Submitted:
14 April 2025
Posted:
15 April 2025
You are already at the latest version
Abstract
Keywords:
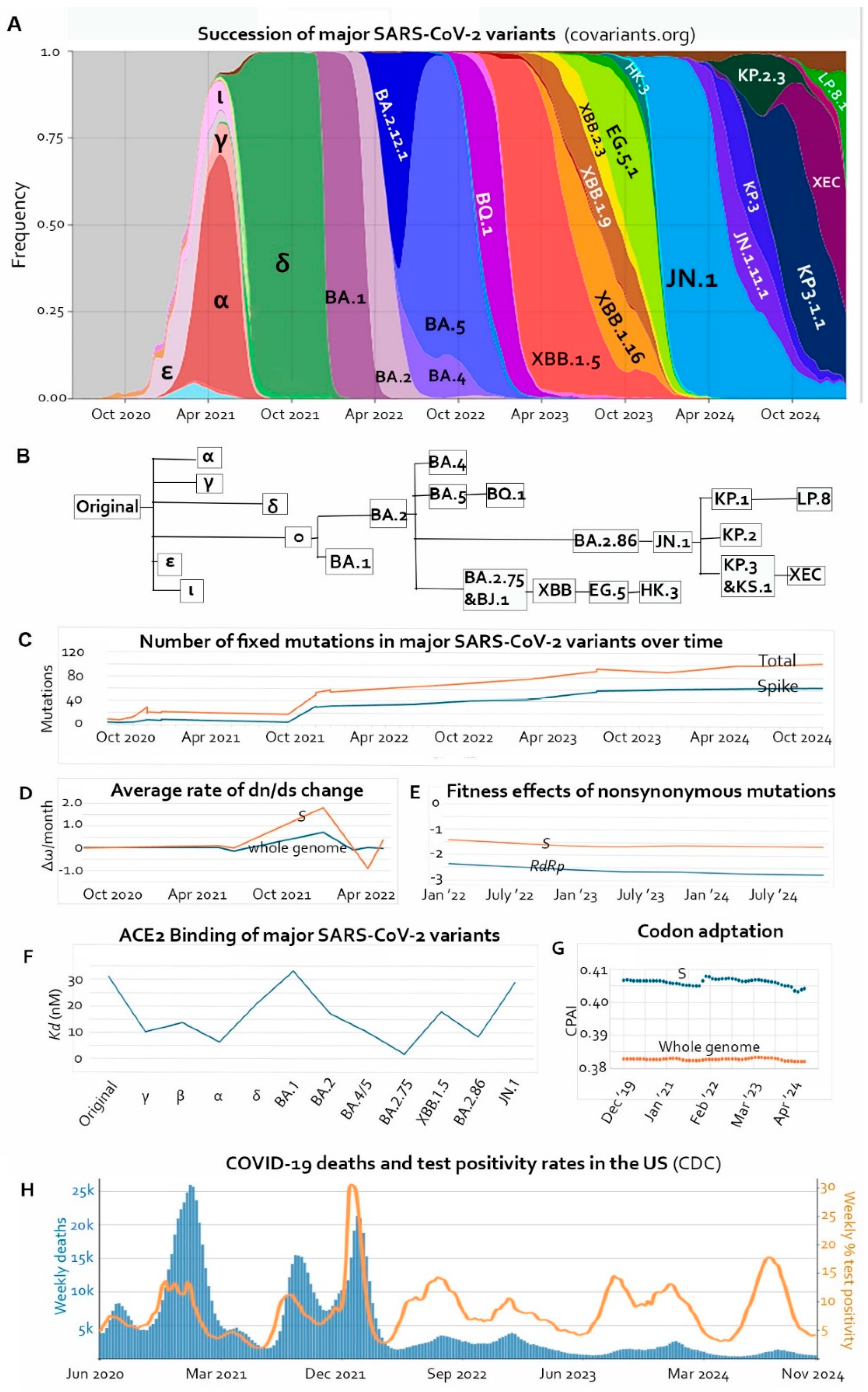
Introduction
1. Accumulation of Fixed Mutations Is Slowing Down
1.1. Cooperative Emergence of Multiple Affinity-Enhancing Mutations Followed by Immune Escape Mutations
1.2. Convergence, Flip, and Reversion
1.3. Natural Restrictions on the Improvement of Receptor-Binding Affinity
2. Deleterious Mutations Lead to Degeneration and Attenuation
2.1. Declining Mutational Fitness Effects
2.2. Declining Codon Adaptation Index in the Human Host
2.3. Phenotypical Attenuation
Conclusion
Acknowledgments
Conflict of Interest
References
- Carossino M, Izadmehr S, Trujillo JD, Gaudreault NN, Dittmar W, Morozov I, et al. ACE2 and TMPRSS2 distribution in the respiratory tract of different animal species and its correlation with SARS-CoV-2 tissue tropism. Microbiol Spectr 2024;12(2):e0327023. [CrossRef]
- Zahradník J, Marciano S, Shemesh M, Zoler E, Harari D, Chiaravalli J, et al. SARS-CoV-2 variant prediction and antiviral drug design are enabled by RBD in vitro evolution. Nat Microbiol 2021;6(9):1188-1198. [CrossRef]
- De Maio N, Walker CR, Turakhia Y, Lanfear R, Corbett-Detig R, Goldman N. Mutation Rates and Selection on Synonymous Mutations in SARS-CoV-2. Genome Biol Evol 2021; 13(5):evab087. [CrossRef]
- Colson P, Chaudet H, Delerce J, Pontarotti P, Levasseur A, Fantini J, et al. Role of SARS-CoV-2 mutations in the evolution of the COVID-19 pandemic. J Infect 2024;88(5):106150. [CrossRef]
- Simmonds P. C→U transition biases in SARS-CoV-2: still rampant 4 years from the start of the COVID-19 pandemic. mBio. 2024;15(12):e0249324. [CrossRef]
- Bloom JD, Neher RA. Fitness effects of mutations to SARS-CoV-2 proteins. Virus Evol 2023;9(2):vead055. Erratum in: Virus Evol. 2024 Mar 26;10(1):veae026. [CrossRef]
- Lippi G, Henry BM. The landscape of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) genomic mutations. J Lab Precis Med 2022; 7:10. [CrossRef]
- Maiti AK. Progressive evolutionary dynamics of gene-specific ω led to the emergence of novel SARS-CoV-2 strains having super-Infectivity and virulence with vaccine neutralization. Int J Mol Sci 2024; 25(12):6306. [CrossRef]
- Lan J, Ge J, Yu J, Shan S, Zhou H, Fan S, et al. Structure of the SARS-CoV-2 spike receptor-binding domain bound to the ACE2 receptor. Nature 2020;581(7807):215-220. [CrossRef]
- Gangavarapu K, Latif AA, Mullen JL, Alkuzweny M, Hufbauer E, Tsueng G, et al. Outbreak.info genomic reports: scalable and dynamic surveillance of SARS-CoV-2 variants and mutations. Nat Methods 2023. [CrossRef]
