Contributions of CYP2D6, CYP2C9 and CYP2C19 to the biotransformation of E- and Z-doxepin in healthy volunteers. Pharmacogenetics 2002,12(7),571–580. [CrossRef]
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA165981686/overview.
- Kaneko, H.; Korenaga, R.; Nakamura, R.; Kawai, S.; Ando, T.; Shiroishi, M. Binding characteristics of the doxepin E/Z-isomers to the histamine H1 receptor revealed by receptor-bound ligand analysis and molecular dynamics study. Journal of Molecular Recognition 2024, 37 (5). [CrossRef]
- Foye, W. O.; Lemke, T. L.; Williams, D. A. Foye’s Principles of Medicinal Chemistry; 2012.
- Cheung, J. M. Y.; Scott, H.; Muench, A.; Grunstein, R. R.; Krystal, A. D.; Riemann, D.; Perlis, M. Comparative short-term safety and efficacy of hypnotics: A quantitative risk–benefit analysis. Journal of Sleep Research 2023, 33 (4). [CrossRef]
- Cheung, J. M. Y.; Scott, H.; Muench, A.; Grunstein, R. R.; Krystal, A. D.; Riemann, D.; Perlis, M. Comparative short-term safety and efficacy of hypnotics: A quantitative risk–benefit analysis. Journal of Sleep Research 2023, 33 (4). [CrossRef]
- Sateia, M. J.; Buysse, D. J.; Krystal, A. D.; Neubauer, D. N.; Heald, J. L. Clinical Practice Guideline for the Pharmacologic Treatment of Chronic Insomnia in Adults: An American Academy of Sleep Medicine Clinical Practice Guideline. Journal of Clinical Sleep Medicine 2017, 13 (02), 307–349. [CrossRef]
- Gillman, P. K. Tricyclic antidepressant pharmacology and therapeutic drug interactions updated. British Journal of Pharmacology 2007, 151 (6), 737–748. [CrossRef]
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA162359940/overview.
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA166163647.
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA165960076/pathway.
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA161749012.
- Altamura, A. C.; Moro, A. R.; Percudani, M. Clinical pharmacokinetics of fluoxetine. Clinical Pharmacokinetics 1994, 26 (3), 201–214. [CrossRef]
- Bergstrom, R. F.; Peyton, A. L.; Lemberger, L. Quantification and mechanism of the fluoxetine and tricyclic antidepressant interaction. Clinical Pharmacology & Therapeutics 1992, 51 (3), 239–248. [CrossRef]
- Von Ammon Cavanaugh, S. Drug-Drug Interactions of Fluoxetine with Tricyclics. Psychosomatics 1990, 31 (3), 273–276. [CrossRef]
- Suckow, R. F.; Roose, S. P.; Cooper, T. B. Effect of fluoxetine on plasma desipramine and 2-hydroxydesipramine. Biological Psychiatry 1992, 31 (2), 200–204. [CrossRef]
- Hashim, I. A. Therapeutic drugs and toxicology testing. In Elsevier eBooks; 2023; pp 375–418. [CrossRef]
- Browne, M.; Horn, E.; Jones, T. T. The benefits of Clomipramine-Fluoxetine combination in obsessive Compulsive disorder. The Canadian Journal of Psychiatry 1993, 38 (4), 242–243. [CrossRef]
- PharmGKB. PharmGKB. https://www.pharmgkb.org/pathway/PA166181117.
- CENTER FOR DRUG EVALUATION AND RESEARCH. Biopharmaceutics review; report; Somaxon Pharmaceuticals, Inc., 2009. https://www.accessdata.fda.gov/drugsatfda_docs/nda/2010/022036Orig1s000ClinPharmR.pdf.
- Somogyi, A.; Gugler, R. Clinical Pharmacokinetics of Cimetidine. Clinical Pharmacokinetics 1983, 8 (6), 463–495. [CrossRef]
- Sutherland, D. L.; Remillard, A. J.; Haight, K. R.; Brown, M. A.; Old, L. The influence of cimetidine versus ranitidine on doxepin pharmacokinetics. European Journal of Clinical Pharmacology 1987, 32 (2), 159–164. [CrossRef]
- Selective serotonin re-uptake inhibitors (SSRIs). In Elsevier eBooks; 2015; pp 317–337. [CrossRef]
- Szegedi, A.; Wetzel, H.; Leal, M.; Härtter, S.; Hiemke, C. Combination treatment with clomipramine and fluvoxamine: drug monitoring, safety, and tolerability data. PubMed 1996, 57 (6), 257–264.
