Submitted:
08 May 2025
Posted:
09 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Animals and Experimental Design
2.2. Ketogenic Substrate Administration
2.3. Blood BHB Measurement
2.4. Induction of Allergic Dermatitis
2.5. Measurement of Ear Thickness
2.6. Histological Preparation and Toluidine Blue (TB) Staining
2.7. Statistical Analysis
3. Results
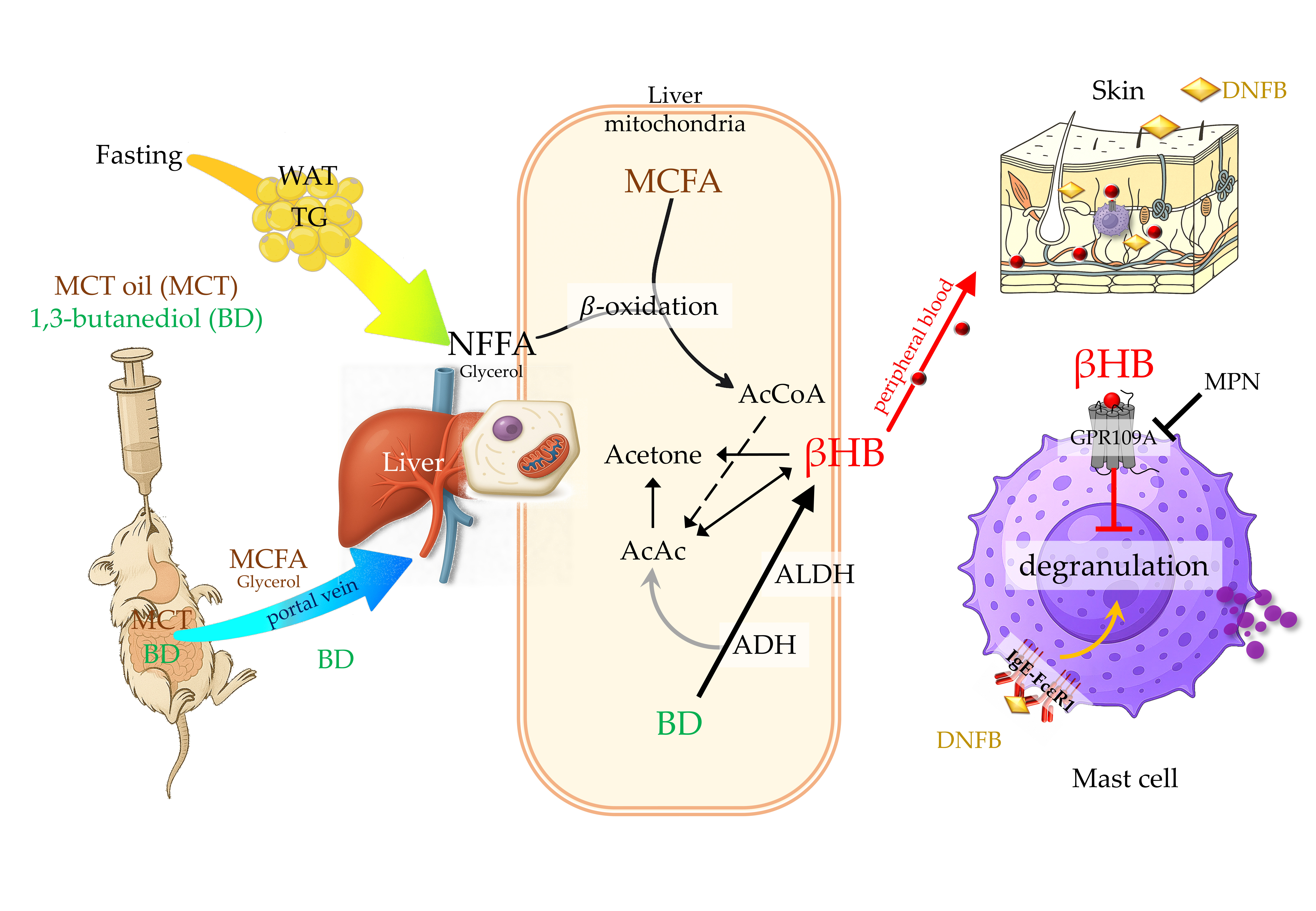
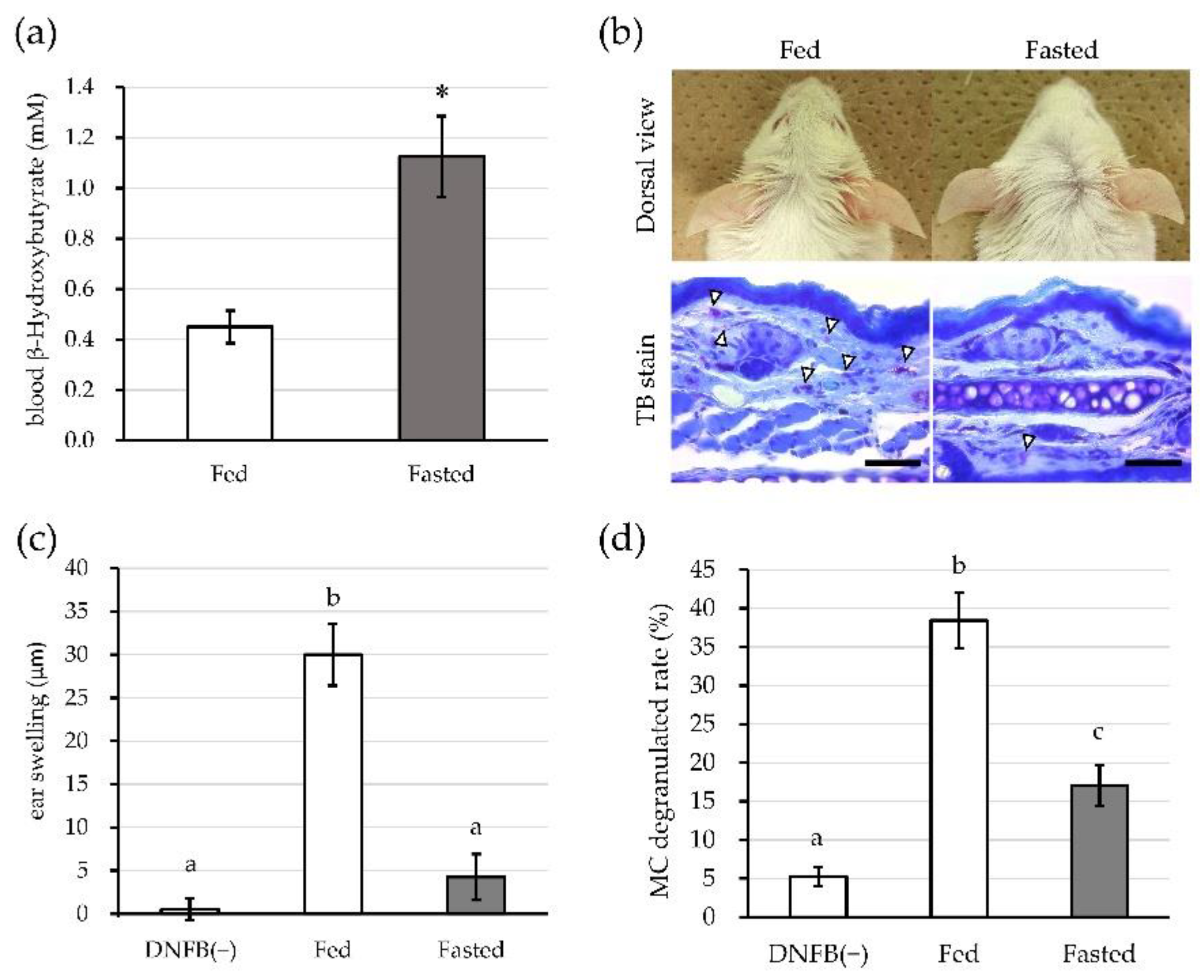
3.1. Fasting Increases BHB Levels and Suppresses Immediate-Type Allergic Response
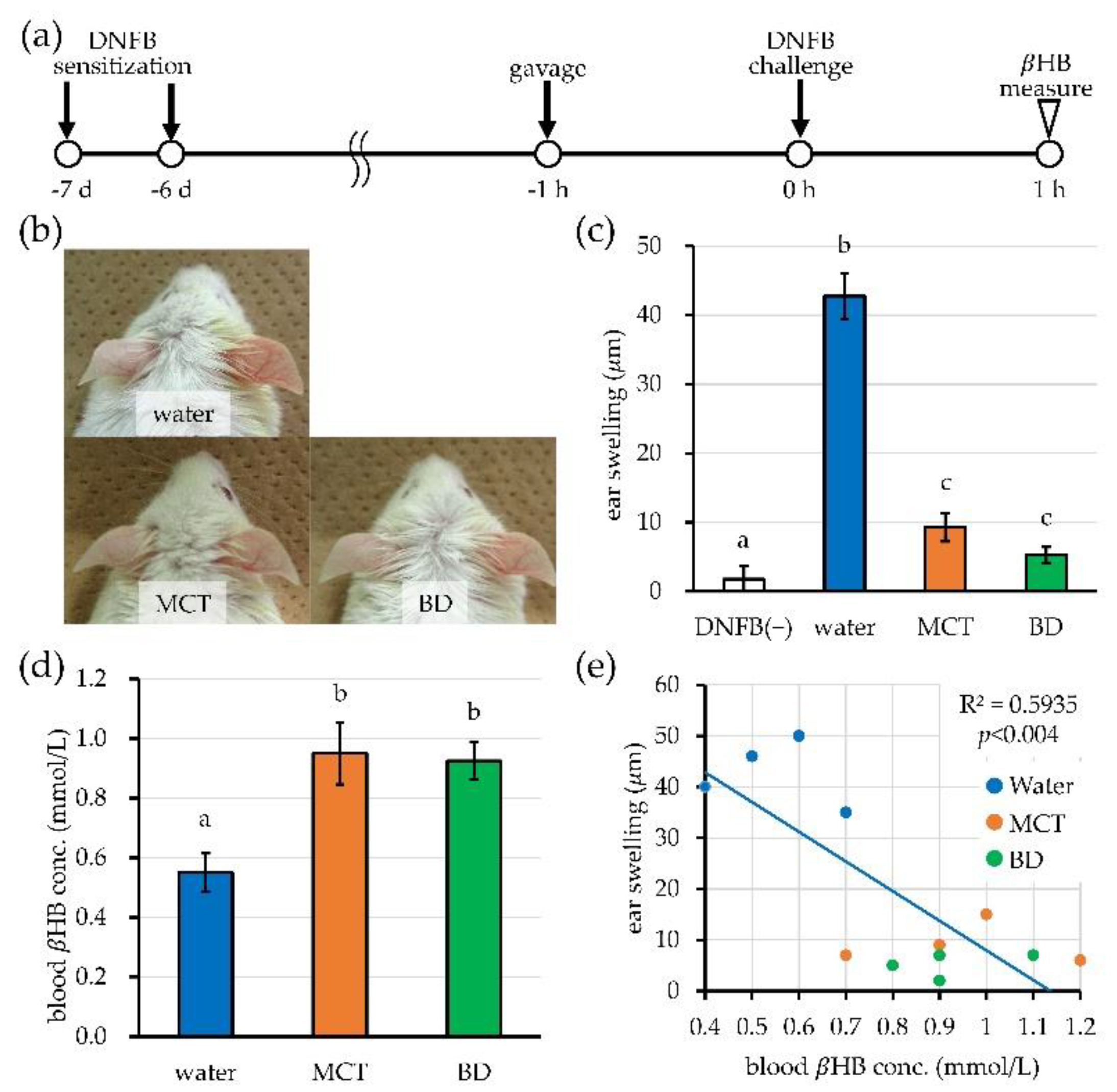
3.2. Nutritional Ketosis via MCT Oil and BD Attenuates Immediate-Type Allergic Response
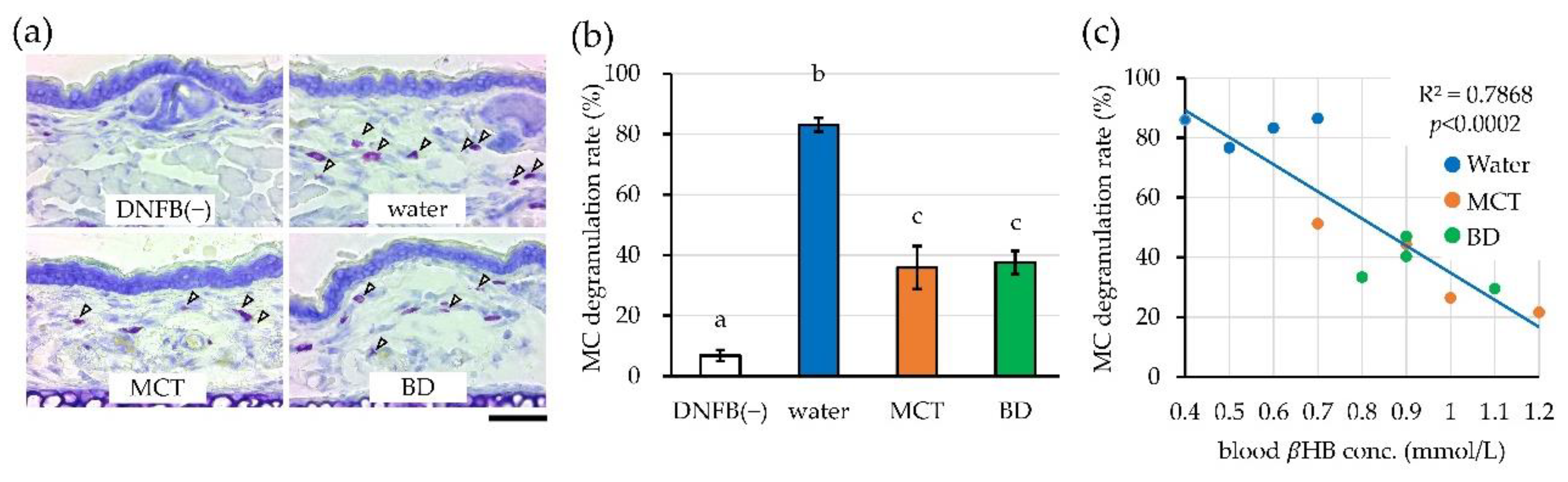
3.3. MCT Oil and BD Suppress MC Degranulation in Association with Elevated BHB Levels
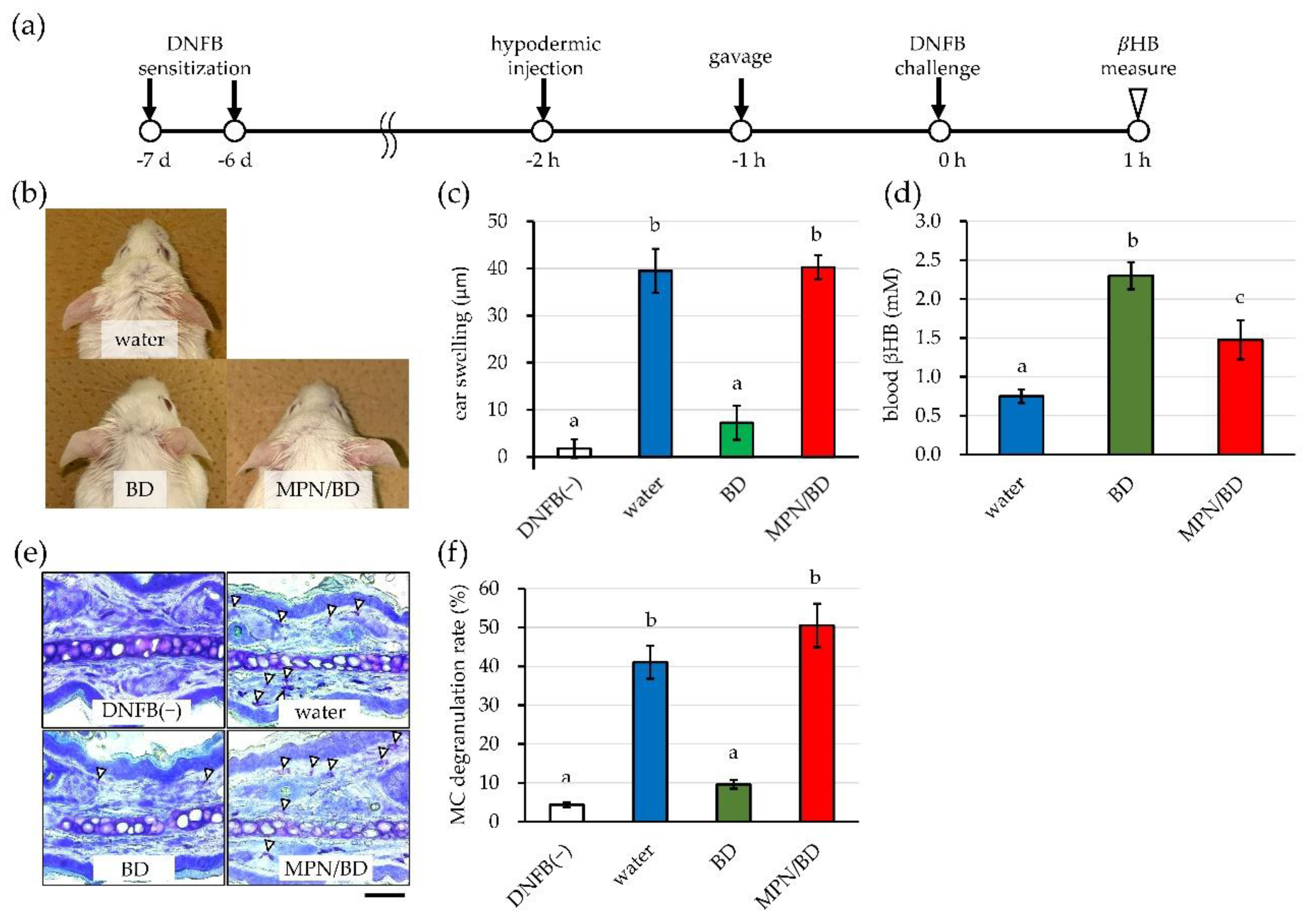
3.4. GPR109A Antagonist Mepenzolate Abolishes the Inhibitory Effects of BD on Allergic Inflammation
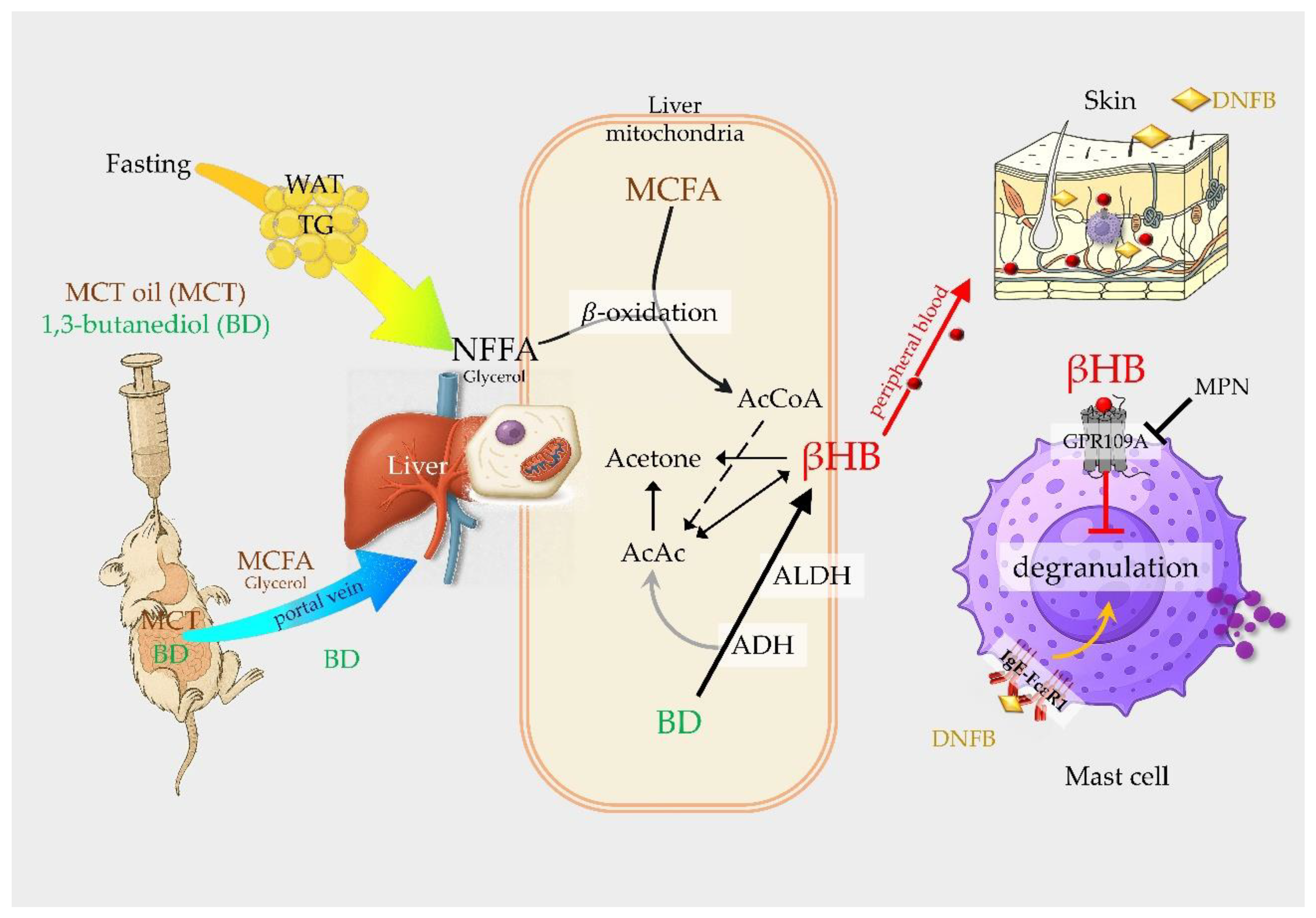
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ACD | Allergic Contact Dermatitis |
| ADH | Alcohol Dehydrogenase |
| ALDH | Aldehyde Dehydrogenase |
| ANOVA | Analysis of Variance |
| BHB | β-Hydroxybutyrate |
| BD | 1,3-Butanediol |