- Wang X, Hu M, Liu B, Xu H, Jin Y, Wang B, Zhao Y, Wu J, Yue J, Ren H. Evaluating the effect of SARS-CoV-2 spike mutations with a linear doubly robust learner. Front Cell Infect Microbiol. 2023 Apr 19;13:1161445. [CrossRef] [PubMed]
- Dejnirattisai W, Huo J, Zhou D, Zahradník J, Supasa P, Liu C, et al. SARS-CoV-2 Omicron-B.1.1.529 leads to widespread escape from neutralizing antibody responses. Cell 2022;185(3):467-484.e15. [CrossRef]
- Ito J, Suzuki R, Uriu K, Itakura Y, Zahradnik J, Kimura KT, et al. Convergent evolution of SARS-CoV-2 Omicron subvariants leading to the emergence of BQ.1.1 variant. Nat Commun 2023;14(1):2671. [CrossRef]
- Liu H, Wei P, Kappler JW, Marrack P, Zhang G. SARS-CoV-2 Variants of concern and variants of interest receptor binding domain mutations and virus infectivity. Front Immunol 2022;13:825256. [CrossRef]
- Wang Q, Mellis IA, Ho J, Bowen A, Kowalski-Dobson T, Valdez R, et al. Recurrent SARS-CoV-2 spike mutations confer growth advantages to select JN.1 sublineages. Emerg Microbes Infect 2024;13(1):2402880. [CrossRef]
- Jian F, Feng L, Yang S, Yu Y, Wang L, Song W, et al. Convergent evolution of SARS-CoV-2 XBB lineages on receptor-binding domain 455-456 synergistically enhances antibody evasion and ACE2 binding. PLoS Pathog 2023;19(12):e1011868. [CrossRef]
- Taylor AL, Starr TN. Deep mutational scanning of SARS-CoV-2 Omicron BA.2.86 and epistatic emergence of the KP.3 variant. Virus Evol 2024;10(1):veae067. [CrossRef]
- Starr TN, Greaney AJ, Hannon WW, Loes AN, Hauser K, Dillen JR, et al. Shifting mutational constraints in the SARS-CoV-2 receptor-binding domain during viral evolution. Science. 2022 Jul 22;377(6604):420-424. [CrossRef]
- Liu C, Zhou D, Dijokaite-Guraliuc A, Supasa P, Duyvesteyn HME, Ginn HM, et al. A structure-function analysis shows SARS-CoV-2 BA.2.86 balances antibody escape and ACE2 affinity. Cell Rep Med 2024;5(5):101553. [CrossRef]
- Yang H, Guo H, Wang A, Cao L, Fan Q, Jiang J, et al. Structural basis for the evolution and antibody evasion of SARS-CoV-2 BA.2.86 and JN.1 subvariants. Nat Commun 2024;15(1):7715. [CrossRef]
- Yang S, Yu Y, Xu Y, Jian F, Song W, Yisimayi A, et al. Fast evolution of SARS-CoV-2 BA.2.86 to JN.1 under heavy immune pressure. Lancet Infect Dis. 2024 Feb;24(2):e70-e72. Erratum in: Lancet Infect Dis. 2024 Mar;24(3):e156. [CrossRef]
- Tamura T, Mizuma K, Nasser H, Deguchi S, Padilla-Blanco M, et al. Virological characteristics of the SARS-CoV-2 BA.2.86 variant. Cell Host Microbe. 2024;32(2):170-180.e12. [CrossRef]
