Submitted:
14 April 2025
Posted:
15 April 2025
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
Statistical Analysis
3. Results
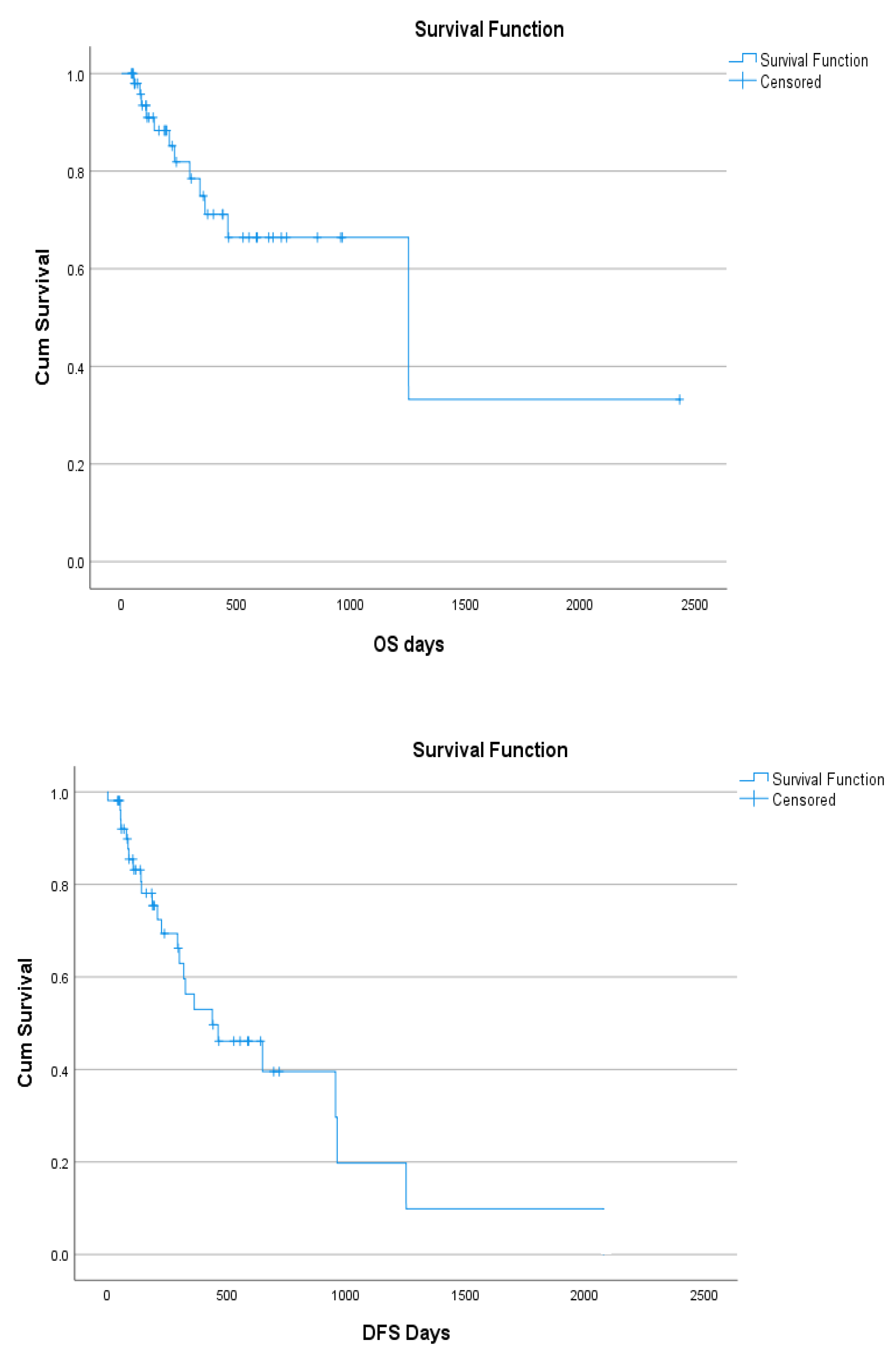
3.1. AML Cohort
| Overall Survival | Disease Free Survival | ||||||||
|
Total (n) |
Dead (n) |
Alive n(%) |
P-value |
Total (n) |
Dead /relapsed (n) |
Alive/disease free (%) | P-value | ||
| WHO Classification | AML-NOS | 34 | 6 | 28 (83) | 0.12 | 37 | 15 | 22 (59.4) | 0.04 |
| AML-MR | 14 | 9 | 5(57.1%) | 11 | 9 | 5(35.7%) | |||
| Prior cytotoxic therapy | 2 | 0 | 2 (100) | 2 | 0 | 2 (100) | |||
| RGA | 3 | 1 | 1 (50) | 2 | 2 | 0 (0) | |||
| Risk stratification | Favorable | 3 | 1 | 2 (66.7) | 0.2 | 3 | 3 | 0 (0) | 0.005 |
| Int | 37 | 5 | 31 (86.1) | 36 | 14 | 22 (61.1) | |||
| Adverse | 14 | 6 | 8 (57.1) | 14 | 8 | 6 (42.9) | |||
| Sequencing | 1st line | 41 | 8 | 32 (80) | 0.81 | 41 | 16 | 24 (60) | 0.84 |
| 2nd line | 12 | 4 | 8 (66.7) | 12 | 8 | 4 (33.3) | |||
| 3rd line | 1 | 0 | 1 (100) | 1 | 1 | 0 (0) | |||
| HMA used | Azacitidine | 44 | 6 | 37 (86) | 0.14 | 43 | 18 | 25 (58.1) | 0.34 |
| Decitabine | 10 | 6 | 4 (40) | 10 | 7 | 3 (30) | |||
| ORR Cycle 1 | Yes | 26 | 4 | 22 (84.6) | 0.003 | 26 | 9 | 17 (65.4) | 0.03 |
| No | 17 | 8 | 9 (52.9) | 17 | 13 | 4 (23.5) | |||
| Not Assessed | 11 | 0 | 10 (100) | 10 | 3 | 7 (70) | |||