| COX-2 | Cyclooxygenase-2 |
| DNFB | 2,4-Dinitrofluorobenzene |
| FcεRI | Fc epsilon receptor I |
| GPR109A | G Protein-Coupled Receptor 109A |
| HDAC | Histone Deacetylase |
| IgE | Immunoglobulin E |
| IL | Interleukin |
| MC | Mast Cell |
| MCT | Medium-Chain Triglyceride |
| MPN | Mepenzolate Bromide |
| NLRP3 | NOD-, LRR- and Pyrin Domain-Containing Protein 3 |
| NFFA | Non-esterified Fatty Acids |
| SEM | Standard Error of the Mean |
| TB | Toluidine Blue |
| TCS | Topical Corticosteroids |
References
- Mraz, V.; Geisler, C.; Bonefeld, C.M. Dendritic Epidermal T Cells in Allergic Contact Dermatitis. Front Immunol 2020, 11, 874. [Google Scholar] [CrossRef] [PubMed]
- Brites, G.S.; Ferreira, I.; Sebastiao, A.I.; Silva, A.; Carrascal, M.; Neves, B.M.; Cruz, M.T. Allergic contact dermatitis: From pathophysiology to development of new preventive strategies. Pharmacol Res 2020, 162, 105282. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Kurashima, Y. Two Sides of the Coin: Mast Cells as a Key Regulator of Allergy and Acute/Chronic Inflammation. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Aitella, E.; De Martinis, M.; Romano, C.; Azzellino, G.; Ginaldi, L. Neurogenic Inflammation in Allergic Contact Dermatitis. Biomedicines 2025, 13. [Google Scholar] [CrossRef]
