1. Introduction
Diels-Alder (DA) reactions [
1], which belong to the general class of cycloaddition reactions, are among the most useful synthetic reactions in Organic Chemistry as they allow the construction of six-membered carbocyclic compounds with high regio- and stereoselectivity in a single synthetic step. [
2,
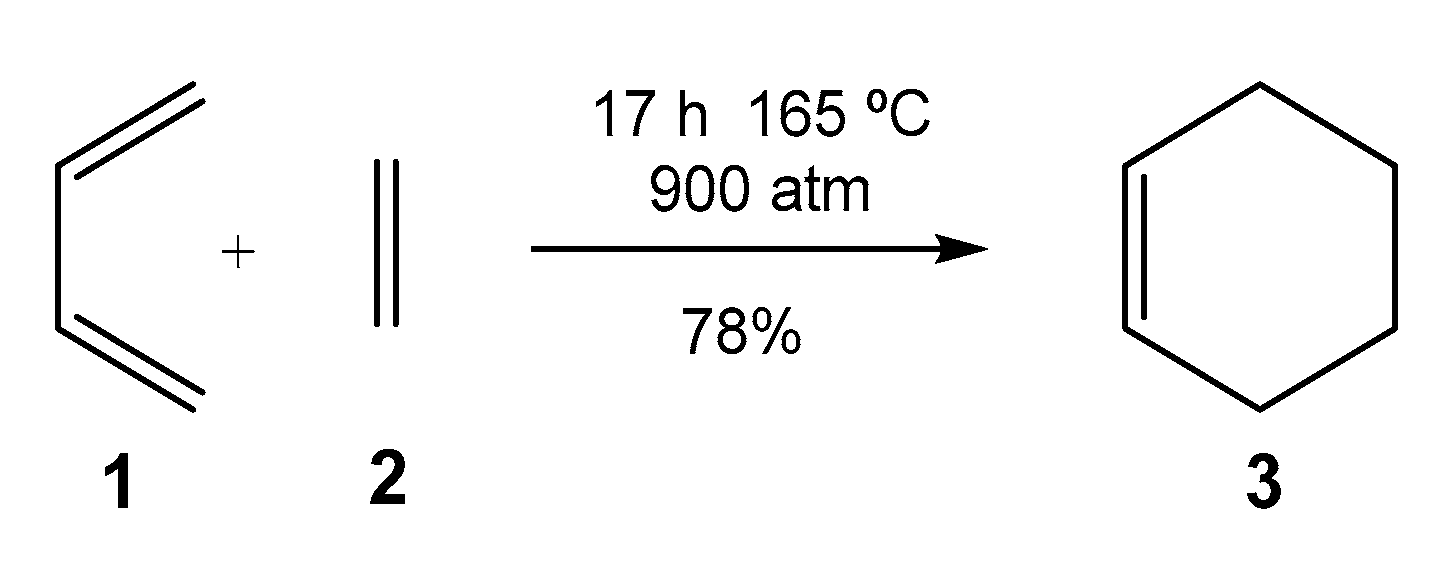
3]. They have been extensively studied both experimentally and theoretically. By varying the nature of the diene and ethylene, many different types of six-membered cyclic structures can be synthesized, but not all possibilities are experimentally feasible. For instance, the DA reaction between butadiene
1 and ethylene
2, selected as the prototype of these cycloaddition reactions [
4,
5], must be forced to take place, for example, after 17 h at 165 °C and 900 atm, cyclohexene
3 is obtained with a 78% yield (see
Scheme 1) [
6].
This DA reaction has a high activation energy of 27.5 kcal·mol
-1, being strongly exothermic by 40 kcal/mol [
6]. The unfavorable negative activation entropy associated with this bimolecular process, -40.6 cal·mol
-1·K
-1 [
7], together with the high temperature required for the reaction, 165 ºC, raises the activation Gibbs free energy to 42.6 kcal·mol
-1.
There are two ways to reduce this highly unfavorable Gibbs free energy of activation: i) by diminishing the unfavorable activation enthalpy associated with the formation of the two new C-C single bonds; and/or ii) by decreasing the unfavorable activation entropy associated with bimolecular processes.
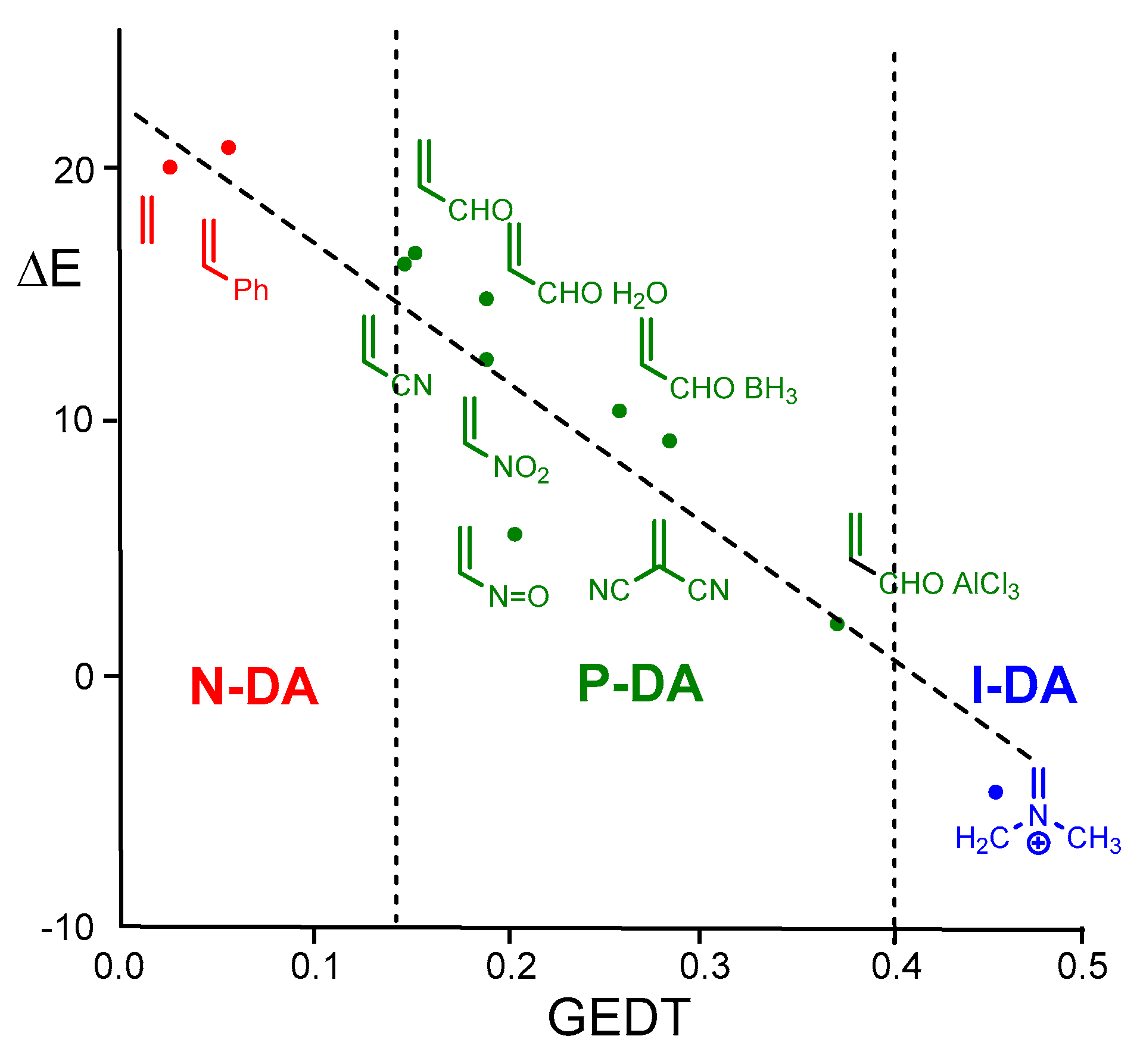
An exhaustive theoretical study of experimentally performed DA reactions allowed the establishment of the definitive role of the global electron density transfer [
8] (GEDT) occurring at the TSs in the decrease of the activation energies of DA reactions [
9,
10]. This discovery enabled the establishment in 2009 of the mechanism for polar DA (P-DA) reactions [
11], in which the favorable nucleophilic/electrophilic interactions taking place at the transition state structures (TSs) are responsible for the feasibility of the experimental DA reaction [
12]. The strong linear correlation found between GEDT and the activation barriers for the DA reactions of Cp
4 with a series of 12 substituted ethylenes of increasing electrophilic character, including an iminium cation, allowed the classification of experimental DA reactions into non-polar DA (N-DA) reactions, which do not take place easily under experimental conditions, polar DA (P-DA) reactions, and ionic DA (I-DA) reactions, in which one of the two reagents is an ionic species (see
Figure 1) [
11]. Notably, I-DA reactions represent the extreme case of P-DA reactions.
On the other hand, the unfavorable entropic costs associated with these bimolecular processes may be reduced through an intramolecular process.
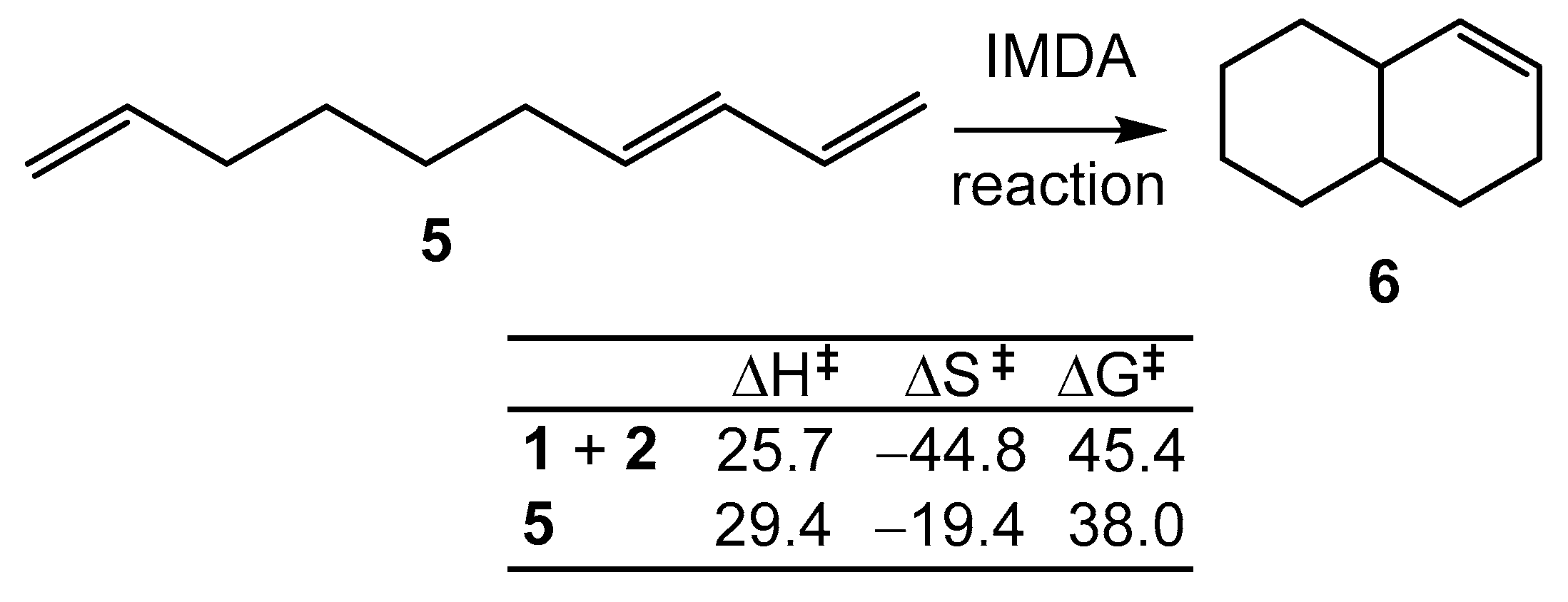
Scheme 2 shows the thermodynamic data for the N-DA reaction between butadiene
1 and ethylene
2 shown in
Scheme 1, along with those for the intramolecular Diels-Alder (IMDA) reaction of (E)-deca-1,3,9-triene (DTE)
5 [13]. The B3LYP/6-311G(d,p) activation enthalpy for the N-DA reaction of
1 with
2, ΔH
≠ = 25.7 kcal·mol
-1, and the computed activation entropy, ΔS
≠ = -44.8 cal·mol
-1·K
-1, were close to the experimental values, 27.5 kcal·mol
-1 and −40.6 cal·mol
-1·K
-1, respectively [
6,
7]. These high values raise the activation Gibbs free energy of this intermolecular N-DA reaction, computed at 165ºC, to 45.4 kcal·mol
-1 (see
Scheme 2). Despite the fact that the computed activation enthalpy associated with the IMDA reaction of
5, ΔH
≠ = 29.4 kcal·mol
-1, was found to be higher than that of the intermolecular one, the low activation entropy associated with this intramolecular process, ΔS
≠ =

19.4 cal·mol
-1·K
-1, reduces the activation Gibbs free energy to 38.0 kcal·mol
-1. This behavior explains the feasibility of non-polar IMDA reactions.
IMDA reactions allow the formation of contiguous cycles in a single synthetic step [
14] with remarkable stereoselectivity [
15,
16,
17]. These reactions are widely used as synthetic routes in the total synthesis of natural products [
15,
16,
17,
18,
19,
20].
