Submitted:
19 April 2025
Posted:
21 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
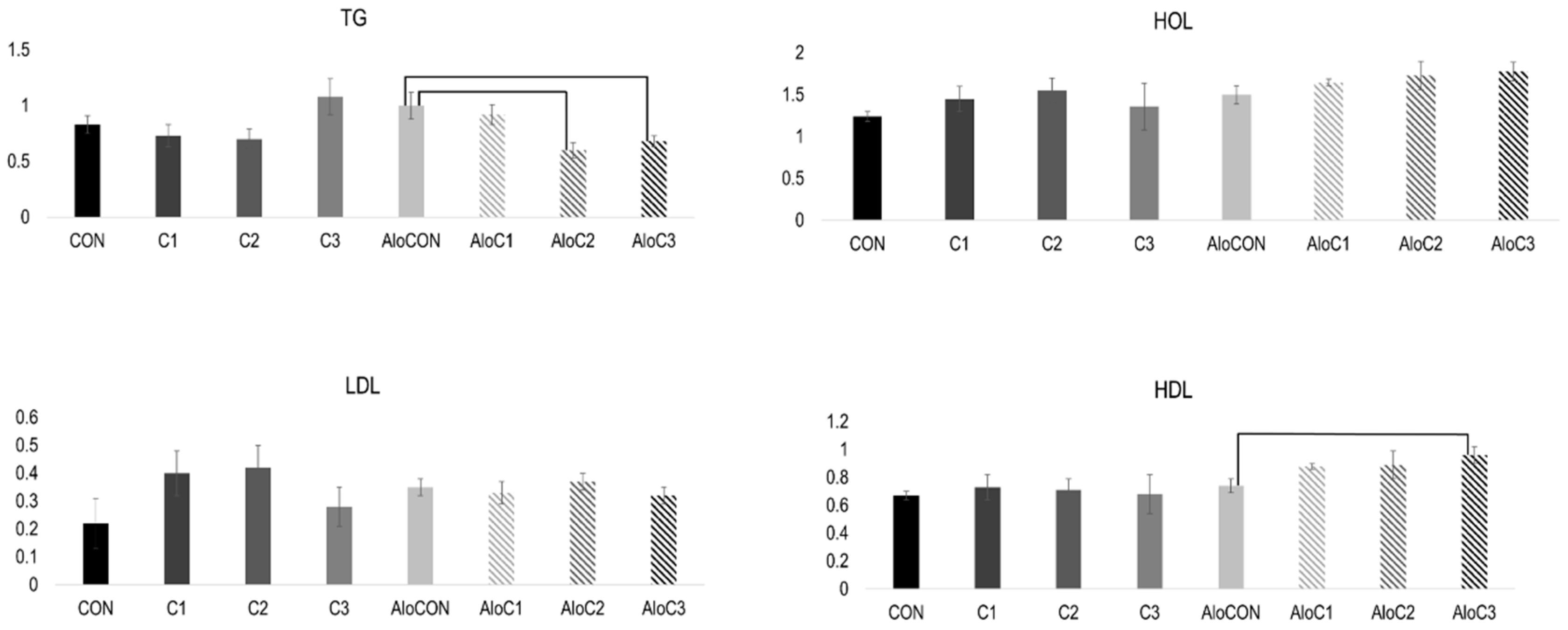
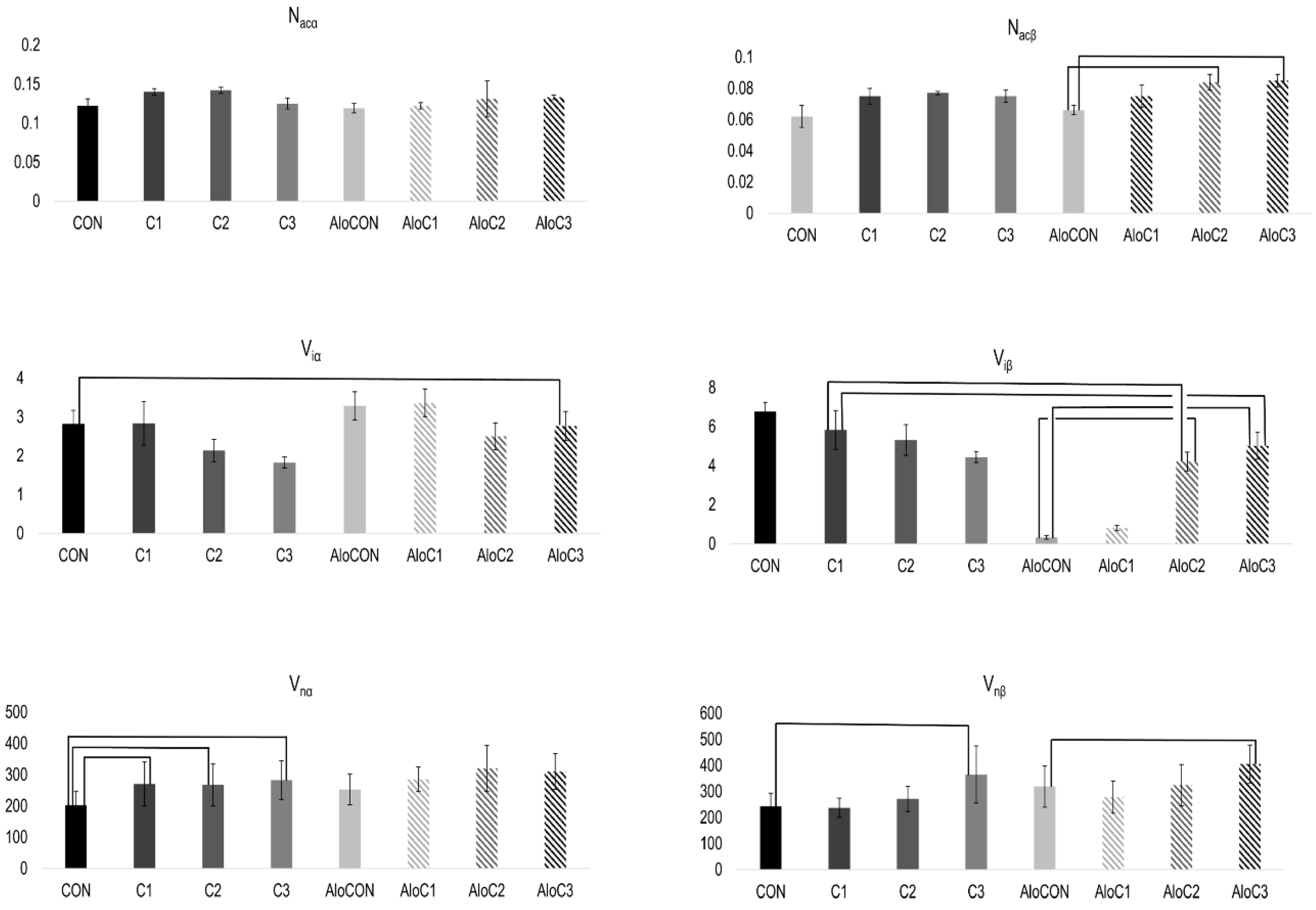
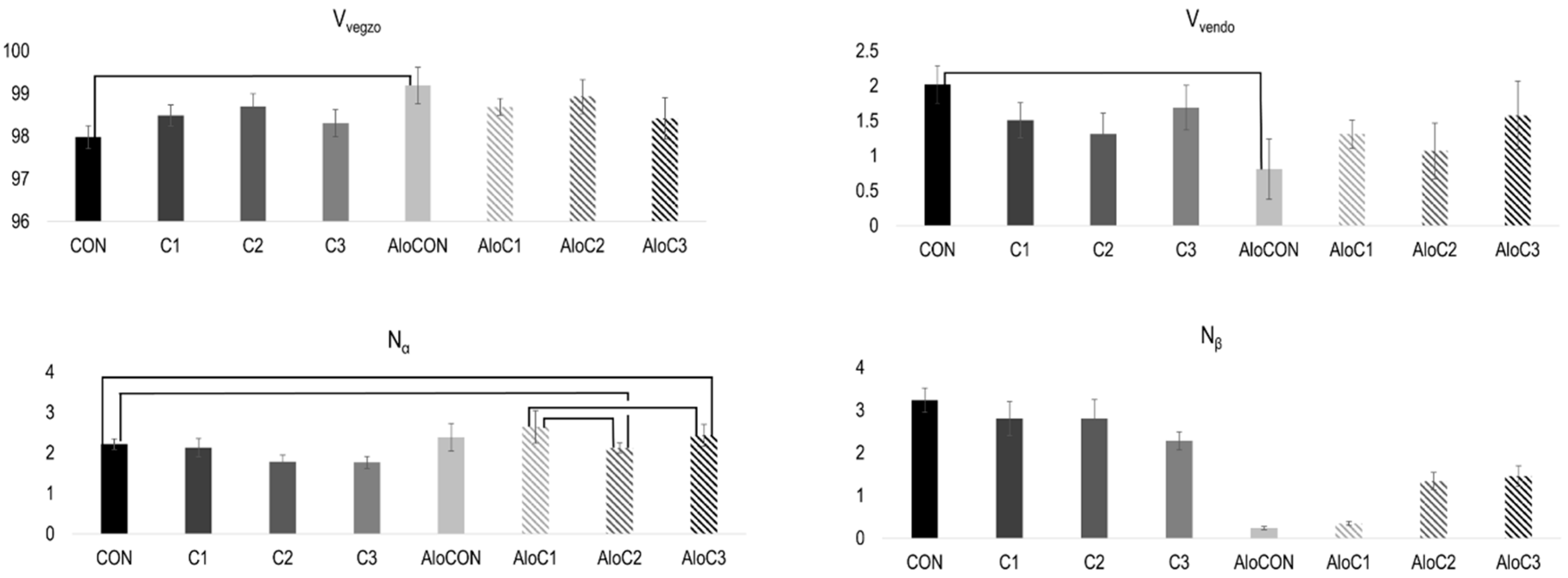
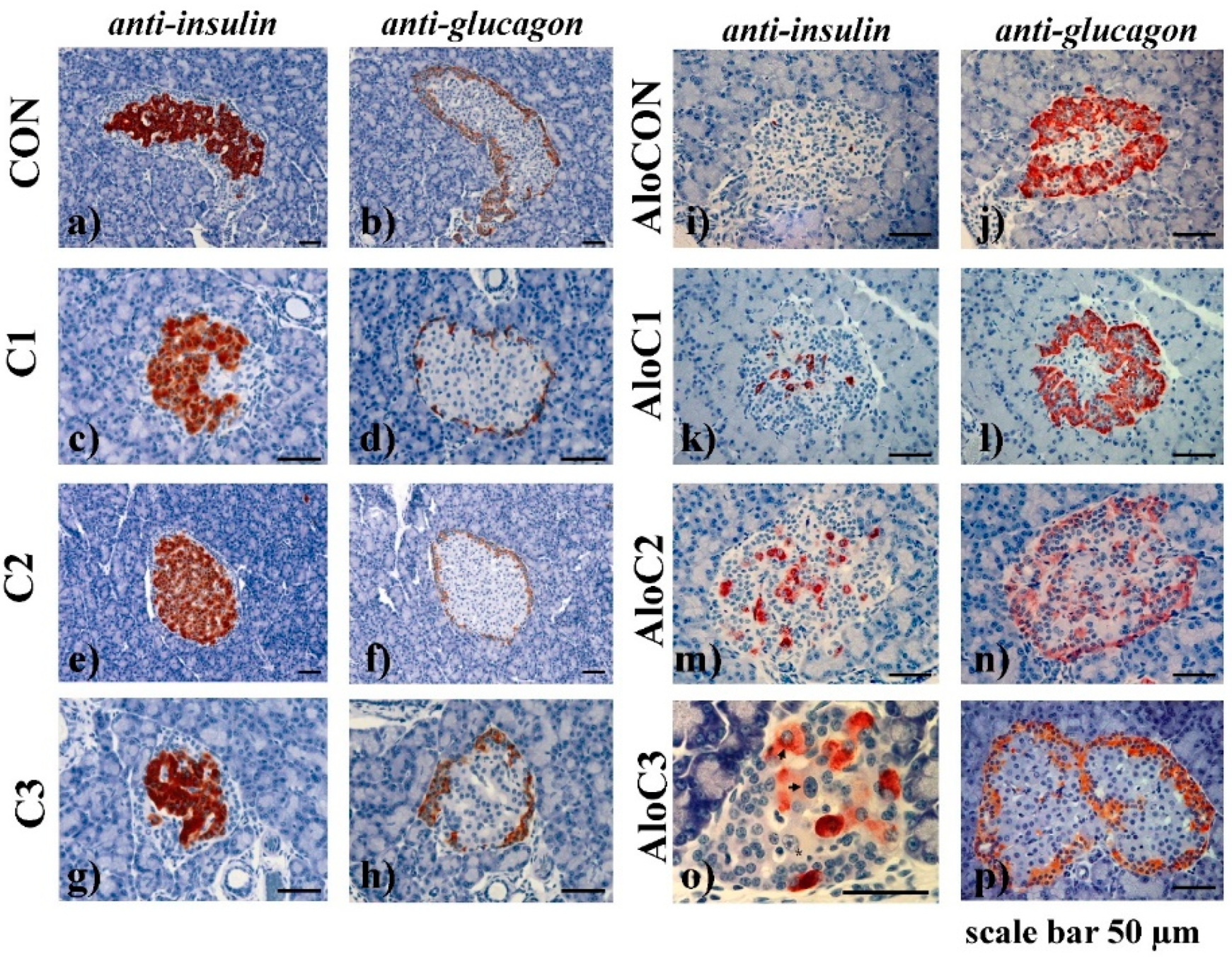
2. Results
3. Discussion
4. Materials and Methods
4.1. Mushroom Samples and Extraction Procedure
4.2. Chemicals
4.3. HPLC Analysis of Phenolics
4.4. In Silico Docking Study
4.5. Inhibition of α-Amylase Activity
4.6. Inhibition of α-Glucosidase Activity
4.7. Inhibition of Dipeptidyl Peptidase (DPP)-4 Activity
4.8. Laboratory Animals
4.9. Experimental Procedures
4.10. Biochemical Parameters Analysis
4.11. Immunohistochemical Analysis
4.12. Morphometrical Analyses
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Popović, M. , Vukmirović, S., Stilinović, N., Čapo, I., Jakovljević, V., 2010. Anti-Oxidative Activity of An Aqueous Suspension of Commercial Preparation of The Mushroom Coprinus comatus. Molecules 15, 4564–4571. [CrossRef]
- Stilinović, N. , Čapo, I., Vukmirović, S., Rašković, A., Tomas, A., Popović, M., Sabo, A., 2020. Chemical composition, nutritional profile and in vivo antioxidant properties of the cultivated mushroom Coprinus comatus. R. Soc. open sci. 7, 200900. [CrossRef]
- Gao, Z. , Kong, D., Cai, W., Zhang, J., Jia, L., 2021. Characterization and anti-diabetic nephropathic ability of mycelium polysaccharides from Coprinus comatus. Carbohydrate Polymers 251, 117081. [CrossRef]