- Numata, T.; Harada, K.; Nakae, S. Roles of Mast Cells in Cutaneous Diseases. Front Immunol 2022, 13, 923495. [Google Scholar] [CrossRef]
- Qi, J.; Gan, L.; Fang, J.; Zhang, J.; Yu, X.; Guo, H.; Cai, D.; Cui, H.; Gou, L.; Deng, J.; et al. Beta-Hydroxybutyrate: A Dual Function Molecular and Immunological Barrier Function Regulator. Front Immunol 2022, 13, 805881. [Google Scholar] [CrossRef]
- Rojas-Morales, P.; Tapia, E.; Pedraza-Chaverri, J. beta-Hydroxybutyrate: A signaling metabolite in starvation response? Cell Signal 2016, 28, 917–923. [Google Scholar] [CrossRef]
- Youm, Y.H.; Nguyen, K.Y.; Grant, R.W.; Goldberg, E.L.; Bodogai, M.; Kim, D.; D’Agostino, D.; Planavsky, N.; Lupfer, C.; Kanneganti, T.D.; et al. The ketone metabolite beta-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat Med 2015, 21, 263–269. [Google Scholar] [CrossRef]
- Boleslawska, I.; Kowalowka, M.; Boleslawska-Krol, N.; Przyslawski, J. Ketogenic Diet and Ketone Bodies as Clinical Support for the Treatment of SARS-CoV-2-Review of the Evidence. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Stubbs, B.J.; Koutnik, A.P.; Goldberg, E.L.; Upadhyay, V.; Turnbaugh, P.J.; Verdin, E.; Newman, J.C. Investigating Ketone Bodies as Immunometabolic Countermeasures against Respiratory Viral Infections. Med 2020, 1, 43–65. [Google Scholar] [CrossRef]
- Shaw, D.M.; Merien, F.; Braakhuis, A.; Plews, D.; Laursen, P.; Dulson, D.K. The Effect of 1,3-Butanediol on Cycling Time-Trial Performance. Int J Sport Nutr Exerc Metab 2019, 29, 466–473. [Google Scholar] [CrossRef] [PubMed]
- Chen, O.; Blonquist, T.M.; Mah, E.; Sanoshy, K.; Beckman, D.; Nieman, K.M.; Winters, B.L.; Anthony, J.C.; Verdin, E.; Newman, J.C.; et al. Tolerability and Safety of a Novel Ketogenic Ester, Bis-Hexanoyl (R)-1,3-Butanediol: A Randomized Controlled Trial in Healthy Adults. Nutrients 2021, 13. [Google Scholar] [CrossRef] [PubMed]
- Poff, A.M.; Rho, J.M.; D’Agostino, D.P. Ketone Administration for Seizure Disorders: History and Rationale for Ketone Esters and Metabolic Alternatives. Front Neurosci 2019, 13, 1041. [Google Scholar] [CrossRef] [PubMed]
- Heidt, C.; Fobker, M.; Newport, M.; Feldmann, R.; Fischer, T.; Marquardt, T. Beta-Hydroxybutyrate (BHB), Glucose, Insulin, Octanoate (C8), and Decanoate (C10) Responses to a Medium-Chain Triglyceride (MCT) Oil with and without Glucose: A Single-Center Study in Healthy Adults. Nutrients 2023, 15. [Google Scholar] [CrossRef]