- Vezmar, S.; Miljkovic, B.; Vucicevic, K.; Timotijevic, I.; Prostran, M.; Todorovic, Z.; Pokrajac, M. Pharmacokinetics and efficacy of fluvoxamine and amitriptyline in depression. Journal of Pharmacological Sciences 2009, 110 (1), 98–104. [CrossRef]
- Niemi, M.; Backman, J. T.; Fromm, M. F.; Neuvonen, P. J.; Kivist, K. T. Pharmacokinetic Interactions with Rifampicin. Clinical Pharmacokinetics 2003, 42 (9), 819–850. [CrossRef]
- Bebchuk, J. M.; Stewart, D. E. Drug Interaction between Rifampin and Nortriptyline: A Case Report. The International Journal of Psychiatry in Medicine 1991, 21 (2), 183–187. [CrossRef]
- Hashmi, H. Z.; Kaur, J.; Stout, S. C.; Drake, T. Doxepin-Associated hypoglycemia in an ambulatory nondiabetic patient. The Primary Care Companion for CNS Disorders 2023, 25 (4). [CrossRef]
- True, B. L.; Perry, P. J.; Burns, E. A. Profound hypoglycemia with the addition of a tricyclic antidepressant to maintenance sulfonylurea therapy [published erratum appears in Am J Psychiatry 1987 Nov;144(11):1521]. American Journal of Psychiatry 1987, 144 (9), 1220–1221. [CrossRef]
- Gupta, B.; Shakarwal, M. K.; Kumar, A.; Jaju, B. P. Modulation of glucose homeostasis by doxepin. PubMed 1992, 14 (1), 61–71.
- Klein, M. D.; Williams, A. K.; Lee, C. R.; Stouffer, G. A. Clinical utility of CYP2C19 genotyping to guide antiplatelet therapy in patients with an acute coronary syndrome or undergoing percutaneous coronary intervention. Arteriosclerosis Thrombosis and Vascular Biology 2019, 39 (4), 647–652. [CrossRef]
- Hicks, J.; Sangkuhl, K.; Swen, J.; Ellingrod, V.; Müller, D.; Shimoda, K.; Bishop, J.; Kharasch, E.; Skaar, T.; Gaedigk, A.; Dunnenberger, H.; Klein, T.; Caudle, K.; Stingl, J. Clinical pharmacogenetics implementation consortium guideline (CPIC) for CYP2D6 and CYP2C19 genotypes and dosing of tricyclic antidepressants: 2016 update. Clinical Pharmacology & Therapeutics 2016, 102 (1), 37–44. [CrossRef]
- Krystal, A. D.; Richelson, E.; Roth, T. Review of the histamine system and the clinical effects of H1 antagonists: Basis for a new model for understanding the effects of insomnia medications. Sleep Medicine Reviews 2013, 17 (4), 263–272. [CrossRef]
- Bertilsson, L.; Mellström, B.; Sjöqvist, F. Pronounced inhibition of noradrenaline uptake by 10-hydroxymetabolites of nortriptyline. Life Sciences 1979, 25 (15), 1285–1292. [CrossRef]
- Balant-Gorgia, A. E.; Gex-Fabry, M.; Balant, L. P. Clinical pharmacokinetics of clomipramine. Clinical Pharmacokinetics 1991, 20 (6), 447–462. [CrossRef]
- Gunning, R.; Chu, C.; Nakhla, N.; Kim, K. C.; Suda, K. J.; Tadrous, M. Major shifts in acid suppression drug utilization after the 2019 ranitidine recalls in Canada and United States. Digestive Diseases and Sciences 2023, 68 (8), 3259–3267. [CrossRef]
- National data. Reported Tuberculosis in the United States, 2023. https://www.cdc.gov/tb-surveillance-report-2023/summary/national.html#:~:text=In%202023%2C%20the%20United%20States,to%20the%20COVID%2D19%20pandemic.
- National Institute of Child Health and Human Development. Tolazamide. Drugs and Lactation Database (LactMed®) - NCBI Bookshelf. https://www.ncbi.nlm.nih.gov/books/NBK501118/.
