Submitted:
17 April 2025
Posted:
17 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.2. Cell Lines
2.3. Antibodies
2.4. Flow Cytometry
3. Results
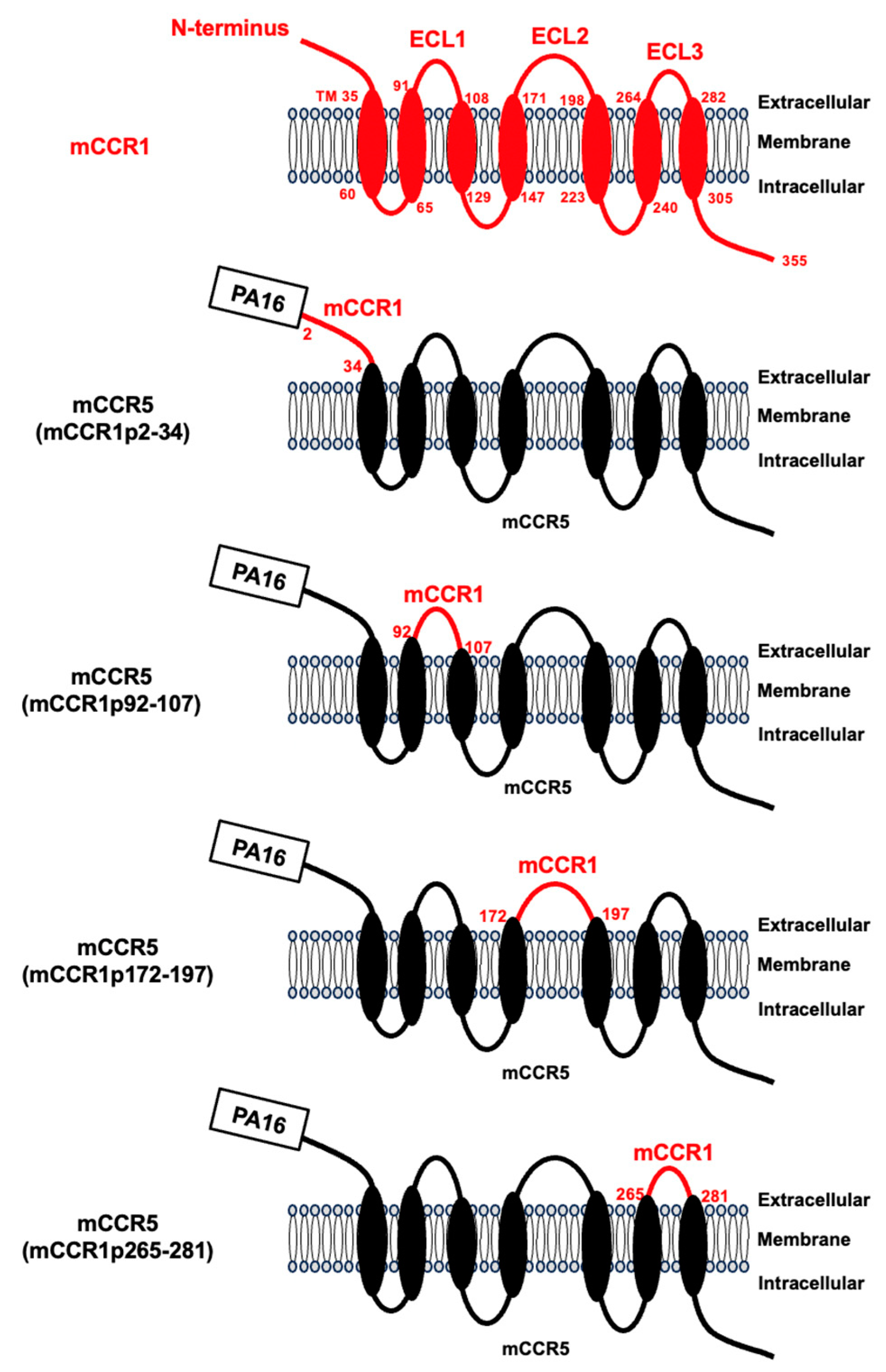
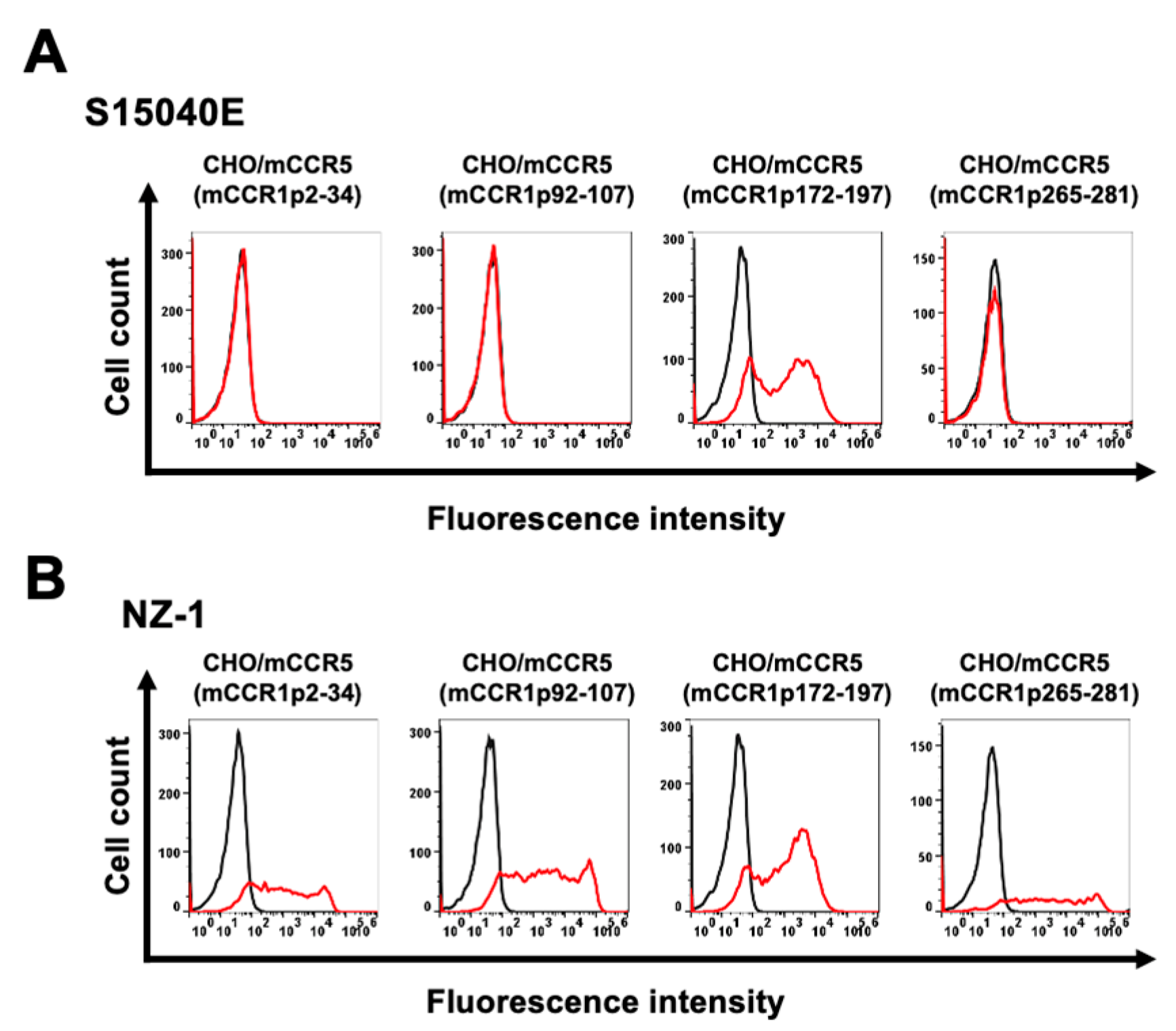
