Submitted:
02 May 2025
Posted:
06 May 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results and Discussion
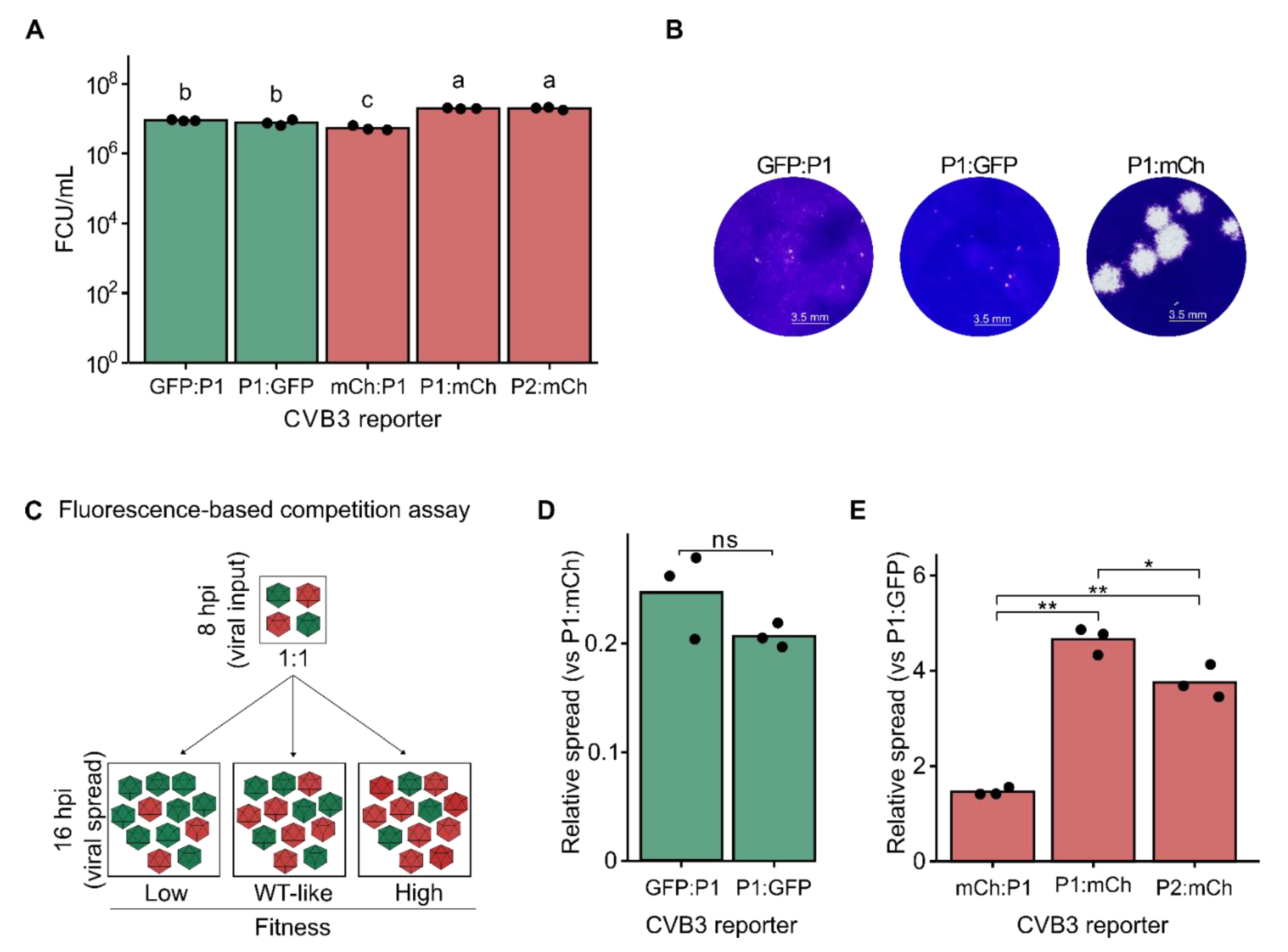
2.1. Insertion Site and Reporter Sequence Determine CVB3 Viability and Fitness
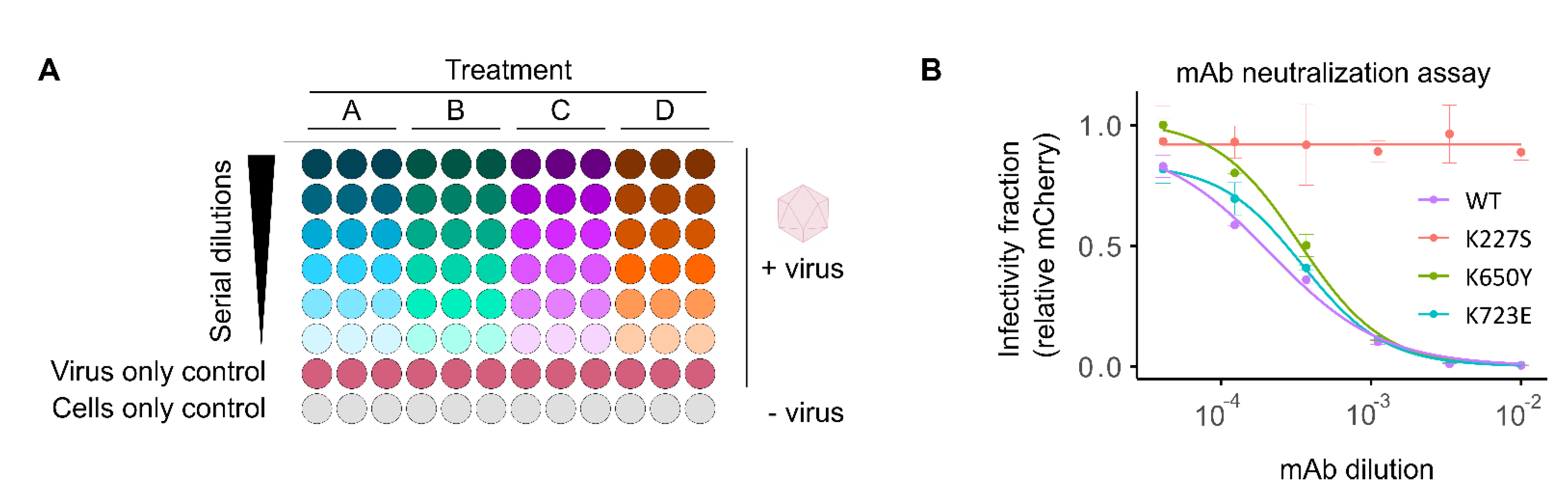
2.2. High-Throughput Screening of Antibody Neutralization
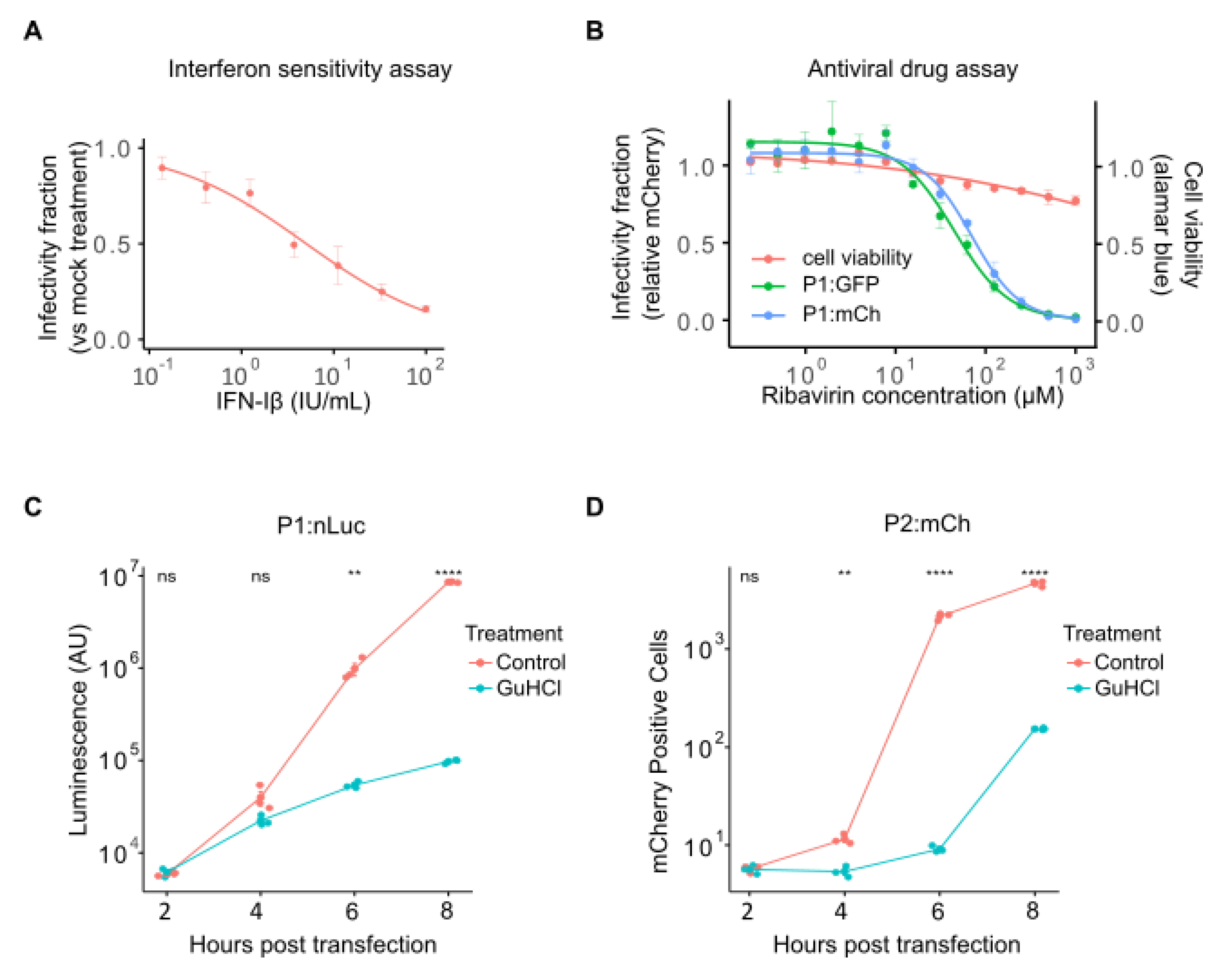
2.3. Analysis of Drug-Response Curves and Mechanism of Action Studies Using Fluorescent Reporter Viruses
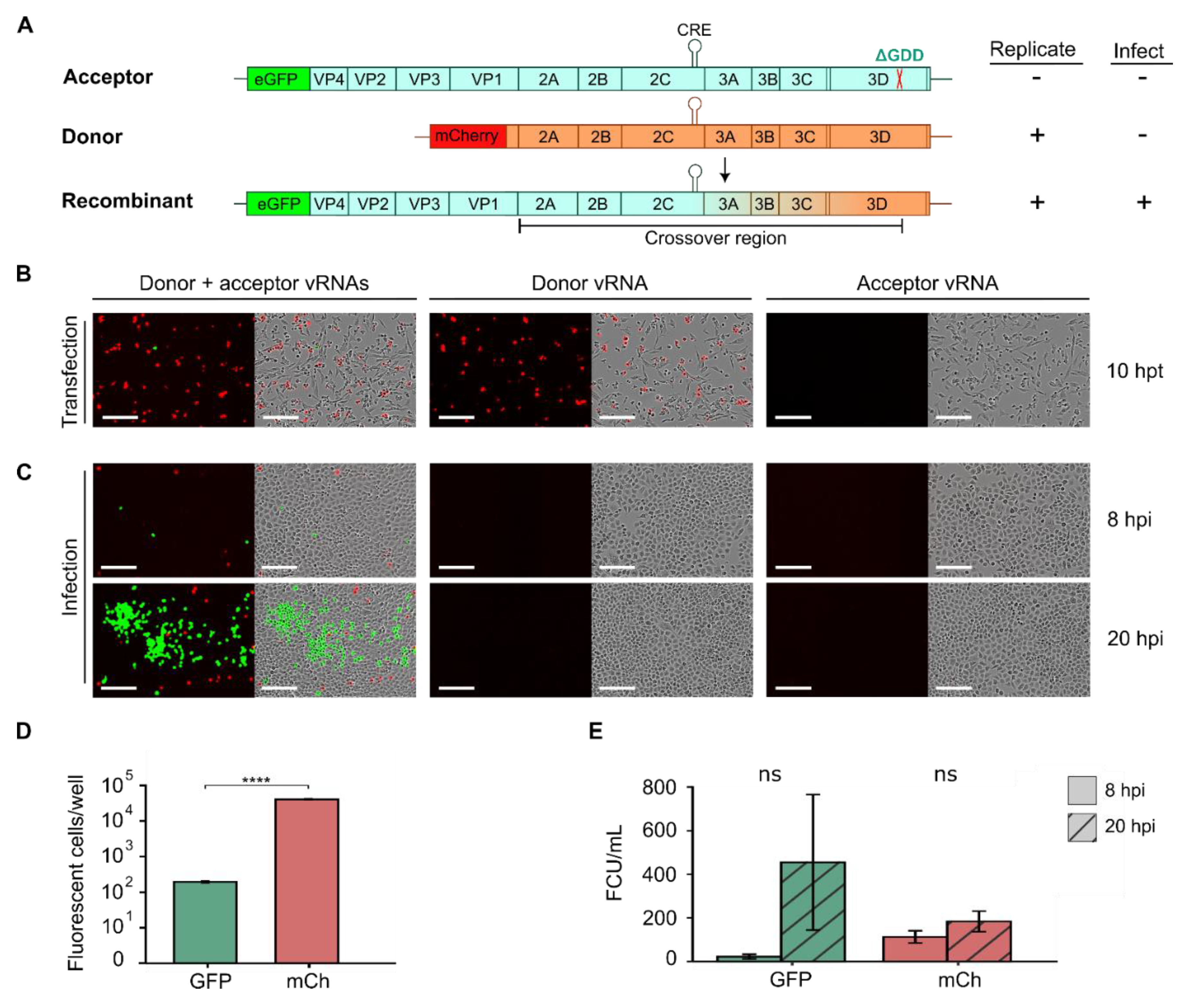
2.4. Tracking RNA Recombination Using Fluorescent Reporter Viruses
3. Methods
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Racaniello, V.R. Picornaviridae: The Viruses and Their Replication. In Fields Virology; Knipe, M.D., Howley, M.P., Eds.; Philadelphia : Wolters Kluwer Health/Lippincott Williams & Wilkins, c2013, 2013; ISBN 9781451105636. [Google Scholar]
- van der Linden, L.; Wolthers, K.C.; van Kuppeveld, F.J.M. Replication and Inhibitors of Enteroviruses and Parechoviruses. Viruses 2015, 7, 4529–4562. [Google Scholar] [CrossRef] [PubMed]
- Bastea, L.I.; Liu, X.; Fleming, A.K.; Pandey, V.; Döppler, H.; Edenfield, B.H.; Krishna, M.; Zhang, L.; Thompson, E.A.; Grandgenett, P.M.; et al. Coxsackievirus and Adenovirus Receptor Expression Facilitates Enteroviral Infections to Drive the Development of Pancreatic Cancer. Nature Communications 2024 15:1 2024, 15, 1–11. [Google Scholar] [CrossRef]