- Nowakowski, P. , Naliwajko, S.K., Markiewicz-Żukowska, R., Borawska, M.H., Socha, K., 2020. The two faces of Coprinus comatus —Functional properties and potential hazards. Phytotherapy Research 34, 2932–2944. [CrossRef]
- Stojković, D. , Reis, F.S., Barros, L., Glamočlija, J., Ćirić, A., Van Griensven, L.J.I.D., Soković, M., Ferreira, I.C.F.R., 2013. Nutrients and non-nutrients composition and bioactivity of wild and cultivated Coprinus comatus (O.F.Müll.) Pers. Food and Chemical Toxicology 59, 289–296. [CrossRef]
- Park, H.J. , Yun, J., Jang, S.-H., Kang, S.N., Jeon, B.-S., Ko, Y.-G., Kim, H.-D., Won, C.-K., Kim, G.-S., Cho, J.-H., 2014. Coprinus comatus Cap Inhibits Adipocyte Differentiation via Regulation of PPARγ and Akt Signaling Pathway. PLoS ONE 9, e105809. [CrossRef]
- Ding, Z. , Lu, Y., Lu, Z., Lv, F., Wang, Y., Bie, X., Wang, F., Zhang, K., 2010. Hypoglycaemic effect of comatin, an antidiabetic substance separated from Coprinus comatus broth, on alloxan-induced-diabetic rats. Food Chemistry 121, 39–43. [CrossRef]
- Ratnaningtyas, N.I. , Hernayanti, H., Ekowati, N., Husen, F., 2022. Ethanol extract of the mushroom Coprinus comatus exhibits antidiabetic and antioxidant activities in streptozotocin-induced diabetic rats. Pharmaceutical Biology 60, 1126–1136. [CrossRef]
- Sabo, A. , Stilinovic, N., Vukmirovic, S., Bukumiric, Z., Capo, I., Jakovljevic, V., 2010. Pharmacodynamic action of a commercial preparation of the mushroom Coprinus comatus in rats. Phytotherapy Research 24, 1532–1537. [CrossRef]