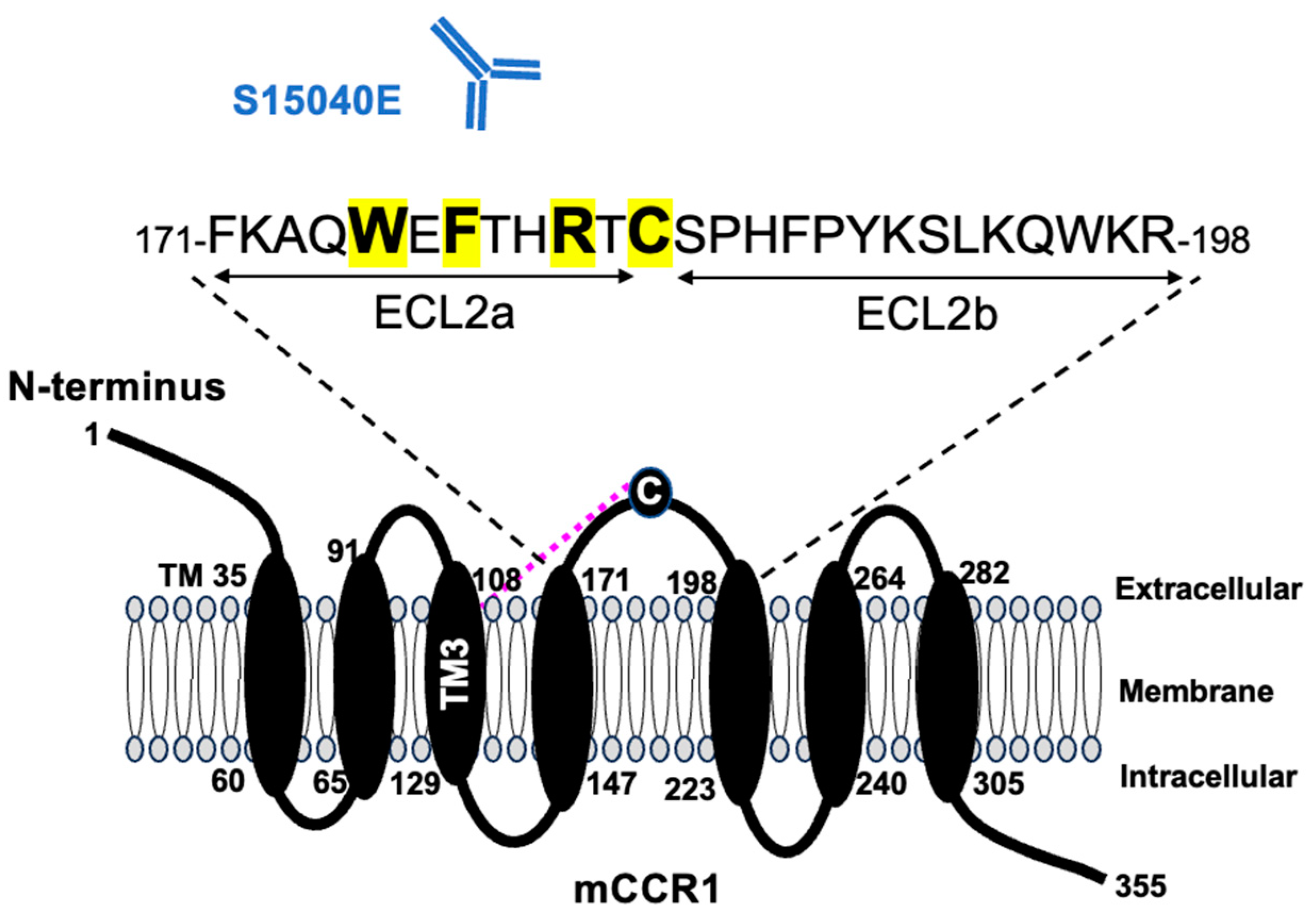
3.1. Determination of the Epitope of an Anti-mCCR1 mAb, S15040E, by Flow Cytometry Using Chimeric Proteins
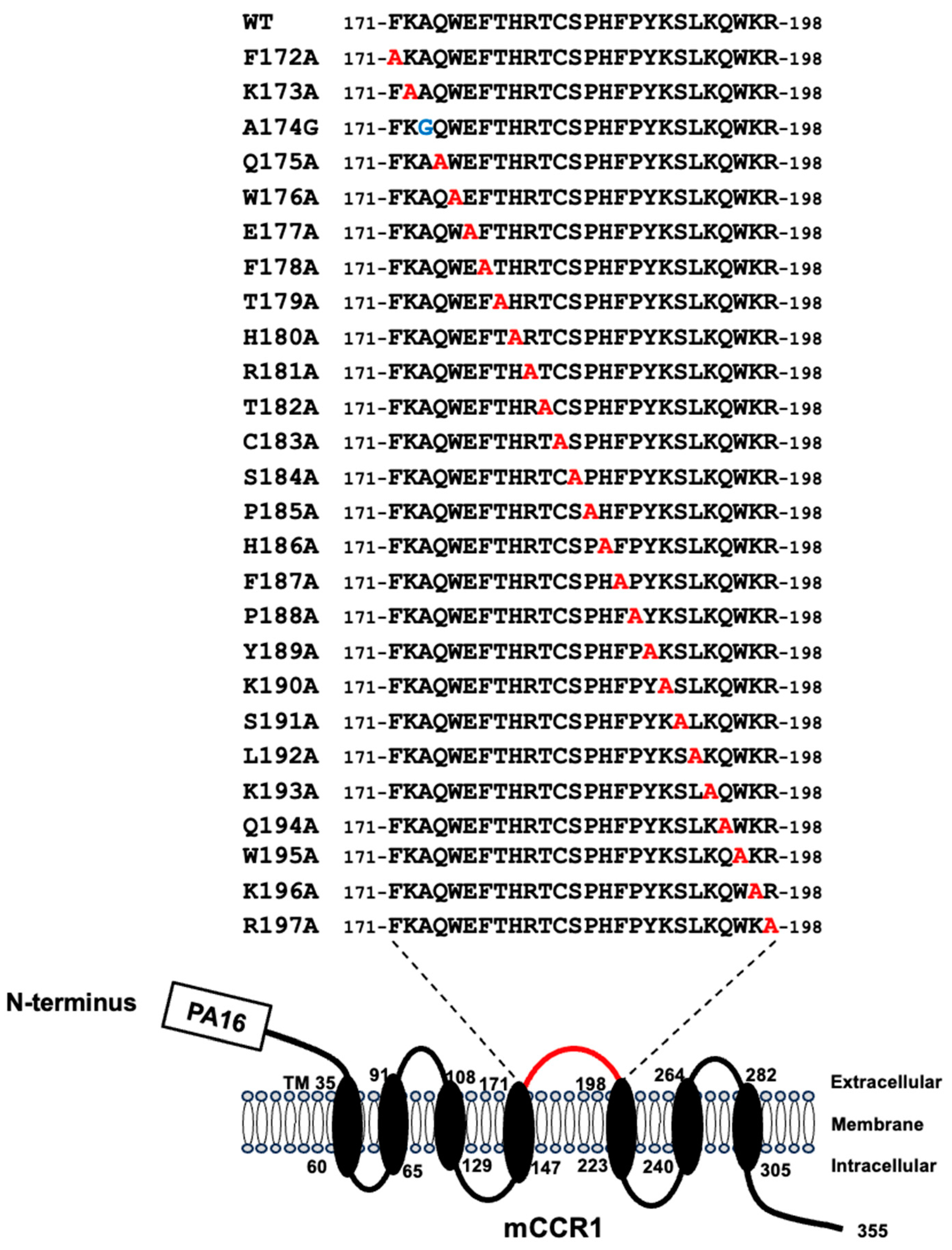
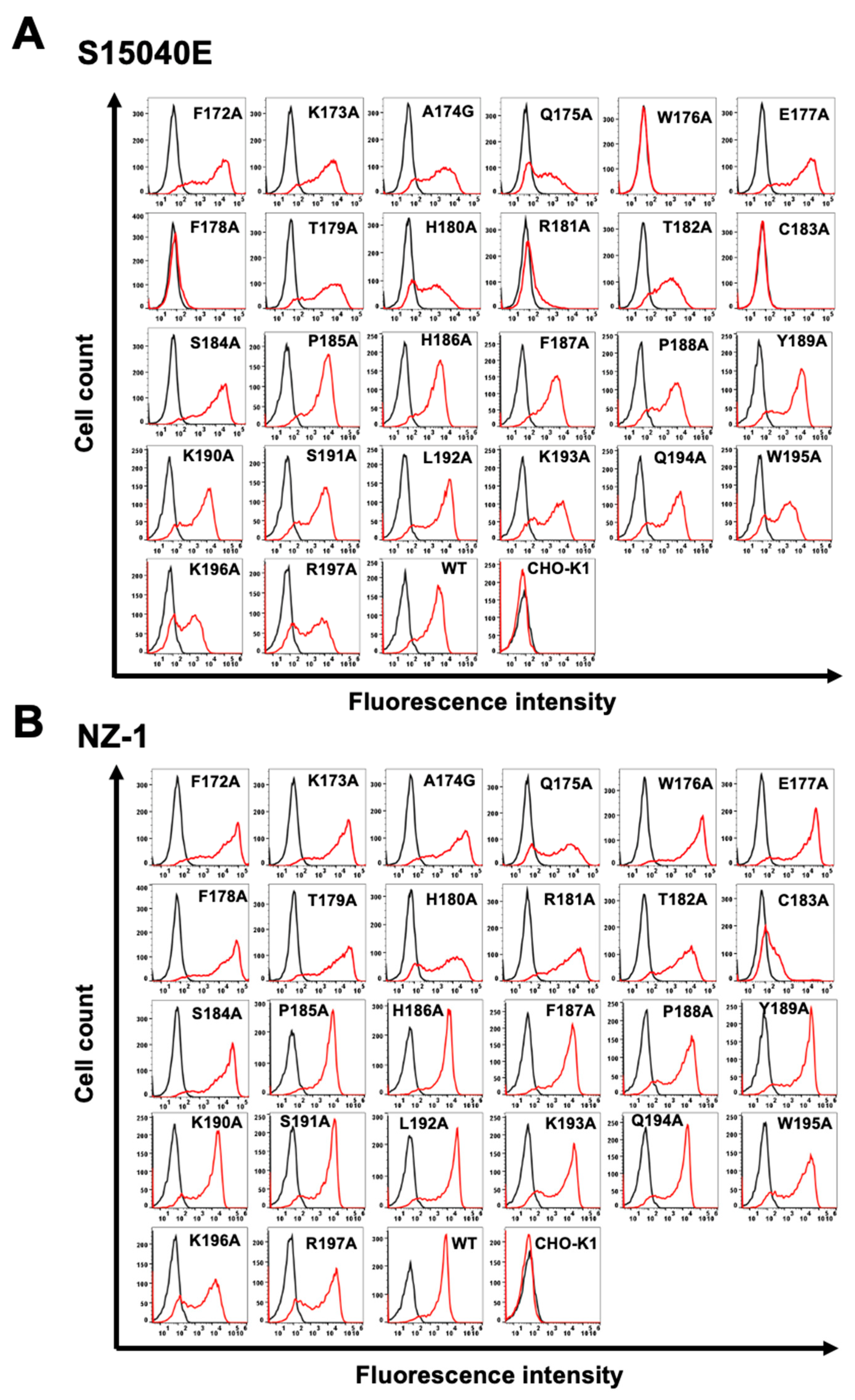
3.2. Determination of the S15040E Epitope by Flow Cytometry Using Alanine Scanning
4. Discussion
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Griffith, J.W.; Sokol, C.L.; Luster, A.D. Chemokines and chemokine receptors: positioning cells for host defense and immunity. Annu Rev Immunol 2014, 32, 659–702. [Google Scholar] [CrossRef] [PubMed]
- Stone, M.J.; Hayward, J.A.; Huang, C.; Z, E.H.; Sanchez, J. Mechanisms of Regulation of the Chemokine-Receptor Network. Int J Mol Sci 2017, 18. [Google Scholar] [CrossRef]
- Defea, K. Beta-arrestins and heterotrimeric G-proteins: collaborators and competitors in signal transduction. Br J Pharmacol, S: (Suppl 1).
