Submitted:
08 August 2025
Posted:
11 August 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Patient and Skin Samples
2.2. Treatments
2.3. Histological Analysis
2.4. Molecular Biology Analysis
2.5. Statistical Analysis
3. Results
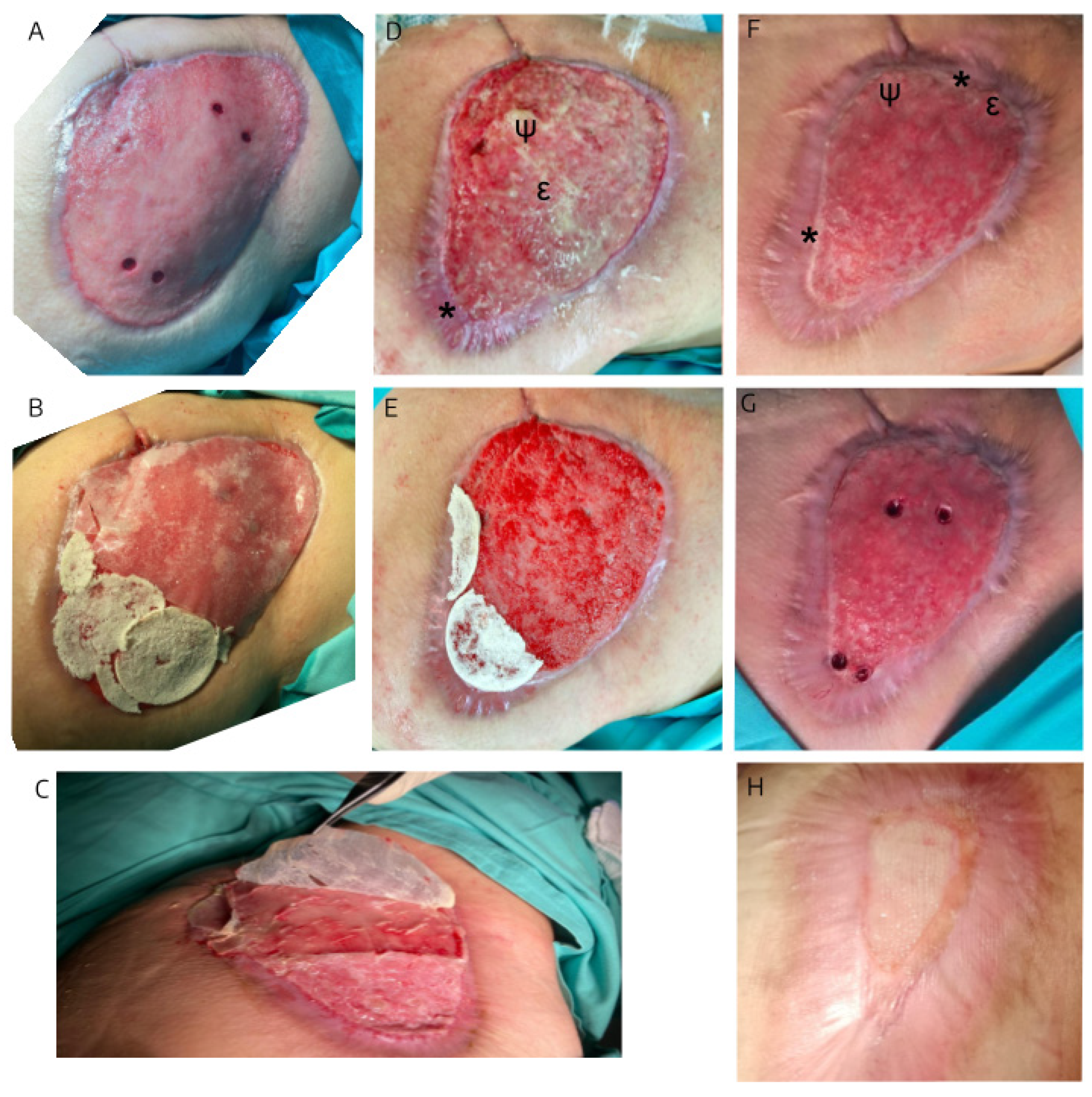
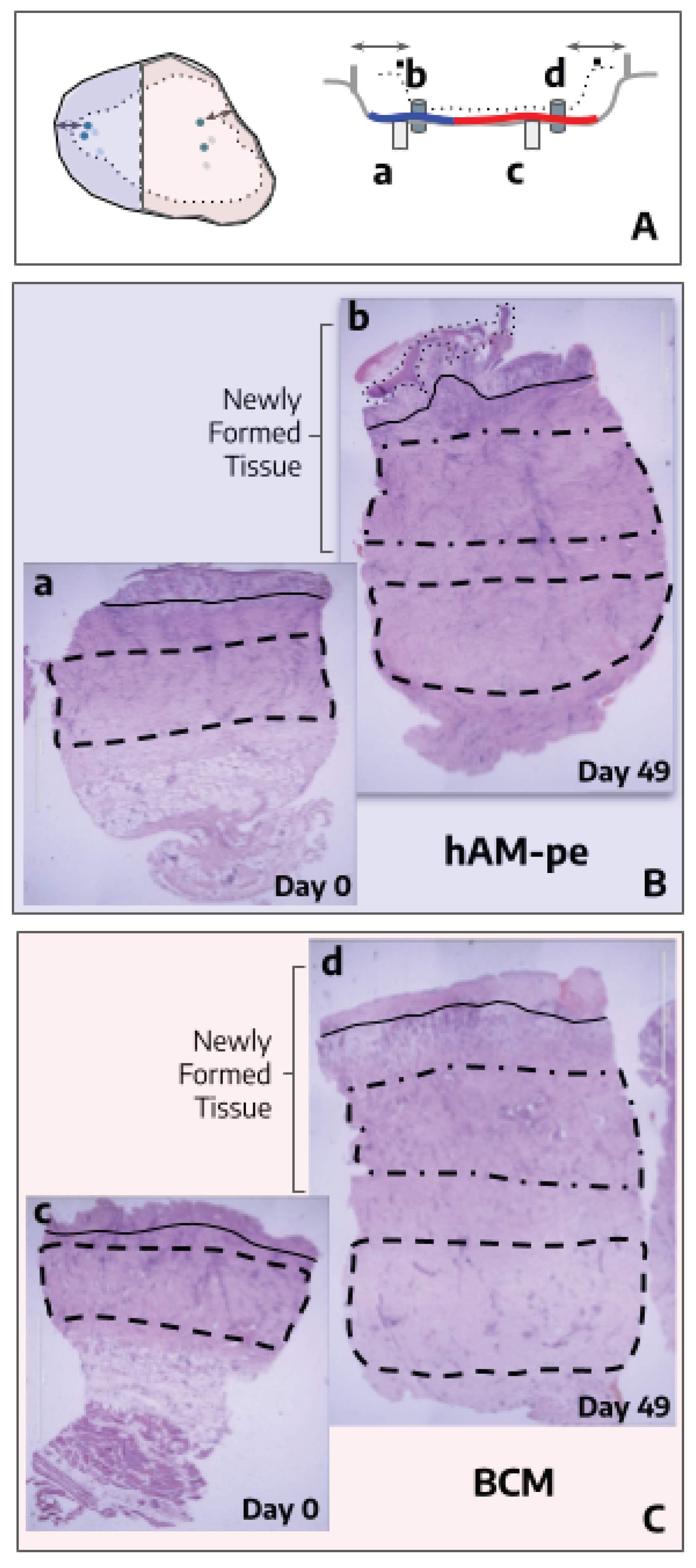
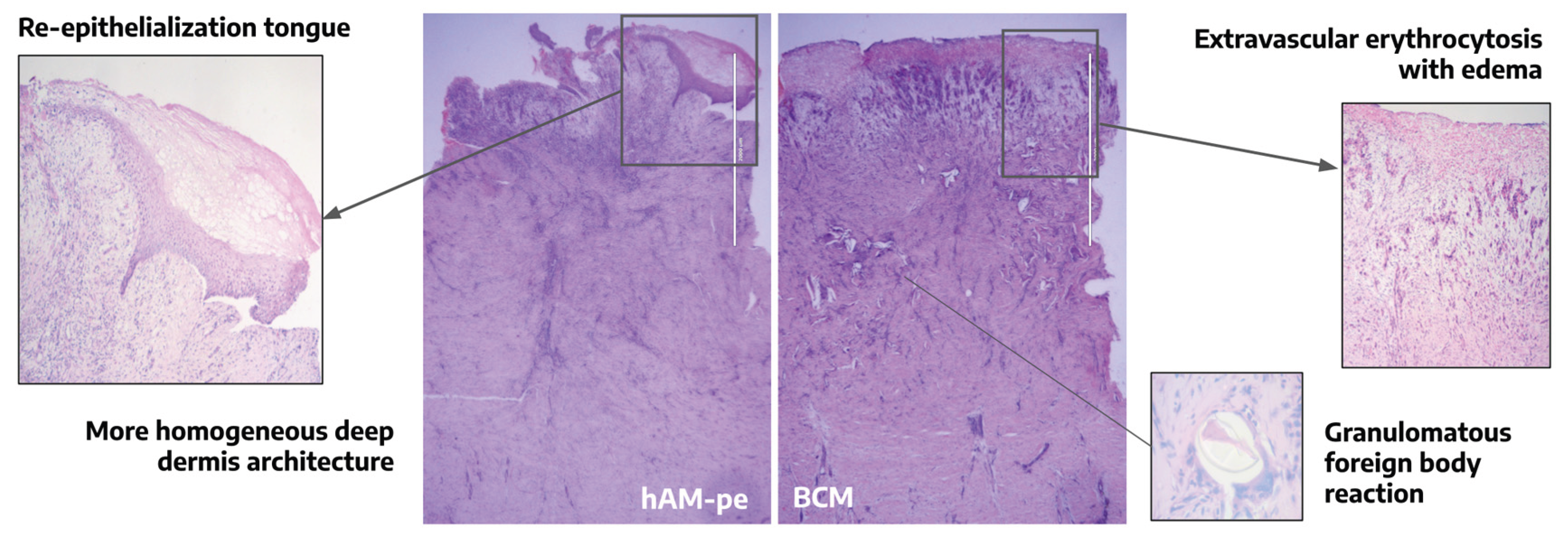
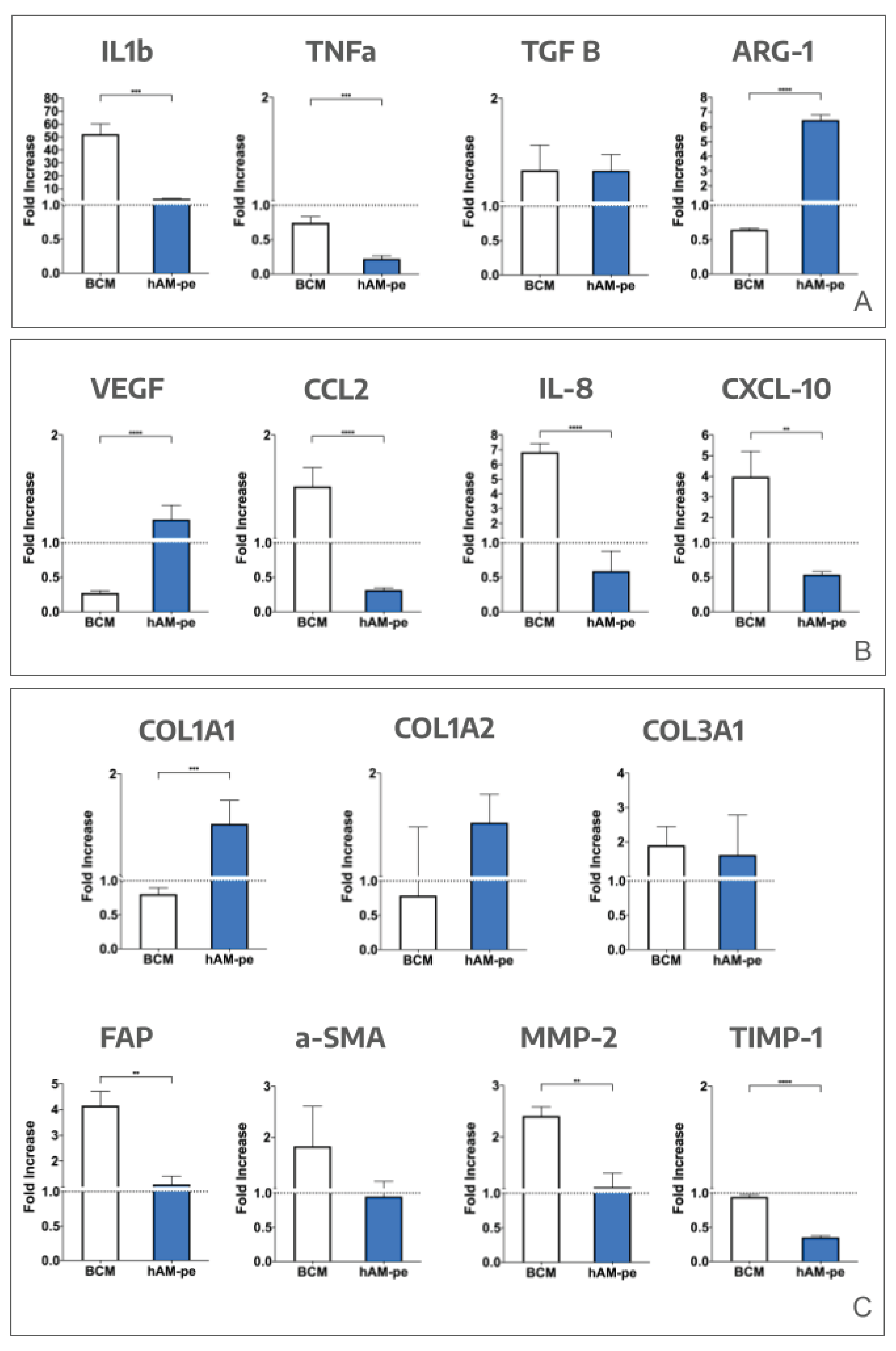
3.1. The Evolution of the Wound Treated with hAM-pe is Associated with a Faster Progression of the Re-Epithelialization Front and a Reduced Local Inflammatory Activity
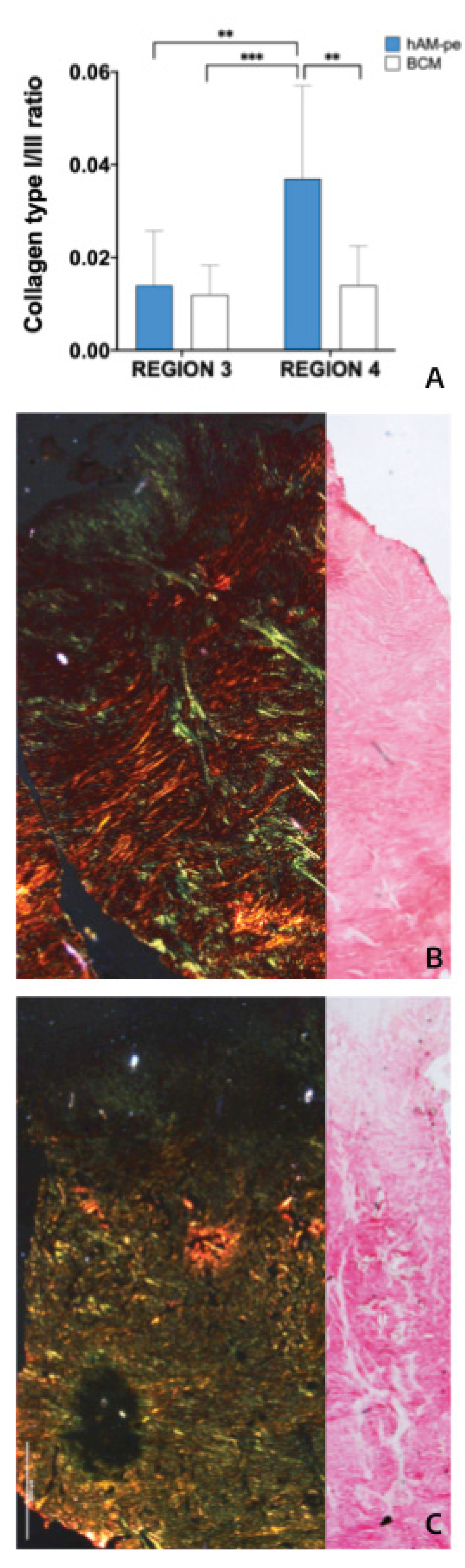
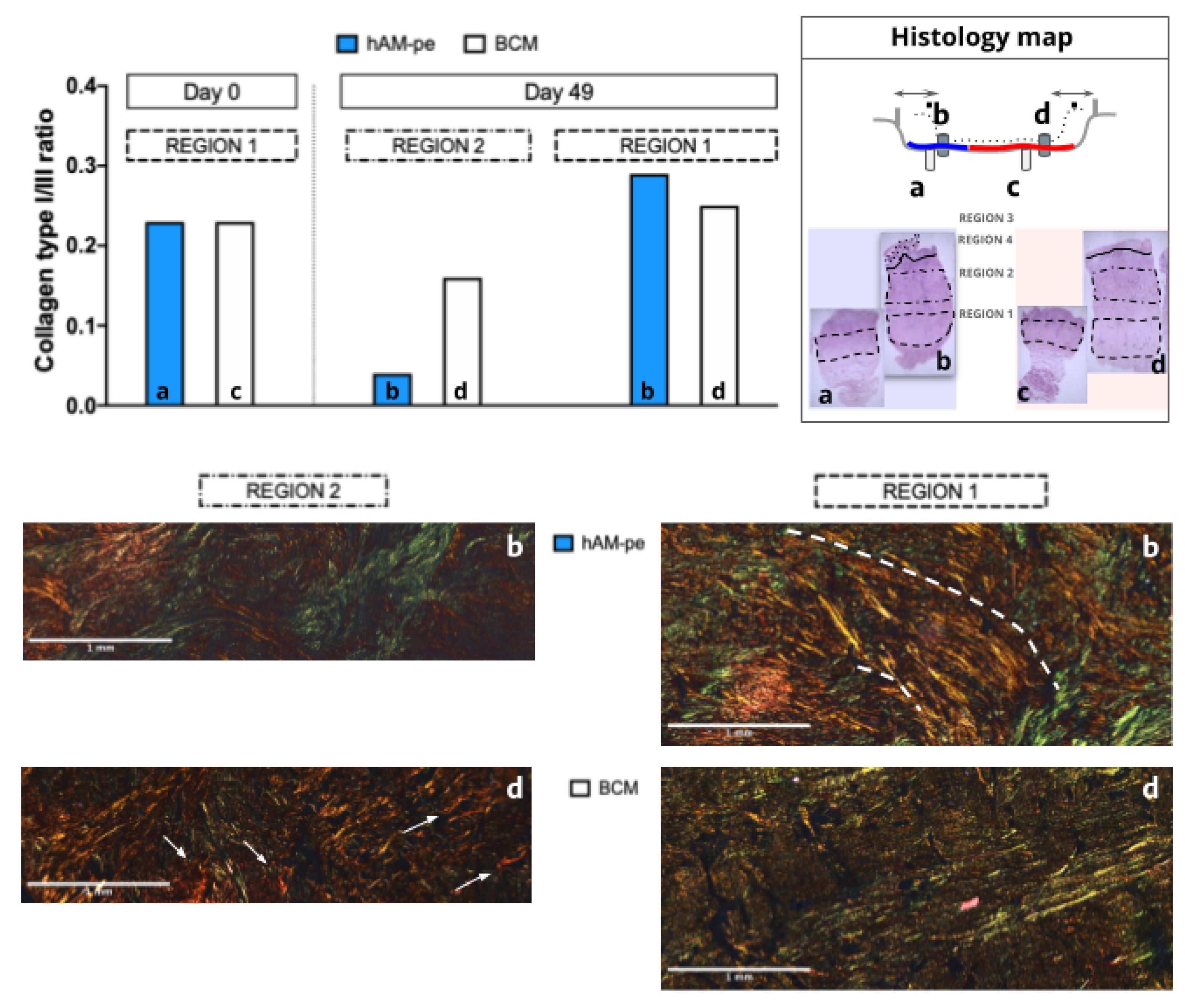
3.2. The Treatment with hAM-pe Improves the Deposition and Organization of the Extracellular Matrix
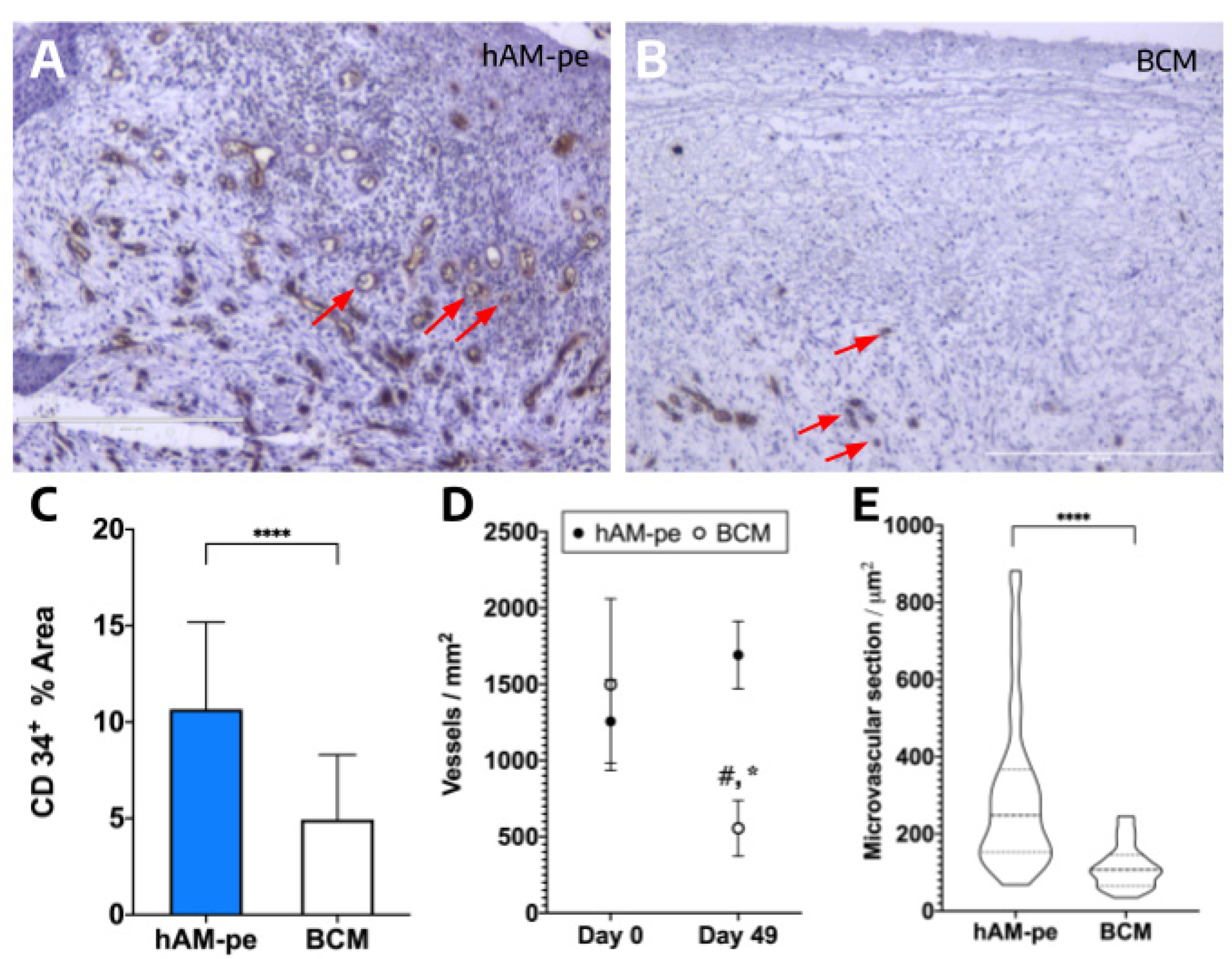
3.3. The hAM-pe Treatment Promotes Angiogenesis and Vascularization in the Repair Tissue
3.4. Tissues Repaired Under the Action of hAM-pe Are Likely in a More Advanced Stage of Wound Healing
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| α-SMA | Alpha-Smooth Muscle Actin |
| ANMAT | National Administration of Drugs, Food, and Medical Technology (Argentina) |
| ANOVA | Analysis of Variance |
| APCs | Antigen-Presenting Cells |
| Arg-1 | Arginase-1 |
| BCM | Bovine Collagen Matrix |
| CCL2 | C-C Motif Chemokine Ligand 2 (Monocyte Chemoattractant Protein-1, MCP-1) |
| CD34 | Cluster of Differentiation 34 (marker of hematopoietic and endothelial progenitor cells) |
| col1a1 | Collagen Type I Alpha 1 Chain |
