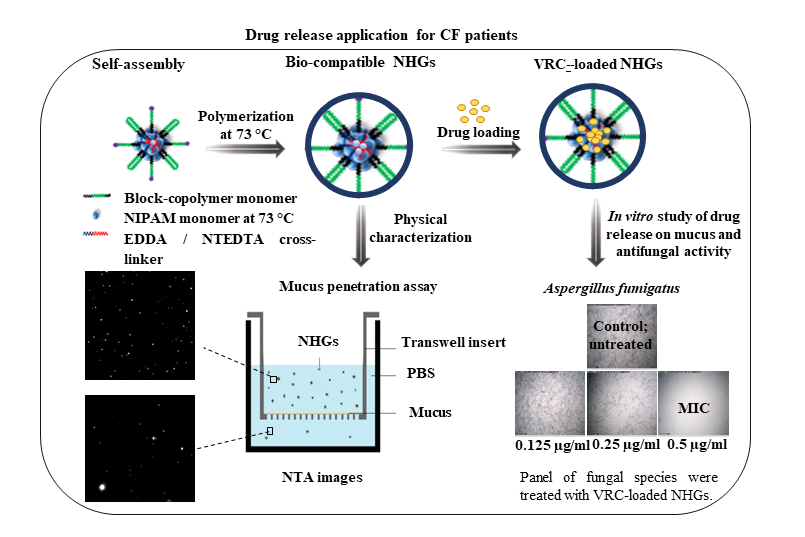
1. Introduction
Cystic fibrosis (CF) is the most common inherited life-limiting disease among Caucasian people affecting 90,000 people worldwide [
1]. This single-gene disease, with autosomal recessive inheritance is caused by a mutation in the CF gene encoding the transmembrane conductance regulator (CFTR) [
2], which forms a chloride channel that is critical to efficient mucus transport. Mutations in CFTR disrupt chloride secretion, sodium reabsorption, and water transport, leading to mucus hyper concentration and decreased mucociliary clearance [
3].
The resultant dehydrated form of mucus is characterized by a reduced mesh size (60−300 nm) [
4] in comparison to physiological mucus (497−503 nm) [
5]. Dehydrated mucus secretions lead to endobronchial infection with a narrow spectrum of distinctive bacteria and fungi and an exaggerated inflammatory response, resulting in development of severe bronchiectasis rather than fibrosis and, eventually, respiratory failure [
6]. Therefore, in mucus-related pathologies such as CF, an overexpression of mucins, accumulation of extracellular DNA as well as cellular debris, and the persistent presence of bacteria and fungi confer mucus stasis, leading to a vicious cycle of infection and inflammation that can be chronically sustained [
7,
8]. This pathological mucus strongly limits the absorption of antibiotics, antifungals and anti-inflammatory drugs, preventing them from reaching target sites [
9,
10,
11,
12,
13].
Despite great advancements in disease management in recent decades, pulmonary failure remains the main cause of morbidity and mortality in CF patients [
2]. Recurrent lung infections that lead to chronic airway inflammation and respiratory failure remain the major prognostic problem accounting for over 90% of mortality [
14]. Clinical research and therapy mostly focus on the role of bacterial species with little attention to the role of fungi in the pathogenesis of lung function decline, despite their frequent isolation in respiratory samples [
15].
Filamentous fungi, especially
Aspergillus,
Scedosporium and
Exophiala species, are frequent lung colonizers in CF patients [
16]. Chronic filamentous fungal colonization is particularly observed at a late stage of the disease and is associated with allergic bronchopulmonary mycosis and chronic infections related to a significant decline in lung function that persists over time [
16,
17,
18,
19,
20,
21,
22,
23]. Chronic antifungal therapy with azole drugs such as voriconazole (VRC) slows down this progression, mainly in patients with advanced disease [
18].
However, the use of azole antifungal agents, including VRC, are often limited by their poor solubility and bioavailability. Azoles are typically hydrophobic, resulting in low aqueous solubility, leading to challenges in formulation and reduced absorption in the gastrointestinal tract. This poor bioavailability necessitates higher doses to achieve therapeutic levels, increasing the risk of adverse effects and drug interactions [
24]. Overcoming these limitations is crucial for improving the efficacy and safety of azole antifungals in chronic therapy. In addition, the pathological CF mucus strongly limits their absorption and their ability to reach target sites in the lung (bronchi, alveoli).
More effective drug delivery to mucosal surfaces are thus needed. For this purpose, drugs and drug carriers must bypass the adhesive mucus barrier [
5]. Nanoparticles (NPs) smaller than the mucus mesh, densely coated with poly(ethylene glycol) (PEG), a muco-inert material, can rapidly diffuse through human mucus secretions [
25]
. These “mucus-penetrating particles (MPP)” demonstrated improved mucosal distribution, enhanced pharmacokinetics, and superior treatment efficacy in applications such as CF lung gene therapy [
26].
This study involves synthesis of series of NHGs based on our established methodology as a platform for loading VRC as a delivery system for treating broncho-pulmonary fungal infections in CF patients. As the mucus presents a diffusional barrier, the NHGs used here are composed of an outer shell of PEO (PEG),which was recently shown to cross lung-generated mucus [
25]. An in vitro model of pulmonary pathological mucus that mimics the chemical composition, structural features, and viscoelastic properties of CF mucus [
27] allowed to study their penetration across this diffusional barrier and to assess their activity against the three major molds affecting CF patients worldwide.