| Response after cycle 1 | CR | 15 | 3 | 12 (80) | 0.02 | 15 | 5 | 10 (66.7) | 0.24 |
| CRi | 5 | 1 | 4 (80) | 5 | 1 | 4 (80) | |||
| PR | 6 | 0 | 6 (100) | 6 | 3 | 3 (50) | |||
| NR | 14 | 8 | 6 (42.9) | 14 | 13 | 1 (7.1) | |||
| IH | 1 | 0 | 1 (100) | 1 | 0 | 1 (100) | |||
| MLFS | 2 | 0 | 2 (100) | 2 | 0 | 2 (100) | |||
| Not assessed | 11 | 0 | 10 (100) | 10 | 3 | 7(70) | |||
| ORR Cycle 2 | Yes | 34 | 3 | 31 (91.2) | 0.001 | 34 | 10 | 24(70) | 0.01 |
| No | 13 | 7 | 6 (46.2) | 13 | 12 | 1 (7.7) | |||
| Not Assessed | 7 | 2 | 4 (66.7) | 6 | 3 | 3 (50) | |||
| Response after cycle 2 | CR | 27 | 3 | 24 (88.9) | 0.01 | 27 | 8 | 19 (70) | 0.06 |
| CRi | 3 | 0 | 3 (100) | 3 | 0 | 3 (100) | |||
| PR | 4 | 0 | 4 (100) | 4 | 2 | 2 (50) | |||
| NR | 12 | 7 | 5 (41.7) | 12 | 12 | 0 (0) | |||
| MLFS | 1 | 0 | 1 (100) | 1 | 0 | 1 (100) | |||
| Not assessed | 7 | 2 | 4 (66.7) | 6 | 3 | 3 (50) | |||
| ORR EOT | Yes | 36 | 2 | 34 (94.4) | 0.001 | 36 | 9 | 27 (75) | 0.001 |
| No | 16 | 10 | 6 (37.5) | 16 | 15 | 1 (6.3) | |||
| Not Assessed | 2 | 0 | 1 (100) | 1 | 1 | 0 (0) | |||
| IDAC cons | Yes | 5 | 1 | 4 (80) | 0.91 | 5 | 1 | 4 (80) | 0.37 |
| No | 48 | 11 | 37 (77.1) | 48 | 24 | 24 (50) | |||
| Allo BMT | Yes | 5 | 1 | 4 (80) | 0.86 | 5 | 1 | 4 (80) | 0.32 |