| col1a2 | Collagen Type I Alpha 2 Chain |
| col3a1 | Collagen Type III Alpha 1 Chain |
| Ct | Threshold Cycles |
| CXCL-10 | C-X-C Motif Chemokine Ligand 10 (Interferon gamma-induced protein 10) |
| DAMPs | Damage-Associated Molecular Patterns |
| DFU | Diabetic Foot Ulcer |
| ECM | Extracellular Matrix |
| FAP | Fibroblast Activation Protein |
| hAM | Human Amniotic Membrane |
| hAM-pe | Lyophilized homogenized human amniotic membrane dressings sterilized by gamma radiation |
| HIV | Human Immunodeficiency Virus |
| IL-1β | Interleukin 1 beta |
| IL-8 | Interleukin 8 |
| INCUCAI | National Central Unique Institute for Ablation and Implant Coordination (Argentina) |
| MMP-1 | Matrix Metalloproteinase-1 |
| MMP-2 | Matrix Metalloproteinase-2 |
| RNA | Ribonucleic Acid |
| ROI | Region of Interest |
| RT-qPCR | Reverse Transcription Quantitative Polymerase Chain Reaction |
| TGF-β | Transforming Growth Factor Beta |
| TIMP-1 | Tissue Inhibitor of Metalloproteinases-1 |
| TNF-α | Tumor Necrosis Factor alpha |
| VAC | Vacuum-Assisted Closure |
| VEGF | Vascular Endothelial Growth Factor |
References
- Robson MC, Steed DL, Franz MG. Wound healing: biologic features and approaches to maximize healing trajectories. Curr Probl Surg. 2001, 38, 72–140. [Google Scholar]
- Dai, C. , Shih, S., & Khachemoune, A. Skin substitutes for acute and chronic wound healing: an updated review. Journal of Dermatological Treatment 2020, 31, 639–648. [Google Scholar] [CrossRef]
