Submitted:
29 April 2025
Posted:
30 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
- -
- Systemic delivery of current drugs is non-specific. The medications circulate throughout the body and do not concentrate specifically at sites of active bone loss. As a result, both healthy and diseased bone receive similar exposure. This lack of lesion specificity means that areas of high bone turnover (the most at-risk regions) are not selectively targeted by the therapy.
- -
- Many osteoporosis drugs, especially bisphosphonates like risedronate, have very low oral absorption. Risedronate, for example, has an oral bioavailability under. Most of the dose is not absorbed from the gastrointestinal tract, which necessitates higher dosing and contributes to variability in patient response. Strict dosing requirements (fasting, water intake, remaining upright) are imposed to maximize absorption and reduce esophageal irritation.
- -
- Due to their pharmacokinetics, bisphosphonates often require weekly or monthly dosing, and anabolic agents may require daily injections. Long-term adherence to these regimens is challenging for patients, and missed doses can reduce effectiveness. Oral bisphosphonates can cause gastrointestinal irritation if instructions are not followed, further complicating compliance.
- -
- Chronic bisphosphonate therapy is associated with side effects such as gastrointestinal (GI) irritation or ulceration of the esophagus and hypocalcemia.
- -
- In rare cases, osteonecrosis of the jaw (ONJ) and atypical femur fractures. RANKL inhibition (denosumab) can lead to suppressed bone remodeling to the extent that, upon discontinuation, there is a rebound increase in bone resorption and risk of vertebral fractures. These risks are exacerbated by the drugs’ lack of targeting, exposing the entire skeleton and other tissues to their effects.
1.1. Design
1.2. Polymer Core
1.3. Targeting Ligands
1.4. Stimuli Responsiveness
1.5. Delivery Method
- -
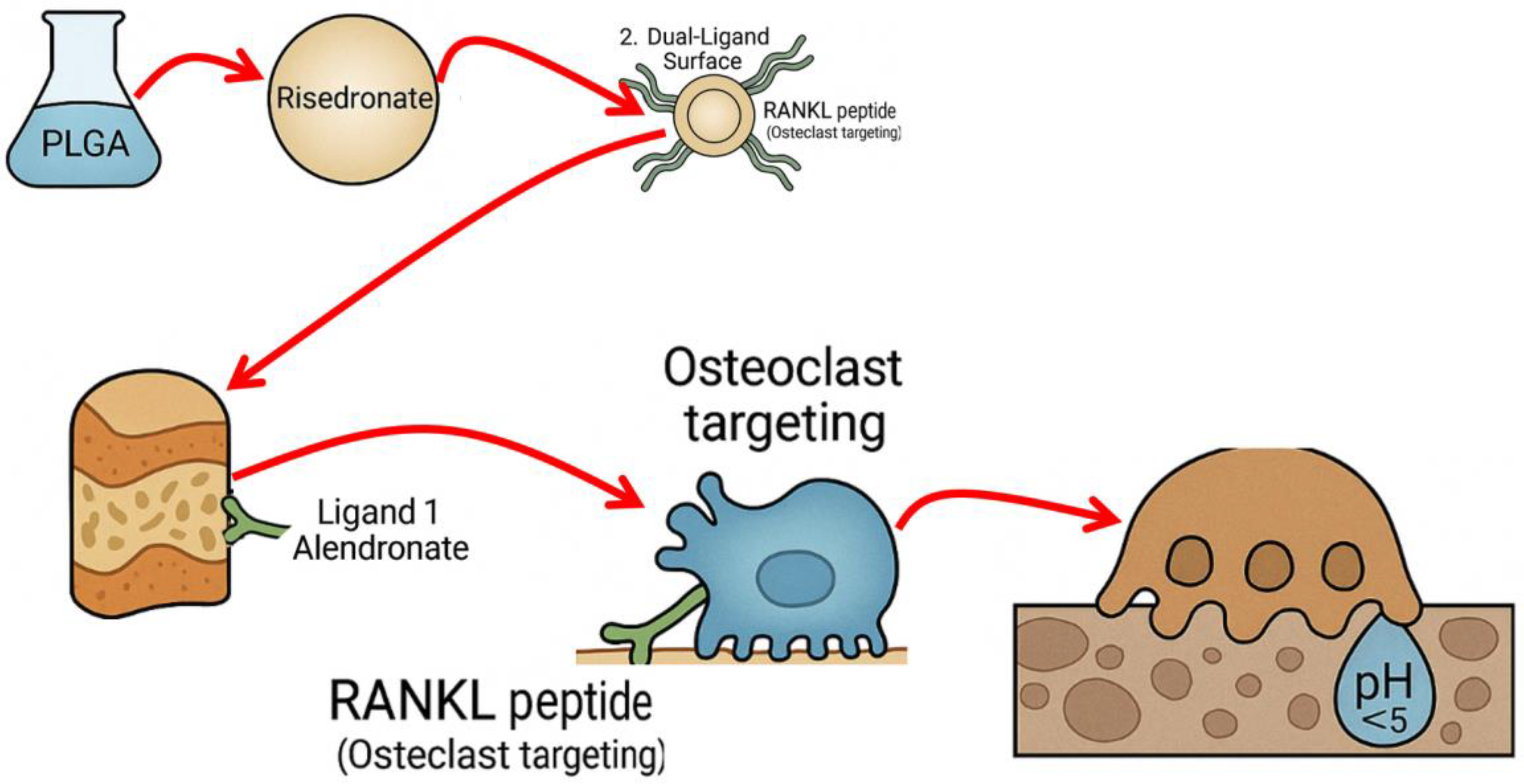
- Dual targeting: On bone surfaces in general (via a mineral-binding ligand) and osteoclast-rich lesions (via a cell-targeting ligand).