- Isaacs, S.R.; Roy, A.; Dance, B.; Ward, E.J.; Foskett, D.B.; Maxwell, A.J.; Rawlinson, W.D.; Kim, K.W.; Craig, M.E. Enteroviruses and Risk of Islet Autoimmunity or Type 1 Diabetes: Systematic Review and Meta-Analysis of Controlled Observational Studies Detecting Viral Nucleic Acids and Proteins. Lancet Diabetes Endocrinol 2023, 11, 578–592. [Google Scholar] [CrossRef]
- Mondal, S.; Sarvari, G.; Boehr, D.D. Picornavirus 3C Proteins Intervene in Host Cell Processes through Proteolysis and Interactions with RNA. Viruses 2023, 15, 2413. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, O.H.; Svedin, E.; Kapell, S.; Nurminen, A.; Hytönen, V.P.; Flodström-Tullberg, M. Enteroviral Proteases: Structure, Host Interactions and Pathogenicity. Rev Med Virol 2016, 26, 251–267. [Google Scholar] [CrossRef]
- Schipper, J.G.; Aloise, C.; Sutter, S.O.; Zwaagstra, M.; Vliet, A.L.W. van; Abdelnabi, R.; Ignacio, B.; Bonger, K.M.; Roelofs, D.; Brand, J.M.A. van den; et al. The Critical Role of Enterovirus 2A Protease in Viral Translation, Replication, and Antagonism of Host Antiviral Responses. bioRxiv, 6393; .37. [Google Scholar] [CrossRef]
- Yang, X.; Cheng, A.; Wang, M.; Jia, R.; Sun, K.; Pan, K.; Yang, Q.; Wu, Y.; Zhu, D.; Chen, S.; et al. Structures and Corresponding Functions of Five Types of Picornaviral 2A Proteins. Front Microbiol 2017, 8. [Google Scholar] [CrossRef]
- Andino, R.; Silvera, D.; Suggett, S.; Achacoso, P.; Miller, C.; Baltimore, D.; Feinberg, M. Engineering Poliovirus as a Vaccine Vector for the Expression of Diverse Antigens. Science (1979) 1994, 265, 1448–1451. [Google Scholar] [CrossRef]
- Ylä-Pelto, J.; Tripathi, L.; Susi, P. Therapeutic Use of Native and Recombinant Enteroviruses. Viruses 2016, 8, 57. [Google Scholar] [CrossRef] [PubMed]