Many theoretical studies on non-polar and polar IMDA reactions are available in the literature [
21,
22,
23,
24,
25,
26,
27,
28,
29,
30]. The ionic IMDA reaction of dieniminium
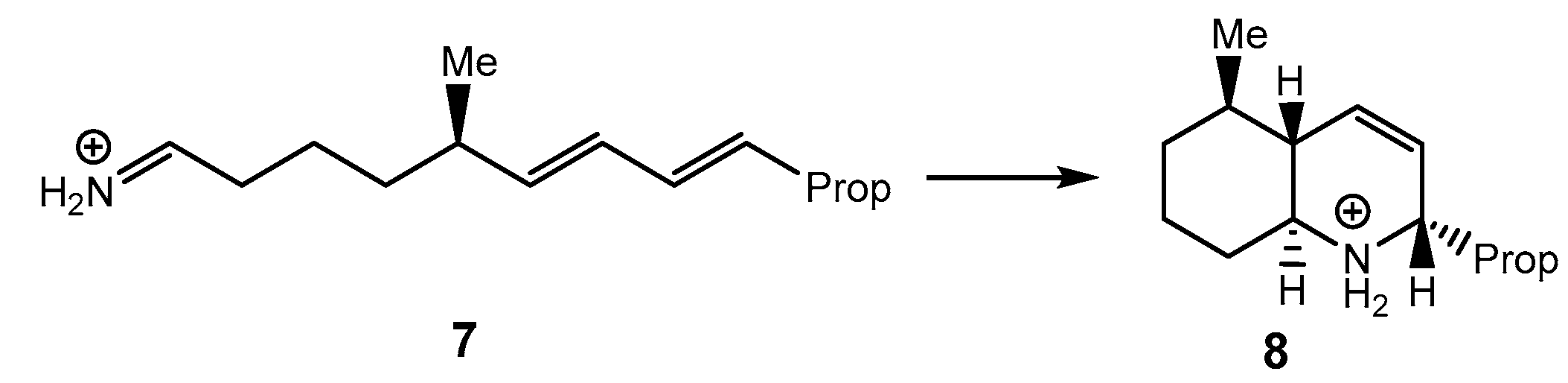
7 [13], experimentally reported by Grieco and Parke [
31], was recently studied within the Molecular Electron Density Theory [
32] (MEDT) (
Scheme 3). The activation enthalpy associated with this IMDA reaction, 8.7 kcal·mol
-1, was close to that of the I-DA reaction between butadiene
1 and ethaniminium
9, 9.3 kcal·mol
-1. However, while the intermolecular I-DA reaction presented an activation Gibbs free energy of 25.3 kcal·mol
-1, this IMDA reaction showed an activation Gibbs free energy of only 12.6 kcal·mol
-1 because of the very low activation entropy associated with the intramolecular process,

14.3 cal·mol
-1·K
-1 [
13]. This ionic IMDA reaction exhibits complete
re/
exo and
si/
endo diastereoselectivity, which is controlled by the most favorable chair conformations adopted by the –(CH
2)
4- chain linking the butadiene and iminium frameworks (see dieniminium
7 in
Scheme 3).
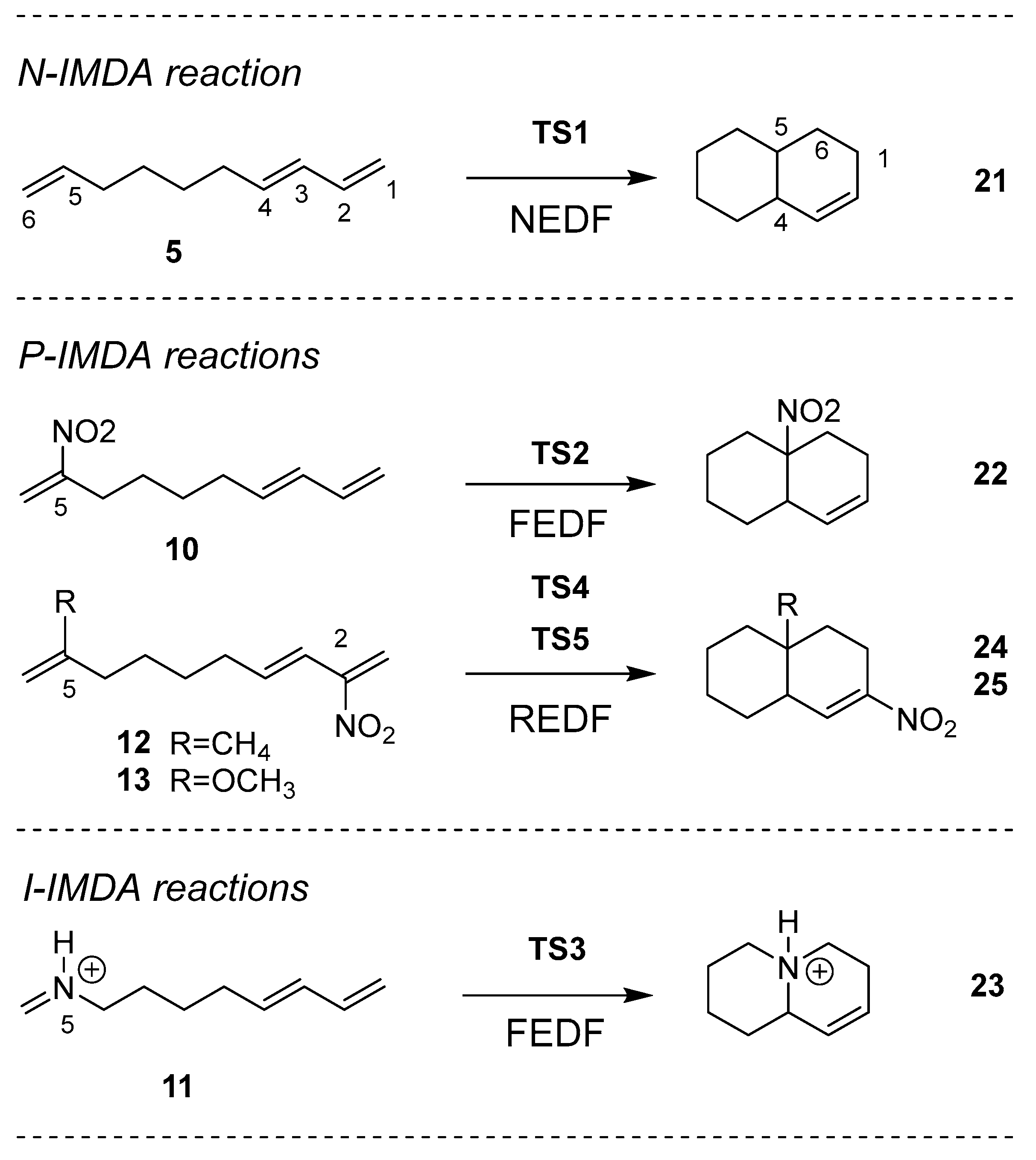
Due to the relevance of the IMDA reactions in organic synthesis, the similarities between the non-polar (N-IMDA), polar (P-IMDA) and ionic (I-IMDA) IMDA reactions of DTEs
5, 10,
12 and
13 and N-methyleneocta-5,7-dien-1-aminium (MODA)
11 shown in
Chart 1, with the N-DA, P-DA and I-DA reactions of pentadienes,
14 and
15, with the ethylenes,
16 –
20, shown in
Chart 2, are studied herein within the MEDT at the M06-2X/6-311G(d,p) computational level. The complete study of the intermolecular DA reactions between the reagents shown in
Chart 2 is reported in the Supplementary Material.
2. Results and Discussion
This MEDT study is divided into five sections: i) initially, the topological analysis of the electron localization function (ELF) of the electronic structure of the four DTEs 5, 10, 12 and MODA 11 is conducted, along with an analysis of the chemical properties of the reagent ground states (GSs); ii) second, the potential energy surfaces (PES) of the IMDA reactions of DTEs 5, 10, 12 and 13 and MODA 11 are explored; iii) third, an ELF topological analysis is achieved on the electronic structure of the five TSs involved in the IMDA reactions; iv) fourth, a comparative analysis of the kinetics of the IMDA reactions relative to the kinetics of the DA reactions is performed; and finally, v) fifth section focuses on an RIAE analysis of the DA reactions of 14 with 16 or 17, and that of 15 with 20, as well as the IMDA reactions of the DTEs 5, 10 and 13.
2.1. Study of the Electronic Structure and Chemical Properties of the DTEs 5, 10, 12 and 13 and MODA 11 in the GS
2.1.1. Study of the Electronic Structure of the DTEs 5, 10, 12 and 13 and MODA 11
Before exploring the PES of the IMDA reactions shown in
Chart 1, the electronic structures at the GS of DTEs
5,
10,
12 and
13 and MODA
11 were investigated through a topological analysis of the ELF [
33]. ELF provides a quantitative characterization of the electron density distribution in a molecule, facilitating a correlation between its electronic structure and reactivity. The attractor positions of the ELF basins, the populations of the most relevant valence basins, and the natural atomic charges of the DTEs
5,
10,
12 and
13 and MODA
11, computed at the M06-2X/6-311G(d,p) level, are shown in
Figure 2. ELF of the compounds
14 –
20, involved in the DA reaction, is analyzed in
Section 1 in the Supplementary Material.
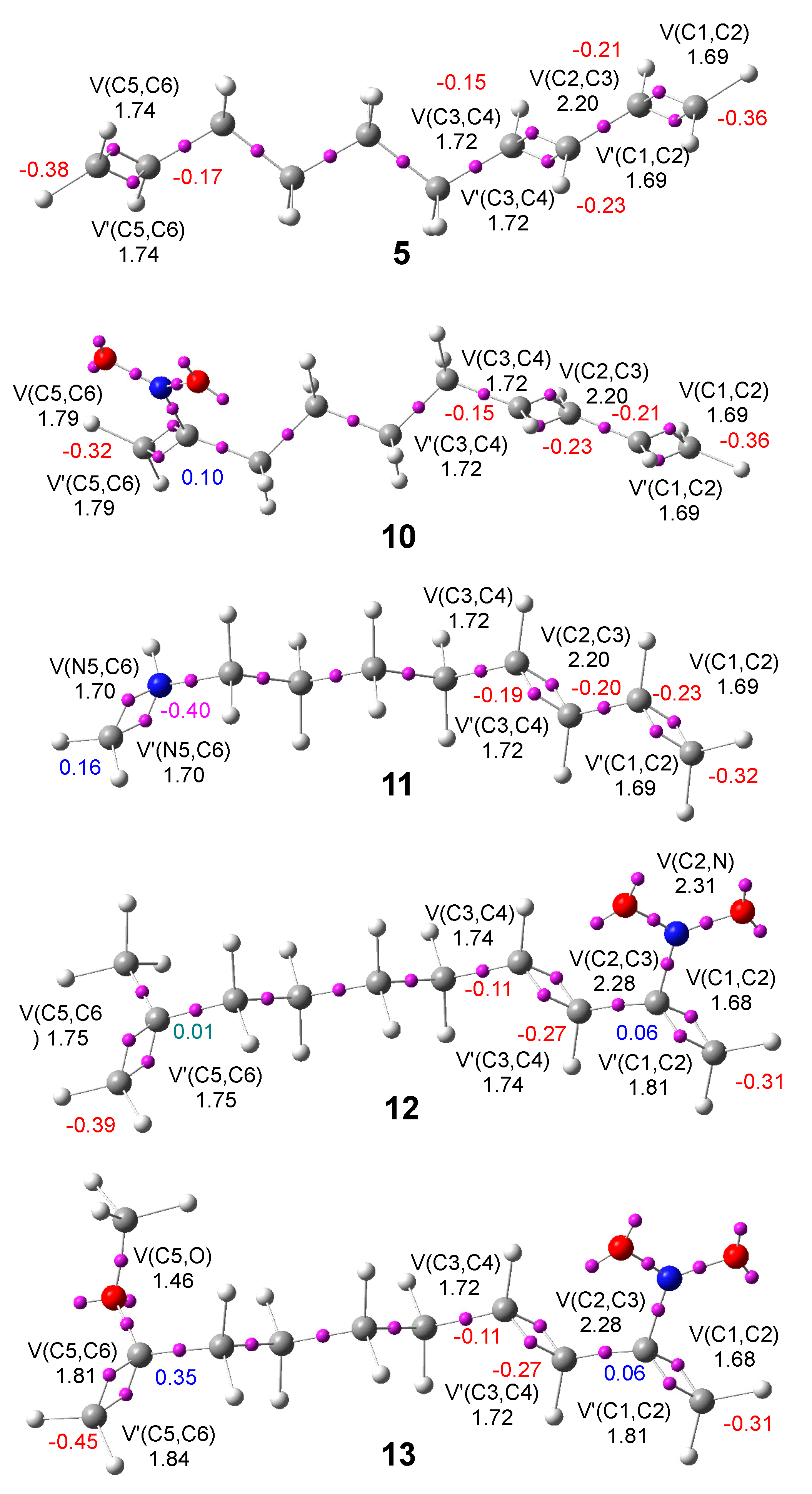
ELF topological analysis of DTE
5 shows the presence of two disynaptic basins, V(C1,C2) and V’(C1,C2), integrating a total of 3.38 e, associated with a depopulated C1-C2 double bond, one V(C2,C3) disynaptic basin integrating 2.20 e, associated with a populated C2-C3 single bond, and two disynaptic basins, V(C3,C4) and V’(C3,C4), integrating a total of 3.44 e, associated with a depopulated C3-C4 double bond, characterizing the conjugated 1,3-butadiene system of DTE
5. On the other hand, the C5-C6 bonding region is characterized by the presence of two disynaptic basins, V(C5,C6) and V’(C5,C6), integrating a total of 3.48 e, associated with a depopulated C5-C6 double bond. The ELF of the diene and ethylene frameworks of
5 is very similar to that of pentadiene
14 and propene
16, as shown in
Figure S1 in the Supplementary Material.
The C1=C2-C3=C4 frameworks of 8-nitro-deca-1,3,9-triene
10 and the MODA cation
11 have ELF topological behaviors closely resembling that of DTE
5 (See
Figure 2). The presence of the electron-withdrawing (EW) NO
2 group on the C5 carbon of
10 causes a slight increase in the population of the C5-C6 bonding region, which is characterized by the presence of two disynaptic basins, V(C5,C6) and V’(C5,C6), integrating a total of 3.58 e. On the other hand, the N5-C6 bonding region of MODA cation
11 shows only a slight depopulation of 0.18 e compared to C5-C6 region in the nitro derivative DTE
10. The ELF of the ethylene frameworks of the DTE
10 and the MODA cation
11 are very similar to those of 2-nitro-propene
17 and iminium cation
18, respectively, as shown in
Figure S1 in the Supplementary Material.
