Submitted:
06 May 2025
Posted:
08 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
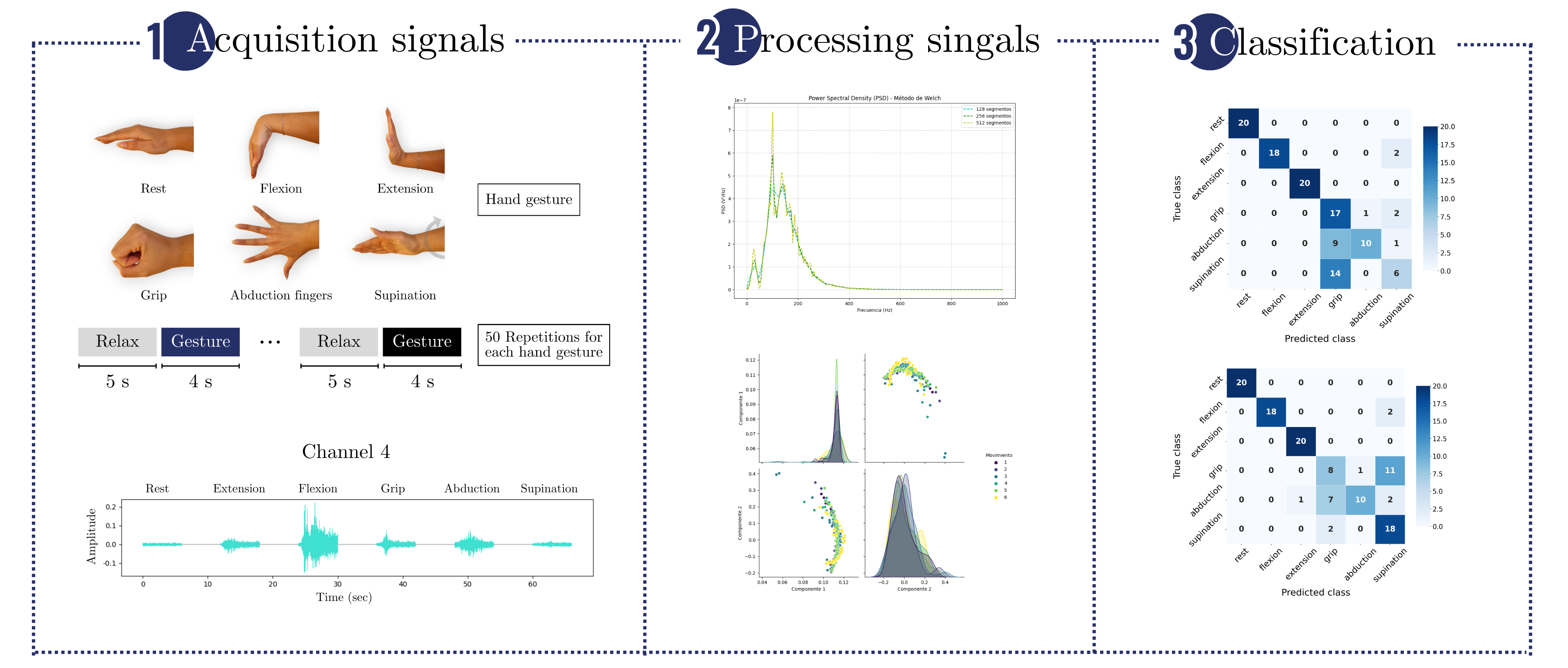
2.1. Dataset
2.1.1. Data from Healthy Individuals
2.1.2. Data Acquisition of EMG Signals from Stroke Patients
| Phase | Duration | Description |
|---|---|---|
| Preparation | ∼ 3 minutes | Electrode placement, signal calibration, and subject familiarization. |
| Gesture task | 4 seconds per trial | The subject maintains the specified gesture while EMG data is recorded. |
| Rest period | 5 seconds | The subject relaxes before the next trial to prevent fatigue. |
| Repetitions | 50 per gesture | Each gesture is repeated 50 times for consistency. |
| Total session time | ∼ 1 hour per patient | Including preparation, trials, and rest periods. |
2.2. Features Extraction
2.2.1. Power Spectral Density
2.2.2. Discrete Wavelet Transform
2.2.3. Principal Component Analysis
2.2.4. Singular Vector Descomposition
2.3. Classification Algorithms
2.3.1. Random Forest
2.3.2. Support Vector Machine
2.3.3. Neural Network
2.4. Metrics
- -
- Accuracy: The proportion of correctly classified instances among all instances, calculated globally across all classes.
- -
- Precision: The proportion of true positive predictions among all positive predictions.
- -
- Recall: The proportion of true positive instances correctly identified
- -
- F1-score: The harmonic mean of precision and recall.
- TP: True positives
- TN: True negatives
- FP: False positives
- FN: False negatives
2.5. Case Study Scenarios
- -
- Single-channel configurations: Channel 1, Channel 2, Channel 3, Channel 4.
- -
- Dual-channel configurations: Channels 1 & 2, Channels 3 & 4.
- -
- Multi-channel configuration: Channels 1, 2, 3 & 4.
2.5.1. Global Performance Evaluation
2.5.2. Robustness Analysis
- (a)
- Single configuration robustness test: A single temporal window and a specific channel configuration are selected based on the performing combination from the global performance analysis. The dataset is split into 40 train-test partitions for this configuration using previously optimized hyperparameters. The accuracy metric is evaluated across all feature extraction methods (PSD, PSD+PCA, PSD+SVD, DWT+PCA, DWT+SVD) and classification algorithms (RF, SVM, NN).
- (b)
- Multiple configurations robustness test: Three different temporal windows (0.5, 2, and 4 seconds) and three channel configurations (1, 2, and 4 channels) are evaluated to assess model stability under varying acquisition conditions. For each configuration, only the feature extraction methods that achieved the highest performance in the global evaluation are considered. The three classification algorithms (RF, SVM, NN) are tested, with 100 train-test partitions generated per setting. Performance is evaluated based on accuracy, recall, and precision.