- Jin, W.P.; Wang, C.; Wu, J.; Guo, J.; Meng, S.L.; Wang, Z.J.; Yu, D.G.; Shen, S. Reporter Coxsackievirus A5 Expressing ILOV Fluorescent Protein or Luciferase Used for Rapid Neutralizing Assay in Cells and Living Imaging in Mice. Viruses 2023, 15. [Google Scholar] [CrossRef]
- Shang, B.; Deng, C.; Ye, H.; Xu, W.; Yuan, Z.; Shi, P.Y.; Zhang, B. Development and Characterization of a Stable EGFP Enterovirus 71 for Antiviral Screening. Antiviral Res 2013, 97. [Google Scholar] [CrossRef]
- Liu, W.; Caglar, M.U.; Mao, Z.; Woodman, A.; Arnold, J.J.; Wilke, C.O.; Cameron, C.E. More than Efficacy Revealed by Single-Cell Analysis of Antiviral Therapeutics. Sci Adv 2019, 5. [Google Scholar] [CrossRef] [PubMed]
- Bakhache, W.; Shen, A.; Symonds-Orr, W.; Freeman, M.C.; Dolan, P.T. Novel Reporter Constructs to Accelerate Antiviral and Therapeutic Discovery for Enterovirus-A71. Antiviral Res 2025, 235, 106094. [Google Scholar] [CrossRef]
- van der Schaar, H.M.; Melia, C.E.; van Bruggen, J.A.C.; Strating, J.R.P.M.; van Geenen, M.E.D.; Koster, A.J.; Bárcena, M.; van Kuppeveld, F.J.M. Illuminating the Sites of Enterovirus Replication in Living Cells by Using a Split-GFP-Tagged Viral Protein. mSphere 2016, 1, e00104–16. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Rodríguez, B.; Buceta, J.; Geller, R. Comprehensive Profiling of Neutralizing Polyclonal Sera Targeting Coxsackievirus B3. Nat Commun 2023, 14, 6417. [Google Scholar] [CrossRef]