| No | 48 | 11 | 37 (77.1) | 48 | 24 | 24 (50) | |||
| Haplo BMT | Yes | 1 | 0 | 1 (100) | 0.51 | 1 | 1 | 0 (0) | 0.91 |
| No | 52 | 12 | 40 (76.9) | 52 | 24 | 28 (53.8) | |||
| Auto BMT | Yes | 1 | 0 | 1 (100) | 0.51 | 1 | 0 | 1 (100) | 0.37 |
| No | 52 | 12 | 40 (76.9) | 52 | 25 | 27 (51.9) | |||
| Ven Maintenance | Yes | 10 | 0 | 10 (100) | 0.02 | 10 | 4 | 6 (60) | 0.18 |
| No | 43 | 12 | 31 (72.1) | 43 | 21 | 22 (51.2) | |||
| WHO Classification |
Overall Survival (OS) (p=0.002) |
Disease Free Survival (DFS) (p=0.12) |
| AML with recurrent genetic abnormalities (n=3) | 66.7% | 33.3% |
| AML with minimal differentiation (n=3) | 66.7% | 66.7% |
| AML without maturation (n=4) | 75% | 50% |
| AML with maturation (n=18) |
100% | 66.7% |
| AML with myelomonocytic differentiation (n=7) |
100% | 66.7% |
| Acute monocytic leukemia (n=2) | 100% | 100% |
| AML with myelodysplasia related changes (MR) (n=14) |
57.1% | 35.7% |
| AML with prior cytotoxic therapy (n=2) |
100% | 100% |
| AML with germline predisposition (n=1) |
100% | 100% |
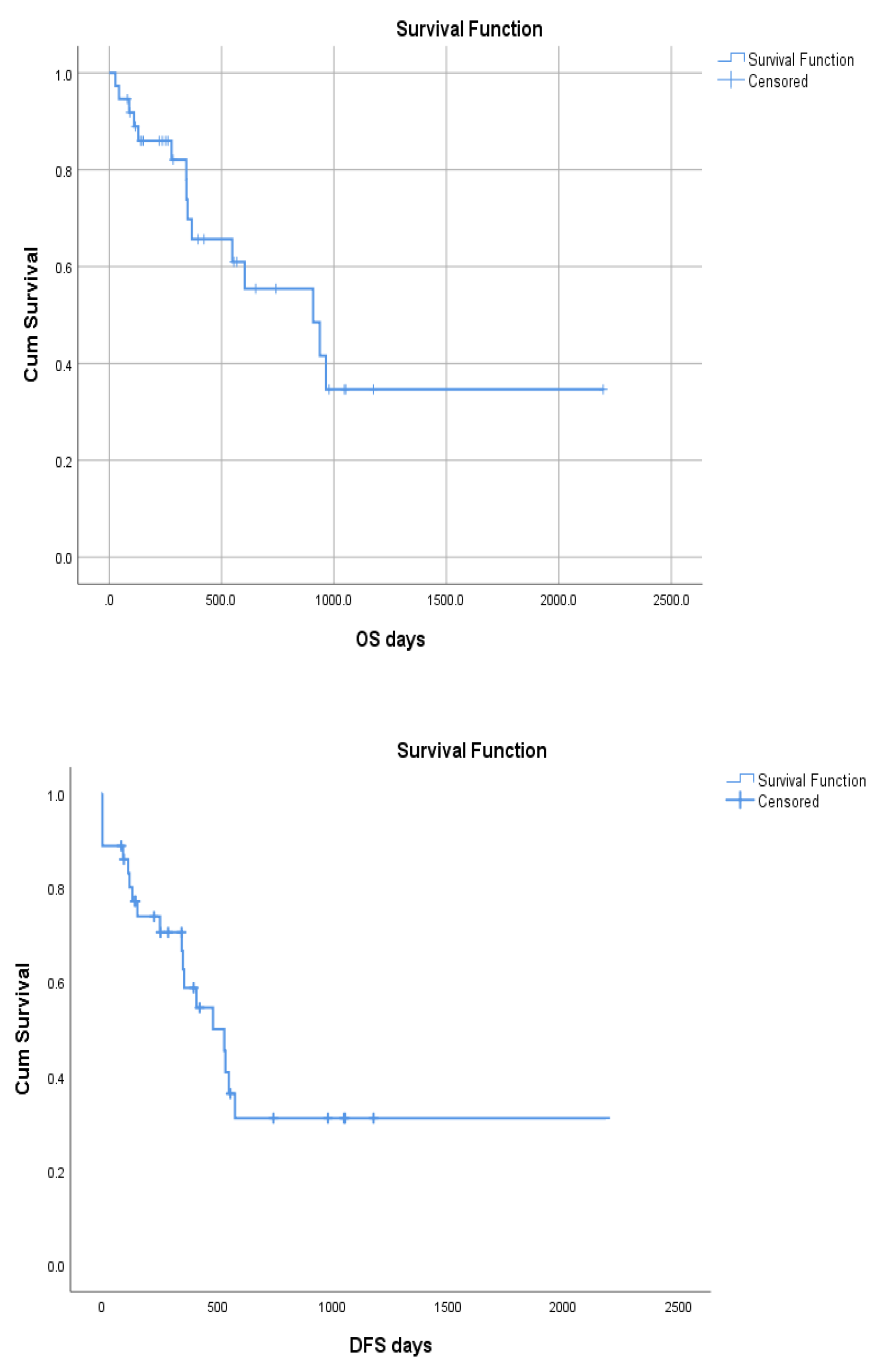
3.2. MDS cohort
| Overall Survival | Disease Free Survival | ||||||||
|
Total (n) |
Dead (n) |
Alive n(%) |
p-value |
Total (n) |
Dead/ Relapsed (n) |
Alive/ Disease free n(%) |
p-value | ||
| Prior disease | MDS-MLD | 5 | 2 | 3 (60) | 0.65 | 5 | 2 | 3 (60) | 0.01 |