- Stilinović, N. , Škrbić, B., Živančev, J., Mrmoš, N., Pavlović, N., Vukmirović, S., 2014. The level of elements and antioxidant activity of commercial dietary supplement formulations based on edible mushrooms. Food Funct. 5, 3170–3178. [CrossRef]
- Tešanović, K. , Pejin, B., Šibul, F., Matavulj, M., Rašeta, M., Janjušević, L., Karaman, M., 2017. A comparative overview of antioxidative properties and phenolic profiles of different fungal origins: fruiting bodies and submerged cultures of Coprinus comatus and Coprinellus truncorum. J Food Sci Technol 54, 430–438. [CrossRef]
- Zhang, Z. , Zhao, L., Qu, H., Zhou, H., Yang, H., Chen, H., 2022. Physicochemical characterization, adsorption function and prebiotic effect of chitin-glucan complex from mushroom Coprinus comatus. International Journal of Biological Macromolecules 206, 255–263. [CrossRef]
- Altındağ, F. , Rağbetli, M.Ç., Özdek, U., Koyun, N., Ismael Alhalboosi, J.K., Elasan, S., 2021. Combined treatment of sinapic acid and ellagic acid attenuates hyperglycemia in streptozotocin-induced diabetic rats. Food and Chemical Toxicology 156, 112443. [CrossRef]
- Parmar, H.S. , Jain, P., Chauhan, D.S., Bhinchar, M.K., Munjal, V., Yusuf, M., Choube, K., Tawani, A., Tiwari, V., Manivannan, E., Kumar, A., 2012. DPP-IV inhibitory potential of naringin: An in silico, in vitro and in vivo study. Diabetes Research and Clinical Practice 97, 105–111. [CrossRef]
- Fajarwati, I. , Solihin, D.D., Wresdiyati, T., Batubara, I., 2023. Self-recovery in diabetic Sprague Dawley rats induced by intraperitoneal alloxan and streptozotocin. Heliyon 9, e15533. [CrossRef]