- Weis, W.I.; Kobilka, B.K. The Molecular Basis of G Protein-Coupled Receptor Activation. Annu Rev Biochem 2018, 87, 897–919. [Google Scholar] [CrossRef]
- Tian, Q.; Yan, Z.; Guo, Y.; Chen, Z.; Li, M. Inflammatory Role of CCR1 in the Central Nervous System. Neuroimmunomodulation 2024, 31, 173–182. [Google Scholar] [CrossRef]
- Scholten, D.J.; Canals, M.; Maussang, D.; et al. Pharmacological modulation of chemokine receptor function. Br J Pharmacol 2012, 165, 1617–1643. [Google Scholar] [CrossRef] [PubMed]
- Murphy, P.M.; Baggiolini, M.; Charo, I.F.; et al. International union of pharmacology. XXII. Nomenclature for chemokine receptors. Pharmacol Rev 2000, 52, 145–176. [Google Scholar] [CrossRef]
- Schall, T.J.; Proudfoot, A.E. Overcoming hurdles in developing successful drugs targeting chemokine receptors. Nat Rev Immunol 2011, 11, 355–363. [Google Scholar] [CrossRef]
- Shao, Z.; Tan, Y.; Shen, Q.; et al. Molecular insights into ligand recognition and activation of chemokine receptors CCR2 and CCR3. Cell Discov 2022, 8, 44. [Google Scholar] [CrossRef] [PubMed]
- Isaikina, P.; Tsai, C.J.; Dietz, N.; et al. Structural basis of the activation of the CC chemokine receptor 5 by a chemokine agonist. Sci Adv 2021, 7. [Google Scholar] [CrossRef]
- Zheng, Y.; Qin, L.; Zacarías, N.V.; et al. Structure of CC chemokine receptor 2 with orthosteric and allosteric antagonists. Nature 2016, 540, 458–461. [Google Scholar] [CrossRef]
- Tan, Q.; Zhu, Y.; Li, J.; et al. Structure of the CCR5 chemokine receptor-HIV entry inhibitor maraviroc complex. Science 2013, 341, 1387–1390. [Google Scholar] [CrossRef] [PubMed]
- Shao, Z.; Shen, Q.; Yao, B.; et al. Identification and mechanism of G protein-biased ligands for chemokine receptor CCR1. Nat Chem Biol 2022, 18, 264–271. [Google Scholar] [CrossRef]
- Wasilko, D.J.; Johnson, Z.L.; Ammirati, M.; et al. Structural basis for chemokine receptor CCR6 activation by the endogenous protein ligand CCL20. Nat Commun 2020, 11, 3031. [Google Scholar] [CrossRef] [PubMed]
- Jaeger, K.; Bruenle, S.; Weinert, T.; et al. Structural Basis for Allosteric Ligand Recognition in the Human CC Chemokine Receptor 7. Cell 2019, 178, 1222–1230.e1210. [Google Scholar] [CrossRef]
- Oswald, C.; Rappas, M.; Kean, J.; et al. Intracellular allosteric antagonism of the CCR9 receptor. Nature 2016, 540, 462–465. [Google Scholar] [CrossRef] [PubMed]
- Sun, D.; Sun, Y.; Janezic, E.; et al. Structural basis of antibody inhibition and chemokine activation of the human CC chemokine receptor 8. Nat Commun 2023, 14, 7940. [Google Scholar] [CrossRef]
- Di Pilato, M.; Kfuri-Rubens, R.; Pruessmann, J.N.; et al. CXCR6 positions cytotoxic T cells to receive critical survival signals in the tumor microenvironment. Cell 2021, 184, 4512–4530.e4522. [Google Scholar] [CrossRef]