- Tate, R.L.; Mehlman, M.A.; Tobin, R.B. Metabolic fate of 1,3-butanediol in the rat: conversion to -hydroxybutyrate. J Nutr 1971, 101, 1719–1726. [Google Scholar] [CrossRef]
- Kirsch, J.R.; D’Alecy, L.G.; Mongroo, P.B. Butanediol induced ketosis increases tolerance to hypoxia in the mouse. Stroke 1980, 11, 506–513. [Google Scholar] [CrossRef]
- Thio, C.L.; Lai, A.C.; Ting, Y.T.; Chi, P.Y.; Chang, Y.J. The ketone body beta-hydroxybutyrate mitigates ILC2-driven airway inflammation by regulating mast cell function. Cell Rep 2022, 40, 111437. [Google Scholar] [CrossRef]
- Nagata, K.; Ando, D.; Ashikari, T.; Ito, K.; Miura, R.; Fujigaki, I.; Goto, Y.; Ando, M.; Ito, N.; Kawazoe, H.; et al. Butyrate, Valerate, and Niacin Ameliorate Anaphylaxis by Suppressing IgE-Dependent Mast Cell Activation: Roles of GPR109A, PGE2, and Epigenetic Regulation. J Immunol 2024, 212, 771–784. [Google Scholar] [CrossRef]
- Singh, V.; Jamwal, S.; Jain, R.; Verma, P.; Gokhale, R.; Rao, K.V. Mycobacterium tuberculosis-driven targeted recalibration of macrophage lipid homeostasis promotes the foamy phenotype. Cell Host Microbe 2012, 12, 669–681. [Google Scholar] [CrossRef]