- Murray, P. J. , & Wynn, T. A. Protective and pathogenic functions of macrophage subsets. Nature reviews immunology 2011, 11, 723–737. [Google Scholar] [PubMed]
- Velnar, T. , Bailey, T., & Smrkolj, V. The wound healing process: an overview of the cellular and molecular mechanisms. Journal of international medical research 2009, 37, 1528–1542. [Google Scholar] [PubMed]
- de Souza, I. C. , Takejima, A. L., Simeoni, R. B., Gamba, L. K., Ribeiro, V. S. T., Foltz, K. M.,... & Guarita-Souza, L. C. Acellular biomaterials associated with autologous bone marrow-derived mononuclear stem cells improve wound healing through paracrine effects. Biomedicines 2023, 11, 1003. [Google Scholar] [CrossRef]
- Hesketh, M. , Sahin, K. B., West, Z. E., & Murray, R. Z. Macrophage phenotypes regulate scar formation and chronic wound healing. International journal of molecular sciences 2017, 18, 1545. [Google Scholar] [PubMed]
- Gardeazabal, L. , & Izeta, A. Elastin and collagen fibres in cutaneous wound healing. Experimental Dermatology 2024, 33, e15052. [Google Scholar] [CrossRef]
- Gauglitz, G. G. , Korting, H. C., Pavicic, T., Ruzicka, T., & Jeschke, M. G. Hypertrophic scarring and keloids: pathomechanisms and current and emerging treatment strategies. Molecular medicine 2011, 17, 113–125. [Google Scholar] [CrossRef]
- Knoedler, S. , Broichhausen, S., Guo, R., Dai, R., Knoedler, L., Kauke-Navarro, M.,... & Rinkevich, Y. Fibroblasts–The cellular choreographers of wound healing. Frontiers in Immunology 2023, 14, 1233800. [Google Scholar] [CrossRef]