2.5.3. Global Performance Evaluation for Stroke Patients Data
2.5.4. Model Generalization Capacity
- (a)
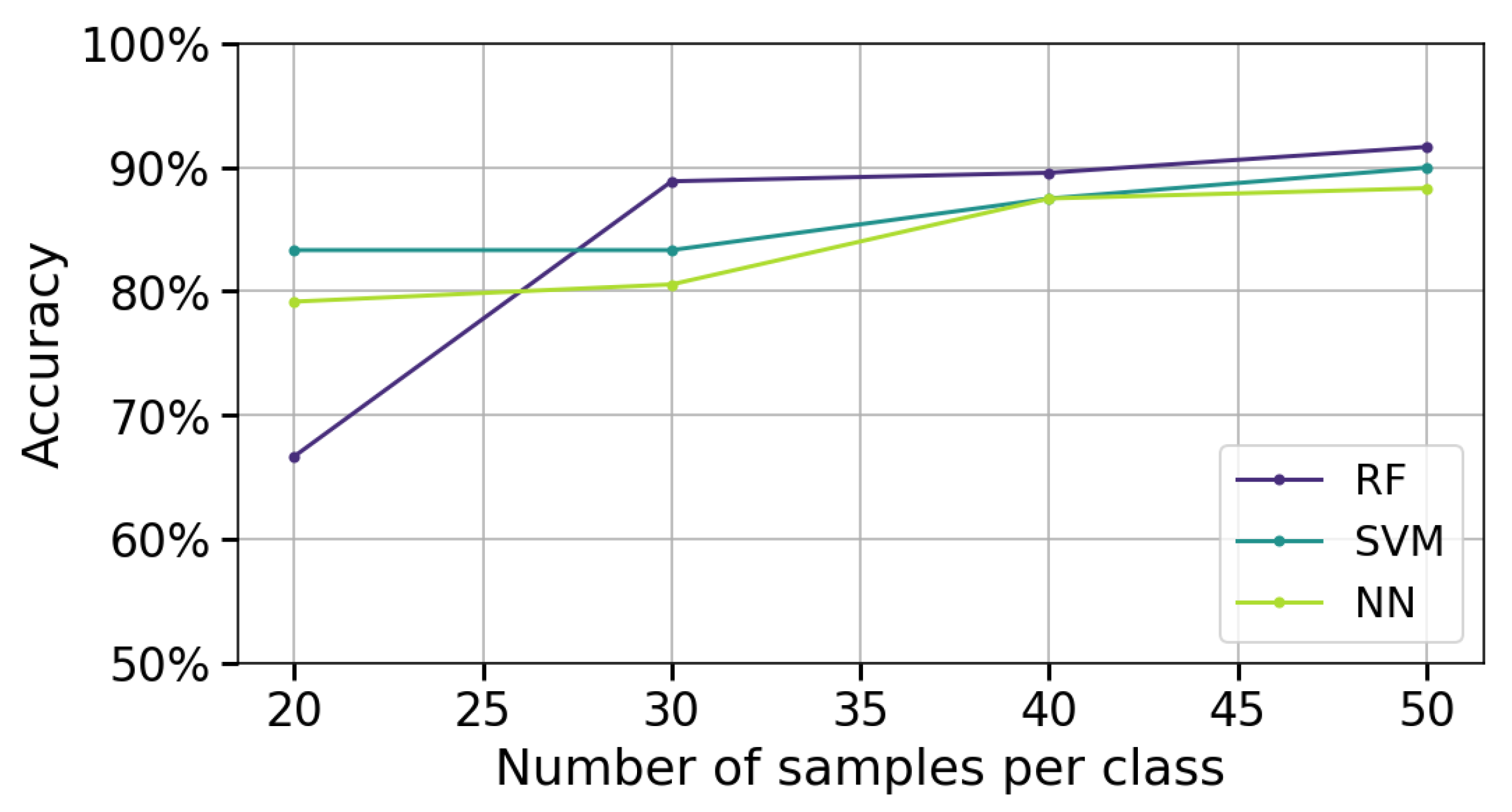
- Intra-patient validation: The temporal and spatial configuration selected from the Global Performance Evaluation on stroke patients’ data is used, but datasets are constructed with 20 and 30 measurements per gesture. The same feature extraction method is applied, and the performance of the three classification algorithms (RF, SVM, NN) is evaluated using accuracy, recall, and precision metrics. This evaluation measures the model’s intra-subject generalization capacity.
- (b)
- Inter-patient validation: The same temporal and spatial configuration, along with the previously selected feature extraction method, is used with datasets containing 50 measurements per gesture. The three classification algorithms are evaluated based on the accuracy metric. This scenario assesses the model’s ability to generalize across different individuals.
3. Results
3.1. Global Performance Evaluation
3.2. Robustness Analysis
- (a)
-
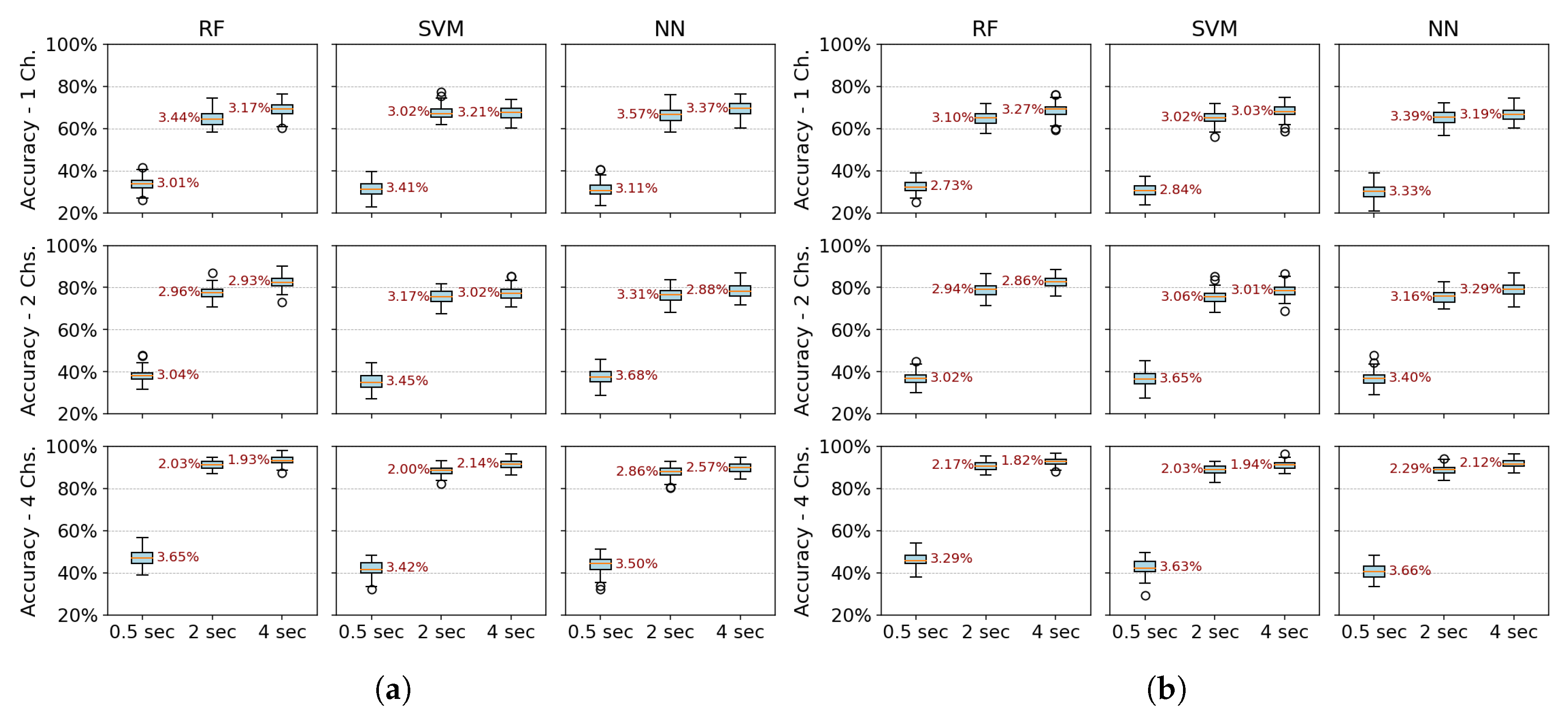
Single configuration robustness test: Table 3.1 shows the mean accuracy and standard deviation calculated to summarize performance. This metric was evaluated over 40 randomly partitioned datasets, with experiments conducted using two channels and a two-second window. All algorithms (RF, SVM, and NN) and feature extraction methods (PSD, PSD+PCA, PSD+SVD, DWT+PCA, and DWT+SVD) were tested.The coefficient of variation (CV) is used to evaluate the robustness of the models, comparing the standard deviation to the