- Xiao, Y.; Dolan, P.T.; Goldstein, E.F.; Li, M.; Farkov, M.; Brodsky, L.; Andino, R. Poliovirus Intrahost Evolution Is Required to Overcome Tissue-Specific Innate Immune Responses. Nat Commun 2017, 8, 375. [Google Scholar] [CrossRef] [PubMed]
- Boersma, S.; Rabouw, H.H.; Bruurs, L.J.M.; Pavlovič, T.; van Vliet, A.L.W.; Beumer, J.; Clevers, H.; van Kuppeveld, F.J.M.; Tanenbaum, M.E. Translation and Replication Dynamics of Single RNA Viruses. Cell 2020, 183, 1930–1945.e23. [Google Scholar] [CrossRef]
- Bou, J.-V.; Geller, R.; Sanjuán, R. Membrane-Associated Enteroviruses Undergo Intercellular Transmission as Pools of Sibling Viral Genomes. Cell Rep 2019, 29, 714–723.e4. [Google Scholar] [CrossRef]
- Corbic Ramljak, I.; Stanger, J.; Real-Hohn, A.; Dreier, D.; Wimmer, L.; Redlberger-Fritz, M.; Fischl, W.; Klingel, K.; Mihovilovic, M.D.; Blaas, D.; et al. Cellular N-Myristoyltransferases Play a Crucial Picornavirus Genus-Specific Role in Viral Assembly, Virion Maturation, and Infectivity. PLoS Pathog 2018, 14, e1007203. [Google Scholar] [CrossRef]
- Álvarez-Rodríguez, B.; Velandia-Álvarez, S.; Toft, C.; Geller, R. Mapping Mutational Fitness Effects across the Coxsackievirus B3 Proteome Reveals Distinct Profiles of Mutation Tolerability. PLoS Biol 2024, 22, 1–25. [Google Scholar] [CrossRef]
- Bakhache, W.; Symonds-Orr, W.; McCormick, L.; Dolan, P.T. Deep Mutation, Insertion and Deletion Scanning across the Enterovirus A Proteome Reveals Constraints Shaping Viral Evolution. Nat Microbiol 2024, 10. [Google Scholar] [CrossRef]
- Chen, P.; Song, Z.; Qi, Y.; Feng, X.; Xu, N.; Sun, Y.; Wu, X.; Yao, X.; Mao, Q.; Li, X.; et al. Molecular Determinants of Enterovirus 71 Viral Entry. Journal of Biological Chemistry 2012, 287. [Google Scholar] [CrossRef] [PubMed]