| MDS-MPN | 1 | 0 | 1 (100) | 1 | 1 | 0 (0) | |||
| No | 31 | 13 | 18 (58.1) | 30 | 17 | 13 (43.3) | |||
| Cytogenetics | Normal | 24 | 8 | 16 (66.7) | 0.12 | 24 | 11 | 13 (54.2) | 0.06 |
| Abnormal | 4 | 2 | 2 (50) | 3 | 3 | 0 (0) | |||
| NA | 9 | 5 | 4 (44.4) | 9 | 6 | 3 (33.3) | |||
| HMA | Azacitidine | 28 | 10 | 18 (64.3) | 0.41 | 27 | 15 | 12 (44.4) | 0.78 |
| Decitabine | 9 | 5 | 4 (44.4) | 9 | 5 | 4 (44.4) | |||
| ORR after cycle 1 | Yes | 12 | 3 | 9 (75) | 0.28 | 12 | 2 | 10 (83.3) | 0.01 |
| No | 11 | 5 | 6 (54.5) | 10 | 8 | 2 (20) | |||
| NA | 14 | 7 | 7 (50) | 14 | 10 | 4 (28.6) | |||
| Response after cycle 1 | Response | 11 | 3 | 10 (90.9) | 0.18 | 13 | 3 | 10 (79.9) | 0.001 |
| No Response | 7 | 4 | 3 (42.8) | 6 | 5 | 1 (16.6) | |||
| Stable disease | 3 | 1 | 2 (66.7) | 3 | 2 | 1 (33.3) | |||
| Not assessed | 9 | 4 | 5 (55.6) | 9 | 6 | 3 (33.3) | |||
| NA | 5 | 3 | 2 (40) | 5 | 4 | 1 (20) | |||
| Febrile neutropenia | Yes | 21 | 10 | 11 (52.4) | 0.24 | 21 | 13 | 8 (38.1) | 0.07 |
| No | 16 | 5 | 11 (68.8) | 15 | 7 | 8 (53.3) | |||
| ORR after cycle 2 | Yes | 19 | 6 | 13 (68.4) | 0.12 | 19 | 6 | 13 (68.4) | 0.009 |
| No | 15 | 9 | 6 (40) | 14 | 12 | 2 (14.3) | |||
| NA | 3 | 0 | 3 (100) | 3 | 2 | 1 (33.3) | |||