- CDC: An official website of the United States government [internet]. Atlanta: Center of Disease Control and Prevention; 2024. Update on SARS-CoV-2 Variant BA.2.86 Being Tracked by CDC; 2024 Nov 27 [Cited 2025 Apr 3]. Available from: https://www.cdc.gov/ncird/whats-new/covid-19-variant-update-2023-11-27.html.
- Liu Y, Zhao X, Shi J, Wang Y, Liu H, Hu YF, et al. Lineage-specific pathogenicity, immune evasion, and virological features of SARS-CoV-2 BA.2.86/JN.1 and EG.5.1/HK.3. Nat Commun 2024;15(1):8728. [CrossRef]
- Liu J, Yu Y, Jian F, Yang S, Song W, Wang P, et al. Enhanced immune evasion of SARS-CoV-2 variants KP.3.1.1 and XEC through N-terminal domain mutations. Lancet Infect Dis 2025;25(1):e6-e7. [CrossRef]
- Li P, Faraone JN, Hsu CC, Chamblee M, Liu Y, Zheng Y-M, et al. Neutralization and spike stability of JN.1-derived LB.1, KP.2.3, KP.3, and KP.3.1.1 subvariants. mBio. 2025:e0046425. [CrossRef]
- Chakraborty C, Bhattacharya M. FLip mutations (L455F + F456L) in newly emerging VOI, JN.1: Its antibody and immune escape. Int Immunopharmacol. 2024;133:112146. [CrossRef]
- Philip AM, Ahmed WS, Biswas KH. Reversal of the unique Q493R mutation increases the affinity of Omicron S1-RBD for ACE2. Comput Struct Biotechnol J 2023;21:1966-1977. [CrossRef]
- Mannar D, Saville JW, Poloni C, Zhu X, Bezeruk A, Tidey K, et al. Altered receptor binding, antibody evasion and retention of T cell recognition by the SARS-CoV-2 XBB.1.5 spike protein. Nat Commun 2024;15(1):1854. [CrossRef]
- Focosi D, Spezia PG, Maggi F. Fixation and reversion of mutations in the receptor-binding domain of SARS-CoV-2 spike protein. Diagn Microbiol Infect Dis 2024;108(2):116104. [CrossRef]
- [3[1]] Sugano A, Murakami J, Kataguchi H, Ohta M, Someya Y, Kimura S, et al. In silico binding affinity of the spike protein with ACE2 and the relative evolutionary distance of S gene may be potential factors rapidly obtained for the initial risk of SARS-CoV-2. Microbial Risk Analysis 2023; 25: 100278. [CrossRef]
- Korber B, Fischer WM, Gnanakaran S, Yoon H, Theiler J, Abfalterer W, et al. Tracking Changes in SARS-CoV-2 Spike: Evidence that D614G Increases Infectivity of the COVID-19 Virus. Cell. 2020 Aug 20;182(4):812-827.e19. [CrossRef]
- Zhang L, Jackson CB, Mou H, Ojha A, Peng H, Quinlan BD, et al. SARS-CoV-2 spike-protein D614G mutation increases virion spike density and infectivity. Nat Commun 2020;11(1):6013. [CrossRef]
- Zhang J, Cai Y, Xiao T, Lu J, Peng H, Sterling SM, et al. Structural impact on SARS-CoV-2 spike protein by D614G substitution. Science 2021;372(6541):525-530. [CrossRef]