- Carrasco, P.; Daròs, J.A.; Agudelo-Romero, P.; Elena, S.F. A Real-Time RT-PCR Assay for Quantifying the Fitness of Tobacco Etch Virus in Competition Experiments. J Virol Methods 2007. [Google Scholar] [CrossRef] [PubMed]
- Mattenberger, F.; Latorre, V.; Tirosh, O.; Stern, A.; Geller, R. Globally Defining the Effects of Mutations in a Picornavirus Capsid. Elife 2021, 10, e64256. [Google Scholar] [CrossRef]
- Geller, R.; Pechmann, S.; Acevedo, A.; Andino, R.; Frydman, J. Hsp90 Shapes Protein and RNA Evolution to Balance Trade-Offs between Protein Stability and Aggregation. Nat Commun 2018, 9, 1781. [Google Scholar] [CrossRef] [PubMed]
- Álvarez-Rodríguez, B.; Velandia-Álvarez, S.; Toft, C.; Geller, R. Mapping Mutational Fitness Effects across the Coxsackievirus B3 Proteome Reveals Distinct Profiles of Mutation Tolerability. PLoS Biol 2024, 22, e3002709. [Google Scholar] [CrossRef]
- Stadnick, E.; Dan, M.; Sadeghi, A.; Chantler, J.K. Attenuating Mutations in Coxsackievirus B3 Map to a Conformational Epitope That Comprises the Puff Region of VP2 and the Knob of VP3. J Virol 2004, 78, 13987–14002. [Google Scholar] [CrossRef]
- Crotty, S.; Maag, D.; Arnold, J.J.; Zhong, W.; Lau, J.Y.N.; Hong, Z.; Andino, R.; Cameron, C.E. The Broad-Spectrum Antiviral Ribonucleoside Ribavirin Is an RNA Virus Mutagen. Nat Med 2000, 6. [Google Scholar] [CrossRef]
- Geller, R.; Andino, R.; Frydman, J. Hsp90 Inhibitors Exhibit Resistance-Free Antiviral Activity against Respiratory Syncytial Virus. PLoS One 2013, 8, e56762. [Google Scholar] [CrossRef]