Finally, the presence of the EW NO
2 group on the C2 carbon of DTEs
12 and
13 induces negligible changes in the C1=C2-C3=C4 bonding region compared to DTE
5. Only the population of V(C2,C3) disynaptic basins are increased by 0.08 e. On the other hand, while the presence of an electron-releasing (ER) CH
3 group decreases the population of the C5-C6 bonding region in compound
12, the presence of the strong ER OCH
3 group on the C5 carbon of compound
13 increases the population of the C5-C6 bonding region by 0.17 e. The ELF of the butadiene frameworks of the DTEs
12 and
13 are very similar to that of 2-nitro-pentadiene
15, while the ethylene frameworks are very similar to those of propene
16 and 2-methoxy-propene
20, respectively (see
Figure S1 in the Supplementary Material).
Thus, the present ELF analysis of DTEs 5, 10, 12 and 13 and MODA 11 shows strong similarity between the electronic structure of the diene and ethylene frameworks of these compounds and those of compounds 14 – 20, which are involved in the selected DA reaction.
The natural atomic charges [
34,
35] of the most relevant centers of DTEs
5,
10,
12 and
13 and MODA
11 are presented in
Figure 2. In general, all carbon atoms belonging to the conjugated double bond framework are negatively charged between -0.11 and -0.45 e. The negative charge at the non-substituted carbon atoms of these DTEs depends on the number of hydrogen atoms attached to them; this behavior is caused by the higher electronegativity of the carbon atom compared to the hydrogen atom.
The presence of the NO
2, CH
3, and OCH
3 groups in DTEs
10,
12, and
13 causes a significant change in the charge on the carbon atom to which they are attached. Thus, the presence of the CH
3 groups at
12 makes the C5 carbon negligibly charged, while the presence of the NO
2 and OCH
3 groups makes the C2 or C5 carbons to which they are attached positively charged (see
Figure 2). This behavior is a consequence of the higher electronegative character of the N nitrogen or O oxygen atoms compared to the C carbon one, rather than the EW or ER character of the corresponding groups. Interestingly, the MODA cation
11, formed by protonation of the N5 nitrogen in the corresponding imine, is negatively charged by -0.40 e.
A comparative analysis of the natural atomic charges of DTEs
5,
10,
12 and
13 and MODA
11 with those of compounds
14 –
20 involved in the selected DA reaction, shows a strong similarity between them (see
Figure S1 in the Supplementary Material).
The present structural analysis of the DTEs 10, 12 and 13 shows that the inclusion of the EW or ER groups on the unsaturated frameworks of these compounds does not substantially modify the electronic structures of these DTEs relative to that of DTE 5. Only the charges of the C2 or C5 carbon atoms to which the activating groups are attached are markedly modified.
2.1.2. Analysis of the Chemical Properties of DTEs 10 ,12 and 13 and MODA 11
A useful method for comprehending reactivity in polar reactions is the analysis of the DFT-based reactivity indices at the GS of the reagents. [
36,
37,
38]. Following, the DFT-based reactivity indices of DTEs
5,
10,
12 and
13 and MODA
11 were analyzed to understand their chemical reactivity. The DFT-based reactivity indices were computed at the B3LYP/6-31G(d) computational level, as it was used to establish electrophilicity and nucleophilicity scales [
37,
38]. The reactivity indices of the cations MODA
11 and iminium
19 were computed in DMSO [
39].
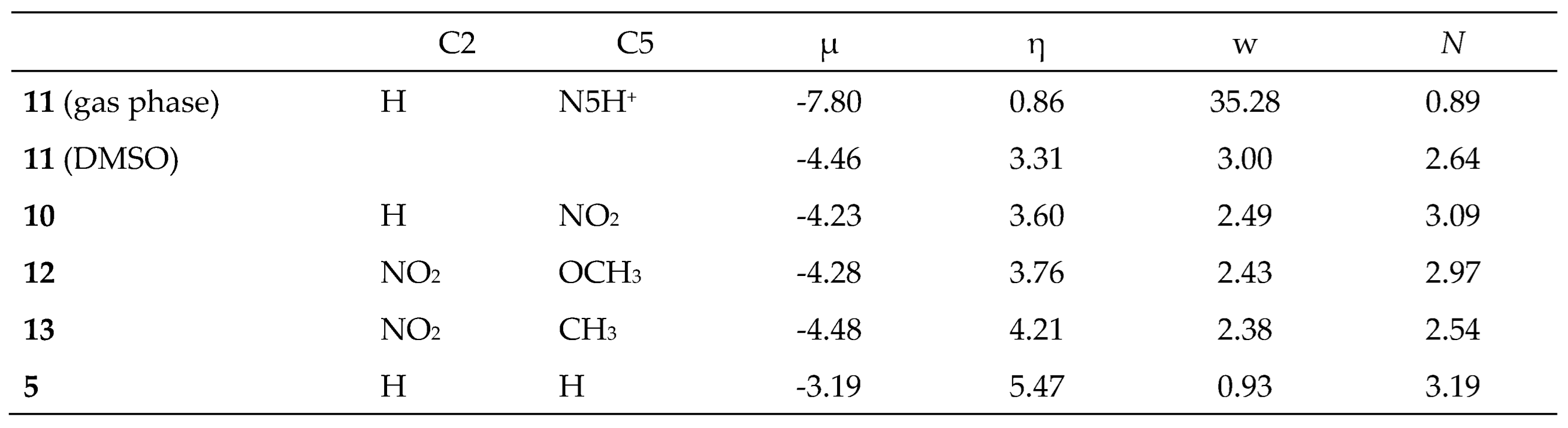
Table 1 summarizes the B3LYP/6-31G(d) global reactivity indices, including electronic chemical potential
μ, chemical hardness
η, as well as the electrophilicity ω and nucleophilicity
N indices of DTEs
5,
10,
12 and
13 and MODA
11. The reactivity indices of the butadiene and ethylene derivatives
14 –
20 are given in
Table S1 in the Supplementary Material.
Previous studies of IMDA reactions have shown that analyzing the electrophilicity ω and nucleophilicity
N indices of the GS reagents provides valuable information about the electrophilicity and nucleophilicity of the butadiene and ethylene frameworks of these complex molecules [
13].
The values of the electrophilicity [
40] w and nucleophilicity [
41]
N indices of DTE
5, 0.93 and 3.19 eV, respectively, allow its classification as a moderate electrophile and a strong nucleophile [
37,
38]. The electrophilicity w and nucleophilicity
N indices of 1,3-pentadiene
14 are 0.91 and 3.20 eV, respectively, and those of propene
16 are 0.60 and 2.32 eV (see
Table S1 in the Supplementary Material). A comparison of these values with those of DTE
5 indicates that its nucleophilicity
N index is associated with the diene framework. The very low electrophilicity w index of propene
16, w = 0.60 eV, indicates that the IMDA reaction of DTE
5 will have a non-polar character, being classified as null electron density flux (NEDF) [
42].
The values of the electrophilicity w and nucleophilicity
N indices of 9-nitro-deca-1,3,9-triene
10, 2.49 and 3.09 eV, respectively, allow its classification as a strong electrophile and a strong nucleophile. While the electrophilicity w index is closer to that of 2-nitro-propene
17, w = 2.43 eV, the nucleophilicity
N index is closer to that of 1,3-pentadiene
14 (see
Table S1 in the Supplementary Material). The analysis of the reactivity indices indicates that along the P-IMDA reaction of DTE
10, the flux of the electron density will occur from the butadiene framework to the ethylene one, in a reaction of forward electron density flux (FEDF) [
42].
The values of the electrophilicity w and nucleophilicity
N indices in DMSO of MODA cation
11, 3.00 and 2.64 eV, respectively, allow its classification as a strong electrophile and a strong nucleophile [
37,
38]. The electrophilicity w index of MODA cation
11 is closer to that of iminium cation
18, w = 2.91 eV (see
Table S1 in the Supplementary Material). Consequently, along the I-IMDA reaction of MODA cation
11, the electron density flux will take place from the butadiene framework to the protonate iminium one, in a reaction of FEDF.
The values of the electrophilicity w and nucleophilicity N indices of 2-nitro-9-methyl-deca-1,3,9-triene 12, 2.38 and 2.54 eV, respectively, allow its classification as a strong electrophile and a moderate nucleophile. The electrophilicity w index of this species is closer to that of 2-nitro-pentadiene 15, w = 2.43 eV, while the nucleophilic N index is closer to that of isobutene 19, N = 2.60 eV. Consequently, along the P-IMDA reaction of DTE 12, the electron density flux will take place from the ethylene framework to the butadiene one, in a reaction of reverse electron density flux, REDF.
The values of the electrophilicity w and nucleophilicity N indices of 2-nitro-9-methoxy-deca-1,3,9-triene 13, 2.43 and 2.97 eV, respectively, allow its classification as a strong electrophile and borderline of strong nucleophile. The nucleophilicity N index of this species is lower than that of 2-methoxy-propene 20, N = 3.24 eV. Consequently, along the P-IMDA reaction of DTE 13, the electron density flux will take place from the ethylene framework to the butadiene one, in a reaction of REDF.
In P-DA reactions involving non-symmetric reagents, the
most favorable reaction path is that involving the two-center interaction
between the most nucleophilic and the most electrophilic centers of the
reagents. In this sense, the analysis of the electrophilicand nucleophilicParr functions [43] has become a powerful tool to study
regioselectivity in P-DA and P-IMDA reactions [44].
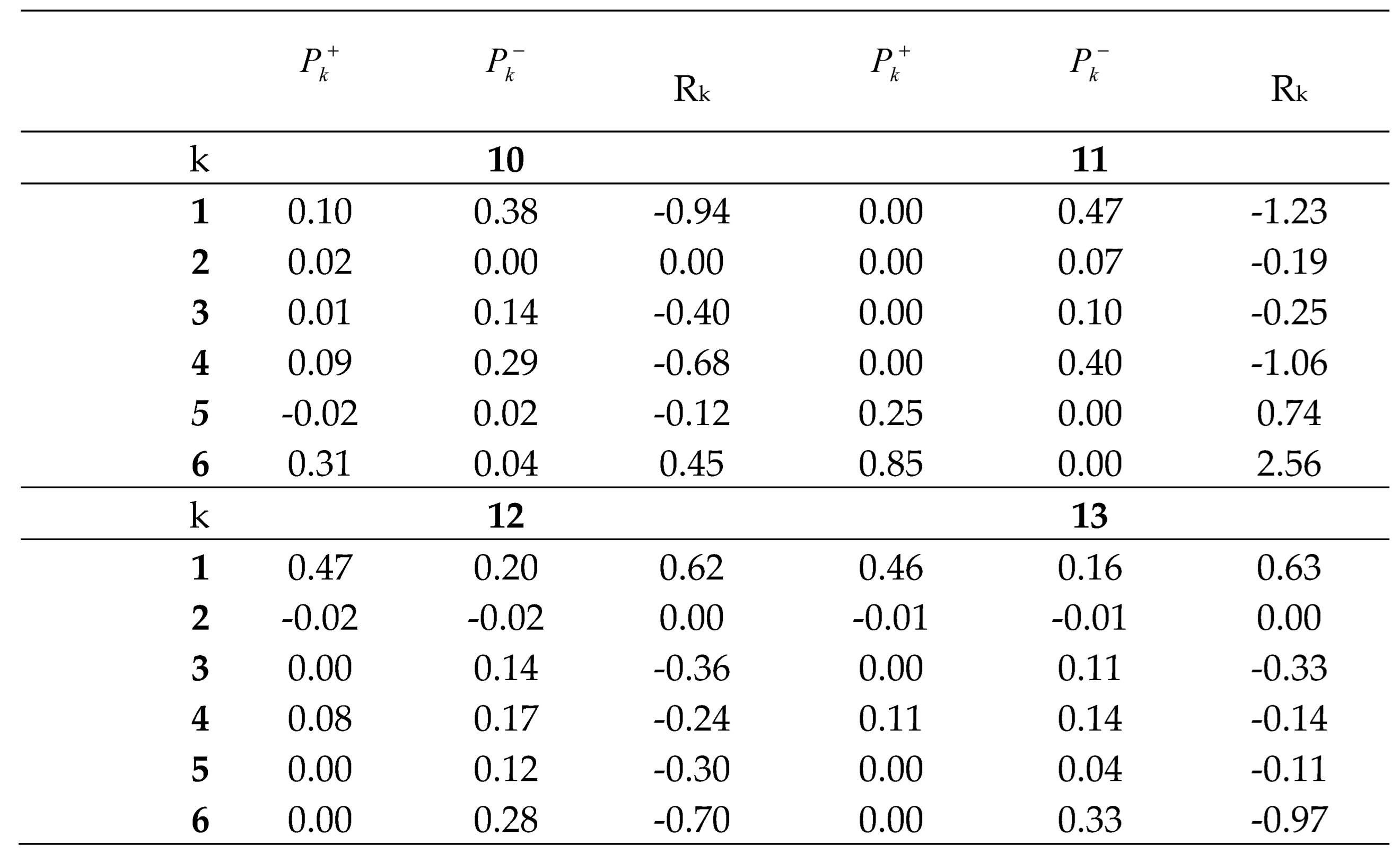
In 2012, Chattaraj et al. proposed a local reactivity
difference, R
k, index to analyze the local electrophilic and/or
nucleophilic activation within a complex organic molecule [45]. Together with the electrophilic and/or
nucleophilic behavior of the center k given by the sign, the magnitude of the R
k
index accounts for the extent of the electronic activation. The R
k
index has become a powerful tool to analyze the electrophilic and nucleophilic
center in a molecule, allowing prediction of the reactivity in an
intramolecular polar reaction [45]. The
electrophilic and
nucleophilic Parr functions,
together with the local reactivity difference R
k indices of DTEs
10,
12, and
13 and MODA
11, involved in the P-IMDA and
I-IMDA reactions, are given in
Table 2 [37].