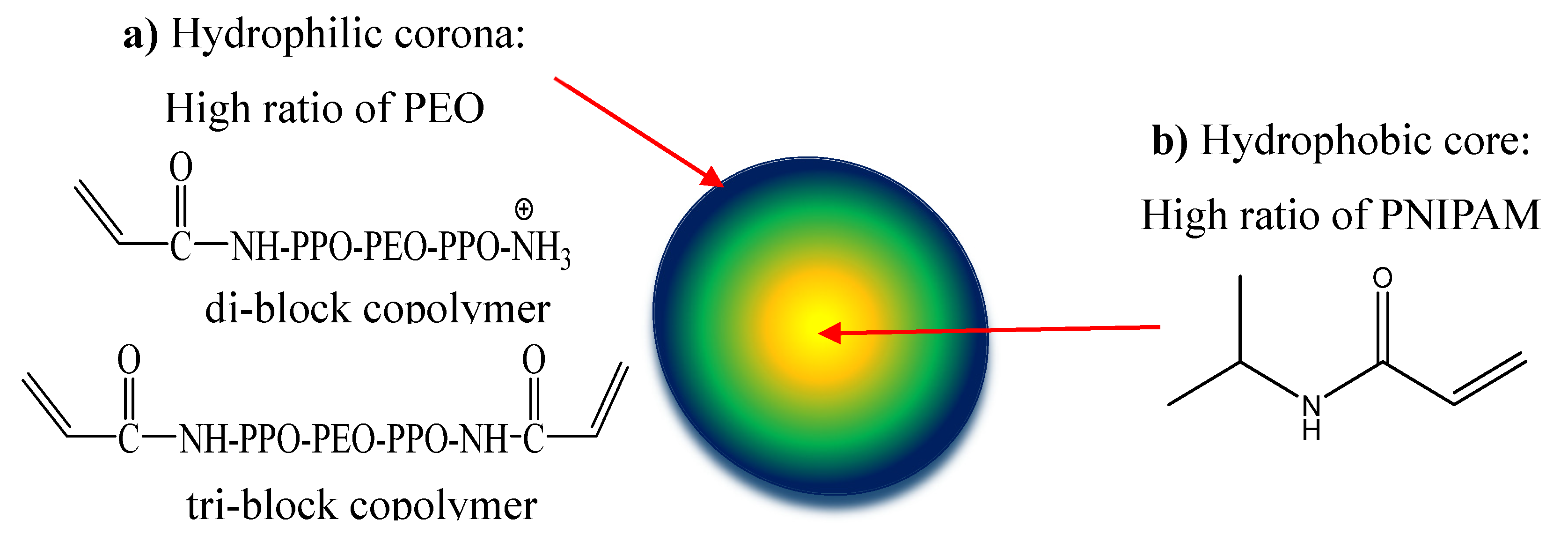
The biocompatible and biodegradable monodisperse NHG libraries, which range in size from 100 to 400 nm, are disclosed in
Figure 1. NHGs are generated by polymerization of mixtures of N-isopropylacrylamide and di-block (hydrophilic-hydrophobic) and tri-block (hydrophobic-hydrophilic-hydrophobic) co-polymer acrylamide macro-monomers made of Jeffamine using recently developed biodegradable cross-linkers [
28]. The schematic structure of the NHG is represented in
Figure 1, notably a radial distribution of hydrophilic PEO (PEG) at high ratio in the outer shell of the NHGs (
Figure 1a in blue) and poly N-isopropyl acrylamide (PNIPAM) at high ratio in the center of the NHGs (
Figure 1b in yellow). NHGs possess two significant advantages: mono-dispersity and biocompatibility. Their unique design allows fine-tuning and optimization of biological applications by adjusting the particle size within the 100–400 nm range, while maintaining the same particle composition. This tunability offers potential for diverse applications, including targeted drug delivery, imaging, and tissue engineering, where particle size plays a critical role in determining the efficacy and specificity of the intended biological interaction.
2. Materials and Methods
Polymerizations were performed in a DAIHAN scientific water bath shaker polymerizer. Jeffamine® ED-2003 (Mn = 1900), acryloyl chloride, polyvinyl-pyrrolidone (PVP360000), potassium persulfate (KPS), ethylene glycol, triethanol amine and Amicon ultrafilter tube (100 kDa cutoff) were purchased from Merck (Jerusalem, Israel), N-isopropylacrylamide (NIPAM, 99.0%) from Acros Organics Co. Ltd. (St. Louis, MI, USA) and Cellulose Ester Spectra/Pore (1000 kDa cutoff) dialysis membrane from Spectrum Laboratories, Inc. (Rancho Dominguez, CA, USA).
Hydrodynamic sizes and size distributions were determined using a Zetasizer 3000 HSA (Malvern Instruments Ltd., Malvern, UK). NP tracking analysis (NTA) was conducted to determine the zeta-potential (ζ and NP number (concentration) using a ZetaView PMX-230 instrument (Particle Metrix GmbH, Germany). An XTT assay kit (Biological Industries, Beit Haemeq, Israel) was used according to the manufacturer’s instructions. A humidified water-jacketed automatic CO2 incubator (US Autoflow, NuAire Inc., Plymouth, MA, USA) was used for cell culture. Dimethyl sulfoxide (DMSO) and VRC were purchased from Merck-Sigma-Aldrich (Argentina), as well as mucin from porcine stomach (type II, powder), sodium alginate, calcium carbonate (CaCO3), D-(+)-gluconic acid δ-lactone (GDL), and sodium chloride (NaCl) used to develop the artificial airway mucus model. All solvents were used of analytical or high performance liquid chromatography (HPLC) grade.
2.1. Synthesis of NHGs
The novel NHGs of PNIPAAM-co-PPO-PEO-PPO (Poly(N-isopropylacrylamide)-co-Poly(propylene oxide)-Poly(ethylene oxide)-Poly(propylene oxide)) were obtained by free-radical polymerization using monomeric mixtures of (Acr)1.1Jeffamine
1900 and NIPAM, at differing ratios for obtaining different particle sizes as previously shown [
28].
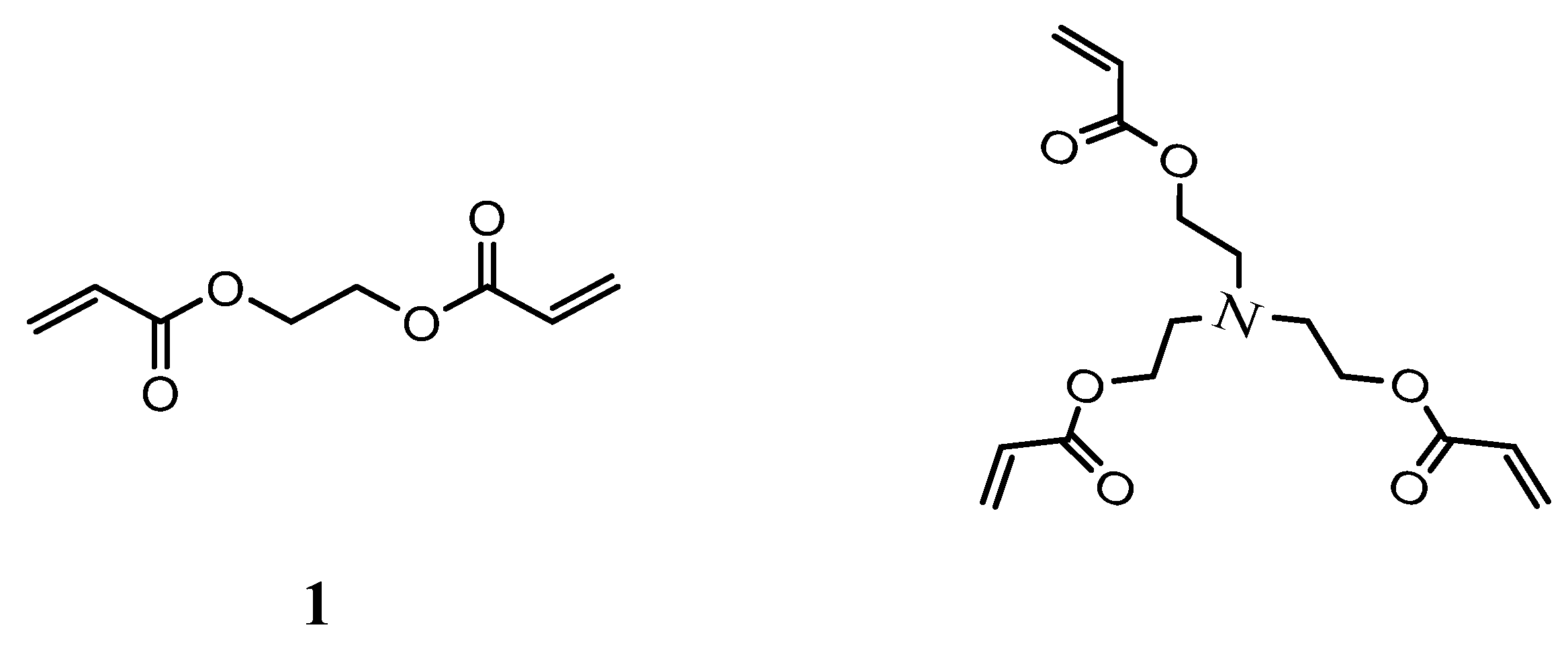
In addition, for obtaining two different families of biodegradable NHGs, the ester functionalized cross-linkers (
Figure 2): Ethane-1,2-diyl diacrylate (EDDA) and Nitrilotris(ethane-2,1-diyl) triacrylate (NTEDTA) were introduced in separate reactions in the presence of potassium persulfate (KPS) as initiator together with the surfactant polyvinylpyrrolidone (PVP
360000) [
28]. These cross-linkers were selected based on their hydrolytically labile ester bonds, which introduce controlled degradability into the polymer backbone under physiological conditions.