- -
- pH Triggered Release: For timing drug release at the right situation – when the nanoparticle senses the acidic conditions indicative of active bone resorption, it will release risedronate, ensuring the drug acts primarily at the site of disease.
- -
- Biocompatible, Oral Formulation: Using PLGA, PEG, and chitosan/polyhistidine yields a fully biocompatible NP. These materials are non-toxic and biodegradable; PLGA breaks down into lactic and glycolic acid (natural metabolites), PEG and chitosan are generally safe in the amounts used. By formulating as an oral dosage form, we aim to improve patient compliance (avoiding injections and possibly reducing dosing frequency).
- -
- Stability: PEGylation and careful surface engineering allows the NP to navigate the body’s defenses (stomach acid, digestive enzymes, immune recognition) to reach the target.
2. Synthesis and Formulation Methods
2.1. Materials
2.2. Preparation of Risedronate-Loaded PLGA Nanoparticles
2.3. Conjugation of Targeting Ligands to NP Surface
Ligand Density and Verification
2.4. pH-Responsive Polymer Coating
2.4.1. Poly(L-histidine) Grafting:
2.4.2. Chitosan Coating
2.5. Characterization of Nanoparticle Properties
Data Confirmation
3. Testing Plan
3.1. In Vitro Testing
Summary, Anticipated Outcomes
3.2. In Vivo Testing
3.2.1. Animal Model
- -
- Treatment Groups: Rats will be randomly divided (with ~8–10 rats per group for statistical power) into the following treatment groups:
- -
- Healthy control (sham + no treatment): to represent normal bone (not osteoporotic, no therapy).
- -
- OVX control (OVX + no treatment): osteoporotic, untreated, to measure the extent of bone loss and serve as a negative control for therapy.
- -
- Free Risedronate (OVX + free drug): osteoporotic rats treated with oral risedronate solution (or suspension) at a dose equivalent to a typical clinical dose scaled to rat size (for example, ~5 mg/kg/week, as a single weekly oral gavage).
- -
- Dual-Targeted pH-Responsive NP (OVX + targeted NP): osteoporotic rats treated with the oral nanoparticle formulation, containing the same equivalent dose of risedronate (5 mg/kg/week) in an enteric capsule or appropriate vehicle.
- -
- Nanoparticle control (OVX + blank NP): (optional) to isolate effects of the NP itself, a group receiving the same NP but without risedronate. This checks biocompatibility, but if animals are limited, we may omit this since PLGA NPs are known to be safe; however, including it is good for interpretation of any effects.
- NP with only bone-targeting ligand (no osteoclast ligand, but still pH-responsive)
- NP with dual targeting but non-pH-responsive (for example, without the pH coat, to see if uncontrolled release is less effective or more toxic)
3.2.2. Pharmacokinetics and Biodistribution Sub-Study
3.2.3. Bone Density and Microarchitecture Outcomes
3.2.4. Biochemical Markers of Bone Turnover
3.2.5. Histological Analysis
3.2.6. Specificity to Lesions
3.2.7. Safety and Off-Target Effects
3.2.8. Data Analysis
3.2.9. Summary, Anticipated Outcomes
4. Discussion
4.1. Future Directions
- Testing in larger animal models (e.g., osteoporotic non-human primates) to see how it works in a system closer to humans.
- Investigating combination therapy: e.g., can we incorporate an anabolic drug into a similar targeted NP (maybe dual delivery)? Or alternate with anabolic therapy in a smart way.