- Ilić, V. , Vukmirović, S., Stilinović, N., Čapo, I., Arsenović, M., Milijašević, B., 2017. Insight into anti-diabetic effect of low dose of stevioside. Biomedicine & Pharmacotherapy 90, 216–221. [CrossRef]
- Marinho, T.D.S. , Martins, F.F., Cardoso, L.E.D.M., Aguila, M.B., Mandarim-de-Lacerda, C.A., 2022. Pancreatic islet cells disarray, apoptosis, and proliferation in obese mice. The role of Semaglutide treatment. Biochimie 193, 126–136. [CrossRef]
- Chen, C.-W. , Guan, B.-J., Alzahrani, M.R., Gao, Z., Gao, L., Bracey, S., Wu, J., Mbow, C.A., Jobava, R., Haataja, L., Zalavadia, A.H., Schaffer, A.E., Lee, H., LaFramboise, T., Bederman, I., Arvan, P., Mathews, C.E., Gerling, I.C., Kaestner, K.H., Tirosh, B., Engin, F., Hatzoglou, M., 2022. Adaptation to chronic ER stress enforces pancreatic β-cell plasticity. Nat Commun 13, 4621. [CrossRef]
- Perez-Frances, M. , Van Gurp, L., Abate, M.V., Cigliola, V., Furuyama, K., Bru-Tari, E., Oropeza, D., Carreaux, T., Fujitani, Y., Thorel, F., Herrera, P.L., 2021. Pancreatic Ppy-expressing γ-cells display mixed phenotypic traits and the adaptive plasticity to engage insulin production. Nat Commun 12, 4458. [CrossRef]
- Ruzittu, S. , Willnow, D., Spagnoli, F.M., 2020. Direct Lineage Reprogramming: Harnessing Cell Plasticity between Liver and Pancreas. Cold Spring Harb Perspect Biol 12, a035626. [CrossRef]
- Tritschler, S. , Theis, F.J., Lickert, H., Böttcher, A., 2017. Systematic single-cell analysis provides new insights into heterogeneity and plasticity of the pancreas. Molecular Metabolism 6, 974–990. [CrossRef]