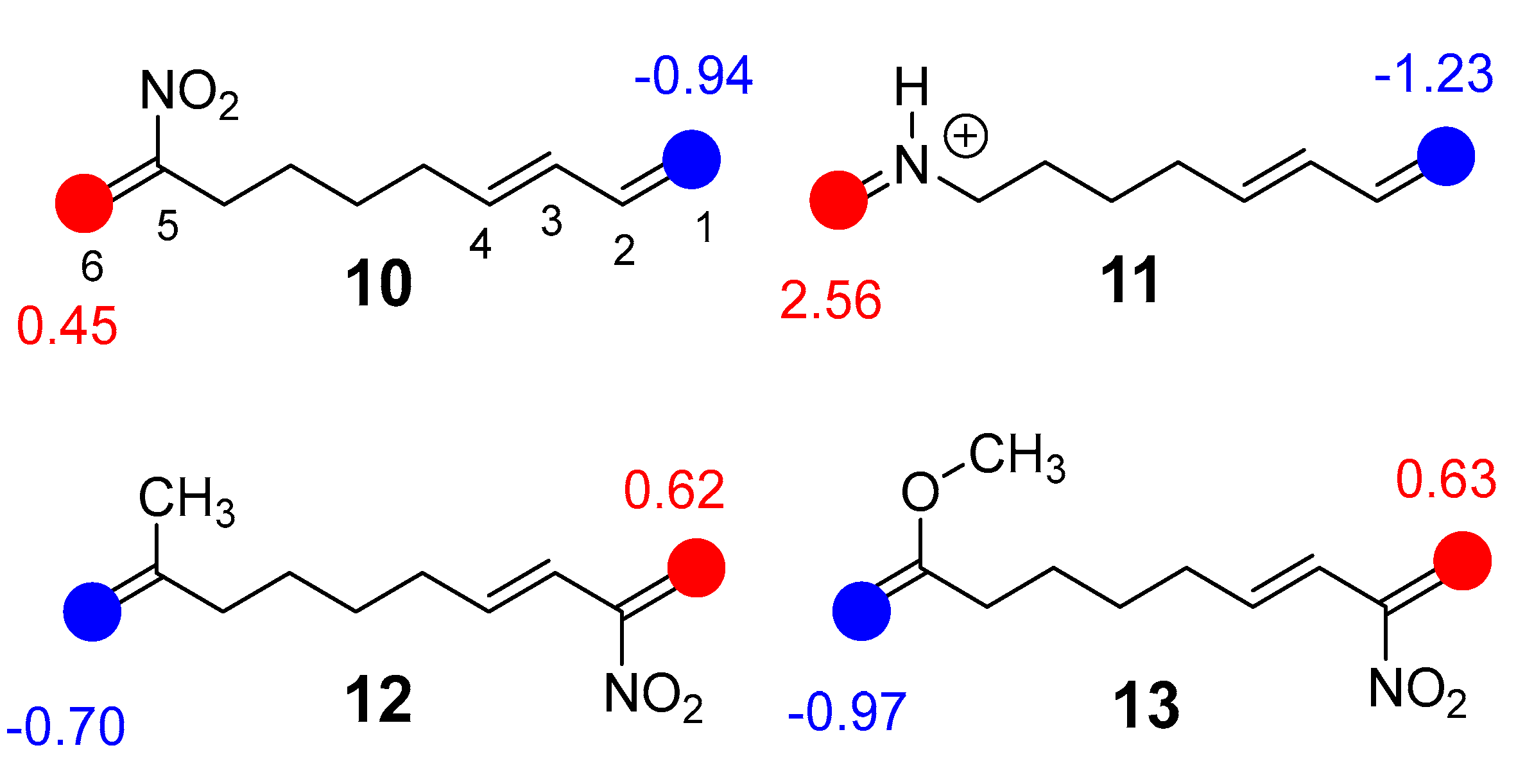
Chart 3
shows a graphical representation of the most electrophilic, lowest R
k
< 0, and nucleophilic, highest R
k > 0, center of DTEs
9
–
12.
The local electrophilic/nucleophilic activation in the butadiene/ethylene system of these DTEs depends on the position of the EW/ER substituents in these unsaturated systems. Thus, the position of the substituent in the butadiene or ethylene framework determines the formation of a mixture of regioisomeric bicyclic compounds in P-IMDA reactions. In the present MEDT study, the selected mono- or disubstitution on the C2 or C5 positions of the deca-1,3,9-triene system, as shown in
Chart 1, activate the C1 and C6 positions, respectively, allowing the most favorable electrophilic/nucleophilic interaction along these P-IMDA reactions (see later).
2.2. Study of the IMDA Reactions of DTEs 5, 10, 12 and 13 and MODA 11
Then, the PESs of the most favorable reaction paths associated with the IMDA reactions of DTEs
5,
10,
12 and
13 and MODA
11 were studied (see
Scheme 4). Along these reaction paths, the –(CH
2)
4- chain adopts a chair conformation diminishing the strain associated with these intramolecular processes [
13]. Along each IMDA reaction, one reagent, one TS, and one bicyclic compound were located and characterized, indicating that these IMDA reactions take place through a one-step mechanism. The M06-2X/6-311G(d,p) gas phase total and relative energies of the stationary points involved in the IMDA reactions of DTEs
5,
10,
12, and
13 and MODA
11 are given in
Table 3, while those involved in the DA reactions of compounds
14 –
20 are given in
Table S2 in the Supplementary Material. The total and relative electronic energies in tetrahydrofuran (THF) are given in
Table S3 in the Supplementary Material.
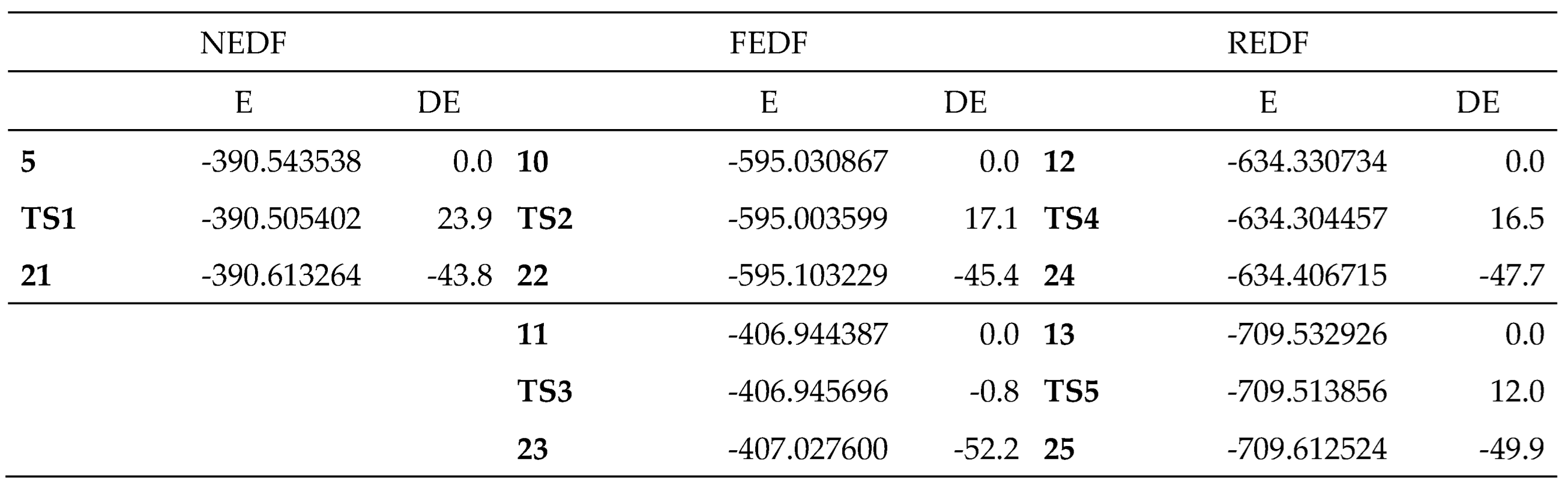
Some appealing conclusions can be obtained from the relative energies given in
Table 3: i) the five IMDA reactions are strongly exothermic ranging from –43.8 (
5) to –52.2 (
11) kcal·mol
-1. Consequently, these IMDA reactions can be considered irreversible, and kinetically controlled; ii) the relative energies of the TSs range from 23.9 kcal·mol
-1 for the N-IMDA reaction of DTE
5 to –0.8 kcal·mol
-1 for the I-IMDA reaction of MODA cation
11; iii) these relative energies indicate that the substitution on the DTE system has a greater impact on the kinetics than on the thermodynamics of the reactions; iv)
TS3 is located 0.8 kcal·mol
-1 below the extended conformation of the DTE system, which has been selected as the energy reference for these IMDA reactions. However, when the PES is calculated in THF, the relative energy becomes positive by 8.3 kcal·mol
-1 due to the strong stabilization of the MODA cation
11 (see
Table S3 in the Supplementary Material); and finally, v) in the REDF P-IMDA reactions of DTEs
12 and
13, the double substitution at the diene and ethylene frameworks is required to increase the nucleophilic character of the ethylene framework and change the nucleophilic character of the butadiene system of DTE
5 to an electrophilic one.
A comparison of the relative energies of the TSs and cycloadducts of the IMDA reactions of DTEs
5,
10,
12 and
13 and MODA
11, given in
Table 3, with those of the DA reactions of compounds
14 –
20, given in
Table S2, provides important insight into the IMDA reactions to be obtained: i) the series of five IMDA and DA reactions exhibit a similar strong exothermic character; ii) the exothermic character of the IMDA reactions is 2 kcal·mol
-1 lower than that of the DA reactions due to the presence of a lower strain associated with the formation of the bicyclic systems; iii) the relative energies of the TSs associated with the intermolecular DA reactions are between 2.9 (
5) and 9.1 (
11) kcal·mol
-1 lower than those associated with the IMDA reaction; iv) these energy differences increase as the activation energies decrease, I,e., the more polar the reactions, the greater difference, and finally v) in spite of this difference, the same trend in the kinetics of the reactions is found in both series of reactions with the substitution on the butadiene and ethylene systems influencing the reactivity.
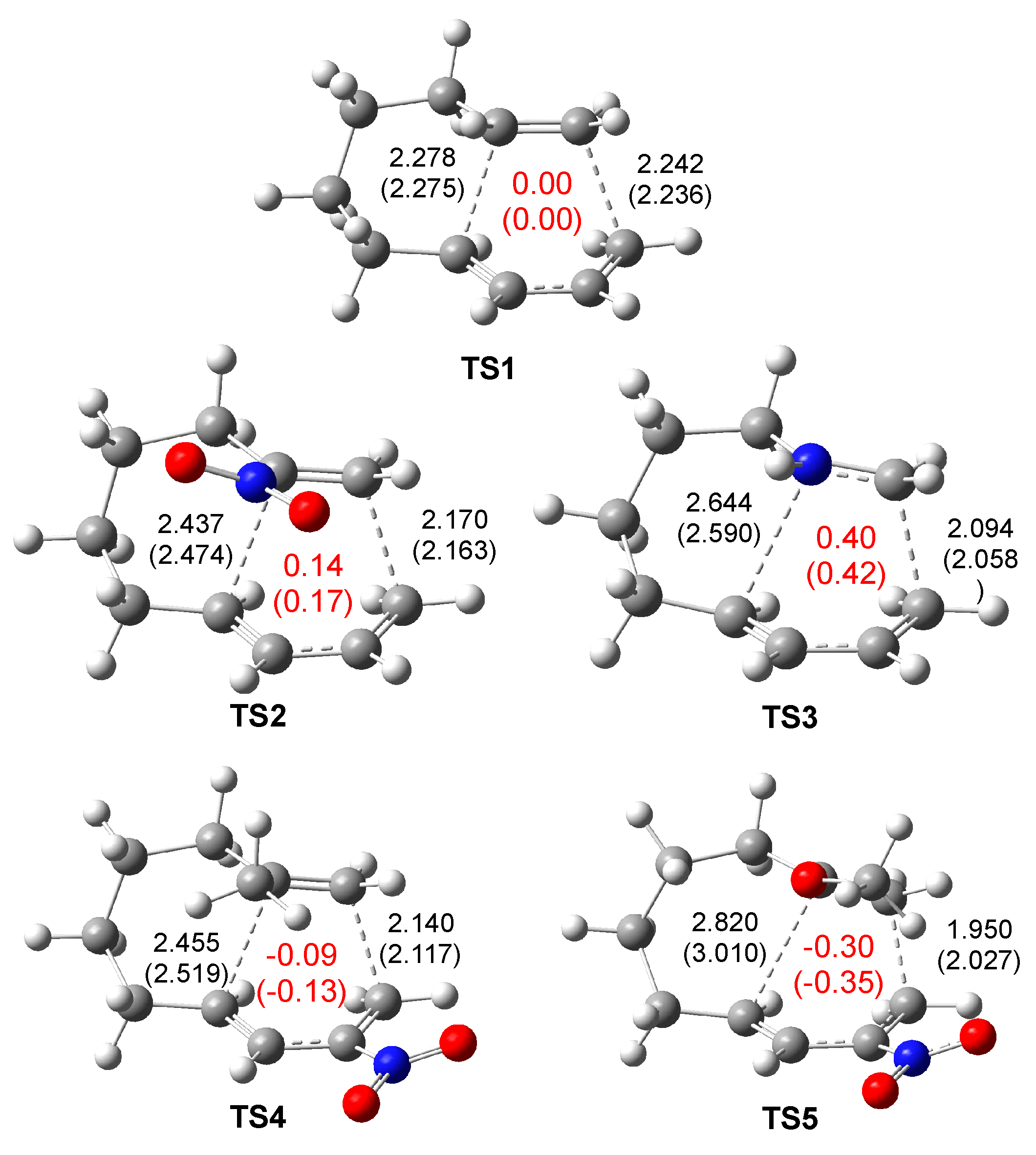
The gas phase geometries of the TSs involved in the IMDA reactions of DTEs
5,
10,
12 and
13 and MODA
11 are given in
Figure 3. In all TSs, the –(CH
2)
4- chain that links the butadiene and ethylene frameworks adopts a chair conformation, reducing the strain energy associated with these intramolecular processes. Note that at GS of the reagents, the –(CH
2)
4- chain adopts an extended
anti conformation.