- Liu J, Yu Y, Yang S, Jian F, Song W, Yu L, Shao F, Cao Y. Virological and antigenic characteristics of SARS-CoV-2 variants LF.7.2.1, NP.1, and LP.8.1. Lancet Infect Dis 2025;25(3):e128-e130. [CrossRef]
- [36 Li Q, Wu J, Nie J, Zhang L, Hao H, Liu S, et al. The Impact of Mutations in SARS-CoV-2 Spike on Viral Infectivity and Antigenicity. Cell 2020;182(5):1284-1294.e9. [CrossRef]
- Sanjuán R, Domingo-Calap P. Genetic Diversity and Evolution of Viral Populations. Encyclopedia of Virology 2021:53–61.
- Liu Y. Attenuation and Degeneration of SARS-CoV-2 Despite Adaptive Evolution. Cureus 2023;15(1):e33316. [CrossRef]
- Venkataraman S, Prasad BVLS, Selvarajan R. RNA Dependent RNA Polymerases: Insights from Structure, Function and Evolution. Viruses 2018;10(2):76. [CrossRef]
- Focosi D, Spezia PG, Maggi F. Subsequent Waves of Convergent Evolution in SARS-CoV-2 Genes and Proteins. Vaccines (Basel) 2024;12(8):887. [CrossRef]
- [4[1]] Huang W, Guo Y, Li N, Feng Y, Xiao L. Codon usage analysis of zoonotic coronaviruses reveals lower adaptation to humans by SARS-CoV-2. Infect Genet Evol 2021;89:104736. [CrossRef]
- Posani E, Dilucca M, Forcelloni S, Pavlopoulou A, Georgakilas AG, Giansanti A. Temporal evolution and adaptation of SARS-CoV-2 codon usage. Front Biosci (Landmark Ed) 2022;27(1):13. [CrossRef]
- Mogro EG, Bottero D, Lozano MJ. Analysis of SARS-CoV-2 synonymous codon usage evolution throughout the COVID-19 pandemic. Virology 2022;568:56-71. [CrossRef]
- Fumagalli SE, Padhiar NH, Meyer D, Katneni U, Bar H, DiCuccio M, et al. Analysis of 3.5 million SARS-CoV-2 sequences reveals unique mutational trends with consistent nucleotide and codon frequencies. Virol J 2023;20(1):31. [CrossRef]
- Wu X, Shan KJ, Zan F, Tang X, Qian Z, Lu J. Optimization and Deoptimization of Codons in SARS-CoV-2 and Related Implications for Vaccine Development. Adv Sci (Weinh). 2023;10(23):e2205445. [CrossRef]
- Padhiar NH, Ghazanchyan T, Fumagalli SE, DiCuccio M, Cohen G, Ginzburg A, et al. SARS-CoV-2 CoCoPUTs: analyzing GISAID and NCBI data to obtain codon statistics, mutations, and free energy over a multiyear period. Virus Evol 2025;11(1):veae115. [CrossRef]
- Wang R, Chen J, Gao K, Hozumi Y, Yin C, Wei GW. Analysis of SARS-CoV-2 mutations in the United States suggests presence of four substrains and novel variants. Commun Biol 2021;4(1):228. [CrossRef]
- Wu H, Xing N, Meng K, Fu B, Xue W, Dong P, et al. Nucleocapsid mutations R203K/G204R increase the infectivity, fitness, and virulence of SARS-CoV-2. Cell Host Microbe 2021;29(12):1788-1801.e6. [CrossRef]