- Aleixandre, A. , Gil, J.V., Sineiro, J., Rosell, C.M., 2022. Understanding phenolic acids inhibition of α-amylase and α-glucosidase and influence of reaction conditions. Food Chemistry 372, 131231. [CrossRef]
- Çayan, F. , Tel-Çayan, G., Deveci, E., Duru, M.E., 2021. HPLC–DAD characterization of phenolic profile and in vitro antioxidant, anticholinesterase, and antidiabetic activities of five mushroom species from Turkey. 3 Biotech 11, 273. [CrossRef]
- Rasouli, H. , Hosseini-Ghazvini, S.M.-B., Adibi, H., Khodarahmi, R., 2017. Differential α-amylase/α-glucosidase inhibitory activities of plant-derived phenolic compounds: a virtual screening perspective for the treatment of obesity and diabetes. Food Funct. 8, 1942–1954. [CrossRef]
- Tuersuntuoheti, T. , Pan, F., Zhang, M., Wang, Z., Han, J., Sun, Z., Song, W., 2022. Prediction of DPP-IV inhibitory potentials of polyphenols existed in Qingke barley fresh noodles: In vitro and in silico analyses. Food Processing Preservation 46. [CrossRef]
- Cao, H. , Ma, S., Guo, H., Cui, X., Wang, S., Zhong, X., Wu, Y., Zheng, W., Wang, H., Yu, J., Ma, L., Chun-chao, H., 2019. Comparative study on the monosaccharide compositions, antioxidant and hypoglycemic activities in vitro of intracellular and extracellular polysaccharides of liquid fermented Coprinus comatus. International Journal of Biological Macromolecules 139, 543–549. [CrossRef]