| Response after cycle 2 | Response | 21 | 6 | 15 (71.4) | 0.03 | 21 | 7 | 14 (66.6) | 0.01 |
| No Response | 8 | 5 | 3 (37.5) | 4 | 3 | 1 (25) | |||
| Stable disease | 6 | 4 | 2 (33.3) | 6 | 6 | 0 (0) | |||
| NA | 2 | 0 | 2 (100) | 2 | 1 | 1 (50) | |||
| ORR EOT | Yes | 17 | 4 | 13 (76.5) | 0.07 | 17 | 3 | 14 (82.4) | 0.001 |
| No | 19 | 11 | 8 (42.1) | 18 | 17 | 1 (5.6) | |||
| NA | 1 | 0 | 1 (100) | 1 | 0 | 1 (100) | |||
| Response EOT | Response | 20 | 4 | 7 (45.4) | 0.62 | 20 | 6 | 14 (70) | 0.001 |
| No Response | 11 | 6 | 5 (45.4) | 10 | 9 | 1 (10) | |||
| Stable disease | 5 | 3 | 2 (40) | 5 | 5 | 0 (0) | |||
| Not assessed | 1 | 0 | 1 (100) | 1 | 0 | 1 (100) | |||
| Consolidation Allo | Yes | 6 | 2 | 4 (66.7) | 0.09 | 6 | 2 | 4 (66.7) | 0.06 |
| No | 30 | 12 | 18 (60) | 29 | 17 | 12 (41.4) | |||
| NA | 1 | 1 | 0 (0) | 1 | 1 | 0 (0) | |||
| Consolidation Haplo | Yes | 2 | 1 | 1 (50) | 0.17 | 2 | 1 | 1 (50) | 0.37 |
| No | 34 | 13 | 21 (61.8) | 33 | 18 | 15 (45.5) | |||
| NA | 1 | 1 | 0 (0) | 1 | 1 | 0 (0) | |||
| Relapse | Yes | 8 | 6 | 2 (25) | 0.001 | 8 | 8 | 0 (0) | 0.01 |
| No | 28 | 8 | 20 (71.4) | 27 | 11 | 16 (59.3) | |||
| Refractory | 1 | 1 | 0 (0) | 1 | 1 | 0 (0) | |||
4. Discussion
5. Conclusions
References
- Tefferi A, Vardiman JW. Myelodysplastic syndromes. New England Journal of Medicine. 2009;361(19):1872-85.