- Tchero, H. , Herlin, C., Bekara, F., Kangambega, P., Sergiu, F., & Teot, L. Failure rates of artificial dermis products in treatment of diabetic foot ulcer: A systematic review and network meta-analysis. Wound Repair and Regeneration 2017, 25, 691–696. [Google Scholar] [CrossRef]
- Leal-Marin, S. , Kern, T., Hofmann, N., Pogozhykh, O., Framme, C., Börgel, M.,... & Gryshkov, O. Human Amniotic Membrane: A review on tissue engineering, application, and storage. Journal of Biomedical Materials Research Part B: Applied Biomaterials 2021, 109, 1198–1215. [Google Scholar] [CrossRef]
- Khosravimelal, S. , Momeni, M., Gholipur, M., Kundu, S. C., & Gholipourmalekabadi, M. Protocols for decellularization of human amniotic membrane. Methods in Cell Biology 2020, arXiv:10.1016/bs.mcb.2019.11.004157, 37–47. [Google Scholar]
- De Angelis, B. , Orlandi, F., Morais D’Autilio, M. F. L., Di Segni, C., Scioli, M. G., Orlandi, A.,... & Gentile, P. Vasculogenic chronic ulcer: tissue regeneration with an innovative dermal substitute. Journal of Clinical Medicine 2019, 8, 525. [Google Scholar] [PubMed]
- Carro, G. V. , Guerbi, X., Berra, M., Rodriguez, M. G., Noli, M. L., Fuentes, M.,... & Berra, A. Homogenized and Lyophilized Amniotic Membrane Dressings for the Treatment of Diabetic Foot Ulcers in Ambulatory Patients. Foot & Ankle International 2024, 10711007241243373. [Google Scholar]