mean. Most methods show a low CV, typically below 5%, with PSD methods demonstrating consistent performance. However, methods like DWT+SVD and DWT+PCA exhibit higher variability, with CVs between 5 - 10% across all algorithms. Notably, the NN algorithm has the highest CV across all feature extraction methods, indicating greater sensitivity to data variability. Dimensionality reduction techniques like PCA improve robustness, as seen in the lower CVs for PSD+PCA (2.9%–3.5%), while DWT methods show higher CVs, especially DWT+SVD (6.1%–10.5%), indicating more variability in performance.
- (b)
-
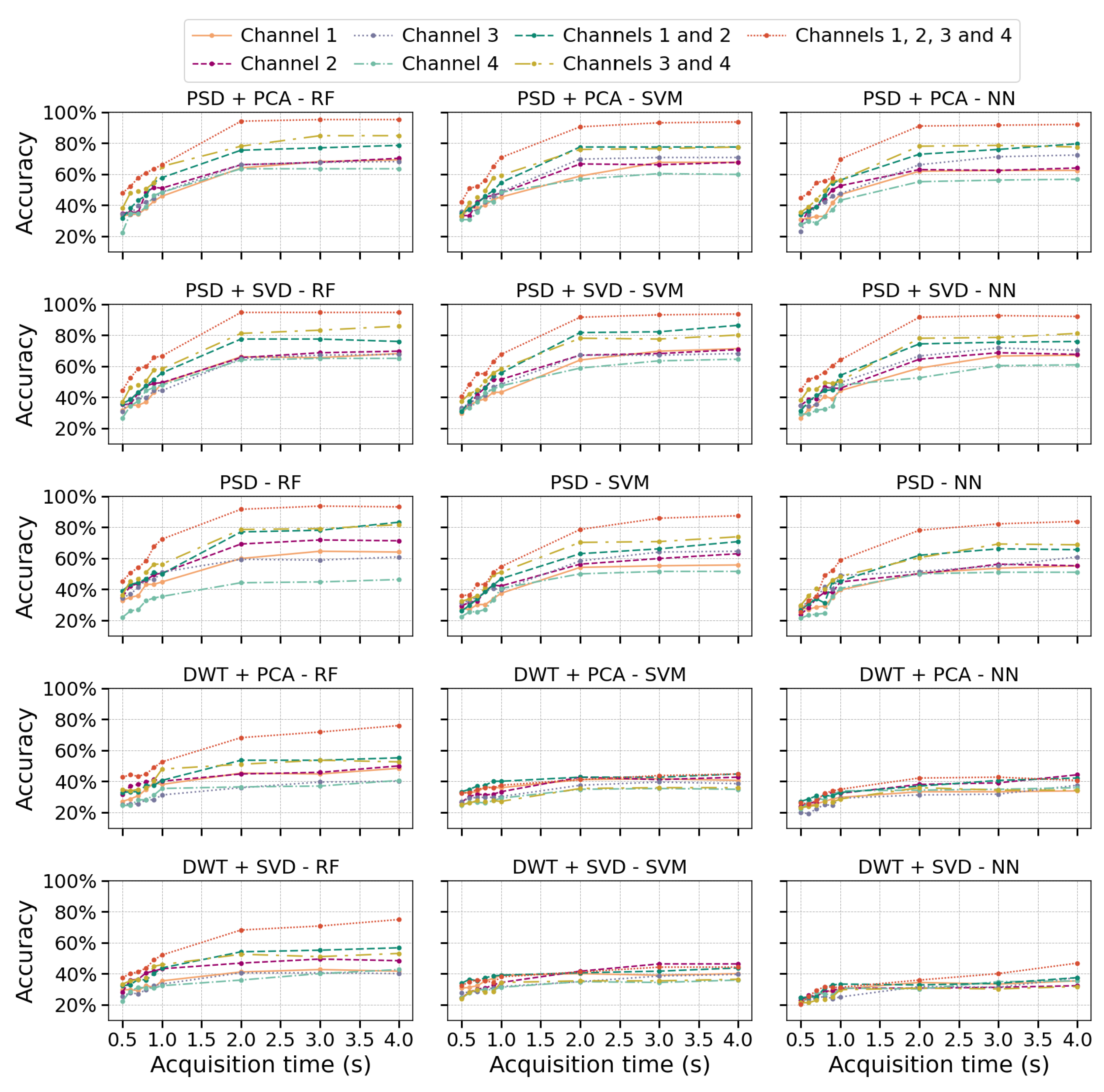
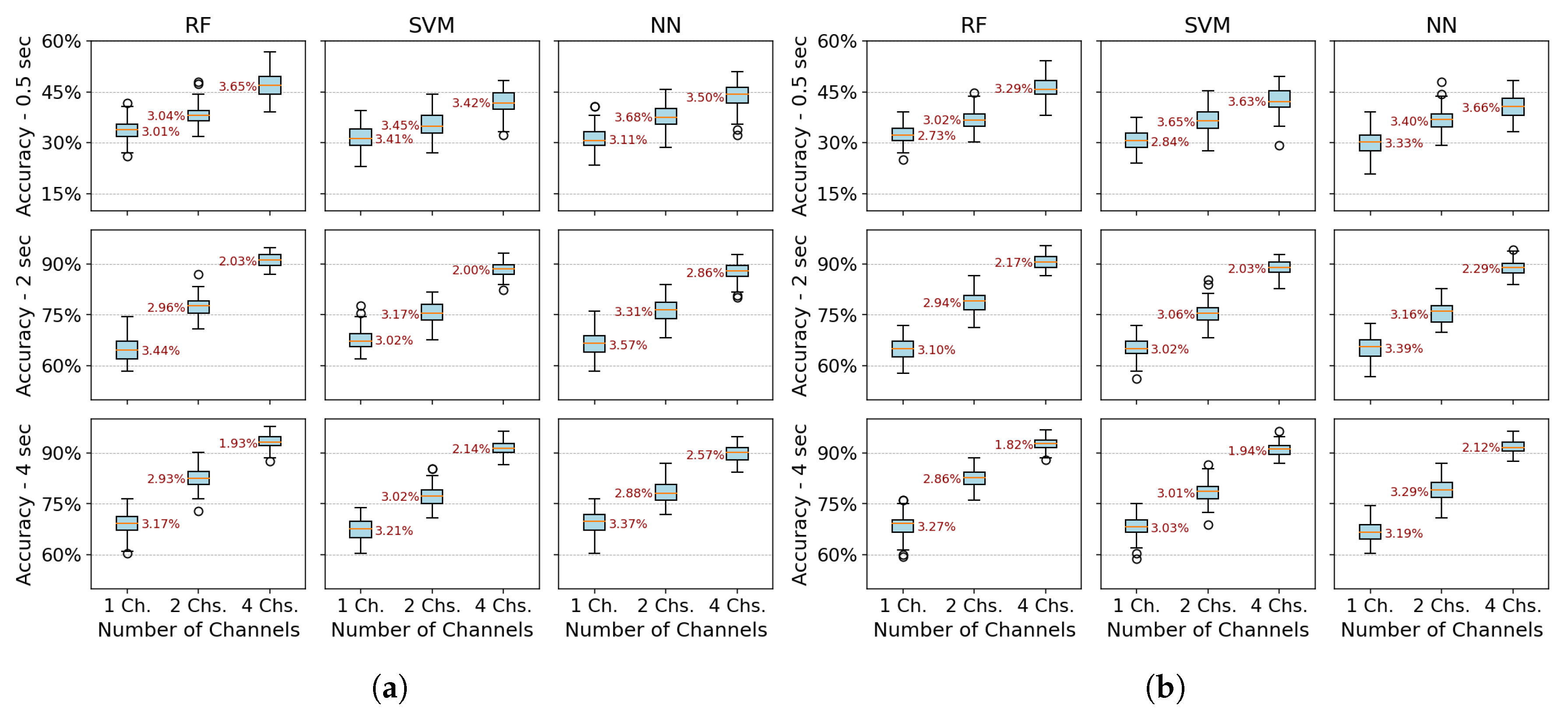
Multiple configuration robustness test: Figure 3 and Figure 4 present boxplots of accuracy for three spatial configurations (single channel, 2 channels, and 4 channels) and three temporal window configurations (0.5, 2, and 4 seconds of acquisition time). For the single-channel setup, channel 3 (extensor carpi radialis) is used, as it demonstrates the best individual performance. In the two-channel configuration, channels 3 and 4 (extensor and flexor carpi radialis muscles, respectively) are employed. The four-channel setup combines channels 3 and 4 (extensor and flexor carpi radialis) with channels 1 and 2 (extensor and flexor carpi ulnaris muscles). The robustness of the models was evaluated using 100 randomly partitioned datasets. The analysis focuses on feature extraction methods that showed the best performance, namely PSD+PCA and PSD+SVD—across all algorithms (RF, SVM, and NN).Figure 3 more clearly demonstrates that accuracy does not