- Estey, E.; Karp, J.E.; Emadi, A.; Othus, M.; Gale, R.P. Recent drug approvals for newly diagnosed acute myeloid leukemia: gifts or a Trojan horse? Leukemia 2020, 34, 671–681. [Google Scholar] [CrossRef] [PubMed]
- DiNardo, C.D.; Pratz, K.W.; Letai, A.; A Jonas, B.; Wei, A.H.; Thirman, M.; Arellano, M.; Frattini, M.G.; Kantarjian, H.; Popovic, R.; et al. Safety and preliminary efficacy of venetoclax with decitabine or azacitidine in elderly patients with previously untreated acute myeloid leukaemia: a non-randomised, open-label, phase 1b study. Lancet Oncol. 2018, 19, 216–228. [Google Scholar] [CrossRef] [PubMed]
- Dinardo, C.D.; Jonas, B.A.; Pullarkat, V.; Thirman, M.J.; Garcia, J.S.; Wei, A.H.; Konopleva, M.; Döhner, H.; Letai, A.; Fenaux, P.; et al. Azacitidine and Venetoclax in Previously Untreated Acute Myeloid Leukemia. N. Engl. J. Med. 2020, 383, 617–629. [Google Scholar] [CrossRef]
- Souers, A.J.; Leverson, J.D.; Boghaert, E.R.; Ackler, S.L.; Catron, N.D.; Chen, J.; Dayton, B.D.; Ding, H.; Enschede, S.H.; Fairbrother, W.J.; et al. ABT-199, a potent and selective BCL-2 inhibitor, achieves antitumor activity while sparing platelets. Nat. Med. 2013, 19, 202–208. [Google Scholar] [CrossRef] [PubMed]
- Pollyea, D.A.; Stevens, B.M.; Jones, C.L.; Winters, A.; Pei, S.; Minhajuddin, M.; D’alessandro, A.; Culp-Hill, R.; Riemondy, K.A.; Gillen, A.E.; et al. Venetoclax with azacitidine disrupts energy metabolism and targets leukemia stem cells in patients with acute myeloid leukemia. Nat. Med. 2018, 24, 1859–1866. [Google Scholar] [CrossRef]
- Testa U, Castelli G, Pelosi E. TP53-mutated myelodysplasia and acute myeloid leukemia. Mediterranean Journal of Hematology and Infectious Diseases. 2023;15(1):e2023038.
- Li, X.; Suh, H.S.; Lachaine, J.; Schuh, A.C.; Pratz, K.; Betts, K.A.; Song, J.; Dua, A.; Bui, C.N. Comparative Efficacy of Venetoclax-Based Combination Therapies and Other Therapies in Treatment-Naive Patients With Acute Myeloid Leukemia Ineligible for Intensive Chemotherapy: A Network Meta-Analysis. Value Heal. 2023, 26, 1689–1696. [Google Scholar] [CrossRef]
- Vardiman, J.W.; Harris, N.L.; Brunning, R.D. The World Health Organization (WHO) classification of the myeloid neoplasms. Blood 2002, 100, 2292–2302. [Google Scholar] [CrossRef]
- Arber, D.A.; Orazi, A.; Hasserjian, R.; Thiele, J.; Borowitz, M.J.; Le Beau, M.M.; Bloomfield, C.D.; Cazzola, M.; Vardiman, J.W. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood 2016, 127, 2391–2405. [Google Scholar] [CrossRef]
- Döhner, H.; Wei, A.H.; Appelbaum, F.R.; Craddock, C.; DiNardo, C.D.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Godley, L.A.; Hasserjian, R.P.; et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood 2022, 140, 1345–1377. [Google Scholar] [CrossRef]
- Döhner, H.; Estey, E.; Grimwade, D.; Amadori, S.; Appelbaum, F.R.; Büchner, T.; Dombret, H.; Ebert, B.L.; Fenaux, P.; Larson, R.A.; et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood 2017, 129, 424–447. [Google Scholar] [CrossRef]