- Stringa P, Romanin D, Lausada N, Papa Gobbi R, Zanuzzi C, Martín P, Abate JC, Cabanne A, Arnal N, Vecchio L, Milesi V, Portiansky E, Gondolesi G, Rumbo M. Gut Permeability and Glucose Absorption Are Affected at Early Stages of Graft Rejection in a Small Bowel Transplant Rat Model. Transplant Direct. 2017, 3, e220. [Google Scholar] [CrossRef]
- Adamska, A. , Pilacinski, S., Zozulinska-Ziolkiewicz, D., Gandecka, A., Grzelka, A., Konwerska, A.,... & Araszkiewicz, A. An increased skin microvessel density is associated with neurovascular complications in type 1 diabetes mellitus. Diabetes and Vascular Disease Research 2019, 16, 513–522. [Google Scholar] [PubMed]
- Rumbo, M. , Sierro, F., Debard, N., Kraehenbuhl, J. P., & Finke, D. Lymphotoxin β receptor signaling induces the chemokine CCL20 in intestinal epithelium. Gastroenterology 2004, 127, 213–223. [Google Scholar] [CrossRef]
- Campelo, M. B. D. , Santos, J. D. A. F., Maia Filho, A. L. M., Ferreira, D. C. L., Sant’Anna, L. B., Oliveira, R. A. D.,... & Arisawa, E. Â. L. Effects of the application of the amniotic membrane in the healing process of skin wounds in rats. Acta cirurgica brasileira 2018, 33, 144–155. [Google Scholar] [PubMed]
- Junqueira, L. C. U. , Bignolas, G., & Brentani, R. R. Picrosirius staining plus polarization microscopy, a specific method for collagen detection in tissue sections. The Histochemical Journal 1979, 11, 447–455. [Google Scholar] [CrossRef]