- Stojkovic, D. , Smiljkovic, M., Ciric, A., Glamoclija, J., Van Griensven, L., Ferreira, I.C.F.R., Sokovic, M., 2019. An insight into antidiabetic properties of six medicinal and edible mushrooms: Inhibition of α-amylase and α-glucosidase linked to type-2 diabetes. South African Journal of Botany 120, 100–103. [CrossRef]
- Deveci, E. , Çayan, F., Tel-Çayan, G., Duru, M.E., 2021. Inhibitory activities of medicinal mushrooms on α-amylase and α-glucosidase-enzymes related to type 2 diabetes. South African Journal of Botany 137, 19–23. [CrossRef]
- Yaribeygi, H. , Atkin, S.L., Sahebkar, A., 2019. Natural compounds with DPP-4 inhibitory effects: Implications for the treatment of diabetes. J of Cellular Biochemistry 120, 10909–10913. [CrossRef]
- Han C, Yuan J, Wang Y, Li L. Hypoglycemic activity of fermented mushroom of Coprinus comatus rich in vanadium. J Trace Elem Med Biol. 2006;20(3):191-6. [CrossRef] [PubMed]
- Yu, J. , Cui, P.-J., Zeng, W.-L., Xie, X.-L., Liang, W.-J., Lin, G.-B., Zeng, L., 2009. Protective effect of selenium-polysaccharides from the mycelia of Coprinus comatus on alloxan-induced oxidative stress in mice. Food Chemistry 117, 42–47. [CrossRef]
- Grimont, A. , Leach, S.D., Chandwani, R., 2022. Uncertain Beginnings: Acinar and Ductal Cell Plasticity in the Development of Pancreatic Cancer. Cellular and Molecular Gastroenterology and Hepatology 13, 369–382. [CrossRef]
- Salaj, N. , Kladar, N., Čonić, B.S., Jeremić, K., Barjaktarović, J., Hitl, M., Gavarić, N., Božin, B., 2020. Stabilization of sunflower and olive oils with savory (Satureja kitaibelii, Lamiaceae). J. Food Nutr. Res. 59.