- Geller, R.; Vignuzzi, M.; Andino, R.; Frydman, J. Evolutionary Constraints on Chaperone-Mediated Folding Provide an Antiviral Approach Refractory to Development of Drug Resistance. Genes Dev 2007, 21, 195–205. [Google Scholar] [CrossRef]
- van der Linden, L.; Vives-Adrián, L.; Selisko, B.; Ferrer-Orta, C.; Liu, X.; Lanke, K.; Ulferts, R.; De Palma, A.M.; Tanchis, F.; Goris, N.; et al. The RNA Template Channel of the RNA-Dependent RNA Polymerase as a Target for Development of Antiviral Therapy of Multiple Genera within a Virus Family. PLoS Pathog 2015, 11, e1004733. [Google Scholar] [CrossRef]
- Xiao, Y.; Rouzine, I.M.; Bianco, S.; Farkov, M.; Brodsky, L.; Andino, R.; Xiao, Y.; Rouzine, I.M.; Bianco, S.; Acevedo, A.; et al. RNA Recombination Enhances Adaptability and Is Required for Virus Spread and Virulence. Cell Host Microbe 2016, 19, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Wang, H.; Shi, J.; Yang, D.; Zhou, G.; Chang, J.; Cameron, C.E.; Woodman, A.; Yu, L. Senecavirus-Specific Recombination Assays Reveal the Intimate Link between Polymerase Fidelity and RNA Recombination. J Virol 2019, 93. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Aponte-Diaz, D.; Sotoudegan, M.S.; Shengjuler, D.; Arnold, J.J.; Cameron, C.E. The Enterovirus Genome Can Be Translated in an IRES-Independent Manner That Requires the Initiation Factors EIF2A/EIF2D. PLoS Biol 2023, 21, e3001693. [Google Scholar] [CrossRef] [PubMed]
- Woodman, A.; Lee, K.-M.; Janissen, R.; Gong, Y.-N.; Dekker, N.H.; Shih, S.-R.; Cameron, C.E. Predicting Intraserotypic Recombination in Enterovirus 71. J Virol 2019, 93. [Google Scholar] [CrossRef]
