Submitted:
09 May 2025
Posted:
12 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals, reagents and culture media
2.2. Morphometric characterization of TiO2 nanoparticles
2.2.1. Transmission Electron Microcopy (TEM)
2.2.2. Dynamic Light Scattering
2.3. Hemocompatibility studies
2.3.1. Blood collection and preparation of erythrocyte suspension
2.3.2. Hemolytic Activity.
2.3.3. Coagulation Assays
2.4. Cell culture and cytotoxicity studies
2.5. Phototoxic behavior
2.6. Genotoxicity
2.7. Statistical Analysis
3. Results
3.1. Transmission elecctronic microscopy (TEM)
3.2. Studies of dynamic light scattering (DLS)
3.2.1. Hydrodynamic diameter of TiO2 and proteins influence
3.2. Hemocompatibility of TiO2
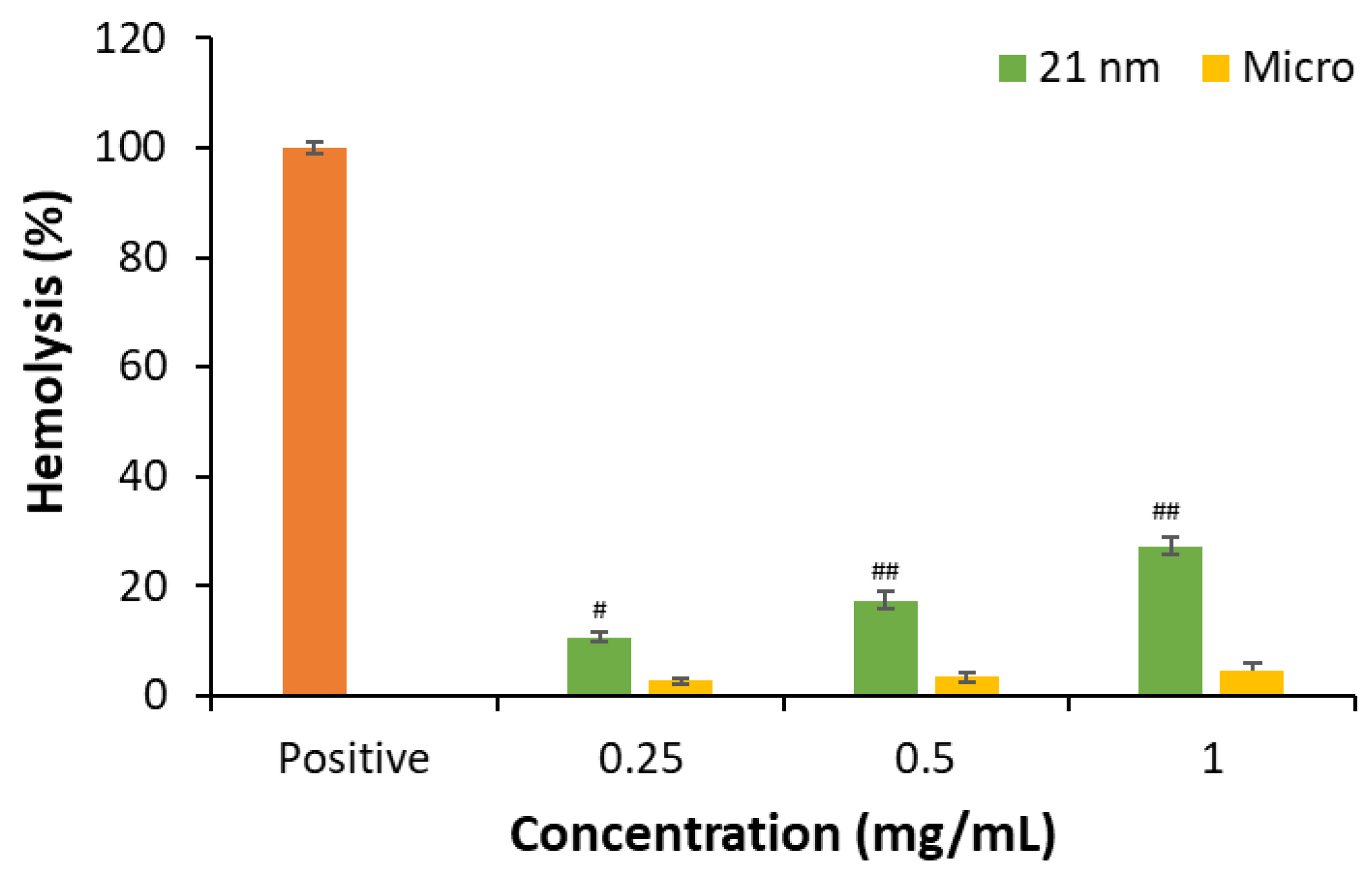
3.2.1. Hemolytic activity of TiO2
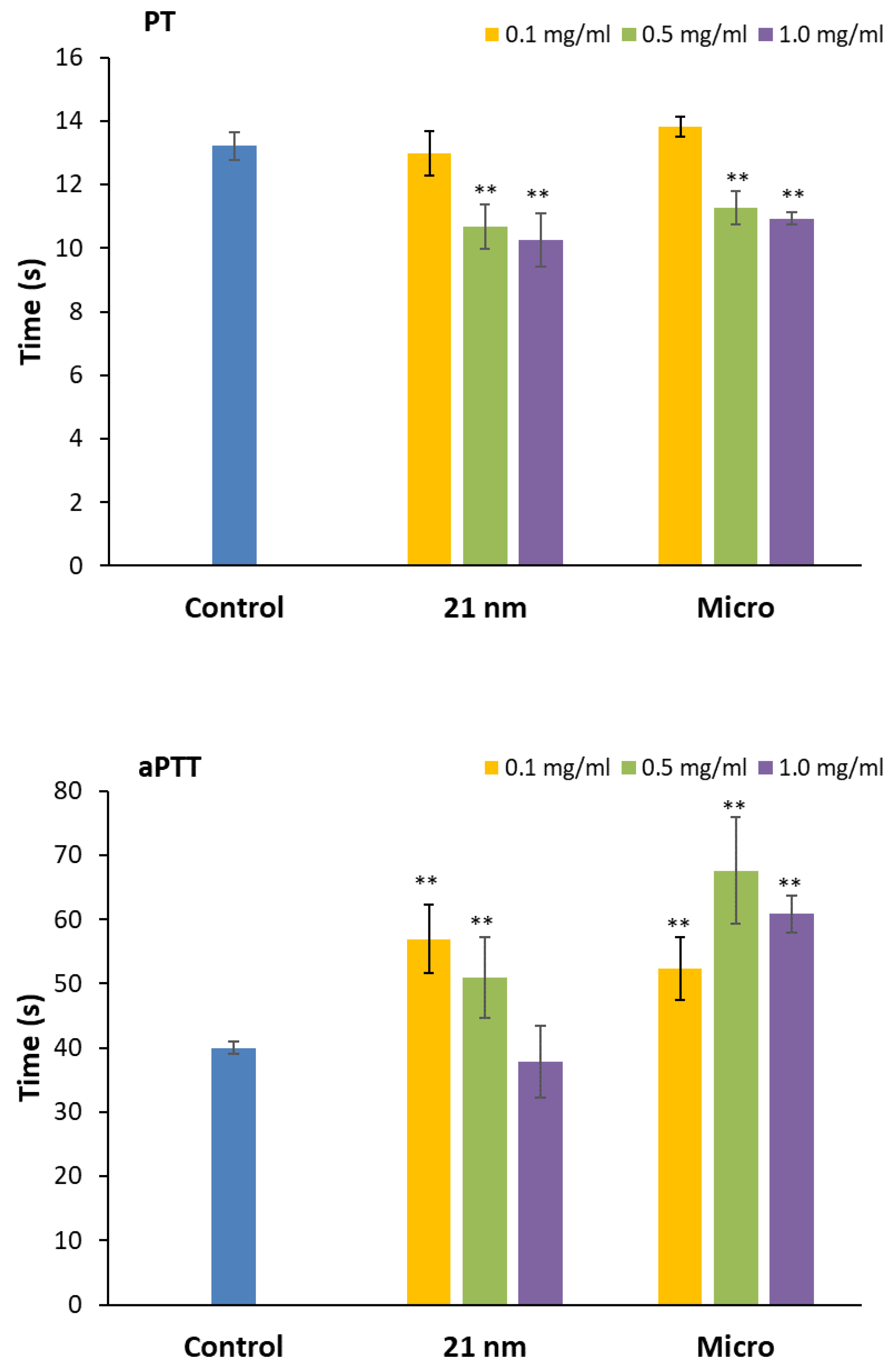