- Greenberg, P.; Cox, C.; LeBeau, M.M.; Fenaux, P.; Morel, P.; Sanz, G.; Sanz, M.; Vallespi, T.; Hamblin, T.; Oscier, D.; et al. International Scoring System for Evaluating Prognosis in Myelodysplastic Syndromes. Blood 1997, 89, 2079–2088. [Google Scholar] [CrossRef] [PubMed]
- Greenberg, P.L.; Tuechler, H.; Schanz, J.; Sanz, G.; Garcia-Manero, G.; Solé, F.; Bennett, J.M.; Bowen, D.; Fenaux, P.; Dreyfus, F.; et al. Revised International Prognostic Scoring System for Myelodysplastic Syndromes. Blood 2012, 120, 2454–2465. [Google Scholar] [CrossRef]
- Heuser, M.; Ofran, Y.; Boissel, N.; Mauri, S.B.; Craddock, C.; Janssen, J.; Wierzbowska, A.; Buske, C. Acute myeloid leukaemia in adult patients: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 697–712. [Google Scholar] [CrossRef] [PubMed]
- Cheson, B.D.; Greenberg, P.L.; Bennett, J.M.; Lowenberg, B.; Wijermans, P.W.; Nimer, S.D.; Pinto, A.; Beran, M.; de Witte, T.M.; Stone, R.M.; et al. Clinical application and proposal for modification of the International Working Group (IWG) response criteria in myelodysplasia. Blood 2006, 108, 419–425. [Google Scholar] [CrossRef]
- Zeidan, A.M.; Platzbecker, U.; Bewersdorf, J.P.; Stahl, M.; Adès, L.; Borate, U.; Bowen, D.T.; Buckstein, R.J.; Brunner, A.M.; E Carraway, H.; et al. Consensus proposal for revised International Working Group response criteria for higher risk myelodysplastic syndromes. Blood 2023, 141, 2047–2061. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.G.; Volpe, V.O.; Wang, C.; Ball, S.; Tobon, K.; Chan, O.; Padron, E.; Kuykendall, A.; Lancet, J.E.; Komrokji, R.; et al. Outcomes by best response with hypomethylating agent plus venetoclax in adults with previously untreated acute myeloid leukemia. Ann. Hematol. 2024, 104, 307–315. [Google Scholar] [CrossRef]
- Juliusson, G.; Antunovic, P.; Derolf, Å.; Lehmann, S.; Möllgård, L.; Stockelberg, D.; Tidefelt, U.; Wahlin, A.; Hoglund, M. Age and acute myeloid leukemia: real world data on decision to treat and outcomes from the Swedish Acute Leukemia Registry. Blood 2009, 113, 4179–4187. [Google Scholar] [CrossRef]
- Dinardo, C.D.; Pratz, K.; Pullarkat, V.; Jonas, B.A.; Arellano, M.; Becker, P.S.; Frankfurt, O.; Konopleva, M.; Wei, A.H.; Kantarjian, H.M.; et al. Venetoclax combined with decitabine or azacitidine in treatment-naive, elderly patients with acute myeloid leukemia. Blood 2019, 133, 7–17. [Google Scholar] [CrossRef]
- Komrokji, R.S.; Singh, A.M.; Al Ali, N.; Chan, O.; Padron, E.; Sweet, K.; Kuykendall, A.; Lancet, J.E.; Sallman, D.A. Assessing the role of venetoclax in combination with hypomethylating agents in higher risk myelodysplastic syndrome. Blood Cancer J. 2022, 12, 1–4. [Google Scholar] [CrossRef]
- Pleyer, L.; Klammer, P.; Drost, M.; Angermann, H.; Keil, F.; Petzer, V.; Heibl, S.; Moritz, J.; Girschikofsky, M.; Stampfl-Mattersberger, M.; et al. Real-World Comparison of Outcomes of Patients Treated with HMA Monotherapy Versus HMA-Ven - Insights from 1634 Patients Included within the Austrian Myeloid Registry of the AGMT Study Group. Blood 2024, 144, 3793–3793. [Google Scholar] [CrossRef]