- Tran, M.T.; Zsengeller, Z.K.; Berg, A.H.; Khankin, E.V.; Bhasin, M.K.; Kim, W.; Clish, C.B.; Stillman, I.E.; Karumanchi, S.A.; Rhee, E.P.; et al. PGC1alpha drives NAD biosynthesis linking oxidative metabolism to renal protection. Nature 2016, 531, 528–532. [Google Scholar] [CrossRef]
- Yoshimura, Y.; Takahashi, M. Effects of Triacetin on AMPK Activation and Immune Responses in Allergic Contact Dermatitis. Allergies 2024, 4, 254–267. [Google Scholar] [CrossRef]
- Fujii, A.; Kimura, R.; Mori, A.; Yoshimura, Y. Sucrose Solution Ingestion Exacerbates Dinitrofluorobenzene-Induced Allergic Contact Dermatitis in Rats. Nutrients 2024, 16, 1962. [Google Scholar] [CrossRef]
- Nakamura, S.; Hisamura, R.; Shimoda, S.; Shibuya, I.; Tsubota, K. Fasting mitigates immediate hypersensitivity: a pivotal role of endogenous D-beta-hydroxybutyrate. Nutr Metab (Lond) 2014, 11, 40. [Google Scholar] [CrossRef]
- Shimazu, T.; Hirschey, M.D.; Newman, J.; He, W.; Shirakawa, K.; Le Moan, N.; Grueter, C.A.; Lim, H.; Saunders, L.R.; Stevens, R.D.; et al. Suppression of oxidative stress by beta-hydroxybutyrate, an endogenous histone deacetylase inhibitor. Science 2013, 339, 211–214. [Google Scholar] [CrossRef]