- Pavlović, N. , Đanić, M., Stanimirov, B., Goločorbin-Kon, S., Stankov, K., Lalić-Popović, M., Mikov, M., 2019. In silico Discovery of Resveratrol Analogues as Potential Agents in Treatment of Metabolic Disorders. CPD 25, 3776–3783. [CrossRef]
- Kladar, N. , Mrđanović, J., Anačkov, G., Šolajić, S., Gavarić, N., Srđenović, B., Božin, B., 2017. Hypericum perforatum : Synthesis of Active Principles during Flowering and Fruitification—Novel Aspects of Biological Potential. Evidence-Based Complementary and Alternative Medicine 2017, 1–11.
| Class of phenols | Phenolic compound | Content [µg/g] |
|---|---|---|
| Phenolic acids | Gallic acid | ND |
| Caffeic acid | 12.88±0.64 | |
| trans-Cinnamic acid | ND | |
| p-Coumaric acid | 6.51±0.65 | |
| Chlorogenic acid | 25.48±1.27 | |
| Rosmarinic acid | ND | |
| Ferulic acid | ND | |
| Flavonoids | Quercetin | ND |
| Rutin | ND | |
| Quercitrin | ND |
| 1X70 (DPP4) | 5NN8 (alpha-glucosidase) | 3BAJ (alpha-amylase) | ||||
| Compound | MolDock Score (kcal/mol) | H bond (kcal/mol) | MolDock Score (kcal/mol) | H bond (kcal/mol) | MolDock Score (kcal/mol) | H bond (kcal/mol) |
| Sitagliptin/ acarbose | -152.99 | -2.16 | -88.60 | -9.63 | -157.24 | -25.16 |
| p-coumaric acid | -90.07 | -4.53 | -63.21 | -4.80 | -70.92 | -3.98 |
| caffeic acid | -80.72 | -9.71 | -67.35 | -7.97 | -81.29 | -9.02 |
| chlorogenic acid | -130.90 | -17.99 | -88.40 | -9.32 | -98.22 | -11.45 |
| Samples | Enzymes | ||
|---|---|---|---|
| α-amylase | α-glucosidase | DPP4 | |
| C. comatus | 13629.74 ± 1026.67 | 757.48 ± 84.45 | 1650.31 ± 145.29 |
| Acarbose | 4.93 ± 0.46 | 42.87 ± 3.96 | n.a. |
| Sitagliptin | n.a. | n.a. | 0.02 ± 0.001 |
| Group | BW start | BW end | ΔBW |
|---|---|---|---|
| CON | 245.17±5.64 | 316.33±5.14 | 71.17±8.35 |
| C1 | 258.67±11.86 | 349.50±18.84 | 90.83±7.56 |
| C2 | 249.67±8.44 | 332.17±19.83 | 82.50±14.22 |
| C3 | 265.50±12.71 | 342.17±19.11 | 76.67±7.31 |
| AloCON | 254.17±4.28 | 296.83±7.05 | 42.67±8.48 |
| AloC1 | 263.17±11.58 | 355.67±26.24 | 92.50±14.89 |
| AloC2 | 261.83±4.62 | 318.50±18.05 | 68.60±17.09 |
| AloC3 | 263.67±10.69 | 337.50±26.60 | 73.83±16.40 |
| Group | BG start | BG end | BG AUC | BG OGTT | ΔBG |
|---|---|---|---|---|---|
| CON | 7.93±0.12 | 6.72±0.16# | 281.74±8.20 | 12.65±0.41 | 5.83±0.32 |
| C1 | 7.80±0.19 | 6.28±0.23# | 264.60±5.50 | 13.53±0.53 | 6.43±0.49 |
| C2 | 7.55±0.13 | 5.48±0.20 | 287.64±3.45 | 12.23±0.74 | 5.60±0.62 |
| C3 | 7.97±0.14 | 6.30±0.11# | 277.73±4.92 | 14.20±0.42 | 7.15±0.39 |
| Group | BG before | BG alo | BG end1 | BG AUC1 | ΔBG1 |
|---|---|---|---|---|---|
| AloCON | 7.62±0.53 | 32.02±0.44 | 24.67±3.53 | 1116.79±72.42 | 7.35±3.22 |
| AloC1 | 8.20±0.35 | 32.53±0.26 | 24.37±0.60 | 1081.85±35.29 | 8.17±0.51 |
| AloC2 | 7.83±0.30 | 30.35±1.44 | 16.98±3.67 | 874.65±170.41 | 13.37±2.31 |
| AloC3 | 7.00±0.13 | 27.86±2.38 | 11.30±2.21* | 681.96±158.69 | 16.45±2.10* |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