- Okello, C.D.; Meshinchi, S.; Tarlock, K.; Warren, E.H.; Towlerton, A.M.; Ddungu, H.; Mulumba, Y.; Geriga, F.; Orem, J.; Balagadde, J.K. Clinical Outcome and Treatment-Related Mortality in Patients with Acute Myeloid Leukemia Treated at the Uganda Cancer Institute. Blood 2022, 140, 8940–8941. [Google Scholar] [CrossRef]
- Yadav, C.; Unni, M.; Paul, M.; Harimadhavan, M.; Ganapathy, R.; Haridas, N.K.; V.S., S.; Sreenaryanan, C.; Yawalkar, R.; Mony, U.; et al. Treatment Challenges in Acute Myeloid Leukemia in Lower-Middle-Income Countries: Navigating Intensity, Affordability, and Infections. Blood 2023, 142, 5180–5180. [CrossRef]
- Fenaux, P.; Ades, L. Review of azacitidine trials in Intermediate-2-and High-risk myelodysplastic syndromes. Leuk. Res. 2009, 33, S7–S11. [Google Scholar] [CrossRef] [PubMed]
- Mishra, R.; Nikoo, M.Z.; Veeraballi, S.; Singh, A. Venetoclax and Hypomethylating Agent Combination in Myeloid Malignancies: Mechanisms of Synergy and Challenges of Resistance. Int. J. Mol. Sci. 2023, 25, 484. [Google Scholar] [CrossRef] [PubMed]
- Bataller, A.; Bazinet, A.; DiNardo, C.D.; Maiti, A.; Borthakur, G.; Daver, N.G.; Short, N.J.; Jabbour, E.J.; Issa, G.C.; Pemmaraju, N.; et al. Prognostic risk signature in patients with acute myeloid leukemia treated with hypomethylating agents and venetoclax. Blood Adv. 2024, 8, 927–935. [Google Scholar] [CrossRef]
- Zhao, L.; Yang, J.; Chen, M.; Xiang, X.; Ma, H.; Niu, T.; Gong, Y.; Chen, X.; Liu, J.; Wu, Y. Myelomonocytic and monocytic acute myeloid leukemia demonstrate comparable poor outcomes with venetoclax-based treatment: a monocentric real-world study. Ann. Hematol. 2024, 103, 1197–1209. [Google Scholar] [CrossRef]
- Dangudubiyyam, S.K.S.S.; Dhawan, R.; Swaminathan, A.; Stitha, P.; Naik, R.D.; Aggarwal, M.; Kumar, P.; Dass, J.; Viswanathan, G.; Tyagi, S.; et al. Challenges and outcomes of treating acute myeloid leukemia (AML) in resource-constrained settings. J. Clin. Oncol. 2022, 40, 7014–7014. [Google Scholar] [CrossRef]
- Sekeres, M.A.; Cutler, C. How we treat higher-risk myelodysplastic syndromes. Blood 2014, 123, 829–836. [Google Scholar] [CrossRef]
- Sallman DA, Xie Z. Frontline treatment options for higher-risk MDS: can we move past azacitidine? Hematology Am Soc Hematol Educ Program. 2023;2023(1):65-72.
- Unglaub, J.M.; Schlenk, R.F.; Middeke, J.M.; Krause, S.W.; Kraus, S.; Einsele, H.; Kramer, M.; Zukunft, S.; Kauer, J.; Renders, S.; et al. Venetoclax-based salvage therapy as a bridge to transplant is feasible and effective in patients with relapsed/refractory AML. Blood Adv. 2025, 9, 375–385. [Google Scholar] [CrossRef]
- Chen, J.; Liu, L.; Ma, R.; Pang, A.; Yang, D.; Chen, X.; Wei, J.; He, Y.; Zhang, R.; Zhai, W.; et al. Outcome of autologous stem cell transplantation in patients with favorable-risk acute myeloid leukemia in first remission. Cancer Cell Int. 2022, 22, 1–6. [Google Scholar] [CrossRef]
- Jimenez-Chillon, C.; Dillon, R.; Russell, N. Optimal Post-Remission Consolidation Therapy in Patients with AML. Acta Haematol. 2023, 147, 147–158. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