improve beyond 2 seconds of acquisition time. Furthermore, the data dispersion—reflected in narrow interquartile ranges—and the standard deviation remain stable after this point, showing no significant variations. With four channels, all algorithms show robust performance (low variability - CV: 2–3%). RF marginally outperforms SVM/NN in two-channel configurations. Single-channel data further amplifies variability, underscoring the impact of spatiotemporal limitations on model stability.Figure 4 reveals a critical interplay between temporal and spatial information in EMG classification. For short acquisition windows (0.5 s), increasing the number of channels from 1 to 4 yields only marginal accuracy gains (∼10%), suggesting that additional data provided by more channels is insufficient to compensate for the lack of relevant temporal dynamics in the signal. Conversely, with longer windows (2–4 s), channel count becomes decisive, boosting accuracy by up to 22% as more dynamic signal features are captured. Notably, shorter windows also exhibit higher data dispersion (wider IQRs), while longer acquisitions coupled with more channels reduce variability, reflecting improved robustness.The comparative analysis reveals temporal acquisition as the dominant factor (Figure 3 and Figure 4). A single channel achieves ∼30% accuracy gain when the temporal window increases from 0.5s to 2s, surpassing any multi-channel advantage at shorter intervals. This underscores that dynamic temporal features are fundamental for discrimination, while channels primarily refine results once sufficient temporal information is attained.