- Exploring other bone diseases: The platform could be adapted to deliver different drugs to bone. For example, in bone metastases or multiple myeloma (where lesions are lytic), a similar NP carrying anti-cancer drugs could target bone tumors. Or in osteomyelitis, carrying antibiotics to infected bones. The dual-target approach can be tweaked (change the second ligand to something that targets tumors or bacteria, etc.).
- Optimizing the oral delivery further, maybe by designing nanoparticles that exploit receptor-mediated transcytosis in the gut (targeting the neonatal Fc receptor or others).
- Cost-benefit analysis and ensuring that adding this technology justifies itself in outcomes, given that oral bisphosphonates are cheap. If our NP can drastically reduce fractures with fewer side effects, it justifies development cost.
4.2. Summary of Expected Impact
- -
- Higher efficacy in preventing bone loss and fractures due to focused drug action on osteoclasts.
- -
- Reduced side effects, as drug action is confined to bones, sparing the GI tract, kidneys, and other tissues from unnecessary exposure.
- -
- Better patient adherence to therapy because of an easier dosing regimen and fewer adverse events.
- -
- A new paradigm for treating not only osteoporosis but potentially other conditions by using multi-targeted nanomedicines that respond to disease microenvironments.
5. Conclusion
References
- C. Wen et al., “Bone Targeting Nanoparticles for the Treatment of Osteoporosis,” Int. J. Nanomedicine, vol. 19, pp. 1363–1383, 2024. [CrossRef]
- Y. Wu et al., “Bone-targeted nano-drug and nano-delivery,” Bone Research, vol. 12, art. 51, 2024. [CrossRef]
- Z. Saifi et al., “Enhancing osteoporosis treatment through targeted nanoparticle delivery of risedronate: In vivo evaluation and bioavailability enhancement,” Pharmaceutics, vol. 15, no. 9, p. 2339, 2023.
- P. Rawat et al., “Design and development of bioceramic based functionalized PLGA nanoparticles of risedronate for bone targeting: In-vitro characterization and pharmacodynamic evaluation,” Pharm. Res., vol. 32, pp. 3149–3158, 2015. [CrossRef]
- Y. Chen et al., “Bone-targeted nanoparticle drug delivery system: an emerging strategy for bone-related disease,” Front. Pharmacol., vol. 13, art. 909408, 2022. [CrossRef]
- M. Ren et al., “An oligopeptide/aptamer-conjugated dendrimer-based nanocarrier for dual-targeting delivery to bone,” J. Mater. Chem. B, vol. 9, no. 12, pp. 2831–2844, 2021. [CrossRef]
- J. Hoque et al., “Bone targeting nanocarrier-assisted delivery of adenosine to combat osteoporotic bone loss,” Biomaterials, vol. 273, p. 120819, 2021. [CrossRef]
- E. Vaculíková et al., “Preparation of risedronate nanoparticles by solvent evaporation technique,” Molecules, vol. 19, pp. 17848–17861, 2014.
- D. A. Ossipov, “Bisphosphonate-modified biomaterials for drug delivery and bone tissue engineering,” Expert Opin. Drug Deliv., vol. 12, pp. 1443–1458, 2015. [CrossRef]
- H. Sahana et al., “Improvement in bone properties by using risedronate adsorbed hydroxyapatite novel nanoparticle-based formulation in a rat model of osteoporosis,” J. Biomed. Nanotechnol., vol. 9, pp. 193–201, 2013. [CrossRef]
- D. K. Khajuria et al., “Risedronate/zinc-hydroxyapatite based nanomedicine for osteoporosis,” Mater. Sci. Eng. C, vol. 63, pp. 78–87, 2016.
- X. Pang et al., “pH-responsive polymer–drug conjugates: design and progress,” J. Control. Release, vol. 222, pp. 116–129, 2016.
- H. Hirabayashi et al., “Bone-specific delivery and sustained release of diclofenac via a bisphosphonic prodrug based on the osteotropic drug delivery system (ODDS),” J. Control. Release, vol. 70, pp. 183–191, 2001.
- X. Xing et al., “Bone-targeted delivery of senolytics to eliminate senescent cells increases bone formation in senile osteoporosis,” Acta Biomater., vol. 157, pp. 352–366, 2023. [CrossRef]
- A. Alhalmi et al., “Nanostructured lipid carrier-based codelivery of raloxifene and naringin: formulation, optimization, in vitro, ex vivo, in vivo assessment, and acute toxicity studies,” Pharmaceutics, vol. 14, no. 9, p. 1771, 2022.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