- Mears HV, Young GR, Sanderson T, Harvey R, Barrett-Rodger J, Penn R, et al. Emergence of SARS-CoV-2 subgenomic RNAs that enhance viral fitness and immune evasion. PLoS Biol 2025;23(1):e3002982. [CrossRef]
- Mautner L, Hoyos M, Dangel A, Berger C, Ehrhardt A, Baiker A. Replication kinetics and infectivity of SARS-CoV-2 variants of concern in common cell culture models. Virol J 2022;19(1):76. [CrossRef]
- Baud D, Qi X, Nielsen-Saines K, Musso D, Pomar L, Favre G. Real estimates of mortality following COVID-19 infection. Lancet Infect Dis. 2020 Jul;20(7):773. [CrossRef]
- CDC: An official website of the United States government [internet]. Atlanta: Center of Disease Control and Prevention; 2024. Trends in United States COVID-19 Deaths, Emergency Department (ED) Visits, and Test Positivity by Geographic Area; 2024 Apr 7 [Cited 2025 Apr 7]. Available from: https://covid.cdc.gov/covid-data-tracker/#trends_weeklydeaths_testpositivity_00.
- CDC: An official website of the United States government [internet]. Atlanta: Center of Disease Control and Prevention; 2024. COVID data tracker weekly review; 2022 Jul 29 [Cited 2025 Apr 10]. Available from: https://archive.cdc.gov/www_cdc_gov/coronavirus/2019-ncov/covid-data/covidview/past-reports/07292022.html?utm_source=chatgpt.com.
- Clarke KEN, Jones JM, Deng Y, Nycz E, Lee A, Iachan R, et al. Seroprevalence of Infection-Induced SARS-CoV-2 Antibodies - United States, September 2021-February 2022. MMWR Morb Mortal Wkly Re. 2022;71(17):606-608.
- Xia Q, Yang Y, Wang F, Huang Z, Qiu W, Mao A. Case fatality rates of COVID-19 during epidemic periods of variants of concern: A meta-analysis by continents. Int J Infect Dis 2024;141:106950. [CrossRef]
- Ong SWX, Chiew CJ, Ang LW, Mak TM, Cui L, Toh MPHS, et al. Clinical and Virological Features of Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) Variants of Concern: A Retrospective Cohort Study Comparing B.1.1.7 (Alpha), B.1.351 (Beta), and B.1.617.2 (Delta). Clin Infect Dis. 2022;75(1):e1128-e1136. [CrossRef]
- Cohen J. Covid’s cold cousins. Science 2024 Jan 12;383(6679):141-145.
| Mutations | Pre-Alpha | Alpha | Beta | Gamma | Delta | Lambda | Omicron |
|---|---|---|---|---|---|---|---|
| HV69-70del | B.1.258, B.1.375 | x | BA.1 | ||||
| Y144del | x | XBB.1.5 | |||||
| R346 | BQ.1.1, XBB.1.5 | ||||||
| K417N/T | x | x | x | ||||
| G446S | BA.1, BA.2.75 | ||||||
| L452R/Q | x | x | BA.4/5 | ||||
| F456L | Multiple XBB and JN.1 sublineages | ||||||
| T478K | x | x | |||||
| E484A/K | x | x | x | ||||
| F490S | x | XBB.1.5 | |||||
| N501Y | x | x | x | x | |||
| D614G | A and B.1 | ||||||
| P681R/H | x | x | x |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