- (c)
-
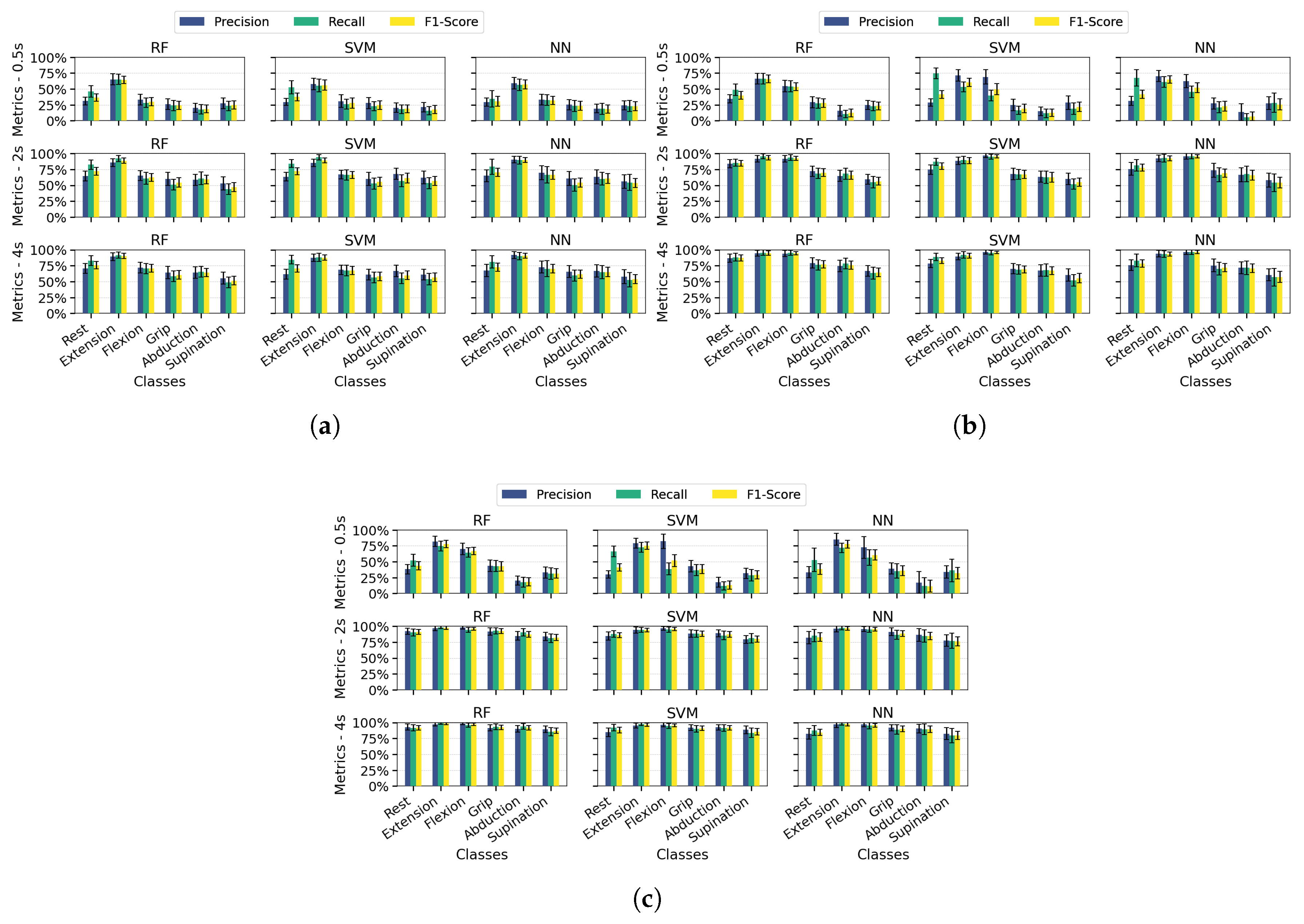
Classification performance by type of motion:Figure 5 presents each class’s average precision, recall, and F1-score values in bar plot format, with error bars indicating the standard deviation across 100 dataset splits. Results are shown for the PSD-PCA method (with PSD-SVD exhibiting similar patterns) under the previously defined temporal windows and channel configurations.Extending the analysis to class-specific performance, wrist extension consistently outperformed other classes across all channel configurations (highest precision/recall/F1), attributable to the distinctive EMG signatures from extensor carpi radialis (Channel 3). While multi-channel configurations improved overall metric consistency, three limitations became apparent: (1) supination exhibited marginally lower performance than other classes even with optimal conditions (≥ 2s windows and multi-channel spatial resolution), (2) finger abduction demonstrated degraded classification in short windows (0.5s) - likely from suboptimal electrode positioning relative to intrinsic hand muscles, and (3) the rest class showed biased performance in SVM/NN (high recall but low precision) due to systematic confusion between inactive states and low-intensity movements. These findings indicate that while proximal muscle movements benefit from channel redundancy, reliable distal movement classification requires both anatomical electrode optimization and appropriate temporal windows.Across most configurations, standard deviations remained relatively stable, indicating consistent performance throughout the evaluation iterations. However, higher deviations occurred in scenarios with unbalanced metrics, mainly when temporal information was limited. Supination demonstrated slightly more significant standard deviations than other classes, suggesting greater variability in movement execution patterns. Among the algorithms, NN showed the least robustness, with higher deviation values associated with individual class performance metrics.