3.2.2. Effect of TiO2 on prothrombin and activated partial thromboplastin time
3.3. Cytotoxity and phototoxicity of TiO2 particles
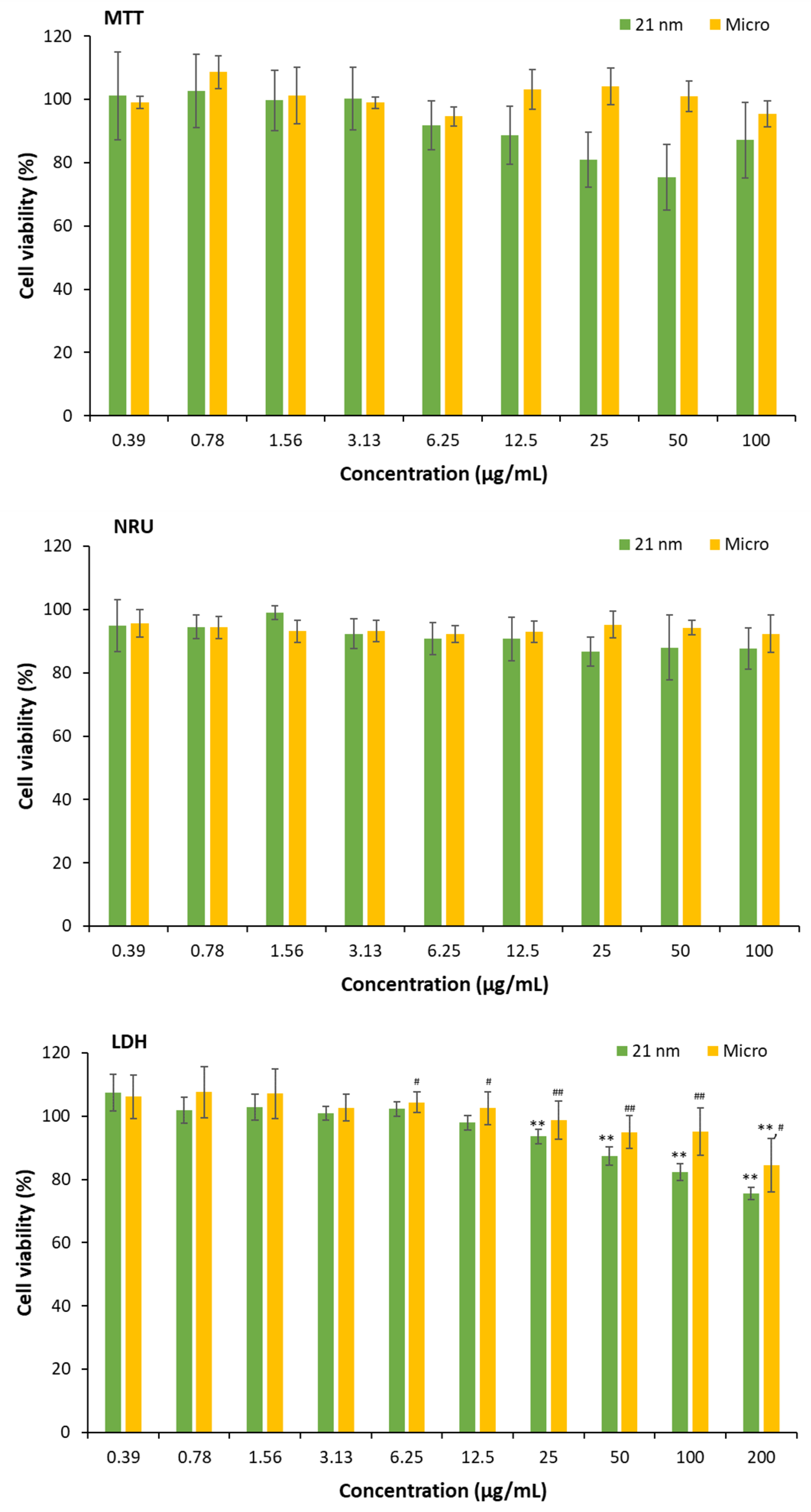
3.3.1. Study of potential interactions or interferences of TiO2 NPs with cytotoxic assays
3.3.2. Cytotoxicity of TiO2 particles on HaCaT cells
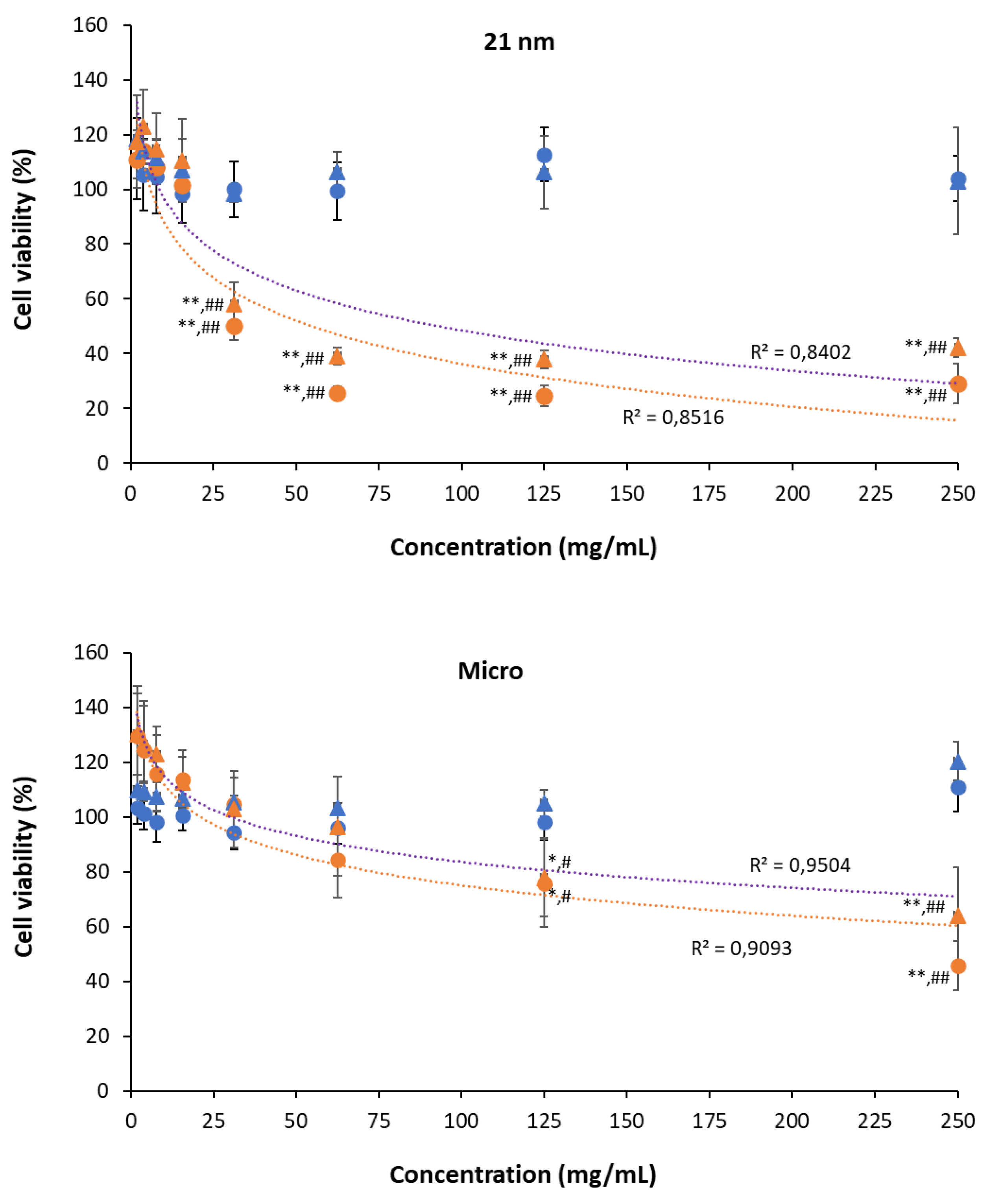
3.3.3. Phototoxicity of TiO2 particles on HaCaT cells
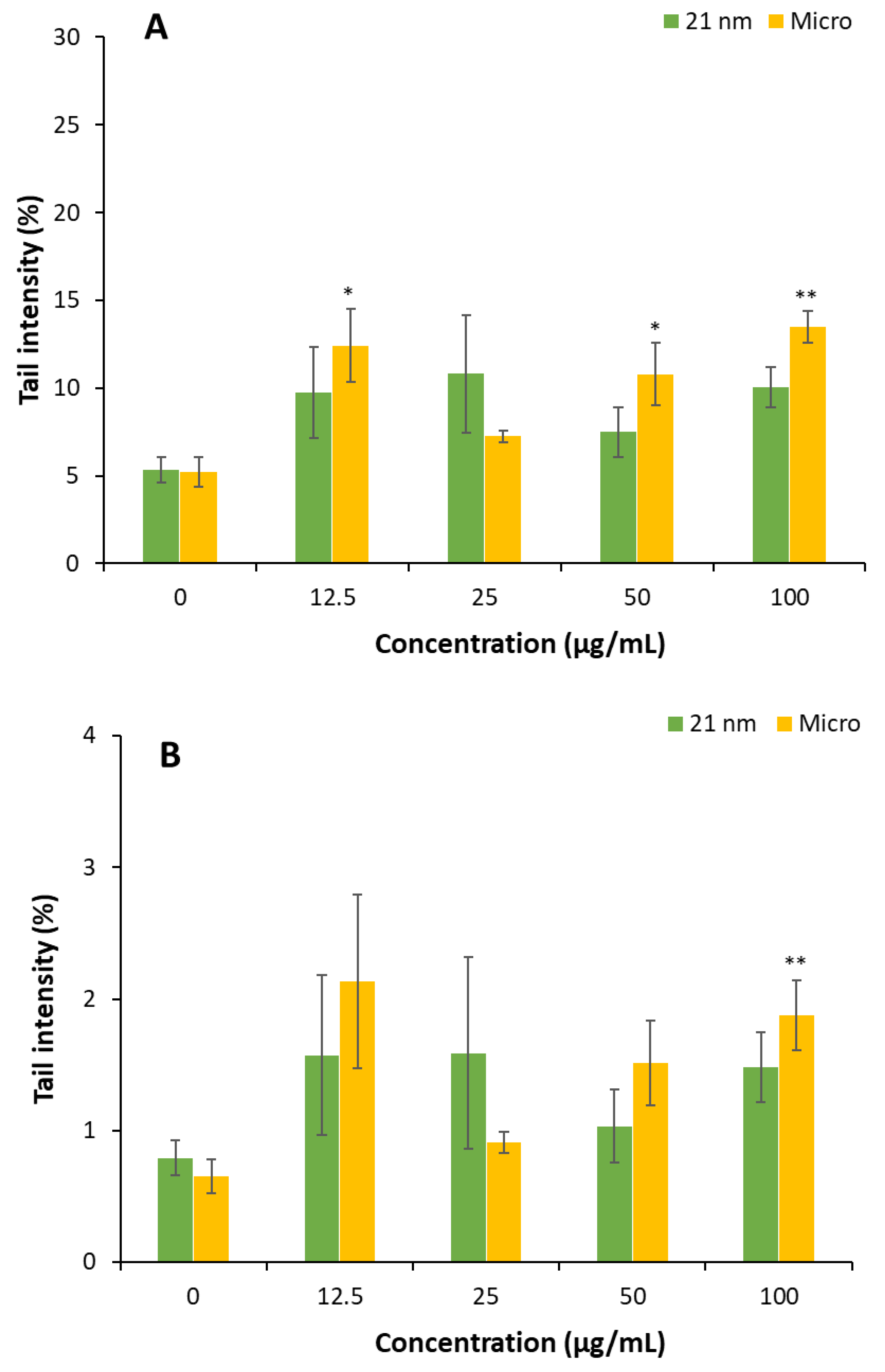
3.4. Genotoxicity of TiO2 particles on HaCaT cells.
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| aPTT | Activated partial thromboplastin time |
| CCiTUB | Centres Científics i Tecnològics de la Universitat de Barcelona” |
| DLS | Dynamic light scattering |
| DMSO | Dimethyl sulfoxide |
| FBS | Fetal Bovine Serum |
| HD | Hydrodynamic diameter |
| LDH | Lactate dehydrogenase |