E1-3/E4-6 series present EDDA/NTEDTA cross-linked NHGs respectively (Table 1). Polymerization was performed at 73 °C in H
2O (10 mL) with PVP and KPS (5 mg each).
2.2. DLS Analysis
Hydrodynamic sizes and size distributions were measured using diluted NHGs (50 µg/3 mL) with a Zetasizer 3000 HSA (Malvern Instruments Ltd., UK) operating with a 4 mW HeNe laser (632.8 nm), a detector positioned at a scattering angle of 90° and a temperature-controlled cuvette. Three measurements consisting of 10 sub runs were performed for each sample.
2.3. Concentration and Zeta Potential (ζ by NTA
NTA was conducted to determine ζ and particle number (concentration) of the NPs using the ZetaView instrument described above. Data were collected and analyzed by ZetaView software (version 8.05.16 SP7). Reported particle numbers correspond to median.
All NHG samples were stored at 4 °C until measurement and were diluted to 1:50,000 in particle-free sterile Milli-Q (MQ) water immediately prior to measurement. The samples were analyzed in light scattering mode using a 488 nm laser. Particles were counted and size-distributed at 25 °C. Instrumental parameters were adjusted as follows: minimum brightness 30 AU, frame rate of 30 per second (10 per cycle), sensitivity 85 AU and shutter 100 AU. Data for two exposures at 11 measurement positions were collected for each sample. The zeta potential was measured twice at 25 °C with sensitivity 80 AU and shutter 150.
2.4. Mucus Penetration Assay for NHGs Monitored by NTA
To further investigate the interaction of nano-formulations with mucus, an assay was set to track the movement of single particles through simulated mucus. For this purpose, we used the previously described (section 2.3) NTA instrument for quantifying the number of NHGs of different size (100, 200 and 400 nm) that were able to cross a mucus layer prepared as described by Pacheco et al. [
27]. To evaluate the NP penetration, an experiment using a dual-chamber model was designed. In the upper compartment, a quantified amount (0.1 mg) of NHGs was placed onto a homogeneous film formed by 100 µL of simulated mucus, supported in a Transwell
® insert with a 1-µm microporous membrane. Meanwhile, the lower receiving compartment was filled with 2 mL of PBS. The same setup without the mucus layer was taken as control. In both cases, the entire setup was incubated at 37 °C for 24 h. At predetermined time intervals (0, 4 and 24 h), 200-µL samples were withdrawn from the lower compartment for analysis. To keep the lower-well volume constant, an appropriate refill of medium (PBS) was performed at each sampling point. For each withdrawn PBS sample, the number of NHGs that managed to pass through the mucus layer was measured by the NTA instrument. Until quantification, samples were preserved at 4 °C, and prior to measurement, dilution in particle-free sterile MQ water was performed, i.e., 1:25 v/v for samples up to 8 h and 1:2000 v/v otherwise (up to 24 h).
2.5. Loading of VRC
VRC-loaded NHGs (VRC-NHGs) were prepared by the solvent evaporation method [
29]. Briefly, VRC (10 mg) was dissolved in chloroform (1 mL) and 200 µL aliquots of the stock solution (10 mg/mL) were dropped into 1 mL of NHGs aqueous solution (10 mg/mL) and mixed overnight at room temperature (RT) to volatilize the chloroform. The obtained NHGs were isolated using an Amicon
® ultrafilter tube by centrifugation at 6,000 rpm for 20 min and repeated washings with DDW until no signal from the filtrate was detected by UV-visible spectroscopy. The loaded amount of drug was determined by a systematic method. A calibration plot (see Figure S1) was obtained using a set of standard solutions prepared with known concentrations of VRC in acetonitrile. The linear portion of this plot served as a reference to calculate the concentration of drug in NHGs.
The VRC-NHGs (1 mL, 10 mg/mL) were lyophilized for loading calculations. The freeze-dried powder was treated with 5 mL MeCN and filtered to obtain a clear solution that was analyzed by UV spectroscopy. The absorbance was measured at 255 nm, enabling determination of the loaded amount of drug by application of the calibration equation derived from the linear portion. This method provides a reliable approach for quantifying encapsulated drug in NHGs [
30]:
Drug loading efficiency (DLE) and drug loading capacity (DLC) were calculated as:
2.6. In Vitro Release of VRC
In vitro drug release studies were performed under sink conditions as previously reported [
31]. Briefly, VRC-NHGs containing equivalent amounts of VRC were transferred in a suspended dialysis bag to a container for urine analysis containing 100 mL phosphate-buffered saline (PBS) at pH 7.4 and 37 °C with continuous orbital agitation. Samples (1 mL) from the PBS bath were taken at specific intervals (1, 2, 4, 6, 24, 48, 72 h) for quantification of VRC and an equal amount of fresh PBS was replaced for keeping constant volume. A solution of free VRC at the same concentration placed in a dialysis bag using the same procedure was used as control. The amount of VRC released at each interval was analyzed by a fluorimeter (λ
ex = 255 nm, λ
em = 372 nm). Assays were done in duplicate and results are expressed as average ±SD.
A number of studies also reported the use of simulated interstitial lung fluid (SILF, pH 7.4) to investigate the release properties of drug formulations due to its close replication of the mucosal fluids in the respiratory system [
32,
33,
34]. A release experiment was also conducted in SILF (pH 7.4) supplemented with 1% w/v Tween
®80 as previously reported [
33].
Dialysis in SILF (pH 7.4) with addition of 1% w/v Tween® 80 was carried out as external medium to investigate the in vitro release profiles from the polymeric matrices of the NHGs. SILF was prepared in 1 L of distilled water (pH 7.4): MgCl2 (98.5 mg), NaCl (6.032 g), KCl (404 mg), Na2HPO4 (436 mg), NaHCO3 (2.59 g), Na2SO4 (83 mg), CaCl2 (359 mg), sodium acetate (607 mg) and trisodium citrate (115 mg).
2.7. Cell Culture
Human lung cancer cell line A549 and embryonic kidney cell line HEK293T were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS, Sigma-Aldrich, NY, USA), 100 μg/mL streptomycin and 100 U/mL penicillin at 37 °C in 5% CO2 atmosphere.