3.3. Global Performance Evaluation for Stroke Patients Data
3.4. Model Generalization Capacity
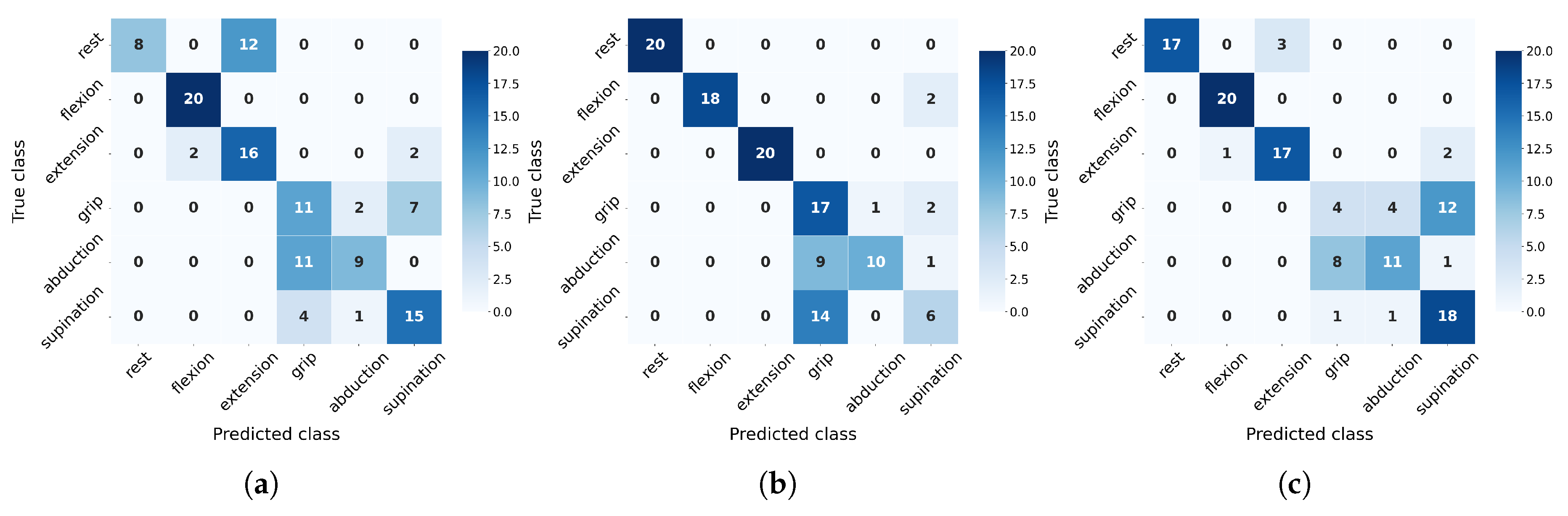
3.5. Evaluation of Cross-Patient Generalization
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EMG | Electromyography |
| TD | Time Domain |
| FD | Frequency Domain |
| TFD | Time-Frequency Domain |
| DWT | Discrete Wavelet Transform |
| PCA | Principal Component Analysis |
| PSD | Power Spectral Density |
| SVD | Singular Value Decomposition |
| RF | Random Forest |
| SVM | Support Vector Machine |
| NN | Neural Network |
| LDA | Linear Discriminant Analysis |
| kNN | k-Nearest Neighbors |
| ANN | Artificial Neural Network |
| MLP | Multi-Layer Perceptron |
Appendix A Hyperparameters
Appendix A.1. Hyperparameters Healthy Individuals
- -
-
RF model, across all feature extraction methods, demonstrates commonalities and distinctions in hyperparameter behavior. One significant observation is the absence of consistent trends in the selection of the partitioning criterion. Both ’gini’ and ’entropy’ are chosen interchangeably, with no evident preference, suggesting that the optimal criterion is largely influenced by the specific dataset and configuration rather than the feature extraction method employed.Regarding the number of estimators, no strong correlation was found with the window duration for any method. However, a recurring trend emerges when examining the influence of channel combinations. Configurations utilizing multiple channels often required a larger number of estimators, likely due to the increased variability and complexity of the feature space introduced by combining signals from different channels.In the vast majority of cases, regardless of the feature extraction method, the number of estimators typically ranged between 100 and 250. This medium-to-high range of trees indicates that RF models generally benefit from larger forests, which can better capture the variability and complexity of the EMG signals across different configurations.
- -
-
SVM models exhibit distinct trends in hyperparameter selection across the various feature extraction methods. For the kernel, rbf predominates across all methods, with occasional use of the poly kernel in PSD–PCA and PSD–SVD. When poly is selected, the degree is typically set to 3 or 5.For the regularization parameter C, values vary depending on the feature extraction method. In PSD–PCA and PSD–SVD, C ranges from approximately 100 to 8000, reflecting lower regularization and a stronger penalization of classification errors. WT–PCA and WT–SVD favor smaller values, typically between 1 and 400, indicating higher regularization and a greater tolerance for misclassifications to maintain simpler decision boundaries. PSD without dimensionality reduction shows a preference for values in the range of 1 to 100, suggesting that raw PSD features generally require higher regularization with lower penalization of errors.For the gamma parameter, PSD–PCA and PSD–SVD typically use values between and . Higher gamma values are observed for channel 4, likely due to its signals exhibiting lower variability or less distinguishable patterns. These higher values create tighter kernel focus, enabling more detailed decision boundaries but increasing the risk of overfitting. A notable trend is that as the number of channels increases, gamma tends to decrease. The additional information from multiple channels provides redundancy and complementarity, allowing the model to generalize better with broader, less localized decision boundaries. In WT–PCA and WT–SVD, gamma values are considerably smaller, often concentrated around . For PSD without dimensionality reduction, most gamma values cluster between and .
- -
-
ANN models exhibit consistent trends in hyperparameter selection across the various feature extraction methods, with notable differences influenced by the data complexity and feature extraction approach. For the regularization parameter alpha, values are concentrated in similar ranges for most methods, typically between and . These ranges reflect varying levels of constraint to balance model complexity and overfitting, with PSD methods favoring more constraint compared to wavelet-transformed features.The network architecture, configurations with 2 or 3 hidden layers dominate across all methods, with frequent patterns such as (128,64,32),(128,64), and (128,128). Networks with a single large layer, such as (256,), are also observed but less frequently, often appearing in cases with simpler data structures or fewer channels. Increasing the number of channels or extending the window duration generally coincides with deeper or more neuron-dense architectures, though simpler configurations (e.g., (128,64)) persist in some multi-channel setups.The activation function relu is consistently predominant across all methods due to its efficiency and robustness in classification tasks. However, logistic and tanh occasionally appear, particularly in PSD-only methods and configurations with shorter windows or fewer channels. These alternatives favor in specific cases where the variability of the signal requires smoother or more gradual activation.The learning rate is generally concentrated in the range of to for all feature extraction methods, striking a balance between convergence speed and stability during training. Solver selection is largely uniform, with adam dominating across all configurations due to its adaptive nature and effectiveness in optimizing complex networks. Occasional use of sgd is observed in specific setups.
Appendix A.2. Hyperparameters Stroke Patients
- -
- For RF models, the optimal number of estimators varies between 98 and 197 across the training datasets, with a trend of using larger forests as the number of measurements decreases. This suggests that with less data, the model compensates by relying on more trees to achieve stable decision boundaries. The criterion alternates between gini and entropy, with no clear preference.