| MTT | Thiazolyl Blue Tetrazolium Bromide |
| NPs | Nanoparticles |
| NR | Neutral Red Solution |
| NRU | Neutral Red Uptake |
| PBS | Phosphate Buffer Saline Solution |
| PDI | Polydispersity index |
| PIF | Photo-irritation-Factor |
| PT | Prothrombin time, |
| RBC | Red blood cells |
| ROS | Reactive Oxygen Species |
| TEM | Transmission Electron Microcopy |
| UV | Ultraviolet |
References
- WHO, Skin cancer. https://www.iarc.who.int/cancer-type/skin-cancer/ (accessed on 08/10/2024).
- Arnold, M., Singh, D., Laversanne, M., Vignat, J., Vaccarella, S., Meheus, F., Cust, A.E., de Vries, E., Whiteman, D.C., Bray, F. Global Burden of Cutaneous Melanoma in 2020 and Projections to 2040. JAMA Dermatol. 2022, 158(5), 495-503. [CrossRef]
- Chaiyabutr, C., Sukakul, T., Kumpangsin, T., Bunyavaree, M., Charoenpipatsin, N., Wongdama, S., Boonchai, W. Ultraviolet filters in sunscreens and cosmetic products-A market survey. Contact Dermatitis. 2021, 85(1), 58-68. [CrossRef]
- National Academies of Sciences, Engineering, and Medicine. Review of Fate, Exposure, and Effects of Sunscreens in Aquatic Environments and Implications for Sunscreen Usage and Human Health. 2022. Washington (DC): National Academies Press (US). [CrossRef]
- Jiménez Reinosa, J., Leret, P., Álvarez-Docio, C.M., del Campo, A., Fernández, J.F. Enhancement of UV absorption behavior in ZnO–TiO2 composites. Bol. Soc. Esp. Cerám. Vidr., 2016, 55 (2), 55-62. [CrossRef]
- Borrás, M.C., Sluyter, R., Barker, P.J., Konstantinov, K., Bakand, S. Y2O3 decorated TiO2 nanoparticles: Enhanced UV attenuation and suppressed photocatalytic activity with promise for cosmetic and sunscreen applications. J. Photochem. Photobiol. B, 2020, 207,111883. [CrossRef]
- Kong, L., Barber, T., Aldinger, J., Bowman, L., Leonard, S., Zhao, J., Ding, M. ROS generation is involved in titanium dioxide nanoparticle-induced AP-1 activation through p38 MAPK and ERK pathways in JB6 cells. Environ Toxicol. 2022, 37(2), 237-244. [CrossRef]
- Wang, Q., Yang, Y., Li, P., Dong, R., Sun, C., Song, G., Wang, Y. Titanium dioxide nanoparticles induce apoptosis through ROS-Ca2+-p38/AKT/mTOR pathway in TM4 cells. J. Appl. Toxicol. 2024, 44(6), 818-832. [CrossRef]
- Hongbo, M., Brennan, A., Diamond, S. A. Photocatalytic reactive oxygen species production and phototoxicity of titanium dioxide nanoparticles are dependent on the solar ultraviolet radiation spectrum. Environmental Toxicology and Chemistry, 2012, 31(9), 2099–2107.