- López De Padilla, C. M. , Coenen, M. J., Tovar, A., De la Vega, R. E., Evans, C. H., & Müller, S. A. Picrosirius red staining: revisiting its application to the qualitative and quantitative assessment of collagen type I and type III in tendon. Journal of Histochemistry & Cytochemistry 2021, 69, 633–643. [Google Scholar]
- Mathew-Steiner, S. S. , Roy, S., & Sen, C. K. Collagen in wound healing. Bioengineering 2021, 8, 63. [Google Scholar] [CrossRef]
- Jain, R. K. K. Molecular regulation of vessel maturation. Nature medicine 2003, 9, 685–693. [Google Scholar] [PubMed]
- Veith, A. P. , Henderson, K., Spencer, A., Sligar, A. D., & Baker, A. B. Therapeutic strategies for enhancing angiogenesis in wound healing. Advanced drug delivery reviews 2019, 146, 97–125. [Google Scholar] [PubMed]
- Shams, F. , Moravvej, H., Hosseinzadeh, S. et al. Overexpression of VEGF in dermal fibroblast cells accelerates the angiogenesis and wound healing function: in vitro and in vivo studies. Sci Rep 2022, 12, 18529. [Google Scholar] [CrossRef]
- Bao, P. , Kodra, A., Tomic-Canic, M., Golinko, M. S., Ehrlich, H. P., & Brem, H. The role of vascular endothelial growth factor in wound healing. Journal of Surgical Research 2009, 153, 347–358. [Google Scholar] [CrossRef]
- Zhao, Y. , Li, M., Mao, J., Su, Y., Huang, X., Xia, W.,... & Zan, T. Immunomodulation of wound healing leading to efferocytosis. Smart Medicine 2024, 3, e20230036. [Google Scholar] [CrossRef]
- Guo, S. A. , & DiPietro, L. A. Factors affecting wound healing. Journal of dental research 2010, 89, 219–229. [Google Scholar]