- Li, A.W.; Yin, E.S.; Antaya, R.J. Topical Corticosteroid Phobia in Atopic Dermatitis: A Systematic Review. JAMA Dermatol 2017, 153, 1036–1042. [Google Scholar] [CrossRef]
- Rathi, S.K.; D’Souza, P. Rational and ethical use of topical corticosteroids based on safety and efficacy. Indian J Dermatol 2012, 57, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, Y.; Tanaka, K.; Asano, T.; Yamakawa, N.; Kobayashi, D.; Ishihara, T.; Hanaya, K.; Shoji, M.; Sugai, T.; Wada, M.; et al. Synthesis and biological comparison of enantiomers of mepenzolate bromide, a muscarinic receptor antagonist with bronchodilatory and anti-inflammatory activities. Bioorg Med Chem 2014, 22, 3488–3497. [Google Scholar] [CrossRef]
- Walker, M.A.; Tian, R. NAD(H) in mitochondrial energy transduction: implications for health and disease. Curr Opin Physiol 2018, 3, 101–109. [Google Scholar] [CrossRef] [PubMed]
- Edenberg, H.J. The genetics of alcohol metabolism: role of alcohol dehydrogenase and aldehyde dehydrogenase variants. Alcohol Res Health 2007, 30, 5–13. [Google Scholar]