- Dréno, B., Alexis, A., Chuberre, B., Marinovich, M. Safety of titanium dioxide nanoparticles in cosmetics. J. Eur. Acad. Dermatol. Venereol. 2019, 33 (7), 34-46. [CrossRef]
- Larese Filon, F., Mauro, M., Adami, G., Bovenzi, M., Crosera, M. Nanoparticles skin absorption: New aspects for a safety profile evaluation. Regul Toxicol Pharmacol. 2015, 72(2), 310-22. [CrossRef]
- SCCS (Scientific Committee on Consumer Safety), Opinion on Titanium dioxide (TiO2), preliminary version of 7 August 2020, final version of 6 October 2020, SCCS/1617/20. https://health.ec.europa.eu/system/files/2021-11/sccs_o_238.pdf.
- Sanches, P.L., Geaquinto, L.R.O., Cruz, R., Schuck, D.C., Lorencini, M., Granjeiro, J.M., Ribeiro, A.R.L. Toxicity Evaluation of TiO2 Nanoparticles on the 3D Skin Model: A Systematic Review. Front. Bioeng. Biotechnol. 2020, 8, 575. [CrossRef]
- Xuan, L., Ju, Z., Skonieczna, M., Zhou, P.K., Huang, R. Nanoparticles-induced potential toxicity on human health: Applications, toxicity mechanisms, and evaluation models. MedComm. 2020, 4(4), e327. [CrossRef]
- Commission Regulation (EU) 2022/63 of 14 January 2022 amending Annexes II and III to Regulation (EC) No 1333/2008 of the European Parliament and of the Council as regards the food additive titanium dioxide (E 171).
- Pokharkar, V., Chandak, S., Pawar, R., Khandke, A. The implications of the EU ban on titanium dioxide: A comprehensive review of safety concerns and alternatives. Ann. Pharm. Fr. 2024, 15, S0003-4509(24)00167-6. [CrossRef]
- Mitjans, M., Marics, L., Bilbao, M., Maddaleno, A.S., Piñero, J.J., Vinardell, M.P. Size Matters? A Comprehensive In Vitro Study of the Impact of Particle Size on the Toxicity of ZnO. Nanomaterials. 2023, 13, 1800. [CrossRef]
- Park, S., Park, J.K. Back to basics: the coagulation pathway. Blood Res. 2024, 59(1), 35. [CrossRef]
- Guadagnini, R., Halamoda Kenzaoui, B., Walker, L., Pojana, G., Magdolenova, Z., Bilanicova, D., Saunders, M., Juillerat-Jeanneret, L., Marcomini, A., Huk, A., Dusinska, M., Fjellsbø, L.M., Marano, F., Boland, S. Toxicity screenings of nanomaterials: challenges due to interference with assay processes and components of classic in vitro tests. Nanotoxicology. 2015, 9(1), 13-24. [CrossRef]
- OECD. OECD Test No. 432: In vitro 3T3 NRU Phototoxicity Test. OECD Guidelines for the Testing of Chemicals, 2019, Section 4. OECD Publishing, Paris. [CrossRef]
- Martínez, V., Galbiati, V., Corsini, E., Martin-Venegas, R., Vinardell, M.P., Mitjans, M. Establishment of an in vitro photoassay using THP-1 cells and IL-8 to discriminate photoirritants from photoallergens. Toxicology in Vitro. 2013, 27, 1920-1927. [CrossRef]
- Singh, N.P., McCoy, M.T., Tice, R.R., Schneider, E.L. A Simple Technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res. 1988, 175, 184–191. [CrossRef]
- Krishan, A., Dandekar, P.D. DAPI Fluorescence in Nuclei Isolated from Tumors. J. Histochem. Cytochem. 2005, 53 (8), 1033–1036. [CrossRef]
- Natarajan, V., Wilson, C.L., Hayward, S.L., Kidambi, S. Titanium Dioxide Nanoparticles Trigger Loss of Function and Perturbation of Mitochondrial Dynamics in Primary Hepatocytes. PLOS ONE. 2015, 10(8), e0134541. [CrossRef]
- Leroux, M.M., Doumandji, Z., Chézeau, L., Gaté, L., Nahle, S., Hocquel, R., Zhernovkov, V., Migot, S., Ghanbaja, J., Bonnet, C., Schneider, R.R., Rihn, B.H., Ferrari, L., Joubert, O. Toxicity of TiO2 Nanoparticles: Validation of Alternative Models. Int. J. Mol. Sci. 2020, 21, 4855. [CrossRef]