- Chen, L. , Wang, J., Li, S., Yu, Z., Liu, B., Song, B., & Su, Y. The clinical dynamic changes of macrophage phenotype and function in different stages of human wound healing and hypertrophic scar formation. International wound journal 2019, 16, 360–369. [Google Scholar] [CrossRef]
- Roberts AB, Sporn MB, Assoian RK, Smith JM, Roche NS, Wakefield LM, Heine UI, Liotta LA, Falanga V, Kehrl JH, et al. Transforming growth factor type beta: rapid induction of fibrosis and angiogenesis in vivo and stimulation of collagen formation in vitro. Proc Natl Acad Sci USA 1986, 83, 4167–4171. [Google Scholar] [CrossRef]
- Pesce, J. T. , Ramalingam, T. R., Mentink-Kane, M. M., Wilson, M. S., El Kasmi, K. C., Smith, A. M.,... & Wynn, T. A. Arginase-1–expressing macrophages suppress Th2 cytokine–driven inflammation and fibrosis. PLoS pathogens 2009, 5, e1000371. [Google Scholar] [CrossRef]
- Mantovani, A. , Biswas, S. K., Galdiero, M. R., Sica, A., & Locati, M. Macrophage plasticity and polarization in tissue repair and remodelling. The Journal of pathology 2013, 229, 176–185. [Google Scholar] [CrossRef]
- Rauchenwald, T. , Handle, F., Connolly, C. E., Degen, A., Seifarth, C., Hermann, M.,... & Ploner, C. Preadipocytes in human granulation tissue: role in wound healing and response to macrophage polarization. Inflammation and Regeneration 2023, 43, 53. [Google Scholar]
- Forde, A. J. , & Kolter, J. (2023). Isolation and Flow Cytometry Analysis of Macrophages from the Dermis. In Tissue-Resident Macrophages: Methods and Protocols (pp. 159–169). New York, NY: Springer US.