| Table 1 | ||||
| Drug | Primary CYP Enzyme | Metabolitea | Comparison of metabolite activity to Parent Drug b | Comparison of parent drug to doxepin c |
| Doxepin | 2D6 | E-Hydroxydoxepin, E-hydroxydesmethyldoxepin | Low | |
| 2C19 | E/Z-Desmethyldoxepin (nordoxepin) * | stronger inhibitor of norepinephrine reuptake and weaker inhibitor of serotonin reuptake | ||
| Clomipramine | 2D6 | Hydroxylated metabolites | Low | less potent at H1 and NE receptors , slightly less potent at α1 ,more potent at Musc, 5-HT receptors |
| 2C19 | Desmethylclomipramine* | stronger inhibitor of norepinephrine reuptake and weaker inhibitor of serotonin reuptake | ||
| Amitriptyline | 2D6 | Hydroxyamitriptyline | Low | Slightly less potent at H1, α1, less potent at NE more potent at 5-HT and Musc receptors. |
| 2C19 | Nortriptyline * | Stronger inhibitor of norepinephrine reuptake and weaker inhibitor of serotonin reuptake | ||
| Nortriptyline | 2D6 | 10-hydroxynortryptiline* | Low | less potent at 5-HT, α1, H1 receptors , more potent at Musc, NE and α1 receptors |
| 2C19 | Desmethylnortryptiline | Low | ||
| Imipramine | 2D6 | Desipramine* | Stronger inhibitor of norepinephrine and weaker inhibitor of serotonin reuptake | less potent at α1, NE, H1 receptors , more potent at 5-HT, Musc |
| 2C19 | Hydroxylated metabolites | Low | ||
| Desipramine | 2D6 | 2-hydroxydesipramine * | Low | Less potent at H1, α1, muscarinic receptors. More potent 5-HT, NE |
| 2C19 | E-Hydroxydoxepin, E-hydroxydesmethyldoxepin | Low | ||
| * Primary active metabolite | ||||
|
a Data adapted from:3,7,8,9 b Data adapted from: 5,6,31,32 c Data adapted from 6 d Abbreviations: 5-HT, serotonin receptor; NE, norepinephrine; H1, Histamine type 1, Musc; acetylcholine muscarinic; α1, α1 adrenoreceptor d | ||||
| Study Name | Interacting Drugs | Results | Author Conclusions | Relevance to Low Dose Doxepin |
|---|---|---|---|---|
| Bergstrom et al. (1992) |
Desipramine 50 mg and Fluoxetine 60mg | There was a 1.5-fold increase in Cmax and no increases in side effects. | Fluoxetine causes an inhibition of tricyclic 2-hydroxylation and may decrease first-pass and systemic metabolism. | Fluoxetine likely decreases doxepin metabolism but at low doses this interaction would not yield safety concerns |
| Von Ammon Cavanaugh (1990) |
Desipramine and Fluoxetine Amitriptyline and Fluoxetine |
There was an increase in plasma levels of desipramine and amitriptyline. The effect was more profound with desipramine than amitriptyline. | ||
| Suckow et al. (1992) |
Desipramine and Fluoxetine | The patient’s depressive symptoms improved, and they were discharged on the combination therapy. |
||
| Browne et al. (1993) |
Clomipramine and Fluoxetine | The combination therapy showed increased benefits without increased side effects. | ||
| 022036Orig1s000ClinPharmR.pdf | Doxepin 6mg and Sertraline 50mg | Doxepin Cmax increased by 32% when given with sertraline. | Not clinically meaningful and requires no dose adjustments | |
| 022036Orig1s000ClinPharmR.pdf | Doxepin 6mg and Cimetidine 300mg bid | There was a 2-fold increase in doxepin exposure | Dose of doxepin should be limited to 3mg when co administered with cimetidine | Although a 2-fold increase in exposure may be clinically significant, doxepin’s therapeutic profile suggest that a low dose with 2x exposure would not yield significant safety concerns. |
| Sutherland et al. (1987) |
Doxepin 50 and Cimetidine 600 mg bid | Plasma concentration of doxepin increased from 4.7 ng/mL to 9 ng/mL | Cimetidine inhibits the biotransformation of doxepin. | |
| Szegedi et al. (1996) |
Clomipramine and Fluvoxamine | Serum clomipramine levels were elevated, and the combination therapy was well tolerated. |
The pharmacokinetic interactions between fluvoxamine and clomipramine may be well tolerated in a majority of patients. | While fluvoxamine may reduce the metabolism of doxepin the impact on treatment and safety would not be harmful. |
| Vezmar et al. (2009) |
Amitriptyline 75mg and Fluvoxamine 100mg | The Cmax of amitriptyline remained unchanged while the Cmax of its metabolite increased from 7 to 17.6. There were no significant differences in adverse effects. Patients with major depression had stronger onset of clinical response. |
pharmacokinetic interaction between fluvoxamine and amitriptyline resulting in impaired metabolism of the latter. However, no significant impact of the interaction on treatment safety was observed. | |
| Bebchuk and Stewart (1991) |
Nortriptyline and Rifampin | Nortriptyline titrated from 10 mg to 175 mg for therapeutic effect Drowsiness at serum level of 671 |
Higher than expected doses of nortriptyline were required to obtain a therapeutic drug level while the patient was receiving rifampin. | Patients on low dose doxepin may not receive therapeutic benefits from doxepin during rifampin treatment |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