2.8. Cytotoxicity
The in vitro cytotoxicity of VRC-NHGs was determined in A549 and HEK293T cell lines by XTT assay and compared to free VRC. For this purpose, 8×103 cells/well were pre-incubated in a 96-well plate for 24 h and incubated for 48 h at 37 °C with 5% CO2 and different concentrations of non-loaded NHGs, free VRC or VRC-NHGs (60 μM VRC). Cells treated with 2% triton were used as positive control and fresh medium as negative control. Subsequently, 50 μL of XTT reagent (with initiator) were added and the cells were incubated for 4 h. The absorbance was read at 570 nm using a TECAN microplate reader.
Cell viability (%) = (I
sample/I
control) × 100, where I
sample is the absorbance of NHG treated wells and I
control is the absorbance of control wells without NHG treatment Experiments were repeated twice. Assays were carried out according to known procedures [
35].
2.9. Antifungal Susceptibility Testing
Antifungal susceptibility testing was performed in the mold isolates most frequently recovered from respiratory secretions of CF patients (
A. fumigatus, A. flavus, A terreus, A. niger, A. nidulans, S. aurantiacum and
E. dermatitidis) against non-loaded NHGs, VRC-NHGs, VRC suspension, and VRC/DMSO following the clinical laboratory standard institute (CLSI) M38 3rd ed. Document [
36]. All clinical fungal isolates are available in the culture collection of the Mycology bank of the Center of Mycology of the School of Medicine (University of Buenos Aires, Argentina).
A. fumigatus ATCC 204305 was used as quality control strain for susceptibility testing.
VRC suspension, VRC/DMSO (5 mg/ml) and VRC-loaded NHGs were prepared in sterile saline buffer. Two-fold dilutions of each drug formulation (32 to 0.06 μg/well) were prepared in 100 μL of RPMI−MOPS in 96-well plates. 5000 spores or 500 yeast cells were added to each well. Results were recorded 48 h post incubation at 37 °C. The MIC was defined as the lowest drug concentration causing complete inhibition of visible growth.
Aiming at developing an optimized method for administration of VRC through the mucus barrier, the antifungal activity of VRC-NHGs was evaluated using the CF mucus composition previously described by Pacheco et al. for drug diffusion assays [
27]. A 16.3 mg/mL NaCl solution was prepared, and a solution of 21 mg/mL alginate sodium salt was dissolved in it using gentle magnetic agitation. Concurrently, a suspension containing 43.7 mg/mL mucin was meticulously prepared in DDW and left to gently agitate overnight. Subsequently, the alginate and mucin solutions were combined at a 1:4 ratio utilizing interconnected luer-lock syringes, and a suspension of 7 mg/mL CaCO
3 in the 16.3 mg/mL NaCl solution was intricately blended at 1:5 proportions. A freshly concocted 70 mg/mL GDL solution in 16.3 mg/mL NaCl was merged with the previously created suspension (comprising alginate, mucin and CaCO
3) at 1:6 proportions, always under aseptic conditions. Lastly, 100 μL of the prepared mucus were accurately pipetted onto the 96-well surface, previously inoculated with fungal conidia. The different VRC formulations (VRC-NHGs, VRC suspension and VRC/DMSO) were added onto the surface of the mucus layer, finalizing the experimental procedure. This antifungal susceptibility procedure was performed according to the abovementioned CLSI document against
A. fumigatus as the most common filamentous fungus isolated from the airways of CF patients [
36].
3. Results and Discussion
3.1. Physico-Chemical Characterization of NHGs and VRC-NHGs
Chronic pulmonary infections caused by opportunistic pathogens affect more than 90% of CF patients. In addition to bacterial colonization, these patients are predisposed to fungal colonization due to the capacity of environmental fungi to colonize the lower respiratory tract and to the frequent cycling of antibiotics required to control the disease.
One of the main goals of the present study was the synthesis of a series of NHGs based on our established methodology as a platform for loading the antifungal drug VRC as a delivery system for an optimized antifungal therapy of broncho-pulmonary infections caused by molds in CF patients. As the mucus presents a diffusional barrier to the antifungal treatment, the NHGs used in this study are composed of a shell of PEG recently shown to be capable of crossing lung-generated mucus [
25].
With this in mind, we developed novel polymeric monodispersed NHGs with a large range of sizes starting from 100 nm up to 400 nm that according to previous results exhibited high biocompatibility both in vitro and in vivo [
28,
30,
37,
38]. This non-toxic profile may support its potential use for treating fungal infections especially in CF patients.
In the present study monomeric mixtures in water at RT spontaneously formed star/flower-like self-assemblies with uncontrollable poly-dispersed sizes around 100–800 nm. Upon heating at 75 °C, NIPAM and the hydrophobic elements of the macro-monomers (PPO) collapse together into well-defined self-assemblies probably with high concentration of the hydrophobic elements in the core (NIPAM and PPO chains) and a high concentration of the hydrophilic elements of the macro-monomeric chains (PEO and NH
2 groups) at the outer shell. These self-assemblies can be polymerized leading to particles with sizes that correlate with those of the intermediate self-assemblies. The size of the assemblies can be tuned/calibrated by combining different ratios of the starting monomeric mixtures at high temperature, as previously reported [
28,
30,
37,
38]. Larger amounts of (Acr)1.1Jeffamine1900 correspond to smaller NHG diameters due to increased hydrophobic interactions during self-assembly.
D
h represents the particle size of pristine and VRC-NHGs as measured by DLS at 25 °C, and the polydispersity index (PDI) represents the relative variance in the particle size distribution [
39,
40]. Each value is the average of three measurements by DLS. The results shown in
Table 2 indicate that both EDDA cross-linked (series E
1-3) and NTEDTA cross-linked NHGs (series E
4-6) are highly monodisperse.
D
H of the VRC-NHGs is a key parameter for their potential clinical use as nanocarriers in CF patients. Carriers in the nano-scale have a greater potential to penetrate the airway mucus of CF patients than microparticles, avoiding steric interactions [
41]. Thus, VRC-NHGs can overcome the mucus barrier for a better treatment outcome. Data summarized in
Table 2 shows that all VRC-NHGs maintain their size upon encapsulation. For instance, the E
1 batch of NHGs and VRC-NHGs span 410 and 424 nm, respectively. In both cases, there is a unimodal narrow size distribution according to the PDI (0.17/0.12 for NHGs/ VRC-NHGs).
The zeta potential measures the electrostatic charge on the surface of particles in a colloidal dispersion (at the shear plane of the particle) [
39,
40]. The potentials of NHGs and VRC-NHGs were examined by NTA at 25 °C, pH 7.4. All potentials measured at neutral pH were clearly negative (
Table 2). This negativity is attributed to the presence of sulfate groups generated at the starting polymeric chains by KPS initiator that are accommodated at the shear plane of the NHGs. A negative zeta potential offers several benefits for NPs. It enhances stability by repelling charges, preventing aggregation, and ensuring a more uniform dispersion, crucial for colloidal stability [
42,
43]. In biomedical applications, NPs with higher negative potential typically show improved bioavailability. Cellular uptake and intracellular trafficking can also be influenced, providing better control over drug release kinetics and targeting specificity, which is particularly advantageous for drug delivery systems [
42]. Moreover, the reduced interaction with cells and tissues due to a high negative
ζ can potentially lower the toxicity of NPs, enhancing their biocompatibility, in particular in biomedical imaging, diagnostics and therapeutics [
43].