- -
- For SVM models, the kernel rbf consistently dominates across all configurations, highlighting its ability to handle the non-linear relationships inherent in EMG data. The regularization parameter C varies from approximately 20 to 61, with a slight tendency to increase as the number of measurements decreases, indicating reduced regularization to better capture the variability in smaller datasets. The gamma parameter ranges from 0.001 to 0.008, with smaller values typically appearing in datasets with fewer measurements, allowing for broader decision boundaries in the higher-dimensional feature space.
- -
- For ANN models, the activation function relu is consistently selected, with alpha values concentrated between and . For the architecture, networks with 2 or 3 layers dominate, with configurations such as (128,128), (128,64), and (128,64,32). The networks tend to become slightly deeper or denser as the number of measurements increases, suggesting that larger datasets allow for more complex models. The learning rate falls within the range of 0.02 to 0.05, balancing convergence speed and stability.
References
- Su, D.; Hu, Z.; Wu, J.; Shang, P.; Luo, Z. Review of adaptive control for stroke lower limb exoskeleton rehabilitation robot based on motion intention recognition. Frontiers in Neurorobotics 2023, 17. [Google Scholar] [CrossRef] [PubMed]
- Young, A.J.; Smith, L.H.; Rouse, E.J.; Hargrove, L.J. A comparison of the real-time controllability of pattern recognition to conventional myoelectric control for discrete and simultaneous movements. Journal of NeuroEngineering and Rehabilitation 2014, 11, 5. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Grefa, W.P.; Portilla-Paguay, G.V.; Ortiz-Cartagena, C.L.; Roman-Huera, C.K. Integration of exoskeletons in the recovery of patients with motor disabilities: towards a new era in physiotherapy. Journal of Economic and Social Science Research 2024, 4, 77–98. [Google Scholar] [CrossRef]
- Fang, C.; He, B.; Wang, Y.; Cao, J.; Gao, S. EMG-Centered Multisensory Based Technologies for Pattern Recognition in Rehabilitation: State of the Art and challenges. Biosensors 2020, 10, 85. [Google Scholar] [CrossRef]
- Phinyomark, A.; Quaine, F.; Charbonnier, S.; Serviere, C.; Tarpin-Bernard, F.; Laurillau, Y. EMG feature evaluation for improving myoelectric pattern recognition robustness. Expert Systems With Applications 2013, 40, 4832–4840. [Google Scholar] [CrossRef]
- Parajuli, N.; Sreenivasan, N.; Bifulco, P.; Cesarelli, M.; Savino, S.; Niola, V.; Esposito, D.; Hamilton, T.J.; Naik, G.R.; Gunawardana, U.; Gargiulo, G.D. Real-Time EMG based Pattern recognition control for hand prostheses: A review on existing methods, challenges and future implementation. Sensors 2019, 19, 4596. [Google Scholar] [CrossRef]
- Merzoug, B.; Ouslim, M.; Mostefai, L.; Benouis, M. Evaluation of Dimensionality Reduction Using PCA on EMG-Based Signal Pattern Classification. Engineering Proceedings 2022, 14, 23. [Google Scholar]
- Nazmi, N.; Rahman, M.A.; Yamamoto, S.; Ahmad, S.; Malarvili, M.; Mazlan, S.; Zamzuri, H. Assessment on Stationarity of EMG Signals with Different Windows Size During Isotonic Contractions. Applied Sciences 2017, 7, 1050. [Google Scholar] [CrossRef]
- Al-Faiz, M.Z.; Miry, A.H. Artificial human arm driven by EMG signal. International Journal of Computer Applications 2012, 47, 1–5. [Google Scholar]
- Phinyomark, A.; Phukpattaranont, P.; Limsakul, C. A review of classification techniques of EMG signals during fatigue conditions. Sensors 2018, 16. [Google Scholar]
- Gokgoz, E.; Subasi, A. Comparison of decision tree algorithms for EMG signal classification using DWT. Biomedical Signal Processing and Control 2015, 18, 138–144. [Google Scholar] [CrossRef]
- Zhao, X.; Lu, Z.; Liu, L. Classification of finger movements using EMG signals based on Random Forest. IEEE Transactions on Neural Systems and Rehabilitation Engineering 2020, 28, 1234–1242. [Google Scholar]
- Dhanjal, J.S.; Amhia, H. A review article of EMG signal classification and feature extraction using machine learning and DWT technique. International Journal of Scientific Research & Engineering Trends 2024, 10, 123–129. [Google Scholar]
- Zhang, H.; Li, J.; Wang, M. EMG-based gesture recognition using neural networks. Journal of Biomedical Signal Processing and Control 2023, 81, 104701. [Google Scholar]
- Lee, K. H.; Min, J. Y.; Byun, S. Electromyogram-Based classification of hand and finger gestures using artificial neural networks. Sensors 2021, 22, 225. [Google Scholar] [CrossRef]
- Palkowski, A.; Redlarski, G. Basic hand gestures classification based on surface electromyography. Computational and Mathematical Methods in Medicine 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Fu, J.; Xiong, L.; Song, X.; Yan, Z.; Xie, Y. Identification of finger movements from forearm surface EMG using an augmented probabilistic neural network. In Proceedings of the 2022 IEEE/SICE International Symposium on System Integration (SII); 547–552. [Google Scholar]
- Shi, W.; Lyu, Z.; Tang, S.; Chia, T.; Yang, C. A bionic hand controlled by hand gesture recognition based on surface EMG signals: A preliminary study. Journal of Applied Biomedicine 2017, 38, 126–135. [Google Scholar] [CrossRef]
- Sharma, S.; Gupta, R. On the Use of Temporal and Spectral Central Moments of Forearm Surface EMG for Finger Gesture Classification. In Proceedings of the 2018 2nd International Conference on Micro-Electronics and Telecommunication Engineering (ICMETE), Ghaziabad, India; 2018; pp. 234–239. [Google Scholar]
- Qi, J.; Jiang, G.; Li, G.; Sun, Y.; Tao, B. Surface EMG hand gesture recognition system based on PCA and GRNN. Neural Computing and Applications 2019, 32, 6343–6351. [Google Scholar] [CrossRef]
- Arteaga, M. V.; Castiblanco, J. C.; Mondragon, I. F.; Colorado, J. D.; Alvarado-Rojas, C. EMG-driven hand model based on the classification of individual finger movements. Biomedical Signal Processing and Control 2020, 58, 101834. [Google Scholar] [CrossRef]
- Fajardo, J. M.; Gomez, O.; Prieto, F. EMG hand gesture classification using handcrafted and deep features. Biomedical Signal Processing and Control 2020, 63, 102210. [Google Scholar] [CrossRef]
- Hahne, J. M.; Graimann, B.; Muller, K. Spatial filtering for robust myoelectric control. IEEE Transactions on Biomedical Engineering 2012, 59, 1436–1443. [Google Scholar] [CrossRef] [PubMed]
- Khushaba, R. N.; Takruri, M.; Miro, J. V.; Kodagoda, S. Towards limb position invariant myoelectric pattern recognition using time-dependent spectral features. Neural Networks 2014, 55, 42–58. [Google Scholar] [CrossRef] [PubMed]
- Palermo, F.; Cognolato, M.; Gijsberts, A.; Muller, H.; Caputo, B.; Atzori, M. Repeatability of grasp recognition for robotic hand prosthesis control based on sEMG data. In Proceedings of the 2017 IEEE International Conference on Rehabilitation Robotics (ICORR); 2017. [Google Scholar]
- Du, Y.; Jin, W.; Wei, W.; Hu, Y.; Geng, W. Surface EMG-Based Inter-Session gesture recognition enhanced by deep domain adaptation. Sensors 2017, 17, 458. [Google Scholar] [CrossRef] [PubMed]
- Fang, Y.; Zhou, D.; Li, K.; Liu, H. ISRMyo-I: A database for sEMG-based hand gesture recognition. IEEE Dataport 2018.