The C1-C6 distances at the TSs range from 1.95 to 2.24 Å, while the C4-C5 distances are in the range from 2.28 to 2.80 Å. While TS1 corresponds to a synchronous C-C single bond formation process, the other four TSs correspond to asynchronous single bond formation processes in which the shorter distance is between the C1 and C6 carbons. This behavior agrees with the analysis of the reactivity difference Rk indices at the GS of the reagents, which indicates that the C1 and C6 carbons are the most nucleophilic/electrophilic centers of these molecules.
For the P-IMDA and I-IMDA reactions, the asynchronicity increases with the increase in the polar character of the reactions (see later). While the C1-C6 distances between the two pairs of interacting atoms decrease with the increase in nucleophilic/electrophilic interactions, the C4-C5 distances increase, i.e., the more polar the reaction, the more asynchronous the TS.
The inclusion of solvent effects of THF in geometrical optimizations does not significantly modify the gas phase geometries (see
Figure 3).
Analysis of the GEDT [
8] at the TSs involved in these IMDA reactions enables the quantification of the polar character of these cycloaddition reactions. The GEDT values calculated at the five TSs are presented in
Figure 3. GEDT values below 0.05 e indicate non-polar processes, while values above 0.20 e denote highly polar processes. Additionally, the sign of the GEDT at the TSs clearly distinguishes the polar DA reactions as FEDF, with GEDT > +0.05 e, or REDF, with GEDT <

0.05 e. Non-polar 32CA reactions, characterized by a negligible GEDT ( ≤ |0.05| e), are classified as NEDF [
42].
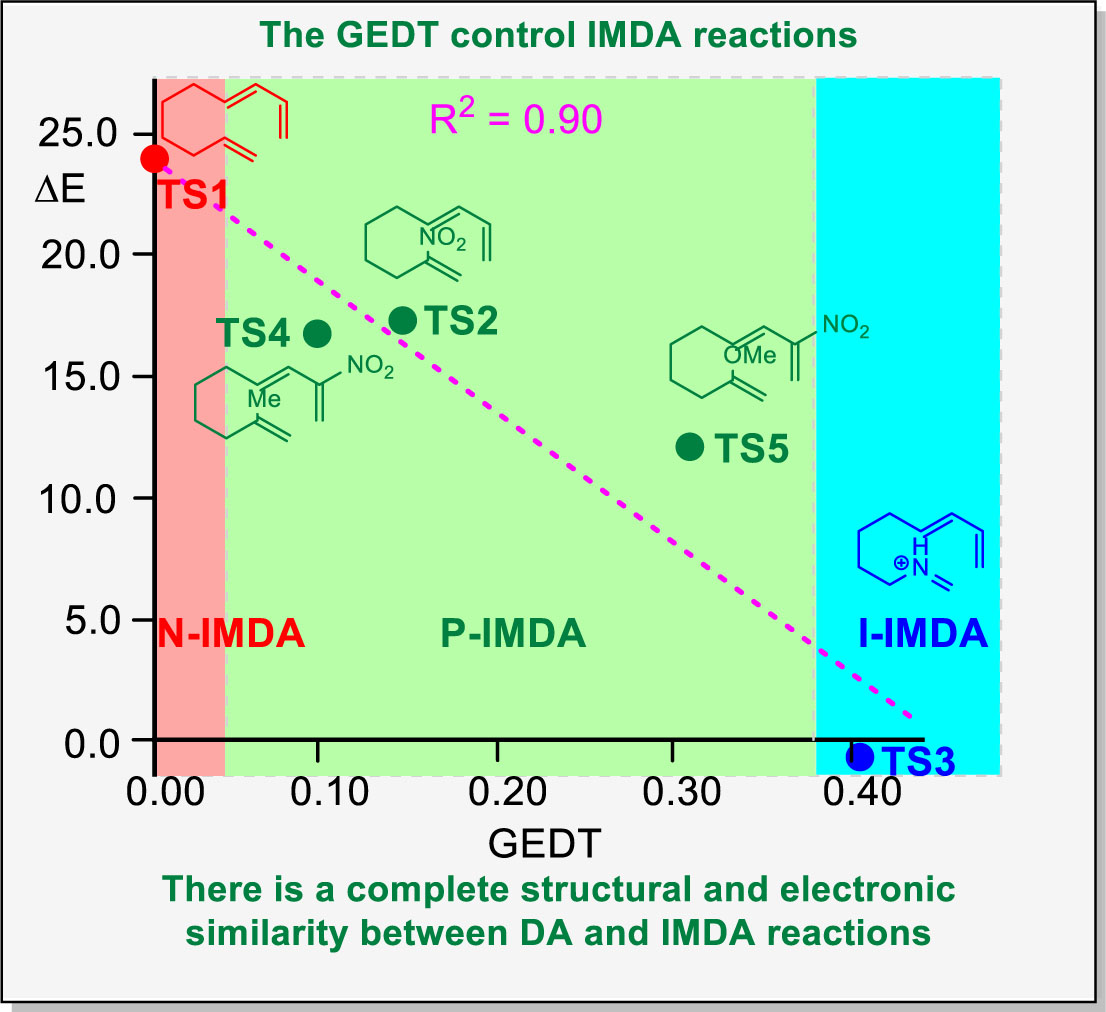
While the GEDT value at
TS1, 0.00 e, indicates that it is associated with an N-IMDA reaction classified as NEDF,
TS3, associated with an I-IMDA reaction, displays the maximum GEDT value, 0.40 e. The positive GEDT values found at
TS2 and
TS3 classify these P-IMDA reactions as FEDF. Finally, the negative GEDT values found at
TS4 and
TS5 allow them to be classified as REDF.
TS5, with GEDT = -0.30 e, corresponds to the most asynchronous TS of the three polar ones. The inclusion of THF solvent effects slightly increases the GEDT at the polar TSs, favoring the electron density transfer process (see
Figure S2 in the Supplementary Material).
Recently, the crucial role of GEDT in TSs of P-DA reactions in reducing activation energies has been established [
12], a behavior that constitutes the foundation of P-DA reactions, as first proposed in 2009 [
11].
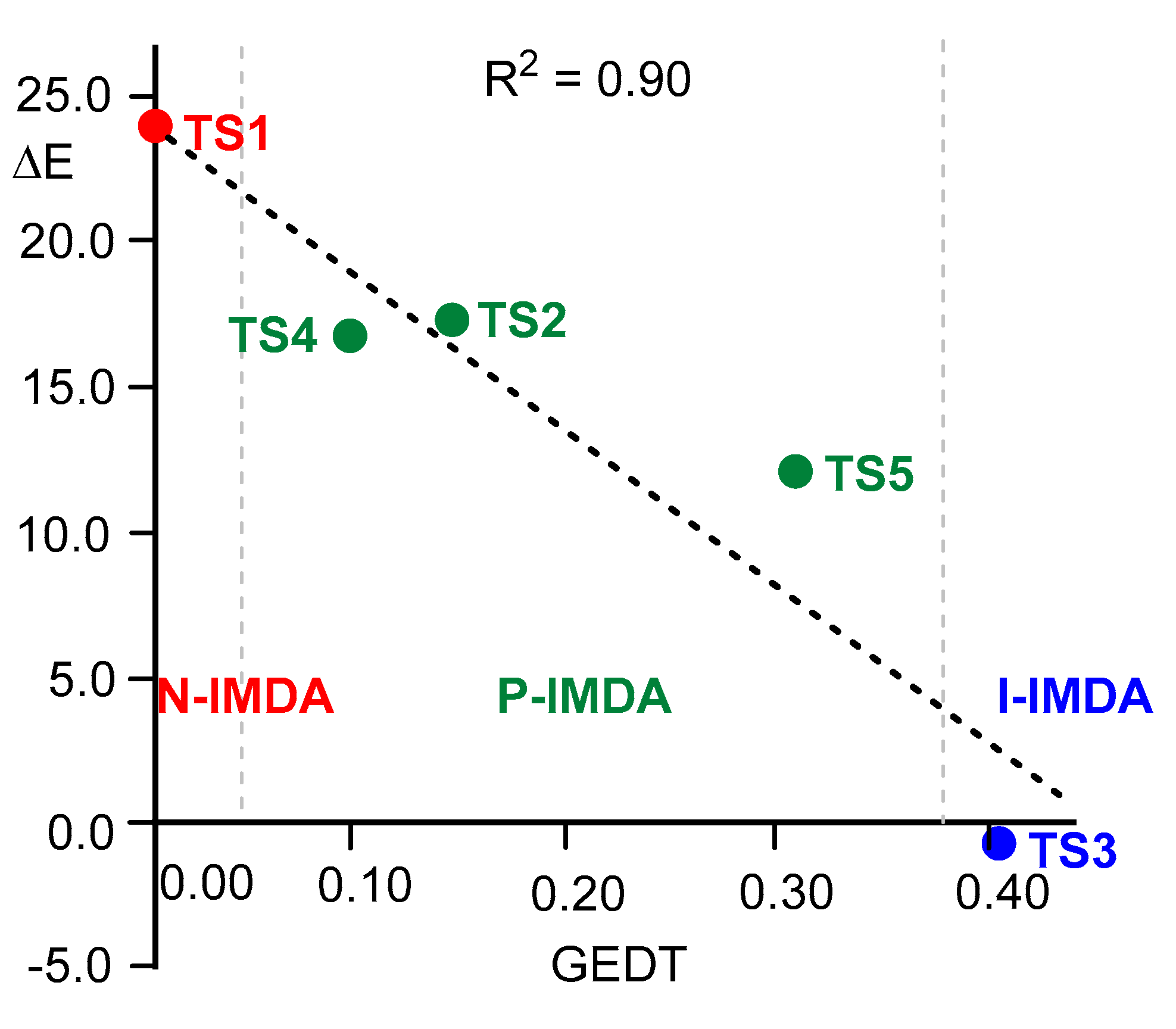
Figure 4 shows the plot of the relative energies of the TSs of five IMDA reactions
vs the GEDT computed at the corresponding TSs. As can be seen, a good linear correlation, R
2 = 0.90, is obtained: the higher the GEDT, the faster the reaction. This graph, which is similar to that obtained for intermolecular DA reactions (see
Figure 1), clearly differentiates N-IMDA, in red, P-IMDA, in green, and I-IMDA, in blue, reactions (see
Figure 4).
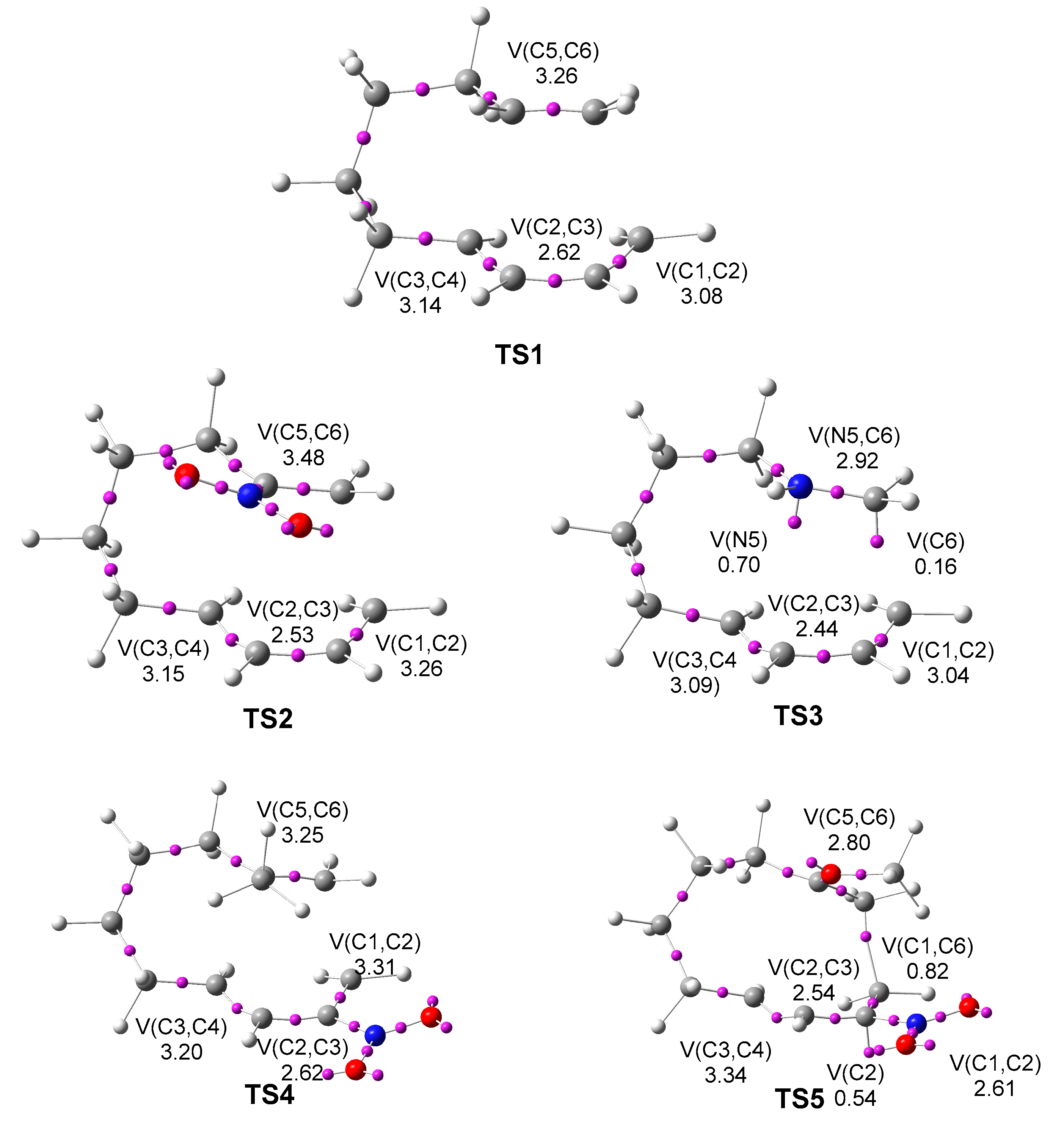
2.3. ELF Topological Analysis of the Electronic Structure of TS1 – TS5
Next, an ELF topological analysis was subsequently conducted on the
TS1 –
TS5 associated with the IMDA reactions. The attractor positions of the ELF basins and key valence basin populations for the five TSs are given in
Figure 5.