3.2. Mucus Penetration Assay of NHGs Monitored by NTA
The goal of this experiment was to evaluate NHGs diffusion through artificial mucus by means of using a dual-chamber setup (
Figure 3a). PBS samples taken from the lower compartment at specific intervals (0, 4 and 24 h) were measured by NTA to quantify the particles reaching that compartment after crossing the mucus layer. Percentages (see
Table 3 and Table S1) were calculated relative to the total number of particles on the receptor media of the same setup without mucus (
Figure 3b). Results are shown in
Table 3 and
Figure 3-b, as percentage of added particles overpassing mucus over time.
Series E
1-3 shows in general better penetration than series E
4-6. As for the size, larger NPs display more penetration for both series. We speculate that smaller particles seem to exhibit more interaction with the mucus film interstices, and therefore spend more time within this layer as compared to larger ones, in contrast to previous reports [
44,
45], therefore the reason for this behavior remains to be elucidated.
3.3. VRC Loading into NHGs
In all loading experiments, the applied amount of VRC was 2 mg into 10 mg of NHGs. The DLE and DLC were calculated according to these parameters. Results in
Table 4 indicate that larger NHGs have better drug loading capacity that is directly proportional to their increased hydrophobicity due to their higher content of PNIPAM in their structure as measured by NMR in a previous report [
28]. Both series display similar loading capacity.
Overall, both NP hydrophobicity and size, as well as the hydrophobic nature of the drug, contribute to the drug loading capacity [
46].
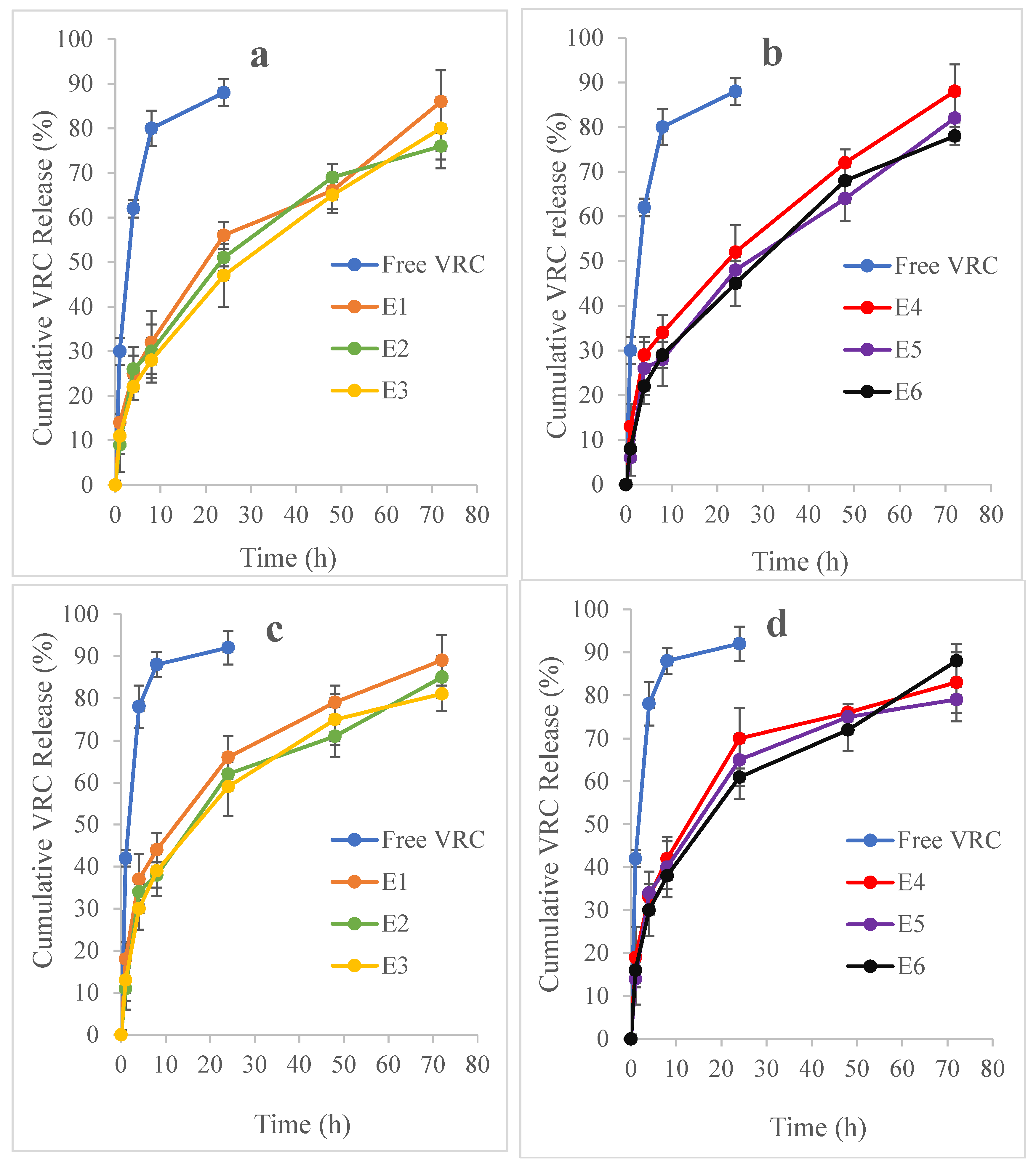
3.4. In Vitro Drug Release Study of VRC-NHGs
To investigate the drug release from the NHGs, the fluorescence of released VRC was measured at predetermined time intervals (λ
ex = 255 nm, λ
em = 372 nm). VRC-NHGs and free VRC as control were dialyzed in PBS (50 mL), as largely studied by others under sink conditions [
31,
47].
VRC-NHGs from both series (
Figure 4a and 4b) showed similar release profiles with approximately 50% of encapsulated VRC released within 24 h and around 80% released after 72 h. In contrast, the free VRC control demonstrated a more rapid release, exceeding 80% after just 8 h, emphasizing the role of NHGs in facilitating a controlled release mechanism.
A release experiment was also conducted in SILF (pH 7.4) supplemented with 1% w/v Tween
®80 as previously reported [
33]. This external medium was utilized to explore the in vitro release profiles of VRC. Here, we dialyzed VRC-NHGs and free VRC as control in SILF (50 mL), as largely studied by others under sink conditions [
32,
33,
34].
In the SILF environment, both series of VRC-NHGs exhibited similar release profiles (
Figure 4c and 4d) with over 60% of encapsulated VRC released within 24 h and ~85% after 72 h. The free VRC control exhibited a release of 85% after 8 h. This further highlights the role of NHGs in enabling a sustained release of VRC.
The slightly faster release of VRC observed in the SILF environment as compared to PBS environment can be attributed to its ionic composition, which closely mimics lung interstitial conditions, enhancing drug solubility and diffusion.