- Jenkins, S. J. , Ruckerl, D., Cook, P. C., Jones, L. H., Finkelman, F. D., Van Rooijen, N.,... & Allen, J. E. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 2011, 332, 1284–1288. [Google Scholar] [PubMed]
- Maschalidi, S. , Mehrotra, P., Keçeli, B.N. et al. Targeting SLC7A11 improves efferocytosis by dendritic cells and wound healing in diabetes. Nature 2022, 606, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Wang, JH. Mechanobiology of tendon. J Biomech. 2006, 39, 1563–1582. [Google Scholar] [CrossRef]
- Somaiah C, Kumar A, Mawrie D, Sharma A, Patil SD, Bhattacharyya J, Swaminathan R, Jaganathan BG. Collagen promotes higher adhesion, survival and proliferation of mesenchymal stem cells. PLoS ONE. 2015, 10, e0145068. [Google Scholar] [CrossRef]
- Merkel JR, DiPaolo BR, Hallock GG, Rice DC. Type I and type III collagen content of healing wounds in fetal and adult rats. Proc Soc Exp Biol Med. 1988, 187, 493–497. [Google Scholar] [CrossRef]
- Volk SW, Wang Y, Mauldin EA, Liechty KW, Adams SL. Diminished type III collagen promotes myofibroblast differentiation and increases scar deposition in cutaneous wound healing. Cells Tissues Organs. 2011, 194, 25–37. [Google Scholar] [CrossRef]
- Kraman, M. , Bambrough, P.J., Arnold, J. N., Roberts, E. W., Magiera, L., Jones, J. O.,... & Fearon, D. T. Suppression of antitumor immunity by stromal cells expressing fibroblast activation protein–α. Science 2010, 330, 827–830. [Google Scholar] [CrossRef]
- Fitzgerald, A. A. , & Weiner, L. M. The role of fibroblast activation protein in health and malignancy. Cancer and Metastasis Reviews 2020, 39, 783–803. [Google Scholar]
- Roman, J. Fibroblasts—Warriors at the Intersection of Wound Healing and Disrepair. Biomolecules 2023, 13, 945. [Google Scholar] [CrossRef] [PubMed]
- García, M. G. S. , Díaz, N. F., López, G. G., Maya, I. Á., Jimenez, C. H., Maldonado, Y. R.,... & Martínez, N.E.D. Evaluation methods for decellularized tissues: A focus on human amniotic membrane. Sci Rep 2024. [Google Scholar] [CrossRef]
- Mogami, H. , Hari Kishore, A., Akgul, Y., & Word, R. A. Healing of preterm ruptured fetal membranes. Scientific reports 2017, 7, 13139. [Google Scholar] [CrossRef]
| Amplicon | Forward primer | Reverse primer |
| β-actin | CCT GGC ACC CAG CAC AAT | GCC GAT CCA CAC GGA GTA CT |
| IL-1β | TAC GAA TCT CCG ACC ACC ACT ACA G | TGG AGG TGG AGA GCT TTC AGT TCA TAT G |
| TNF-ɑ | AAC CTC CTC TCT GCC ATC AA | CCA AAG TAG ACC TGC CCA GA |
| TGF-β | ACC CAC AAC GAA ATC TAT GAC | GCT CCA CTT TTA ACT TGA GCC |
| Arginase 1 | GTT TCT CAA GCA GAC CAG CC | GCT CAA GTG CAG CAA AGA GA |
| VEGF | CAC TGC CTG GAA GAT TCA | TGG TTT CAA TGG TGT GAG GA |
| CCL-2 | CGC CTC CAG CAT GAA AGT CT | ATG AAG GTG GCT GCT ATG AGC |
| IL-8 | CAC CGG AAG GAA CCA TCT CA | GGA AGG CTG CCA AGA GAG C |
| CXCL-10 | TCC ACG TGT TCA GAT CAT TGC | TGA TGG CCT TCG ATT CTG G |
| COL1A1 | CGA AGA CAT CCC ACC AAT CAC | TCA TCG CAC AAC ACC TTG C |
| COL1A2 | ACC TCA GGG TGT TCA AGG TG | CTT CTC CAG CGG TAC CAG AG |
| COL3A1 | CTG GTC CTG TTG GTC CAT CT | ACC TTT GTC ACC TCG TGG AC |
| FAP | ATG AGC TTC CTC GTC CAA TTC A | AGA CCA CCA GAG AGC ATA TTT TG |
| ɑ-SMA | AGG GAG TAA TGG TTG GAA TGG | TGA TGA TGC CGT GTT CTA TCG |
| MMP-1 | TCG CTG GGA GCA AAC ACA | TTG GCA AAT CTG GCG TGA A |
| MMP-2 | CCT CTC CAC TGC CTT CGA TA | GCC TGG GAG GAG TAC AGT CA |
| TIMP-1 | CGC TGA CAT CCG GTT CGT | GTG GAA GTA TCC GCA GAC ACT CT |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