- Du, X.; Li, F.; Zhang, C.; et al. Eosinophil-derived chemokine (hCCL15/23, mCCL6) interacts with CCR1 to promote eosinophilic airway inflammation. Signal Transduct Target Ther 2021, 6, 91. [Google Scholar] [CrossRef]
- Niu, B.; Tian, T.; Wang, L.; et al. CCL9/CCR1 axis-driven chemotactic nanovesicles for attenuating metastasis of SMAD4-deficient colorectal cancer by trapping TGF-β. Acta Pharm Sin B 2024, 14, 3711–3729. [Google Scholar] [CrossRef]
- Ouchida, T.; Isoda, Y.; Nakamura, T.; et al. Establishment of a Novel Anti-Mouse CCR1 Monoclonal Antibody C(1)Mab-6. Monoclon Antib Immunodiagn Immunother 2024, 43, 67–74. [Google Scholar] [CrossRef]
- Suzuki, H.; Tanaka, T.; Li, G.; et al. Development of a Sensitive Anti-Mouse CCR5 Monoclonal Antibody for Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024, 43, 96–100. [Google Scholar] [CrossRef]
- Kato, Y.; Kaneko, M.K.; Kuno, A.; et al. Inhibition of tumor cell-induced platelet aggregation using a novel anti-podoplanin antibody reacting with its platelet-aggregation-stimulating domain. Biochem Biophys Res Commun 2006, 349, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Kobayashi, H.; Suzuki, H.; Tanaka, T.; Kaneko, M.K.; Kato, Y. Epitope Mapping of an Anti-Mouse CCR8 Monoclonal Antibody C(8)Mab-2 Using Flow Cytometry. Monoclon Antib Immunodiagn Immunother 2024, 43, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Larsen, O.; van der Velden, W.J.C.; Mavri, M.; et al. Identification of a conserved chemokine receptor motif that enables ligand discrimination. Sci Signal 2022, 15, eabg7042. [Google Scholar] [CrossRef]
- Jørgensen, A.S.; Larsen, O.; Uetz-von Allmen, E.; et al. Biased Signaling of CCL21 and CCL19 Does Not Rely on N-Terminal Differences, but Markedly on the Chemokine Core Domains and Extracellular Loop 2 of CCR7. Front Immunol 2019, 10, 2156. [Google Scholar] [CrossRef]
- Barington, L.; Rummel, P.C.; Lückmann, M.; et al. Role of Conserved Disulfide Bridges and Aromatic Residues in Extracellular Loop 2 of Chemokine Receptor CCR8 for Chemokine and Small Molecule Binding. J Biol Chem 2016, 291, 16208–16220. [Google Scholar] [CrossRef] [PubMed]
- Rummel, P.C.; Thiele, S.; Hansen, L.S.; et al. Extracellular disulfide bridges serve different purposes in two homologous chemokine receptors, CCR1 and CCR5. Mol Pharmacol 2013, 84, 335–345. [Google Scholar] [CrossRef]
- Blanpain, C.; Doranz, B.J.; Bondue, A.; et al. The core domain of chemokines binds CCR5 extracellular domains while their amino terminus interacts with the transmembrane helix bundle. J Biol Chem 2003, 278, 5179–5187. [Google Scholar] [CrossRef]
- Smith, J.S.; Lefkowitz, R.J.; Rajagopal, S. Biased signalling: from simple switches to allosteric microprocessors. Nat Rev Drug Discov 2018, 17, 243–260. [Google Scholar] [CrossRef]
- Barnes, P.J. Chemokine receptor CCR1: new target for asthma therapy. Trends Pharmacol Sci 2022, 43, 539–541. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