- Kaczmarek, P.; Mańkowski, T.; Tomczyński, J. PUTEMG—A surface Electromyography Hand Gesture Recognition Dataset. Sensors 2019, 19, 3548. [Google Scholar] [CrossRef]
- Cognolato, M.; Gijsberts, A.; Gregori, V.; Saetta, G.; Giacomino, K.; Hager, A. M.; Gigli, A.; Faccio, D.; Tiengo, C.; Bassetto, F.; Caputo, B.; Brugger, P.; Atzori, M.; Müller, H. Gaze, visual, myoelectric, and inertial data of grasps for intelligent prosthetics. Scientific Data 2020, 7, 1. [Google Scholar] [CrossRef]
- Cote-Allard, U.; Gagnon-Turcotte, G.; Phinyomark, A.; Glette, K.; Scheme, E.; Laviolette, F.; Gosselin, B. A transferable adaptive domain adversarial neural network for virtual reality augmented EMG-Based gesture recognition. IEEE Transactions on Neural Systems and Rehabilitation Engineering 2021, 29, 546–555. [Google Scholar] [CrossRef]
- Pradhan, A.; He, J.; Jiang, N. Gesture recognition and biometrics electromyography (GRABMyo) dataset. 2022.
- Chen, X.; Yang, H.; Zhang, D.; Hu, X.; Xie, P. Hand gesture recognition based on High-Density myoelectricity in forearm flexors in humans. Sensors 2024, 24, 3970. [Google Scholar] [CrossRef]
- Ozdemir, M. A.; Kisa, D. H.; Guren, O.; Akan, A. Dataset for multi-channel surface electromyography (sEMG) signals of hand gestures. Data in Brief 2022, 41, 107921. [Google Scholar] [CrossRef]
- Kyranou, I.; Szymaniak, K.; Nazarpour, K. EMG Dataset for Gesture Recognition with Arm Translation. Scientific Data 2025, 12, 1. [Google Scholar] [CrossRef]
- Jahromi, M.G.; Parsaei, H.; Zamani, A.; Dehbozorgi, M. Comparative Analysis of Wavelet-Based Feature Extraction for Intramuscular EMG Signal Decomposition. J. Med. Signals Sens. 2017, 7, 365–378. [Google Scholar]
- Chowdhury, R.H.; Reaz, M.B.I.; Ali, M.A.B.; Bakar, A.A.A.; Chellappan, K.; Chang, T.G. Surface Electromyography Signal Processing and Classification Techniques. Sensors 2013, 13, 12431–12466. [Google Scholar] [CrossRef] [PubMed]
- Tallapragada, N.D.; Sagare, N.V. Multichannel EMG-Based Gesture Recognition Utilizing Advanced Machine Learning Techniques: A Random Forest Classifier for High-Precision Signal Classification. World Journal of Advanced Research and Reviews 2024, 24, 323–332. [Google Scholar] [CrossRef]
- Subasi, A. Classification of EMG Signals Using PSO Optimized SVM for Diagnosis of Neuromuscular Disorders. Computers in Biology and Medicine 2013, 43, 576–586. [Google Scholar] [CrossRef] [PubMed]
- Kehri, V.; N, A. R. EMG Signal Analysis for Diagnosis of Muscular Dystrophy Using Wavelet Transform, SVM and ANN. Biomedical & Pharmacology Journal 2018, 11, 1583–1591. [Google Scholar]
- Liu, X.; Zhang, Y. Hyperparameter optimization of neural networks for EMG classification. Artificial Intelligence in Medicine 2024, 123, 101819. [Google Scholar]

| Kernel | {rbf, poly} |
| Gamma | logspace(-5,0,50) |
| C | logspace(-1,4,80) |
| Degree | {3, 4, 5} |
| Hidden layers | (64) , (128), (256), (64,64), (128,128), | |||
| (256,128), (128,64), (128,64,32) | ||||
| Function activation | {tanh, relu, logistic} | |||
| Solver | {adam, sgd} | |||
| PSD | PSD + PCA | PSD + SVD | WT + PCA | WT + SVD | ||
| Mean accuracy ± Std |
RF | 78.5% ± 2.9% | 78.4% ± 2.7% | 80.1% ± 2.9% | 47.7% ± 2.3% | 49.0% ± 3.0% |
| SVM | 64.1% ± 3.4% | 75.8% ± 2.9% | 78.8% ± 3.1% | 43.4% ± 2.5% | 38.1% ± 3.3% | |
| NN | 61.1% ± 3.7% | 76.2% ± 3.1% | 77.7% ± 3.1% | 32.3% ± 2.9% | 31.4% ± 3.3% |
| Accuracy training | Accuracy validation | |
| RF | 67% | 66% |
| SVM | 83% | 76% |
| NN | 79% | 73% |
| Accuracy training | Accuracy validation | |
| RF | 89% | 80% |
| SVM | 83% | 78% |
| NN | 81% | 81% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