- Woodman, A.; Arnold, J.J.; Cameron, C.E.; Evans, D.J. Biochemical and Genetic Analysis of the Role of the Viral Polymerase in Enterovirus Recombination. Nucleic Acids Res 2016, 44, 6883–6895. [Google Scholar] [CrossRef]
- Kempf, B.J.; Peersen, O.B.; Barton, D.J. Poliovirus Polymerase Leu420 Facilitates RNA Recombination and Ribavirin Resistance. J Virol 2016, 90, 8410–8421. [Google Scholar] [CrossRef]
- Lowry, K.; Woodman, A.; Cook, J.; Evans, D.J. Recombination in Enteroviruses Is a Biphasic Replicative Process Involving the Generation of Greater-than Genome Length “Imprecise” Intermediates. PLoS Pathog 2014, 10. [Google Scholar] [CrossRef]
- Hochstein, N.; Webb, D.; Hösel, M.; Seidel, W.; Auerochs, S.; Doerfler, W. Human CAR Gene Expression in Nonpermissive Hamster Cells Boosts Entry of Type 12 Adenovirions and Nuclear Import of Viral DNA. J Virol 2008, 82. [Google Scholar] [CrossRef]
- Zhang, H.-L.; Dong, H.-L.; Zhang, Y.-N.; Xu, L.-L.; Deng, C.-L.; Li, X.-F.; Li, X.-D.; Ye, H.-Q.; Yuan, Z.-M.; Qin, C.-F.; et al. Visualization of Chikungunya Virus Infection in Vitro and in Vivo. Emerg Microbes Infect 2019, 8, 1574–1583. [Google Scholar] [CrossRef]
- Revuelta, J.; Rusu, L.; Frances-Gomez, C.; Trapero, E.; Iglesias, S.; Pinilla, E.C.; Blázquez, A.B.; Gutiérrez-Adán, A.; Konuparamban, A.; Moreno, O.; et al. Synthetic Heparan Sulfate Mimics Based on Chitosan Derivatives Show Broad-Spectrum Antiviral Activity. Commun Biol 2025, 8. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