- Carrillo-Romero, J., Mentxaka, G., García-Salvador, A., Katsumiti, A., Carregal-Romero, S., Goñi-de-Cerio, F. Assessing the Toxicity of Metal- and Carbon-Based Nanomaterials In Vitro: Impact on Respiratory, Intestinal, Skin, and Immune Cell Lines. Int. J. Mol. Sci. 2024, 25(20), 10910. [CrossRef]
- Niska, K., Muszyńska, B., Kowalski, S., Tomaszewska, E., Narajczyk, M., Pawłowska, M., Majewski, P.W., Augustin, E., Inkielewicz-Stepniak, I. Cytotoxicity of ZnO nanoparticles in human melanocyte cells in the presence or absence of UV radiation: A preliminary comparative study with TiO2 nanoparticles. Toxicol In Vitro. 2025, 106, 106051. [CrossRef]
- Fornaguera, C., Calderó, G., Mitjans, M., Vinardell, M.P., Solans, C., Vauthier, C. Interactions of PLGA nanoparticles with blood components: protein adsorption, coagulation, activation of the complement system and hemolysis studies. Nanoscale. 2015, 7(14), 6045-58. [CrossRef]
- Murugadoss, S., Brassinne, F., Sebaihi, N., Petry, J., Cokic, S.M., Van Landuyt, K.L., Godderis, L., Mast, J., Lison, D., Hoet, P.H., van den Brule, S. Agglomeration of titanium dioxide nanoparticles increases toxicological responses in vitro and in vivo. Part. Fibre Toxicol. 2020, 17, 10. [CrossRef]
- Allouni, Z. E., Gjerdet, N. R., Cimpan, M. R. i Høl, P. J. The effect of blood protein adsorption on cellular uptake of anatase TiO2 nanoparticles. Int. J. Nanomedicine. 2015, 10, 687-695. [CrossRef]
- https://www.malvernpanalytical.com/en/learn/knowledge-center/insights/polydispersity-what-does-it-mean-for-dls-and-chromatography. 9/10/2024.
- Preedia Babu, E., Subastri, A., Suyavaran, A., Premkumar, K., Sujatha, V., Aristatile, B., Alshammari, G.M., Dharuman, V., Thirunavukkarasu, C. Size Dependent Uptake and Hemolytic Effect of Zinc Oxide Nanoparticles on Erythrocytes and Biomedical Potential of ZnO-Ferulic acid Conjugates. Sci. Rep. 2017, 7, 4203. [CrossRef]
- Yedgar, S., Barshtein, G., Gural, A. Hemolytic Activity of Nanoparticles as a Marker of Their Hemocompatibility. Micromachines (Basel). 2022, 13(12), 2091. [CrossRef]
- Lozano-Fernández, T., Dobrovolskaia, M., Camacho, T., González-Fernández, A., Simón-Vázquez, R. Interference of Metal Oxide Nanoparticles with Coagulation Cascade and Interaction with Blood Components. Part. Part. Syst. Charact. 2019, 36, 1800547. [CrossRef]
- Huang, H.H., Chen, Z.H., Nguyen, D.T., Tseng, C.M., Chen, C.S., Chang, J.H. Blood Coagulation on Titanium Dioxide Films with Various Crystal Structures on Titanium Implant Surfaces. Cells. 2022, 11(17), 2623. [CrossRef]
- Kushida, T., Saha, K., Subramani, C., Nandwana, V., Rotello, V.M. Effect of nano-scale curvature on the intrinsic blood coagulation system. Nanoscale. 2014, 6(23), 14484-7. [CrossRef]
- Ilinskaya, A.N., Dobrovolskaia, M.A. Nanoparticles and the blood coagulation system. Part II: safety concerns. Nanomedicine (Lond). 2013, 8(6), 969-81. [CrossRef]
- Martínez-Gutierrez, F. , Thi, E.P., Silverman, J.M., de Oliveira, C.C., Svensson, S.L., Vanden Hoek, A., Sánchez, E.M., Reiner, N.E., Gaynor, E.C., Pryzdial, E.L., Conway, E.M., Orrantia, E., Ruiz, F., Av-Gay, Y., Bach, H. Antibacterial activity, inflammatory response, coagulation and cytotoxicity effects of silver nanoparticles. Nanomedicine. 2012, 8(3), 328-36. [CrossRef]
- Soddu, L., Trinh, D.N., Dunne, E., Kenny, D., Bernardini, G., Kokalari, I., Marucco, A., Monopoli, M.P., Fenoglio, I. Identification of physicochemical properties that modulate nanoparticle aggregation in blood. Beilstein J. Nanotechnol. 2020, 11, 550-567. [CrossRef]