3.5. Cytotoxicity Studies of NHGs and VRC-NHGs
Ensuring the safety and compatibility of the formulation with lung cells is essential for developing a pulmonary drug delivery system, as inhalation of toxic substances may deplete lung surfactant and trigger phagocytic cell activation [
48].
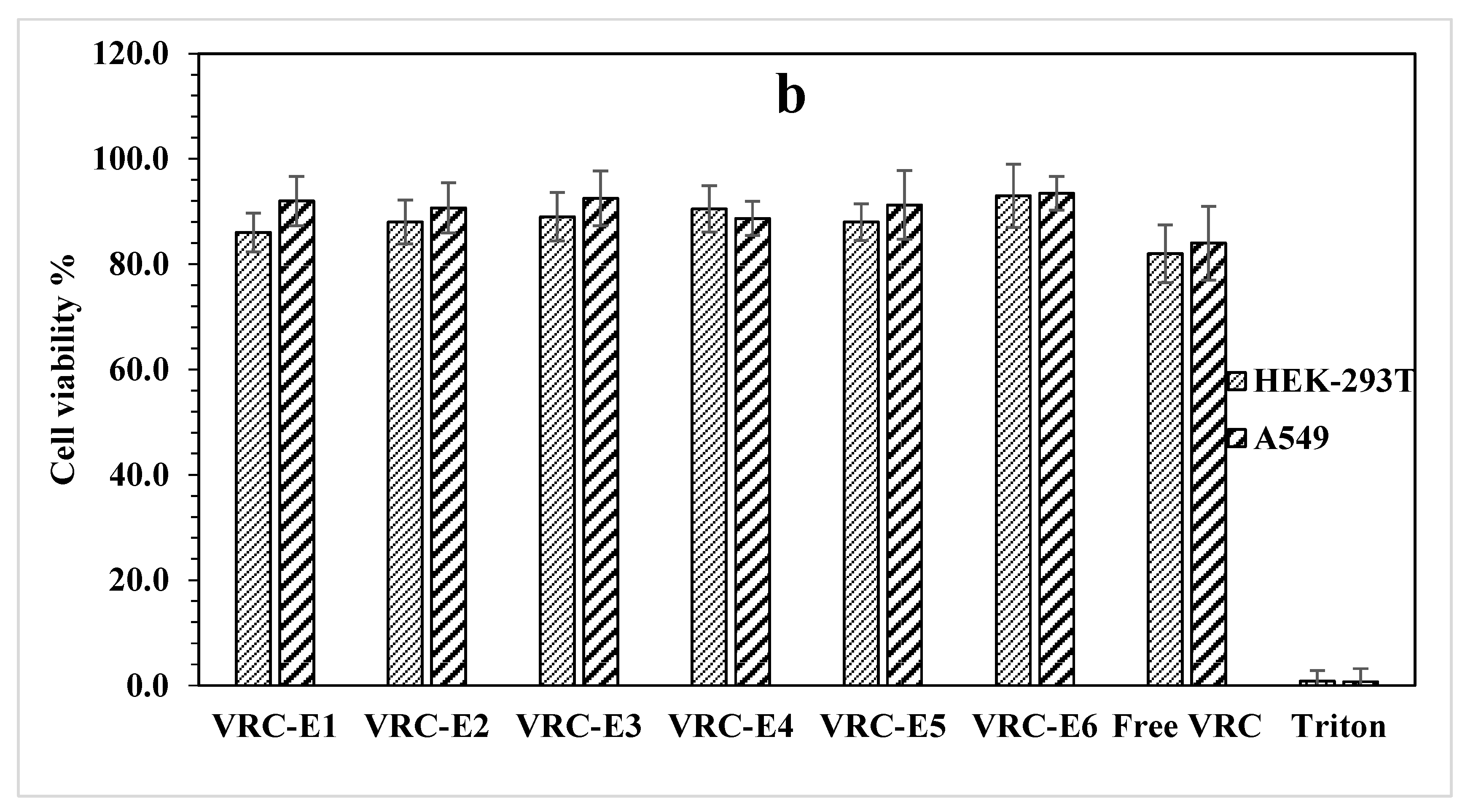
Both NHGs and VRC-NHGs were tested for cell toxicity using XTT viability assay and compared to the relevant controls on HEK-293T and A549 cell lines.
The results demonstrate that NHGs (Figure 5a) are devoid of toxicity at concentrations of 2 mg/mL on A549 and HEK293T cell lines after incubating 48 h. Both cell lines show beyond 90% viability. This concentration is by far higher than those reported for most of the NPs reported as having low toxicity [
28]
. VRC-NHGs are non-toxic with 20 µg/mL of VRC on A549 and HEK-293T after incubating for 48 h (Figure 5b). Both cell lines show beyond 80% viability. The viability of the control group (free VRC: 20 µg/mL) was 80–84%.
These results show that VRC-NHGs could be considered as a “safe nano-formulation” where cell viability is not compromised. Cell viability above 70% is considered safe in vitro assays according to the ISO 10993-5 guideline [
49].
3.6. Antifungal Effect of VRC-NHGs
The antifungal studies showed that only 400-nm VRC-NHGs (see E
1 and E
4 in
Table 5) maintain high activity, similar to free VRC in DMSO but higher than the aqueous suspension. Moreover, despite the introduction of a mucus barrier for
A. Fumigatus denoted with a star, the antifungal effect was similar in all cases (see
Table 5—
A. fumigatus and * A. fumigatus). The non-loaded NHGs did not display any antifungal activity against any of the studied strains (data not shown). Activity decreases proportionally with size, resulting in MIC similar to VRC suspension (see E
2, E
3, E
5 and E
6 in
Table 5).
The higher or similar activity of the VRC-DMSO can be attributed to the fact that under the standard conditions used for the MIC assays (48h incubation), about 25% of the VRC is still loaded in the NHGs and is not available for inhibition. By using an in vitro model of pulmonary pathological mucus which mimics the chemical composition, structural features, and viscoelastic properties of CF mucus [
27], we studied their penetration across this diffusional barrier and assessed the antimicrobial capacity of the developed NHGs against the three major molds affecting CF patients worldwide.
Our results with a series of NHGs of different size suggest that the 400-nm NHGs (E
1 and E
4) are able to penetrate through the mucus better as shown also for the non-loaded NHGs in
Figure 3, serving as a sustained release depot, improving the antifungal efficacy of VRC against the tested major molds. It should be further explored whether these mucus-penetrating NHGs, containing different hydrophobic therapeutics effectively bypass the mucus barrier allowing a better drug distribution and absorption at mucosal surfaces.
4. Conclusions
Ester-functionalized cross-linked NHGs with tunable sizes (100-400 nm), and devoid of toxicity, were prepared by free radical polymerization. The present study demonstrates that the newly developed NHGs are effective for controlled drug delivery, as evidenced by the in vitro controlled release of VRC-NHGs under different sink conditions.