The five TSs show the depopulation of the C1-C2, C3-C4 and C5-C6 bonding regions, which are characterized by the presence of only one V(C1,C2), V(C3,C4) and V(C5,C6) disynaptic basin, integrating less than 3.48 e. Note that these bonding regions in DTEs 5, 10, 12 and 13 and MODA 11 are characterized by the presence of two disynaptic basin, V(Cx,Cy) and V’(Cx,Cy), integrating more than 3.50 e. Together with these depopulations, the C2-C3 bonding regions are slightly populated, as shown by the presence of one V(C2,C3) disynaptic basin integrating more than 2.53 e. Note that at the final cycloadducts, the C2-C3 bonding regions become C2-C3 double bonds.
TS3 also shows the presence of two new V(N5) and V(C6) monosynaptic basins integrating 0.70 and 0.16 e, respectively. The electron density of these new monosynaptic basins comes from the depopulation of the N5-C6 bonding region in MODA cation 11, as well as from the high GEDT taking place at TS3. On the other hand, TS5 shows the presence of a new V(C1,C6) disynaptic basin integrating 0.82 e, indicating that the formation of the new C1-C6 single bond has already begun at this TS.
The present ELF topological analysis Indicates that while the more energetic TS1, TS2, and TS4 are more delayed, the more favorable TS3 and TS5 are more advanced, i.e., the easier it is to reach the TS, the more advanced the reaction.
An IRC analysis of the highly asynchronous
TS5 indicates that this P-IMDA reaction is associated with a
two-stage one-step mechanism [
46] in which the formation of the C4-C5 single bond begins when the formation of the C1-C6 single bond is practically completed.
2.4. Comparative Analysis of the Kinetics of the IMDA Reactions with Respect to the Kinetics of the DA Reactions
Next, the relative reaction rate constants (RRRCs), k
r, computed using the Eyring-Polanyi equation [
47], for the DA reactions of compounds
14 –
20 and the IMDA reactions of DTEs
5,
10,
12, and
13 and MODA
11 were analyzed. To this end, all gas phase stationary points involved in these intermolecular and intramolecular DA reactions were optimized in THF, and then the thermodynamic data were obtained by frequency calculations carried out in THF at 66 ºC. The electronic energies and the thermodynamic data of the stationary points involved in the DA reactions of compounds
14 –
20, and the IMDA reactions of DTEs
5,
10,
12 and
13 and MODA
11 are given in
Tables S3 and S4, respectively, in the Supplementary Material.
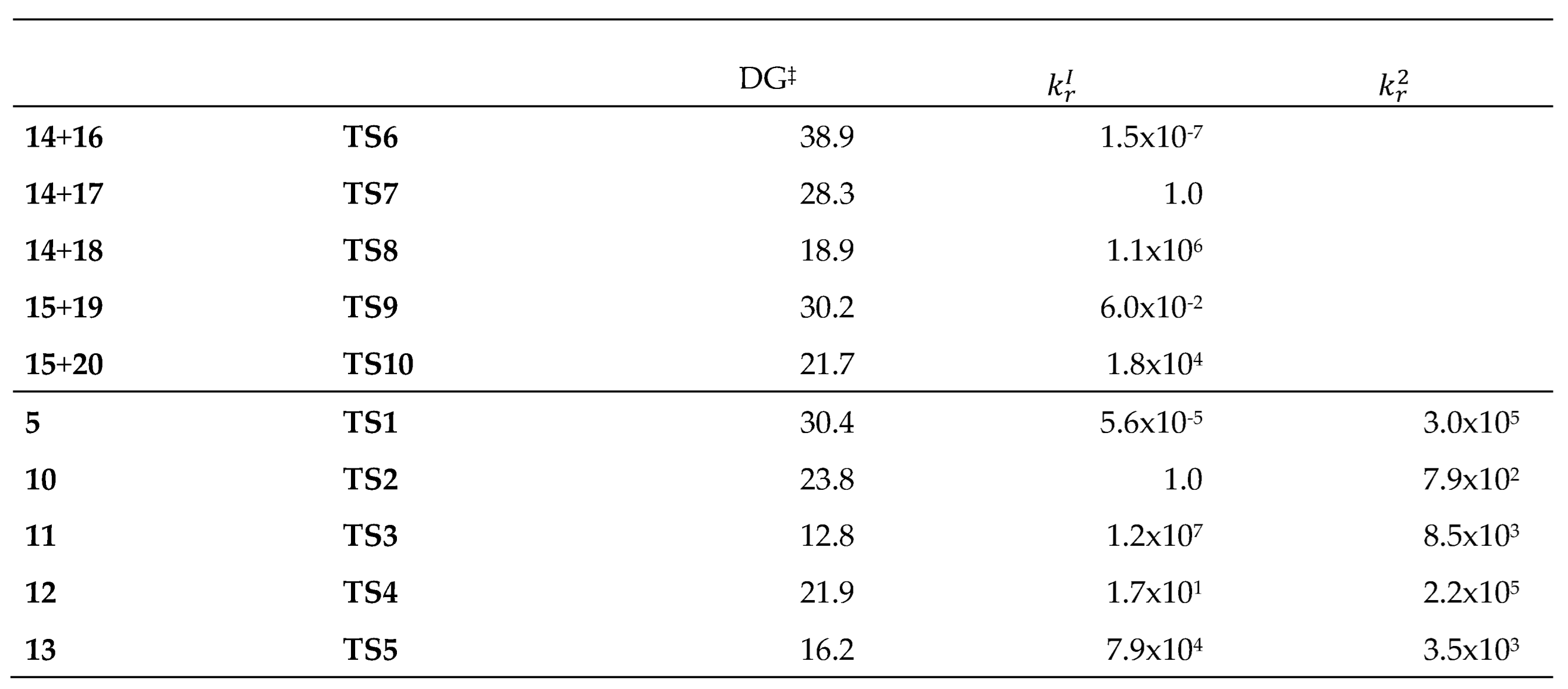
Table 4 summarizes the calculated activation Gibbs free energies for
TS1 –
TS10. Additionally, this table includes the RRRCs,
, with respect to the P-DA reaction between pentadiene
14 and 2-nitropropene
17, via
TS7, and with respect to the P-IMDA reaction of the 9-nitro derivative DTE
10, via
TS2. On the other hand,
, represents the RRRCs of each IMDA reaction with respect to the corresponding intermolecular DA reaction.
The RRRCs for the DA reactions with respect to the P-DA reaction between pentadiene 14 and 2-nitropropene 17 via TS7 range from 1.5x10-7 for the N-DA reaction between pentadiene 14 and propene 16 via TS6 to 1.1x106 for I-DA reaction between 14 and iminium cation 18 via TS8. On the other hand, the RRRCs for the IMDA reactions with respect to the P-IMDA reaction of the 9-nitro derivative DTE 10 via TS2 range from 5.6x10-5 for the N-IMDA reaction of DTE 5 via TS1 to 1.2x107 for the I-IMDA reaction of MODA cation 11 via TS3.
Some appealing conclusions can be drawn from the kinetic data given in
Table 4: i) the N-DA and N-IMDA reactions involving non-substituted unsaturated compounds are highly unfavorable; the N-DA reaction between pentadiene
14 and propene
16 via
TS6 is 1.5x10
-7 times slower than the P-DA reaction between
14 and 2-nitropropene
17 via
TS7. Despite the unfavorable character of the N-IMDA reaction of DTE
5, this reaction is 3.0x10
5 times faster than the N-DA between pentadiene
14 and propene
16; ii) the I-IMDA of MODA cation
11 via
TS3 is 1.2x10
7 times faster than the P-IMDA reaction of the 9-nitro derivative DTE
10 via
TS2, as a consequence of the higher GEDT taking place at the ionic process (see
Figure 4); iii) the double substitution in the P-IMDA reaction of the 2-nitro-9-methyl derivative DTE
13 via
TS5 is 7.9x10
4 times faster than the P-IMDA reaction of 9-nitro derivative DTE
10 via
TS2, again in agreement with the higher polar character of the former; and finally, iv) a similar trend is observed for the intermolecular P-DA and I-DA reactions, indicating that the substitution on the unsaturated compounds involved in the DA and IMDA reactions causes similar kinetic effects (see
Table 4).
The present comparative analysis of the kinetic parameters of the five DA and IMDA reactions shows that the substitution on the unsaturated compounds involved in these cycloaddition reactions produces a similar effect on the kinetics of these reactions. However, the IMDA reactions are approximately 10
4 times faster than the intermolecular DA reactions because of the less unfavorable activation entropies associated with the intramolecular processes (see
Tables S3 and S4 in the Supplementary Material). This behavior reduces the activation Gibb free energies associated with the intramolecular reaction by 4.5 and 8.5 kcal·mol
−1 compared to the intermolecular ones (see
Table 4).
2.5. Comparative RIAE Analysis of the DA Reactions of 14 with 16 or 17, and that of 15 with 20, and the IMDA Reactions of the DTEs 5, 10 and 13
Finally, to determine the electronic effects of the substituents on the butadiene or ethylene frameworks of the DA reactions of pentadiene
14 with propene
16 or 2-nitropropene
17, and those of 2-nitropentadiene
15 with vinyl ether
20, as well as the IMDA reactions of DTE
5, 9-nitro derivative DTE
10, and the 9-methyl-2-nitro derivative DTE
13 in the activation energies, a comparative relative interacting atomic energy [
48] (RIAE) analysis of the corresponding TSs was performed. The RIAE analysis provides a rationalization of the energy costs associated with the changes in electron density at the TSs, on which MEDT is based [
32]. The theoretical background of the RIAE is given in Supporting Information. The M06-2X/6-311G(d,p) gas-phase ξ
total, ξ
intra-atomic, and ξ
interatomic energies of the butadiene, ethylene and the –(CH
2)
4– chain frameworks of the TSs are given in
Table 5.
A recent RIAE study the P-DA reaction of cyclopentadiene
4 with the series of cyanoethylenes
26 has shown that the electronic stabilization of the electrophilic framework, resulting from GEDT in P-DA reactions, is the main factor responsible for the reduction in the activation energies [
12]. In polar processes, although the nucleophilic framework is destabilized by the loss of electron density, the greater stabilization of the electrophilic framework upon receiving the electron density is responsible for the reduction in activation energies [
12].
The RIAE analysis of TS6 associated with the N-DA reaction of pentadiene 14 with propene 16 shows that the destabilization of both the ethylene framework, ξ = 12.4 kcal·mol-1, and the butadiene framework, ξ = 8.5 kcal·mol-1, are responsible for the high RIAE activation energies, ξ = 20.9 kcal·mol-1, associated with this non-polar reaction. Although the ξ interatomic energies are stabilizing at the two frameworks, the more destabilizing ξ intra-atomic energies are responsible for the very high RIAE activation energy ξ.
The inclusion of a strong EW NO2 group in 2-nitropropene 17 decreases the ξ total energies of electrophilic ethylene framework of TS7 to -23.1 kcal·mol-1. Despite the increase in the unfavorable ξ total energies of the nucleophilic butadiene framework to 34.0 kcal·mol-1, the RIAE activation energy decreases to 10.9 kcal·mol-1.
The inclusion of a strong ER OCH3 group in 2-methoxypropene 20 and a strong EW NO2 group in 2-nitropentadiene 15 causes a more remarkable effect in the RIAE activation energy of TS10 in the P-DA reaction of 15 with 20. While the ξ total energies of the nucleophilic ethylene framework increase to 43.4 kcal·mol-1, those of the electrophilic butadiene framework decrease considerably to -36.3 kcal·mol-1. These effects reduce the RIAE activation energy of this P-DA reaction to 7.1 kcal·mol-1.
The molecular structure of the TSs of the three selected IMDA reactions is divided into three frameworks: a) the ethylene, Et, b) the butadiene, Bu, and c) the –(CH
2)
4- saturated hydrocarbon chain, CH
2, linking the two unsaturated frameworks (see
Table 5). A comparative analysis of the ξ
interatomic, ξ
intra-atomic and ξ
total energies of the three DA and the three IMDA reactions shows that both types of reactions experience a similar electronic effect with the substitution at the ethylene and the butadiene frameworks. The RIAE activation energies of the TSs of the IMDA reaction are 3.6 and 6.1 kcal·mol
-1 higher in energy than those of the intermolecular DA reactions. In general, the unfavorable ξ
intra-atomic energies associated with the –(CH
2)
4- chain are responsible for these energy differences. The N-IMDA rection of DTE
5 presents the most unfavorable RIAE activation energy, 24.5 kcal·mol
-1. The ξ
total energies associated with the ethylene and butadiene frameworks are destabilized by ca. 17 kcal·mol
-1. As in the P-DA reactions, the inclusion of a strong EW NO
2 group at the ethylene framework of 9-nitro derivative DTE
10, or a strong EW NO
2 group at the butadiene framework combined with a strong ER OCH
3 group at the ethylene framework of 9-metoxy-2-nitro derivative DTE
13, reduces the RIAE activation energies associated with
TS2 and
TS5 to 17.0 and 11.7 kcal·mol
-1, respectively. Analysis of the ξ
total energies of
TS2 and
TS5 further shows that the stabilization of the electrophilic ethylene at
TS2 or electrophilic butadiene framework at
TS5 is the factor responsible for the acceleration observed in these IMDA reactions. The GEDT taking place at the TSs is also represented.