- Fu, W., Liu, M., Wang, Y., Yang, H., Ye, A., Wu, J., Li, Y., Yu, Z., Qiu, Y., Xu, L. Nano titanium dioxide induces HaCaT cell pyroptosis via regulating the NLRP3/caspase-1/GSDMD pathway. Toxicol Lett. 2024, 402, 27-37. [CrossRef]
- Horie, M., Sugino, S., Kato, H., Tabei, Y., Nakamura, A., Yoshida, Y. Does photocatalytic activity of TiO2 nanoparticles correspond to photo-cytotoxicity? Cellular uptake of TiO2 nanoparticles is important in their photo-cytotoxicity. Toxicol. Mech. Methods. 2016, 26(4), 284-94. [CrossRef]
- Yin, J.J., Liu, J., Ehrenshaft, M., Roberts, J.E., Fu, P.P., Mason, R.P., Zhao, B. Phototoxicity of nano titanium dioxides in HaCaT keratinocytes--generation of reactive oxygen species and cell damage. Toxicol. Appl. Pharmacol. 2012, 263(1), 81-8. [CrossRef]
- Xiong, S., George, S., Ji, Z., Lin, S., Yu, H., Damoiseaux, R., France, B., Ng, K.W., Loo, S.C. Size of TiO2 nanoparticles influences their phototoxicity: an in vitro investigation. Arch. Toxicol. 2013, 87(1), 99-109. [CrossRef]
- Kim, I.Y., Lee, T.G., Reipa, V., Heo, M.B. Titanium Dioxide Induces Apoptosis under UVA Irradiation via the Generation of Lysosomal Membrane Permeabilization-Dependent Reactive Oxygen Species in HaCat Cells. Nanomaterials. 2021, 11(8), 1943. [CrossRef]
- Park, Y.H., Jeong, S.H., Yi, S.M., Choi, B.H., Kim, Y.R., Kim, I.K., Kim, M.K., Son, S.W. Analysis for the potential of polystyrene and TiO2 nanoparticles to induce skin irritation, phototoxicity, and sensitization. Toxicol. In Vitro. 2011, 25(8),1863-9. [CrossRef]
- Tang, Y., Cai, R., Cao, D., Kong, X., Lu, Y. Photocatalytic production of hydroxyl radicals by commercial TiO2 nanoparticles and phototoxic hazard identification. Toxicology. 2018, 406-407, 1-8. [CrossRef]
- Carriere, M., Arnal, M.E., Douki, T. TiO2 genotoxicity: An update of the results published over the last six years. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2020, 854-855, 503198. [CrossRef]
- Charles, S., Jomini, S., Fessard, V., Bigorgne-Vizade, E., Rousselle, C., Michel, C. Assessment of the in vitro genotoxicity of TiO2 nanoparticles in a regulatory context. Nanotoxicology. 2018, 12(4), 357-374. [CrossRef]
- Fenoglio, I., Ponti, J., Alloa, E., Ghiazza, M., Corazzari, I., Capomaccio, R., Rembges, D., Oliaro-Bosso, S., Rossi, F. Singlet oxygen plays a key role in the toxicity and DNA damage caused by nanometric TiO2 in human keratinocytes. Nanoscale. 2013, 5(14), 6567-76. [CrossRef]
- Rashid, M.M., Forte Tavčer, P., Tomšič, B. Influence of Titanium Dioxide Nanoparticles on Human Health and the Environment. Nanomaterials. 2021, 11, 2354. [CrossRef]
- Johnston, H.J., Hutchison, G.R., Christensen, F.M., Peters, S., Hankin, S., Stone, V. Identification of the mechanisms that drive the toxicity of TiO2 particulates: the contribution of physicochemical characteristics. Part. Fibre. Toxicol. 2009, 6, 33. [CrossRef]

| Particle Size | Incubation Time | PBS* | PBS + BSA* | PBS + Fib* | DMEM* |
|---|---|---|---|---|---|
| 21 nm | 2 hours | 458.0 ± 6.0 | 362.8 ± 16.4 | 403. 5 ± 33.8 | 311.4 ± 7.2¥ |
| 24 hours | 485.4 ± 15.1 | 348.1 ± 6.6¥¥ | 404.7 ± 57.1 | 288.2 ± 17.2¥¥ | |
| Micro | 2 hours | 478.5 ± 20.5 | 236.3 ± 20.6¥¥ | 402.5 ± 16.18 | 280.6 ± 12.4¥¥ |
| 24 hours | 606.3 ± 11.4$$ | 158.8± 10.9¥¥, $$ | 428.4 ± 5.1¥¥ | 280.0 ± 2.9¥¥ |
| Particle Size | MTT | NRU |
|---|---|---|
| 21 nm | 4.6 ± 0.5 | 3.0 ± 0.5 |
| Micro | 1.3 ± 0.3 | 1.3 ± 0.3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