The 400-nm VRC-NHGs (E1 and E4) exhibit high antifungal activity, comparable to free VRC in DMSO and superior to an aqueous VRC suspension. Notably, even in the presence of a mucus barrier, the antifungal effect remained consistent. This research showcases the significant advancements in the field of biodegradable NHGs and their potential for diverse biological applications such as the therapy of lung disorders caused by fungi in CF patients. These findings shall serve as a basis for upcoming projects aimed at further exploring the applications and advantages of these promising NHGs.
Acknowledgements
This work was supported by grants from the Ministry of Science, Technology and Innovation of the Argentine Republic (MINCyT) and the Ministry of Innovation, Science and Technology of the State of Israel (MoST) within the framework of the first Argentine-Israel Scientific Research Program. SDC part of his PhD dissertation. MC, RJG and MLC are staff members of CONICET. MFL and KHA thank CONICET for their doctoral scholarships. SDC is indebted to Bar Ilan University for the PhD scholarship. We are indebted for the support of the Marcus Center for Medicinal Chemistry at Bar Ilan University and to the Israel Science Foundation grant N01482/22
Conflicts of Interest
The authors declare no conflict of interest with respect to this manuscript.
References
- A. Warris, A. Bercusson, D. Armstrong-James, Aspergillus colonization and antifungal immunity in cystic fibrosis patients, Med. Mycol. 57 (2019) S118–S126. [CrossRef]
- C.M. Farinha, I. Callebaut, Molecular mechanisms of cystic fibrosis—how mutations lead to misfunction and guide therapy, Biosci. Rep. (2022). [CrossRef]
- N.L. Turcios, Cystic fibrosis lung disease: An overview, Respir. Care (2020). [CrossRef]
- J.S. Suk, S.K. Lai, Y.Y. Wang, L.M. Ensign, P.L. Zeitlin, M.P. Boyle, J. Hanes, The penetration of fresh undiluted sputum expectorated by cystic fibrosis patients by non-adhesive polymer nanoparticles, Biomaterials (2009). [CrossRef]
- T. Yu, J. Chisholm, W.J. Choi, A. Anonuevo, S. Pulicare, W. Zhong, M. Chen, C. Fridley, S.K. Lai, L.M. Ensign, J.S. Suk, J. Hanes, Mucus-Penetrating Nanosuspensions for Enhanced Delivery of Poorly Soluble Drugs to Mucosal Surfaces, Adv. Healthc. Mater. (2016). [CrossRef]
- G.C. Hansson, Mucus and mucins in diseases of the intestinal and respiratory tracts, J. Intern. Med. (2019). [CrossRef]
- J. V. Fahy, B.F. Dickey, Airway Mucus Function and Dysfunction, N. Engl. J. Med. (2010). [CrossRef]
- O.W. Meldrum, S.H. Chotirmall, Mucus, microbiomes and pulmonary disease, Biomedicines (2021). [CrossRef]
- R.A. Cone, Barrier properties of mucus, Adv. Drug Deliv. Rev. (2009). [CrossRef]
- J.P. Pearson, P.I. Chater, M.D. Wilcox, The properties of the mucus barrier, a unique gel—how can nanoparticles cross it?, Ther. Deliv. (2016). [CrossRef]
- X. Murgia, B. Loretz, O. Hartwig, M. Hittinger, C.M. Lehr, The role of mucus on drug transport and its potential to affect therapeutic outcomes, Adv. Drug Deliv. Rev. (2018). [CrossRef]
- H.H. Sigurdsson, J. Kirch, C.M. Lehr, Mucus as a barrier to lipophilic drugs, Int. J. Pharm. (2013). [CrossRef]
- M. Boegh, H.M. Nielsen, Mucus as a barrier to drug delivery—Understanding and mimicking the barrier properties, Basic Clin. Pharmacol. Toxicol. (2015). [CrossRef]
- J. King, S.F. Brunel, A. Warris, Aspergillus infections in cystic fibrosis, J. Infect. (2016). [CrossRef]
- S.H. Chotirmall, N.G. McElvaney, Fungi in the cystic fibrosis lung: Bystanders or pathogens?, Int. J. Biochem. Cell Biol. (2014). [CrossRef]
- M.T. Martín-Gómez, Taking a look on fungi in cystic fibrosis: More questions than answers, Rev. Iberoam. Micol. (2020). [CrossRef]
- M.T. Montagna, G. Barbuti, F. Paglionico, G. Lovero, R. Iatta, O. De Giglio, T. Cuna, C. Coretti, T. Santostasi, A. Polizzi, A. Manca, G. Caggiano, J.P. Bouchara, Retrospective analysis of microorganisms isolated from cystic fibrosis patients in Southern Italy, 2002-2010, J. Prev. Med. Hyg. (2011). [CrossRef]
- C. Viñado, R.M. Girón, E. Ibáñez, A. García-Ortega, I. Pérez, D. Polanco, J. Pemán, A. Solé, Filamentous fungi in the airway of patients with cystic fibrosis: Just spectators?, Rev. Iberoam. Micol. (2021). [CrossRef]
- D. Seidel, A. Meißner, M. Lackner, E. Piepenbrock, J. Salmanton-García, M. Stecher, S. Mellinghoff, A. Hamprecht, L. Durán Graeff, P. Köhler, M.P. Cheng, J. Denis, I. Chedotal, J. Chander, D.L. Pakstis, I. Los-Arcos, M. Slavin, M.T. Montagna, G. Caggiano, M. Mares, J. Trauth, U. Aurbach, M.J.G.T. Vehreschild, J.J. Vehreschild, R.F. Duarte, R. Herbrecht, H. Wisplinghoff, O.A. Cornely, Prognostic factors in 264 adults with invasive Scedosporium spp. and Lomentospora prolificans infection reported in the literature and FungiScope®, Crit. Rev. Microbiol. (2019). [CrossRef]
- M. Pihet, J. Carrere, B. Cimon, D. Chabasse, L. Delhaes, F. Symoens, J.P. Bouchara, Occurrence and relevance of filamentous fungi in respiratory secretions of patients with cystic fibrosis—A review, in: Med. Mycol., 2009. [CrossRef]
- B. Cimon, J. Carrère, J.F. Vinatier, J.P. Chazalette, D. Chabasse, J.P. Bouchara, Clinical significance of Scedosporium apiospermum in patients with cystic fibrosis, Eur. J. Clin. Microbiol. Infect. Dis. (2000). [CrossRef]
- Ö. Güngör, Z. Tamay, N. Güler, Z. Erturan, Frequency of fungi in respiratory samples from Turkish cystic fibrosis patients, Mycoses (2013). [CrossRef]
- S. Ziesing, S. Suerbaum, L. Sedlacek, Fungal epidemiology and diversity in cystic fibrosis patients over a 5-year period in a national reference center, Med. Mycol. (2016). [CrossRef]
- P. Lakhani, A. Patil, S. Majumdar, Challenges in the Polyene- and Azole-Based Pharmacotherapy of Ocular Fungal Infections, J. Ocul. Pharmacol. Ther. (2019). [CrossRef]