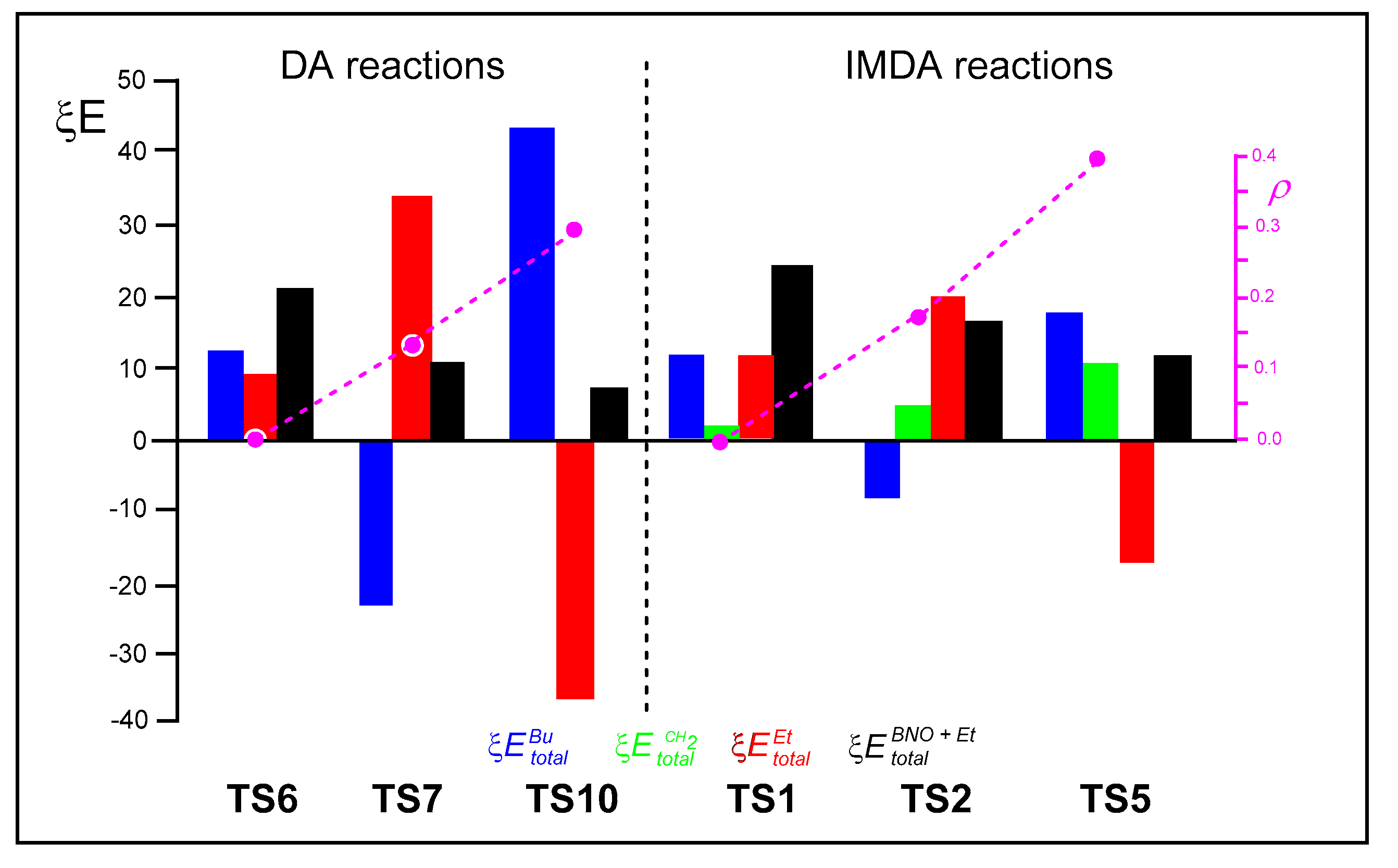
Finally,
Figure 6 shows a graphical representation of the ξ
energies of the Bu, Et and CH
2 frameworks at the six TSs, shown in blue, red and green, respectively, and the ξ
and ξ
energies, shown in black, which correspond to the RIAE activation energies of these DA and IMDA reactions, respectively.
An analysis of the data presented in
Figure 6 permits us to obtain some interesting conclusions: i) in both DA and IMDA reactions, the RIAE activation energies decrease with increasing GEDT. This decrease is more effective for the intermolecular processes; ii) a comparison of the ξ
total energies associated with the ethylene and butadiene frameworks in the inter- and intramolecular polar reactions indicates these energies are higher in the intermolecular DA reactions, suggesting more effective nucleophilic/electrophilic interactions in the intermolecular processes, despite the fact that GEDT values are higher in IMDA reactions; iii) in non-polar cycloaddition reactions, both ethylene and butadiene frameworks are destabilized at the TSs, explaining the high activation energies associated with these non-polar reactions; iv) in polar reactions, although the nucleophilic frameworks are strongly destabilized compared to the non-polar reactions, the electrophilic frameworks are even more strongly stabilized, reducing the RIAE activation energies of polar reactions; and finally, v) in IMDA reactions, the ξ
total energies, associated with the strain of the chain, increase as the RIAE activation energies decrease, due to the more advanced character of the IMDA reaction.
3. Computational Methods
The M06-2X functional [
49], together with the standard 6-311G(d,p) basis set [
50], which includes d-type polarization for second-row elements and p-type polarization functions for hydrogen atoms, was used throughout this MEDT study The TSs were identified by the presence of a single imaginary frequency. Optimizations were performed using the Berny algorithm [
51,
52]. Intrinsic reaction coordinate [
53] (IRC) paths were traced to map of the energy profiles linking each TS to the two corresponding minima on the PES.
Solvent effects of THF were considered in the thermodynamic calculations by full optimization of the
in-vacuo structures at the same computational level using the polarizable continuum model (PCM) [
54,
55] in the framework of the self-consistent reaction field (SCRF) [
56,
57,
58]. Values of M06-2X/6-311G(d,p) enthalpies, entropies, and Gibbs free energies in THF were calculated with standard statistical thermodynamics at 66 ºC and 1 atm through PCM frequency calculations at the solvent-optimized structures. Except for thermodynamic data, all other results were obtained in
in-vacuo for an overall coherence in interpreting the results obtained from different quantum chemical tools.
DFT-based reactivity indices [
36,
37,
38] were calculated using the equations in reference 37. The GEDT [
8] values were computed using the equation GEDT(f) = Σq
f, where q corresponds to the natural charges [
34,
35] of the atoms belonging to one of the two frameworks (f) at the TS geometries.
The Gaussian 16 suite of programs was used to perform the calculations [
59]. Molecular geometries and ELF basin attractors were visualized by using the GaussView program [
60]. ELF analyses [
33] of the M06-2X/6-311G(d,p) monodeterminantal wavefunctions were done by using the TopMod [
61] package with a cubical grid of step size of 0.1 Bohr. The Interacting Quantum Atoms [
62] (IQA) analysis for the RIAE study [
58] was performed with the AIMAll package [
63] using the corresponding M06-2X/6-311G(d,p) monodeterminantal wavefunctions.
4. Conclusions
The IMDA reactions of DTEs 5, 10, 12, and 13, and MODA 11, have been studied within the MEDT framework at the M06-2X/6-311G(d,p) computational level and compared with the intermolecular DA reactions of the unsaturated compounds 14 – 20 to better understand the behavior of the intramolecular processes.
An ELF topological analysis of the GS electronic structure of DTEs 5, 10, 12 and 13, and MODA 11 shows a strong similarity with that of the derivatives 14 – 20. The inclusion of EW or ER groups in these compounds does not modify markedly the electronic structure of non-substituted compounds.
Analysis of the electrophilicity w and nucleophilicity N indices of DTEs DTEs 5, 10, 12 and 13, and MODA 11 indicates that they are closely related to those of the butadiene and ethylenes derivatives 14 – 20. On the other hand, the analysis of the local reactivity difference Rk indices permits the study of the regioselectivity in intramolecular polar processes helping to identify the most nucleophilic and electrophilic centers of these complex molecules.
Analysis of the relative energies of the stationary points involved in the most favorable reaction path associated with the IMDA reactions of DTEs 5, 10, 12 and 13, and MODA 11 leads to two significant conclusions: i) the five IMDA reactions are strongly exothermic in the range –43.8 (5) to –52.2 (11) kcal·mol-1, while the relative energies of the TSs range from 23.9 for the N-IMDA reaction of DTE 5 via TS1 kcal·mol-1 to –0.8 kcal·mol-1 for the I-IMDA reaction of MODA 11 via TS3. The trend in these relative energies closely resembles that of DA reactions; and ii) the REDF P-IMDA reactions of DTEs 12 and 13 demand double substitution at the diene and ethylene frameworks to facilitate the polar reactions.
Analysis of the geometries, GEDT values, and electronic structure of the five TSs involved in the IMDA reactions shows a strong similarity to those found in the intermolecular DA reactions, indicating that the substituents have similar electronic effects in both types of DA reactions.
A comparative RIAE analysis of the intermolecular and intramolecular DA reactions allows for the establishment of the effects of the EW and ER groups in these polar reactions. The non-polar reactions are highly disfavored due to the unfavorable ξintra-atomic energies associated with the butadiene and ethylene frameworks demanded to reach the TSs. The polar character of the TS enhances the feasibility of these DA reactions. As the butadiene framework of DTE 5 is nucleophilically activated, the ethylene framework must be electrophilically activated by the presence of an EW NO2 group. The RIAE analysis shows that although the nucleophilic frameworks are destabilized due to the loss of electron density, the electrophilic frameworks are further stabilized due to the gain of electron density, thereby reducing the RIAE activation energies.
The present MEDT study shows the strong electronic similarity between the intramolecular and intermolecular DA reactions. The non-substituted compounds demand a high energy cost to reach the non-polar TSs. To favor the bonding changes in these reactions, at least one of the two interacting frameworks should be electrophilically activated by the presence of an EW group, such as NO2, to increase the GEDT of the reaction. The same trend is observed in both types of DA reactions. The main difference between them lies in the unfavorable activation entropy associated with intermolecular DA reactions. Thus, the lower activation entropy associated with the intramolecular processes makes them approximately 104 times faster than intermolecular ones, despite the fact that the activation enthalpies associated with the latter are lower.
Scheme 1.
DA reaction between butadiene 1 and ethylene 2.
Scheme 1.
DA reaction between butadiene 1 and ethylene 2.
Figure 1.
Plot of the activation barriers (ΔE in kcal·mol-1)) vs. GEDT, e, R2 = 0.89, for the DA reactions of Cp 4 with a series of substituted ethylenes of increasing electrophilic character. The classification of the DA reactions, based on GEDT, is included.
Figure 1.
Plot of the activation barriers (ΔE in kcal·mol-1)) vs. GEDT, e, R2 = 0.89, for the DA reactions of Cp 4 with a series of substituted ethylenes of increasing electrophilic character. The classification of the DA reactions, based on GEDT, is included.
Scheme 2.
IMDA reaction of (E)-deca-1,3,9-triene 5. Relative enthalpies and Gibbs free energies are given in kcal·mol-1, while relative entropies are given in cal·mol-1·K-1.
Scheme 2.
IMDA reaction of (E)-deca-1,3,9-triene 5. Relative enthalpies and Gibbs free energies are given in kcal·mol-1, while relative entropies are given in cal·mol-1·K-1.
Scheme 3.
ionic IMDA reaction of dieniminium 7.
Scheme 3.
ionic IMDA reaction of dieniminium 7.
Chart 1.
DTEs 5, 10, 12 and 13 and MODA 11 used in the study of the N-IMDA, P-IMDA, and I-IMDA reactions.
Chart 1.
DTEs 5, 10, 12 and 13 and MODA 11 used in the study of the N-IMDA, P-IMDA, and I-IMDA reactions.