- Vatamaniuk, M.Z.; Horyn, O.V.; Vatamaniuk, O.K.; Doliba, N.M. Acetylcholine affects rat liver metabolism via type 3 muscarinic receptors in hepatocytes. Life Sci 2003, 72, 1871–1882. [Google Scholar] [CrossRef]
- Li, J.H.; Gautam, D.; Han, S.J.; Guettier, J.M.; Cui, Y.; Lu, H.; Deng, C.; O’Hare, J.; Jou, W.; Gavrilova, O.; et al. Hepatic muscarinic acetylcholine receptors are not critically involved in maintaining glucose homeostasis in mice. Diabetes 2009, 58, 2776–2787. [Google Scholar] [CrossRef] [PubMed]
- Hanson, J.; Gille, A.; Zwykiel, S.; Lukasova, M.; Clausen, B.E.; Ahmed, K.; Tunaru, S.; Wirth, A.; Offermanns, S. Nicotinic acid- and monomethyl fumarate-induced flushing involves GPR109A expressed by keratinocytes and COX-2-dependent prostanoid formation in mice. J Clin Invest 2010, 120, 2910–2919. [Google Scholar] [CrossRef] [PubMed]
- Mukai, K.; Tsai, M.; Saito, H.; Galli, S.J. Mast cells as sources of cytokines, chemokines, and growth factors. Immunol Rev 2018, 282, 121–150. [Google Scholar] [CrossRef] [PubMed]
- Nestle, F.O.; Di Meglio, P.; Qin, J.Z.; Nickoloff, B.J. Skin immune sentinels in health and disease. Nat Rev Immunol 2009, 9, 679–691. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