- F. Wan, S.S.R. Bohr, S.N. Kłodzińska, H. Jumaa, Z. Huang, T. Nylander, M.B. Thygesen, K.K. Sørensen, K.J. Jensen, C. Sternberg, N. Hatzakis, H. Mørck Nielsen, Ultrasmall TPGS-PLGA Hybrid Nanoparticles for Site-Specific Delivery of Antibiotics into Pseudomonas aeruginosa Biofilms in Lungs, ACS Appl. Mater. Interfaces (2020). [CrossRef]
- J.S. Suk, A.J. Kim, K. Trehan, C.S. Schneider, L. Cebotaru, O.M. Woodward, N.J. Boylan, M.P. Boyle, S.K. Lai, W.B. Guggino, J. Hanes, Lung gene therapy with highly compacted DNA nanoparticles that overcome the mucus barrier, J. Control. Release (2014). [CrossRef]
- D.P. Pacheco, C.S. Butnarasu, F. Briatico Vangosa, L. Pastorino, L. Visai, S. Visentin, P. Petrini, Disassembling the complexity of mucus barriers to develop a fast screening tool for early drug discovery, J. Mater. Chem. B (2019). [CrossRef]
- S.D. Cemal, G. Kazimirsky, Y. Shadkchan, L. Eswaran, R. Abramovitch, N. Abudi, M.L. Cuestas, N. Osherov, G. Byk, Biocompatible Narrow Size Nanohydrogels for Drug Delivery, Nanomedicine: NBM (2025), in press. [CrossRef]
- H. Yang, Q. Wang, W. Chen, Y. Zhao, T. Yong, L. Gan, H. Xu, X. Yang, Hydrophilicity/hydrophobicity reversable and redox-sensitive nanogels for anticancer drug delivery, Mol. Pharm. (2015). [CrossRef]
- S. Palakkal, D. Logviniuk, G. Byk, Tuning the size and hydrophobicity of nanohydrogels exploiting a self-assembly assisted polymerization mechanism for controlled drug delivery, J. Nanoparticle Res. (2020). [CrossRef]
- F. Shi, J. Ding, C. Xiao, X. Zhuang, C. He, L. Chen, X. Chen, Intracellular microenvironment responsive PEGylated polypeptide nanogels with ionizable cores for efficient doxorubicin loading and triggered release, J. Mater. Chem. (2012). [CrossRef]
- V. Gupta, F. Ahsan, Influence of PEI as a core modifying agent on PLGA microspheres of PGE 1, a pulmonary selective vasodilator, Int. J. Pharm. (2011). [CrossRef]
- G. Costabile, I. D’Angelo, G. Rampioni, R. Bondì, B. Pompili, F. Ascenzioni, E. Mitidieri, R. D’Emmanuele Di Villa Bianca, R. Sorrentino, A. Miro, F. Quaglia, F. Imperi, L. Leoni, F. Ungaro, Toward Repositioning Niclosamide for Antivirulence Therapy of Pseudomonas aeruginosa Lung Infections: Development of Inhalable Formulations through Nanosuspension Technology, Mol. Pharm. (2015). [CrossRef]
- D.S. Pellosi, I. d’Angelo, S. Maiolino, E. Mitidieri, R. d’Emmanuele di Villa Bianca, R. Sorrentino, F. Quaglia, F. Ungaro, In vitro/in vivo investigation on the potential of Pluronic® mixed micelles for pulmonary drug delivery, Eur. J. Pharm. Biopharm. (2018). [CrossRef]
- N. Gulati, R. Rastogi, A.K. Dinda, R. Saxena, V. Koul, Characterization and cell material interactions of PEGylated PNIPAAM nanoparticles, Colloids Surfaces B Biointerfaces (2010). [CrossRef]
- Clinical Laboratory Standard Institute (CLSI), Reference Method for Broth Dilution Antifungal Susceptibility Testing of Filamentous Fungi, 3rd ed. CLSI standard M38, Wayne, PA Clin. Lab. Stand. Inst. (2017).
- R. Khandadash, V. Machtey, I. Shainer, H.E. Gottlieb, Y. Gothilf, Y. Ebenstein, A. Weiss, G. Byk, Novel biocompatible hydrogel nanoparticles: generation and size-tuning of nanoparticles by the formation of micelle templates obtained from thermo-responsive monomers mixtures, J. Nanoparticle Res. (2014). [CrossRef]
- L. Eswaran, G. Kazimirsky, G. Byk, New Biocompatible Nanohydrogels of Predefined Sizes for Complexing Nucleic Acids, Pharmaceutics (2023). [CrossRef]
- S.C. Yang, S.Y.R. Paik, J. Ryu, K.O. Choi, T.S. Kang, J.K. Lee, C.W. Song, S. Ko, Dynamic light scattering-based method to determine primary particle size of iron oxide nanoparticles in simulated gastrointestinal fluid, Food Chem. (2014). [CrossRef]
- F.H. Yang, Q. Zhang, Q.Y. Liang, S.Q. Wang, B.X. Zhao, Y.T. Wang, Y. Cai, G.F. Li, Bioavailability enhancement of paclitaxel via a novel oral drug delivery system: Paclitaxel-loaded glycyrrhizic acid micelles, Molecules (2015). [CrossRef]
- L.M. Ensign, C. Schneider, J.S. Suk, R. Cone, J. Hanes, Mucus penetrating nanoparticles: Biophysical tool and method of drug and gene delivery, Adv. Mater. (2012). [CrossRef]
- L. Li, H. Wang, J. Ye, Y. Chen, R. Wang, D. Jin, Y. Liu, Mechanism Study on Nanoparticle Negative Surface Charge Modification by Ascorbyl Palmitate and Its Improvement of Tumor Targeting Ability, Molecules (2022). [CrossRef]
- M. Gumustas, C.T. Sengel-Turk, A. Gumustas, S.A. Ozkan, B. Uslu, Effect of Polymer-Based Nanoparticles on the Assay of Antimicrobial Drug Delivery Systems, in: Multifunct. Syst. Comb. Deliv. Biosensing Diagnostics, 2017. [CrossRef]
- Y. Cu, W.M. Saltzman, Controlled surface modification with poly(ethylene)glycol enhances diffusion of PLGA nanoparticles in human cervical Mucus, Mol. Pharm. (2009). [CrossRef]
- X. Murgia, P. Pawelzyk, U.F. Schaefer, C. Wagner, N. Willenbacher, C.M. Lehr, Size-Limited Penetration of Nanoparticles into Porcine Respiratory Mucus after Aerosol Deposition, Biomacromolecules (2016). [CrossRef]
- S. Shen, Y. Wu, Y. Liu, D. Wu, High drug-loading nanomedicines: Progress, current status, and prospects, Int. J. Nanomedicine (2017). [CrossRef]
- S.A. Abouelmagd, B. Sun, A.C. Chang, Y.J. Ku, Y. Yeo, Release kinetics study of poorly water-soluble drugs from nanoparticles: Are we doing it right?, Mol. Pharm. (2015). [CrossRef]
- J.S. Patil, S. Sarasija, Pulmonary drug delivery strategies: A concise, systematic review, Lung India (2012). [CrossRef]
- I. INTERNATIONAL STANDARD, ISO 10993-5:2009—Biological evaluation of medical devices- Part 5: Tests for in vitro cytotoxicity, Int. Organ. Stand. Geneva, Switz. (2009).
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).