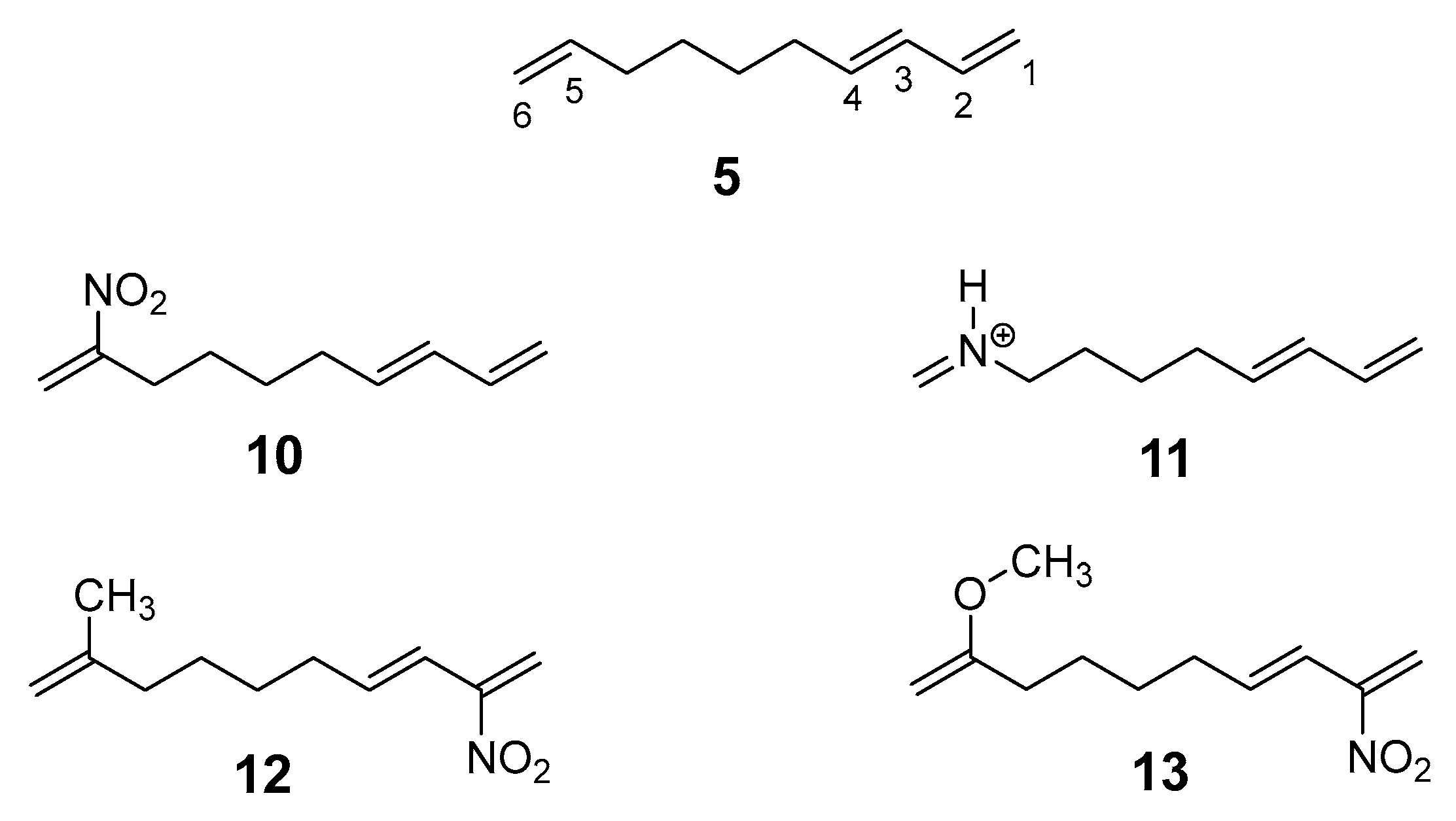
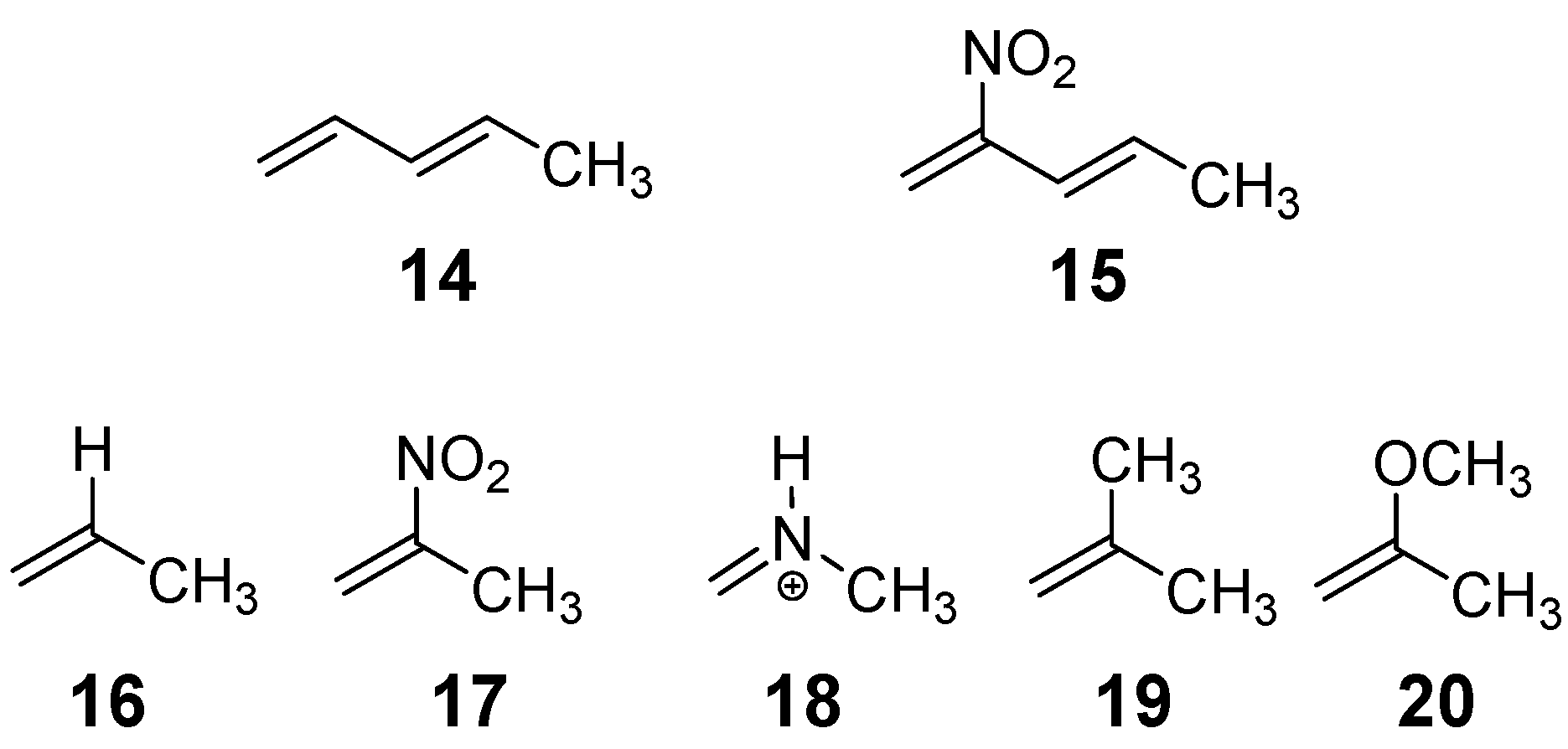
Chart 2.
Dienes, 14 and 15, and ethylenes, 16 – 20, involved in the studied DA reactions.
Chart 2.
Dienes, 14 and 15, and ethylenes, 16 – 20, involved in the studied DA reactions.
Figure 2.
ELF basin attractor positions and the populations of the most relevant valence basins of DTEs 5, 10, 12, and 13 and MODA 11, calculated at the M06-2X/6-311G(d,p) level in the gas phase. Natural atomic charges are reported as the average number of electrons, e. Negative charges are highlighted in red, positive charges in blue, and negligible charges in green.
Figure 2.
ELF basin attractor positions and the populations of the most relevant valence basins of DTEs 5, 10, 12, and 13 and MODA 11, calculated at the M06-2X/6-311G(d,p) level in the gas phase. Natural atomic charges are reported as the average number of electrons, e. Negative charges are highlighted in red, positive charges in blue, and negligible charges in green.
Chart 3.
Most electrophilic, in red, and nucleophilic, in blue, centers, together with the corresponding Rk values of DTEs 10, 12, and 13 and MODA 11.
Chart 3.
Most electrophilic, in red, and nucleophilic, in blue, centers, together with the corresponding Rk values of DTEs 10, 12, and 13 and MODA 11.
Scheme 4.
N-IMDA, 5, P-IMDA, 10, 12 and 13, and I-IMDA, 11, reactions.
Scheme 4.
N-IMDA, 5, P-IMDA, 10, 12 and 13, and I-IMDA, 11, reactions.
Figure 3.
M06-2X/6-311G(d,p) gas phase optimized geometries of TS1 – TS5 associated with the IMDA reactions of DTEs 5, 10 ,12 and 13 and MODA 11. GEDT values are shown in red and expressed as the average number of electrons, e. Bond lengths are given in Angstroms.
Figure 3.
M06-2X/6-311G(d,p) gas phase optimized geometries of TS1 – TS5 associated with the IMDA reactions of DTEs 5, 10 ,12 and 13 and MODA 11. GEDT values are shown in red and expressed as the average number of electrons, e. Bond lengths are given in Angstroms.
Figure 4.
Plot of the relative energies, ΔE in kcal·mol-1, of TSs of the IMDA reactions vs the GEDT expressed as the average number of electrons e, computed at the corresponding TSs. The three groups of N-IMDA, P-IMDA, and I-IMDA reactions are shown in red, green, and blue, respectively.
Figure 4.
Plot of the relative energies, ΔE in kcal·mol-1, of TSs of the IMDA reactions vs the GEDT expressed as the average number of electrons e, computed at the corresponding TSs. The three groups of N-IMDA, P-IMDA, and I-IMDA reactions are shown in red, green, and blue, respectively.
Figure 5.
ELF basin attractor positions and the populations of the most relevant valence basins of TS1 – TS5, calculated at the M06-2X/6-311G(d,p) level in the gas phase.
Figure 5.
ELF basin attractor positions and the populations of the most relevant valence basins of TS1 – TS5, calculated at the M06-2X/6-311G(d,p) level in the gas phase.
Figure 6.
Graphical representation of the sets of the M06-2X/6-311G(d,p) total energies, ξ, ξ, ξ, ξand the ξ for the TSs. The ξand ξ energies correspond to the RIAE activation energies of these DA and IMDA reactions, respectively. ξ energies for the Bu, Et and CH2 frameworks are shown in blue, red and green, respectively, while the black bar represents the ξ and ξ energies. ξ energies are reported in kcal mol-1. The GEDT values at the TSs, displayed in pink, are expressed as an average number of electrons e.
Figure 6.
Graphical representation of the sets of the M06-2X/6-311G(d,p) total energies, ξ, ξ, ξ, ξand the ξ for the TSs. The ξand ξ energies correspond to the RIAE activation energies of these DA and IMDA reactions, respectively. ξ energies for the Bu, Et and CH2 frameworks are shown in blue, red and green, respectively, while the black bar represents the ξ and ξ energies. ξ energies are reported in kcal mol-1. The GEDT values at the TSs, displayed in pink, are expressed as an average number of electrons e.
Table 1.
B3LYP/6-31G(d) electronic chemical potential μ, chemical hardness η, and electrophilicity ω and nucleophilicity N indices, calculated at the B3LYP/6-31G(d) for DTEs 5, 10, 12 and 13 and MODA 11, expressed in eV. The reactivity indices of the MODA cation 11 are also computed in DMSO.
Table 1.
B3LYP/6-31G(d) electronic chemical potential μ, chemical hardness η, and electrophilicity ω and nucleophilicity N indices, calculated at the B3LYP/6-31G(d) for DTEs 5, 10, 12 and 13 and MODA 11, expressed in eV. The reactivity indices of the MODA cation 11 are also computed in DMSO.
Table 2.
Electrophilicand nucleophilicParr functions, and
reactivity difference Rk indices of DTEs 10, 12 and 13
and MODA 11.
Table 2.
Electrophilicand nucleophilicParr functions, and
reactivity difference Rk indices of DTEs 10, 12 and 13
and MODA 11.
Table 3.
M06-2X/6-311G(d,p) gas phase total, E in a.u., and relative, DE, in kcal·mol-1, energies of the stationary points involved in the IMDA reactions of DTEs 5, 10, 12 and 13 and MODA 11.
Table 3.
M06-2X/6-311G(d,p) gas phase total, E in a.u., and relative, DE, in kcal·mol-1, energies of the stationary points involved in the IMDA reactions of DTEs 5, 10, 12 and 13 and MODA 11.
Table 4.
M06-2X/6-311G(d,p) activation Gibbs free energies, ΔG‡ in kcal·mol−1, calculated in tetrahydrofuran at 66 ºC, along with the RRRCs, kr, for the DA reactions of compounds 14 – 20, and for the IMDA reactions of DTEs 5, 10, 12 and 13 and MODA 11. represents the RRRCs relative to TS7, while represents the RRRCs of the IMDA reactions relative to the corresponding DA reactions.
Table 4.
M06-2X/6-311G(d,p) activation Gibbs free energies, ΔG‡ in kcal·mol−1, calculated in tetrahydrofuran at 66 ºC, along with the RRRCs, kr, for the DA reactions of compounds 14 – 20, and for the IMDA reactions of DTEs 5, 10, 12 and 13 and MODA 11. represents the RRRCs relative to TS7, while represents the RRRCs of the IMDA reactions relative to the corresponding DA reactions.
Table 5.
M06-2X/6-311G(d,p) Gas-phase total energies, ξ, intra-atomic energies, ξ, and interatomic energies, ξ calculated using M06-2X/6-311G(d,p) level for the ethylene, Et, butadiene, Bu, and the –(CH2)4– chain, CH2, frameworks at the selected TSs relative to their GSs. The sum of the ξ energies of both frameworks, denoted as ξ, represents the RIAE activation energies.
Table 5.
M06-2X/6-311G(d,p) Gas-phase total energies, ξ, intra-atomic energies, ξ, and interatomic energies, ξ calculated using M06-2X/6-311G(d,p) level for the ethylene, Et, butadiene, Bu, and the –(CH2)4– chain, CH2, frameworks at the selected TSs relative to their GSs. The sum of the ξ energies of both frameworks, denoted as ξ, represents the RIAE activation energies.
| TS |
f(x) |
|
|
|
|
| TS6 |
Et |
36.2 |
-23.8 |
12.4 |
20.9 |
| Bu |
38.3 |
-29.8 |
8.5 |
| TS7 |
Et |
31.8 |
-54.9 |
-23.1 |
10.9 |
| Bu |
52.8 |
-18.8 |
34.0 |
| TS10 |
Et |
95.9 |
-52.4 |
43.4 |
7.1 |
| Bu |
43.5 |
-79.7 |
-36.3 |
| TS1 |
Et |
29.3 |
-17.6 |
11.7 |
24.5 |
| CH2
|
5.2 |
-3.4 |
1.8 |
| Bu |
36.2 |
-25.2 |
11.0 |
| TS2 |
Et |
36.7 |
-44.7 |
-8.0 |
17.0 |
| CH2
|
5.8 |
-1.4 |
4.4 |
| Bu |
41.3 |
-20.6 |
20.6 |
| TS5 |
Et |
81.1 |
-63.2 |
17.8 |
11.7 |
| CH2
|
3.9 |
6.9 |
10.8 |
| Bu |
42.0 |
-59.0 |
-17.0 |
 19.4 cal·mol-1·K-1, reduces the activation Gibbs free energy to 38.0 kcal·mol-1. This behavior explains the feasibility of non-polar IMDA reactions.
19.4 cal·mol-1·K-1, reduces the activation Gibbs free energy to 38.0 kcal·mol-1. This behavior explains the feasibility of non-polar IMDA reactions. 14.3 cal·mol-1·K-1 [13]. This ionic IMDA reaction exhibits complete re/exo and si/endo diastereoselectivity, which is controlled by the most favorable chair conformations adopted by the –(CH2)4- chain linking the butadiene and iminium frameworks (see dieniminium 7 in Scheme 3).
14.3 cal·mol-1·K-1 [13]. This ionic IMDA reaction exhibits complete re/exo and si/endo diastereoselectivity, which is controlled by the most favorable chair conformations adopted by the –(CH2)4- chain linking the butadiene and iminium frameworks (see dieniminium 7 in Scheme 3). 0.05 e. Non-polar 32CA reactions, characterized by a negligible GEDT ( ≤ |0.05| e), are classified as NEDF [42].
0.05 e. Non-polar 32CA reactions, characterized by a negligible GEDT ( ≤ |0.05| e), are classified as NEDF [42].